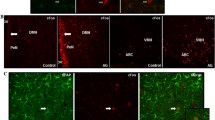
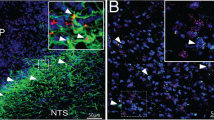
Abstract
Brain-derived neurotrophic factor (BDNF) is essential for maintaining energy and glucose balance within the central nervous system. Because the study of its metabolic actions has been limited to effects in neuronal cells, its role in other cell types within the brain remains poorly understood. Here we show that astrocytic BDNF signaling within the ventromedial hypothalamus (VMH) modulates neuronal activity in response to changes in energy status. This occurs via the truncated TrkB.T1 receptor. Accordingly, either fasting or central BDNF depletion enhances astrocytic synaptic glutamate clearance, thereby decreasing neuronal activity in mice. Notably, selective depletion of TrkB.T1 in VMH astrocytes blunts the effects of energy status on excitatory transmission, as well as on responses to leptin, glucose and lipids. These effects are driven by increased astrocytic invasion of excitatory synapses, enhanced glutamate reuptake and decreased neuronal activity. We thus identify BDNF/TrkB.T1 signaling in VMH astrocytes as an essential mechanism that participates in energy and glucose homeostasis.







Similar content being viewed by others
Data availability
All data associated with this study are presented in the paper or the extended data figures. Source data are provided with this paper.
References
Fu, L. Y. & van den Pol, A. N. Agouti-related peptide and MC3/4 receptor agonists both inhibit excitatory hypothalamic ventromedial nucleus neurons. J. Neurosci. 28, 5433–5449 (2008).
Huang, E. J. & Reichardt, L. F. Trk receptors: roles in neuronal signal transduction. Annu. Rev. Biochem. 72, 609–642 (2003).
Tran, P. V. et al. Diminished hypothalamic bdnf expression and impaired VMH function are associated with reduced SF-1 gene dosage. J. Comp. Neurol. 498, 637–648 (2006).
Unger, T. J., Calderon, G. A., Bradley, L. C., Sena-Esteves, M. & Rios, M. Selective deletion of Bdnf in the ventromedial and dorsomedial hypothalamus of adult mice results in hyperphagic behavior and obesity. J. Neurosci. 27, 14265–14274 (2007).
Rios, M. et al. Conditional deletion of brain-derived neurotrophic factor in the postnatal brain leads to obesity and hyperactivity. Mol. Endocrinol. 15, 1748–1757 (2001).
Chen, Z. Y. et al. Genetic variant BDNF (Val66Met) polymorphism alters anxiety-related behavior. Science 314, 140–143 (2006).
Han, J. C. et al. Brain-derived neurotrophic factor and obesity in the WAGR syndrome. N. Engl. J. Med. 359, 918–927 (2008).
Speliotes, E. K. et al. Association analyses of 249,796 individuals reveal 18 new loci associated with body mass index. Nat. Genet. 42, 937–948 (2010).
Haber, M., Zhou, L. & Murai, K. K. Cooperative astrocyte and dendritic spine dynamics at hippocampal excitatory synapses. J. Neurosci. 26, 8881–8891 (2006).
Hirrlinger, J., Hulsmann, S. & Kirchhoff, F. Astroglial processes show spontaneous motility at active synaptic terminals in situ. Eur. J. Neurosci. 20, 2235–2239 (2004).
Lippman, J. J., Lordkipanidze, T., Buell, M. E., Yoon, S. O. & Dunaevsky, A. Morphogenesis and regulation of Bergmann glial processes during Purkinje cell dendritic spine ensheathment and synaptogenesis. Glia 56, 1463–1477 (2008).
Pannasch, U. et al. Connexin 30 sets synaptic strength by controlling astroglial synapse invasion. Nat. Neurosci. 17, 549–558 (2014).
Dietrich, M. O. & Horvath, T. L. Feeding signals and brain circuitry. Eur. J. Neurosci. 30, 1688–1696 (2009).
Sternson, S. M., Shepherd, G. M. & Friedman, J. M. Topographic mapping of VMH–> arcuate nucleus microcircuits and their reorganization by fasting. Nat. Neurosci. 8, 1356–1363 (2005).
Valenstein, E. S., Cox, V. C. & Kakolewski, J. W. Modification of motivated behavior elicited by electrical stimulation of the hypothalamus. Science 159, 1119–1121 (1968).
Danbolt, N. C. Glutamate uptake. Prog. Neurobiol. 65, 1–105 (2001).
Lehre, K. P. & Danbolt, N. C. The number of glutamate transporter subtype molecules at glutamatergic synapses: chemical and stereological quantification in young adult rat brain. J. Neurosci. 18, 8751–8757 (1998).
Hanson, E. et al. Astrocytic glutamate uptake is slow and does not limit neuronal NMDA receptor activation in the neonatal neocortex. Glia 63, 1784–1796 (2015).
Baxter, G. T. et al. Signal transduction mediated by the truncated trkB receptor isoforms, trkB.T1 and trkB.T2. J. Neurosci. 17, 2683–2690 (1997).
Holt, L. M. et al. Astrocyte morphogenesis is dependent on BDNF signaling via astrocytic TrkB.T1. eLife https://doi.org/10.7554/eLife.44667 (2019).
Ohira, K. et al. A truncated tropomyosin-related kinase B receptor, T1, regulates glial cell morphology via Rho GDP dissociation inhibitor 1. J. Neurosci. 25, 1343–1353 (2005).
Rose, C. R. et al. Truncated TrkB-T1 mediates neurotrophin-evoked calcium signalling in glia cells. Nature 426, 74–78 (2003).
Baydyuk, M. et al. TrkB receptor controls striatal formation by regulating the number of newborn striatal neurons. Proc. Natl Acad. Sci. USA 108, 1669–1674 (2011).
Du, J., Feng, L., Yang, F. & Lu, B. Activity- and Ca(2+)-dependent modulation of surface expression of brain-derived neurotrophic factor receptors in hippocampal neurons. J. Cell Biol. 150, 1423–1434 (2000).
Haapasalo, A. et al. Regulation of TRKB surface expression by brain-derived neurotrophic factor and truncated TRKB isoforms. J. Biol. Chem. 277, 43160–43167 (2002).
Meyer-Franke, A. et al. Depolarization and cAMP elevation rapidly recruit TrkB to the plasma membrane of CNS neurons. Neuron 21, 681–693 (1998).
Carim-Todd, L. et al. Endogenous truncated TrkB.T1 receptor regulates neuronal complexity and TrkB kinase receptor function in vivo. J. Neurosci. 29, 678–685 (2009).
Yacoubian, T. A. & Lo, D. C. Truncated and full-length TrkB receptors regulate distinct modes of dendritic growth. Nat. Neurosci. 3, 342–349 (2000).
Lindberg, D., Chen, P. & Li, C. Conditional viral tracing reveals that steroidogenic factor 1-positive neurons of the dorsomedial subdivision of the ventromedial hypothalamus project to autonomic centers of the hypothalamus and hindbrain. J. Comp. Neurol. 521, 3167–3190 (2013).
Nguyen, N. L. et al. Separate and shared sympathetic outflow to white and brown fat coordinately regulates thermoregulation and beige adipocyte recruitment. Am. J. Physiol. Regul. Integr. Comp. Physiol. 312, R132–R145 (2017).
Ruffin, M. & Nicolaidis, S. Electrical stimulation of the ventromedial hypothalamus enhances both fat utilization and metabolic rate that precede and parallel the inhibition of feeding behavior. Brain Res. 846, 23–29 (1999).
Bingham, N. C., Anderson, K. K., Reuter, A. L., Stallings, N. R. & Parker, K. L. Selective loss of leptin receptors in the ventromedial hypothalamic nucleus results in increased adiposity and a metabolic syndrome. Endocrinology 149, 2138–2148 (2008).
Dhillon, H. et al. Leptin directly activates SF1 neurons in the VMH, and this action by leptin is required for normal body-weight homeostasis. Neuron 49, 191–203 (2006).
Elmquist, J. K., Ahima, R. S., Elias, C. F., Flier, J. S. & Saper, C. B. Leptin activates distinct projections from the dorsomedial and ventromedial hypothalamic nuclei. Proc. Natl Acad. Sci. USA 95, 741–746 (1998).
Satoh, N. et al. Sympathetic activation of leptin via the ventromedial hypothalamus: leptin-induced increase in catecholamine secretion. Diabetes 48, 1787–1793 (1999).
Scott, M. M. et al. Leptin targets in the mouse brain. J. Comp. Neurol. 514, 518–532 (2009).
Komori, T., Morikawa, Y., Nanjo, K. & Senba, E. Induction of brain-derived neurotrophic factor by leptin in the ventromedial hypothalamus. Neuroscience 139, 1107–1115 (2006).
Allen, N. J. et al. Astrocyte glypicans 4 and 6 promote formation of excitatory synapses via GluA1 AMPA receptors. Nature 486, 410–414 (2012).
Baldwin, K. T. & Eroglu, C. Molecular mechanisms of astrocyte-induced synaptogenesis. Curr. Opin. Neurobiol. 45, 113–120 (2017).
Chung, W. S., Allen, N. J. & Eroglu, C. Astrocytes control synapse formation, function, and elimination. Cold Spring Harb. Perspect. Biol. 7, a020370 (2015).
Elmariah, S. B., Oh, E. J., Hughes, E. G. & Balice-Gordon, R. J. Astrocytes regulate inhibitory synapse formation via Trk-mediated modulation of postsynaptic GABAA receptors. J. Neurosci. 25, 3638–3650 (2005).
Yoon, B. E. & Lee, C. J. GABA as a rising gliotransmitter. Front Neural Circuits 8, 141 (2014).
Mederos, S. & Perea, G. GABAergic-astrocyte signaling: a refinement of inhibitory brain networks. Glia 67, 1842–1851 (2019).
Takano, T. et al. Chemico-genetic discovery of astrocytic control of inhibition in vivo. Nature 588, 296–302 (2020).
Chee, M. J., Myers, M. G. Jr., Price, C. J. & Colmers, W. F. Neuropeptide Y suppresses anorexigenic output from the ventromedial nucleus of the hypothalamus. J. Neurosci. 30, 3380–3390 (2010).
Klockener, T. et al. High-fat feeding promotes obesity via insulin receptor/PI3K-dependent inhibition of SF-1 VMH neurons. Nat. Neurosci. 14, 911–918 (2011).
Ono, T. et al. Glucoresponsive neurons in rat ventromedial hypothalamic tissue slices in vitro. Brain Res. 232, 494–499 (1982).
Choi, Y. H., Fujikawa, T., Lee, J., Reuter, A. & Kim, K. W. Revisiting the ventral medial nucleus of the hypothalamus: the roles of SF-1 neurons in energy homeostasis. Front Neurosci. 7, 71 (2013).
Williams, K. W. & Elmquist, J. K. From neuroanatomy to behavior: central integration of peripheral signals regulating feeding behavior. Nat. Neurosci. 15, 1350–1355 (2012).
Choi, Y. H. et al. Melanocortin receptors mediate leptin effects on feeding and body weight but not adipose apoptosis. Physiol. Behav. 79, 795–801 (2003).
Halassa, M. M., Fellin, T., Takano, H., Dong, J. H. & Haydon, P. G. Synaptic islands defined by the territory of a single astrocyte. J. Neurosci. 27, 6473–6477 (2007).
Nagy, J. I. & Rash, J. E. Connexins and gap junctions of astrocytes and oligodendrocytes in the CNS. Brain Res. Brain Res. Rev. 32, 29–44 (2000).
Genoud, C., Knott, G. W., Sakata, K., Lu, B. & Welker, E. Altered synapse formation in the adult somatosensory cortex of brain-derived neurotrophic factor heterozygote mice. J. Neurosci. 24, 2394–2400 (2004).
Martinez, A. et al. TrkB and TrkC signaling are required for maturation and synaptogenesis of hippocampal connections. J. Neurosci. 18, 7336–7350 (1998).
Otal, R., Martinez, A. & Soriano, E. Lack of TrkB and TrkC signaling alters the synaptogenesis and maturation of mossy fiber terminals in the hippocampus. Cell Tissue Res. 319, 349–358 (2005).
Genoud, C. et al. Plasticity of astrocytic coverage and glutamate transporter expression in adult mouse cortex. PLoS Biol. 4, e343 (2006).
Murai, K. K., Nguyen, L. N., Irie, F., Yamaguchi, Y. & Pasquale, E. B. Control of hippocampal dendritic spine morphology through ephrin-A3/EphA4 signaling. Nat. Neurosci. 6, 153–160 (2003).
Garcia-Caceres, C. et al. Astrocytic insulin signaling couples brain glucose uptake with nutrient availability. Cell 166, 867–880 (2016).
Horvath, T. L. et al. Synaptic input organization of the melanocortin system predicts diet-induced hypothalamic reactive gliosis and obesity. Proc. Natl Acad. Sci. USA 107, 14875–14880 (2010).
Kim, J. G. et al. Leptin signaling in astrocytes regulates hypothalamic neuronal circuits and feeding. Nat. Neurosci. 17, 908–910 (2014).
Nuzzaci, D. et al. Postprandial hyperglycemia stimulates neuroglial plasticity in hypothalamic POMC neurons after a balanced meal. Cell Rep. 30, 3067–3078 (2020).
Fagan, M. P. et al. Essential and sex-specific effects of mGluR5 in ventromedial hypothalamus regulating estrogen signaling and glucose balance. Proc. Natl Acad. Sci. USA 117, 19566–19577 (2020).
Felsted, J. A. et al. α2δ-1 in SF1(+) neurons of the ventromedial hypothalamus is an essential regulator of glucose and lipid homeostasis. Cell Rep. 21, 2737–2747 (2017).
McClellan, K. M., Parker, K. L. & Tobet, S. Development of the ventromedial nucleus of the hypothalamus. Front. Neuroendocrinol. 27, 193–209 (2006).
Acknowledgements
We thank B. Xu for the floxed TrkB animals and the Danbolt laboratory for the anti-GLAST antibody. We thank the HMS Electron Microscopy Facility and M. Ericsson for electron microscopy imaging, consultation and services. We thank the Vanderbilt Hormone Assay and Analytical Services Core and Lipid Core supported by NIH grants DK059637 and DK020593, specifically D. Edgerton, E. Allen and C. Harris for assisting in tissue and serum catecholamine and lipid analysis. We thank the Imaging, Genomics and Circuits Behavior Cores at the Tufts Center for Neuroscience Research. This work was supported by grants by the National Institute of Neurological Disorders and Stroke and the National Institute of Diabetes and Digestive and Kidney Diseases awarded to M.R. (1R21NS091871 and 1R01DK117935-01) and D.A. (1F31DK118789-01A1), the Synapse Neurobiology Training Program awarded to D.A. (5T32NS061764-09) and the Training Program in Nutrition, Obesity and Metabolic Disorders awarded to A.M. (T32DK124170).
Author information
Authors and Affiliations
Contributions
D.A., M.R. and C.D. designed experiments. D.A., A.M., S.C. and J.F. conducted experiments. D.A., A.M. and M.R. analyzed and interpreted the results. D.A. and M.R. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks Michela Matteoli and the other, anonymous, reviewers for their contribution to the peer review of this work. Primary handling editors: Christoph Schmitt and Ashley Castellanos-Jankiewicz.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Intrinsic excitability of VMH neurons is not regulated by energy status or BDNF signaling.
a. Amplitude of sEPSCs in VMH neurons of fed (n = 21) and fasted WT (n = 21) and fed BDNF2L/2LCK:Cre mice (n = 19) (4–6 mice per group), Ordinary One-way ANOVA, p = 0.3. b. Amplitude of sEPSCs in VMH neurons of in WT Fed + aCSF (n = 13), Fasted + aCSF (n = 13) and Fasted + BDNF conditions (n = 12) (4 mice per group), Ordinary One-way ANOVA, p = 0.136. c. Input-output curves from VMH neurons in fed (n = 19) and fasted WT (n = 18) and fed BDNF2L/2LCK-Cre mice (n = 17) (3–4 mice per group). Two-way repeated measures ANOVA: Interaction, p = 0.7; Genotype, p = 0.3. d. Representative Traces showing resulting hyperpolarization or evoked action potentials in response to current injection steps of -120 mV and 120 mV. Data represented as mean + /- SEM.
Extended Data Fig. 2 Energy status and BDNF regulate astrocytic glutamate uptake at VMH synapses.
a. Representative traces of raw NMDAR responses (light purple), responses +100 uM DL-TBOA (dark purple) and responses + 50 uM APV (light gray). For following panels, WT Fed (n = 8), WT Fasted (n = 7) and Fed BDNF 2 L/2 L CK:Cre (n = 10). b. Amplitude (pA) of NMDAR responses before and after DL-TBOA application. c. Charge transfer (pA*ms) of NMDAR responses before and after DL-TBOA application. d. Decay (weighted tau) of NMDAR responses before and after DL-TBOA application. Two-way ANOVA: Genotype, p = 0.01; p = 0.0005; Interaction of genotype and DL-TBOA, p = 0.09. Bonferroni multiple comparisons, *, p = 0.001. Data represented as mean + /- SEM.
Extended Data Fig. 3 Energy status regulates astrocytic glutamate uptake at VMH synapses via BDNF signaling.
a. Representative traces of raw NMDAR responses (light purple), responses +100 uM DL-TBOA (dark purple) and responses + 50 uM APV (light gray). For following panels, Fed + aCSF (n = 6), Fasted + aCSF (n = 7), Fasted + BDNF (n = 6). b. Amplitude (pA) of NMDAR responses before and after DL-TBOA application. c. Charge transfer (pA*ms) of NMDAR responses before and after DL-TBOA application. d. Decay (weighted tau) of NMDAR responses before and after DL-TBOA application. Two-way ANOVA: Condition, p = 0.007; DL-TBOA, p = 0.02; Interaction of condition and DL-TBOA, p = 0.0009. Bonferroni multiple comparisons, *, p < 0.0001. Data represented as mean + /- SEM.
Extended Data Fig. 4 TrkB.T1 in VMH astrocytes is an essential regulator of body weight under standard chow conditions.
a. Expression of the neuronal marker b3 tubulin (FT n = 5, Astrocyte n = 7) and b. GLT-1 in mRNA isolated from VMH astrocytes and flowthrough (FT n = 3, Astrocyte n = 3). c. Immunolabeling of TrkB.T1 in the VMH of a TrkB F/F animal injected unilaterally with AAV5–GFAP-GFP–Cre 4 weeks post-surgery. Scale bar 15 uM. d. Colocalization of AAV5-GFAP-GFP–Cre with GFP signal with astrocytes (Sox9 + cells) and exclusion from neurons (NeuN + cells). Scale bar 50 uM. e. Colocalization of AAV5-GFAP-driven GFP signal with astrocytic (Sox9), neuronal (NeuN) and microglial (Iba)-specific markers. Scale bar 150 nM. f. Image showing that viral spread is limited to the VMH. g. Western blot and analysis showing TrkB.T1 expression in Control and TrkB.T1 KD mice within the VMH (n = 6) and the DMH of mice (n = 5). Data collected from one experiment. Student’s two-sided t-test, *, p = 0.01. Data represented as mean + /- SEM.
Extended Data Fig. 5 Depletion of TrkB.T1 from VMH astrocytes leads to increased body weight and alterations in locomotor activity, sympathetic tone and leptin insensitivity.
a. Body weights of TrkB.T1 KD (n = 10) and control males (n = 13). Two-way RM ANOVA: Genotype, p = 0.02; Time, p < 0.0001; Time x Genotype Interaction, p < 0.0001; Bonferroni multiple comparisons, *, p < 0.05. b. Body weights of TrkB.T1 KD (n = 11) and control females (n = 8). Two-way RM ANOVA: Genotype, p = 0.04; Time, p < 0.0001; Time x Genotype Interaction, p < 0.0001; Bonferroni multiple comparisons, *, p < 0.05, #, p = 0.09. c. Percentage body weight gain in TrkB.T1 KD (n = 7) and wild-type C57Bl6 males (n = 4) delivered AAV5-GFAP-GFP or AAV5-GFAP-GFP-Cre to the VMH. Two-way RM ANOVA: Genotype, p = 0.01; Bonferroni multiple comparisons, *, p < 0.5. d. Fine movements per hour of TrkB.T1 KD and control animals over 6 days (n = 7–9). Students two-sided t-test, *, p = 0.009. e – g. Norepinephrine levels in tissues from TrkB.T1 KD and control animals (n = 7). Student’s two-sided t-test, *, p = 0.01. h. Experimental design and daily weight change in TrkB.T1 KD and control male mice 4 weeks post-surgery in response to IP administration of vehicle for 3 days followed by administration of leptin (3 ug/g) for 3 days. Data are represented as mean + /- SEM.
Extended Data Fig. 6 The use of serotype AAV2 and a CMV promoter to knockdown TrkB is specific to neurons.
a. Diagram showing experimental approach for depleting TrkB from neurons bilaterally in the VMH of adult mice. b. Co-immunolabeling of VMH showing colocalization of AAV2-CMV-driven GFP signal with the neuronal marker NeuN but not with the astrocytic marker Sox9 or the microglial marker Iba1. Scale bar 50 nM. c. TrkB.T1 and TrkB.FL expression in floxed TrkB mice delivered AAV2-CMV-GFP (control) or AAV2-CMV-GFP–Cre (TrkB KD) to the VMH (n = 6). Student’s two-sided t-test, *, p = 0.01, **, p = 0.0016. Data represented as mean + /- SEM. d. Representative western blot showing viral knockdown of TrkB in VMH. Data collected from one experiment.
Extended Data Fig. 7 TrkB in VMH neurons is not required for the regulation of energy balance under chow conditions but is essential for glycemic control.
A. Percent body weight gain in Neuronal TrkB KD and control mice (n = 6). Two-way RM ANOVA: Genotype, p = 0.88; Time, p < 0.0001; Interaction, p = 0.85; Subjects (matching), p < 0.0001. B. Body weights of Neuronal TrkB KD and control mice (n = 7). Two-way RM ANOVA: Genotype, p = 0.44; Time, p < 0.0001; Interaction, p 0.76; Subjects (matching), p < 0.0001. C. Average weekly food intake weeks 3–6 post-surgery in neuronal TrkB KD (n = 8) and control (n = 6) mice. Student’s two-sided t-test, NS. D. Core body temperatures in neuronal TrkB KD (n = 8) and control (n = 6) mice. Student’s two-sided t-test, NS. E. Basic movements per hour of neuronal TrkB KD (n = 8) and control (n = 6) mice recorded over 6 days. F. Serum levels of leptin (pg/mL) in fed animals (n = 6). Students two-sided t-test, *, p = 0.02. G. Norepinephrine levels in indicated tissues in neuronal TrkB KD (n = 6) and control (n = 5) mice. Student’s two-sided t-test, *, p = 0.05. H. Glucose tolerance test of neuronal TrkB KD (n = 8) and control (n = 6) mice. Two-way RM ANOVA; Time, p < 0.0001; Genotype, p = 0.1; Interaction, p = 0.01. Bonferroni multiple comparisons, *, p = 0.04. I. GTT area under the curve. Students two-sided t-test, p = 0.08. J. Insulin tolerance test (n = 7). Two-way RM ANOVA; Time, p < 0.0001; Genotype, p = 0.65; Interaction, p = 0.48. Bonferroni multiple comparisons. K. ITT area under the curve. Students t-test, NS. Data represented as mean + /- SEM.
Extended Data Fig. 8 Selective depletion of TrkB.T1 from VMH astrocytes in adult mice does not alter VMH neuronal excitatory synapse density.
a. Representative images of VMH co-immunolabeled with anti-PDS95 and anti-vGlut2. Arrows indicate PSD95 and vGlut2 colocalization (scale bar 15 uM). b. Density of excitatory synapses (colocalization of vGlut2 and PSD95) in the VMH of TrkB.T1 KD (n = 8) and control mice (n = 7). Students two-tailed t-test, NS. Data are represented as mean + /- SEM.
Extended Data Fig. 9 Selective depletion of TrkB.T1 from VMH astrocytes in adult mice leads to increased glutamate uptake at synapses.
a. Representative traces of raw NMDAR responses (light purple), responses + 100 uM DL-TBOA (dark purple), and responses + 50 uM APV (light gray). For following panels, Control Fed (n = 9), Control Fasted (n = 7), TrkB.T1 Fed (n = 7), TrkB.T1 Fasted (n = 9). b. Amplitude (pA) of NMDAR responses before and after DL-TBOA application. c. Charge transfer (pA*ms) of NMDAR responses before and after DL-TBOA application. d. Decay (weighted tau) of NMDAR responses before and after DL-TBOA application. Two-way ANOVA: Genotype, p = 0.03; Treatment, p < 0.0001; Genotype x Treatment Interaction, p = 0.01. Bonferroni multiple comparisons, *, p = 0.02. **, p = 0.005, ***, p < 0.0001. Data represented as + /- SEM.
Supplementary information
Supplementary Information
Supplementary Table 1
Source data
Source Data Fig. 1
Statistical Source Data.
Source Data Fig. 2
Statistical Source Data.
Source Data Fig. 2
Unprocessed western blots.
Source Data Fig. 3
Statistical Source Data.
Source Data Fig. 3
Unprocessed western blots.
Source Data Fig. 4
Statistical Source Data.
Source Data Fig. 5
Statistical Source Data.
Source Data Fig. 6
Statistical Source Data.
Source Data Fig. 6
Unprocessed western blots.
Source Data Fig. 7
Statistical Source Data.
Source Data Extended Data Fig. 1
Statistical Source Data.
Source Data Extended Data Fig. 2
Statistical Source Data.
Source Data Extended Data Fig. 3
Statistical Source Data.
Source Data Extended Data Fig. 4
Statistical Source Data.
Source Data Extended Data Fig. 4
Unprocessed western blots.
Source Data Extended Data Fig. 5
Statistical Source Data.
Source Data Extended Data Fig. 6
Statistical Source Data.
Source Data Extended Data Fig. 6
Unprocessed western blots.
Source Data Extended Data Fig. 7
Statistical Source Data.
Source Data Extended Data Fig. 8
Statistical Source Data.
Source Data Extended Data Fig. 9
Statistical Source Data.
Rights and permissions
About this article
Cite this article
Ameroso, D., Meng, A., Chen, S. et al. Astrocytic BDNF signaling within the ventromedial hypothalamus regulates energy homeostasis. Nat Metab 4, 627–643 (2022). https://doi.org/10.1038/s42255-022-00566-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s42255-022-00566-0
- Springer Nature Limited
This article is cited by
-
A conceptual framework for astrocyte function
Nature Neuroscience (2023)
-
Astroglial clean-up of satiety synapses
Nature Metabolism (2022)