Abstract
Soil microbial community composition plays a key role in the decomposition of organic matter, while the quality of exogenous organic matter (EOM: rice straw, roots and pig manure) can influence soil chemical and biological properties. However, the evidences of the effect of combination of crop residues and pig manure on the changes in soil microbial community and enzymes activities are scarce. A greenhouse pot experiment was conducted to investigate the potential effect of EOM by analyzing soil properties, enzyme activities and microbial communities. The experiment consisted of eight treatments: CK (control), S (1% (w/w) rice straw), R (1% (w/w) rice root), SR (1% (w/w) rice straw + 1% (w/w) rice root), and added 1% (w/w) pig manure to CK, S, R and SR, respectively. Results showed that the straw treatment significantly increased the microbial biomass (carbon and nitrogen) and total carbon and nitrogen contents, cellulase and β-1,4-glucosidase activities, bacteria (i.e., gram-positive bacteria and gram-negative bacteria) PLFAs contents relative to CK regardless of whether pig manure was added. Moreover, the interaction between crop residues (e.g., straw and roots) and pig manure significantly influenced the contents of microbial biomass nitrogen and microbial biomass phosphorus, and the ratio of gram-positive bacteria to gram-negative bacteria. Redundance analysis confirmed that pH, nitrate nitrogen, ammonium nitrogen and dissolve organic carbon contents were significantly associated with soil microbial community under crop residues without pig manure addition. Furthermore, the experiment results showed that pig manure application not only provided more abundant nutrients (C, N and P) but also induced higher microbial and enzymatic activity compared with no pig manure addition. Our findings suggest that the combination of above-ground straw and pig manure is a better option for improving the functions of soil ecosystem.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Land use changes such as converting conventional tillage to no-tillage or minimal tillage, and paddy fields to upland field cultivation can cause changes in the soil properties and function1,2. Doubts remain uncertain how crop residues (i.e., root and straw) returning affect the availability of soil nutrients and the stability of soil carbon (C) supply under the land use change, and what difference in the contribution to soil fertility and soil organism community between above-ground straw and below-ground roots. The residues (i.e., root and straw) of previous crops, as an important and abundant biomass resource, not only directly increases the C input in agro-ecosystems, but also affects soil physical, chemical and biological properties and crop growth. However, the adverse effects of crop residues return on the quality of cultivated land, and the emergence, growth and development of crops have also been reported3. Thus, it is necessary to optimize fertilization methods, such as combining crop residues and animal wastes, to compensate for the deficiency of nitrogen in crop residues and to eliminate the competition with crops for nutrients during the decomposition of the residues.
The crop residues are increasing every year and annually around 360 MT in the world, of which the production of rice straw is about 234 MT in China3. Specifically, rice straw is rich in nutrients that can be degraded by microorganisms, and can contribute with 2.1 ~ 2.2 t C ha−1, 31 ~ 42 kg N ha−1, 8 kg P ha−1 and 34 ~ 61 kg K ha−1 per crop cycle by returning to the field4,5. Although the contribution of above-ground rice straw to SOC sequestration is significant, rice roots have been reported to contribute to SOC one point five to three fold than that of rice straw6,7. Generally, the decomposition rate of rice roots is lower than straw, this may be explained by the higher content of recalcitrant components such as lignin and lower content of decomposable components such as non-structural carbohydrates6,8. Conversely, the improper disposal, utilization, and management practices of crop residues could lead to environmental problem such as greenhouse gases emissions, air pollution and public health issues. In addition, the intensification of livestock has resulted in a large increase in the production of animals waste, but animals manure (e.g., pig manure) contains a large amount of organic matter, nitrogen (N) and phosphorus (P)9, which may largely compensate for the imbalances of soil nutrients caused by high intensity agricultural production10,11. It is well-known that organic fertilizer such as pig manure is considered worldwide as a promising and inexpensive source of nutrients to meet crop nutrient demand, maintain soil fertility and achieve high crop yield10. Thus, the rational use of crop residues and animal wastes not only reduces nutrients loss but also alleviates various environmental problems (e.g., greenhouse gases emissions, water and air pollution).
Organic matter decomposition and soil nutrient turnover cannot be achieved without the participation of extracellular enzymes, while the activity of extracellular enzymes can serve to evaluate soil health status12,13. Previous research work has reported that soil extracellular enzymes are synthetized and secreted by soil microorganisms, and are direct drivers of organic matter decomposition13. Soil enzymes may be a good indicator of soil biological changes as they respond rapidly to variables in soil fertility14. Moreover, soil enzyme activity may be a rate-limiting step in mineralization process and has a rapid response to soil C and nutrient availability with fertilizer15. In the cycling of nutrients, some soil enzymes participate in the elemental biochemical process, such as cellulase and β-1,4-glucosidase (BG) play an important role in degrading cellulose, producing glucose and releasing available nutrients, while N-acetyl-β-Dglucosaminidase (NAG) and Urease can represent soil N turnover, and acid phosphomonoesterase (AcP) is involved in organic phosphorus hydrolysis16,17,18,19. These enzymes are sensitive indicators for the variables of soil properties and are useful for the sustainable management of soil quality14. Therefore, soil extracellular enzymes participate in the decomposition of organic matter and modulate the rate-limiting steps of N and P mineralization in soils.
Soil microorganism is an important regulator of organic materials decomposition, while the addition of exogenous organic matter (EOM) affects microbial community composition12,20,21. Moreover, soil microorganism also plays an important role in the ecosystem processes, such as soil structural formtion and nutrients turnover, thereby maintaining soil agroecosystem functioning and sustainability12,22. The variation in critical species engaged in nutrient cycling can also impact soil fertility, for example, bacteria dominate in the initial stages of degrading organic matter, while fungi and actinomycetes can degrade the recalcitrant lignocellulose components and dominate in the later stage23,24. Several studies have reported that EOM (e.g., pig manure and crop residues) addition significantly affected microbial community composition and diversity9,25,26. Additionally, changes in microbial community structure and activity are important for SOM pools since the secretions (e.g., soil enzyme) from different microbial groups are involved in the dynamics of C in soil21. In general, crop residue inputs to soil are considered to provide substrates that are more readily utilizable than native SOM for microorganisms12,20, this might be related to a large proportion of chemical complex and recalcitrant to decomposition in native SOM27. As we all know, pig manure could strongly influence microbial communities by shifting oligotrophic organisms (common in exclusively mineral fertilizer or soils without fertilizer) toward microbes decomposing complex organic compounds28. This occurs because pig manure is rich in organic matter and nutrients, which are conducive to the metabolism and growth of microorganisms9. Therefore, it is necessary to further explore the effects of crop residues and their combined addition with pig manure on soil microbial community composition and diversity.
Returning crop residues (i.e., rice straw and root) and pig manure can provide nutrients and organic matter to meet the growth and metabolic requirement of crops, and maintain soil fertility. However, whether pig manure addition can wake soil enzyme activity and microbial community composition response to crop residues remains unclear. Therefore, it is crucial to investigate the potential impact of EOMs (i.e., pig manure, above-ground straw and below-ground root) on microbial community composition, as well as the involved enzymatic processes that control the availability of most limiting nutrients for microbial metabolic requirements. In this study, EOMs were collected to devise a greenhouse pot experiment, we hypothesized that the combination addition of crop residues (i.e., rice straw and root) and pig manure would change microbial community composition and enzyme activity. Specifically, the objectives of this study were (1) to determine the effect of crop residues (i.e., above-ground straw and below-ground root and their combination with pig manure on soil properties, enzyme activities and soil microbial community, (2) to estimate the relationships between soil microbial community and soil properties and enzyme activities, (3) to explore the benefits of above-ground straw and below-ground root in combination with pig manure to optimize its agronomic performance.
Materials and methods
Preparation of experimental soil
The soil for this study came from the Zengcheng Teaching and Research Farm (23°14′22″N, 113° 37′57″E), South China Agriculture University, Guangzhou, China. The climatic condition of the sampling site belongs to a typical subtropical monsoon climate and the soil is classified as sandy clay, consisting of sand (57.56%), silt (6.13%) and clay (36.31%), as well as the main soil properties are shown in Table 1.
On 15 November, 2021, the soil was collected in the surface soil (0 ~ 20 cm) from paddy fields after rice harvest. Soil samples were transported to the laboratory, and the visible stones and plant roots were removed. Meanwhile, we also collected the post-harvest straw and roots from the same plot, and transported to the laboratory for cleaning. The collected samples including rice straw, roots and animal manure (pig manure) were oven-dried at 105℃, and then crushed, homogenized, ground and stored in a fine powder, and through a 2-mm sieve before the pot experiment. The properties of straw, roots and pig manure are shown in Table 1.
Experiment design
The greenhouse pot experiment started on December 8, 2021 and conducted in a greenhouse at South China Agriculture University, Guangzhou, China. Each plastic pot (height = 200 mm, a bottom diameter = 160 mm and a top diameter = 213 mm) contains 3.5 kg of naturally air-dried soil through a 2-mm sieve, and a filter paper beneath the bottom to prevent the soil loss. Subsequently, we set up eight treatments with six replicates: unamended control soil (denoted by CK), soil + 1% (w/w) rice straw (denoted by S), soil + 1% (w/w) rice root (denoted by R), soil + 1% (w/w) rice straw + 1% (w/w) rice roots (denoted by SR), and added 1% (w/w) pig manure to CK, S, R and SR, respectively. Maize was planted as the test crop in each plastic pot, and the average greenhouse temperature was maintained at 25 °C, the growing process of maize lasted 136 days. Watering was done little and frequently in the beginning to ensure smooth seed germination. During growth, a moderate amount of deionized water was sprayed on the surface of each pot weekly so that the soil moisture content was sufficient for maize growth and metabolism.
Soil sampling
On April 22, 2022, soil samples were destructively collected at the end of the maturation period by drilling three cores (3 cm in diameter) from each plastic pot, and then six cores from two replicates were mixed to form a composite one. Each soil sample composite was homogenized and passed through a 2-mm sieve, and the sieved soil is divided into three parts. A portion of the soil sample was stored at 4 °C for subsequent content analysis of soil microbial biomass carbon (MBC), microbial biomass nitrogen (MBN), microbial biomass phosphorus (MBP), dissolving organic carbon (DOC), ammonium nitrogen (NH4+−N) and nitrate nitrogen (NO3−–N). A portion of soil sample was air-dried for determining soil pH, and contents of soil total carbon (TC), total nitrogen (TN), total phosphorus (TP) and available phosphorus (AP), and enzyme activities. The last portion of soil sample was freeze-dried to extract the phospholipid fatty acids (PLFA).
Soil properties and microbial biomass analysis
Soil pH was determined using the digital pH meter (Seven2Go, Mettler-Toledo Instruments (Shanghai) Co., Ltd, Shanghai, China) in a 1:2.5soil /water suspension after shaking at 250 rpm for 5 min. TC and TN contents were determined using a Vario micro-Cube elemental analyzer (Analyzer Vario MICRO cube, Elementar, Germany). Soil NO3−–N and NH4+−N were extracted by 2 M KCl and their contents were determined using a colorimetric method by an AutoAnalyser III continuous Flow Analyzer (Bran + Luebbe, AA3 AutoAnalyzer, German). MBC and MBN contents were determined using the chloroform fumigation extraction method and the extracted liquid was determined by a TOC analyzer (Multi C/N 3000, Analytik Jena, Germany)29. The TP content was determined by the antimony molybdenum anti-colorimetric method after perchloric acid digestion, MBP was extracted with 0.5 M NaHCO3 and its content was measured using the chloroform fumigation extraction method, AP was also extracted with 0.5 M NaHCO3, all of the production were detected by antimony molybdenum anti-colorimetric method described by Bray and Kurtz30. DOC was extracted with 0.5 M K2SO4 after shaking at 200 rpm for 1 h, then the filtrates were used to determine the DOC content in the Vario TOC element analyzer (Elementar, Hanau, Germany).
Soil enzyme activities analysis
Soil enzyme (cellulase, β-1,4-glucosidase [BG], Urease, N-acetyl-β-D-glucosaminidase [NAG] and acid phosphomonoesterase [AcP]) activities were determined by using an enzyme activity detection kit (Nanjing Mol Farming Biotechnology Co., Ltd.). The substrates for enzymes were as follows: Cellulase, BG, Urease, NAG and AcP were assayed with the 3.5-dinitrosalicylic acid (DNS)31, ρ-nitrophenyl-β-D-glucopyranoside32, urea33, ρ-nitrophenyl-N-acetyl-β-d-glucosaminide34, and 4-methylumbelliferyl-phosphate33, respectively. For detailed determination of the above five soil enzymes refers to the enzyme activity detection kit procedures.
Analysis of PLFA
Microbial community structure was assessed by using phospholipid fatty acids (PLFA) analysis21,35. Briefly, the PLFAs were extracted from 8 g of freeze-dried soil with chloroform–methanol–citrate buffer mixture (1:2:0.8), followed by elution with chloroform, acetone and methanol. After the organic phase separation, purification, and methyl esterification, the PLFAs were analyzed using an Agilent 7890A GC (Version 6.2, MIDI Inc., Newark, Delaware, USA). MIDI software (“Sherlock Microbial Identification System”) was used to identify the individual PLFAs peaks and quantify the peak areas with reference to the internal standard peak (19:0) of known concentration. The biomarkers mainly included gram-positive bacteria (a11:0, a13:0, i14:0, i15:0, a15:0, i15:1 ω6c, i16:0, i17:0, a17:0, i17:1 ω9c and i18:0), the gram-negative bacteria (16:1 ω9c, 16:1 ω7c, 17:1 ω8c, 18:1 ω7c, 18:1 ω5c, cy17:0 ω7c and cy19:0 ω7c), saprophytic fungi (18:1 ω9c and 18:2 ω6c), arbuscular mycorrhizal fungi (16:1 ω5c) and actinomycetes (16:0 10-methyl, 17:0 10-methyl, 17:1 ω7c 10-methyl, 18:0 10-methyl and 20:0 10-methyl). The sum of gram-positive bacteria (G+), gram-negative bacteria (G−) and non-specific bacteria (14:0, 15:0, 15:0 DMA, 16:0, 16:3 ω6c and 18:0) was used as total bacteria. All the above PLFAs were considered to be representative of the total PLFAs of the soil microbial community21. The PLFAs abundance was expressed in nmol g−1 dry soil.
Statistical analysis
All values were based on the weight of oven-dried soil (105℃) and the data were averaged from three replicates with standard deviation. The effect of EOM on soil properties, enzyme activities, microbial biomass and community was investigated by two-way analysis of variance (Two-way ANOVA) using the “car” package in R software. When the interactions between crop residues and pig manure were significant, they were further analyzed by using a one-way ANOVA (Duncan or multiple comparison) and independent-samples t-test, respectively. The only main effect was compared when the interaction was not significant. The significance level was set at p < 0.05.
Pearson correlation analysis was performed to analyze the relationship between soil microbial community and enzyme activities, and the “corrplot” package in R was used to analyze soil properties, microbial biomass, enzyme activity and microbial community. Redundancy analysis (RDA) was conducted by using the “vegan” package in R software to explore the relationships between soil microbial community, soil chemical and biological properties.
Results
Soil properties and microbial biomass under the EOM additions
Two-way ANOVA showed that crop residues and pig manure addition, and their interactions significantly influenced the contents of MBN and MBP (p < 0.05, Figs. 1e and f). TC and MBN contents significantly increased in both S and SR treatments compared with CK regardless of whether pig manure was added (Figs. 1a and e). Pig manure addition increased TC and MBN contents in all the treatments compared with the corresponding treatments without pig mnaure, except for R treatment (Figs. 1a and e). Compared with CK, MBC content significantly increased and reduced in both S treatment and R treatment with pig manure, respectively (Fig. 1d), while significantly increased in SR treatment without pig manure (Fig. 1d). There were no significant differences in TP, DOC, AP, NH4+–N and NO3−–N contents, and pH value among crop residues treatments compared with CK regardless of whether pig manure was added, except for DOC and NO3−–N contents in SR treatment without pig manure (Figs. 1c, 2a–d and 3, and Figs. 2a and d). However, pig manure addition increased contents of TP, DOC, AP and NH4+–N, and pH value in all the treatments compared with the corresponding treatments without pig manure (Figs. 1c, 2a–c and 3). Compared with CK, TP and MBP contents were not significantly influenced by crop residues regardless of whether pig manure was added (Figs. 1c and f), while TN content significantly increased in crop residue treatment without pig manure, and MBP content was significantly higher in S treatment with pig manure, respectively (Figs. 1b and f).
Effect of exogenous organic matter (EOM) on the contents of soil total nutrients (carbon, nitrogen and phosphorus) and microbial biomass (carbon, nitrogen and phosphorus), and the plots were labeled without pig manure (without), with pig manure (with). The data were expressed by mean ± standard error (n = 3). Pig manure × Treatment: interaction between crop residues without and with pig manure. The different lowercase and uppercase letters denote the significant difference among different treatments without and with pig manure addition at 0.05 level, respectively. The two-way ANOVA results were indicated by NS non-significant, *p < 0.05, **p < 0.01, ***p < 0.001.
Effect of EOM on contents of soil available nutrients (DOC, AP, NH4+–N and NO3−–N), and the plots were labeled without pig manure (without), with pig manure (with). The data were expressed by mean ± standard error (n = 3). Pig manure × Treatment: interaction between crop residues without and with pig manure. The different lowercase and uppercase letters denote the significant difference among different treatments without and with pig manure addition at 0.05 level, respectively. The two-way ANOVA results were indicated by NS non-significant, *p < 0.05, **p < 0.01, ***p < 0.001.
Effect of EOM on soil pH, and the plots were labeled without pig manure (without), with pig manure (with). The data were expressed by mean ± standard error (n = 3). Pig manure × Treatment: interaction between crop residues without and with pig manure. The different lowercase and uppercase letters denote the significant difference among different treatments without and with pig manure addition at 0.05 level, respectively. The two-way ANOVA results were indicated by NS non-significant, *p < 0.05, **p < 0.01, ***p < 0.001.
Soil enzyme activities and microbial community composition under EOM additions
Compared with CK, both S and SR significantly increased cellulase and BG activities (Figs. 4a and b) regardless of whether pig manure was added. Urease and AcP activities were significantly higher in the S treatment with pig manure than that in CK (Figs. 4c and e). Urease and NAG activities were significantly higher in S and SR treatment without pig manure than those in CK (Figs. 4c and d), while the AcP activity significantly increased in SR treatment without pig manure (Fig. 4e). Pig manure addition increased activities of BG, urease, NAG and AcP in all the treatments compared with the corresponding treatments without pig manure, except for AcP activity in R treatment (Figs. 4b–e).
Effect of EOM on soil enzymes activities, and the plots were labeled without pig manure (without), with pig manure (with). The data were expressed by mean ± standard error (n = 3). Pig manure × Treatment: interaction between crop residues without and with pig manure. The different lowercase and uppercase letters denote the significant difference among different treatments without and with pig manure addition at 0.05 level, respectively. The two-way ANOVA results were indicated by NS non-significant, *p < 0.05, **p < 0.01, ***p < 0.001.
Two-way ANOVA showed that crop residues and pig manure addition, and their interactions significantly influenced G+/G− ratio (p < 0.05, Fig. 5f). Compared with CK, the S treatment with pig manure significantly increased the contents of total PLFA, and actinomycetal, G+, G-, fungi and bacteria PLFAs (Figs. 5a, b,d, e, g, h), and the S treatment without pig manure significantly increased G+ and bacteria PLFAs contents (Figs. 5d and h), respectively, while S and SR treatment without pig manure significantly increased AMF PLFA content, G+/G- ratio, and significantly reduced F/B ratio, respectively (Figs. 5c, f and i). Pig manure addition increased total PLFA, and actinomycetal, AMF, G+, G−, fungi and bacteria PLFAs contents in all the treatments compared with the corresponding treatments without pig manure (Figs. 5a–e, g and h).
Effect of EOM on PLFA content of soil microbial communities, and the plots were labeled without pig manure (without), with pig manure (with). The data were expressed by mean ± standard error (n = 3). Pig manure × Treatment: interaction between crop residues without and with pig manure. The different lowercase and uppercase letters denote the significant difference among different treatments without and with pig manure addition at 0.05 level, respectively. The two-way ANOVA results were indicated by NS non-significant, *p < 0.05, **p < 0.01, ***p < 0.001.
Relationship among soil microbial communities, soil properties and enzyme activities
The results of Pearson correlation analysis (Table 2) showed that BG activity was significantly positively correlated with G+, AMF PLFAs contents and G+/G− ratio, while significantly negatively correlated with F/B ratio; cellulase activity was significantly positively correlated with G+/G− ratio, and negatively correlated with F/B ratio; urease and NAG activities were significantly positively correlated with AMF PLFA content and G+/G− ratio, and negatively correlated with F/B ratio under crop residues without pig manure addition, respectively. For crop residues with pig manure addition, BG activity was significantly positively correlated with G+, G−, actinomycetal and bacteria PLFAs contents; AcP activity was significantly positively correlated with G+, G− and bacteria PLFAs contents, respectively.
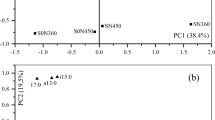
The changes in soil microbial communities under crop residues with or without pig manure were analyzed by the redundancy analysis (RDA) (Fig. 6). RDA indicated that 37.16% and 9.70% of the variability in microbial community composition could be explained by the first and second principal components under crop residues with pig manure treatment, respectively (Fig. 6b and Table 3). Similarly, the changes in soil microbial community composition under crop residues without pig manure treatment was shown with the first and second principal components (RDA1 and RDA2) accounting for 54.40% and 15.89% of the variance, respectively (Fig. 6a and Table 3). This analysis also revealed that pH, the contents of NO3−–N, NH4+–N and DOC were significantly associated with soil microbial community composition under crop residues without pig manure addition (Fig. 6a and Table 3), whereas the correlation between explanatory variables and microbial community composition under crop residues with pig manure addition was not significant (Fig. 6b and Table 3).
Redundancy analysis (RDA) between soil microbial communities and soil environmental parameters under EOM addition, and the plots were labeled without pig manure (a), and with pig manure (b). NO3−–N nitrate nitrogen; NH4+–N ammonia nitrogen, DOC dissolved organic carbon, TP total phosphorus, MBC microbial biomass carbon, G+ gram-positive bacterial, G− gram-negative bacteria, AMF arbuscular mycorrhizal fungi, G+/G− the ratio of gram-positive bacteria to gram-negative bacteria, AcP acid phosphomonoesterase.
Discussion
Effect of EOM on soil properties and microbial biomass
Returning crop residues to field can provide abundant essential nutrients such as carbon (C), nitrogen (N) and phosphorus (P), thereby enhancing soil fertility36,37. Our results indicated that both S and SR treatment significantly increased TC and MBN contents regardless of whether pig manure was added (Figs. 1a, e), and S treatment with pig manure and SR without pig manure significantly increased MBC content (Fig. 1d). These results suggest that crop straw can influence soil microbial biomass and activities by altering the supply of C and other nutrients. We also found that MBC significantly increased in S treatment and significantly reduced in R treatment with pig manure, while significantly increased in SR treatment without pig manure compared with CK, respectively (Figs. 1d). The above results might be attributed to the carbon to nitrogen ratio (C/N) and the biochemical composition of crop residues. Previous studies reported that crop residues with high C/N of rice straw might cause soil microorganisms to compete with the crops for N, while high C/N might also reduce the accumulation of available N through microbial immobilization processes38,39. Conversely, the root with a low C/N (R treatment) might enhance the mineralization, thereby decreasing MBC and MBN contents (Figs. 1d, e). Our study found the interaction between crop residues and pig manure significantly influenced MBN and MBP contents (Figs. 1e, f), suggesting that pig manure addition might alter soil biological properties and regulated the keystone microbial groups involved in crop residues decomposition40.
Moreover, the DOC content increased mainly due to the release of soluble organic matter and some nutrients (e.g., N, P) from the C inputs or crop residues decomposition, which stimulated microbial activities and growth41. In our study, pig manure addition increased TP, DOC, AP and NH4+–N contents, and pH value in all the treatments (Figs. 1c, 2a–c, 3), suggesting that pig manure addition improves soil fertility by increasing the organic matter, nutrients availability (e.g., available N and P), and elevating soil pH. In addition, pig manure is enriched with high organic matter, N and P nutrient contents10, and could further increase DOC and TP contents to higher levels after applied to the soil.
Effect of EOM on soil enzyme activities
Soil enzymes are considered as indicators of soil fertility, and are involved in SOM transformation and nutrients turnover42. Changes in soil fertility depend on the changes in soil enzymes activities, and some hydrolytic enzymes such as cellulase, BG, urease, NAG and AcP are associated with soil C, N and P cycling12. Compared with CK, both S and SR significantly increased cellulase and BG activities (Figs. 4a, b) regardless of whether pig manure was added, and urease activities significantly increased in S with and without pig manure, and NAG activities were significantly higher in S and SR treatment without pig manure (Figs. 4c, d), respectively. This may have occurred because the microbial population and microbial biomass C or N were increased by EOM addition, which provided organic matter that is used as a substrate for soil enzymes43,44. It was recently reported that the straw with easily decomposable matter could improve the availability of P in soils by providing energy materials for microbial activities and abundant substrates for enzymatic processes45. Our study revealed that the AcP activity significantly increased in the S treatment with pig manure and SR treatment without pig manure than that in CK, respectively (Fig. 4e). Pig manure addition increased BG, urease, NAG and AcP activities in all the treatments, except for AcP activity in R treatment (Figs. 4b–e). This is probably because pig manure addition affected the unstable pool of soil organic nutrients, thereby influencing enzyme activities46. However, soil enzymes activities are regulated by soil C and N levels, and the lower decomposition rates of roots might limit the substrate use efficiency for soil enzymes in R treatment. Therefore, crop residues and pig manure addition might affect soil enzyme activities by influencing the quantity and quality of C inputs.
Effect of EOM on soil microbial community composition
Compared with CK, the presence of rice straw increased the total PLFA, and actinomycetal, G+, G−, fungi, bacteria and AMF PLFAs contents, and G+/G− ratio, and reduced F/B ratio, respectively. These phenomena were attributed to the preference of different microorganisms for C substrates. Specifically, total PLFA significantly increased in the S treatment with pig manure compared with CK (Fig. 5a), this is probably because crop residues (e.g., straw, roots) and pig manure could provide energy and substrates for microbial metabolisms and growth12,46. Previous studies have reported that actinomycetal are efficient decomposers of C-poor compounds, so they exhibit the highest activity when soil is C-poor due to N limitation47. We observed a significant increase in actinomycetal in S treatment with pig manure compared with CK (Fig. 5b), this is probably because the straw and pig manure with a higher C/N ratio might cause N limitation by soil microorganism competing with crops for N. Our results indicated that AMF PLFA content significantly increased in S and SR treatment without pig manure (Fig. 5c), probably because AMF could also recycle and distribute nutrients from older mycelia to newly grown mycelia48, so they are less affected by active nutrients. Our results also showed that the interaction between crop residues and pig manure significantly increased the G+/G− ratio in S and SR treatment without pig manure (Fig. 5f), suggesting that the presence of rice straw increased the relative abundance of G+ bacteria more than that of G− bacteria (Figs. 5d, e). This might be because G+ bacteria are more stress-tolerant than G− bacteria, and G+ bacteria prefer to use recalcitrant substrates, whereas G− bacteria prefer to use relative labile residue C49,50.
Bacteria and fungi are the two main decomposers, of which bacteria are better adapted to the metabolism of easily decomposed organic matter, and fungi have an advantage in soil with high contents of recalcitrant organic matter (e.g., cellulose and lignin)51,52. In our study, bacteria and fungi PLFAs contents significantly increased in S treatment with pig manure while bacteria PLFA content significantly increased only in S treatment without pig manure (Figs. 5g, h), indicating that pig manure could provide abundant available nutrients for bacteria and fungi, whereas straw was readily degradable to release nutrients that can meet the metabolic requirement of bacteria. The F/B ratio is capable to characterize the most active microbial community for degrading crop residues53. In addition, our results found that the S and SR treatment significantly decreased the F/B ratio under no pig manure (Fig. 5i), and a significantly negative correlation existed between the F/B ratio and the C, N acquisition enzymes (e.g., cellulase, BG, urease and NAG) under straw without pig manure (Table 1). These results indicate that enzymatic hydrolysis released available nutrients for microbial growth and metabolism, resulting in a higher level of bacterial community than the fungal community.
Potential relationships between soil microbial communities and soil enzymes activities and soil properties
In our study, crop residues with or without pig manure treatment significantly affected the content of total PLFAs and altered microbial community composition (Figs. 5, 6). These changes might attribute to the organic materials containing labile and recalcitrant organic C54, which could promote microbial metabolism and growth. In addition, the increases in microbial biomass (C, N and P) and available nutrients (C, N and P) contents were observed under crop residues combined with pig manure addition (Figs. 1, 2), which could provide energy and substrates for microbial activity. This result was further confirmed by RDA analysis, which revealed that the relationships between microbial community composition and soil properties reached 46.86% and 70.39% of the variability under crop residues with and without pig manure treatment, respectively (Fig. 6). Specifically, the changes in the microbial community composition were dependent upon pH, NO3−–N, NH4+–N and DOC contents under crop residues without pig manure treatment, whereas the correlation between explanatory variables and microbial community composition under crop residues with pig manure addition was not significant (Fig. 6b; Table 3), indicating that microbial community was not limited by the available nutrients under pig manure addition. Moreover, we also found that the higher microbial biomass (C, N and P) and available nutrients (C, N and P) contents were observed in pig manure treatment compared with the treatments without pig manure (Fig. 1, 2). This was probably because the relatively abundant energy and substrates were enough to maintain the microbial metabolism and activity.
Generally, nutrient turnover was related to microbial community composition and extracellular enzymes activities, and the correlation between microbial community composition and extracellular enzymes was significant46,55,56. Moreover, Tasoff et al.57 have predicted the increase in enzyme production when simple nutrients are scarce and complex nutrients are abundant by using economics of microbial metabolism. In our study, the F/B ratio was significantly negatively correlated with the C, N acquisition enzymes (i.e., cellulase, BG, urease and NAG) activities under straw treatment without pig manure (Table 1), indicating that changes in microbial community composition might depend on soil condition such as C and N availability58, as well as competition for available nutrients with C, N acquisition enzymes. However, the G+/G− ratio was significantly positively correlated with the C, N acquisition enzymes (i.e., cellulase, BG, urease and NAG) under straw without pig manure (Table 1), suggesting that straw addition shifted the microbial community towards a more G+ bacteria-dominated stage59. Previous study also found that G+ bacteria are capable of secreting enzymes to decompose recalcitrant C, but that they require higher N levels60. Furthermore, organic matter mineralization is necessary to provide energy and substrates for G+ with the depletion of available nutrients, whereas the recalcitrant compounds are more readily degraded by extracellular enzymes61,62. Fungi could degrade cellulose or recalcitrant components (e.g., recalcitrant C, organic N and P polymers) by secreting soil extracellular enzymes51,63. In addition, mycorrhizal fungi could form symbiotic relationships with plant roots facing nutrients deficiency stress, thus releasing mineralized nutrients such as N and P64,65. In our study, AMF PLFA content was significantly positively with BG, urease and NAG activities under crop residues without pig manure (Table 1), indicating that the mineralization process requires mycorrhizal fungi to produce extracellular enzymes to decompose C and N compounds, and then facilitate microbial metabolisms and growth. Bacteria play an important role in organic matter decomposition, where G+ bacteria are more suitable for soil with low substrate availability66. The early catabolism of G+ bacteria is dependent on more labile substrates, and the labile substrates require BG involvement in the crop residues mineralization67. In addition, we also observed the positive correlation between G+ bacteria PLFA content and BG activity (Table 1). These results indicated that G+ bacteria play more important roles in the mineralization process of crop residues treatment without no pig manure addition.
The relationship between microbial community composition and enzyme activities exhibited difference under crop residues treatments with pig manure addition. That’s probably because pig manure can increase soil fertility by releasing large amounts of organic matter, N and P that are beneficial to microorganisms68,69,70. Moreover, DOC, as an unstable C fraction, is considered to be the dominant source of substrates and energy for microorganisms71. We found that pig manure significantly increased DOC content (Fig. 2a) and BG activity (Fig. 4b). The higher BG activity further promoted the mineralization of crop residues with increasing energy and substrates. Although crop residues decomposition is a complex process, pig manure provides durable energy and nutrients for a good microbial community structure. Our finding indicated that the BG activity was significantly positively with the PLFAs contents of G+ bacteria, G− bacteria, actinomycetal and bacteria under crop residues with pig manure (Table 2), this might be a direct effect of pig manure. It has been confirmed that pig manure addition can increase the total P and soluble P contents in soil, and provide a good nutrient source to supplement soil P for crops growth70. Moreover, the increase in moderately unstable and stable P is possibly due to the adsorption or precipitation process, some of which may also come from pig manure and may not be readily absorbed by plants72. In our study, the PLFAs contents of bacteria (including G+ bacteria and G− bacteria) were significantly and positively correlated with AcP activity (Table 2), suggesting that pig manure could stimulate the activity of AcP by providing good P sources for microorganisms, as well as C, N sources. Thus, pig manure not only provides rich nutrients (C, N and P) for microorganisms but also induces the higher of enzyme activities to maintain microbial activities and diversity.
Conclusions
This study showed that exogenous organic matter addition increased microbial biomass and enzymes activities. Relative to the below-ground roots (R) treatment, the high carbon to nitrogen ratio (C/N) of straw could reduce the accumulation of available nutrients through microbial immobilization process. Moreover, exogenous organic matter addition could provide substrates for soil enzymes by influencing the quantity and quality of carbon inputs. Compared with CK, the presence of straw increased the contents of total PLFA, and actinomycetal, gram-positive bacteria, gram-negative bacteria, fungi, bacteria and AMF PLFAs, gram-positive bacteria to gram-negative bacteria ratio (G+/G−), and reduced fungi to bacteria ratio (F/B), respectively, indicating the preference of different microorganisms for C substrates. Redundance analysis confirmed that pH, nitrate nitrogen (NO3−–N), ammonium nitrogen (NH4+–N) and dissolve organic carbon (DOC) contents were dominant factors in regulating the changes of soil microbial community under crop residue treatment without the pig manure, whereas the nutrients were relatively abundant to maintain microbial metabolism and activity under crop residues with the pig manure treatment. Overall, the combination of above-ground straw and pig manure was more beneficial to improve soil carbon and nitrogen availability, increase enzyme activities and facilitate microbial biomass and activity compared with no pig manure. Our findings suggest that the combination of above-ground straw and pig manure is a better alternative for improving the functions of soil ecosystem, especially for no-tillage with straw management system.
Data availability
The dataset generated in the course of the current study can be obtained from the corresponding author upon reasonable request.
References
Liu, X. et al. Soil carbon sequestration in paddy field and its simultaneous mineralization to supply available nutrients for the crops are affected by no-tillage with straw management: A meta-analysis. Appl. Soil Ecol. 188, 104850. https://doi.org/10.1016/j.apsoil.2023.104850 (2023).
Wang, Q. C. et al. Converting rice paddy to upland fields decreased plant lignin but increased the contribution of microbial residue to SOC. Geoderma 425, 116079. https://doi.org/10.1016/j.geoderma.2022.116079 (2022).
Jin, Z. Q. et al. Effect of straw returning on soil organic carbon in rice-wheat rotation system: A review. Food Energy Secur. 9, 66. https://doi.org/10.1002/fes3.200 (2020).
Trong Hung, D., Hughes, H. J., Keck, M. & Sauer, D. Rice-residue management practices of smallholder farms in Vietnam and their effects on nutrient fluxes in the soil-plant system. Sustainability 11, 1641. https://doi.org/10.3390/su11061641 (2019).
Otero-Jiménez, V., del Pilar Carreno-Carreno, J., Barreto-Hernandez, E., van Elsas, J. D. & Uribe-Velez, D. Impact of rice straw management strategies on rice rhizosphere microbiomes. Appl. Soil Ecol. 167, 104036. https://doi.org/10.1016/j.apsoil.2021.104036 (2021).
Lu, Y., Watanabe, A. & Kimura, M. Carbon dynamics of rhizodeposits, root- and shoot-residues in a rice soil. Soil Biol. Biochem. 35, 1223–1230. https://doi.org/10.1016/S0038-0717(03)00184-6 (2003).
Hooker, B. A., Morris, T. F., Peters, R. & Cardon, Z. G. Long-term effects of tillage and corn stalk return on soil carbon dynamics. Soil Sci. Soc. Am. J. 69, 188–196. https://doi.org/10.2136/sssaj2005.0188 (2005).
Puget, P. & Drinkwater, L. E. Short-term dynamics of root-and shoot-derived carbon from a leguminous green manure. Soil Sci. Soc. Am. J. 65, 771–779. https://doi.org/10.2136/sssaj2001.653771x (2001).
Li, P. et al. Responses of microbial communities to a gradient of pig manure amendment in red paddy soils. Sci. Total Environ. 705, 135884. https://doi.org/10.1016/j.scitotenv.2019.135884 (2020).
Cai, A. et al. Manure acts as a better fertilizer for increasing crop yields than synthetic fertilizer does by improving soil fertility. Soil Till. Res. 189, 168–175. https://doi.org/10.1016/j.still.2018.12.022 (2019).
Mallmann, F. J. K. et al. Modeling field-scale vertical movement of zinc and copper in a pig slurry-amended soil in Brazil. J. Hazard. Mater. 243, 223–231. https://doi.org/10.1016/j.jhazmat.2012.10.022 (2012).
Zhao, S. et al. Changes in soil microbial community, enzyme activities and organic matter fractions under long-term straw return in north-central China. Agric. Ecosyst. Environ. 216, 82–88. https://doi.org/10.1016/j.agee.2015.09.028 (2016).
Burns, R. G. et al. Soil enzymes in a changing environment: Current knowledge and future directions. Soil Biol. Biochem. 58, 216–234. https://doi.org/10.1016/j.soilbio.2012.11.009 (2013).
Bandick, A. K. & Dick, R. P. Field management effects on soil enzyme activities. Soil Biol. Biochem. 31, 1471–1479. https://doi.org/10.1016/S0038-0717(99)00051-6 (1999).
Zhang, Y. et al. Stoichiometric analyses of soil nutrients and enzymes in a Cambisol soil treated with inorganic fertilizers or manures for 26 years. Geoderma 353, 382–390. https://doi.org/10.1016/j.geoderma.2019.06.026 (2019).
Pamidipati, S. & Ahmed, A. Cellulase stimulation during biodegradation of lignocellulosic residues at increased biomass loading. Biocatal. Biotrans. 37, 261–267. https://doi.org/10.1080/10242422.2018.1508284 (2019).
Sinsabaugh, R. L. et al. Stoichiometry of soil enzyme activity at global scale. Ecol. Lett. 11, 1252–1264 (2008).
Kandeler, E., Poll, C., Frankenberger, W. T., Tabatabai, M. A. & Dick, R. Nitrogen cycle enzymes. Meth. Enzymol. 9, 211–245. https://doi.org/10.2136/sssabookser9.c10 (2011).
Turner, B. L., Hopkins, D. W., Haygarth, P. M. & Ostle, N. β-Glucosidase activity in pasture soils. Appl. Soil Ecol. 20, 157–162. https://doi.org/10.1016/s0929-1393(02)00020-3 (2002).
Paterson, E., Sim, A., Osborne, S. M. & Murray, P. J. Long-term exclusion of plant-inputs to soil reduces the functional capacity of microbial communities to mineralise recalcitrant root-derived carbon sources. Soil Biol. Biochem. 43, 1873–1880. https://doi.org/10.1016/j.soilbio.2011.05.006 (2011).
Wu, G. et al. Oxidases and hydrolases mediate soil organic matter accumulation in chernozem of northeastern China. Geoderma 403, 115206. https://doi.org/10.1016/j.geoderma.2021.115206 (2021).
Saleem, M., Hu, J. & Jousset, A. More than the sum of its parts: microbiome biodiversity as a driver of plant growth and soil health. Annu. Rev. Ecol. Evol. S 50, 145–168 (2019).
Paterson, E. et al. Labile and recalcitrant plant fractions are utilised by distinct microbial communities in soil: Independent of the presence of roots and mycorrhizal fungi. Soil Biol. Biochem. 40, 1103–1113. https://doi.org/10.1016/j.soilbio.2007.12.003 (2008).
Marschner, P., Umar, S. & Baumann, K. The microbial community composition changes rapidly in the early stages of decomposition of wheat residue. Soil Biol. Biochem. 43, 445–451. https://doi.org/10.1016/j.soilbio.2010.11.015 (2011).
Lundquist, E., Jackson, L., Scow, K. & Hsu, C. Changes in microbial biomass and community composition, and soil carbon and nitrogen pools after incorporation of rye into three California agricultural soils. Soil Biol. Biochem. 31, 221–236. https://doi.org/10.1016/S0038-0717(98)00093-5 (1999).
Guo, Z. et al. Fertilization regime has a greater effect on soil microbial community structure than crop rotation and growth stage in an agroecosystem. Appl. Soil Ecol. 149, 103–510. https://doi.org/10.1016/j.apsoil.2020.103510 (2020).
Kemmitt, S. J. et al. Mineralization of native soil organic matter is not regulated by the size, activity or composition of the soil microbial biomass—A new perspective. Soil Biol. Biochem. 40, 61–73. https://doi.org/10.1016/j.soilbio.2007.06.021 (2008).
Hartmann, M., Frey, B., Mayer, J., Mäder, P. & Widmer, F. Distinct soil microbial diversity under long-term organic and conventional farming. ISME J. 9, 1177–1194. https://doi.org/10.1038/ismej.2014.210 (2015).
Joergensen, R. G. The fumigation-extraction method to estimate soil microbial biomass: Calibration of the kEC value. Soil Biol. Biochem. 28, 25–31. https://doi.org/10.1016/0038-0717(95)00102-6 (1996).
Bray, R. H. & Kurtz, L. T. Determination of total, organic, and available forms of phosphorus in soils. Soil Sci. 59, 39–46 (1945).
Miller, G. L., Blum, R., Glennon, W. E. & Burton, A. L. Measurement of carboxymethylcellulase activity. Anal. Biochem. 1, 127–132. https://doi.org/10.1016/0003-2697(60)90004-X (1960).
Marx, M.-C., Wood, M. & Jarvis, S. A microplate fluorimetric assay for the study of enzyme diversity in soils. Soil Biol. Biochem. 33, 1633–1640. https://doi.org/10.1016/S0038-0717(01)00079-7 (2001).
Tabatabai, M. Soil enzymes. In: Weaver R. W., Angle G. S. and Bottomley P. S. (eds.). Method of Soil Analysis, Part 2-Microbiological and Biochemical Properties. Soil Sci Soc Am, Madison, 775–833. https://doi.org/10.2136/sssabookser5.2.c37 (1994).
Saiya-Cork, K., Sinsabaugh, R. & Zak, D. The effects of long term nitrogen deposition on extracellular enzyme activity in an Acer saccharum forest soil. Soil Biol. Biochem. 34, 1309–1315. https://doi.org/10.1016/S0038-0717(02)00074-3 (2002).
Bossio, D. A. & Scow, K. Impacts of carbon and flooding on soil microbial communities: Phospholipid fatty acid profiles and substrate utilization patterns. Microb. Ecol. 35, 265–278. https://doi.org/10.1007/s002489900082 (1998).
Nie, J., Zhou, J.-M., Wang, H.-Y., Chen, X.-Q. & Du, C.-W. Effect of long-term rice straw return on soil glomalin, carbon and nitrogen. Pedosphere 17, 295–302. https://doi.org/10.1016/S1002-0160(07)60036-8 (2007).
Singh, R., Yadav, D. B., Ravisankar, N., Yadav, A. & Singh, H. Crop residue management in rice–wheat cropping system for resource conservation and environmental protection in north-western India. Environ. Dev. 22, 3871–3896. https://doi.org/10.1007/s10668-019-00370-z (2020).
Singh, B., Rengel, Z. & Bowden, J. W. Carbon, nitrogen and sulphur cycling following incorporation of canola residue of different sizes into a nutrient-poor sandy soil. Soil Biol. Biochem. 38, 32–42. https://doi.org/10.1016/j.soilbio.2005.03.025 (2006).
Li, F. et al. Fate of nitrogen from green manure, straw, and fertilizer applied to wheat under different summer fallow management strategies in dryland. Biol. Fertil. Soils 51, 769–780. https://doi.org/10.1007/s00374-015-1023-2 (2015).
Kong, Y. et al. DNA stable-isotope probing delineates carbon flows from rice residues into soil microbial communities depending on fertilization. Appl. Environ. Microbiol. 86, e02151-e2119. https://doi.org/10.1128/AEM.02151-19 (2020).
Wang, W., Lai, D. Y. F., Wang, C., Pan, T. & Zeng, C. Effects of rice straw incorporation on active soil organic carbon pools in a subtropical paddy field. Soil Till. Res. 152, 8–16. https://doi.org/10.1016/j.still.2015.03.011 (2015).
Li, T. et al. Influence of green manure and rice straw management on soil organic carbon, enzyme activities, and rice yield in red paddy soil. Soil Till. Res. 195, 104–428. https://doi.org/10.1016/j.still.2019.104428 (2019).
Li, H. et al. Variations in soil bacterial taxonomic profiles and putative functions in response to straw incorporation combined with N fertilization during the maize growing season. Agric. Ecosyst. Environ. 283, 106–578. https://doi.org/10.1016/j.agee.2019.106578 (2019).
Akhtar, K. et al. Changes in soil enzymes, soil properties, and maize crop productivity under wheat straw mulching in Guanzhong, China. Soil Till. Res. 182, 94–102. https://doi.org/10.1016/j.still.2018.05.007 (2018).
Yang, C. & Lu, S. Straw and straw biochar differently affect phosphorus availability, enzyme activity and microbial functional genes in an Ultisol. Sci. Total Environ. 805, 150–325. https://doi.org/10.1016/j.scitotenv.2021.150325 (2022).
Ozlu, E., Sandhu, S. S., Kumar, S. & Arriaga, F. J. Soil health indicators impacted by long-term cattle manure and inorganic fertilizer application in a corn-soybean rotation of South Dakota. Sci. Rep. 9, 1–11. https://doi.org/10.1038/s41598-019-48207-z (2019).
MacKenzie, M. D. & Quideau, S. A. Microbial community structure and nutrient availability in oil sands reclaimed boreal soils. Appl. Soil Ecol. 44, 32–41. https://doi.org/10.1016/j.apsoil.2009.09.002 (2010).
Ren, G. et al. Soil organic carbon and nitrogen fractions as affected by straw and nitrogen management on the North China Plain. Agric. Ecosyst. Environ. 342, 108248. https://doi.org/10.1016/j.agee.2022.108248 (2023).
Song, D. et al. Soil nutrient and microbial activity responses to two years after maize straw biochar application in a calcareous soil. Ecotoxicol. Environ. Saf. 180, 348–356. https://doi.org/10.1016/j.ecoenv.2019.04.073 (2019).
Song, D. et al. Combined biochar and nitrogen fertilizer change soil enzyme and microbial activities in a 2-year field trial. Eur. J. Soil Biol. 99, 103212. https://doi.org/10.1016/j.ejsobi.2020.103212 (2020).
Fontaine, S. et al. Fungi mediate long term sequestration of carbon and nitrogen in soil through their priming effect. Soil Biol. Biochem. 43, 86–96. https://doi.org/10.1016/j.soilbio.2010.09.017 (2011).
Wang, H. et al. Quality of fresh organic matter affects priming of soil organic matter and substrate utilization patterns of microbes. Sci. Rep. 5, 1–13. https://doi.org/10.1038/srep10102 (2015).
Li, P. et al. Rice straw decomposition affects diversity and dynamics of soil fungal community, but not bacteria. J. Soils Sed. 18, 248–258. https://doi.org/10.1007/s11368-017-1749-6 (2018).
Zhang, H., Ding, W., Yu, H. & He, X. Linking organic carbon accumulation to microbial community dynamics in a sandy loam soil: result of 20 years compost and inorganic fertilizers repeated application experiment. Biol. Fertil. Soils 51, 137–150. https://doi.org/10.1007/s00374-014-0957-0 (2015).
Ågren, G., Hyvönen, R., Berglund, S. & Hobbie, S. Estimating the critical N:C from litter decomposition data and its relation to soil organic matter stoichiometry. Soil Biol. Biochem. 67, 312–318. https://doi.org/10.1016/j.soilbio.2013.09.010 (2013).
Meng, C. et al. Global meta-analysis on the responses of soil extracellular enzyme activities to warming. Sci. Total Environ. 705, 135992. https://doi.org/10.1016/j.scitotenv.2019.135992 (2020).
Tasoff, J., Mee, M. T. & Wang, H. H. An economic framework of microbial trade. PLoS One 10, e0132907. https://doi.org/10.1371/journal.pone.0132907 (2015).
Prosser, J. I. et al. The role of ecological theory in microbial ecology. Nat. Rev. Microbiol 5, 384–392. https://doi.org/10.1038/nrmicro1643 (2007).
Lu, W. & Zhang, H. Response of biochar induced carbon mineralization priming effects to additional nitrogen in a sandy loam soil. Appl. Soil Ecol. 96, 165–171. https://doi.org/10.1016/j.apsoil.2015.08.002 (2015).
Treseder, K. K., Kivlin, S. N. & Hawkes, C. V. Evolutionary trade-offs among decomposers determine responses to nitrogen enrichment. Ecol. Lett. 14, 933–938. https://doi.org/10.1111/j.1461-0248.2011.01650.x (2011).
Brant, J. B., Sulzman, E. W. & Myrold, D. D. Microbial community utilization of added carbon substrates in response to long-term carbon input manipulation. Soil Biol. Biochem. 38, 2219–2232. https://doi.org/10.1016/j.soilbio.2006.01.022 (2006).
Waldrop, M. P. & Firestone, M. K. Microbial community utilization of recalcitrant and simple carbon compounds: Impact of oak-woodland plant communities. Oecologia 138, 275–284. https://doi.org/10.1007/s00442-003-1419-9 (2004).
Schneider, T. et al. Who is who in litter decomposition? Metaproteomics reveals major microbial players and their biogeochemical functions. ISME J. 6, 1749–1762. https://doi.org/10.1038/ismej.2012.11 (2012).
Kayama, M. & Yamanaka, T. Growth characteristics of ectomycorrhizal seedlings of Quercus glauca, Quercus salicina, and Castanopsis cuspidata planted on acidic soil. Trees 28, 569–583. https://doi.org/10.1007/s00468-013-0973-y (2014).
Smith, S. E., Smith, F. A. & Jakobsen, I. Mycorrhizal fungi can dominate phosphate supply to plants irrespective of growth responses. Plant Physiol. 133, 16–20. https://doi.org/10.1104/pp.103.024380 (2003).
Kieft, T., Ringelberg, D. & White, D. Changes in ester-linked phospholipid fatty acid profiles of subsurface bacteria during starvation and desiccation in a porous medium. Appl. Environ. Microbiol. 60, 3292–3299. https://doi.org/10.1128/aem.60.9.3292-3299.1994 (1994).
Zhu, Z. et al. Microbial stoichiometric flexibility regulates rice straw mineralization and its priming effect in paddy soil. Soil Biol. Biochem. 121, 67–76. https://doi.org/10.1016/j.soilbio.2018.03.003 (2018).
Mandal, A., Patra, A. K., Singh, D., Swarup, A. & Masto, R. E. Effect of long-term application of manure and fertilizer on biological and biochemical activities in soil during crop development stages. Bioresour. Technol. 98, 3585–3592. https://doi.org/10.1016/j.biortech.2006.11.027 (2007).
Wang, Q. et al. Improvement of pig manure compost lignocellulose degradation, organic matter humification and compost quality with medical stone. Bioresour. Technol. 243, 771–777. https://doi.org/10.1016/j.biortech.2017.07.021 (2017).
Ye, D. et al. P accumulation of Polygonum hydropiper, soil P fractions and phosphatase activity as affected by swine manure. Appl. Soil Ecol. 86, 10–18. https://doi.org/10.1016/j.apsoil.2014.10.002 (2015).
Tian, J. et al. Effects of land use intensity on dissolved organic carbon properties and microbial community structure. Eur. J. Soil Biol. 52, 67–72. https://doi.org/10.1016/j.ejsobi.2012.07.002 (2012).
Malik, M. A., Marschner, P. & Khan, K. S. Addition of organic and inorganic P sources to soil-effects on P pools and microorganisms. Soil Biol. Biochem. 49, 106–113. https://doi.org/10.1016/j.soilbio.2012.02.013 (2012).
Acknowledgements
The authors would like to thank all the participants colleagues involved in this work. We also thank the reviewers and editors for their feedback, which helped us improve the manuscript.
Funding
This study was funded by Key-Area Research and Development Program of Guangdong Province(Grant no. 2021B0202030002); Science and Technology Planning Project of Guangdong Province of China (Grant no. 2019B030301007); Guangdong Provincial Special Project of Rural Revitalization Strategy (Document No. (2021) 12); the Innovation Team Construction Project of Modern Agricultural Industry Technology System of Guangdong Province (2022KJ105); the Joint Team Project of Guangdong Laboratory for Lingnan Modern Agriculture (Grant no. NT2021010).
Author information
Authors and Affiliations
Contributions
X.L.: Writing-original draft, Visualization, Data curation, Formal analysis, Investigation, Methodology, Writing-review & editing. Q.C.: Data curation, Formal analysis, Investigation, Methodology. H.Z.: Investigation, Methodology. Y.C.: Data curation, Writing-review & editing. F.Y.: Investigation, Methodology. Y.C.: Data curation, Investigation. J.Z.: Conceptualization, Writing-review & editing, Funding acquisition, Project administration, Supervision. All authors have reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Liu, X., Chen, Q., Zhang, H. et al. Effects of exogenous organic matter addition on agricultural soil microbial communities and relevant enzyme activities in southern China. Sci Rep 13, 8045 (2023). https://doi.org/10.1038/s41598-023-33498-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-023-33498-0
- Springer Nature Limited
This article is cited by
-
Lanthanide-doped zinc gallium oxide (ZnGa2O4) nanocrystals and their engineered photoluminescence
Macromolecular Research (2024)