Abstract
Tube-building larvae of non-biting midges, or chironomids, are considered bioindicators of water pollution. The larvae use benthic particles to make their tubes and create a respiratory current with the movement of their bodies inside the tubes. The tube length of the chironomid larvae varies depending on several physicochemical properties of the aquatic medium. Here we study the role of physicochemical parameters on the tube length from different field sites and in the laboratory. It appears that among different physicochemical factors, dissolved oxygen (DO) has a major role in determining the tube length of the larvae. A quantitative relationship between oxygen concentration and the tube length of larvae is presented here. Our study reveals a longer tube in aquatic media with oxygen deficiency and a shorter tube in those with higher oxygen. This result may help to assess the quality of water bodies and, in particular the status of DO.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Chironomids (Diptera: Chironomidae) play a major role in the aquatic food chain by maintaining a link between producer and primary carnivores. They are also considered as important biological tools for environment assessment. Chironomids are primary consumers and a source of food for the higher trophic levels1. Tube building is an important characteristic feature of chironomid larvae and the tubes protect the larvae from the effects of heavy metals and other pollutants to some extent2,3. Larvae make their tubes with salivary silk and soft benthic particles4. The composition of the tube of the larvae is similar to the benthic particles of the surrounding medium. The quality of building materials is more important than the size of particles, in upstream of lotic water5. Tubes help to collect food particles, assist larvae in producing respiratory currents, act as a refuge from predators and inhibit chemical toxicants6,7,8. Halpern et. al. reported tubes of these larvae protect them from predation by damselflies9.Tube building properties also can modify the quality of the substrate used for tube building10. As a bio-indicator species, larvae are used to understand the quality of the aquatic body. Chironomus plumosus and Polypedilum sp. have a great influence on de-nitrification and nitrogen turnover in lake deposits11. Different physicochemical factors such as water temperature, alkalinity, and benthic soil organic carbon have been found to influence the seasonal variation of body protein and the growth of chironomid larvae12,13. At the same time, some environmental parameters also influenced the tube-building ability of the chironomids. During the study, a strong relation between the tube length and DO was observed. As well as this feature will help the person who is involved in freshwater aquaculture to measure the DO by a standard scale without using any expensive instrument or titrimetric method. Keeping these features in mind, here an attempt has been made to find out the relationship between tube length and dissolved oxygen (DO).The work seems to be unique and novel for its own kind.
Materials and methods
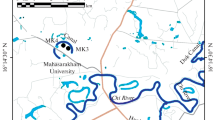
The Kanchrapara wastewater canal (KWC) (22°93 N, 88°47 E) and Kanchrapara fish culturing pond (KFP) (22°94 N, 88°45 E), Kanchrapara, North 24 Parganas, West Bengal, India, are chosen for this experiment in natural conditions to study the relationship between the tube length of chironomid larvae and different physicochemical parameters of the aquatic bodies. The distance between two water bodies is approximately five kilometres, and these water bodies may be an ideal model for the urban aquatic ecosystem due to their different quality of water. KWC is enriched with a high organic and nutritive content. Because of the fish culture in KFP, nutrient quality is also maintained in a controlled manner. In KFP, Wolffia sp. and Lemna minor, are observed mainly during the monsoon as floating vegetation. Characteristics of the aquatic bodies are presented in Table 1.
Physicochemical parameters like DO (dissolved oxygen), free CO2, water temperature, salinity, hardness, alkalinity, ammonia, nitrate, nitrite, iron, and benthic soil organic carbon are measured from the collected water and benthic soil samples following the APHA methods14 between 2015 and 2019. To measure the tube length of the larvae of Chironomus striatipennis Kieffer (1910) tubes were kept on blotting paper and measured by a standard measuring scale during the abundance study of these two sites. C. striatipennis is considered a model chironomid due to its high abundance in both the study sites.
Based on the observations under natural conditions, laboratory experiments were conducted to observe the relationship between DO and tube length of Chironomus larvae. For the laboratory study, larvae of Chironomus striatipennisKieffer (1910) are obtained from laboratory culture. Approximately 300 newly hatched larvae are placed into three experimental trays (TARSONS) (10ʺ × 8ʺ × 6ʺ) containing finely washed and autoclaved silt (@ 0.02 g soil per larva) at the bottom (Supplementary information 1). The larvae are reared up to the 2nd instar stage with filtered pond water (2 inches high in the tray) and DO is maintained with an aquarium air pump. The larvae are acclimatised to the artificial conditions and have reached the second instar stage in their tube. At this point, water from the experimental trays is replaced with treated pond water. The pond water is collected from the study area during sample collection and DO is measured. Treatment (25%, 50%, 75% grade of dilution) is done by mixing distilled water (boiled and cooled) to confirm the minimization of oxygen concentration, as boiling removes dissolved oxygen from water. DO is measured by the Winkler method15. Three differently treated waters are poured into three separate trays carefully. Artificial aeration is turned off and trays are covered with a cotton cloth to minimise the oxygenation of water from the air.
The control is maintained in another experimental tray following the same protocol with untreated pond water. DO is maintained by the aquarium air pump.
The level of DO and the tube length of the larvae are monitored at every 24-h interval for 7 consecutive days in both control and treated trays. The experiment was replicated twelve times.
Statistical analysis
To investigate the effect of aquatic physicochemical parameters on tube length, stepwise linear regression analysis is performed, considering tube length as a dependent variable while all aquatic physicochemical parameters are independent variables. In the step-wise method, the last model is considered with its highest R2 value along with standardised coefficients (β) of different parameters (Supplementary information 2). R2 is a statistical measure that represents the proportion of the variance for a dependent variable i.e. explained by the independent variable (s) in a regression model. A simple linear regression along with ANOVA was also performed to justify the relationship between DO and tube length of the larvae in both natural and laboratory conditions. A graphical model is also constructed for a more straightforward interpretation of the result. Statistical analyses are done using Microsoft Excel (2016) and SPSS software version 26 (SPSS Inc., Chicago, IL, USA).
Results
The stepwise linear regression revealed a significant negative relationship between tube length and DO (Model IV: β = − 2.779, Model VI: β = − 2.044; p < 0.001). A positive relation between tube length and free carbon dioxide (F CO2) also exists. Water temperature (W TEMP) and total hardness (TH), nitrate and iron of the water also shows the relationship with the tube length. But the priority is given to DO because of its high negative relation with tube length in both models (Table 2).
During the study period, the DO values of these two aquatic bodies were recorded in their natural state (Tables 3 and 4).
Results are also recorded from the experimental setup in the laboratory condition along with control. After running the experiment for seven days, the amount of DO and length of the tubes of every experimental setup are considered (Table 5).
A simple linear regression model is constructed between the DO (independent variable) and tube length (dependent variables) of the chironomid larvae in natural conditions as well as laboratory conditions (Figs. 1 and 2).
Under both natural and laboratory conditions (Figs. 1 and 2), a strong linear relationship is observed between the DO and the tube length. R (R for natural conditions: 0.650; R for laboratory conditions: 0.971) is the correlation coefficient value which is significantly high in both conditions. The R2 values (R2 for the natural condition: 0.423; R2 for laboratory condition: 0.943) indicate that 42% of the variance in tube length in the case of natural condition and 94% of the variance in tube length in laboratory condition can be accounted for by the dissolve oxygen measure. In both natural and laboratory conditions, the standard error is observed with a nominal value. The linear relationship is also confirmed by the significant F value (natural condition F = 69.04, p < 0.05; laboratory condition F = 772.81, p < 0.05). The scatter plot and linear regression both show significant inverse relationships between DO and larval tube lengths.
The result showed that in all the cases, the tube length of Chironomus striatipennis increased with the decrease of DO. Since KWC is a wastewater canal, the fluctuation of oxygen in KWC is much higher than in KFP throughout the year. The effect of fluctuating DO on the tube length of larvae is higher in KWC than in KFP. Similar effects have also been observed in laboratory conditions. Graphical representations are made to compare the tube length and DO of water at the two study sites and also with the laboratory data (Figs. 3 and 4).
Discussion
Chironomids have the ability to survive and reproduce in polluted environments, and thus they are included in many ecological studies where approaches may be taxonomic or functional16. The diversity of most macroinvertebrates is controlled by the oxygen level of water, but chironomids may survive in hypoxic conditions where the oxygen concentration may be less than 3 mg l−117. The current study demonstrates that changing seasons, as well as anthropogenic activities, have a significant impact on the levels of DO in aquatic bodies. As observed from the result, DO highly influences the tube length of the chironomid larvae. Since KWC is a wastewater canal, the average oxygen level is lower (5.24 ± 1.14 mg l−1) than KFP (6.63 ± 1.28 mg l−1) which is a normal fish culturing pond. It has also been observed that the average tube length of the chironomid larvae of KWC (8.66 ± 0.88 mm) is higher than KFP (7.68 ± 0.62 mm), which indicates that a low concentration of DO promotes the building of longer tubes in natural conditions. Similar observations were also observed in laboratory conditions. When the oxygen level (7.03 ± 0.41 mg l−1) in the experiment was kept in the normal range, there was negligible variation in tube length (7.61 ± 0.31 mm). But when the concentration of oxygen is gradually reduced by dilution, the tube length starts to increase accordingly, which is explained graphically in Fig. 4. The regression model of both the experimental conditions also supports the hypothesis that the tube length has an inverse relationship with DO. The scatter plot and simple linear regression confirmed the inverse relationship between DO and tube length (Figs. 1 and 2).
Chironomid larvae are able to grow in the polluted water of a wastewater pond as dominant macroinvertebrates18. It is observed that those larvae living in the sand tubes are more susceptible to chemical pollutants than the larvae living in silt tubes7. Sand particles are bigger than silt and are not suitable for the survival of larvae19. Chironomus riparius larvae make their tubes from different external particles and their own proteins20. Midge larvae are the inhabitants of sediments, and at the same time, sediment is the depository of different inorganic, organic, and heavy metals. In such cases, the tube of chironomid larvae may act as a defensive structure, which protects them from the adverse effects of undesirable pollutants and may increase their tolerance against such chemicals21,22,23.
Larvae can thrive in benthic sediments with high decaying organic content and very low DO concentrations in water bodies24. In poor DO concentration, larvae can survive due to the presence of haemoglobin in their body tissue fluid, which plays an important physiological role in increasing respiratory efficiency, as was observed in Chironomus plumosus. Longer tube length may help larvae generate better respiratory currents so that they can cope with a low DO environment.
Tube length is crucial for living in water because primarily tubes protect them from outer environmental factors like predators, and pollution. It was observed during this study that when the DO of water is low, larvae make elongated tubes to reach the upper layer of water, where the DO level is comparatively high. To get their required amount of oxygen, the larvae increase the tube length towards the water surface and increase the DO in tube water by undulating the body and other structures, creating a current inside the tube25,26. On contrary, when the DO level of the surrounding water of chironomid is sufficient, they can manage their normal physiological activities with the available oxygen. They need not to elongate their tube length. That’s why their tube length is inversely related to the DO of their surrounding medium.
If tube length does not increase in size in hypoxic water, larvae will not be able to meet their oxygen demand. If the DO of water decreases, tube length will increase and vice versa. Behavioural and physiological adaptations of chironomids larvae make them successful to live in a hypoxic environment. Thus, in hypoxic conditions, larvae with longer tubes are able to gather more oxygen from the upper layer of water and get more space to create a current of water to increase the amount of O2 inside the tube by undulating the preanal papillae, anal gill, ventral gills. This would explain why the tube length of chironomids depends on the DO of water. Hence by measuring the tube length with a standard measuring scale, one may get an idea about the quality of water, especially DO, before doing any chemical analysis. The work seems to be unique and novel for its own kind.
Conclusion
The role of physicochemical parameters of water on the tube length of chironomid larvae is considered in the present study. Among different parameters, dissolved oxygen has a major role in determining the tube length of larvae. Under both natural and laboratory conditions, a strong linear relationship is observed between the DO and the tube length. The value of R (correlation coefficient) is high both in natural as well as in laboratory conditions (0.650 and 0.971 respectively). The present study reveals a longer tube in aquatic media with the hypoxic condition and a shorter tube in those with normal DO in the surrounding medium. This result may help to assess the quality of water bodies and, in particular the status of DO. By measuring the tube length with a standard measuring scale, one may get an idea about the approximate level of DO in the aquatic bodies, without doing any chemical analysis or using the costly instrument (Supplementary Information).
Data availability
All data used in this manuscript are original and collected from field and laboratory studies.
References
Armitage, P., Cranston, P. S. & Pinder, L. C. V. The Chironomidae, The Biology and Ecology of Non-Biting Midges 8–34 (Chapman & Hall India, 1995).
Chaloner, D. T. & Wotton, R. S. Tube building by larvae of three species of midge (Diptera: Chironomidae). J. North Am. Benthol. Soc. 63, 65–69 (1996).
Vedamanikam, V. J. & Shazili, N. A. M. The chironomid larval tube, a mechanism to protect the organism from environmental disturbances?. Toxicol. Environ. Chem. 91(1), 171–176 (2009).
Case, S. T. & Wieslander, L. Secretory proteins of Chironomus tentans salivary glands: Structural motifs and assembly characteristics of a novel biopolymer. In Results and Problems in Cell Differentiation Vol. 19 (ed. Case, S. T.) 187–236 (Springer, 1992).
Brennan, A. & McLachlan, A. J. Tubes and tube-building in a lotic Chironomid (Diptera) community. Hydrobiologia 67, 173–178 (1979).
Walshe, B. M. The function of haemoglobin in Chironomus plumosus under natural conditions. J. Exp. Biol. 27, 73–95 (1950).
McLachlan, A. J. Some effects of tube shape on the feeding of Chironomus plumosus L. (Diptera: Chironomidae). J. Anim. Ecol. 46(1), 139–146 (1976).
Hershey, A. E. Tubes and foraging behaviour in larval Chironomidae: Implications for predator avoidance. Oecologia 73, 236–241 (1987).
Halpern, M., Gasith, A. & Broza, M. Does the tube of a benthic Chironomid larva play a role in protecting its dweller against chemical toxicants?. Hydrobiologia 470, 49–55 (2002).
Fukuhara, H. & Sakamoto, M. Enhancement of inorganic nitrogen and phosphate release from lake sediment by Tubificid worms and Chironomid larvae. Oikos 48(3), 312–320 (1987).
Svensson, J. & Leonardson, L. Effect of bioturbation by tube-dwelling chironomid larvae on oxygen uptake and denitrification in eutrophic lake sediments. Freshw. Biol. 35(2), 289–300 (2003).
Frouz, J., Matena, J. & Ali, A. Survival strategies of Chironomids (Diptera: Chironomidae) living in temporary habitats: A review. Eur. J. Entomol. 100, 459–465 (2003).
Nath, S., Podder, R. & Modak, B. K. Seasonal trend of body protein and growth of Chironomid larvae. Proc. Zool. Soc. 70(1), 88–91 (2017).
APHA (American Public Health Association American Water Works Association and Water Pollution Control Federation). Standard Methods for the Examination of Water and Wastewater 23rd edn. (APHA, 2017).
Butler, I. B., Schoonen, M. A. A. & Rickard, D. T. Removal of dissolved oxygen from water: A comparison of four common techniques. Talanta 41(2), 211–215 (1994).
Tucker, C. S. Carbon dioxide. In For Fish Farmers (eds Wellborn, T. L. & MacMillan, J. R.) 84–92 (Mississippi Co-operative Extension Service, 1984).
Pinder, L. C. V. The habitats of Chironomid larvae. In The Chironomidae: Biology and Ecology of Non-biting Midges (eds Armitage, P. D. et al.) 8–34 (Chapman & Hall India, 1995).
Broza, M., Halpern, M. & Inbar, M. Non-biting midges (Diptera: Chironomidae) in waste stabilization ponds: As an intensifying nuisance in Israel. Water Sci. Technol. 42, 71–74 (2000).
Burton, C. S. & Rodgers, J. H. Development of formulated reference sediments for freshwater and estuarine sediment testing. Environ. Toxicol. Chem. 13, 1163–1174 (1994).
Edgar, W. D. & Meadows, P. S. Case construction, movement, spatial distribution and substrate selection in the larva of Chironomus riparius Meigen. J. Exp. Biol. 50, 247–253 (1969).
Winner, R. W., Bossel, M. W. & Farrel, M. P. Insect community structure as an index of heavy-metal pollution in lotic ecosystems. Can. J. Fish. Aquat. Sci. 37, 647–655 (1980).
Krantzberg, G. & Stokes, P. M. Metal regulation, tolerance and body burdens in the larvae of the genus Chironomus. Can. J. Fish. Aquat. Sci. 16, 389–398 (1989).
Ingersoll, C. G. et al. Toxicity and bioaccumulation of sediment associated contaminants using freshwater invertebrates: A review of methods and applications. Environ. Toxicol. Chem. 14, 185–194 (1995).
Rasmussen, A. Least oxygen dependent. University of Florida book of insect records, Department of Entomology and Nematology University of Florida, Gainesville. 46–48 (University of Florida, 1996).
Konstantinov, A. S. Ecological factors affecting respiration in chironomid larvae. Limnologica 8, 127–134 (1971).
PanisInt, L., Goddeeris, B. & Verheyen, R. On the relationship between vertical microdistribution and adaptations to oxygen stress in littoral Chironomidae (Diptera). Hydrobiologia 318, 61–67 (1996).
Acknowledgements
The authors are thankful to the Department of Science and Technology and Biotechnology, Government of West Bengal, India [Grant No. 1242(Sanc.)/ST/P/S&T/1G-13/2014] for providing financial support as a Major Research Project. Thanks to the co-investigator of the project Dr Sudipto Das, Durgapur Govt. College. Thank you to the Principal, Government General Degree College Singur, and HOD, Department of Zoology of the Government General Degree College Singur, and the HOD of the Department of Zoology at Sidho-Kanho-Birsha University in Purulia for providing laboratory facilities. The authors are thankful to Dr Sovan Roy, Senior Scientific Officer at the Department of Science and Technology and Biotechnology of the Government of West Bengal, for his constant encouragement throughout this work.
Author information
Authors and Affiliations
Contributions
R.P.: Prepared the experimental set-up and performed the experiments; wrote the first draft; did the statistical analysis. S.N.: Guided and designed the experiments, arranged and performed experiments, and helped in writing the manuscript. B.K.M.: Provide advice and manage the literature searches. L.W.: Co-wrote the manuscript. B.M.: Helped in preparing the manuscript and statistical analysis.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Podder, R., Nath, S., Modak, B.K. et al. Tube length of chironomid larvae as an indicator for dissolved oxygen in water bodies. Sci Rep 12, 19971 (2022). https://doi.org/10.1038/s41598-022-23953-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-23953-9
- Springer Nature Limited