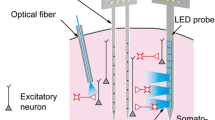
Abstract
Neural circuitry is typically modulated via invasive brain implants and tethered optical fibres in restrained animals. Here we show that wide-field illumination in the second near-infrared spectral window (NIR-II) enables implant-and-tether-free deep-brain stimulation in freely behaving mice with stereotactically injected macromolecular photothermal transducers activating neurons ectopically expressing the temperature-sensitive transient receptor potential cation channel subfamily V member 1 (TRPV1). The macromolecular transducers, ~40 nm in size and consisting of a semiconducting polymer core and an amphiphilic polymer shell, have a photothermal conversion efficiency of 71% at 1,064 nm, the wavelength at which light attenuation by brain tissue is minimized (within the 400–1,800 nm spectral window). TRPV1-expressing neurons in the hippocampus, motor cortex and ventral tegmental area of mice can be activated with minimal thermal damage on wide-field NIR-II illumination from a light source placed at distances higher than 50 cm above the animal’s head and at an incident power density of 10 mW mm–2. Deep-brain stimulation via wide-field NIR-II illumination may open up opportunities for social behavioural studies in small animals.






Similar content being viewed by others
Data availability
The main data supporting the results in this study are available within the paper and its Supplementary Information. The raw and analysed datasets generated during the study are too large to be publicly shared, yet they are available for research purposes from the corresponding authors on reasonable request. Source data are provided with this paper.
Code availability
The custom MATLAB code used in this study is available at https://github.com/XiangWu96/Wu_NBME_21_code.
References
Fenno, L., Yizhar, O. & Deisseroth, K. The development and application of optogenetics. Annu. Rev. Neurosci. 34, 389–412 (2011).
Tsai, H.-C. et al. Phasic firing in dopaminergic neurons is sufficient for behavioral conditioning. Science 324, 1080–1084 (2009).
Montgomery, K. L. et al. Wirelessly powered, fully internal optogenetics for brain, spinal and peripheral circuits in mice. Nat. Methods 12, 969–974 (2015).
Lozano, A. M. et al. Deep brain stimulation: current challenges and future directions. Nat. Rev. Neurol. 15, 148–160 (2019).
Carvalho-de-Souza, J. L. et al. Photosensitivity of neurons enabled by cell-targeted gold nanoparticles. Neuron 86, 207–217 (2015).
Jiang, Y. et al. Rational design of silicon structures for optically controlled multiscale biointerfaces. Nat. Biomed. Eng. 2, 508–521 (2018).
DiFrancesco, M. L. et al. Neuronal firing modulation by a membrane-targeted photoswitch. Nat. Nanotechnol. 15, 296–306 (2020).
Hong, G. & Lieber, C. M. Novel electrode technologies for neural recordings. Nat. Rev. Neurosci. 20, 330–345 (2019).
Hong, G., Antaris, A. L. & Dai, H. Near-infrared fluorophores for biomedical imaging. Nat. Biomed. Eng. 1, 0010 (2017).
Ledesma, H. A. et al. An atlas of nano-enabled neural interfaces. Nat. Nanotechnol. 14, 645–657 (2019).
Salatino, J. W., Ludwig, K. A., Kozai, T. D. Y. & Purcell, E. K. Glial responses to implanted electrodes in the brain. Nat. Biomed. Eng. 1, 862–877 (2017).
Fan, Z. et al. Using the tube test to measure social hierarchy in mice. Nat. Protoc. 14, 819–831 (2019).
Kim, T.-I. et al. Injectable, cellular-scale optoelectronics with applications for wireless optogenetics. Science 340, 211–216 (2013).
Lin, J. Y., Knutsen, P. M., Muller, A., Kleinfeld, D. & Tsien, R. Y. ReaChR: a red-shifted variant of channelrhodopsin enables deep transcranial optogenetic excitation. Nat. Neurosci. 16, 1499–1508 (2013).
Chuong, A. S. et al. Noninvasive optical inhibition with a red-shifted microbial rhodopsin. Nat. Neurosci. 17, 1123–1129 (2014).
Rajasethupathy, P. et al. Projections from neocortex mediate top-down control of memory retrieval. Nature 526, 653–659 (2015).
Marshel, J. H. et al. Cortical layer-specific critical dynamics triggering perception. Science 365, eaaw5202 (2019).
Chen, R. et al. Deep brain optogenetics without intracranial surgery. Nat. Biotechnol. 39, 161–164 (2021).
Gong, X. et al. An ultra-sensitive step-function opsin for minimally invasive optogenetic stimulation in mice and macaques. Neuron 107, 38–51 (2020).
Bedbrook, C. N. et al. Machine learning-guided channelrhodopsin engineering enables minimally invasive optogenetics. Nat. Methods 16, 1176–1184 (2019).
Chen, S. et al. Near-infrared deep brain stimulation via upconversion nanoparticle-mediated optogenetics. Science 359, 679–684 (2018).
Miyazaki, T. et al. Large timescale interrogation of neuronal function by fiberless optogenetics using lanthanide micro-particles. Cell Rep. 26, 1033–1043.e5 (2019).
Wu, X. et al. Sono-optogenetics facilitated by a circulation-delivered rechargeable light source for minimally invasive optogenetics. Proc. Natl Acad. Sci. USA 116, 26332–26342 (2019).
Munshi, R. et al. Magnetothermal genetic deep brain stimulation of motor behaviors in awake, freely moving mice. eLife 6, e27069 (2017).
Chen, R., Romero, G., Christiansen, M. G., Mohr, A. & Anikeeva, P. Wireless magnetothermal deep brain stimulation. Science 347, 1477–1480 (2015).
Alexander, G. M. et al. Remote control of neuronal activity in transgenic mice expressing evolved G protein-coupled receptors. Neuron 63, 27–39 (2009).
Magnus, C. J. et al. Ultrapotent chemogenetics for research and potential clinical applications. Science 364, eaav5282 (2019).
Smith, A. M., Mancini, M. C. & Nie, S. Second window for in vivo imaging. Nat. Nanotechnol. 4, 710–711 (2009).
Nelidova, D. et al. Restoring light sensitivity using tunable near-infrared sensors. Science 368, 1108–1113 (2020).
Grandl, J. et al. Temperature-induced opening of TRPV1 ion channel is stabilized by the pore domain. Nat. Neurosci. 13, 708–714 (2010).
Bashkatov, A. N., Genina, E. A., Kochubey, V. I. & Tuchin, V. V. Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000 nm. J. Phys. D 38, 2543–2555 (2005).
Miao, Q. et al. Molecular afterglow imaging with bright, biodegradable polymer nanoparticles. Nat. Biotechnol. 35, 1102–1110 (2017).
Gracheva, E. O. et al. Molecular basis of infrared detection by snakes. Nature 464, 1006–1011 (2010).
Huang, H., Delikanli, S., Zeng, H., Ferkey, D. M. & Pralle, A. Remote control of ion channels and neurons through magnetic-field heating of nanoparticles. Nat. Nanotechnol. 5, 602–606 (2010).
Yao, J., Liu, B. & Qin, F. Rapid temperature jump by infrared diode laser irradiation for patch-clamp studies. Biophys. J. 96, 3611–3619 (2009).
Davis, H. C. et al. Nanoscale heat transfer from magnetic nanoparticles and ferritin in an alternating magnetic field. Biophys. J. 118, 1502–1510 (2020).
Moiseenkova-Bell, V. Y., Stanciu, L. A., Serysheva, I. I., Tobe, B. J. & Wensel, T. G. Structure of TRPV1 channel revealed by electron cryomicroscopy. Proc. Natl Acad. Sci. USA 105, 7451–7455 (2008).
Berridge, M. J., Bootman, M. D. & Llewelyn Roderick, H. Calcium signalling: dynamics, homeostasis and remodelling. Nat. Rev. Mol. Cell Biol. 4, 517–529 (2003).
Jiang, Y. et al. Heterogeneous silicon mesostructures for lipid-supported bioelectric interfaces. Nat. Mater. 15, 1023–1030 (2016).
Shapiro, M. G., Homma, K., Villarreal, S., Richter, C.-P. & Bezanilla, F. Infrared light excites cells by changing their electrical capacitance. Nat. Commun. 3, 736 (2012).
Matthes, R. et al. Revision of guidelines on limits of exposure to laser radiation of wavelengths between 400 nm and 1.4 μm. Health Phys. 79, 431–440 (2000).
Chen, X., Chen, Y., Xin, H., Wan, T. & Ping, Y. Near-infrared optogenetic engineering of photothermal nanoCRISPR for programmable genome editing. Proc. Natl Acad. Sci. USA 117, 2395–2405 (2020).
Yang, X. et al. Bioinspired neuron-like electronics. Nat. Mater. 18, 510–517 (2019).
Owen, S. F., Liu, M. H. & Kreitzer, A. C. Thermal constraints on in vivo optogenetic manipulations. Nat. Neurosci. 22, 1061–1065 (2019).
Gradinaru, V. et al. Targeting and readout strategies for fast optical neural control in vitro and in vivo. J. Neurosci. 27, 14231–14238 (2007).
Lee, J., Darlington, T. R. & Lisberger, S. G. The neural basis for response latency in a sensory-motor behavior. Cereb. Cortex 30, 3055–3073 (2020).
Dhaka, A., Viswanath, V. & Patapoutian, A. Trp ion channels and temperature sensation. Annu. Rev. Neurosci. 29, 135–161 (2006).
Zeng, X. et al. Visualization of intra‐neuronal motor protein transport through upconversion microscopy. Angew. Chem. Int. Ed. 58, 9262–9268 (2019).
Zheng, W. et al. Lanthanide-doped upconversion nano-bioprobes: electronic structures, optical properties, and biodetection. Chem. Soc. Rev. 44, 1379–1415 (2015).
Zimprich, A. et al. A robust and reliable non-invasive test for stress responsivity in mice. Front. Behav. Neurosci. 8, 125 (2014).
Choi, C. The mazes with minds of their own. Nature 555, 127–128 (2018).
Ibsen, S., Tong, A., Schutt, C., Esener, S. & Chalasani, S. H. Sonogenetics is a non-invasive approach to activating neurons in Caenorhabditis elegans. Nat. Commun. 6, 8264 (2015).
Nakatsuji, H. et al. Thermosensitive ion channel activation in single neuronal cells by using surface-engineered plasmonic nanoparticles. Angew. Chem. Int. Ed. 54, 11725–11729 (2015).
Lyu, Y., Xie, C., Chechetka, S. A., Miyako, E. & Pu, K. Semiconducting polymer nanobioconjugates for targeted photothermal activation of neurons. J. Am. Chem. Soc. 138, 9049–9052 (2016).
Lodola, F., Martino, N., Tullii, G., Lanzani, G. & Antognazza, M. R. Conjugated polymers mediate effective activation of the mammalian ion channel transient receptor potential vanilloid 1. Sci. Rep. 7, 8477 (2017).
Li, N. et al. Spatiotemporal constraints on optogenetic inactivation in cortical circuits. eLife 8, e48622 (2019).
Haas, H. L. & Buzsaki, G. (eds) Synaptic Plasticity in the Hippocampus (Springer, 1988).
Wang, Y. et al. Tetherless near-infrared control of brain activity in behaving animals using fully implantable upconversion microdevices. Biomaterials 142, 136–148 (2017).
Chan, K. Y. et al. Engineered AAVs for efficient noninvasive gene delivery to the central and peripheral nervous systems. Nat. Neurosci. 20, 1172–1179 (2017).
Madisen, L. et al. A toolbox of Cre-dependent optogenetic transgenic mice for light-induced activation and silencing. Nat. Neurosci. 15, 793–802 (2012).
Chen, K.-T., Wei, K.-C. & Liu, H.-L. Theranostic strategy of focused ultrasound induced blood–brain barrier opening for CNS disease treatment. Front. Pharmacol. 10, 86 (2019).
Su, Y. et al. Novel NanoLuc substrates enable bright two-population bioluminescence imaging in animals. Nat. Methods 17, 852–860 (2020).
Lyu, Y. et al. Enhancing both biodegradability and efficacy of semiconducting polymer nanoparticles for photoacoustic imaging and photothermal therapy. ACS Nano 12, 1801–1810 (2018).
Zhu, X. et al. Temperature-feedback upconversion nanocomposite for accurate photothermal therapy at facile temperature. Nat. Commun. 7, 10437 (2016).
Jang, M., Ruan, H., Judkewitz, B. & Yang, C. Model for estimating the penetration depth limit of the time-reversed ultrasonically encoded optical focusing technique. Opt. Express 22, 5787–5807 (2014).
Martelli, F., Del Bianco, S. & Ismaelli, A. Light Propagation Through Biological Tissue and Other Diffusive Media: Theory, Solutions, and Software (Society of Photo-Optical Instrumentation Engineers, 2009).
Yaroslavsky, A. N. et al. Optical properties of selected native and coagulated human brain tissues in vitro in the visible and near infrared spectral range. Phys. Med. Biol. 47, 2059–2073 (2002).
Nachabé, R. et al. Validation of interventional fiber optic spectroscopy with MR spectroscopy, MAS-NMR spectroscopy, high-performance thin-layer chromatography, and histopathology for accurate hepatic fat quantification. Invest. Radiol. 47, 209–216 (2012).
Prahl, S. A. Tabulated Molar Extinction Coefficient for Hemoglobin in Water http://omlc.ogi.edu/spectra/hemoglobin/summary.html (1998).
Mason, M. G., Nicholls, P. & Cooper, C. E. Re-evaluation of the near infrared spectra of mitochondrial cytochrome c oxidase: implications for non invasive in vivo monitoring of tissues. Biochim. Biophys. Acta 1837, 1882–1891 (2014).
Bhattacharya, M. & Dutta, A. Computational modeling of the photon transport, tissue heating, and cytochrome C oxidase absorption during transcranial near-infrared stimulation. Brain Sci. 9, 179 (2019).
Welsher, K., Sherlock, S. P. & Dai, H. Deep-tissue anatomical imaging of mice using carbon nanotube fluorophores in the second near-infrared window. Proc. Natl Acad. Sci. USA 108, 8943–8948 (2011).
Welch, A. J. & van Gemert, M. J. C. (eds.). Optical-Thermal Response of Laser-Irradiated Tissue (Springer, 1995).
Tuchin, V. V. Light scattering study of tissues. Phys. Usp. 40, 495–515 (1997).
Sabino, C. P. et al. The optical properties of mouse skin in the visible and near infrared spectral regions. J. Photochem. Photobiol. B 160, 72–78 (2016).
Samatham, R., Phillips, K. G. & Jacques, S. L. Assessment of optical clearing agents using reflectance-mode confocal scanning laser microscopy. J. Innov. Opt. Health Sci. 3, 183–188 (2010).
Bashkatov, A. N. & Genina, E. A. Optical properties of human cranial bone in the spectral range from 800 to 2000 nm. Proc. SPIE 6163, 616310 (2006).
Ascenzi, A. & Fabry, C. Technique for dissection and measurement of refractive index of osteones. J. Biophys. Biochem. Cytol. 6, 139–142 (1959).
Ugryumova, N., Matcher, S. J. & Attenburrow, D. P. Measurement of bone mineral density via light scattering. Phys. Med. Biol. 49, 469–483 (2004).
Binding, J. et al. Brain refractive index measured in vivo with high-NA defocus-corrected full-field OCT and consequences for two-photon microscopy. Opt. Express 19, 4833–4847 (2011).
Nathanson, J. L., Yanagawa, Y., Obata, K. & Callaway, E. M. Preferential labeling of inhibitory and excitatory cortical neurons by endogenous tropism of adeno-associated virus and lentivirus vectors. Neuroscience 161, 441–450 (2009).
Hong, G. et al. Near-infrared-fluorescence-enhanced molecular imaging of live cells on gold substrates. Angew. Chem. Int. Ed. 50, 4644–4648 (2011).
Fu, T.-M. et al. Stable long-term chronic brain mapping at the single-neuron level. Nat. Methods 13, 875–882 (2016).
Lein, E. S. et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature 445, 168–176 (2007).
Gradinaru, V., Mogri, M., Thompson, K. R., Henderson, J. M. & Deisseroth, K. Optical deconstruction of parkinsonian neural circuitry. Science 324, 354–359 (2009).
Cohen, J. Statistical Power Analysis for the Behavioral Sciences (Routledge, 2013).
Acknowledgements
We thank W. T. Newsome, M. Z. Lin, X. Chen, L. Luo, H. Dai, D. Jiang and J. R. Sanes for helpful discussions; the Stanford Animal Histology Services for help with preparation of histologic specimens; G.H. acknowledges startup support from the Wu Tsai Neurosciences Institute of Stanford University, a National Institutes of Health (NIH) Pathway to Independence Award (National Institute on Aging 5R00AG056636-04), a National Science Foundation (NSF) CAREER Award (2045120), the Rita Allen Foundation Scholars Program, a gift from the Spinal Muscular Atrophy (SMA) Foundation, and seed grants from the Wu Tsai Neurosciences Institute and the Bio-X Initiative of Stanford University. X.W. acknowledges support from the Stanford Graduate Fellowship. K.S.O. acknowledges the NeuroTech training programme supported by the National Science Foundation under Grant No. 1828993. This work was performed in part at the Stanford Nano Shared Facilities (SNSF) and Cell Sciences Imaging Facility (CSIF) of Stanford University. K.P. thanks Nanyang Technological University (startup grant: M4081627) and Singapore Ministry of Education Academic Research Fund Tier 2 (MOE2016-T2-1-098) for financial support. Some schematics were created with BioRender.com.
Author information
Authors and Affiliations
Contributions
X.W., Y.J., K.P. and G.H. conceived and designed the project; X.W., Y.J., N.J.R., F.Y., Q.Z., R.Y., J.L., S.C., W.R., A.S. and K.S.O. performed the experiments; X.W., Y.J., N.J.R., F.Y., Q.Z., R.Y., J.L., S.C., W.R., K.P. and G.H. analysed the data and wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Biomedical Engineering thanks Thomas McHugh and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Main Supplementary Information
Supplementary notes, figures, tables, video captions and references.
Supplementary Video 1
Distant 1,064 nm illumination induces mouse circling through the scalp in a freely behaving mouse.
Supplementary Video 2
Mouse trajectory in a Y maze during the pre-test.
Supplementary Video 3
Mouse trajectory in a Y maze during the post-test.
Source data
SD for Fig. 1
Source data.
SD for Fig. 2
Source data.
SD for Fig. 3
Source data.
SD for Fig. 4
Source data.
SD for Fig. 5
Source data.
SD for Fig. 6
Source data.
Rights and permissions
About this article
Cite this article
Wu, X., Jiang, Y., Rommelfanger, N.J. et al. Tether-free photothermal deep-brain stimulation in freely behaving mice via wide-field illumination in the near-infrared-II window. Nat. Biomed. Eng 6, 754–770 (2022). https://doi.org/10.1038/s41551-022-00862-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-022-00862-w
- Springer Nature Limited
This article is cited by
-
White-light activatable organic NIR-II luminescence nanomaterials for imaging-guided surgery
Nature Communications (2024)
-
Nanotechnology for vision restoration
Nature Reviews Bioengineering (2024)
-
Biomimetic NIR-II fluorescent proteins created from chemogenic protein-seeking dyes for multicolor deep-tissue bioimaging
Nature Communications (2024)
-
Near-infrared II fluorescence imaging
Nature Reviews Methods Primers (2024)
-
In vivo ultrasound-induced luminescence molecular imaging
Nature Photonics (2024)