Abstract
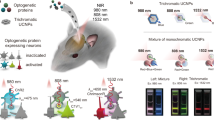
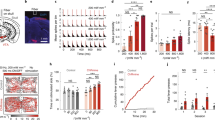
Optogenetics has revolutionized the experimental interrogation of neural circuits in the past decade and holds potential for the treatment of neurological disorders. However, optogenetic stimulation of deep brain neurons requires the insertion of invasive optical fibers because the activating blue-green light cannot penetrate deep inside brain tissue. Here we describe a minimally invasive technique for the stimulation of deep brain neurons by transcranial near-infrared light (NIR), where upconversion nanoparticles (UCNPs) are used as optogenetic actuators to locally convert NIR into visible light. We detail the protocol to use locally injected UCNPs to stimulate dopamine neurons in the ventral tegmental area (VTA) of anesthetized mice by transcranial NIR.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Fenno L, Yizhar O, Deisseroth K (2011) The development and application of optogenetics. Annu Rev Neurosci 34:389–412
Lin JY, Knutsen PM, Muller A, Kleinfeld D, Tsien RY (2013) ReaChR: a red-shifted variant of channelrhodopsin enables deep transcranial optogenetic excitation. Nat Neurosci 16(10):1499–1508
Hamblin MR (2016) Shining light on the head: photobiomodulation for brain disorders. BBA Clin 6:113–124
Chuong AS et al (2014) Noninvasive optical inhibition with a red-shifted microbial rhodopsin. Nat Neurosci 17(8):1123–1129
Klapoetke NC et al (2014) Independent optical excitation of distinct neural populations. Nat Methods 11(3):338–346
Rajasethupathy P et al (2015) Projections from neocortex mediate top-down control of memory retrieval. Nature 526(7575):653–659
Yizhar O, Fenno LE, Davidson TJ, Mogri M, Deisseroth K (2011) Optogenetics in neural systems. Neuron 71(1):9–34
Zhang F et al (2008) Red-shifted optogenetic excitation: a tool for fast neural control derived from Volvox carteri. Nat Neurosci 11(6):631–633
Chen S et al (2018) Near-infrared deep brain stimulation via upconversion nanoparticle-mediated optogenetics. Science 359(6376):679–684
Chen G, Qiu H, Prasad PN, Chen X (2014) Upconversion nanoparticles: design, nanochemistry, and applications in theranostics. Chem Rev 114(10):5161–5214
Zhou B, Shi B, Jin D, Liu X (2015) Controlling upconversion nanocrystals for emerging applications. Nat Nanotechnol 10(11):924–936
Wanat MJ, Willuhn I, Clark JJ, Phillips PE (2009) Phasic dopamine release in appetitive behaviors and drug addiction. Curr Drug Abuse Rev 2(2):195–213
Boyden ES, Zhang F, Bamberg E, Nagel G, Deisseroth K (2005) Millisecond-timescale, genetically targeted optical control of neural activity. Nat Neurosci 8(9):1263–1268
Gunaydin LA et al (2014) Natural neural projection dynamics underlying social behavior. Cell 157(7):1535–1551
Acknowledgments
This work was supported by Human Frontier Science Program Postdoctoral Fellowship (LT 000579/201) (S.C.), JSPS (Japan Society for the Promotion of Science) Postdoctoral Fellowship (16F16386) (S.C.), RIKEN Special Postdoctoral Researchers Program (S.C.), Grant-in-Aid for Young Scientists from MEXT (the Ministry of Education, Culture, Sports, Science and Technology of Japan) (16K18373, 18K14857) (S.C.), RIKEN Incentive Research Project Grant for Individual Germinating Research (S.C.), Narishige Neuroscience Research Foundation Grant (S.C.), and Nakatani Foundation Grant Program (S.C.).
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media, LLC, part of Springer Nature
About this protocol
Cite this protocol
Chen, S. (2020). Near-infrared Deep Brain Stimulation in Living Mice . In: Niopek, D. (eds) Photoswitching Proteins . Methods in Molecular Biology, vol 2173. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0755-8_4
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0755-8_4
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0754-1
Online ISBN: 978-1-0716-0755-8
eBook Packages: Springer Protocols