Abstract
For long-lasting artificial hip joint implants, it is necessary to reduce the wear of the acetabular liner composed of ultra-high-molecular-weight polyethylene (UHMWPE) and to eliminate periprosthetic osteolysis. An articular cartilage-mimicking technology has been developed for nanoscale surface modification by grafting poly(2-methacryloyloxyethyl phosphorylcholine) (MPC) onto a highly cross-linked UHMWPE (X-UHMWPE) using photoinduced polymerization. The thickness of the poly(MPC) graft layer is 100–200 nm. This treatment increases the surface hydrophilicity. Other hydrophilic polymers grafted onto the X-UHMWPE are not suitable for long-term functioning under biological conditions. Studies of the tribological and biological effects with poly(MPC) grafted onto the X-UHMWPE substrate revealed that this grafting decreases the production of wear particles and bone resorption responses. The poly(MPC)-grafted X-UHMWPE has been introduced onto an artificial hip joint as a liner for lubrication. This artificial hip joint has been used clinically since 2011 and has been implanted in more than 20 000 patients. This technology has also been applied to the surface modification of PMPC on poly(ether ether ketone) (PEEK), using self-initiated photoinduced grafting, for the development of a new type of artificial joint. This articular cartilage-mimicking technology, which is applied to obtain highly lubricating surfaces, is therefore suitable for preparing artificial hip joint substrates.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
Progress in the development of medical devices has been strongly correlated to changes in human society and has helped to provide a good quality of life. Many medical devices, including single-use devices and long-term implantable artificial organs, have been developed and are being used for treating serious medical conditions. Polymer science and technology have played an extremely important role in the development of these medical devices. Various conventional polymeric materials have been employed to fabricate such devices and developed to resolve specific biological problems when they are used in medical treatments.1, 2, 3, 4 For example, polysulfone and cellulose acetate are currently used as hollow fiber materials for a hemodialyzer. Although blood dialysis therapy using the hemodialyzer is successful for extending the life of patients with kidney disease, thrombus formation and immunoreactions because of activation of the complementary system are currently major issues associated with this treatment. Furthermore, expanded polytetrafluoroethylene has been applied for the fabrication of artificial valves and artificial vascular prostheses. However, implantation of these devices requires permanent administration of an antiplatelet reagent to prevent clot formation. Thus, new polymeric materials with good blood compatibility and antithrombus function are strongly required.
Total hip arthroplasty (THA) with an artificial hip joint is one of the most effective treatments for severe arthritis patients; the number of these treatments has been increasing due to the increase in the population of the elderly.5, 6, 7 Improvements have been made in implant design and surgical techniques. However, aseptic loosening following periprosthetic osteolysis remains a serious problem, which limits patient survival and clinical success. Up to 20% of patients implanted with artificial hip joints with conventional PE develop aseptic loosening within 10 years, and approximately half of these patients become disabled due to pain and loss of function.8, 9 The only therapeutic intervention is revision surgery. The number of these incidents has been increasing and is projected to double by the year 2026. This will lead to an increasing social and economic impact10, 11 unless some limiting mechanism can be successfully implemented to prevent periprosthetic osteolysis.
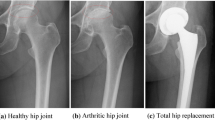
The most widely used bearing couple for an artificial joint system for THA involves the combination of an ultra-high-molecular-weight polyethylene (UHMWPE) acetabular liner and a cobalt–chromium (Co–Cr) alloy femoral head (Figure 1). It is known that UHMWPE has good mechanical properties with low friction. However, when creating a lubrication interface between UHMWPE and the Co–Cr alloy, wear of the polymer occurs and small particles are produced, even under biological conditions. Periprosthetic osteolysis is triggered by the host inflammatory response to the UHMWPE wear particles generated from the interface,12 which induces phagocytosis by macrophages and the subsequent secretion of bone resorption cytokines.13 Hence, various studies have been conducted with the aim of reducing the formation of UHMWPE wear particles. Among these, cross-linking of UHMWPE through gamma-ray irradiation leads to a 42–93% reduction in the wear rate compared with conventional PE in vivo.14, 15, 16 Clinical results involving wear resistance of the polymer (X-UHMWPE) are now in medium-term follow-up. The reported percentage of reductions in the rate of femoral head penetration have ranged from 23 to 95%, depending on which conventional PE was used as the control.17 Another strategy for reducing the formation of wear particles is to change the femoral head material from metal to ceramics to reduce friction against the PE liner.18, 19, 20
From the viewpoint of the interfacial chemistry of polymeric materials, the surface lubrication of PE is expected to be improved by combination with other polymers at the interface. Considering that the healthy human cartilage joint surface is covered with a nanoscale phospholipid layer,21 grafting a phospholipid-like layer onto the liner surface may lead to an ideal lubricity, resembling that of the physiological joint surface. This may be an ideal surface for the design of a joint interface that is suitable for long-term use under biological conditions. In this review, the basic concepts and scientific background for highly lubricating interfaces prepared on PE will be introduced, and their performance in an artificial hip joint will be discussed. In addition, the processes for the development of an artificial hip joint installed with this novel lubrication interface will be explained.
Articular cartilage lubrication
Learning the functions of nature and mimicking them in artificial systems are important in the development of novel technologies. Every human joint is covered with cartilage to provide essential lubrication. Generally, the articular cartilage consists of a collagen network, hyaluronic acid and proteoglycan subunits.22 The proteoglycan subunits form a hydrogel-like surface layer, along with the joint synovia. Although the binding that exists between the proteoglycan subunits and the hyaluronic acid has been detected,23 the binding between the hyaluronic acid and the collagen network is not yet well understood. The friction coefficients of joints increased when the hydrogel-like layer on the cartilage surface was removed.24 When this hydrogel layer was removed, the joint surface was only lubricated by the joint synovia or hyaluronic acid; for example, following the loss of the hydrogel composed of the proteoglycan subunits, the friction coefficient of the joints increases and cannot be recovered. This indicates not only that the proteoglycan aggregates bond with the collagen network through physical adsorption but also that the hydrophilic macromolecules on the joint surface play an important role in reducing friction. A previous study has reported that the natural water-soluble macromolecules present on the cartilage surface are likely to have a hydrophilic brush-like structure; a part of this proteoglycan aggregate brush bonds with the collagen network on the cartilage surface (Figure 2).25 The remainder of the proteoglycan aggregates float freely in the joint synovia.
Although the lubrication mechanism of human joints has been extensively studied for a long time, this mechanism is still not clearly understood. The lubrication of joints is considered to mainly involve hydration lubrication.26, 27 That is, extremely hydrophilic macromolecules reduce friction by promoting the formation of a fluid film that is retained by attractive forces of the water molecules, indicating that these attractive interactions are critical for achieving the low friction necessary for smooth motion of the joints.28 The lubrication of human joints appears to occur by hydration lubrication because the structure of the surface layer of the joints is similar to that of the gelled material. A new mechanism known as ‘surface gel hydration lubrication’ has been proposed to describe joint lubrication in cases where such hydrophilic macromolecules are involved.25
Water is attracted by the hydrophilic macromolecules in the surface layer and has an important role in lubrication. Because most macromolecules are flexible, they cannot support a load by themselves. It is thus likely that the water in the surface layer supports most of the load. Frictional forces arise as a result of the adhesion of macromolecules to the counter surface. The time-dependent properties of these friction forces can be interpreted as follows (Figure 3).29 Under a load, water exudes slowly from the surface layer, with or without sliding. As a result of water loss, the thickness of the surface layer is reduced, and the water content in the surface layer decreases. Consequently, the degree of adhesion of this surface to the opposite bearing surface increases along with the frictional force. Therefore, it may be concluded that the amount of friction essentially depends on the water content in the surface layer and that ‘hydration lubrication’ leads to low friction and wear.
The lubrication of an artificial hip joint mainly occurs through a mechanism that differs from that of natural human joints and that is also considerably less effective. The bearing couple is structurally rigid and composed of nonporous materials, which do not allow for the biophysical mechanisms responsible for lubrication in natural joints. When artificially restoring joint function, it is important for the same level of lubrication as in natural joints to be achieved. The primary advantage of this bearing couple is that it allows for the design of anatomically accurate hip joints. Therefore, substituting or modifying the bearing surfaces of artificial joints with a hydrogel structure should increase lubrication to levels that match those of articular cartilage under physiological conditions. Because a bearing surface with a hydrogel structure composed of hydrophilic macromolecules should be very similar to that of articular cartilage, these materials will allow for hydration lubrication and are well suited for this application.
Biomimetic surface modification with grafting of a hydrophilic polymer
Polymer brush surface prepared by surface-initiated living radical polymerization
Surface modification is one of the important technologies for preparing new multifunctional materials. In artificial hip joints, the modification of bearing surfaces with a hydrophilic polymer possessing a brush-like structure is assumed to be similar to that of articular cartilage. The hydration lubrication interface of such joints can also be considered to mimic natural joint cartilage in vivo. The grafting of polymers onto various surfaces has long been studied and has an important role in many areas of biomaterial science and technology. Recent efforts have focused on the synthesis of ‘polymer brushes’, in which the polymer chains stretch out from the surface of the substrate.30, 31 There are two primary methods for modifying a planar substrate with an organic polymer: physical coating or chemical grafting (to and from the surface). When physical coating techniques, such as spin and dip coating, are applied, the polymer is simply adsorbed onto the substrate surface and may diffuse away when the substrate is immersed into a good solvent for the dissociation of the polymer. By contrast, the ‘grafting from’ approach can be used to synthesize high-density polymer brushes,32 which can dramatically change conformation in solvents by varying the graft density (Figure 4). At low graft densities, these brushes will be in a ‘mushroom-like’ conformation, with the coil dimensions being similar to that of their free chains. With increasing graft density, the graft chains are forced to stretch away from the substrate, forming a ‘polymer brush.’ Such high-density brushes are considerably thicker and range in size from a few nanometers to several micrometers. This significant increase in thickness is attributed to the fact that the ‘grafting from’ layers have substantially higher grafting densities than those of the ‘grafting to’ layers.
Takahara et al.33, 34, 35, 36 and Klein et al.37, 38, 39 prepared polymer brush surfaces of polyelectrolytes and evaluated the lubricity at the interfaces. The surface-initiated living polymerization provides well-defined polymer brush surfaces, where the chain density and chain length of graft polymers could be regulated. They observed that extremely hydrophilic polyelectrolyte brush surfaces in water showed high lubrication characteristics. However, they found that there are some effects from inorganic salts present in the medium. These phenomena were discussed by considering the effects of the salts on the polymer conformation.
Photoinduced graft polymerization of hydrophilic monomers onto PE in aqueous medium
For the industrial manufacturing of highly lubricated surfaces, a conventional radical polymerization is necessary. The grafting of a hydrophilic polymer onto PE is performed using a photoinitiated ‘grafting-from’ polymerization with the corresponding monomer, such as the zwitterionic 2-methacryloyloxyethyl phosphorylcholine (MPC).40, 41 The methacrylate monomer MPC, with an electrically neutral phospholipid polar group (phosphorylcholine group) in the side chain, is the basic unit for creating novel polymer biomaterials, following the design and development by Ishihara et al.42, 43, 44 MPC is currently produced on an industrial scale worldwide and is commercially available. The photoinitiated graft polymerization reaction, which uses a typical water-insoluble photoinitiator, benzophenone (BP), is shown in Figure 5. The photochemical radical formation with this reaction is a well-known and widely used photoinduced reaction with polymers. First, a physically coated layer of BP on the PE surface is excited by ultraviolet irradiation. This excites the BP to its triplet state, which abstracts a hydrogen atom from the -CH2- group. The generated radical is capable of initiating the graft polymerization of the monomer. This technique has several important benefits. These benefits include the direct grafting of the polymer chains on the PE, resulting in the formation of C–C covalent bonds between the polymer chains and the PE substrate, high mobility of the polymer chains, high density and the ability to control the length of the graft introduced on the polymer.
A comparison among the surface modification methods of X-UHMWPE has been made to understand the effect of polymer chains at the interface on friction stability. Here, two types of MPC polymer are used. The first is poly(MPC-co-n-butyl methacrylate) (MPC unit mole fraction is 0.30). The poly(MPC-co-n-butyl methacrylate is used for surface coatings on implantable artificial hearts.44 The second MPC polymer is poly(MPC-co-3-methacryloyloxypropyl trimethoxysilane) for chemical reacting on the surface. First, a plasma treatment was applied to the surface of the X-UHMWPE substrate, and then the poly(MPC-co-3-methacryloyloxypropyl trimethoxysilane) was reacted with the hydroxyl groups generated on the surface. The dynamic friction coefficients of the physically hydrophilic polymer-coated X-UHMWPE increased to the level of the dynamic friction coefficient of untreated X-UHMWPE (Figure 6).45 These results indicate that the hydrophilic polymer-coated layers are detached from the X-UHMWPE surface. Therefore, the physical attachment by molecular interactions is ineffective in the case of large and multidirectional loads. By contrast, the hydrophilic polymer-grafted X-UHMWPE exhibited an extremely low and stable dynamic friction coefficient and volumetric wear compared with those of untreated X-UHMWPE and hydrophilic polymer-coated X-UHMWPE. The surface hydrophilic graft polymer layer is bound with the substrate through chemical covalent bonding. Therefore, the benefits of the surface modification layer used in artificial joints are expected to be retained long term.
(a) Illustration of various modification modes on the substrate with hydrophilic polymers and (b) coefficient of dynamic friction of the untreated cross-linked ultra-high-molecular-weight polyethylene (X-UHMWPE), physical coating with poly(2-methacryloyloxyethyl phosphorylcholine) (MPC)-co-n-butyl methacrylate) (PMB), chemical reaction with poly(MPC-co-3-methacryloyloxypropyl trimethoxysilane) (PMSi) and chemical grafting of poly(MPC) on X-UHMWPE as a function of loads in the ball-on-plate friction test.45 Data are expressed as the mean±standard deviation.
Kyomoto and Ishihara et al. prepared hydrophilic polymer layers bearing various types of charges, namely, non-ionic, cationic, anionic or zwitterionic, on X-UHMWPE surfaces using the photoinduced radical polymerization procedure explained above.46 These monomers are oligo(ethylene glycol) monomethacrylate (OEGMA), 2-(N,N-dimethylaminoethyl) methacrylate (DMAEMA), 2-(methacryloylethyl) phosphoric acid (MPA), and MPC, respectively. The employed approach only renders the surface of the X-UHMWPE substrate susceptible to modification and does not affect its bulk properties.47, 48 The hydrophilicity of the polymer-grafted X-UHMWPE surfaces increased gradually with the photoirradiation time (Figure 7).49 For example, the poly(MPC)-graft X-UHMWPE shows a low contact angle of ~30°. The hydrophilic polymer-grafted X-UHMWPE surface is imparted with wettability from the presence of a hydrophilic polymer layer. The hydrated layer clearly affects the friction response; the dynamic friction coefficients of the hydrophilic polymer-grafted X-UHMWPE surface are considerably lower than those of the untreated X-UHMWPE surface. This result is attributed to the hydrophilicity of the surface, as is also evident from the reduction in the static water contact angles of the hydrophilic polymer-grafted surfaces.46
Static contact angles by water of various hydrophilic polymer-grafted X-UHMWPE surfaces as a function of photoirradiation time.46 The substrates are (a) poly(oligo(ethylene glycol) monomethacrylate(OEGMA)), (b) poly(2-(N,N-dimethylaminoethyl) methacrylate (DMAEMA)), (c) poly(2-(methacryloylethyl) phosphoric acid (MPA)) and (d) poly(2-methacryloyloxyethyl phosphorylcholine) (MPC)). Data are expressed as the mean±standard deviation.
The improvement in the dynamic friction coefficients of the hydrophilic polymer-grafted surface with an increase in loading has revealed some very interesting observations. The hydrophilic polymer-grafted layer cannot follow Amonton’s first law, F=mN, where F is the frictional force, m is the friction coefficient and N is the normal force, which can be stated as the frictional force is proportional to the normal force of two dry solid surfaces sliding against one another; that is, a variable friction coefficient of hydrophilic polymer-grafted surface indicates that the bearing surface or interface does not provide solid lubrication.50 The viscoelastic X-UHMWPE substrate is slightly deformed by the loads. Consequently, a low friction coefficient may be necessary to accumulate a larger volume of water in the thin film over the larger contact area of the concave surface.45
Characteristics of poly(MPC)-grafted PE
Moro and Ishihara et al. have fabricated an artificial hip joint based on the concept of ‘hydration lubrication’ by grafting poly(MPC) onto the surface of X-UHMWPE (poly(MPC)-grafted X-UHMWPE). This material was designed to reduce wear and suppress bone resorption.51, 52, 53 As mentioned above, the hydrogel-like cartilage surfaces can be assumed to have a brush-like structure; a part of the proteoglycan aggregate brush is bonded with the collagen network on the cartilage surface.25 The bearing surface with grafted poly(MPC) in artificial hip joints is assumed to have a brush-like structure, similar to that of articular cartilage (Figure 8). Molecular simulations of the poly(MPC) chain revealed that the polymer chain assumes a stretched conformation in water.44 This is because the hydrophilic phosphorylcholine units face a water-contacting surface with the hydrophobic backbone embedded with the phosphorylcholine group. In addition, the volume of the phosphorylcholine group is extremely large compared with the methacrylate unit. Both of these effects operate cooperatively, and the conformation of the poly(MPC) is rod-shaped and rigid. Therefore, the grafted chains of poly(MPC) on the X-UHMWPE may stand up from the substrate with associated water molecules when they are in aqueous medium. The hydration lubrication interface can therefore be considered to mimic natural joint cartilage in vivo.
Schematic illustration of a poly(2-methacryloyloxyethyl phosphorylcholine(MPC))-grafted surface and molecular simulation of the poly(MPC) chain.44
An important characteristic of MPC polymers is their excellent biocompatibility. They provide unique surface properties of high lubricity and low friction, anti-protein adsorption and cell adhesion resistance.43, 44, 54, 55, 56, 57, 58, 59, 60 MPC polymers are one of the most readily available biocompatible and hydrophilic polymers studied thus far, and they have potential applications in a wide variety of fields, including biology, biomedical science and surface chemistry. These polymers can form a thin film of free water under physiological conditions.44, 55, 61, 62 The use of MPC polymers has already led to the development of several medical devices. These devices have been employed in clinical practice with the approval of the Ministry of Health, Labour and Welfare of Japan and the Food and Drug Administration (FDA) of the United States; therefore, the efficacy and safety of MPC polymers as a biomaterial are well established.63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80
Surface properties of hydrophilic polymers grafted on X-UHMWPE: comparison of the chemical structure of graft polymer chains
The graft polymerization process resulted in the formation of uniform polyelectrolyte layers on the X-UHMWPE surface, as observed from the cross-sectional transmission electron microscopy images in Figure 9a; the thickness of these layers was 100–150 nm. As shown in Figure 9b, the dynamic friction coefficients for the polyelectrolyte-grafted X-UHMWPE samples ranged from 0.01 to 0.05, representing a 40–85% reduction compared with those of untreated X-UHMWPE.
(a) Cross-sectional transmission electron microscopy images and (b) dynamic friction coefficients in the ball-on-plate friction test under various lubrication conditions for untreated cross-linked ultra-high-molecular-weight polyethylene (X-UHMWPE) and various hydrophilic polymer-grafted X-UHMWPE samples.46 Data are expressed as the mean±standard deviation. DMAEMA, 2-(N,N-dimethylaminoethyl) methacrylate; MPA, 2-(methacryloylethyl) phosphoric acid; MPC, 2-methacryloyloxyethyl phosphorylcholine; OEGMA, oligo(ethylene glycol) monomethacrylate.
The effects of the chemical structure of graft polymer chains on lubricity have been reported.46 The dynamic friction coefficients of the poly(OEGMA)- and poly(MPC)-grafted X-UHMWPE surfaces, with respect to different lubricants, do not differ significantly. For all lubricants, the dynamic friction coefficient of the poly(MPC)-grafted X-UHMWPE is significantly less than that of poly(OEGMA)-grafted X-UHMWPE; a similar relation is observed with the values of the static water contact angles of the two types of polyelectrolyte-grafted X-UHMWPE surfaces. The dynamic friction coefficients of the poly(DMAEMA)- and poly(MPA)-grafted X-UHMWPE dramatically increased in a simulated body fluid and bovine serum, respectively. In addition, untreated X-UHMWPE also exhibited increased friction in bovine serum lubricant compared with that in water. The surface zeta potentials of the untreated X-UHMWPE and the poly(OEGMA)- and poly(MPC)-grafted X-UHMWPE are close to zero. By contrast, the surface zeta potential of the poly(DMAEMA)-grafted X-UHMWPE with cationic grafted polymer layers is strongly positive, whereas that of the poly(MPA)-grafted X-UHMWPE sample with anionic grafted polymer layers is strongly negative. The amounts of bovine serum albumin adsorbed on the surface of the poly(OEGMA)-, poly(MPA)- and poly(MPC)-grafted X-UHMWPE with nonionic, anionic and zwitterionic polymer layers were significantly smaller. However, in the case of the poly(DMAEMA)-grafted X-UHMWPE, the amount of adsorbed bovine serum albumin was higher. These findings indicate that the type of charge (nonionic, cationic, anionic or zwitterionic) of the graft polymer layer affects the hydration and friction kinetics of the X-UHMWPE-bearing surface. The dynamic friction coefficient measured during a ball-on-plate friction test using water as the lubricant depends on the hydrophilicity of the surface (that is, on the static-water contact angles), as shown in Figures 7 and 9b. The surfaces of all the polyelectrolyte-grafted X-UHMWPE exhibited considerably higher lubricity than did the surface of the untreated X-UHMWPE when water was used as a lubricant. This result is because the water molecules in the hydration layers of the hydrophilic polymer or polyelectrolytes act as highly efficient lubricants.46, 81, 82 However, when other lubricants were used, the surface of the polyelectrolyte-grafted X-UHMWPE samples with cationic and anionic polymer layers exhibited characteristics that were significantly different from those of the hydrophilic polymer- and zwitterionic polymer-grafted X-UHMWPE, even though all the surfaces were highly hydrophilic. The poly(DMAEMA)-grafted X-UHMWPE exhibits a higher dynamic friction coefficient in bovine serum, which contains proteins such as albumin and γ-globulin, than in water or simulated body fluid during the ball-on-plate friction test.33 Poly(DMAEMA) has positively charged –NH+(CH3)2 groups at neutral pH; this group, in turn, attaches to negatively charged molecules. For example, albumin molecules have negative charges at physiological pH values (pH 7.4). This implies that the presence of protein molecules at the bearing interface increases the resistance to sliding motion. Because proteins are adsorbed on the Co–Cr–Mo alloy counterface,83, 84 the high resistance to sliding motion can be attributed to either a stronger adhesive interaction or to the interpenetration of the adsorbed protein films formed on both the poly(DMAEMA)-grafted X-UHMWPE and the Co–Cr–Mo alloy surfaces. However, the poly(DMAEMA)-grafted X-UHMWPE cups exhibit high wear resistance at high loads during the hip simulator test despite the fact that they show a high dynamic friction coefficient in bovine serum during the ball-on-plate friction test. This may be because the protein film adsorbed from the bovine serum is ‘squeezed out’ of the bearing interface at high loads.85 By contrast, the surface of the poly(MPA)-grafted X-UHMWPE exhibits considerably lower lubricity in simulated body fluid during the ball-on-plate friction test. The chemical structure of the negatively charged PMPA is characterized by the presence of a large number of trap sites for positively charged inorganic ions. Hence, the poor lubricity of the surface of the poly(MPA)-grafted X-UHMWPE can be attributed to the shrinkage or bridging of the negatively charged polyelectrolyte chains; this reduces the mobility of the chains in solutions containing positively charged inorganic ions.33, 46, 81
A long-term hip simulator test has been conducted to understand the stability and durability of the hydrophilic polymer-grafted X-UHMWPE liners. This hip simulation test was conducted under ISO 14242-3 conditions to compare X-UHMWPE with polymer-grafted X-UHMWPE acetabular liners against a 26-mm Co-Cr alloy femoral head. Bovine calf serum (25 vol%), diluted in distilled water, was used as a lubricant. The amount of wear on the specimen after 106 cycles as evaluated by this test corresponds to that of the human life-time of 10 years under normal conditions.
The poly(OEGMA)- and poly(MPC)-grafted X-UHMWPE cups exhibit high wear resistance during the hip simulator tests (Figure 10), as well as low dynamic friction coefficients, during the ball-on-plate friction tests (Figure 9b). In particular, the highly hydrated surface layer of the poly(MPC)-grafted X-UHMWPE provides extremely efficient lubrication under all conditions. Moreover, the surface zeta-potential of the surface of poly(MPC)-grafted X-UHMWPE is close to zero because the ionic group in the MPC unit forms an inner salt, and the electrostatic effects are diminished. Therefore, the zwitterionic-grafted polymer layers only attract water molecules and repel proteins and positively charged inorganic ions. This characteristic is similar to that of the nonionic polymer-grafted surfaces. It has been reported that the water molecules adsorbed on the surfaces of highly hydrophilic and zwitterionic polymer (that is, poly(MPC)) brushes act as lubricants to reduce the friction between the brushes and the counter surface.39 Recent efforts to identify hydrophilic polymers, including polyelectrolytes, have focused on tribological studies of surface-attached molecules, seeking to emulate those found at the cartilage surface.41, 82, 86 However, the wear properties of the poly(DMAEMA)- and poly(MPA)-grafted X-UHMWPE during the hip joint simulator tests demonstrate that the mechanism of action of the polyelectrolyte-grafted layer cannot be explained merely in terms of fluid-film lubrication. Therefore, it is assumed that the primary factor responsible for the low friction and the high wear resistance is the high degree of hydration of the polymer layer, as is the case with the zwitterionic poly(MPC)-grafted layer; water molecules in the hydration layers act as highly efficient lubricants.41, 82, 87 From these fundamental studies, it can be confirmed that poly(MPC)-grafting is a promising approach for modifying the surface of X-UHMWPE to obtain stable and highly lubricating systems. This justifies the placing of the poly(MPC) grafting layer on the acetabular liner of artificial hip joints.
(a) Picture of hip simulator testing device and (b) time course of gravimetric wear of various hydrophilic polymer-grafted cross-linked ultra-high-molecular-weight polyethylene (X-UHMWPE) cups during hip simulator test.46 Data are expressed as the means±standard deviations. DMAEMA, 2-(N,N-dimethylaminoethyl) methacrylate; MPA, 2-(methacryloylethyl) phosphoric acid; MPC, 2-methacryloyloxyethyl phosphorylcholine; OEGMA, oligo(ethylene glycol) monomethacrylate.
Poly(MPC)-grafted X-UHMWPE for artificial hip joints
Material properties of poly(MPC)-grafted X-UHMWPE
In Figure 11, a time course of the gravimetric wear of X-UHMWPE cups without and with poly(MPC)-grafting during 20 million cycles of loading is shown. Virtually no clear reduction of weight is observed, even after 2 × 107 cycles; that is, there is no significant wear after ~20 years of normal life.88 This effect is independent of whether the X-UHMWPE substrate is crosslinked and of the material used for the femoral heads.53 The poly(MPC)-grafted X-UHMWPE cups exhibit a greater than 90% reduction in the steady wear rate after these hip simulator tests compared with the untreated X-UHMWPE specimen. Furthermore, a poly(MPC)-grafted X-UHMWPE liner showed a continuous weight gain during 7 × 107 cycles, confirming that the poly(MPC) graft layer is maintained even after 7 × 107 cycles. The increased wear resistance is likely due to the hydrated lubricating layer formed by poly(MPC) grafting. One of the important functions of a nanoscale layer of poly(MPC) with cartilage-mimicking structures on a X-UHMWPE liner surface may be to provide hydrophilicity and lubricity identical to those of the physiological joint surface. This suggests that this approach may be promising for extending the longevity of artificial hip joints for THA.50 It is assumed that the hydrated bearing surfaces of artificial joints modified with poly(MPC) exhibit fluid-film lubrication (that is, hydration lubrication25, 89) and suggests that such artificial hip joints mimic cartilage, or the surface-active phospholipid layer on the cartilage, in natural joints.47 Although the poly(MPC) layer has no direct analog with the cartilage surface, which contains a surface-active phospholipid layer, the findings support the hypothesis that highly hydrated macromolecules on such surfaces have a role in chondroprotection and lubrication. Finally, a nanoscale poly(MPC) layer on a X-UHMWPE surface can confer high durability on the acetabular cup bearings used in THA. These observations may have implications for future studies on surface modification using cartilage-like, or surface-active phospholipid layer-like layers, which will be of great importance in the design of lubricated surfaces for artificial joints.
Time course of gravimetric wear of cross-linked ultra-high-molecular-weight polyethylene (X-UHMWPE) cups without and with poly(2-methacryloyloxyethyl phosphorylcholine (MPC))-grafting during 20 million cycles of loading. Data are expressed as the means±standard deviations.88
The poly(MPC)-grafted X-UHMWPE cups show a slight increase in weight. This behavior is partially attributed to the absorption of the medium in the tested cups being greater than that in the load-soak controls, which is in line with the standard ISO14242. The weight loss in the tested cups is corrected, when using the gravimetric method, by subtracting the weight gain in the case of the load-soak controls.
Generally, increased crosslinking in the X-UHMWPE degrades its mechanical properties, producing a trade-off between wear-resistance and mechanical properties.90, 91 It is desirable to reduce wear while maintaining the mechanical properties required for proper in vivo function. The advantage of photoinduced radical graft polymerization results from the fact that the grafted poly(MPC) only gives a high lubricity on the surface and has no effect on the bulk properties of the X-UHMWPE substrate. This indicates that photoscission occurs as a sub-reaction and/or the photoinduced radical graft polymerization proceeds only on the surface of the X-UHMWPE substrates, whereas the properties of the substrates remain unchanged.
Biological performance
The production of wear particles in THA is recognized as the main factor behind the initiation of periprosthetic osteolysis and aseptic loosening.92, 93 The inflammatory cellular response to particles is hypothesized to be dependent upon factors such as particle number, size, shape, surface area and materials.94 With the poly(MPC)-grafted X-UHMWPE cups, the number of wear-generated particles isolated from the lubricants of the hip simulator tests decreased significantly. The wear particles collected from these poly(MPC)-grafted X-UHMWPE liners are on the scale of sub-micrometers, independent of poly(MPC) grafting. However, because the wear-generated particles are produced in extremely small amounts, they cannot be classified according to size. The majority of the wear-generated particles from the untreated X-UHMWPE surfaces have sizes in the range of 0.1 to 1.0 μm. In addition to enhancing the wear resistance of the cups, reducing the bone resorptive responses to the wear-generated particles is also important for preventing periprosthetic osteolysis. Such responses are dependent not only on the total number of wear-generated particles but also on the proportion of particles that are within the most biologically active size range. It has been shown that polymer particles (polystyrene (PSt) particles of ~0.50 μm in diameter) covered with poly(MPC) are biologically inert with respect to phagocytosis by macrophages and subsequent bone resorptive actions (Figure 12).51 Concentrations of bone-resorptive factors, such as tumor necrosis factor-α, interleukin-1, interleukin-6 and prostaglandin E2, increased with the addition of polymer particles; however, poly(MPC)-modified particles did not induce such increases in these concentrations. That is, the poly(MPC) grafting could suppress such biological responses. An increasing number of studies are exploring potential pharmacological modifications for the adverse host response to wear particles using agents such as cytokine antagonists, cyclooxygenase-2 inhibitors and osteoprotegerin, or anti-receptor activators of nuclear factor-kappa B ligand antibody. However, they may cause serious side effects because these must be taken for a long period after surgery and they are also not currently targeted to the site of the problem.
In vitro and in vivo bone resorptions in mouse calvariae. (a) Concentrations of tumor necrosis factor-α (TNF-α), interleukin (IL)-1, IL-6 and prostaglandin E2 (PGE2) in the supernatants of the mouse macrophage-like cell line J774 culture exposure to polystyrene (PSt) particles and those with poly(2-methacryloyloxyethyl phosphorylcholine (MPC)) grafting (poly(MPC)-grafted PSt); *Significant difference from control (P<0.01), (b) bone resorption induced by an injection of the PSt or poly(MPC)-grafted PSt particles into mouse calvariae; representative histological findings of injected sites where osteoclasts were stained red with tartrate-resistant acid phosphatase.51
Clinical applications
Aquala, poly(MPC)-grafted X-UHMWPE, is a new generation X-UHMWPE fabricated using a patented grafting process of MPC on the PE developed by research group at the University of Tokyo and commercialized by KYOCERA Medical Corporation (Osaka, Japan). Takatori et al. performed multicenter clinical trials of the poly(MPC)-grafted X-UHMWPE cups in 80 consecutive patients who underwent THA for a Charnley Class A or Class B painful, non-infectious hip disorder between 2007 and 2009 at the University of Tokyo and various hospitals in Japan.95 Close monitoring of the clinical performance of the cups and accurate quantification of their wear rates are essential for the early recognition of unforeseen problems. With respect to the safety of these implants, clinical trials demonstrated that THA using this liner shows good clinical results, and no adverse events associated with the liner were observed after implantation. Therefore, this liner should be considered to be a safe option in hip replacements. Osteolysis was not observed, and revision surgery was not necessary for up to 7 years post surgery. Considering the efficacy of poly(MPC) grafting in reducing wear, it has reported that the steady-state femoral head penetration rate of poly(MPC)-grafted X-UHMWPE acetabular liners was ~0.002 mm per year, which is less than 10% of that of conventional X-UHMWPE used clinically at present (Figure 13).96 On the basis of the results of the clinical trials and other related evidence, the Japanese government (Ministry of Health, Labor and Welfare) approved the clinical use of poly(MPC)-grafted X-UHMWPE acetabular liners in artificial hip joints in April 2011. In addition, from the data collected to date for over 20 000 clinical applications (March 2015) of poly(MPC)-grafted X-UHMWPE liner in Japan, it has also been found that neither wear nor any osteolysis were observed during follow-up assessment periods that spanned a minimum of 5 years.
Femoral head penetration at 6 months, 12 months and 36 months after surgery. The values of femoral head penetration are calculated using the position at 3 weeks as the original position. Standard deviation bars are displayed.96 X-UHMWPE, cross-linked ultra-high-molecular-weight polyethylene.
Future perspectives
The quality of implanted artificial joints is becoming increasingly important with the need for functional, durable and natural joint-like artificial implants. The author believes that an important research goal for the future is the creation of an artificial joint interface that mimics the natural joint cartilage. Fortunately, the author has proposed a way to achieve this goal, which is to use poly(MPC)-grafted X-UHMWPE liners. Considering that poly(MPC)-grafted X-UHMWPE liners show enhanced lubrication, one can expect improved long-term clinical results from their use. In addition to its effects in providing high wear resistance, the poly(MPC) grafting on X-UHMWPE liner also provides high oxidative stability and excellent mechanical properties for life-long orthopedic bearings. That is, this surface modification can be applied to any type of polymeric material, composites, metals and ceramics. For example, a study is currently underway for the fabrication of a highly lubricious nanoscale modified surface using poly(MPC) grafting onto the surface of an antioxidative X-UHMWPE substrate involving vitamin E blending.97, 98
In 2009, Kyomoto and Ishihara discovered a special photochemical reaction with the super-engineering plastic poly(ether ether ketone) (PEEK).99 Recently, PEEK has been the focus of major attention as new type of polymeric biomaterial, where the potential has been recognized for the substitution of metal and ceramic biomaterials. It has good mechanical properties, chemical stability, and as a biomaterial, the fabrication of PEEK is much easier than that of ceramic and metal biomaterials. However, considerable functionalization of the surface of PEEK will be required for the development of implantable medical devices based on it. PEEK has aromatic ketone units in the main chain, which is the same chemical structure as BP. Consequently, PEEK may be activated by photoirradiation to induce radical formation at the surface. If so, when the monomer exists in the reaction system, polymerization of the monomer is spontaneously initiated at the PEEK surface, leading to grafting and layer formation there (Figure 14). The graft polymerization of MPC has been carried out as follows: the PEEK substrate was immersed in an aqueous solution of MPC and irradiated with light at 365 nm for 60 min at 60 °C. A 100- to 150-nm-thick poly(MPC) layer was generated using a very easy procedure. Surface characterization and evaluation of biological response are currently underway.100, 101, 102, 103 This will be a new polymeric biomaterial that possesses good mechanical and biological properties.
Schematic representation of self-initiated photoinduced graft polymerization of 2-methacryloyloxyethyl phosphorylcholine (MPC) from the poly(ether ether ketone) PEEK surface and a cross-sectional transmission electron microscopy image of the poly(MPC)-grafted PEEK substrate.99 [PEEK]T indicates the triplet state of the PEEK unit.
Such a functional, durable and natural surface will find widespread application in medical devices, such as artificial joints, and will also be important for biomaterials and bioengineering sciences. The surface of articular cartilages consists of a collagen network, hyaluronic acid and proteoglycan subunits. However, the functions of the articular cartilage surface have not yet been fully elucidated. A bioengineered surface based on a new hydrogel-like biomaterial will help provide information on this. In addition, biotribological studies should help to elucidate properties, such as hydration lubrication of the articular cartilage surfaces. Recent advances in regenerated tissue engineering have provided us with regenerated cartilage. The poly(MPC)-grafting substrate can be used for evaluating the performance of this regenerated cartilage as a counter surface of lubrication. The author believes that such polymers and bioengineered surfaces will be key for joint arthroplasty and that this issue will be addressed by future scientists working on biomaterials and in biotribological science.
References
Ratner, B. D., Hoffman, A. S., Schoen, F. J. & Lemons, J. E. (eds) Biomaterials Science 3rd edn (Academic Press, Amsterdam, Netherland, 2013).
Silver, F. H. & Christiansen, D. L. Biomaterials Science and Biocompatibility, Springer-Verlag,, New York, NY, (1999).
Santin, M. & Phillips, G. J. Biomimetic, Bioresponsive and Bioactive Materials, Wiley, Hoboken, NJ, (2012).
Tsuruta, T., Hayashi, T., Kataoka, K., Ishihara, K. & Kimura, Y. (eds) Biomedical Applications of Polymeric Materials, (CRC Press, Boca Raton, FL, 1993).
Kurtz, S., Mowat, F., Ong, K., Chan, N., Lau, E. & Halpern, M. Prevalence of primary and revision total hip and knee arthroplasty in the United States from 1990 through 2002. J. Bone Joint Surg. Am. 87, 1487–1497 (2005).
Kurtz, S., Ong, K., Lau, E., Mowat, F. & Halpern, M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J. Bone Joint Surg. Am. 89, 780–785 (2007).
Merx, H., Dreinhofer, K., Schrader, P., Sturmer, T., Puhl, W. & Gunther, K. P. International variation in hip replacement rates. Ann. Rheum. Dis. 62, 222–226 (2003).
Dumbleton, J. H., Manley, M. T. & Edidin, A. A. A literature review of the association between wear rate and osteolysis in total hip arthroplasty. J. Arthroplasty 17, 649–661 (2002).
Manley, M. T., D’Antonio, J. A., Capello, W. N. & Edidin, A. A. Osteolysis: a disease of access to fixation interfaces. Clin. Orthop. 405, 129–137 (2002).
Kim, S. Changes in surgical loads and economic burden of hip and knee replacements in the US: 1997–2004. Arthritis Rheum. 59, 481–488 (2008).
Kurtz, S. M, Ong, K. L., Schmier, J., Mowat, F., Saleh, K., Dybvik, E., Kärrholm, J., Garellick, G., Havelin, L. I., Furnes, O., Malchau, H. & Lau, E. Future clinical and economic impact of revision total hip and knee arthroplasty. J. Bone Joint Surg. Am 89, 144–151 (2007).
Jacobs, J. J., Roebuck, K. A., Archibeck, M., Hallab, N. J. & Glant, T. T. Osteolysis: basic science. Clin. Orthop. 393, 71–77 (2001).
Glant, T. T., Jacobs, J. J., Molnar, G., Shanbhag, A. S., Valyon, M. & Galante, J. O. Bone resorption activity of particulate-stimulated macrophages. J. Bone Miner. Res. 8, 1071–1079 (1993).
Atienza, C. Jr. & Maloney, W. J. Highly cross-linked polyethylene bearing surfaces in total hip arthroplasty. J. Surg. Orthop. Adv 17, 27–33 (2008).
Collier, J. P., Currier, B. H., Kennedy, F. E., Currier, J. H., Timmins, G. S., Jackson, S. K. & Brewer, R. L. Comparison of cross-linked polyethylene materials for orthopaedic applications. Clin. Orthop. Relat. Res 414, 289–304 (2003).
Muratoglu, O. K., Bragdon, C. R., O’Connor, D. O., Jasty, M. & Harris, W. H. A novel method of cross-linking ultra-high-molecular-weight polyethylene to improve wear, reduce oxidation, and retain mechanical properties. Recipient of the 1999 HAP Paul Award. J. Arthroplasty 16, 149–160 (2001).
Jacobs, C. A., Christensen, C. P., Greenwald, A. S. & McKellop, H. Clinical performance of highly cross-linked polyethylenes in total hip arthroplasty. J. Bone Joint Surg. Am 89, 2779–2786 (2007).
Capello, W. N., Dantonio, J. A., Feinberg, J. R. & Manley, M. T. Alternative bearing surfaces: alumina ceramic bearings for total hip arthroplasty. Instr. Course Lect 54, 171–176 (2005).
Oonishi, H., Kim, S. C., Takao, Y., Kyomoto, M., Iwamoto, M. & Ueno, M. Wear of highly cross-linked polyethylene acetabular cup in Japan. J. Arthroplasty 21, 944–949 (2006).
Wroblewski, B. M., Siney, P. D. & Fleming, P. A. Low-friction arthroplasty of the hip using alumina ceramic and cross-linked polyethylene. A 17-year follow-up report. J. Bone Joint Surg. 87, 1220–1221 (2005).
Kirk, T. B., Wilson, A. S & Stachowiak, G. W. The morphology and composition of the superficial zone of mammalian articular cartilage. J. Orthopaedic Rheumatol 6, 21–28 (1993).
Mow, V. C., Ratcliffe, A. & Poole, A. R. Cartilage and diarthrodial joints as paradigms for hierarchical materials and structures. Biomaterials 13, 67–97 (1992).
Buckwalter, J. A. & Rosenberg, L. Structural changes during development in bovine fetal epiphyseal cartilage. Coll. Relat. Res 3, 489–504 (1983).
Obara, T., Mabuchi, K., Iso, T. & Yamaguchi, T. Increased friction of animal joints by experimental degeneration and recovery by addition of hyaluronic acid. Clin. Biomech. 12, 246–252 (1997).
Ishikawa, Y., Hiratsuka, K. & Sasada, T. Role of water in the lubrication of hydrogel. Wear 261, 500–504 (2006).
Longfield, M. D., Dowson, D., Walker, P. S. & Wright, V. "Boosted lubrication" of human joints by fluid enrichment and entrapment. Biomed. Eng 4, 517–522 (1969).
Ikeuchi, K., Kusaka, J., Yamane, D. & Fujita, S. Time-dependent wear process between lubricated soft materials. Wear 1229, 656–659 (1999).
Wright, V. & Dowson, D. Lubrication and cartilage. J. Anat. 121, 107–118 (1976).
Yoshida, H., Morita, Y., Ikeuchi, K. in Tribological Research and Design for Engineering Systems (eds Dowson, D., Priest, M., Dalmaz, G. & Lubrecht, A. A.) 425–428 (Elsevier, Cambridge, UK, 2003).
Milner, S. T. Polymer brushes. Science 251, 905–914 (1991).
Nagasaki, Y. & Kataoka, K. An intelligent polymer brush. Trends Polym. Sci. 4, 59–64 (1996).
Edmondson, S., Osborne, V. L. & Huck, W. T. S. Polymer brushes via surface-initiated polymerizations. Chem. Soc. Rev. 33, 14–22 (2004).
Kobayashi, M. & Takahara, A. Tribological properties of hydrophilic polymer brushes under wet conditions. Chem. Rec. 10, 208–216 (2010).
Kobayashi, M., Terada, M. & Takahara, A. Polyelectrolyte brushes: a novel stable lubrication system in aqueous conditions. Faraday Discuss. 156, 403–412 (2012).
Kobayashi, M., Ishihara, K. & Takahara, A. Neutron reflectivity study of the swollen structure of polyzwitterion and polyeletrolyte brushes in aqueous solution. J. Biomater. Sci. Polym. Ed. 25, 1673–1686 (2014).
Kobayashi, M., Tanaka, H., Minn, M., Sugimura, J. & Takahara, A. Interferometry study of aqueous lubrication on the surface of polyelectrolyte brush. ACS Appl. Mater. Interfaces 6, 20365–20371 (2014).
Raviv, U., Giasson, S., Kampf, N., Gohy, J. F., Jérôme, R. & Klein, J. Normal and frictional forces between surfaces bearing polyelectrolyte brushes. Langmuir 24, 8678–8687 (2008).
Chen, M., Briscoe, W. H., Armes, S. P., Cohen, H. & Klein, J. Robust, biomimetic polymer brush layers grown directly from a planar mica surface. Chemphyschem 8, 1303–1306 (2007).
Chen, M., Briscoe, W. H., Armes, S. P., Cohen, H. & Klein, J. Lubrication at physiological pressures by polyzwitterionic brushes. Science 323, 1698–1701 (2009).
Ishihara, K., Iwasaki, Y., Ebihara, S., Shindo, Y. & Nakabayashi, N. Photoinduced graft polymerization of 2-methacryloyloxyethyl phosphorylcholine on polyethylene membrane surface for obtaining blood cell adhesion resistance. Colloid. Surf. B Biointerfaces 18, 325–335 (2000).
Kitano, K., Inoue, Y., Konno, T., Matsuno, R., Takai, M. & Ishihara, K. Nanoscale evaluation of lubricity on well-defined polymer brush surfaces using QCM-D and AFM. Colloid. Surf. B: Biointerfaces 74, 350–357 (2009).
Ishihara, K., Ueda, T. & Nakabayashi, N. Preparation of phospholipid polymers and their properties as polymer hydrogel membranes. Polym. J. 22, 355–360 (1990).
Ueda, T., Oshida, H., Kurita, K., Ishihara, K. & Nakabayashi, N. Preparation of 2-methacryloyloxyethyl phosphorylcholine copolymers with alkyl methacrylates and their blood compatibility. Polym. J. 24, 1259–1269 (1992).
Ishihara, K. & Fukazawa, K. in Phosphorus-based Polymers: From Synthesis to Applications (eds Monge, S. & David, G.) 68–96 (RSC Publishing, Cambridge, UK, 2014).
Kyomoto, M., Moro, T., Miyaji, F., Hashimoto, M., Kawaguchi, H., Takatori, Y., Nakamura, K. & Ishihara, K. Effects of mobility/immobility of surface modification by 2-methacryloyloxyethyl phosphorylcholine polymer on the durability of polyethylene for artificial joints. J. Biomed. Mater. Res. A 90, 362–371 (2009).
Kyomoto, M., Moro, T., Saiga, K., Hashimoto, M., Ito, H., Kawaguchi, H., Takatori, Y. & Ishihara, K. Biomimetic hydration lubrication with various polyelectrolyte layers on cross-linked polyethylene orthopedic bearing materials. Biomaterials 33, 4451–4459 (2012).
Kyomoto, M., Moro, T., Miyaji, F., Hashimoto, M., Kawaguchi, H., Takatori, Y., Nakamura, K. & Ishihara, K. Effect of 2-methacryloyloxyethyl phosphorylcholine concentration on photo-induced graft polymerization of polyethylene in reducing the wear of orthopaedic bearing surface. J. Biomed. Mater. Res. A 86, 439–447 (2008).
Kyomoto, M., Moro, T., Konno, T., Takadama, H., Kawaguchi, H., Takatori, Y., Nakamura, K., Yamawaki, N. & Ishihara, K. Effects of photo-induced graft polymerization of 2-methacryloyloxyethyl phosphorylcholine on physical properties of cross-linked polyethylene in artificial hip joints. J. Mater. Sci. Mater. Med 18, 1809–1815 (2007).
Kyomoto, M., Moro, T., Takatori, Y., Kawaguchi, H. & Ishihara, K. Cartilage-mimicking, high-density brush structure improves wear resistance of crosslinked polyethylene: a pilot study. Clin. Orthop. Relat. Res 469, 2327–2336 (2011).
Czichos, H. in Tribology: A Systems Approach to the Science and Technology of Friction, Lubrication and Wear (ed Czichos, H) 1–13 (Elsevier, New York, NY, 1978).
Moro, T., Takatori, Y., Ishihara, K., Konno, T., Takigawa, Y., Matsushita, T., Chung, U. I., Nakamura, K. & Kawaguchi, H. Surface grafting of artificial joints with a biocompatible polymer for preventing periprosthetic osteolysis. Nat. Mater. 3, 829–837 (2004).
Moro, T., Takatori, Y., Ishihara, K., Nakamura, K. & Kawaguchi, H. 2006 Frank Stinchfield Award: Grafting of biocompatible polymer for longevity of artificial hip joints. Clin. Orthop. Relat. Res 453, 58–63 (2006).
Moro, T., Kawaguchi, H., Ishihara, K., Kyomoto, M., Karita, T., Ito, H., Nakamura, K. & Takatori, Y. Wear resistance of artificial hip joints with poly(2-methacryloyloxyethyl phosphorylcholine) grafted polyethylene: comparisons with the effect of polyethylene cross-linking and ceramic femoral heads. Biomaterials 30, 2995–3001 (2009).
Feng, W., Zhu, S., Ishihara, K. & Brash, J. L. Adsorption of fibrinogen and lysozyme on silicon grafted with poly(2-methacryloyloxyethyl phosphorylcholine) via surface-initiated atom transfer radical polymerization. Langmuir 21, 5980–5987 (2005).
Ishihara, K., Nomura, H., Mihara, T., Kurita, K., Iwasaki, Y. & Nakabayashi, N. Why do phospholipid polymers reduce protein adsorption? J. Biomed. Mater. Res. 39, 323–330 (1998).
Ishihara, K., Ziats, N. P., Tierney, B. P., Nakabayashi, N. & Anderson, J. M. Protein adsorption from human plasma is reduced on phospholipid polymers. J. Biomed. Mater. Res. 25, 1397–1407 (1991).
Lewis, A. L. Phosphorylcholine-based polymers and their use in the prevention of biofouling. Colloid. Surf. B: Biointerfaces 18, 261–275 (2000).
Ishihara, K. Bioinspired phospholipid polymer biomaterials for making high performance artificial organs. Sci. Technol. Adv. Mater. 1, 131–138 (2000).
Iwasaki, Y. & Ishihara, K. Cell membrane-inspired phospholipid polymers for developing medical devices with excellent biointerfaces. Sci. Technol. Adv. Mater. 13, 046101(10p) (2012).
Goda, T., Ishihara, K. & Miyahara, Y. A critical update on 2-methacryloyloxyethyl phosphorylcholine (MPC) polymer science. J. Appl. Polym. Sci. 132, 41766(10p) (2015).
Kitano, H., Imai, M., Mori, T., Gemmei-Ide, M., Yokoyama, Y. & Ishihara, K. Structure of water in the vicinity of phospholipid analog copolymers as studied by vibrational spectroscopy. Langmuir 19, 10260–10266 (2003).
Morisaku, T., Watanabe, J., Konno, T., Takai, M. & Ishihara, K. Hydration of phosphorylcholine groups attached to highly swollen polymer hydrogels studied by thermal analysis. Polymer 24, 4652–4657 (2008).
Snyder, T. A., Tsukui, H., Kihara, S., Akimoto, T., Litwak, K. N., Kameneva, M. V., Yamazaki, K. & Wagner, W. R. Preclinical biocompatibility assessment of the EVAHEART ventricular assist device: coating comparison and platelet activation. J. Biomed. Mater. Res. A 81, 85–92 (2007).
Myers, G. J., Gardiner, K., Ditmore, S. N., Swyer, W. J., Squires, C., Johnstone, D. R., Power, C. V., Mitchell, L. B., Ditmore, J. E. & Cook, B. Clinical evaluation of the Sorin synthesis oxygenator with integrated arterial filter. J. Extra. Corpor. Technol 37, 201–206 (2005).
Myers, G. J., Johnstone, D. R., Swyer, W. J., McTeer, S., Maxwell, S. L., Squires, C., Ditmore, S. N., Power, C. V., Mitchell, L. B., Ditmore, J. E., Aniuk, L. D., Hirsch, G. M. & Buth, K. J. Evaluation of Mimesys phosphorylcholine (PC)-coated oxygenators during cardiopulmonary bypass in adults. J. Extra. Corpor. Technol 35, 6–12 (2003).
Kandzari, D. E. & Leon, M. B. Overview of pharmacology and clinical trials program with the zotarolimus-eluting endeavor stent. J. Interv. Cardiol. 19, 405–413 (2006).
Sakurai, R., Hongo, Y., Yamasaki, M., Honda, Y., Bonneau, H. N., Yock, P. G., Cutlip, D., Popma, J. J., Zimetbaum, P., Fajadet, J., Kuntz, R. E., Wijns, W. & Fitzgerald, P. J., ENDEAVOR II Trial Investigators Detailed intravascular ultrasound analysis of Zotarolimus-eluting phosphorylcholine-coated cobalt-chromium alloy stent in de novo coronary lesions (results from the ENDEAVOR II trial). Am. J. Cardiol. 100, 818–823 (2007).
Kandzari, D. E., Leon, M. B., Popma, J.J., Fitzgerald, P. J., O'Shaughnessy, C., Ball, M. W., Turco, M., Applegate, R. J., Gurbel, P. A., Midei, M. G., Badre, S. S., Mauri, L., Thompson, K. P., LeNarz, L. A. & Kuntz, R. E., ENDEAVOR III Trial Investigators Comparison of zotarolimus-eluting and sirolimus-eluting stents in patients with native coronary artery disease: a randomized controlled trial. J. Am. Coll. Cardiol. 48, 2440–2447 (2006).
Abizaid, A., Popma, J. J., Tanajura, L. F., Hattori, K., Solberg, B., Larracas, C., Feres, F., Costa Jde, R. Jr. & Schwartz, L. B. Clinical and angiographic results of percutaneous coronary revascularization using a trilayer stainless steel-tantalum-stainless steel phosphorylcholine-coated stent: the TriMaxx trial. Catheter Cardiovasc. Interv. 70, 914–919 (2007).
Abizaid, A., Lansky, A. J, Fitzgerald, P. J., Tanajura, L. F., Feres, F., Staico, R., Mattos, L., Chaves, A., Centemero, M., Sousa, A. G., Sousa, J. E., Zaugg, M. J. & Schwartz, L. B. Percutaneous coronary revascularization using a trilayer metal phosphorylcholine-coated zotarolimus-eluting stent. Am. J. Cardiol. 99, 1403–1408 (2007).
Han, S. H., Ahn., T. H., Kang, W. C., Oh, K. J., Chung, W. J., Shin, M. S., Koh, K. K., Choi, I. S. & Shin, E. K. The favorable clinical and angiographic outcomes of a high-dose dexamethasone-eluting stent: randomized controlled prospective study. Am. Heart J. 152, 887.e1–e7 (2006).
Kwok, O. H., Chow, W. H., Law, T. C., Chiu, A., Ng, W., Lam, W. F., Hong, M. K. & Popma, J. J. First human experience with angiopeptin-eluting stent: a quantitative coronary angiography and three-dimensional intravascular ultrasound study. Catheter Cardiovasc. Interv. 66, 541–546 (2006).
Airoldi, F., Di Mario, C., Ribichini, F., Presbitero, P., Sganzerla, P., Ferrero, V., Vassanelli, C., Briguori, C., Carlino, M., Montorfano, M., Biondi-Zoccai, G. G., Chieffo, A., Ferrari, A. & Colombo, A. 17-Beta-estradiol eluting stent versus phosphorylcholine-coated stent for the treatment of native coronary artery disease. Am. J. Cardiol. 96, 664–667 (2005).
Rodriguez, A., Rodríguez Alemparte, M., Fernández Pereira, C., Sampaolesi, A., da Rocha Loures Bueno, R., Vigo, F., Obregón, A. & Palacios, I. F., LASMAL investigators Latin American randomized trial of balloon angioplasty vs coronary stenting for small vessels (LASMAL): immediate and long-term results. Am. J. Med. 118, 743–751 (2005).
Bakhai, A., Booth, J., Delahunty, N., Nugara, F., Clayton, T., McNeill, J., Davies, S. W., Cumberland, D. C., Stables, R. H. & Stent, SV Investigators. The SV stent study: a prospective, multicentre, angiographic evaluation of the BiodivYsio phosphorylcholine coated small vessel stent in small coronary vessels. Int. J. Cardiol. 102, 95–102 (2005).
Shinozaki, N., Yokoi, H., Iwabuchi, M., Nosaka, H., Kadota, K., Mitsudo, K. & Nobuyoshi, M. Initial and follow-up results of the BiodivYsio phosphorylcholine coated stent for treatment of coronary artery disease. Circ. J. 69, 295–300 (2005).
Hausleiter, J., Kastrati, A., Mehilli, J., Schühlen, H., Pache, J., Dotzer, F., Glatthor, C., Siebert, S., Dirschinger, J. & Schömig, A, ISAR-SMART-2 Investigators A randomized trial comparing phosphorylcholine-coated stenting with balloon angioplasty as well as abciximab with placebo for restenosis reduction in small coronary arteries. J. Intern. Med. 256, 388–397 (2004).
Boland, J. L., Corbeij, H. A., Van Der Giessen, W., Seabra-Gomes, R., Suryapranata, H., Wijns, W., Hanet, C., Suttorp, M. J., Buller, C., Bonnier, J. J., Colombo, A., Van Birgelen, C., Pieper, M., Mangioni, J. A., Londero, H., Carere, R. G., Hamm, C. W., Bonan, R., Bartorelli, A., Kyriakides, Z. S., Chauhan, A., Rothman, M., Grinfeld, L., Oosterwijk, C., Serruys, P. W. & Cumberland, D. C. Multicenter evaluation of the phosphorylcholine-coated BiodivYsio stent in short de novo coronary lesions: The SOPHOS study. Int. J. Cardiovasc. Intervent 3, 215–225 (2000).
Kuiper, K. K. & Nordrehaug, J. E. Early mobilization after protamine reversal of heparin following implantation of phosphorylcholine-coated stents in totally occluded coronary arteries. Am. J. Cardiol. 85, 698–702 (2000).
Grenadier, E., Roguin, A., Hertz, I., Peled, B., Boulos, M., Nikolsky, E., Amikam, S., Kerner, A., Cohen, S. & Beyar, R. Stenting very small coronary narrowings (<2 mm) using the biocompatible phosphorylcholine-coated coronary stent. Catheter Cardiovasc. Interv. 55, 303–308 (2002).
Raviv, U., Giasson, S., Kampf, N., Gohy, J. F., Jérôme, R. & Klein, J. Lubrication by charged polymers. Nature 425, 163–165 (2003).
Goda., T., Matsuno, R., Konno, T., Takai, M. & Ishihara, K. Photografting of 2-methacryloyloxyethyl phosphorylcholine from polydimethylsiloxane: tunable protein repellency and lubrication property. Colloid. Surf. B Biointerfaces 63, 64–72 (2008).
Serro, A. P., Gispert, M. P., Martins, M. C., Brogueira, P., Colaço, R. & Saramago, B. Adsorption of albumin on prosthetic materials: implication for tribological behavior. J. Biomed. Mater. Res. A 78, 581–589 (2006).
Crockett, R., Roba, M., Naka, M., Gasser, B., Delfosse, D., Frauchiger, V. & Spencer, N. D. Friction, lubrication, and polymer transfer between UHMWPE and CoCrMo hip-implant materials: a fluorescence microscopy study. J. Biomed. Mater. Res. A 89, 1011–1018 (2009).
Fang, H. W., Hsieh, M. C., Huang, H. T., Tsai, C. Y. & Chang, M. H. Conformational and adsorptive characteristics of albumin affect interfacial protein boundary lubrication: from experimental to molecular dynamics simulation approaches. Colloid. Surf. B Biointerfaces 68, 171–177 (2009).
Briscoe, W. H., Titmuss, S., Tiberg, F., Thomas, R. K., McGillivray, D. J. & Klein, J. Boundary lubrication under water. Nature 444, 191–194 (2006).
Kobayashi, M., Terayama, Y., Hosaka, N., Kaido, M., Suzuki, A., Yamada, N., Torikai, N., Ishihara, K. & Takahara, A. Friction behavior of high-density poly(2-methacryloyloxyethyl phosphorylcholine) brush in aqueous media. Soft Matter 3, 740–746 (2007).
Moro, T., Takatori, Y., Kyomoto, M., Ishihara, K., Hashimoto, M., Ito, H., Tanaka, T., Oshima, H., Tanaka, S. & Kawaguchi, H. Long-term hip simulator testing of the artificial hip joint bearing surface grafted with biocompatible phospholipid polymer. J. Orthop. Res. 32, 369–376 (2014).
Raviv, U. & Klein, J. Fluidity of bound hydration layers. Science 297, 1540–1543 (2002).
McKellop, H., Shen, F. W., Lu, B., Campbell, P. & Salovey, R. Development of an extremely wear-resistant ultra high molecular weight polyethylene for total hip replacements. J. Orthop. Res. 17, 157–167 (1999).
Muratoglu, O. K., Bragdon, C. R., O'Connor, D. O., Jasty, M., Harris, W. H., Gul, R. & McGarry, F. Unified wear model for highly crosslinked ultra-high molecular weight polyethylenes (UHMWPE). Biomaterials 20, 1463–1470 (1999).
Harris, W. H. The problem is osteolysis. Clin. Orthop. Relat. Res 311, 46–53 (1995).
Bozic, K. J., Kurtz, S. M., Lau, E., Ong, K., Vail, T. P. & Berry, D. J. The epidemiology of revision total hip arthroplasty in the United States. J. Bone Joint Surg. Am 91, 128–133 (2009).
Tipper, J. L., Galvin, A. L., Williams, S., McEwen, H. M., Stone, M. H., Ingham, E. & Fisher, J. Isolation and characterization of UHMWPE wear particles down to ten nanometers in size from in vitro hip and knee joint simulators. J. Biomed. Mater. Res. A 78, 473–480 (2006).
Takatori, Y., Moro, T., Kamogawa, M., Oda, H., Morimoto, S., Umeyama, T., Minami, M., Sugimoto, H., Nakamura, S., Karita, T., Kim, J., Koyama, Y., Ito, H., Kawaguchi, H. & Nakamura, K. Poly(2-methacryloyloxyethyl phosphorylcholine)-grafted highly cross-linked polyethylene liner in primary total hip replacement: one-year results of a prospective cohort study. J. Artif. Organs 16, 170–175 (2013).
Takatori, Y., Moro, T., Ishihara, K., Kamogawa, M., Oda, H., Umeyama, T., Kim, Y. T., Ito, H., Kyomoto, M., Tanaka, T., Kawaguchi, H. & Tanaka, S. Clinical and radiographic outcomes of total hip replacement with poly(2-methacryloyloxyethyl phosphorylcholine)-grafted highly cross-linked polyethylene liners: three-year results of a prospective consecutive series. Mod. Rheumatol 25, 286–291 (2015).
Kyomoto, M., Moro, T., Yamane, S., Watanabe, K., Hashimoto, M., Takatori, Y., Tanaka, S. & Ishihara, K. Poly(2-methacryloyloxyethyl phosphorylcholine) grafting and vitamin E blending for high wear resistance and oxidative stability of orthopedic bearings. Biomaterials 35, 6677–6686 (2014).
Kyomoto, M., Moro, T., Takatori, Y., Tanaka, S. & Ishihara, K. Multidirectional wear and impact-to-wear tests of phospholipid-polymer-grafted and vitamin E-blended crosslinked polyethylene: A pilot study. Clin. Orthop. Relat. Res 473, 942–951 (2015).
Kyomoto, M. & Ishihara, K. Self-initiated surface graft polymerization of 2-methacryloyloxyethyl phosphorylcholine on poly(ether-ether-ketone) by photoirradiation. ACS Appl. Mater. Interfaces 1, 537–542 (2009).
Kyomoto, M., Moro, T., Takatori, Y., Kawaguchi, H., Nakamura, K. & Ishihara, K. Self-initiated surface grafting with poly(2-methacryloyloxyethyl phosphorylcholine) on poly(ether-ether-ketone). Biomaterials 31, 1017–1024 (2010).
Kyomoto, M., Moro, T., Yamane, S., Hashimoto, M., Takatori, Y. & Ishihara, K. Poly(ether-ether-ketone) orthopedic bearing surface modified by self-initiated surface grafting of poly(2-methacryloyloxyethyl phosphorylcholine). Biomaterials 34, 7829–7839 (2013).
Tateishi, T., Kyomoto, M., Kakinoki, S., Yamaoka, T. & Ishihara, K. Reduced platelets and bacteria adhesion on poly(ether ether ketone) by photoinduced and self-initiated graft polymerization of 2-methacryloyloxyethyl phosphorylcholine. J. Biomed. Mater. Res. A 102, 1342–1349 (2014).
Kyomoto, M., Moro, T., Yamane, S., Watanabe, K., Takatori, Y., Tanaka, S. & Ishihara, K. Smart PEEK modified by self-initiated surface graft polymerization for orthopeadic bearings. Reconst. Rev 4, 36–45 (2014).
Acknowledgements
This review summarizes the research that won the Award of the Society of Polymer Science, Japan (2013). I express sincere appreciation to Dr Toru Moro, Dr Yoshio Takatori, Dr Sakae Tanaka, Dr Hiroshi Kawaguchi and Dr Kozo Nakamura of the University of Tokyo, and Dr Masayuki Kyomoto of KYOCERA Medical Corporation. Additionally, I thank all of the staff: Dr Junji Watanabe, Dr Tomohiro Konno, Dr Ryosuke Matsuno, Dr Yuuki Inoue, Dr Kyoko Fukazawa and students belonging to the laboratory for providing a fine support to this research. This research was carried out with excellent cooperation with Medical School of the University of Tokyo and KYOCERA Medical Corporation and supported financially by Japan Science and Technology Agency (JST) and Japan Agency for Medical Research and Development (AMED).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ishihara, K. Highly lubricated polymer interfaces for advanced artificial hip joints through biomimetic design. Polym J 47, 585–597 (2015). https://doi.org/10.1038/pj.2015.45
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/pj.2015.45
- Springer Nature Limited
This article is cited by
-
Composite lubricating layer with enhanced wear-resistant properties between HXLPE and Cu/ZrO2 ceramic friction interface
Journal of Materials Science (2024)
-
Plastic–elastic Model for Water-based Lubrication Considering Surface Force
Chinese Journal of Mechanical Engineering (2022)
-
Optimization of micromachining operation for particle reinforced UHMWPE composites
Archives of Civil and Mechanical Engineering (2022)
-
Updates in biomaterials of bearing surfaces in total hip arthroplasty
Arthroplasty (2021)
-
Biomimetic cartilage-lubricating polymers regenerate cartilage in rats with early osteoarthritis
Nature Biomedical Engineering (2021)