Abstract
Purpose of Review
This study aims to communicate updated information on the recent innovations in grapefruit byproduct valorization.
Recent Finding
Grapefruit is an important fruit of the citrus genus which has commercial importance and its processing generates waste in bulk, mainly in the form of peels, seeds, and pomace, which only leads to a strenuous waste stream that ends up in landfills causing environmental issues if overlooked. However, grapefruit byproducts are rich in high-value compounds including dietary fiber, polyphenols, pectin, and essential oil, which therefore could be valorized for different applications in the food sector and other realms as well. In line with the United Nations Sustainable Development Goals (UN-SDGs) to ensure sustainable consumption and production patterns, the valorization of these byproducts in the most efficient and environment-friendly manner is of great importance for the future.
Summary
The valorization of grapefruit byproducts can be addressed through environmentally friendly extraction procedures that allow recovery of target high-value compounds and open new vistas for their applications. Overall, this work describes an updated tapestry of reports about the characteristics and compositions of grapefruit byproducts. In parallel, it offers an updated vision of high-value compounds and the various extraction techniques used for their extraction have been discussed. Comprehensively, the current review summarizes the latest advancements in the application of high-value compounds from grapefruit waste in the numerous areas of the food, pharma, and cosmetics realm, along with the utilization for development of environmentally sustainable materials, fostering a sustainable economy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
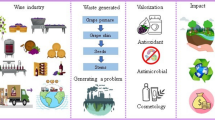
Grapefruit (Citrus paradisi) is a commercially important cultivar of the citrus family that encompasses a myriad of bioactive chemicals. Originating in Barbados in the eighteenth century, it is reported to be an accidental cross between the sweet orange (C. sinensis) and the pomelo (C. maxima). It is widely distributed throughout the world’s subtropical and tropical regions [1,2,3]. During the marketing year 2022/2023, global grapefruit production amounted to about 6.81 million metric tons [4], with China ranking top among the producers followed by Mexico, South Africa, and the U.S. The grapefruit market is flourishing due to its rich nutrients and health-promoting properties [5]. Grapefruit varieties include Marsh Seedless, Duncan, Red Blush, Flame, Foster, Star Ruby, Thompson, and White Marsh [6]. Mainly consumed in the form of juice and segments, grapefruit can be transformed into various confectionery items. However, growing recognition and increased grapefruit production also draw attention to the wastes and byproducts generated from its processing. These byproducts mainly include peels, seeds, and pomace, and account for more than 50% of the total fruit weight like other citrus fruits [7]. Improper disposal of this organic waste, therefore, can invoke the release of greenhouse gases like methane and volatile compounds during decomposition in landfills which contributes to climate change [8]. Therefore, eco-friendly valorization of grapefruit processing waste is crucial.
Grapefruit byproduct Valorization Towards the Circular Economy
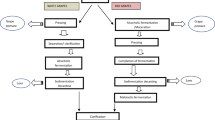
To promote sustainable development, the United Nations Assembly adopted the "2030 Agenda" in 2015, outlining 17 major goals, also known as the Sustainable Development Goals, or SDGs [9]. The conversion of food wastes and byproducts into valuable products presents a significant potential and overall opportunity to support sustainable development, adhering to the circular economy approach, upon which the futurologists and policymakers stress to implement. Opposite to the conventional “cradle-to-grave” or “take-make-waste” approach, the circular economy follows a “cradle-to-cradle” approach [10, 11]. Deemed as the blueprint for a sustainable future, the circular economy is a closed-loop system and is generally opposite to the linear economy that makes waste. Governments and the food industries are implementing policies to reduce food waste, with studies highlighting the potential of transforming byproducts into value-added products to contribute to meeting the ‘zero waste’ target. Henceforth, adequate waste management in the most systematic and eco-friendly manner is of great interest for the future. By efficiently utilizing grapefruit byproducts, we can adopt a sustainable consumption and production pattern that aligns with the Sustainable Development Goals (SDGs), in particular SDG 12 (responsible consumption and reproduction) [12, 13]. This article provides an updated overview of grapefruit byproducts as natural resources of bioactive compounds, and their extraction methods, alongside valorization strategies for their comprehensive and effective utilization in food, pharma, and cosmetic industries to promote sustainability (Fig. 1).
Grapefruit Processing byproducts
Peels
Grapefruit peels are important byproducts accounting for around 35.0–41.0% of the total fruit [14]. Proximate analysis for crude protein, fat, ash, crude fiber, and carbohydrate, reports around 9.27–10.71%, 6.13–6.64%, 3–3.97%, 7.55, and 60.22–71.86, respectively [15, 16]. Grapefruit peels, which contain moisture and sugars and are perishable, can pose environmental problems and, therefore seek apt utilization. They are the source of polyphenols, which are confirmed to possess diverse bioactivities, beneficial to human health. TPC was reported to be 77.3 mg of gallic acid equivalent/g peels [17]. In particular, naringin is the most abundant flavonoid present in peels followed by isonaringin, and hesperidin, which can be used as functional ingredients in food and therapeutically [18]. Apart from this, peels account for 59.77% of the insoluble dietary fiber (IDF) fraction [19, 20], and can also be harnessed to yield pectin, EO, and peel extract, making them a valuable resource for valorization.
Seeds
Grapefruit seeds are the premier repositories for limonoids (triterpenoid dilactones chemically related to limonin), from which 77% are neutral while 2% are acidic limonoids [21]. The number of seeds present in grapefruits varies with varieties, with Duncan having around 50–70, while Marsh seedless, has very few to no seeds, as the name illustrates [22]. The chemical composition might vary among the seeds from different cultivars and geographical niches, though oil content lies from 40.2 to 45.5%, similar to that of seeds from the same genus. Grapefruit seed oil consists of both saturated and unsaturated or omega fatty acids, with palmitic acid, oleic acid, and linoleic acid as key constituents accounting for more than 20% of most grapefruit seed oils [23], apart, fat-soluble bioactive compounds such as tocopherols, carotenoids, and phytosterols are present in seeds. Different fatty acids were quantified in the oil such as linoleic acid (40.78–40.95%), palmitic acid (28.19–28.66%), and oleic acid (20.74–20.78%), linolenic (5.45–5.71%), stearic (3.77–3.81%), and palmitoleic (0.40–0.75%) acids [24]. Seeds are often transformed into extracts for various food and pharma applications [21]. The typical proximate for grapefruit seed divulges 36.54% oil content, 3.90% protein, 8.50% fiber, and 5.03% ash content [25].
Pomace
Following the juice extraction, the solid residue left is called pomace. Proximate analysis of grapefruit pomace unveiled 7.69 ± 0.02 crude protein, 50.33 ± 2.1 carbohydrate, 6.13 ± 0.01 crude fat, 2.16 ± 0.01 ash, 24.79 ± 2.4 as nonfibrous carbohydrates such as starch and sugars [16]. Like other citrus fruits, grapefruit pomace is a sound source of dietary fiber, along with health-promoting compounds, and hence therefore can be used to improve the nutritional content of foods. Since pomace is indexed by its high moisture and soluble sugar content, it makes it highly susceptible biomass to fermentation and microbial degradation. The conventional management route follows dried pellet production at a commercial scale. However, ascribable to the reported dietary fiber and polyphenols present, grapefruit pomace can be explored for edible packaging [26], nutritional content improvement [27], and as a functional agent [16, 28]. Pectin from grapefruit pomace with significant emulsifying and gelling properties can be used as a food additive [29].
High-value Components from Grapefruit byproducts
Dietary Fiber
Often referred to as the “seventh nutrient”, dietary fiber (DF) is an important component of the human diet [30]. The total dietary fiber of grapefruit waste (peel and pomace) is reported as 90.34%, with 7.03% as soluble and 83.31% as insoluble fraction [31]. DF mainly comprises soluble dietary fibers (SDF) and insoluble dietary fibers (IDF) and are linked with various health benefits including improving digestive health, serum-lipid concentrations, and reduced risk of cancers [32, 33]. SDF content in grapefruit peel is reported as 3.62 ± 0.13%, however, extraction with modifications in microwave-assisted extraction increased yield significantly (7.94 ± 0.20%) [19]. In recent times investigations have been conducted to improve the IDF/SDF ratio from grapefruit byproducts. SDF from grapefruit peel IDF (GP-IDF-SDF) obtained with microwave and enzymatic methods given 9.2 ± 0.36% yield, with excellent glucose adsorption, water and oil retention capacity 14.49 ± 0.068 mg/g, 13.43 ± 1.19 g/g, and 22.10 ± 0.85 g/g, respectively [34]. Arabinose (100.72 mg/g db) equates to the main monosaccharide in grapefruit peel SDF, followed by glucose (84.00), fructose (31.40), galactose (17.60), and rhamnose (10.85). Compared to orange and lemon peels, grapefruit peels contain a higher amount of uronic acid (UA) (130.72 GUAE/g db), alluding to higher pectic polysaccharide contents [33], ascribable to which, it could find applications in the pharmaceutical industry for its beneficial effects on gastrointestinal health.
Pectin
Pectin, a soluble fiber, finds extensive use as a thickener, stabilizer, and gelling agent and replacement for fat or sugar in low-calorie foods. Pectin is a key ingredient in various pharmaceutical formulations, particularly in producing dietary supplements and oral dosage forms like tablets and capsules. Global pectin consumption as an additive has reached more than 60,000 tonnes, with industry experts projecting a steady 5.8% compound annual growth rate by 2024 in the global pectin market [35, 36]. Grapefruit peel pectin comprises shorter side chains and is richer in rhamnogalacturonan I backbones, which also imparts its valuable bioactivities [33, 35]. Different innovative extraction approaches can be useful tools to increase pectin yield and quality. DES (betaine-citric acid) based extraction from grapefruit peels provided a significant yield of 36.47% pectin with higher RG-I value, more arabinan side chains, and bigger Mw and Mn values, better emulsifying activity and stability than conventional HCl-extracted pectin [36], while microwave-assisted High-Pressure CO2/H2O system (147 °C, 3 min, and 10 mL g−1) of 27.53% [37], and monosonication-assisted with 17.10% pectin yield [38].
Phenolic Compounds
Phenolic compounds are natural bioactive molecules that are ubiquitous in fruits and are of significant merit due to their health-promoting bioactivities. Grapefruit byproducts have been investigated and affirmed as a good source of phenolic antioxidants. Grapefruit byproducts are rich in flavonoids. Among these, high levels of bioactive flavanones glycosides, namely, naringin and narirutin, and their aglycones, naringenin has been reported in peel and seed residues [39]. Apart, eritrocin, poncirin, neoponcirin, and neohesperidin are also effective antioxidants attributable to their ability to stabilize and inhibit free radicals [40, 41]. The HPLC analysis also pointed out the presence of phenolic acids (resveratrol, gallic acid, ellagic acid, and caffeic acid), and tannin (catechin) in grapefruit byproducts [42]. Levels of polymethoxylated flavones, sinensetin, nobiletin, and tangeretin range from 1.03 to 3.45 mg/g DW in fresh grapefruit peels, while dried (oven or freeze-dried) peels exhibit lower concentrations [39]. Peels also contain ferulic, sinapic, p-coumaric, and caffeic acids (32.3, 31.9, 13.1, and 5.6 mg/100 g, respectively) [43]. The presence of hydroxyl groups on phenolic rings and their ability to attract free radicals with available hydrogen atoms is deemed to be the reason for the antioxidant activity of these compounds [44]. Extraction of different bioactive compounds can be done with different approaches such as microwave-assisted (MAE) [45], ultrasound-assisted (UAE) [46], and enzyme-assisted extractions (EAE) [47]. Nishad et al. [47] divulged higher TPC yields of 2116.71 and 3170.35 mg GAE/100 g with UAE and EAE, respectively, compared with conventional solvent extraction (CSE) of 1528 mg GAE/100 g [20]. Garcia-Castello et al. [46] also reported better yields for UAE (on average TPC 50% and TAA 66% higher) with lower temperatures and extraction times, compared to CSE. Among individual phenolic compounds, naringin was the most abundant flavonoid in the UAE (24 − 36 mg/ g dw) followed by hesperidin (0.72 − 1.14 mg/g dw) and narirutin (0.42 − 0.98 mg/g dw). Grapefruit peel waste may potentially turn out to be a good source of flavonoids, especially naringin, that could be used for food fortification [48] or as therapeutic agents for pharmacological propositions [39].
Essential oil
Grapefruit essential oil obtained from peels is one of the primary grapefruit byproducts, known for its characteristic aroma with wide applications. The major components of essential oil (EO) are terpene oxides, including alcohols, ethers, aldehydes, ketones, and esters, which are attributed to the aroma [49]. Grapefruit EO, primarily extracted through cold pressing, is now being explored through steam distillation and hydrodistillation. GC–MS analysis identified 25 compounds, with D-limonene being the main component. [50,51,52]. Other compounds include β-Phellandrene (4.17%), β.-Myrcene (2.51%), and o-Cymene (1.18%) [51, 53]. Grapefruit EO is promoted to exhibit apoptotic, antioxidant, olfactory stimulation, antibacterial, antifungal, insecticidal, acaricidal, and repellency properties [49]. Denkova-Kostova et al. [52] reported 87.5% DPPH free radical inhibition at a concentration of 1 mg/cm3 for grapefruit EO and also highlighted antimicrobial activity against saprophytic microorganisms, spore-forming bacteria, yeast, and fungi. Apart, EO sensibility to external agents like ultraviolet light, high temperatures, and water, may affect their composition, which facilitates the loss of some specific properties, especially D-limonene is prone to oxidation, which is why encapsulation is feasible for stability, controlled release and enhancement the various characteristics of EO [54,55,56].
Seed Extract
Grapefruit seed extract (GSE) is a natural product containing tocopherols, citric and ascorbic acids, and flavonoids, with significant antioxidant and antimicrobial properties [57]. Phenolic acids, i.e., trans-ferulic acid, rosmarinic acid, trans-2-hydroxycinnamic acid, and flavonoids, are deemed to be the chief antioxidant active ingredients responsible for antioxidant activity [23, 58]. GSE is a natural food preservative to maintain food quality of various types of foods, including meat, fish, poultry, fruits, cheese, and vegetables [59]. It can be applied directly and can be used to fabricate various composite functional films [60, 61]. The antimicrobial activity of GSE equates to the bacterial membrane disruption and liberation of the bacterial cytoplasmic contents within a relatively short time [23, 59]. GSE also finds applications in the pharma and health sector for wound healing applications [62], oral healthcare [63], treating urinary tract infections [64], gastritis and gastric ulcers [65], improving kidney activity, purifying the blood, and help keep cholesterol levels under control. Owing to the reported biological properties, GSE can be explored for the line-up of cosmetic products, including facial cleansers, creams, and serums. [66].
Minerals and Vitamins
Micronutrients include minerals and vitamins because small amounts of these components are needed for the body. Peels of the grapefruit grown in Turkey are reported for minerals, including potassium (K) (111 – 117), calcium (Ca) (34.8 – 38.9), phosphorus (P) (19 – 22.5), magnesium (Mg) (9.50 – 11.1) (mg 100 g−1), respectively [67]. In another study, Saleem et al. [68] quantified the micro- and macro-elements and reported for Fe (3.53), Mn (0.36), Cu (0.14), Zn (0.14), Mg (79.33), K (984.33), and Ca (801) (mg/100 g). Reported different values are attributable to the differences in soil conditions, climate, and cultivars. The vitamin C content of grapefruit peels was found to be 113.3 mg/100 g [17], while Vitamin E, which is a fat-soluble vitamin, has been confirmed. Further authors have quantified a-, γ-, δ- tocopherol as 380.00, 43.41, and 9.08 mg/kg, respectively [25].
Various studies indicate that all these high-value components are interesting for the food, pharma, and cosmetic industries, and their reutilization would promote a circular and sustainable economy around the grapefruit industry. This strategy is relevant to harmonize with the SDGs, specifically to minimize grapefruit waste, in line with the circular economy model which pivots on bringing the waste back into the streamline of production so that it goes back into the production loop and can either become the resource for the next cycle of production or is channeled for an independent new product. However, to maximize the environmental benefits of reusing these wastes, it is required the application of green extraction techniques to obtain optimal production yields ecologically and economically.
Extraction Technologies for Extraction of Various Components
The extraction of bioactive compounds from food processing waste is a critical step. Conventional methods have drawbacks like higher energy expenditure and toxic solvents, prompting the search for efficient technologies like ultrasound, microwave, enzyme-assisted, and supercritical fluid [69,70,71,72,73,74]. Extraction of functional and bioactive compounds from grapefruit byproducts using various techniques has been framed in Table 1 and discussed hereunder.
Ultrasound-assisted Extraction
Ultrasound follows the acoustic cavitation principle. When ultrasound propagates through any medium it generates a series of compressions and rarefactions in the molecules of the medium, such alternate pressure changes induce the formation, growth, and consequently implosion of air bubbles in a liquid medium. Such violent collapse produces extremely high pressures and temperatures at the surface of cell membranes of bio matrices, due to which cell destruction occurs, causing localized damage to the plant tissues termed ‘erosion’. This event creates microchannels, making the intercellular content more available to the solvent, and increasing the yield of extraction [86, 87]. Ultrasound applications are versatile and can be used to treat grapefruit byproducts for extraction of polyphenols, pectin, polysaccharides, dietary fibers, and oils. For instance, Garcia-Castello et al. [46] reported a higher TPC range for UAE (29.4 to 80.0 mg GAE/g dw) than for the conventional approach (25.3 to 55.8 mg GAE/g dw) from grapefruit solid wastes. In the latest investigation, Islam et al. [75] reported TPC (78.5 mg GAE/g dw), TFC (53.5 mg naringin/g dw), naringin (40.8 mg/g dw), and TAA by DPPH (25.5 mM Trolox/g dw), and FRAP assay (17.45 Fe [II]/g dw), for treated 2 g grapefruit peel powder with ultrasonication. Ultrasound is a rapid process, provides higher extraction yields, easy to operate, and requires less investment costs, and versatility, strengthens its ability to be implemented at the industrial level, among others.
Microwave-assisted Extraction
Microwave-assisted extraction functions the application of non-ionizing electromagnetic waves with frequencies ranging from 300 MHz to 300 GHz to a sample matrix that induces changes in the cell structures. Responsible mechanisms for energy transfer in MAE involve ionic conduction and dipole rotation [69, 70]. Ionic conduction in particular pertains to the movement of ions through a solution, eliciting a homogeneous heat in the media ascribable to the resistance of the solvent to the ionic migration upon application of electromagnetic waves. Dipole rotation is enacted by the interaction of dipoles with polar components and elicits the dipoles to realign with the applied field, instigating coerced molecular movements that produce heat [88]. The moisture content in the sample significantly influences MAE, as water evaporation increases intracellular pressure, breaking cell walls, and leaching high-value compounds [89]. Taşan & Akpınar [79] reported a 20.93% pectin yield from grapefruit peels using MAE (pH 1, 30 ml/g solvent/solid ratio, 90 s) with significantly lower extraction times than conventional extraction without compromising on yield and quality of pectin. In a different study, a 17.19 ± 0.35% increased yield of soluble DF was obtained with MAE treatment of grapefruit peels [19]. Merits for the technology include operational ease and low running costs. Apart, higher outputs can be generated with a punctilious selection of operating conditions such as temperature, solid-to-liquid ratio, extraction duration, microwave power, and stirring [90].
Supercritical fluid Extraction
The supercritical fluid extraction (SFE) approach follows the solvation of compounds of interest in a solvent maintained typically above its critical pressure and temperature. CO2 is commonly used for SFE applications and behaves as supercritical fluid above the critical temperature and pressure and shows improved productivity for solubilizing non-polar compounds. Elevating the temperature and pressure of supercritical fluid beyond 4000 psi significantly improves solubility for highly efficient extractions in shorter periods [91]. Supercritical fluid carries target compounds past the pressure release valve into the separator, where lower pressure separates CO2 from the extracted compounds and routed back into the CO2 tank to be used again. Priyadarsani et al. [92] reported for 93% extraction efficiency of lycopene from grapefruit endocarp at 305 bar pressure, 35 g/min CO2 flow rate, 135 min of extraction time, and 70 °C temperature. Latestly, Yaldiz et al. [93] reported TPC values of 79.60 mg GAE/g from grapefruit peel waste when treated at 150 bar, 70 °C, and a cosolvent ratio of 20% (v/v) ethanol. Short extraction time, a low amount of solvent requirement, and rapid solvent recovery, account for the advantages of the technology [94], while the major setbacks are the high cost of the equipment and operation and optimization complexity.
Enzyme-assisted Extraction
This approach involves exclusive enzymes (such as cellulases, amylases, pectinases) to break down bound chemicals, and resultantly enhance the extraction through cell wall breakdown and polysaccharide hydrolysis. Phenolic compounds are entangled within the cell wall polysaccharides like cellulose, hemicellulose, and pectin and are linked by hydrophobic interactions and hydrogen bonds. In particular grapefruit peel flavonoids, are covalently linked by a glycosidic bond with sugar moieties through an OH group (O-glycosides) or carbon–carbon bonds (C-glycosides) [20, 95]. Enzymes pounce upon the internal spots of the amorphous region of the polysaccharide chains which prompts small oligosaccharides generation of uneven length that facilitates easy release of entrapped molecules [96]. Pectinase and cellulase (5, 6, 7 U g−1 enzyme concentration) used to extract phenolic compounds at different temperatures (40–60 °C) and time (6–24 h) combinations from grapefruit peels enhanced the extraction yields (p < 0.05) [97].
Natural Deep eutectic Solvents
Natural deep eutectic solvents (NADES) are sustainable solvents fabricated by blending a hydrogen bond acceptor (e.g. choline-chloride) with a hydrogen bond donor (such as sugars, alcohols, and amines), and up to 50% (v/v) water; at a precise ration to develop a liquid solvent mixture. DESs are liquid at low temperatures, miscible with water, non-flammable, and highly viscous [18]. NADES are widespread in nature and are more based on biological than chemical concepts since ionic liquids or deep eutectic solvents might exist in nature with specific physiological functions [98]. In the latest investigation, Lin et al. [36] extracted pectin from grapefruit peels with betaine-citric acid (BC-P) at an L/S ratio of 25 mL/g, 2.0 pH, and 85 °C for 120 min with a higher yield (36.47%), compared to HCl-extracted pectin (HCl-P, 8.76%) under a pH of 2.0. BC-P exhibited a higher RG-I (Rhamnogalacturonan I) value, Mw, and more arabinan side chains, than HCl-P. The authors also stated higher viscosity, emulsifying activity, and stability compared to HCl-P and commercial pectin. NADES is a versatile method for extracting bioactive compounds from grapefruit byproducts, offering high solubilization strength and easy extraction. Drawbacks such as high viscosity and very low vapor pressure are there, but anti-solvents can be used to confront the issue, moreover, liquid–liquid or solid–liquid extraction can be performed [99].
Pooling Technologies
Numerous studies have reported the integration of novel extraction technologies with beneficial and efficient results [100]. The food industry is heavily focused on lowering manufacturing costs, either by increasing process speed or increasing yield. This refers to either using one thriving technique or combining two or more techniques to achieve its goal. For grapefruit peel, Peng et al. [20] combined microwave (600 W, 85 °C, and 37 min) and enzymatic treatment (8% cellulose, 60 °C, 2 h) (ME-BP) to free insoluble bound phenolic compounds. The obtained results showed higher values for the combined treatment (12.46 ± 0.028 GAE mg/100 g) compared to enzymatic (7.17 ± 0.044 GAE mg/100 g) and microwave (9.95 ± 0.049 GAE mg/100 g) alone.
Purification and Detection of Bioactive Compounds
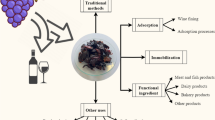
Impurities are introduced during the extraction process and must be isolated immediately to validate the safety and purity of the compound of interest. Different methodologies for purification and characterization are employed as post-extraction treatments. In particular, citrus waste comprises different polysaccharides and other phytochemicals in tandem with bioactive compounds. These compounds are also extracted during the solid–liquid extraction process. To separate a particular bioactive compound, it should be availing in concentrated form in the solution, which can be done by the polarity and pH of the solvent engaged. Non-polar solvents are commonly utilized for lipid fractions, whereas, polar ones are preferable to isolate the ionic compounds [101, 102]. Solid-phase extraction is carried out for the removal of carbohydrates. Sugars, polar nonphenolic compounds, and organic acids could interfere with the total polyphenol content analysis, therefore crude polyphenolic extracts from grapefruit solid wastes are quite commonly purified by using C18 chromatography cartridges [46]. Fractionalization of polyphenols is brought about with methanol and/or acetone from the polyphenol concentrate [101]. In particular, for flavonoid, i.e., naringenin, Chen et al. [103] reported improved performance of CMIPs when employed to enrich naringenin in grapefruit peel extract compared with the common adsorbent materials including AB-8, D101, cationic exchange resin, and active carbon. Flavonoids can also be adsorbed with Indion PA 800 and later desorbed using ethanol. The purification phase culminates in different scaled filtrations, while the identification and characterization of bioactive phytochemicals from grapefruit byproducts is done using different chromatographic and spectrophotometric techniques, such as HPLC–DAD [39], HPLC–MS [103, 104], thin layer chromatography (TLC) [105, 106], ultrahigh performance liquid chromatography (UPLC) [107], gas chromatography-mass spectrometry (GC‒MS) [53], nuclear magnetic resonance (NMR) and UV-spectrophotometry [75]. The schematic representation of extraction to the identification of bioactive compounds utilizing innovative approaches is illustrated in Fig. 2.
Grapefruit byproduct Utilization
Food sector
Bioactive ingredients work well as an integrant to produce nutritious goods and nutraceuticals with improved technological and biofunctional qualities. The following section catalogs and discusses the uses of various grapefruit byproducts for different food applications (Table 2).
Food Packaging
The rejection of synthetic materials and the shift toward renewable and eco-friendly materials for packaging has fueled research for green alternatives [123, 124]. Moreover, the global market for edible coatings and films has been projected to grow at a rate of 7.70% (CAGR) during the next five years to reach a value of 4.54 billion US$ in 2028 [125]. Grapefruit waste yields pectin, essential oils, and seed extracts that can be used in bio-based coatings and films. Moreover, active ingredients may enhance the flavors, colors, antimicrobial, and antioxidant properties of films, ultimately improving food quality [126]. In a study, Zanganeh et al. [127] fabricated a Lallemantia iberica seed mucilage (LISM) coating incorporated with grapefruit EO (0–2% v/v), which reduced microbial growth and lipid oxidation. The authors also reported that lamb with 2% v/v EO concentration had better quality maintenance and extended shelf life (> 9 days). Chiabrando & Giacalone [128] tested the potential of alginate-based grapefruit EO coating for fresh-cut kiwifruit quality preservation. The results showed a lowered respiration rate, increased firmness, and vitamin C content, and curbed yeast and mold flourishing. Roy & Rahim [60] tested the antioxidant potential of grapefruit seed extract (GSE) after addition to a poly(vinyl alcohol)-based film. The obtained results showed escalated DPPH scavenging activity of 50.3% with GSE in comparison to the control film (0.7%), while ABTS activity reached 90.2% with GSE incorporation.
Baek et al. [129] investigated the potential of sodium alginate nanoparticle-based grapefruit seed extract (GSE) coating for safety and quality maintenance of shrimp stored at 4 °C for 8 days. Upon coating on shrimp, nanoparticles (1% alginate + 1% GSE) prevented the microbiological limit from exceeding during 8 days of storage, while uncoated shrimp exceeded the limit on the 4th day. Additionally, nanoparticles markedly attenuated the TVB-N values and showed the lowest weight loss when compared to other samples. Chitosan coating with GSE (1.0% w/w) inactivated Salmonella by 2.0 ± 0.3 log CFU without affecting the lycopene content, color, or sensorial properties of fruits, as reported by Won et al. [130]. Bionanocomposites based on halloysite-encapsulating grapefruit seed oil (GO) (2.5 wt%) showed marked mold prevention and better preservation of the fruit texture and appearance for strawberries [131]. Citrus peels are among the major industrial sources for pectin extraction, and grapefruit peel pectin is another component that needs to be studied further for use as a packaging film/coating biomaterial.
Antimicrobial Agent
The use of natural antimicrobials as food preservatives prevents the extremities of physical and chemical processing [132]. In a study, grapefruit EO was tested against food-borne pathogens. The results revealed bacteriostatic properties against most of the tested bacterial strains. Essential oil at concentrations up to 25 mg/mL effectively suppressed Salmonella parathypi A, Vibrio vulnificus, and Seratia liquefaciens growth. Grapefruit EO, when used in a nanoemulsion system, ameliorated bacteriostatic potency [51].
Grapefruit seed extract (GSE) in particular has a broad antimicrobial spectrum against a variety of microbial strains. Choi et al. [133] reported MIC values of GSE against the food-borne pathogens B. subtilis, C. albicans, E. coli O157:H7, P. aeruginosa, S. enteritidis, and S. aureus in the range 0.0061 to 0.7813 μL/mL, and promoted GSE as an efficacious natural additive that prolonged the shelf-life of fresh Makgeolli with no significant loss in quality. Antibacterial mechanism of GSE accounts for the bacterial membrane disruption and liberation of cytoplasmic contents [59], while antifungal action is attributed to causing loss of spore contents and damage to the thick cell wall and cell membrane of the spore [58]. Although the conventional use of GSE in foods is its sole application, it is also used to fabricate edible coatings with antimicrobial properties. Carboxymethyl cellulose (CMC)-based films incorporated with chitin nanocrystals (ChNCs) and GSE displayed strong antimicrobial efficacy against both gram-negative bacteria (E. coli) and gram-positive bacteria (L. monocytogenes) Oun and Rahim [57], due to the release of polyphenol compounds (e.g., naringin, limonin), that breach the cell membrane and bind to cellular proteins, thereby impairing function [60].
Food Additives
Food additives are used for shelf-life extension, nutritional quality improvement, and appearance. Grapefruit byproducts and derivatives can be used as a natural substitute for chemical additives in various roles. Soluble dietary fiber from grapefruit peels added to blueberry jam formulation enhanced the stability of jam while maintaining the color, texture, and spreadability of jam [34]. Soluble dietary fibers (SDF) obtained by microwave-ultrasonic treatment from grapefruit peels were used for bread formation and improved the structural, functional, and in vitro digestion properties of the prepared bread. The authors also highlighted its low glucose release rate and potential as a functional food ingredient [19]. In another investigation, Ukom et al. [134] utilized grapefruit peel powder (3.7–5 g) in cake preparation that improved the DPPH, ABTS, and FRAP percentages up to 2–threefold over the control samples. Grapefruit peel nanofibrillated cellulose (GNFC) has been used as a fat substitute in the preparation of ice cream with lower gross energy and calorie content. Furthermore, GNFC incorporation demonstrated digestion impediments of 21.70% and 59.53% for protein and fat, respectively [116]. Kaanin-Boudraa et al. [107] used grapefruit EO as an alternative to vitamin E in margarine and found that it was more resistant to oxidation than control samples. Functional foods can be considered to be those fortified, enriched, or enhanced foods that are destined to provide additional health benefits. Ajtonty et al. [48] prepared functional chocolate fortified with grapefruit peel extract and confirmed it as a satisfactory carrier for naringin. Qin et al. [118] improved SDF quality from grapefruit peel by using superfine grinding combined with L. paracasei fermentation and used SDF to prepare functional yogurt, which exhibited lower syneresis, higher gel strength and hardness, and stronger odor characteristics compared to control yogurt. Moreover, functional drinks enriched with grapefruit peel and pomace showed improved phytochemical profile, which therefore could be promoted as a nutraceutical product with multiple benefits to the consumers [16].
Prebiotics
Prebiotics can help to nourish gut bacteria and eventually promote health-promoting bacteria that colonize the gastrointestinal tract, healthy digestive and therefore, immune systems [135]. Grapefruit albedo and flavedo peel flour evaluated with two lactic acid bacteria strains (P. pentosaceus UAM21 and A. viridans UAM22) showed the viability of the employed strains during the fermentation period employing the alternative carbon sources. Short-chain fatty acid production confirmed the prebiotic potential of grapefruit peel flours authenticating their potential as a functional ingredient in foods [120]. GSLSDF-1 obtained from the grapefruit peel sponge layer soluble dietary fiber (GSLSDF) possessed a low molecular weight and crystallinity, a loose and porous microstructure, and a high glucose content. GSLSDF-1 showed a better prebiotic activity, increasing the relative abundances of Lactobacillus, Bacteroides, Bifidobacterium, and Faecalibacterium. Furthermore, GSLSDF-1 promoted the production of short-chain fatty acids (SCFAs) by modulating the SCFAs synthesis pathway of intestinal microorganisms, while the NH3-N synthesis of intestinal microorganisms was inhibited by GSLSDF-1 [5]. Qin et al. [30] reported that ultrafine ground SDF from grapefruit peel more effectively promoted bacteria proliferation and stimulated probiotic strains to produce more short-chain fatty acids, compared to untreated SDF when tested for in vitro prebiotic activity. These investigations confirm grapefruit peel is an apt and sustainable contender for developing products with prebiotic properties.
Encapsulating Agents
Encapsulation is an apt tailored option to improve the stability, bioavailability, quality, safety, and applicability of the bioactive compounds [136]. Encapsulation finds wide applications in the food and pharma sector, conferring micro (1–1000 μm) and nano (1- several hundred nm) levels of fabrications. Calvo and Santagapita [122] encapsulated grapefruit lycopene in alginate-based beads to improve its stability and shelf life. Alginate beads comprising trehalose with β-cyclodextrin retained a higher lycopene content (> 80%) after freezing and drying. In a separate study, Ko et al. [137] extracted flavanones (naringin, narirutin, naringenin, hesperidin, and hesperetin) from grapefruit peels, treated the extracts with 60% β-cyclodextrin and analyzed the extracts using field emission-scanning electron microscopy (FE-SEM). The results showed that encapsulation in β-cyclodextrin improved the solubilization. Nishad et al. [77] encapsulated grapefruit peel phenolics (GPP) into the nano-emulsion-based delivery system and reported extended oxidative stability of mustard oil.
Cosmetic Industry
The cosmetic industry is shifting towards safer, natural compounds due to health concerns over harmful chemical constituents. Consumers are demanding natural, organic, and certified organic ingredients, therefore high-value compounds from grapefruit waste can be explored in cosmetics and toiletries [138, 139] (Table 3).
Flavonoids present in grapefruit peels could preferably be used in cosmetics due to reported anti-inflammatory activity, as they prevent the release of arachidonic acid caused by oxidative processes of membrane lipids [146]. Naringin, hesperidin, and isonaringin are important flavonoids present in grapefruit peels and seed residues [39] that possess anti-carcinogenic, anti-oxidative, anti-aging, antimicrobial, anti-inflammatory, and free radical scavenging activity [147]. It has been reported that naringin scavenges free radicals in vitro [142] and, therefore could be used to develop skin creams and topical lotions. Phenolic acid i.e., such as resveratrol present in grapefruit byproducts is known to safeguard against photo-oxidative damage to the skin [42], while gallic acid, which is a major phenolic acid in grapefruit different parts, followed by chlorogenic acid, caffeic acid, and ferulic acid are often associated with preservation of hair color, strength, and growth. Limonene is a predominant component (93.33%) of monoterpenes present in light-phase grapefruit EO [49], therefore can be explored against acne due to its antibacterial properties. Cosmetic composition developed with natural extracts from rosemary and grapefruit seeds and pulp showed no bacterial colonies neither after one day nor after three months of storage period, divulging good antibacterial properties, being quite a satisfactory substitute for the parabens [148]. Grapefruit peel ethanolic extract with a concentration of 2% has the best anti-aging activity attributable to significant antioxidant activity [149]. Ha et al. [150] tested the antimicrobial effects of (GSE) against human skin pathogens: Malassezia furfur, M. restricta, Propionibacterium acnes, Trichophyton mentagrophytes, and T. rubrum and reported MIC values of 3.91, 3.91, 0.004, 0.024, and 0.012 µl/ml, respectively. The study indicated GSE as a promising source of antibacterial agents that could be utilized in skin and hair care products and alternative medicine for certain skin ailments. Grapefruit EO contains limonene, myrcene, and α-pinene that could be looked into as fragrance ingredients in cosmetics [49].
Pharma and Health Sector
Grapefruit byproducts, rich in phytochemicals and value-added compounds, are being explored for nutraceutical, pharmacological effects, and drug development. They offer a lucrative, sustainable, and cost-effective source of biologically active compounds [6, 22, 42]. Pectin from grapefruit peels can be utilized as a natural prophylactic for the active elimination of toxic metals from the digestive and respiratory systems, in tablet formulations as a carrier material in colon-specific drug delivery systems. Pectin from grapefruit peels is also reported for inhibitory activity against pancreatic cholesterol esterase, pancreatic lipase, and α-glucosidase [73]. Grapefruit IntegroPectin, a potent antioxidant, has potential as a therapeutic and preventive agent for treating oxidative stress-related brain disorders and may also aid in cancer research. [151]. Dietary fibers are often associated with preventive action against constipation, elevating smooth bowel movements, helping with diabetes, and lowering cholesterol. Grapefruit pomace with an SDF/total DF ratio (w/w) of 0.76, followed by peels (0.15) [22] can be used as a reliable source of dietary fibers. Recently, Qin et al. [30] reported that ultrafine ground SDF from grapefruit peels effectively promoted bacteria proliferation and stimulated probiotic strains to produce more short-chain fatty acids. Apart from this, vitamin C is present in grapefruit peels, while vitamin E or tocopherol, a fat-soluble vitamin possibly found in seed oil can be harnessed for topical applications [44]. These byproducts can be exploited for the potential possibilities in the pharma and health sector (Table 4).
Hydroxycinnamic acids are present in grapefruit peels, which as dietary supplements are known to reduce inflammation, improve digestion, and promote cardiovascular health [43]. Naringin, abundantly present in grapefruit peels encompasses antioxidant, anticancer, and anti-osteoporosis, and serves as a facilitator for the absorption of other drugs [158]. It is also used for lipid-lowering functions and to treat obesity and diabetes [18]. Grapefruit peel flavonoids, naringin, and hesperidin have shown potential as neuroprotective agents in animal models of Parkinson's disease and neurodegenerative diseases, though clinical use is still a long way off [39].
Arsène et al. [159] highlighted the antibacterial properties of grapefruit peel that can be effectively used against antibiotic resistance and for developing new drugs for treating bacterial diseases. Bokhary et al. [160] prepared Al2O3 nanoparticles using grapefruit extract, which showed antioxidant, anti-inflammatory, and immunomodulatory potentials. Fabricated Al2O3 nanoparticles displayed a potential to curtail the production of pro-inflammatory cytokines interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α), as well as the signaling pathway of the transcription factor NF-B, in addition to lowering NO and O2 generation. Han et al. [161] reported for antibacterial activity of GSE against methicillin-sensitive Staphylococcus aureus (MSSA), methicillin-resistant S. aureus (MRSA), and vancomycin-resistant S. aureus (VRSA) in the disk and microdilution MIC tests highlighting its potential as a natural substitute to traditional antibiotics to fight multidrug-resistant pathogens. Antimicrobial properties of GSE are ascribable to involve bacterial membrane disruption and release of cytoplasmic contents in a relatively short time. Quercetin and naringenin, significant GSE flavonoids, show potent anti-inflammatory and antiviral effects via NFκB, TLR, and IL-6 signaling [59]. The presence of flavonoids in GSE is also equated with anticancer activity against various human breast cancers and is associated with the ability to inhibit platelet aggregation, thereby lowering the risk of coronary thrombosis and myocardial infarction [21]. GSE has significant gastroprotective properties against gastric lesions by preserving antioxidizing enzyme activity, reducing lipid peroxidation, enhancing gastric blood flow, and influencing plasma gastrin levels [21].
Development of Environmentally Sustainable Materials
Biosorbents
SDG’s 6th goal is associated with clean water and sanitation. Researchers have been focusing on a new process for the remediation of heavy metals, dyes, pesticides, and organic and inorganic pollutants from water [162, 163]. Numerous water-soluble and insoluble monomers and polymers are found in grapefruit peels. Glucose, fructose, sucrose, and some xylose are present in the water-soluble fraction, whereas between 50 and 70 percent of the insoluble fraction is made up of lignin, pectin, cellulose, and hemicellulose. The carboxyl and hydroxyl functional groups are abundant in these polymers [164]. Grapefruit peels can be used as biosorbents in their natural state [165] or can be amended with physical (drying, grinding, heat treatment) and/or chemical modifications (graft co-polymerization, deamination, saponification, disulfide treatment, pyrolysis, protonation), to ameliorate the potential and adsorption capacity [166]. The maximum uptakes of Cd(II) and Ni(II) by grapefruit peel are found to be 42.09 and 46.13 mg/g, respectively. The kinetics of the biosorption process are found to follow the pseudo-second-order kinetic model [164]. Grapefruit peels pretreated with H2O2 (1 M) showed high levels of uptake, qmax of 37.4270 mg/g and 39.0628 mg/g for dye mixture and Cr (VI), respectively [167]. Apart, the adsorption of ciprofloxacin pollutants (CIP) using modified waste grapefruit peel was investigated by Fu et al. [168]. Obtained results showed a maximum uptake of 1.71 mmol·g−1 under optimal experimental conditions, with the adsorption process following pseudo-second-order kinetics, and fitting the Langmuir isotherm model. The latest investigations covering the utilizing grapefruit byproducts for fabricating biosorbents have been tabulated in Table 5.
Biofertilizer
Although citrus biofertilizers have been reported to improve the quantities of native nutrients such as nitrogen (N), carbon (C), and potassium (K) in the soil which enhances the growth and development of plants [174, 175], in particular, reports on the utilization of the grapefruit peels for the same are scarce. Grapefruit peel utilization for the co-composting of poultry manure is conferred to increase the C/N ratio, and thus minimize N losses caused by increased pH [44]. Biochar materials made from grapefruit peels have the potential to be used as a soil amendment. They can help immobilize lead and copper in the surrounding soil and enhance the establishment of vegetation in the treated soil. [176].
Bioethanol
SDG’s 7th Goal contemplates affordable and clean energy. Bioethanol is a clean potential biofuel [177], and grapefruit peel represents a lucrative biomass for bioethanol production as it comprises cellulose, pectin, and hemicellulose that can be hydrolyzed by enzymes to monomer sugars, for bioethanol production [178]. In one study, glucose, fructose, galactose, arabinose, xylose, and galacturonic acid (GA) were produced using the enzymes pectinase, cellulase, and beta-glucosidase, with a pH range of 3.8–4.8 found to be optimal for sugar yields from peel hydrolysis [179]. However, D-limonene in grapefruit peels could act as a native inhibitor, which before bioethanol production, must be removed due to its antibacterial action against yeast or bacteria used in fermentation to produce ethanol [180]. Teke et al. [181] employed an ultrasound-assisted pretreatment (14.6 °C, 25.81 W/cm2) to extract D-Limonene with a validation yield of 134 ± 4.24 mg/100 g dry CPW, and reported bioethanol yield with a 66% increase. Following grapefruit peel fermentation in an immobilized cell reactor (ICR), Choi et al. [182] observed reduced ethanol concentrations and created a D-limonene removal column (LRC) that effectively eliminated this inhibitor from the fruit waste. Yeast fermentation using an LRC in conjunction with an ICR produced yields of 90.7% and ethanol concentrations of 21.6 g/L, which were twelve times higher than those obtained from ICR fermentation alone.
Conclusion and Future Directions
Grapefruit processing generates important volumes of disposals such as peels, pomace, and seeds that could be attractive raw materials for the recovery of compounds such as dietary fiber, polyphenols, flavonoids, essential oil, and pectin. These could be natural alternatives to cope with the high demand for natural compounds for the development of healthy matrices for different industries, favoring a circular economy model. This manuscript also spotlights advanced and innovative extraction techniques for processing grapefruit byproducts, allowing an efficient, easier, quick, cost-effective, and appreciable recovery of bioactive compounds that could be used in food, cosmetic, or pharma sectors for various applications. The recovery of byproducts from grapefruit can not only benefit the environment, but could commence businesses, and will keep valuable resources and materials in the economy. Future works should focus on optimizing the extraction parameters and researchers should prioritize the gap between in-vitro trials and commercial-scale applications for the sustainable valorization of grapefruit byproducts. However, a multidisciplinary approach with the collaboration of academics, engineers, economists, and policymakers, is critical to understand and pave the valorization route to a level of innovation where it is possible to achieve a broader landscape of zero waste and a sustainable society. Moreover, consonance of capital investments, policy amendments, and commercial, social, and consumer acceptance is vital for unraveling the true potential of grapefruit waste.
Data Availability
No datasets were generated or analysed during the current study.
References
Louzada ES, Ramadugu C. Grapefruit: history, use, and breeding. Horttechnology. 2021;31(3):243–58. https://doi.org/10.21273/HORTTECH04679-20.
Hung WL, Suh JH, Wang Y. Chemistry and health effects of furanocoumarins in grapefruit. J Food Drug Anal. 2017;25(1):71–83. https://doi.org/10.1016/j.jfda.2016.11.008.
Spreen TH, Gao Z, Fernandes Jr W, Zansler ML. Global economics and marketing of citrus products. In: The Genus Citrus;2020, pp.471–493. Woodhead Publishing. https://doi.org/10.1016/B978-0-12-812163-4.00023-1
Statista. 2023. https://www.statista.com/statistics/577836/world-grapefruit-production/. Accessed 7 Mar 2024.
Du X, Chen J, Hu T, Xu Y, Wu J, Peng J, Li L. Identification and structural characterization of key prebiotic fraction of soluble dietary fiber from grapefruit peel sponge layer and its regulation effect on gut microbiota. Int J Biol Macromol. 2024;259, 129274. https://doi.org/10.1016/j.ijbiomac.2024.129274
Cristóbal-Luna JM, Álvarez-González I, Madrigal-Bujaidar E, Chamorro-Cevallos G. Grapefruit and its biomedical, antigenotoxic and chemopreventive properties. Food Chem Toxicol. 2018;112:224–34. https://doi.org/10.1016/j.fct.2017.12.038.
Maqbool Z, Khalid W, Atiq HT, Koraqi H, Javaid Z, Alhag SK, Al-Shuraym LA, Bader DMD, Almarzuq M, Afifi M, Al-Farga A. Citrus waste as source of bioactive compounds: extraction and utilization in health and food industry. Molecules. 2023;28(4):1636. https://doi.org/10.3390/molecules28041636.
Esparza I, Jiménez-Moreno N, Bimbela F, Ancín-Azpilicueta C, Gandía LM. Fruit and vegetable waste management: Conventional and emerging approaches. J Environ Manage. 2020;265:110510. https://doi.org/10.1016/j.jenvman.2020.110510.
United Nations. Sustainable development goals. 2015. https://sustainabledevelopment.un.org. Accessed 7 Mar 2024.
Khajuria A, Atienza VA, Chavanich S, Henning W, Islam I, Kral U, ... & Li J. Accelerating circular economy solutions to achieve the 2030 agenda for sustainable development goals. Circular Economy. 2022;1(1), 100001. https://doi.org/10.1016/j.cec.2022.100001
Facchini F, Silvestri B, Digiesi S, Lucchese A. Agri-food loss and waste management: Win-win strategies for edible discarded fruits and vegetables sustainable reuse. Innov Food Sci Emerg Technol. 2023;83:103235. https://doi.org/10.1016/j.ifset.2022.103235.
Campos DA, Gómez-García R, Vilas-Boas AA, Madureira AR, Pintado MM. Management of fruit industrial by-products—A case study on circular economy approach. Molecules. 2020;25(2):320. https://doi.org/10.3390/molecules25020320.
Scarano P, Sciarrillo R, Tartaglia M, Zuzolo D, Guarino C. Circular economy and secondary raw materials from fruits as sustainable source for recovery and reuse. A review. Trends Food Sci Technol. 2022;122:157–70. https://doi.org/10.1016/j.tifs.2022.02.003.
Nadeem M, Asghar M. Variability in peel composition and quality evaluation of peel oils of citrus varieties. J Agric Res. 2016;54(4):747–56.
Edet U, Ebana R, Ekanemesang U, Ikon G, Edem E, Mbim E. Phytochemical screening, nutrient analysis, anti-termite and antimicrobial activity of Citrus paradis peel powder. J Appl Life Sci Int. 2016;9(4):1–9. https://doi.org/10.9734/JALSI/2016/29921.
Ali SM, Imran A, Arshad MU, Ahmed RS, Imran M. Physicochemical, antioxidant and enzymes activities of grape fruit peel and pomace enriched functional drinks. Cell Mol Biol. 2021;67(1):125–31. https://doi.org/10.14715/cmb/2021.67.1.19.
Sir Elkhatim KA, Elagib RA, Hassan AB. Content of phenolic compounds and vitamin C and antioxidant activity in wasted parts of Sudanese citrus fruits. Food Sci Nutr. 2018;6(5):1214–9. https://doi.org/10.1002/fsn3.660.
El Kantar S, Rajha HN, Boussetta N, Vorobiev E, Maroun RG, Louka N. Green extraction of polyphenols from grapefruit peels using high voltage electrical discharges, deep eutectic solvents and aqueous glycerol. Food Chem. 2019;295:165–71. https://doi.org/10.1016/j.foodchem.2019.05.111.
Gan J, Huang Z, Yu Q, Peng G, Chen Y, Xie J, ... & Xie M. Microwave assisted extraction with three modifications on structural and functional properties of soluble dietary fibers from grapefruit peel. Food Hydrocolloids. 2020;101, 105549. https://doi.org/10.1016/j.foodhyd.2019.105549
Peng G, Gan J, Dong R, Chen Y, Xie J, Huang Z, ... & Yu Q. Combined microwave and enzymatic treatment improve the release of insoluble bound phenolic compounds from the grapefruit peel insoluble dietary fiber. Lwt. 2021;149:111905. https://doi.org/10.1016/j.lwt.2021.111905
Drozdowicz D, Brzozowski T, Pajdo R, Kwiecien S, Pawlik M, Konturek PC, ... & Pawlik WW. Gastroprotective Activity of Grapefruit (Citrus paridisi) Seed Extract Against Acute Gastric Lesions. In: Nuts and Seeds in Health and Disease Prevention; 2011, pp. 553–560. Academic Press. https://doi.org/10.1016/B978-0-12-375688-6.10066-0
Khalil MN, Farghal HH, Farag MA. Outgoing and potential trends of composition, health benefits, juice production and waste management of the multi-faceted Grapefruit Citrus Χ paradisi: A comprehensive review for maximizing its value. Critical Rev Food Sci Nutr. 2022;62(4):935–956. https://doi.org/10.1080/10408398.2020.1830364This paper reviews the grapefruit and its byproducts, explores novel aspects, and presents the full potential of this valued multi-faceted citrus fruit.
Roy S, Zhang W, Biswas D, Ramakrishnan R, Rhim JW. Grapefruit seed extract-added functional films and coating for active packaging applications: A review. Molecules. 2023;28(2):730. https://doi.org/10.3390/molecules28020730.
Yilmaz E, Aydeniz Guneser B, Ok S. Valorization of grapefruit seeds: cold press oil production. Waste and Biomass Valorization. 2019;10:2713–24. https://doi.org/10.1007/s12649-018-0286-x.
Anwar F, Naseer R, Bhanger MI, Ashraf S, Talpur FN, Aladedunye FA. Physico-chemical characteristics of citrus seeds and seed oils from Pakistan. J Am Oil Chem Soc. 2008;85(4):321–30. https://doi.org/10.1007/s11746-008-1204-3.
Chen L, Jiang X, Sun Y, Gan D, Liu W, Wu Y, Hao X. Composite optimization and characterization of dietary fiber-based edible packaging film reinforced by nanocellulose from grapefruit peel pomace. Int J Biol Macromol. 2023;253:127655. https://doi.org/10.1016/j.ijbiomac.2023.127655.
Asif M, Khan MKI, Khan MI, Maan AA, Helmick H, Kokini JL. Effects of citrus pomace on mechanical, sensory, phenolic, antioxidant, and gastrointestinal index properties of corn extrudates. Food Biosci. 2023;55:103012. https://doi.org/10.1016/j.fbio.2023.103012.
Kohajdová Z, Karovičová J, Jurasová M. Influence of grapefruit dietary fibre rich powder on the rheological characteristics of wheat flour dough and on biscuit quality. Acta Aliment. 2013;42(1):91–101. https://doi.org/10.1556/aalim.42.2013.1.9.
Zhang L, Cui J, Zhao S, Liu D, Zhao C, Zheng J. The structure-function relationships of pectins separated from three citrus parts: Flavedo, albedo, and pomace. Food Hydrocolloids. 2023;136: 108308. https://doi.org/10.1016/j.foodhyd.2022.108308.
Qin X, Dong X, Tang J, Chen Y, Xie J, Cheng Y, ... & Yu Q. Comparative analysis of dietary fibers from grapefruit peel prepared by ultrafine grinding: Structural properties, adsorption capacities, in vitro prebiotic activities. Food Bioscience. 2023;56:103163. https://doi.org/10.1016/j.fbio.2023.103163
Akşit Z, Gençcelep H. Investigation of tomato, quince and grapefruit waste; compositions and functional properties. Hittite J Sci Eng. 2021;8(1):19–26. https://doi.org/10.17350/HJSE19030000208.
Deng M, Lin Y, Dong L, Jia X, Shen Y, Liu L, ... & Zhang R. Physicochemical and functional properties of dietary fiber from pummelo (Citrus grandis L. Osbeck) and grapefruit (Citrus paradisi Mcfad) cultivars. Food Biosci. 2021;40:100890. https://doi.org/10.1016/j.fbio.2021.100890
Wang L, Xu H, Yuan F, Pan Q, Fan R, Gao Y. Physicochemical characterization of five types of citrus dietary fibers. Biocatal Agric Biotechnol. 2015;4(2):250–8. https://doi.org/10.1016/j.bcab.2015.02.003.
Xie J, Peng G, Hu X, Xie J, Chen Y, Dong R, ... & Yu Q. Physicochemical Characteristics of Soluble Dietary Fiber Obtained from Grapefruit Peel Insoluble Dietary Fiber and Its Effects on Blueberry Jam. Foods. 2022;11(22);3735. 10.3390/foods11223735
Wang W, Ma X, Jiang P, Hu L, Zhi Z, Chen J, ... & Liu D. Characterization of pectin from grapefruit peel: a comparison of ultrasound-assisted and conventional heating extractions. Food Hydrocolloids. 2016;61:730–739. https://doi.org/10.1016/j.foodhyd.2016.06.019
Lin X, Liu Y, Wang R, Dai J, Wang L, Zhang J. Extraction of pectins from renewable grapefruit (Citrus paradisi) peels using deep eutectic solvents and analysis of their structural and physicochemical properties. Int J Biol Macromol. 2024;254:127785. https://doi.org/10.1016/j.ijbiomac.2023.127785.
Öztürk T, Özbek HN, Koçak Yanık D. Environmentally Friendly Approach to Pectin Extraction from Grapefruit Peel: Microwave-Assisted High-Pressure CO2/H2O. Foods. 2024;13(3):476. https://doi.org/10.3390/foods13030476.
Hu W, Chen S, Wu D, Zhu K, Ye X. Manosonication assisted extraction and characterization of pectin from different citrus peel wastes. Food Hydrocolloids. 2021;121:106952. https://doi.org/10.1016/j.foodhyd.2021.106952.
Castro-Vazquez L, Alañón ME, Rodríguez-Robledo V, Pérez-Coello MS, Hermosín-Gutierrez I, Díaz-Maroto MC, ... & Arroyo-Jimenez MDM. Bioactive flavonoids, antioxidant behaviour, and cytoprotective effects of dried grapefruit peels (Citrus paradisi Macf.). Oxid Med Cell Longev. 2016. https://doi.org/10.1155/2016/8915729
Zarate-Vilet N, Wisniewski C, Gué E, Delalonde M. Towards a better identification of naringin and narirutin dispersion state in grapefruit peel press liquor. Chem Eng Res Des. 2020;159:205–14. https://doi.org/10.1016/j.cherd.2020.03.023.
Zarate-Vilet N, Gué E, Servent A, Delalonde M, Wisniewski C. Filtration-compression step as downstream process for flavonoids extraction from citrus peels: Performances and flavonoids dispersion state in the filtrate. Food Bioprod Process. 2020;120:104–13. https://doi.org/10.1016/j.fbp.2020.01.001.
Ademosun AO, Oboh G, Passamonti S, Tramer F, Ziberna L, Boligon AA, Athayde ML. Phenolics from grapefruit peels inhibit HMG-CoA reductase and angiotensin-I converting enzyme and show antioxidative properties in endothelial EA. Hy 926 cells. Food Sci Human Wellness. 2015;4(2):80–5. https://doi.org/10.1016/j.fshw.2015.05.002.
Singh B, Singh JP, Kaur A, Singh N. Phenolic composition, antioxidant potential and health benefits of citrus peel. Food Res Int. 2020;132:109114. https://doi.org/10.1016/j.foodres.2020.109114.
Zarate-Vilet N, Gué E, Delalonde M, Wisniewski C. Valorization of Grapefruit (Citrus× paradisi) Processing Wastes. In: Mediterranean Fruits Bio-wastes: Chemistry, Functionality and Technological Applicationspp, 2022;179–220. Cham: Springer International Publishing. https://doi.org/10.1007/978-3-030-84436-3_8
Wang Z, Shang Q, Wang W, Feng X. Microwave-assisted extraction and liquid chromatography/mass spectrometry analysis of flavonoids from grapefruit peel. J Food Process Eng. 2011;34(3):844–59. https://doi.org/10.1111/j.1745-4530.2009.00513.x.
Garcia-Castello EM, Rodriguez-Lopez AD, Mayor L, Ballesteros R, Conidi C, Cassano A. Optimization of conventional and ultrasound assisted extraction of flavonoids from grapefruit (Citrus paradisi L.) solid wastes. LWT-Food Sci Technol. 2015;64(2):1114–22. https://doi.org/10.1016/j.lwt.2015.07.024.
Nishad J, Saha S, Dubey AK, Varghese E, Kaur C. Optimization and comparison of non-conventional extraction technologies for Citrus paradisi L. peels: a valorization approach. J Food Sci Technol. 2019;56:1221–33. https://doi.org/10.1007/s13197-019-03585-0.
Ajtony Z, Sik B, Csuti A. Examining the Naringin Content and Sensory Characteristics of Functional Chocolate Fortified with Grapefruit Peel Extract. Plant Foods Hum Nutr. 2023;78(3):533–8. https://doi.org/10.1007/s11130-023-01091-5.
Deng W, Liu K, Cao S, Sun J, Zhong B, Chun J. Chemical composition, antimicrobial, antioxidant, and antiproliferative properties of grapefruit essential oil prepared by molecular distillation. Molecules. 2020;25(1):217. https://doi.org/10.3390/molecules25010217.
Tran TH, Dao TP, Le XT, Huynh BL, Minh LTN. Volatile compounds of grapefruit (Citrus Grandis (L.) Osbeck) peel essential oil by cold pressing and hydrodistillation methods. In: IOP Conference Series: Earth and Environmental Science, 2023: (Vol. 1241, No. 1, p. 012070). IOP Publishing. https://doi.org/10.1088/1755-1315/1241/1/012070
Özogul Y, Özogul F, Kulawik P. The antimicrobial effect of grapefruit peel essential oil and its nanoemulsion on fish spoilage bacteria and food-borne pathogens. LWT. 2021;136: 110362. https://doi.org/10.1016/j.lwt.2020.110362.
Denkova-Kostova R, Teneva D, Tomova T, Goranov B, Denkova Z, Shopska V, ... & Hristova-Ivanova Y. Chemical composition, antioxidant and antimicrobial activity of essential oils from tangerine (Citrus reticulata L), grapefruit (Citrus paradisi L.), lemon (Citrus lemon L.) and cinnamon (Cinnamomum zeylanicum Blume). Zeitschrift für Naturforschung C. 2021;76(5–6):175–185. https://doi.org/10.1515/znc-2020-0126
El Houda AKN, Ali B, Ahmed A, Salah O, Yazid FC. Chemical composition, antimicrobial and insecticidal activities of Citrus paradisi peel essential oil from Algeria. Journal of microbiology, biotechnology and food sciences. 2020;9(6):1093–8. https://doi.org/10.15414/jmbfs.2020.9.6.1093-1098.
Li C, Pei J, Xiong X, Xue F. Encapsulation of grapefruit essential oil in emulsion-based edible film prepared by plum (pruni domesticae semen) seed protein isolate and gum acacia conjugates. Coatings. 2020;10(8):784. https://doi.org/10.3390/coatings10080784.
Dudeja I, Tandon R, Kalia A. Grapefruit peel essential oil nanoemulsions: preparation, characterisation, stability and antifungal potential against Penicillium digitatum of Kinnow. Int J Food Sci Technol. 2023;58(5):2790–9. https://doi.org/10.1111/ijfs.16311.
Gupta S, Saning A, Laodheerasiri S, Chuenchom L, Watcharin W. Nanoencapsulated grapefruit essential oil with carrageenan: Facile preparation, characterization and cytotoxicity studies. In: AIP Conference Proceedings, 2023;(Vol. 2720, No. 1). AIP Publishing. https://doi.org/10.1063/5.0136867
Oun AA, Rhim JW. Preparation of multifunctional carboxymethyl cellulose-based films incorporated with chitin nanocrystal and grapefruit seed extract. Int J Biol Macromol. 2020;152:1038–46. https://doi.org/10.1016/j.ijbiomac.2019.10.191.
Kim T, Kim JH, Oh SW. Grapefruit seed extract as a natural food antimicrobial: A review. Food Bioprocess Technol. 2021;14(4):626–33. https://doi.org/10.1007/s11947-021-02610-5.
Seo DS, Lee JS, Shin YC, Jang Y. Multipotent Antiviral Effects of Deacetylated Chitosan Supplemented with Grapefruit Seed Extract against Influenza and Parainfluenza Viruses. Appl Sci. 2023;13(17):9938. https://doi.org/10.3390/app13179938.
Roy S, Rhim JW. Antioxidant and antimicrobial poly (vinyl alcohol)-based films incorporated with grapefruit seed extract and curcumin. J Environ Chem Eng. 2021;9(1):104694. https://doi.org/10.1016/j.jece.2020.104694.
Roy S, Rhim JW. Fabrication of pectin/agar blended functional film: Effect of reinforcement of melanin nanoparticles and grapefruit seed extract. Food Hydrocolloids. 2021;118:106823. https://doi.org/10.1016/j.foodhyd.2021.106823.
Koneru A, Dharmalingam K, Anandalakshmi R. Cellulose based nanocomposite hydrogel films consisting of sodium carboxymethylcellulose–grapefruit seed extract nanoparticles for potential wound healing applications. Int J Biol Macromol. 2020;148:833–42. https://doi.org/10.1016/j.ijbiomac.2020.01.018.
Choi YS, Lee JS, Lee HG. Nanoencapsulation of Grapefruit Seed Extract and Cinnamon Oil for Oral Health: Preparation, In Vitro, and Clinical Antimicrobial Activities. J Agric Food Chem. 2023;71(14):5646–54. https://doi.org/10.1021/acs.jafc.2c05518.
Osungunna MO, Onawunmi GO. Antimicrobial Activity of Grapefruit (Citrusparadisi) Seed Extract and Juice Against Bacteria Associated with Urinary Tract Infections. Univ Lagos J Basic Med Sci. 2022;3(6). https://www.jbms.unilag.ng/index.php/jbms/article/view/173/156.
Kang S, Guo Y, Rao J, Jin H, You HJ, Ji GE. In vitro and in vivo inhibition of Helicobacter pylori by Lactobacillus plantarum pH3A, monolaurin, and grapefruit seed extract. Food Funct. 2021;12(21):11024–32. https://doi.org/10.1039/D1FO01480C.
Hsouna AB, Sadaka C, Generalić Mekinić I, Garzoli S, Švarc-Gajić J, Rodrigues F, ... & Mnif W. The chemical variability, nutraceutical value, and food-industry and cosmetic applications of citrus plants: A critical Review. Antioxidants. 2023;12(2):481. https://doi.org/10.3390/antiox12020481.
Czech A, Zarycka E, Yanovych D, Zasadna Z, Grzegorczyk I, Kłys S. Mineral content of the pulp and peel of various citrus fruit cultivars. Biol Trace Elem Res. 2020;193:555–63. https://doi.org/10.1007/s12011-019-01727-1.
Saleem M, Durani AI, Alharthy RD, Ahmed M, Shafiq MI, Hameed A, ... Bashir W. Ultrasound-assisted extraction of micro-and macroelements in fruit peel powder mineral supplement for osteoporosis patients and their determination by flame atomic absorption spectrometry. J Chem. 2021, 1–10. https://doi.org/10.1155/2021/5151560.
Belwal T, Chemat F, Venskutonis PR, Cravotto G, Jaiswal DK, Bhatt ID, Devkota HP, Luo Z. Recent advances in scaling-up of non-conventional extraction techniques: Learning from successes and failures. TrAC, Trends Anal Chem. 2020;127:115895. https://doi.org/10.1016/j.trac.2020.115895.
Chemat F, Abert-Vian M, Fabiano-Tixier AS, Strube J, Uhlenbrock L, Gunjevic V, Cravotto G. Green extraction of natural products. Origins, current status, and future challenges. TrAC, Trends Anal Chem. 2019;118:248–63. https://doi.org/10.1016/j.trac.2019.05.037.
Dueñas M, García-Estévez I. Agricultural and food waste: Analysis, characterization and extraction of bioactive compounds and their possible utilization. Foods. 2020;9(6):817. https://doi.org/10.3390/foods9060817.
Brito TBN, Ferreira ML, Fai AE. Utilization of agricultural by-products: Bioactive properties and technological applications. Food Rev Intl. 2020;38:1305–29. https://doi.org/10.1080/87559129.2020.1804930.
Picot-Allain MCN, Amiri-Rigi A, Abdoun-Ouallouche K, Aberkane L, Djefal-Kerrar A, Mahomoodally MF, Emmambux MN. Assessing the bioactivity, cytotoxicity, and rheological properties of pectin recovered from citrus peels. Food Biosci. 2022;46:101550. https://doi.org/10.1016/j.fbio.2022.101550.
Kumar M, Dahuja A, Tiwari S, Punia S, Tak Y, Amarowicz R, Bhoite AG, Singh S, Joshi S, Panesar PS, Saini RP, Pihlanto A, Tomar M, Sharifi-Rad J, Kaur C. Recent trends in extraction of plant bioactives using green technologies: A review. Food Chem. 2021;353:129431. https://doi.org/10.1016/j.foodchem.2021.129431.
Islam M, Malakar S, Dwivedi U, Kumar N, Prabakar PK, Kishore A, Kumar A. Impact of different drying techniques on grapefruit peels and subsequent optimization of ultrasonic extraction conditions for bioactive compounds. J Food Proc Eng. 2023;e14331. https://doi.org/10.1111/jfpe.14331
Stabrauskiene J, Marksa M, Ivanauskas L, Bernatoniene J. Optimization of naringin and naringenin extraction from Citrus× paradisi L. using hydrolysis and excipients as adsorbent. Pharmaceutics. 2022;14(5):890. https://doi.org/10.3390/pharmaceutics14050890.
Nishad J, Dutta A, Saha S, Rudra SG, Varghese E, Sharma RR, ... Kaur C. Ultrasound-assisted development of stable grapefruit peel polyphenolic nano-emulsion: Optimization and application in improving oxidative stability of mustard oil. Food Chem. 2021;334:127561. https://doi.org/10.1016/j.foodchem.2020.127561
Binghe Z, Chuxin W, Zeming Z, Yangcang X. Study on the extraction process of pectin from grapefruit peel based on steam explosion technology. In: IOP Conference Series: Earth and Environmental Science. 2021;(Vol. 792, No. 1, p. 012008). IOP Publishing. https://doi.org/10.1088/1755-1315/792/1/012008
Taşan NT, Akpınar Ö. Microwave Assisted Extraction of Pectin from Grapefruit Peel and Optimization of Extraction Conditions. Turkish J Agriculture-Food Sci Technol. 2020;8(7):1528–35. https://doi.org/10.24925/turjaf.v8i7.1528-1535.3401.
Wang W, Wu X, Chantapakul T, Wang D, Zhang S, Ma X, Ding T, Ye X, Liu D. Acoustic cavitation assisted extraction of pectin from waste grapefruit peels: A green two-stage approach and its general mechanism. Food Res Int. 2017;102:101–10. https://doi.org/10.1016/j.foodres.2017.09.087.
Wang W, Ma X, Xu Y, Cao Y, Jiang Z, Ding T, Ye X, Liu D. Ultrasound-assisted heating extraction of pectin from grapefruit peel: Optimization and comparison with the conventional method. Food Chem. 2015;178:106–14. https://doi.org/10.1016/j.foodchem.2015.01.080.
Jin Y, Yang N. Array-induced voltages assisted extraction of pectin from grapefruit (Citrus paradisi Macf.) peel and its characterization. Int J Biol Macromol. 2020;152:1205–12. https://doi.org/10.1016/j.ijbiomac.2019.10.215.
Trujillo-Juárez LG, Hernández-Meléndez Ó, Gimeno M, Gracia-Fadrique J, Bárzana E. Extraction of essential oil from waste grapefruit peel using a pilot-scale twin-screw extruder. ACS Food Science & Technology. 2021;1(7):1198–205. https://doi.org/10.1021/acsfoodscitech.1c00098.
Uysal B, Sozmen F, Aktas O, Oksal BS, Kose EO. Essential oil composition and antibacterial activity of the grapefruit (Citrus Paradisi. L) peel essential oils obtained by solvent-free microwave extraction: comparison with hydrodistillation. Int J Food Sci Technol. 2011;46(7):1455–61. https://doi.org/10.1111/j.1365-2621.2011.02640.x.
Karaman E, Yılmaz E, Tuncel NB. Physicochemical, microstructural and functional characterization of dietary fibers extracted from lemon, orange and grapefruit seeds press meals. Bioactive carbohydrates and dietary fibre. 2017;11:9–17. https://doi.org/10.1016/j.bcdf.2017.06.001.
Kumar K, Srivastav S, Sharanagat VS. Ultrasound assisted extraction (UAE) of bioactive compounds from fruit and vegetable processing by-products: A review. Ultrason Sonochem. 2021;70:105325. https://doi.org/10.1016/j.ultsonch.2020.105325.
Khadhraoui B, Ummat V, Tiwari BK, Fabiano-Tixier AS, Chemat F. Review of ultrasound combinations with hybrid and innovative techniques for extraction and processing of food and natural products. Ultrason Sonochem. 2021;76:105625. https://doi.org/10.1016/j.ultsonch.2021.105625.
Ekezie FGC, Sun DW, Cheng JH. Acceleration of microwave-assisted extraction processes of food components by integrating technologies and applying emerging solvents: A review of latest developments. Trends Food Sci Technol. 2017;67:160–72. https://doi.org/10.1016/j.tifs.2017.06.006.
Gomez L, Tiwari B, Garcia-Vaquero M. Emerging extraction techniques: Microwave-assisted extraction. In: Sustainable seaweed technologies, 2020.pp. 207–224. Elsevier. https://doi.org/10.1016/B978-0-12-817943-7.00008-1
Alvi T, Asif Z, Khan MKI. Clean label extraction of bioactive compounds from food waste through microwave-assisted extraction technique-A review. Food Biosci. 2022;46:101580. https://doi.org/10.1016/j.fbio.2022.101580.
Ray A, Dubey KK, Marathe SJ, Singhal R. Supercritical fluid extraction of bioactives from fruit waste and its therapeutic potential. Food Biosci. 2023;52:102418. https://doi.org/10.1016/j.fbio.2023.102418.
Priyadarsani S, Patel AS, Kar A, Dash S. Process optimization for the supercritical carbondioxide extraction of lycopene from ripe grapefruit (Citrus paradisi) endocarp. Sci Rep. 2021;11(1):10273. https://doi.org/10.1038/s41598-021-89772-6.
Yaldiz B, Saglam-Metiner P, Cakmak B, Kaya E, Deliogullari B, Yesil-Celiktas O. Essential Oil and Supercritical Carbon Dioxide Extract of Grapefruit Peels Formulated for Candida albicans Infections: Evaluation by an in Vitro Model to Study Fungal-Host Interactions. ACS Omega. 2022;7(42):37427–35. https://doi.org/10.1021/acsomega.2c04189.
Dhakane-Lad J, Kar A. Supercritical CO2 extraction of lycopene from pink grapefruit (Citrus paradise Macfad) and its degradation studies during storage. Food Chem. 2021;361:130113. https://doi.org/10.1016/j.foodchem.2021.130113.
Nadar SS, Rao P, Rathod VK. Enzyme assisted extraction of biomolecules as an approach to novel extraction technology: a review. Food Res Int. 2018;108:309–30. https://doi.org/10.1016/j.foodres.2018.03.006.
Gligor O, Mocan A, Moldovan C, Locatelli M, Crișan G, Ferreira IC. Enzyme-assisted extractions of polyphenols–A comprehensive review. Trends Food Sci Technol. 2019;88:302–15. https://doi.org/10.1016/j.tifs.2019.03.029.
Multari S, Licciardello C, Caruso M, Anesi A, Martens S. Flavedo and albedo of five citrus fruits from Southern Italy: physicochemical characteristics and enzyme-assisted extraction of phenolic compounds. J Food Measurement Characterization. 2021;15:1754–62. https://doi.org/10.1007/s11694-020-00787-5.
Mišan A, Nađpal J, Stupar A, Pojić M, Mandić A, Verpoorte R, Choi YH. The perspectives of natural deep eutectic solvents in agri-food sector. Crit Rev Food Sci Nutr. 2020;60(15):2564–92. https://doi.org/10.1080/10408398.2019.1650717.
Ozturk B, Parkinson C, Gonzalez-Miquel M. Extraction of polyphenolic antioxidants from orange peel waste using deep eutectic solvents. Sep Purif Technol. 2018;206:1–13. https://doi.org/10.1016/j.seppur.2018.05.052.
Wen L, Zhang Z, Sun DW, Sivagnanam SP, Tiwari BK. Combination of emerging technologies for the extraction of bioactive compounds. Crit Rev Food Sci Nutr. 2020;60(11):1826–41. https://doi.org/10.1080/10408398.2019.1602823.
Mahato N, Sinha M, Sharma K, Koteswararao R, Cho MH. Modern extraction and purification techniques for obtaining high purity food-grade bioactive compounds and value-added co-products from citrus wastes. Foods. 2019;8(11):523. https://doi.org/10.3390/foods8110523.
Nayak A, Bhushan B. An overview of the recent trends on the waste valorization techniques for food wastes. J Environ Manag. 2019:233:352–370. https://doi.org/10.1016/j.jenvman.2018.12.041This paper presents new vistas for valorization of food wastes and byproducts.
Chen X, Sheng Y, Che J, Reymick OO, Tao N. Integration of covalent organic frameworks and molecularly imprinted polymers for selective extraction of flavonoid naringenin from grapefruit (Citrus× paradisi Macf) peels. Food Chem X. 2024;21:101107. https://doi.org/10.1016/j.fochx.2023.101107.
Scurria A, Sciortino M, Albanese L, Nuzzo D, Zabini F, Meneguzzo F, ... Ciriminna R. Flavonoids in lemon and grapefruit IntegroPectin. ChemistryOpen. 2021;10(10):1055–1058. https://doi.org/10.1002/open.202100223
Hassan BA, Hamed FM, Abdulridha MM, Baqer FM. Phytoconstituents Screened of Grapefruit Peels with Antimicrobial Properties of Naringin and Naringenin Extracted and Isolated from its. Ann Romanian Soc Cell Biol. 2022;26(01):1160–1168. http://annalsofrscb.ro/index.php/journal/article/view/10962. Accessed 14 Feb 2024.
Sharif M, Ansari F, Hassan NU, Sultana K, Ali Q. Explore the antiproliferative phytocompounds from ethanolic extracts of Citrus paradisi against liver cancer cell line by chemical analysis using TLC and FT-IR spectroscopy. Brazilian J Biol. 2022;82. https://doi.org/10.1590/1519-6984.256856
Kaanin-Boudraa G, Brahmi F, Wrona M, Nerín C, Moudache M, Mouhoubi K, ... Boulekbache-Makhlouf L. Response surface methodology and UPLC-QTOF-MSE analysis of phenolic compounds from grapefruit (Citrus✕ paradisi) by-products as novel ingredients for new antioxidant packaging. Lwt. 2021;151:112158. https://doi.org/10.1016/j.lwt.2021.112158
Viscusi G, Adami R, Gorrasi G. Fabrication of rice flour films reinforced with hemp hurd and loaded with grapefruit seed oil: A simple way to valorize agro-waste resources toward low cost materials with added value. Ind Crops Prod. 2021;170:113785. https://doi.org/10.1016/j.indcrop.2021.113785.
Xue F, Gu Y, Wang Y, Li C, Adhikari B. Encapsulation of essential oil in emulsion based edible films prepared by soy protein isolate-gum acacia conjugates. Food Hydrocolloids. 2019;96:178–89. https://doi.org/10.1016/j.foodhyd.2019.05.014.
Jha P. Effect of grapefruit seed extract ratios on functional properties of corn starch-chitosan bionanocomposite films for active packaging. Int J Biol Macromol. 2020;163:1546–56. https://doi.org/10.1016/j.ijbiomac.2020.07.251.
Bof MJ, Jiménez A, Locaso DE, Garcia MA, Chiralt A. Grapefruit seed extract and lemon essential oil as active agents in corn starch–chitosan blend films. Food Bioprocess Technol. 2016;9(12):2033–45. https://doi.org/10.1007/s11947-016-1789-8.
Ramakrishnan SR, Jo Y, Nam HA, Gu SY, Baek ME, Kwon JH. Implications of low-dose e-beam irradiation as a phytosanitary treatment on physicochemical and sensory qualities of grapefruit and lemons during postharvest cold storage. Sci Hortic. 2019;245:1–6. https://doi.org/10.1016/j.scienta.2018.09.058.
Abdel-Naeem HH, Elshebrawy HA, Imre K, Morar A, Herman V, Pașcalău R, Sallam KI. Antioxidant and antibacterial effect of fruit peel powders in chicken patties. Foods. 2022;11(3):301. https://doi.org/10.3390/foods11030301.
Haskaraca G, Juneja VK, Mukhopadhyay S, Kolsarici N. The effects of grapefruit seed extract on the thermal inactivation of Listeria monocytogenes in sous-vide processed döner kebabs. Food Control. 2019;95:71–6. https://doi.org/10.1016/j.foodcont.2018.07.006.
Kim JH, Kwon KH, Oh SW. Effects of malic acid or/and grapefruit seed extract for the inactivation of common food pathogens on fresh-cut lettuce. Food Sci Biotechnol. 2016;25(6):1801–4. https://doi.org/10.1007/s10068-016-0274-5.
Yu B, Zeng X, Wang L, Regenstein JM. Preparation of nanofibrillated cellulose from grapefruit peel and its application as fat substitute in ice cream. Carbohyd Polym. 2021;254:117415. https://doi.org/10.1016/j.carbpol.2020.117415.
Sayari N, Sila A, Balti R, Abid E, Hajlaoui K, Nasri M, Bougatef A. Antioxidant and antibacterial properties of Citrus paradisi barks extracts during turkey sausage formulation and storage. Biocatal Agric Biotechnol. 2015;4(4):616–23. https://doi.org/10.1016/j.bcab.2015.10.004.
Qin X, Yang C, Si J, Chen Y, Xie J, Tang J, ... Yu Q. Fortified yogurt with high-quality dietary fiber prepared from the by-products of grapefruit by superfine grinding combined with fermentation treatment. Lwt. 20233b;188:115396. https://doi.org/10.1016/j.lwt.2023.115396
Khan AA, Butt MS, Randhawa MA, Karim R, Sultan MT, Ahmed W. Extraction and characterization of pectin from grapefruit (Duncan cultivar) and its utilization as gelling agent. Int Food Res J. 2014;21(6):2195–9.
Parra-Matadamas A, Mayorga-Reyes L, Pérez-Chabela M. In vitro fermentation of agroindustrial by-products: grapefruit albedo and peel, cactus pear peel and pineapple peel by lactic acid bacteria. Int Food Res J. 2015;22(2):859.
Cava EL, Gerbino E, Sgroppo SC, Gómez-Zavaglia A. Pectin hydrolysates from different cultivars of pink/red and white grapefruits (Citrus Paradisi [macf.]) as culture and encapsulating media for lactobacillus plantarum. J Food Sci. 2019;84(7):1776–83. https://doi.org/10.1111/1750-3841.14671.
Calvo TRA, Santagapita PR. Pink grapefruit lycopene encapsulated in alginate-based beads: stability towards freezing and drying. Int J Food Sci Technol. 2019;54(2):368–75. https://doi.org/10.1111/ijfs.13946.
Sid S, Mor RS, Kishore A, Sharanagat VS. Bio-sourced polymers as alternatives to conventional food packaging materials: a review. Trends Food Sci Technol. 2021;115:87–104. https://doi.org/10.1016/j.tifs.2021.06.026.
Bayram B, Ozkan G, Kostka T, Capanoglu E, Esatbeyoglu T. Valorization and application of fruit and vegetable wastes and by-products for food packaging materials. Molecules. 2021;26(13):4031. https://doi.org/10.3390/molecules26134031.
Bhat ZF, Bhat HF, Manzoor M, Abdi G, Aadil RM, Hassoun A, Aït-Kaddour A. Enhancing the lipid stability of foods of animal origin using edible packaging systems. Food Chemistry: X. 2024;101185. https://doi.org/10.1016/j.fochx.2024.101185
Falleh H, Jemaa MB, Saada M, Ksouri R. Essential oils: A promising eco-friendly food preservative. Food Chem. 2020;330:127268. https://doi.org/10.1016/j.foodchem.2020.127268.
Zanganeh H, Mortazavi SA, Shahidi F, Alizadeh Behbahani B. Evaluation of the chemical and antibacterial properties of Citrus paradise essential oil and its application in Lallemantia iberica seed mucilage edible coating to improve the physicochemical, microbiological and sensory properties of lamb during refrigerated storage. J Food Meas Charact. 2021;15(6):5556–71. https://doi.org/10.1007/s11694-021-01129-9.
Chiabrando V, Giacalone G. Effects of citrus essential oils incorporated in alginate coating on quality of fresh-cut Jintao kiwifruit. J Food Nutr Res. 2019;58:177–86.
Baek JH, Lee SY, Oh SW. Enhancing safety and quality of shrimp by nanoparticles of sodium alginate-based edible coating containing grapefruit seed extract. Int J Biol Macromol. 2021;189:84–90. https://doi.org/10.1016/j.ijbiomac.2021.08.118.
Won JS, Lee SJ, Park HH, Song KB, Min SC. Edible coating using a chitosan-based colloid incorporating grapefruit seed extract for cherry tomato safety and preservation. J Food Sci. 2018;83(1):138–46. https://doi.org/10.1111/1750-3841.14002.
Viscusi G, Lamberti E, D’Amico F, Tammaro L, Gorrasi G. Fabrication and characterization of bio-nanocomposites based on halloysite-encapsulating grapefruit seed oil in a pectin matrix as a novel bio-coating for strawberry protection. Nanomaterials. 2022;12(8):1265. https://doi.org/10.3390/nano12081265.
Kaur R, Kaur L. Encapsulated natural antimicrobials: a promising way to reduce microbial growth in different food systems. Food Control. 2021;123:107678. https://doi.org/10.1016/j.foodcont.2020.107678.
Choi JS, Lee YR, Ha YM, Seo HJ, Kim YH, Park SM, Sohn JH. Antibacterial effect of grapefruit seed extract (gse) on makgeolli-brewing microorganisms and its application in the preservation of fresh makgeolli. J Food Sci. 2014;79(6):M1159–67. https://doi.org/10.1111/1750-3841.12469.
Ukom AN, Ezenwigbo MC, Ugwuona FU. Grapefruit peel powder as a functional ingredient in cake production: effect on the physicochemical properties, antioxidant activity and sensory acceptability of cakes during storage. Int J Gastronomy Food Sci. 2022;28:100517. https://doi.org/10.1016/j.ijgfs.2022.100517.
de Oliveira FL, Arruda TYP, Morzelle MC, Pereira APA, Casarotti SN.Fruit by-products as potential prebiotics and promising functional ingredients to produce fermented milk. Food Research International. 2022;111841. https://doi.org/10.1016/j.foodres.2022.111841
Comunian TA, Silva MP, Souza CJ. The use of food by-products as a novel for functional foods: Their use as ingredients and for the encapsulation process. Trends Food Sci Technol. 2021;108:269–80. https://doi.org/10.1016/j.tifs.2021.01.003.
Ko MJ, Kwon HL, Chung MS. Optimum conditions for extracting flavanones from grapefruit peels and encapsulation of extracts. Korean Journal of Food Science and Technology. 2014;46(4):465–9. https://doi.org/10.9721/KJFST.2014.46.4.465.
de la Luz Cadiz-Gurrea M, del Carmen Villegas-Aguilar M, Leyva-Jiménez FJ, Pimentel-Moral S, Fernandez-Ochoa A, Alañón ME, Segura-Carretero A. Revalorization of bioactive compounds from tropical fruit by-products and industrial applications by means of sustainable approaches. Food Res Int. 2020;138:109786. https://doi.org/10.1016/j.foodres.2020.109786.
Michalak M. Plant-derived antioxidants: Significance in skin health and the ageing process. Int J Mol Sci. 2022;23(2):585. https://doi.org/10.3390/ijms23020585.
El Kady WM, Ayoub IM, El Mehrate AK, Emad M, Tarek M, El Gdeily A, ... Fathallah N. Valorization of Citrus peels: GC/MS‐based metabolites profiling, multivariate analysis, and antiaging potential. Archiv der Pharmazie. 2024;e2300742. https://doi.org/10.1002/ardp.202300742
Lukitaningsih E, Nur S, Qonithah F, Zulbayu A, Kuswahyuning R, Rumiyati R. In vitro anti-wrinkle and tyrosinase inhibitory activities of grapefruit peel and strawberry extracts. Majalah Obat Tradisional. 2020;25(3):182–9. https://doi.org/10.22146/mot.59194.
Ahmed H, Yousif E, Ahmed A, Yusop R, Zainulabdeen K, Ahmed DS, ... Husain A. The photostabilizing technology of grapefruit peel extract on polystyrene thin films: concept generation using morphological and physical properties. Progress in Color, Colorants and Coatings. 2024;17(2):159–173. https://doi.org/10.30509/pccc.2023.167205.1251
Zhang X, Xu H, Hua J, Zhu Z, Wang M. Protective effects of grapefruit essential oil against staphylococcus aureus-induced inflammation and cell damage in human epidermal keratinocytes. Chem Biodivers. 2022;19(6):e202200205. https://doi.org/10.1002/cbdv.202200205.
Yoon YC, Choi HJ, Park JH, Diniyah N, Shin HA, Kim MY. Combination of grapefruit and rosemary extracts has skin protective effect through MMPs, MAPKs, and the NF-κB signaling pathway In Vitro and in Vivo UVB-exposed model. Korean J Plant Resour. 2019;32(6):633–43. https://doi.org/10.7732/kjpr.2019.32.6.633.
Kiefer S, Weibel M, Smits J, Juch M, Tiedke J, Herbst N. Citrus flavonoids with skin lightening effects–Safety and efficacy studies. Int J Appl Sci. 2010;132:46–54. http://pascal-francis.inist.fr/vibad/index.php?action=getRecordDetail&idt=24061416. Accessed 14 Feb 2024.
Čižmárová B, Hubková B, Tomečková V, Birková A. Flavonoids as promising natural compounds in the prevention and treatment of selected skin diseases. Int J Mol Sci. 2023;24(7):6324. https://doi.org/10.3390/ijms24076324.
Gollavilli H, Hegde AR, Managuli RS, Bhaskar KV, Dengale SJ, Reddy MS, ... Mutalik S. Naringin nano-ethosomal novel sunscreen creams: Development and performance evaluation. Colloids and Surfaces B: Biointerfaces. 2020;193:111122. https://doi.org/10.1016/j.colsurfb.2020.111122
Petkova-Parlapanska K, Nancheva V, Diankov S, Hinkov I, Karsheva M. Rheological properties of cosmetic compositions containing rosemary and grapefruit pulp and seeds extracts. J Chem Technol Metall. 2014;49:487–93.
Zebua NF, Febrika N. Antioxidant activity and antiaging gel formulation grapefruit peel (Citrus maxima Merr.) ethanolic extract. http://repository.usu.ac.id/handle/123456789/64531. Accessed 14 Feb 2024.
Ha YM, Lee BB, Bae HJ, Je KM, Kim SR, Choi JS, Choi IS. Anti-microbial activity of grapefruit seed extract and processed sulfur solution against human skin pathogens. J Life Sci. 2009;19(1):94–100. https://doi.org/10.5352/JLS.2009.19.1.094.
Nuzzo D, Scordino M, Scurria A, Giardina C, Giordano F, Meneguzzo F, ... Di Liberto V. Protective, antioxidant and antiproliferative activity of grapefruit integropectin on SH-SY5Y cells. Int J Mol Sci. 2021;22(17):9368. https://doi.org/10.3390/ijms22179368
Ademosun AO, Oboh G, Adebayo AA, Olasehinde TA, Fasakin OW. Grapefruit peel extract mitigates paroxetine-induced erectile dysfunction in rats through stimulation of erectile response, antioxidant status, and inhibition of key enzymes related with impaired penile erection. J Food Biochem. 2022;46(11):e14193. https://doi.org/10.1111/jfbc.14193.
Miya G, Nyalambisa M, Oyedeji O, Gondwe M, Oyedeji A. Chemical profiling, toxicity and anti-inflammatory activities of essential oils from three grapefruit cultivars from KwaZulu-Natal in South Africa. Molecules. 2021;26(11):3387. https://doi.org/10.3390/molecules26113387.
Ahmed OM, Ashour MB, Fahim HI, Ahmed NA. Citrus limon and paradisi fruit peel hydroethanolic extracts prevent the progress of complete Freund’s adjuvant-induced arthritis in male Wistar rats. Adv Anim Vet Sci. 2018a;6(10), 443–455. https://doi.org/10.17582/journal.aavs/2018/6.10.443.460
Ahmed OM, Ashour MB, Fahim HI, Ahmed NA. The role of Th1/Th2/Th17 cytokines and antioxidant defense system in mediating the effects of lemon and grapefruit peel hydroethanolic extracts on adjuvant-induced arthritis in rats. J Appl Pharma Sci. 2018;8(10):069–81. https://doi.org/10.7324/JAPS.2018.81010.
Salah M, Abdul-Hamid M. Protective effect of orange and grapefruit peel extracts against testicular toxicity induced by sulfasalazine in male albino rats. Egypt J Histol. 2014;37(3):429–41. https://doi.org/10.1097/01.EHX.0000452106.79509.22.
Tsujiyama I, Mubassara S, Aoshima H, Hossain SJ. Anti-histamine release and anti-inflammatory activities of aqueous extracts of citrus fruits peels. Orient Pharm Exp Med. 2013;13:175–80. https://doi.org/10.1007/s13596-012-0093-z.
Jiang H, Zhang M, Lin X, Zheng X, Qi H, Chen J, ... Xiao G. Biological Activities and Solubilization Methodologies of Naringin. Foods. 2023;12(12):2327. https://doi.org/10.3390/foods12122327
Arsène MM, Podoprigora IV, Davares AK, Razan M, Das MS,Senyagin AN. Antibacterial activity of grapefruit peel extracts and green-synthesized silver nanoparticles. Veterinary World. 2021;14(5):1330. https://doi.org/10.14202/vetworld.2021.1330-1341
Bokhary KA, Maqsood F, Amina M, Aldarwesh A, Mofty HK, Al-Yousef HM. Grapefruit extract-mediated fabrication of photosensitive aluminum oxide nanoparticle and their antioxidant and anti-inflammatory potential. Nanomaterials. 2022;12(11):1885. https://doi.org/10.3390/nano12111885.
Han HW, Kwak JH, Jang TS, Knowles JC, Kim HW, Lee HH, Lee JH. Grapefruit seed extract as a natural derived antibacterial substance against multidrug-resistant bacteria. Antibiotics. 2021;10(1):85. https://doi.org/10.3390/antibiotics10010085.
Osman, A. I., El-Monaem, E. M. A., Elgarahy, A. M., Aniagor, C. O., Hosny, M., Farghali, M., ... & Eltaweil, A. S. (2023). Methods to prepare biosorbents and magnetic sorbents for water treatment: a review. Environmental Chemistry Letters, 1–62. https://doi.org/10.1007/s10311-023-01603-4
Saravanan A, Karishma S, Kumar PS, Thamarai P, Yaashikaa PR. Recent insights into mechanism of modified bio-adsorbents for the remediation of environmental pollutants. Environmental Pollution. 2023;122720. https://doi.org/10.1016/j.envpol.2023.122720
Torab-Mostaedi M, Asadollahzadeh M, Hemmati A, Khosravi A. Equilibrium, kinetic, and thermodynamic studies for biosorption of cadmium and nickel on grapefruit peel. J Taiwan Inst Chem Eng. 2013;44(2):295–302. https://doi.org/10.1016/j.jtice.2012.11.001.
Shahsavan-Davoudi MH, Ziarati P. Green method for cadmium removal from pharmaceutical effluent laboratories by grapefruit peel. J Med Discov. 2020;5(3):1–13. https://doi.org/10.24262/jmd.5.3.20036
Nyika J, Dinka MO. The potential of reusing fruit bio-adsorbents for water purification: A minireview. Mater Today: Proc. 2023. https://doi.org/10.1016/j.matpr.2023.07.285
Rosales E, Meijide J, Tavares T, Pazos M, Sanromán MA. Grapefruit peelings as a promising biosorbent for the removal of leather dyes and hexavalent chromium. Process Saf Environ Prot. 2016;101:61–71. https://doi.org/10.1016/j.psep.2016.03.006.
Fu Y, Yang Z, Xia Y, Xing Y, Gui X. Adsorption of ciprofloxacin pollutants in aqueous solution using modified waste grapefruit peel. Energy Sources, Part A: Recovery, Utilization, and Environmental Effects. 2021;43(2):225–34. https://doi.org/10.1080/15567036.2019.1624877.
Zhang W, Song J, He Q, Wang H, Lyu W, Feng H, ... Chen L. Novel pectin based composite hydrogel derived from grapefruit peel for enhanced Cu (II) removal. J Hazard Mater. 2020;384:121445. https://doi.org/10.1016/j.jhazmat.2019.121445
Imran M, Islam A, Farooq MU, Ye J, Zhang P. Characterization and adsorption capacity of modified 3D porous aerogel from grapefruit peels for removal of oils and organic solvents. Environ Sci Pollut Res. 2020;27:43493–504. https://doi.org/10.1007/s11356-020-09085-5.
Romero-Cano LA, González-Gutiérrez LV, Baldenegro-Pérez LA, Carrasco-Marín F. Grapefruit peels as biosorbent: characterization and use in batch and fixed bed column for Cu (II) uptake from wastewater. J Chem Technol Biotechnol. 2017;92(7):1650–8. https://doi.org/10.1002/jctb.5161.
Inkoua S, Li C, Zhang L, Li X, Zhang S, Xiang J, ... Hu X. Hydrothermal carbonization of grapefruit peels in swine manure-derived liquid: Property of resulting hydrochars and activated carbons. Fuel. 2023;351:128889. https://doi.org/10.1016/j.fuel.2023.128889
Yu H, Gu L, Chen L, Wen H, Zhang D, Tao H. Activation of grapefruit derived biochar by its peel extracts and its performance for tetracycline removal. Biores Technol. 2020;316:123971. https://doi.org/10.1016/j.biortech.2020.123971.
Chojnacka K. Valorization of biorefinery residues for sustainable fertilizer production: a comprehensive review. Biomass Conv Bioref. 2023;13(16):14359–88. https://doi.org/10.1007/s13399-023-04639-2.
Zema DA, Calabrò PS, Folino A, Tamburino VINCENZO, Zappia G, Zimbone SM. Valorisation of citrus processing waste: A review. Waste Manage. 2018;80:252–73. https://doi.org/10.1016/j.wasman.2018.09.024.
Navarro MA. Grapefruit Peel Biochar Applications for Immobilizing Copper and Lead in Soil. 2022. https://www.proquest.com/openview/eb3c430e44d9394b150a62550a402b87/1?pq-origsite=gscholar&cbl=18750&diss=y.
Singh A, Singhania RR, Soam S, Chen CW, Haldar D, Varjani S, ... Patel AK. Production of bioethanol from food waste: Status and perspectives. Bioresource technology. 2022;127651. https://doi.org/10.1016/j.biortech.2022.127651
John I, Muthukumar K, Arunagiri A. A review on the potential of citrus waste for D-Limonene, pectin, and bioethanol production. Int J Green Energy. 2017;14(7):599–612. https://doi.org/10.1080/15435075.2017.1307753.
Wilkins MR, Widmer WW, Grohmann K, Cameron RG. Hydrolysis of grapefruit peel waste with cellulase and pectinase enzymes. Biores Technol. 2007;98(8):1596–601. https://doi.org/10.1016/j.biortech.2006.06.022.
Wikandari R, Sanjaya AP, Millati R, Karimi K, Taherzadeh MJ. Fermentation inhibitors in ethanol and biogas processes and strategies to counteract their effects. In: Biofuels: alternative feedstocks and conversion processes for the production of liquid and gaseous biofuels, 2019. pp. 461–499). Academic Press. https://doi.org/10.1016/B978-0-12-816856-1.00020-8
Teke GM, De Vos L, Smith I, Kleyn T, Mapholi Z. Development of an ultrasound-assisted pre-treatment strategy for the extraction of d-Limonene toward the production of bioethanol from citrus peel waste (CPW). Bioprocess Biosyst Eng. 2023;46(11):1627–37. https://doi.org/10.1007/s00449-023-02924-y.
Choi IS, Lee YG, Khanal SK, Park BJ, Bae HJ. A low-energy, cost-effective approach to fruit and citrus peel waste processing for bioethanol production. Appl Energy. 2015;140:65–74. https://doi.org/10.1016/j.apenergy.2014.11.070.
Acknowledgements
The authors are thankful to I. K. Gujral Punjab Technical University, Kapurthala, Punjab, India, for providing the necessary infrastructure and support for the study.
Author information
Authors and Affiliations
Contributions
SC: Conceptualization, methodology, investigation, visualization, data curation, writing—original draft, manuscript editing, and review. BS: Project administration, supervision, conceptualization, methodology, revision of manuscript, manuscript editing, and review.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Conflict of interest
The authors declare no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Competing of Interest
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chaudhary, S., Singh, B. From Bin to Benefit: Sustainable Valorization of Grapefruit (Citrus paradisi) Byproducts Towards the Circular Economy. Curr Food Sci Tech Rep 2, 271–296 (2024). https://doi.org/10.1007/s43555-024-00033-9
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43555-024-00033-9