Abstract
Effects of foliage extracts of goat weed, Ageratum conyzoides (Family: Asteraceae) on feeding, oviposition and egg hatchability of diamondback moth, Plutella xylostella were assessed under standard laboratory conditions. Bioassays using aqueous, methanol and hexane crude extracts were conducted in no-choice and choice conditions. The larval feeding on 0.5% hexane extract treated leaf surface reduced to more than half relative to control in the choice condition; however, complete inhibition of feeding was recorded at 2% and higher concentration of extract. In no-choice bioassays, the larval feeding consistently decreased with increasing concentration of the hexane extract. Larval feeding on methanol extract treated leaves was also significantly reduced, although in the concentration dependent manner in both choice and no-choice bioassays. Likewise, the aqueous extract also reduced larval feeding at 3%, 4% and 5% concentrations of extracts in both no-choice and choice bioassays. The Antifeedant Index (AFI) for hexane extract was 100 at 2% concentration, while AFI values were 78.4 and 41.5 for 5% concentration of methanol and aqueous extracts, respectively. The egg laying on the leaf surface treated with hexane and methanol extracts was significantly reduced in the concentration dependent manner, as compared to the control in both choice and no-choice bioassays. However, the aqueous extract did not affect egg laying. The egg hatchability was completely inhibited at higher concentrations of the hexane extract, but not at any concentration of the methanol and aqueous extracts. The oviposition Deterrent Index values for the hexane and methanol extracts were statistically similar, however. These results indicate that the crude hexane and methanol extracts of A. conyzoides foliage could potentially be used for the management of diamondback moth.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The diamondback moth (DBM), Plutella xylostella (Lepidoptera: Plutellidae) is an invasive species which is considered to have European origin but now distributed throughout the world. This is one of the most destructive pest of crucifer plants (Li et al. 2016a; Thorsteinson 1953; Zalucki et al. 2012). DBM has also been reported to infest pea plant, Pisum sativum (L.) (Fabaceae) in Kenya (Henniges-Janssen et al. 2014). Management cost along with economic loss in crop production due to this insect has been estimated US $ 4-US $5 billion annually (Furlong et al. 2013; Li et al. 2016b; Verkerk and Wright 1996; Zalucki et al. 2012). Chemicals control strategies for the management of this pest has failed to produce a satisfactory result, since insect has developed resistance to all the classes of insecticides (Agboyi et al. 2016; Furlong et al. 2013; Talekar and Shelton 1993). This insect has also been reported to develop field resistance against biopesticide, Bacillus thuringiensis (Shelton et al. 1993; Tabashnik et al. 1990). Moreover, indiscriminate use of insecticides has led to environmental hazards (Mahmood et al. 2016). Therefore, this is necessary to search an alternative approach for the sustainable management of DBM.
Plants are important natural sources of bioactive compounds and many of them have been reported to possess insecticidal properties (Boulogne and Petit 2012; Isman 1995; Kraikrathok et al. 2013; Morallo-Rejesus 1986). These secondary compounds may provide an alternative to the chemical control (Ibrahim et al. 2004). Ageratum conyzoides (Asteraceae), also known as ‘goatweed’, has been reported to cause adverse effect on the biology of insects like the ovarian inhibition in Dysdercus flavidus (Fagonee and Umrit 1981), precocious metamorphosis and delayed development in Heliothis zea (Bowers and Feldlaufer 1982), acute toxicity against Callosobruchus maculatus (Bouda et al. 2001) and antijuvenile activity against Anopheles stephensi and Culex quinquefasciatus (Saxena et al. 1994). These findings indicate presence of bioactive compound in A. conyzoides that may be exploited for insect control. There is hardly any report about the bioactivity of A. conyzoides on behavioural responses of P. xylostella. Any information regarding bioactivity of A. conyzoides against P. xylostella may be helpful in developing alternative strategy to chemical control. With this objective, present study was initiated to evaluate the effect of A. conyzoides foliage extracts on feeding and oviposition behaviour of P. xylostella.
Materials and methods
Diamondback moth was reared in the laboratory on cauliflower (Brassica oleraceae: Brassicaceae) leaves under standard conditions in a Biological Oxygen Demand Incubator (BOD), maintaining 25 ± 2 °C temperature, 65 ± 5% Relative Humidity (RH) and 14 Dark (D): 10 Light (L) photoperiod regime. The cauliflower (variety Poosi special) seeds were obtained from National Seeds Bank, Indian Agriculture Research Institute, Pusa, New Delhi. These seeds were grown in the field plots of Zoology Department, University of Delhi, India under pesticide free condition, following standard farm practices. The freshly excised leaves of these plants were used to maintain culture of P. xylostella, and also for conducting various bioassays.
Preparation of foliar extracts
The young leaves of A. conyzoides were collected in April from Rishikesh, Uttarakhand, India. The collected leaves were thoroughly washed with water and air-dried under the shade inside a room maintaining at 40 °C. The dried leaves were ground into a fine powder form in an electric grinder (Philips HL1643/04). The powdered material was stored in refrigerator for preparation of extracts. Later, aqueous extract was prepared by immersing 10 g of leaf powder in 100 ml of distilled water in a glass beaker, and kept at room temperature for 24 h. Next day, beaker was shaken and liquid suspension was filtered through Whatman paper (No. 1) to obtain 10% aqueous extract. The methanol and hexane leaf extracts were prepared by immersing 100 g of leaf powder in 500 ml of the respective solvent (methanol or hexane) in separate conical flasks. These were kept undisturbed for 24 h at the normal room temperature. The flasks were shaken and the liquid suspension was decanted in the glass beaker. The process was repeated three times, each time immersing with respective solvent for 24 h, and liquid suspension was pooled in the same beaker every time. The filtrate was subsequently filtered using Whatman filter paper (No. 1) that was evaporated in Rotary Evaporator (Büchi Rotary Evaporator, Model R-200) at 40 °C under reduced pressure. The obtained crude extracts were refrigerated for further use. However, the aqueous extract was always prepared fresh every time before use.
The control solution for methanol extract was prepared by mixing 9.0 ml of water and 1 ml of methanol, whereas, for hexane extract this was 8 ml of water and 2 ml of hexane. One gram of crude methanol or hexane extract was dissolved in 10 ml of control solution for preparation of stock solution. Different concentrations of extracts (5%, 4%, 3%, 2% and 1%) were prepared by serial dilutions of stock solution. One drop (50 μl) of Triton X was added in the extract as emulsifier, before use.
Antifeedant bioassays
Feeding bioassays were conducted in no-choice and choice conditions. In no-choice feeding bioassays, circular leaf discs (2.5 cm dia) were cut using sharp edged razor from freshly excised cauliflower leaves, obtained from the Departmental plot. These discs were dipped in respective extract concentrations for 5 s and air dried for 5 min. A single leaf disc, control or treated, was placed in the centre of a Petri dish (9 cm dia × 4 cm ht), lined with a moist tissue paper. Five overnight moulted fourth instar DBM larvae, starved for 4 h, were released on the leaf disc. The Petri dish was transferred to the BOD incubator set at standard conditions (25 ± 2 °C, 65 ± 5% RH, 14D: 10 L). After 18 h of feeding, the leaf discs were taken out, and the area consumed by larvae was measured using a graph sheet. Each experiment was replicated five times with different batches of larvae for each extract concentration along with the control. The choice bioassays were performed in similar way, except both the control and treated leaf discs were placed in the same Petri dish at a distance of 3 cm.
The Antifeedant Index (AFI) was calculated using formula (Isman 1992):
where, C and T represent control and treated leaf area consumed by the larvae respectively.
Oviposition bioassays
Both no-choice and choice bioassays were performed with freshly emerged male and female moths. These moths were released in a mating cage for 24 h and used for oviposition bioassays next day. Oviposition cage was made of a rectangular plexiglass (45 cm × 20 cm × 20 cm) with windows (10 cm × 10 cm) on side walls, covered with muslin cloth. The cage had also an opening (14 cm × 14 cm) in the middle of the front wall, fitted with a muslin cloth sleeve to facilitate the handling of moths. The bottom of the cage was lined with tissue paper. The oviposition cage was marked in 3 equal sectors, i.e. two sides and middle. Two diet cups having 10% sucrose solution, soaked in cotton swabs were placed on cage floor for feeding of the adults. Freshly excised tender cauliflower leaves of same size were painted on both sides using a fine hair brush with the plant extract of desired concentration and air dried at room temperature for 5 min. Similarly, the leaf surfaces painted with control solution served as control. Bouquet of leaf, applied with either extract or control solution, was prepared by dipping its petiole into water, filled in a reagent bottle to prevent wilting. The mouth of bottle was plugged with cotton. In choice bioassays, both control and treated leaf bouquets were placed inside the oviposition cage at opposite end sectors. Five pairs of mated male and female moths were released in the cage just before the onset of dark phase. The oviposition cage was kept in BOD incubator set at standard conditions (25 ± 2 °C, 65 ± 5% RH, 14D: 10 L). After 24 h, bouquets of treated and/or control leaf were removed, and eggs laid were counted separately. The experiments were replicated five times, each replicate with fresh 5 pairs of males and females from different batches of the stock culture. The sides of the treated and control leaf bouquets were randomized in the oviposition cage during the experiment to avoid the side effects. The Oviposition Deterrent Index (ODI) was calculated, described by Huang and Renwick (1994), as follows.
where, Cn and Tn represents number of eggs laid on control and treated leaves, respectively.
Similar procedure was adopted for no-choice bioassays, except that single leaf bouquet was placed, either control or treated, at centre of the oviposition cage.
Egg hatchability bioassays
The leaf containing overnight laid eggs was removed and cut into pieces with sharp edged razor, each piece having 20 eggs. The leaf pieces were dipped separately into respective extract concentrations or control solution for 20 s, and air dried at room temperature for 5 min. Each leaf piece with eggs was placed in a plastic container (10 cm × 10 cm), lined with a moist blotting paper and kept in BOD incubator as mentioned earlier. The containers were inspected twice daily at 8 a.m. and 8 p.m., for 5 days to record egg hatched. Each experiment was replicated five times with different batches of eggs.
Statistical analysis
Experimental data were subjected to one way analysis of variance (1 way-ANOVA) followed by Tukey’s pair-wise multiple comparison test, if ANOVA indicated a significant effect. 2- way ANOVA was used to test the effect of two factors followed by Tukey’s test, if ANOVA indicated a significant difference (F values obtained were described only for the interaction between the concentration and the extract type). In choice bioassays, paired t-test was used to compare control and treatment groups (antifeedant and ovipositional bioassays). All statistical analyses were performed on the computer using Sigma Stat 2.0, Jandel Scientific Software, 1995.
Results
Effect on feeding Reponses
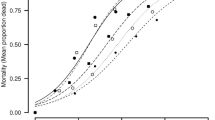
The difference in mean area consumption by DBM larvae on aqueous, methanol and hexane extract treated leaf disc was statistically significant as compared to control, in both no-choice and choice bioassays (P < 0.05; Tables 1 and 2). There was also a positive correlation of extract concentrations with antifeedant index (AFI) values (Methanol: R2 = 0.911; Hexane: R2 = 0.807) (Table, 3a, 3b; Figs. 1, 2). The AFI value at 2% concentration of the hexane extract was 100, since no consumption of leaf was recorded (Table 3b). However, AFI values did not differ significantly between aqueous and methanol extracts, applied on leaf surface at different concentrations (F (4, 40) = 0.744; P > 0.05; Table 3a).
Effect on ovipositional responses
Concentration dependent deterrent effect of the aqueous, methanol and hexane crude foliage extracts was observed on the oviposition behaviour of P. xylostella females, in both no-choice and choice bioassays as compared to control (P < 0.05; Table 4a,b). There was no difference between the eggs laid on leaf surface treated with aqueous and methanol extracts (F (10, 72) = 2.579; P > 0.05; Table 4a). However, a significant difference between the eggs laid on methanol and hexane treated leaf surface was observed at higher concentrations (P < 0.05; Table 4a). The ODI values were positively related with concentrations of both methanol and hexane extracts. However, when data were subjected to 2-way ANOVA, ODI values were not significant between different concentrations of methanol and hexane extracts (F (4, 40) = 1.242; Table 5).
Effect on egg hatchability
The ovicidal effect of hexane extract on the hatchability of P. xylostella eggs was observed. Hatchability significantly decreased at lower concentrations of extracts as compared to control, and was completely inhibited at 4% and 5% concentration of hexane extract (Table 6). However, inhibition effect of aqueous and methanol extract on the hatchability was not observed in this study.
Discussion
The leaf extracts of A. conyzoides strongly inhibited feeding of P. xylostella larvae. The larvae took ‘test bite’ and maintained feeding, if it was the control leaf. By contrast, both in no-choice and choice bioassays, larvae stopped feeding after initial ‘test bite’ and moved away if the leaf was smeared with an extract. Thereafter, larvae showed wandering behaviour in the Petri dish. However, after a period of starvation, larvae accepted the treated leaf disc and resumed feeding. The feeding of DBM larvae on cauliflower leaves has been attributed to glucosinolates, which act as the feeding stimulant (Badenes-Pérez et al. 2011, 2013). Such responses are mediated by sensory receptors, located on mouth parts of larvae (Chapman 1995). The inhibition of larval feeding may be either due to the masking of stimulant compounds in leaf, which provide neural inputs to brain for initiation of feeding, or due to presence of deterrent chemicals in the extract that inhibit feeding. Such antifeedant effect from the extracts of other plants, belonging to Asteraceae family, has also been reported earlier against many lepidopterans (Facknath and Lalljee 2008; González-Coloma et al. 2005; Passreiter and Isman 1997; Susurluk et al. 2007; Tandon et al. 1998) and coleopteran pests (Amelot et al. 2003). The crude hexane extract of A. conyzoides has been found to strongly suppress the feeding of Helicoverpa armigera larvae with a deterrent index (DI50) of 0.21 (Ragesh et al. 2016). However, such deterrent effects of the non-polar extract of A. conyzoides were not observed against Spodoptera litura larvae (Singh and Rao 2000). This may be because of differences in susceptibilities of various insects towards goatweed plant extracts. Such susceptibility differences of closely related insect species towards extracts of the same plant have also been observed in earlier studies (Champagne et al. 1992; Isman 1993; Xie et al. 1994).
Reduced egg laying by P. xylostella females on host cauliflower leaf surface treated with all the tested extracts, i.e. aqueous, methanol and hexane of A. conyzoides foliage were observed. This may be either due to the presence of repellent in the extract, restricting arrival of female moths on oviposition substrates, or due to the presence of deterrent chemicals in the extracts that inhibit egg laying by gravid females on arrival or both. Such reduction in egg laying by Callosobruchus maculatus females was observed, when the volatile oil from dried leaves of A. conyzoides was applied on cowpea (Gbolade et al. 1999). Liu et al. (2005) also recorded a significant reduction in egg laying by P. xylostella females on cabbage leaves smeared with the methanol foliage extract of Chrysanthemum morifolium (Asteraceae) in a choice bioassay. Also, olfactometer bioassay revealed that this reduction in the oviposition was due to the repellent effect of the methanol extract of C. morifolium (Liu et al. 2005, 2006). Oviposition deterrent effects (ODE) of A. conyzoides crude hexane extract were also reported on Helicoverpa armigera females (Ragesh 2011). Hough-Goldstein and Hahn (1992) reported deterrent effect of aqueous extracts of Tanacetum vulgare (Asteraceae) on egg laying of P. xylostella. Such ODE on oviposition of diamondback moth has been also been recorded in earlier studies from plant extracts of Chinaberry (Chen et al. 1996), Neem (Charleston et al. 2005) and Yam bean (Basukriadi and Wilkins 2014). The inhibition occurs through chemoreception for which the receptors are located on the antennae, proboscis, ovipositor and tarsi of insects, transmitting signals to the central nervous system for information processing (Schoonhoven et al. 2005). Present study clearly indicates that A. conyzoides possesses chemicals that inhibit feeding of larvae and oviposition of gravid P. xylostella females.
The effect of hexane extract of A. conyzoides on hatching of P. xylostella eggs indicates the presence of inhibitory compounds in extract that adversely affect development. Such ovicidal activity of essential oil from foliage of Artemisia abrotanum L. and Tanacetum vulgare L., both belonging to Asteraceae, on P. xylostella egg has also been observed by Sangha et al. (2017). Ovicidal effect has also been reported from lipid extract of A. conyzoides on Dysdercus flavidus eggs (Fagonee and Umrit 1981) and neem oil on Dysdercus koenigii eggs (Bhathal et al. 1991).
Present study shows that A. conyzoides has a potential for its use in the management of P. xylostella. Its antifeedant property will deter larval feeding, and reduce damage to the plants. Moreover, oviposition repellent and deterrent effects of the extract will reduce the number of eggs laid on plant surface, which along with its ovicidal effect will decelerate the population growth rate and in turn, reduce future damage. However, experiments have to be conducted in Greenhouse to assess the influence of environment factor and implication of the effect of these extract/s on benefic insects for its applicability in the field. This management system for DBM will be environmental friendly and sustainable with minimum chance of the insect developing resistance to plant derivatives.
References
Agboyi LK, Ketoh GK, Martin T, Glitho IA (2016) Pesticide resistance in Plutella xylostella (Lepidoptera: Plutellidae) populations from Togo and Benin. Int J Trop Insect Sc 36(4):204–210. https://doi.org/10.1017/S1742758416000138
Amelot MEA, Avendaño M, Aubert L, Avila JL (2003) Repellency and feeding deterrence activity of Ageratum conyzoides against the stored grain pests Tribolium castaneum and Sitophilus oryzae. Active Plant Parts Compos Cienc 11(1):61–76
Badenes-Pérez FR, Reichelt M, Gershenzon J, Heckel DG (2011) Phylloplane location of glucosinolates in Barbarea spp. (Brassicaceae) and misleading assessment of host suitability by a specialist herbivore. New Phytol 189(2):549–556. https://doi.org/10.1111/j.1469-8137.2010.03486.x
Badenes-Pérez FR, Reichelt M, Gershenzon J, Heckel DG (2013) Interaction of glucosinolate content of Arabidopsis thaliana mutant lines and feeding and oviposition by generalist and specialist lepidopterans. Phytochemistry 86:36–43. https://doi.org/10.1016/j.phytochem.2012.11.006
Basukriadi A, Wilkins RM (2014) Oviposition deterrent activities of Pachyrhizus erosus seed extract and other natural products on Plutella xylostella (Lepidoptera : Plutellidae). J Insect Sci 14(1):244. https://doi.org/10.1093/jisesa/ieu106
Bhathal SS, Singh D, Dhillon RS, Nayyar K (1991) Ovicidal effect of neem oil and plant extract of Ageratum conyzoides linn. on Dysdercus koenigii fab. J Insect Sci 4(2):185–186
Bouda H, Tapondjou LA, Fontem DA, Gumedzoe MYD (2001) Effect of essential oils from the leaves of Ageratum conyzoides, Lantana camara and Chromolaena odorata on the mortality of Sitophilus zeamais (Coleoptera: Curculionidae). J Stored Prod Res 37(2):103–109
Boulogne I, Petit P (2012) Insecticidal and antifungal chemicals produced by plants. A review: Environ Chem Lett 10(4):325–347. https://doi.org/10.1007/s10311-012-0359-1
Bowers WS, Feldlaufer MF (1982) In vitro inactivation of Tenebrio molitor corpora allata by a synthetic precocene analogue. Gen Comp Endocrinol 47(1):120–124
Champagne DE, Koul O, Isman MB, Scudder GGE, Towers GHN (1992) Biological activity of limonoids from the Rutales. Phytochemistry 31(2):377–394. https://doi.org/10.1016/0031-9422(92)90003-9
Chapman RF (1995) Mechanics of food handling by chewing insects. In: Chapman RF, de Boer G (eds) Regulatory mechanisms in insect feeding. Chapman & Hall, New York, pp 3–31
Charleston DS, Kfir R, LEM V, Dicke M (2005) Behavioural responses of diamondback moth Plutella xylostella (Lepidoptera: Plutellidae) to extracts derived from Melia azedarach and Azadirachta indica. Bull Entomol Res 95(5):457–465. https://doi.org/10.1079/BER2005377
Chen CC, Chang SJ, Cheng LL, Hou RF (1996) Deterrent effect of the chinaberry extract on oviposition of the diamondback moth, Plutella xylostella (L.) (Lep., Yponomeutidae). J Appl Ent 120:165–169. https://doi.org/10.1111/j.1439-0418.1996.tb01585.x
Facknath S, Lalljee B (2008) Study of various extracts of Ayapana triplinervis for their potential in controlling three insect pests of horticultural crops. Tropicultura 26(2):119–124
Fagonee I, Umrit G (1981) Anti-gonadotropic hormones from the goat weed, Ageratum conyzoides. Insect Sci Appl 1(4):373–376
Furlong MJ, Wright DJ, Dosdall LM (2013) Diamondback moth ecology and management: problems, progress, and prospects. Annu Rev Entomol 58:517–541. https://doi.org/10.1146/annurev-ento-120811-153605
Gbolade A, Onayade O, Ayinde BA (1999) Insecticidal activity of Ageratum conyzoides L. volatile oil against Callosobruchus maculatus F. in seed treatment and fumigation laboratory tests. Insect Sci Applic 19(2–3):237–240. https://doi.org/10.1017/S1742758400019536
González-Coloma A, Guadaño A, Tonn CE, Sosa ME (2005) Antifeedant/insecticidal terpenes from Asteraceae and Labiatae species native to Argentinean semi-arid lands. Z Naturforsch C 60(11–12):855–861
Henniges-Janssen K, Heckel DG, Groot AT (2014) Preference of diamondback moth larvae for novel and original host plant after host range expansion. Insects 5(4):793–804. https://doi.org/10.3390/insects5040793
Hough-Goldstein JA, Hahn SP (1992) Antifeedant and oviposition deterrent activity of an aqueous extract of Tanacetum vulgare L. on two cabbage pests. Environ Entomol 21(4):837–844. https://doi.org/10.1093/ee/21.4.837
Huang X, Renwick JAA (1994) Cardenolides as oviposition deterrents to two Pieris species: structure-activity relationships. J Chem Ecol 20(5):1039–1051. https://doi.org/10.1007/BF02059740
Ibrahim MA, Oksanen EJ, Holopainen JK (2004) Effects of limonene on growth and physiology of cabbage and carrot plants. J Sci Food Agric 84(11):1319–1326. https://doi.org/10.1002/jsfa.1819
Isman MB (1992) A physiological perspective. In: Roitberg BD, Isman MB (eds) Insect chemical ecology: an evolutionary approach. Chapman and Hall, New York, pp 156–176
Isman MB (1993) Growth inhibitory and antifeedant effects of azadirachtin on six noctuids of regional economic importance. Pestic Sci 38:57–63. https://doi.org/10.1002/ps.2780380109
Isman MB (1995) Leads and prospects for the development of new botanical insecticides. Rev Toxicol 3:1–20 AGR:IND20587890
Kraikrathok C, Ngamsaeng S, Bullangpoti V, Pluempanupat W, Koul O (2013) Bioefficacy of some Piperaceae plant extracts against Plutella xylostella L. (Lepidoptera:Plutellidae). Commun Agric Appl Biol Sci 78(2):305–310
Li Z, Feng X, Liu SS, You M, Furlong MJ (2016a) Biology, ecology and Management of Diamondback Moth in China. Annu Rev Entomol 61:277–296
Li Z, Zalucki MP, Yonow T, Kriticos DJ (2016b) Population dynamics and management of diamondback moth (Plutella xylostella) in China: the relative contributions of climate, natural enemies and cropping patterns. Bull Entomol Res 102(2):197–214. https://doi.org/10.1017/S0007485315001017
Liu SS, Li YH, Liu YQ, Zalucki MP (2005) Experience-induced preference for oviposition repellents derived from a non-host plant by a specialist herbivore. Ecol Lett 8(7):722–729. https://doi.org/10.1111/j.1461-0248.2005.00776.x
Liu SS, Li YH, Lou YG (2006) Non-host plant extracts reduce oviposition of Plutella xylostella (Lepidoptera: Plutellidae) and enhance parasitism by its parasitoid Cotesia plutellae (Hymenoptera: Broconidae). Bull Entomol Res 96(4):373–378. https://doi.org/10.1079/BER2006441
Mahmood I, Imadi SR, Shazadi K, Gul A (2015) Effects of pesticides on environment: pp. 253-269. In: Hakeem KR et al (eds) Plant, Soil and Microbes. Springer International Publishing, Cham. https://doi.org/10.1007/978-3-319-27455-3_13
Morallo-Rejesus M (1986) Botanical pesticides against the diamondback moth, pp. 241-255. In: Talekar, NS, and Griggs TD (eds) Diamondback moth management. Proceedings of the first international workshop, 11-15, March, 1985, Asian Vegetables Research and Develoment Centre; Taiwan
Passreiter CM, Isman MB (1997) Antifeedant bioactivity of sesquiterpene lactones from Neurolaena lobata and their antagonism by gamma aminobutyric acid. Biochem Syst Ecol 25(5):371–377. https://doi.org/10.1016/S0305-1978(97)00023-9
Ragesh PR (2011) Biological activity of Ageratum conyzoides (L.) (Asteraceae) extracts against gram podborer, Helicoverpa armigera (Hubner) (Lepidoptera: Noctuidae). Ph.D. Thesis, University of Delhi, Delhi, India
Ragesh PR, Bhutia TN, Ganta S, Singh AK (2016) Repellent, antifeedant and toxic effects of Ageratum conyzoides (Linnaeus) (Asteraceae) extract against Helicoverpa armigera (Hübner) (Lepidoptera:Noctuidae). Arch Phytopathol Plant Prot 49(1-4):19–30. https://doi.org/10.1080/03235408.2016.1147123
Sangha JS, Astatkie T, Cutler GC (2017) Ovicidal, larvicidal, and behavioural effects of some plant essential oils on diamondback moth (Lepidoptera: Plutellidae). Can Entomol 00:1–10. https://doi.org/10.4039/tce.2017.13
Saxena RC, Jayashree S, Padma S, Dixit OP (1994) Evaluation of growth disrupting activity of Ageratum conyzoides crude extract on Culex quinquefasciatus. J Environ Biol 15(1):67–74
Schoonhoven LM, van Loon JJA, Dicke M (2005) Insect-plant biology, 2nd edn. Oxford University Press, Oxford
Shelton AM, Robertson JL, Tang JD, Perez C, Eigenbrode SD, Preisler HK, Wilsey WT, Cooley RJ (1993) Resistance of diamondback moth (Lepidoptera: Plutellidae) to Bacillus thuringiensis subspecies in the field. J Econ Entomol 86:697–705
Singh S, Rao PJ (2000) Effect of Ageratum conyzoides on development and reproduction of Spodoptera litura. Indian J Entomol 62(3):231–238
Susurluk H, Caliskan Z, Gurkan O, Kirmizigul S, Goren N (2007) Antifeedant activity of some Tanacetum species and bioassay guided isolation of the secondary metabolites of Tanacetum cadmium ssp. (Compositae). Ind Crop Prod 26(2):220–228. https://doi.org/10.1016/j.indcrop.2007.04.002
Tabashnik BE, Cushing NL, Finson N, Johnson MW (1990) Field development of resistance to Bacillus thuringiensis in diamondback moth (Lepidoptera: Plutellidae). J Econ Entomol 84:49–55
Talekar NS, Shelton AM (1993) Biology, ecology, and management of the diamondback moth. Annu Rev Entomol 38:275–301. https://doi.org/10.1146/annurev.en.38.010193.001423
Tandon M, Shukla YN, Tripathi AK, Singh SC (1998) Insect antifeedant principles from Vernonia cinerea. Phytother Res 12(3):195–199. https://doi.org/10.1002/(SICI)1099-1573(199805)12:3<195::AID-PTR223>3.0.CO;2-%23
Thorsteinson AJ (1953) The chemotactic responses that determine host specificity in an oligophagous insect Plutella maculipennis (Curt.) Lepidoptera:Plutellidae. Can J Zool 31(1):52–72. https://doi.org/10.1139/z53-006
Verkerk RHJ, Wright DJ (1996) Multitrophic interactions and management of the diamondback moth: a review. Bull Entomol Res 86(3):205–216. https://doi.org/10.1017/S0007485300052482
Xie YS, Isman MB, Gunning P, MacKinnon S, Arnason JT, Taylor DR, Sanchez P, Hasbun C, Towers GHN (1994) Biological activity of extracts of Trichilia species and the limonoid Hirtin against lepidopteran larvae. Biochem Syst Ecol 22(2):129–136. https://doi.org/10.1016/0305-1978(94)90003-5
Zalucki MP, Shabbir A, Silva R, Adamson D, Shu-Sheng L, Furlong MJ (2012) Estimating the economic cost of one of the world's major insect pests, Plutella xylostella (Lepidoptera: Plutellidae): just how long is a piece of string? J Econ Entomol 105(4):1115–1129. https://doi.org/10.1603/EC12107
Acknowledgements
The authors wish to thank University Grant Commission, Council for Scientific and Industrial Research and University of Delhi to provide financial support for this study. We also thank to National Seed Bank, Indian Agriculture Research Institute, New Delhi to provide Cauliflower seeds of required cultivar.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vats, T.K., Rawal, V., Mullick, S. et al. Bioactivity of Ageratum conyzoides (L.) (Asteraceae) on feeding and oviposition behaviour of diamondback moth Plutella xylostella (L.) (Lepidoptera: Plutellidae). Int J Trop Insect Sci 39, 311–318 (2019). https://doi.org/10.1007/s42690-019-00042-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42690-019-00042-5