Abstract
Liverworts are considered as the earliest-divergent clade of land plants, while in phylogenetics mosses are the more advanced sister group to a clade that existed between hornworts and tracheophytes. In the present studies, a comparative analysis was done to understand the biochemical and physiological changes during transition from desiccated to hydrated phases of Semibarbula orientalis (moss) and Riccia gangetica (Liverwort). In dehydrated state, resurrection plants enter in quiescent phase and stop all molecular, biochemical and physiological processes until the following rehydration. Biochemical (chl content, proline accumulation, expression of antioxidant enzymes) and biophysical (fluorescence values, density of active reaction centers, specific and phenomenological fluxes, and performance of PSII) studies were done to unravel the mechanism of reactivation during the transition from desiccation-to-hydration. In both bryophytes, SOD, CAT and POD remained in inactive form in the fully desiccated state. In contrast, upon the availability of water, rapid upregulation in the activity of antioxidant enzymes was observed in S. oreintalis as compared with R. gangetica. Even upto 15 min of rehydration, significant accumulation of proline reflects its role to prevent hypo-osmotic shock in S. oreintalis. Rehydration quickly activated photosynthetic machinery in S. oreintalis than R. gangetica. The results clearly reflect that S. oreintalis have more efficient survival strategies at biochemical and physiological levels as compared with R. gangetica.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Water scarcity is one of the greatest dilemmas which greatly affect the distribution of plants across the globe. The frequent and longer drought episodes due to global climate change negatively influence the ecosystem and its components. Bryophytes, the first land plants, are believed to be the richest group with resurrection abilities (de León and Montesano 2013). They grow at diverse environmental places such as tree trunks, walls, rock faces, hills, and even at extreme hot and cold regions (Dey and De 2012). The absence of well define vascular system, epidermis, thick cuticle and small size make the plants suitable for experimentation to unravel the mechanism of various abiotic stresses (Onele et al. 2018). During the entire life cycle, these plants encounter numerous dehydration and rehydration cycles, which are regulated by several molecular, biochemical and physiological pathways (Proctor et al. 2007).
Due to the severity of drought, plants deal with many complications such as overproduction of reactive oxygen species (ROS), ion toxicity, osmotic stress, protein degradation, enzyme inactivation, nutrition imbalance, membrane disintegration, bleaching in pigments, DNA damage, and ultimately cell death (Todaka et al. 2015; Cruz de Carvalho et al. 2017; Forni et al. 2017; Zhanassova et al. 2021). Oxidative damage is one of the most deleterious results of desiccation (Mundree et al. 2000; Mihailova et al. 2022). To ameliorate these impediments, resurrection plants are well equipped with enzymatic and non-enzymatic antioxidants such as SOD (superoxide dismutase), POD (peroxidase), CAT (catalase), proline etc. SOD is a metalloenzyme and a vital component of the ROS scavenging system. It converts superoxides into molecular oxygen and H2O2 (Scandalios 1993). To abate the toxic effect of H2O2 produced, dismutation is also a major step to prevent enzymes of the Calvin cycle (Kaiser 1979). At this time POD and CAT play a major role to control H2O2 level in cells. The activity of antioxidants is also associated with increased concentration of proline (Ashraf and Foolad, 2007), which significantly act as osmoregulatory and ROS scavengers (Szabados and Savouré 2010; Ghosh et al. 2022). Accumulation of proline is a positive sign of peak desiccation tolerance among plants (Verslues et al. 2014). The relative water content (RWC) represents the level of cellular water which indicates the physiological status of water during dehydration and rehydration (Sánchez-Rodríguez et al. 2010).
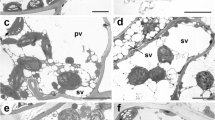
The chlorophyll a fluorescence (chlF) is an impactful technique to study various physiological responses of plants during water reactivation of the desiccated thalli (Schlensog and Schroeter 2001; Kalaji et al. 2014).The quick rehydration induces a metabolic imbalance in the resurrection plant which activates the antioxidant system to protect cells from oxidative damage (Rakić et al. 2014). Desiccation alters the primary photochemistry of PS-II, Fv/Fm and performance indexes (Bhatt et al. 2019; Soni 2020). In contrary to this, the bryophytes shows quick physiological activation after rewatering. To unravel these protective strategies in bryophytes, two different clades were taken for the experiment. Liverworts are considered as an earliest-divergent clade of land plants, which are characterized by the presence of oil bodies, polar organizers, elaters and lacking stomata, (Brown and Lemmon 2011; Crandall-Stotler and Stotler 2012; Duckett and Ligrone 1995; Flores et al. 2018, 2020; Ligrone et al. 2012). Vascular and mechanical tissues are also absent in the seta of sporophytes of liverworts. On the other hand in phylogenetics, mosses are the more advanced sister group to a clade that existed between hornworts and tracheophytes, having septate and multicellular rhizoids, filamentous protonemata, a spiral leafy mature gametophyte and the presence of stomata (Chang and Graham 2011; Harris et al. 2020; Ligrone et al. 2012; Merced and Renzaglia 2017; Qiu, 2008; Qiu et al. 2007). In the present studies, comparative analyses were done to understand the biochemical and physiological changes during watering of desiccated thalli of S. orientalis (Fig. 1) and R. gangetica (Fig. 2).
Material and method
Plants material and culture conditions
Well hydrated rosettes of moss S. orientalis and Liverwort R. gangetica were collected from the 3 locations of forest area near Udaipur city, India (24.6367° N, 73.6801° E) during July 2019. The 5 large size patches of each plants were collected with the upper crusts of soil by cluster sampling method and maintained in the laboratory for 2 weeks under 12/12 light and dark conditions at 65–70% relative humidity. After 2 weeks, plants were subjected to desiccation by ceasing water supply for 1 month. Thereafter, biochemical and physiological analyses were done to understand the changes during transition from desiccation to rehydration phases in both thalli. Desiccated thalli were rehydrated by spraying water and measurements were conducted during 0 (fully desiccated), 05, 15, 30, 60, 90, 120, 150 min.
Relative water content (RWC)
RWC is measured at an assortment of times to perceive plant water status. For this purpose, 1 g of both plant samples i.e. R. gangetica and S. orientalis were collected to determine RWC as per the following formula (Turner 1981)
where FW is fresh weight at the time of sample collection, DW is dry weight and TW is the turgid weight of plant samples.
Chlorophyll contents
Arnon (1949) method was used to determine the total chlorophyll content in R. gangetica and moss S. orientalis by crushing fresh leaves in 80% acetone. The extract was collected in a 3ml vial and cold centrifuged (4 °C) at 5000 rpm for 5 min. The supernatant was analyzed through spectrophotometer at 663 and 645nm against blank acetone for measurements of chl.a and b respectively. The total chlorophyll content was calculated by using absorbance taken with the help of the following formula (Arnon 1949)
where A is the absorbance at 645 and 663 nm.
H2O2 quantification
H2O2 content was determined by method described by Velikova et al. (2000). 1 g of samples was homoginated in 0.1% TCA and cold centrifuged (4 °C) at 10,000 rpm for 10 min. 75 μl TCA, 0.5 M KI and 5 mM K2HPO4 were added to form reaction mixture. Samples were kept at 25 °C till 20 min. and the optical density was noted at 390 nm. Reaction mixture without KI is used as a blank. The peroxide content was calculated with help of standard curve of pure H2O2 standards, whose absorbance was measured at the same wavelength.
SOD assay
The SOD (EC 1.15.1.1) was assayed by Kono (1978) method. The experimental plant samples of R. gangetica and S. orientalis were homogenized with 50 mM phosphate buffer (pH 7.0) and cold centrifuged at 10,000 rpm for 10 min. The supernatant was mixed with 50 mM potassium buffer (pH 10), 500 µl p-nitrobluetetrazolium chloride (NBT), 100 µl Triton and 100 µl hydroxylamine-HCl to form a reaction mixture. The absorbance was recorded at 540 nm by spectrophotometer and the specific activity was expressed in terms of unit ml−1g−1 protein.
POD assay
POD (EC 1.11.1.7) activities were measured by assessing the mixture containing 2 ml of 0.05 mM phosphate buffer (pH 7.0) and 1.0 ml guaiacol (1%) and enzyme extract prepared by samples of both plants R. gangetica and moss S. orientalis at various time intervals. 0.2 ml of 0.3% H2O2 was instantly mixed to initiate the reaction. The absorbance was taken at 470 nm for every 15 s up to 3 min and the data were expressed in terms of absorbance s−1 mg−1 protein according to Racusen and Foote (1965) method.
CAT assay
CAT (EC 1.11.1.6) activity was estimated by Teranishi et al. (1974) method during rewatering of desiccated plants of R. gangetica and S. orientalis from each variety and treatment. The reaction mixture was prepared of 50 mM potassium buffer (pH 7.0), 20 mM peroxide, and 0.1 ml enzyme solutions. At last, addition of 2 ml of Titanium reagent blocked the reaction and further, it was centrifuged at 10,000 rpm for 10 min. The optical density was observed at 410 nm and the Catalase activity was expressed as unit g−1 FW
Proline estimation
For determination of proline, fresh thallus of both plants were harvested and homogenated in 10 ml of 3% sulphosylicylic acid and then filtered. The filtered solution was mixed with an acidic nin-hydrin solution, and treated with 100 °C for 60 min in a water bath. The reaction was ended by incubating the mixture in ice for 5 min. Absorbance was taken at 520 nm after the addition of toluene. The proline content in tissues of thallus was calculated from a standard curve of proline by using Bates et al. (1973) method.
Chlorophyll fluorescence analysis
The O–J–I–P transients were measured in the dark chamber by using the Plant Efficiency Analyzer, PEA (Hansatech Instruments, Kings Lynn, UK). Plant materials were fixed in leaf clip of 4mm diameter and exposed to the red light of 3000 µmol m−2 s−1 by using three high-intensity light-emitting diodes. The fluorescence was measured after 0, 05, 15, 30, 60, 90, 120, 150 minutes of re-watering and analyzed according to the JIP-test measurements as per the formula (Strasser and Strasser, 1995; Tsimilli-Michael and Strasser, 2001)
Statistical analysis
All experiments were conducted in triplicates for both plants R. gangetica and S. orientalis. The three mean values of all experiments were compared at p < 0.05 by using One-way analysis of variance (ANOVA) and after obtaining significance differences the two groups were compared by bonferroni correction method at p < 0.05 by using M.S. Excel.
Results and discussion
Water is one of the most abundant and vital compounds on earth, which is required for the survival of all living organisms. However, there are few organisms which belongs to bryophytes, lichens and angiosperms that have the potential to stay alive after complete loss of water content. The process of anhydrobiosis is evolved when plants were migrated from aquatic to land regions during the evolution. During dehydrated state, resurrection organisms enter a quiescent phase, and stop all molecular, biochemical and physiological processes until they follow rehydration. Bryophytes, commonly known as the amphibians of the plant kingdom, developed the remarkable ability to grow at terrestrial habitats through developing various survival strategies from molecular to physiological levels. In the present work, comparative studies were performed between desiccated thalli of R. gangetica (liverwort) and S. orientalis (moss) to analyze the biochemical and biophysical strategies required for reactivation upon watering.
The effect of water stress on relative water content (RWC) has been investigated by several researchers. Under severe drought stress, some specific plants encountered irreversible damage when RWC drops below 20%. In the present study, fully desiccated thalli of resurrection bryophytes S. orientalis and R. gangetica showed 0% RWC which rapidly increased after watering (Fig. 3a). The RWC reached upto 82.3% and 79.12% in 150 min after rewatering S. oreintalis and R. gangetica, respectively (Fig. 3a). The RWC reached up to the control level in both the plants within 150 min. Low RWC of plants allied with falling in water potential and accumulation of some organic solutes such as glucose, sucrose, glucosinolates, proline and some other osmoprotectants which play a major role to retain the metabolic activities of plants (Mattson and Haack 1987; Verslues et al. 2006; Wanjiku and Bohne 2017). During desiccation, slow growth and photosynthetic shutdown reduce their metabolic requirements to adjust the cellular, metabolic and physiological status of vegetative tissues in resurrection plants (Challabathula et al. 2016). Coupling of the photosynthetic shutdown with other metabolic adjustments minimize damage and also protect photosynthetic machinery in resurrection plants (Farrant et al. 2015; Urano et al. 2017), but due to this premature photosynthesis inhibition plants faces energy deficiency which is maintained by using alternate reserve energy sources or by reducing energy expenditure (Asami et al. 2018) which may protect plants from irreversible damage. The speedy recovery in RWC is the indication of fast water retention capacity which helps S. orientalis and R. gangetica both plants to survive under desiccation.
In bryophytes with daily cycles of dehydration/rehydration, neoxanthin stabilizes the structure of chls by binding with light-harvesting chlorophyll protein (LHCP). The present study showed, rewatering markedly enhanced the chl contents in both bryophytes. After 150 min of rewatering, the chlorophyll concentration reached up to 2.70 mg/g FW and 2.45 mg/g FW in S. oreintalis and R. gangetica, respectively (Fig. 3b). The chl content was only 1.28% and 0.68% lower to the control level after 150 min of rehydration in moss and R. gangetica, respectively. Although, the chl reformation was much faster in moss(Fig. 3b), this water- induced rapid increment in chl contents shows the poikilochlorophyllous activation by the rapid re-synthesis of inactive chl molecules into active forms (Bewley 1979; Csintalan et al. 1996).
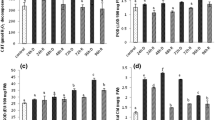
Formation of ROS during respiration and photosynthesis in mitochondria, peroxisomes and chloroplasts results in 'oxidative burst' (You and Chan 2015). A light mediated overexpression of chl. molecules are the main cause of excess ROS production during desiccated state (Apel and Hirt 2004; Blokhina et al. 2003; Fryer et al. 2003; Wang et al. 2010). The H2O2 concentration was highest in fully desiccated thalii (Fig. 4) of both the plants. The peroxide concentration was decreased with increasing time after re-watering, however, a wide variation in ROS scavenging enzymes was observed during the transition of desiccation to a fully hydrated state in both bryophytes. It was observed that SOD, CAT and POD remained inactive during the fully desiccated phase and became active upon the availability of water. A progressive reduction was observed in the H2O2 content just after 15 min of rewatering in S. orientalis and R. gangetica due to the higher activity of ROS-scavenging enzymes (Fig. 4).
The mechanism of desiccation tolerance and reactivation of resurrection plants is a typical phenomenon, dominantly dependent on rehydration-based cellular repairing (Oliver et al. 2004; Oliver and Derek Bewley 2010). At the time of rehydration, plants show oscillatory, fast, vulnerable and unstable recovery which induced maximum antioxidant action for defending cells from ROS, these antioxidant activities are the clear indication of metabolic imbalance (Rakić et al. 2014; Mansoor et al. 2022) in both plants during reactivations. Upon watering, the activity of SOD and CAT initially increased and thereafter slightly declined in S. oreintalis (Fig. 5a, b). The highest SOD and CAT activity was observed after 15 min of watering (Fig. 5a, b), the higher concentration of the H2O2 enhanced the CAT activity in R. gangetica. Initial increment in the activities of SOD and CAT during moderate rehydration is a consequence of shifting in redox potential by increased production of ROS (Sgherri et al. 2004; Jovanović et al. 2011). With the course of time, the SOD and CAT activities decreased up to the control level in S. orientalis (Fig. 5a, b), and due to the significant drop in peroxide concentration similar results were obtained in Syzygium maire (van der Walt et al. 2022). But the SOD activity slight enhanced at 60 min, therefore the amount of H2O2 was higher in R. gangetica as compare to the moss. Peroxidases are capable of eliminating the access level of H2O2 formed during non-enzymatic or enzymatic dismutation which also plays a vital role in cell wall remodeling (Rakić et al. 2014). Therefore, changes in peroxidase activity have been frequently correlated to the response of resistance or susceptibility of organisms to environmental stresses. A temporary electrolytic leakage through the plasma membrane is coordinated with high activities of SOD, CAT and POD during the initial state of rehydration (Sgherri et al. 2004; Jovanović et al. 2011). In the present study, the availability of water continuously declined the activity of POD in S. oreintalis (Fig. 5c). SOD and CAT activity continuously declined after the watering process in R. gangetica (Fig. 5a, b). On the other hand, expression of POD initially increased up to 15 min and thereafter declined with increasing watering period in R. gangetica (Fig. 5c). The lowering POD activities may be due to the higher oxidation within ascorbate–glutathione cycle (Veljovic-Jovanovic et al. 2006; Rakić et al. 2014).
Proline is the most important osmolyte which allocates cells to hold more water and also act as a metal chelator, antioxidant and signaling molecule which contributes to osmoregulation in plants under various types of biotic and abiotic stresses to stabilize subcellular complexes and redox buffering (Anwar Hossain et al. 2014; Ashraf and Foolad 2007; De Ronde et al. 2004; Gururani et al. 2015; Parvaiz and Satyawati, 2008; Wang et al. 2015). Therefore, proline accumulation is the positive sign of desiccation tolerance. In the present investigation, control plants contain very low amount of proline in both the plants which was evidently increased in desiccated thalii of S. orientalis and R. gangetica. Proline concentration was higher in desiccated thalii of R. gangetica as compare to S. orientalis. But it was slightly increased after 15 min of watering and then continuously decreased in S. oreintalis (Fig. 5d). After a long term, desiccating plants quickly rehydrated, this initial phase generate metabolic imbalance and plants faces hypo-osmotic shock during hydration (Zhou et al. 2022). To prevent plant from this shock, proline concentration enhanced after 15 min of watering in the moss, which is great sign of adaptation to cope the stress conditions. In contrast, proline content rapidly dropped after watering in R. gangetica (Fig. 5d) and finally reached up to the control level. These data collaborated with previous research works, that the proline accumulation is directly proportional to the ability of drought tolerance of plants (Efeoǧlu et al. 2009). Our results showed that the S. orientalis has advanced strategies to deals with the osmotic stress by a slow drop of proline during rewatering.
Water-induced quick reactivation of photosynthetic apparatus has been observed in many plant species (Heber et al. 2011; Soni 2020). Upon watering, maximal fluorescence (Fm) increased several folds in both the thalli and mimic to the control level, however it was higher in S. oreintalis than R. gangetica (Fig. 6a). The comparative rapid increase of Fm in S. oreintalis indicates that the watering process quickly converts inactive chl into active chl molecules (Fig. 6b). Similarly the concentration of active PSII reaction centers (RC/CS) markedly enhanced in S. oreintalis as compared to R. gangetica (Fig. 6b). In hydrated thalii of R. gangetica, the numbers of active RC/CS were unable to touch the control level even after 150 min. Results indicate that PSII reaction centers remained in their inactive form during the desiccation state of both bryophytes. Traces of water transformed inactive forms of reactions centers into an active one.
Upon watering, phenomenological fluxes [ABS/CS (absorbance per cross section) and ET/CS (electron transfer per cross section)] remarkably increased in S. oreintalis (Table 1). The rapid increase of ABS/CS and ET/CS in S. oreintalis is due to the increased amount of chl molecules and functional light-harvesting complexes (LHCs). Despite the rapid increase in chl molecules, no significant variations in ABS/CS and ET/CS were observed in R. gangetica (Table 1), which may be due to the lack of functional LHCs. At the beginning of rehydration-fast recovery, zeaxanthin plays a crucial role in non-radiative energy dissipation when ET/CS lower in R. gangetica energy dissipated in the form of heat which must be recognized by the de-epoxidation state of the xanthophyll cycle and non-photochemical quenching (Augusti et al. 2001).
Performance indexes (PI) introduced by Strasser et al. (2004), provides in-depth information about the overall photosynthetic potential of PSI and PSII. The watering process significantly increased the PI per cross section (PIcs) in S. oreintalis (Fig. 6c). In contrary to this, PIcs slightly increased after 15 min of watering and thereafter remained constant due to the ceased ET/CS till 150 min and unable to recover in R. gangetica (Fig. 6c) which shows higher reactivation potential of moss.
Conclusion
Our study focused on the biochemical and physiological reactivation strategies of desiccated bryophytes. The increased ROS concentration in fully desiccated plants shows the higher intensity of stress. Meanwhile, chlorophyll and RWC enhanced after re-watering both the plants. The retardation in H2O2 concentration was supported by the activity of antioxidants (SOD, CAT and POD). The activity of antioxidants enhanced when H2O2 was higher and it reshuffled up to the control level after rehydration. All reaction centers become active after 150 min of hydration in S. orientalis therefore, the Fm reached up to the control level while the R. gangetica takes more time to be active physiologically. It has also been shown by comparative analyses of PIcs of both the plants. The biochemical (chl content, proline accumulation, expression of antioxidant enzymes) and biophysical (fluorescence values, density of active reaction centers, ABS/CS, ET/CS, performance of PSII and PSI) studies during the desiccation-to-hydrated state demonstrate that S. oreintalis has developed high potential to grow at land habitats during the evolution as compared with R. gangetica.
References
Anwar Hossain M, Hoque MA, Burritt DJ, Fujita M (2014) Proline protects plants against abiotic oxidative stress: biochemical and molecular mechanisms. In: oxidative damage to plants: antioxidant networks and signaling
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in beta vulgaris. Plant Physiol. https://doi.org/10.1104/pp.24.1.1
Asami P, Mundree S, Williams B (2018) Saving for a rainy day: control of energy needs in resurrection plants. Plant Sci 271:62–66
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot. https://doi.org/10.1016/j.envexpbot.2005.12.006
Augusti A, Scartazza A, Navari-Izzo F et al (2001) Photosystem II photochemical efficiency, zeaxanthin and antioxidant contents in the poikilohydric Ramonda serbica during dehydration and rehydration. Photosynth Res. https://doi.org/10.1023/A:1010692632408
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil. https://doi.org/10.1007/BF00018060
Bewley JD (1979) Physiological aspects of desiccation tolerance. Annu Rev Plant Physiol. https://doi.org/10.1146/annurev.pp.30.060179.001211
Bhatt U, Singh H, Kumar D, Soni V (2019) Rehydration induces quick recovery of photosynthesis in desiccation tolerant moss Semibarbula orientalis. J Plant Sci Res 35
Blokhina O, Virolainen E, Fagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot 91:179–194
Brown RC, Lemmon BE (2011) Dividing without centrioles: Innovative plant microtubule organizing centres organize mitotic spindles in bryophytes, the earliest extant lineages of land plants. AoB Plants. https://doi.org/10.1093/aobpla/plr028
Challabathula D, Puthur JT, Bartels D (2016) Surviving metabolic arrest: photosynthesis during desiccation and rehydration in resurrection plants. Ann N Y Acad Sci. https://doi.org/10.1111/nyas.12884
Chang Y, Graham SW (2011) Inferring the higher-order phylogeny of mosses (Bryophyta) and relatives using a large, multigene plastid data set. Am J Bot. https://doi.org/10.3732/ajb.0900384
Crandall-Stotler B, Stotler RE (2012) Morphology and classification of the Marchantiophyta. In: Bryophyte biology
Cruz de Carvalho R, Catalá M, Branquinho C et al (2017) Dehydration rate determines the degree of membrane damage and desiccation tolerance in bryophytes. Physiol Plant. https://doi.org/10.1111/ppl.12511
Csintalan Z, Tuba Z, Lichtenthaler HK, Grace J (1996) Reconstitution of photosynthesis upon rehydration in the desiccated leaves of the Poikilochlorophyllous shrub Xerophyta scabrida at elevated CO2. J Plant Physiol. https://doi.org/10.1016/S0176-1617(96)80263-X
de León IP, Montesano M (2013) Activation of defense mechanisms against pathogens in mosses and flowering plants. Int J Mol Sci 14:3178–3200
De Ronde JA, Cress WA, Krüger GHJ et al (2004) Photosynthetic response of transgenic soybean plants, containing an Arabidopsis P5CR gene, during heat and drought stress. J Plant Physiol. https://doi.org/10.1016/j.jplph.2004.01.014
Dey A, De JN (2012) Antioxidative potential of bryophytes: stress tolerance and commercial perspectives: a review. Pharmacologia. https://doi.org/10.5567/pharmacologia.2012.151.159
Duckett JG, Ligrone R (1995) The formation of catenate foliar gemmae and the origin of oil bodies in the liverwort odontoschisma denudatum (Mart.) dum. (Jungermanniales): a light and electron microscope study. Ann Bot. https://doi.org/10.1006/anbo.1995.1114
Efeoǧlu B, Ekmekçi Y, Çiçek N (2009) Physiological responses of three maize cultivars to drought stress and recovery. S Afr J Bot. https://doi.org/10.1016/j.sajb.2008.06.005
Farrant JM, Cooper K, Hilgart A et al (2015) A molecular physiological review of vegetative desiccation tolerance in the resurrection plant Xerophyta viscosa (Baker). Planta 242:407–426
Flores JR, Catalano SA, Muñoz J, Suárez GM (2018) Combined phylogenetic analysis of the subclass Marchantiidae (Marchantiophyta): towards a robustly diagnosed classification. Cladistics. https://doi.org/10.1111/cla.12225
Flores JR, Suárez GM, Hyvönen J (2020) Reassessing the role of morphology in bryophyte phylogenetics: combined data improves phylogenetic inference despite character conflict. Mol Phylogenet Evol. https://doi.org/10.1016/j.ympev.2019.106662
Forni C, Duca D, Glick BR (2017) Mechanisms of plant response to salt and drought stress and their alteration by rhizobacteria. Plant Soil. https://doi.org/10.1007/s11104-016-3007-x
Fryer MJ, Ball L, Oxborough K et al (2003) Control of ascorbate peroxidase 2 expression by hydrogen peroxide and leaf water status during excess light stress reveals a functional organisation of Arabidopsis leaves. Plant J. https://doi.org/10.1046/j.1365-313X.2003.01656.x
Ghosh UK, Islam MN, Siddiqui MN et al (2022) Proline, a multifaceted signalling molecule in plant responses to abiotic stress: understanding the physiological mechanisms. Plant Biol 24:227–239
Gururani MA, Venkatesh J, Ganesan M et al (2015) In Vivo assessment of cold tolerance through chlorophyll-a fluorescence in transgenic zoysiagrass expressing mutant phytochrome A. PLoS One 10(5):e0127200. https://doi.org/10.1371/journal.pone.0127200
Harris BJ, Harrison CJ, Hetherington AM, Williams TA (2020) Phylogenomic evidence for the monophyly of bryophytes and the reductive evolution of stomata. Curr Biol. https://doi.org/10.1016/j.cub.2020.03.048
Heber U, Soni V, Strasser RJ (2011) Photoprotection of reaction centers: thermal dissipation of absorbed light energy vs charge separation in lichens. Physiol Plant. https://doi.org/10.1111/j.1399-3054.2010.01417.x
Jovanović Ž, Rakić T, Stevanović B, Radović S (2011) Characterization of oxidative and antioxidative events during dehydration and rehydration of resurrection plant Ramonda nathaliae. Plant Growth Regul. https://doi.org/10.1007/s10725-011-9563-4
Kaiser WM (1979) Reversible inhibition of the calvin cycle and activation of oxidative pentose phosphate cycle in isolated intact chloroplasts by hydrogen peroxide. Planta. https://doi.org/10.1007/BF00388364
Kalaji HM, Schansker G, Ladle RJ et al (2014) Frequently asked questions about in vivo chlorophyll fluorescence: practical issues. Photosynth Res 122:121–158
Kono Y (1978) Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys. https://doi.org/10.1016/0003-9861(78)90479-4
Ligrone R, Duckett JG, Renzaglia KS (2012) Major transitions in the evolution of early land plants: a bryological perspective. Ann Bot 109:851–871
Mansoor S, Ali Wani O, Lone JK et al (2022) Reactive oxygen species in plants: from source to sink. Antioxidants 11:225
Mattson WJ, Haack RA (1987) The role of drought in outbreaks of plant-eating insects. Bioscience. https://doi.org/10.2307/1310365
Merced A, Renzaglia KS (2017) Structure, function and evolution of stomata from a bryological perspective. Bryophyt Divers Evol 39:7–20
Mihailova G, Vasileva I, Gigova L et al (2022) Antioxidant defense during recovery of resurrection plant Haberlea rhodopensis from drought-and freezing-induced desiccation. Plants 11:175
Mundree SG, Whittaker A, Thomson JA, Farrant JM (2000) An aldose reductase homolog from the resurrection plant Xerophyta viscosa baker. Planta. https://doi.org/10.1007/s004250000331
Oliver MJ, Dowd SE, Zaragoza J et al (2004) The rehydration transcriptome of the desiccation-tolerant bryophyte Tortula ruralis: transcript classification and analysis. BMC Genom. https://doi.org/10.1186/1471-2164-5-89
Oliver MJ, Derek Bewley J (2010) Desiccation-tolerance of plant tissues: a mechanistic overview. In: Horticultural reviews
Onele AO, Chasov A, Viktorova L et al (2018) Biochemical characterization of peroxidases from the moss Dicranum scoparium. S Afr J Bot. https://doi.org/10.1016/j.sajb.2018.08.014
Parvaiz A, Satyawati S (2008) Salt stress and phyto-biochemical responses of plants—a review. Plant Soil Environ 54:89
Proctor MCF, Oliver MJ, Wood AJ et al (2007) Desiccation-tolerance in bryophytes: a review. Bryologist 110:595–621
Qiu YL (2008) Phytogeny and evolution of charophytic algae and land plants. J Syst Evol. https://doi.org/10.3724/SP.J.1002.2008.08035
Qiu YL, Li B, Wang B et al (2007) A nonflowering land plant phylogeny inferred from nucleotide sequences of seven chloroplast, mitochondrial, and nuclear genes. Int J Plant Sci. https://doi.org/10.1086/513474
Racusen D, Foote M (1965) Protein synthesis in dark-grown bean leaves. Can J Bot. https://doi.org/10.1139/b65-091
Rakić T, Lazarević M, Jovanović ŽS et al (2014) Resurrection plants of the genus Ramonda: prospective survival strategies—unlock further capacity of adaptation, or embark on the path of evolution? Front Plant Sci 4:550
Sánchez-Rodríguez E, Mm R-W, Cervilla LM et al (2010) Genotypic differences in some physiological parameters symptomatic for oxidative stress under moderate drought in tomato plants. Plant Sci. https://doi.org/10.1016/j.plantsci.2009.10.001
Scandalios JG (1993) Oxygen stress and superoxide dismutases. Plant Physiol 101:7
Schlensog M, Schroeter B (2001) A new method for the accurate in situ monitoring of chlorophyll a fluorescence in lichens and bryophytes. Lichenol 33:443–452
Sgherri C, Stevanovic B, Navari-Izzo F (2004) Role of phenolics in the antioxidative status of the resurrection plant Ramonda serbica during dehydration and rehydration. Physiol Plant. https://doi.org/10.1111/j.1399-3054.2004.00428.x
Soni V (2020) Rehydration quickly assembles photosynthetic complexes in desiccation tolerant Riccia gangetica. Biomed J Sci Tech Res. https://doi.org/10.26717/bjstr.2020.30.004891
Strasser BJ, Strasser RJ (1995) Measuring fast fluorescence transients to address environmental questions: the JIP-test. In: Photosynthesis: from light to biosphere
Strasser RJ, Tsimilli-Michael M, Srivastava A (2004) Analysis of the chlorophyll a fluorescence transient. In: Papageorgiou GC, Govindjee (eds) Chlorophyll a fluorescence. Advances in photosynthesis and respiration, vol 19. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-3218-9_12
Szabados L, Savouré A (2010) Proline: a multifunctional amino acid. Trends Plant Sci 15:89–97
Teranishi Y, Tanaka A, Osumi M, Fukui S (1974) Catalase activities of hydrocarbon-utilizing candida yeasts. Agric Biol Chem. https://doi.org/10.1080/00021369.1974.10861301
Todaka D, Shinozaki K, Yamaguchi-Shinozaki K (2015) Recent advances in the dissection of drought-stress regulatory networks and strategies for development of drought-tolerant transgenic rice plants. Front Plant Sci 6:84
Tsimilli-Michael M, Strasser RJ (2001) Fingerprints of climate changes on the photosynthetic apparatus’ behaviour, monitored by the JIP-test. In: “Fingerprints” of climate change
Turner NC (1981) Techniques and experimental approaches for the measurement of plant water status. Plant Soil. https://doi.org/10.1007/BF02180062
Urano K, Maruyama K, Jikumaru Y et al (2017) Analysis of plant hormone profiles in response to moderate dehydration stress. Plant J. https://doi.org/10.1111/tpj.13460
van der Walt K, Burritt DJ, Nadarajan J (2022) Impacts of rapid desiccation on oxidative status, ultrastructure and physiological functions of Syzygium maire (Myrtaceae) zygotic embryos in preparation for cryopreservation. Plants 11:1056
Velikova V, Yordanov I, Edreva A (2000) Oxidative stress and some antioxidant systems in acid rain-treated bean plants protective role of exogenous polyamines. Plant Sci. https://doi.org/10.1016/S0168-9452(99)00197-1
Veljovic-Jovanovic S, Kukavica B, Stevanovic B, Navari-Izzo F (2006) Senescence- and drought-related changes in peroxidase and superoxide dismutase isoforms in leaves of Ramonda serbica. J Exp Bot 57:1759–1768
Verslues PE, Agarwal M, Katiyar-Agarwal S et al (2006) Methods and concepts in quantifying resistance to drought, salt and freezing, abiotic stresses that affect plant water status. Plant J. https://doi.org/10.1111/j.1365-313X.2005.02593.x
Verslues PE, Lasky JR, Juenger TE et al (2014) Genome-wide association mapping combined with reverse genetics identifies new effectors of low water potential-induced proline accumulation in Arabidopsis. Plant Physiol. https://doi.org/10.1104/pp.113.224014
Wang X, Chen S, Zhang H et al (2010) Desiccation tolerance mechanism in resurrection fern-ally Selaginella tamariscina revealed by physiological and proteomic analysis. J Proteome Res. https://doi.org/10.1021/pr100767k
Wang H, Tang X, Wang H, Shao HB (2015) Proline accumulation and metabolism-related genes expression profiles in Kosteletzkya virginica seedlings under salt stress. Front Plant Sci. https://doi.org/10.3389/fpls.2015.00792
Wanjiku JG, Bohne H (2017) Growth and drought responses of three Prunus spinosa L. ecotypes. Afr J Hort Sci 12:40–50
You J, Chan Z (2015) Ros regulation during abiotic stress responses in crop plants. Front Plant Sci 6:1092
Zhanassova K, Kurmanbayeva A, Gadilgereyeva B et al (2021) ROS status and antioxidant enzyme activities in response to combined temperature and drought stresses in barley. Acta Physiol Plant. https://doi.org/10.1007/s11738-021-03281-7
Zhou X, Zheng Y, Wang L et al (2022) SYP72 interacts with the mechanosensitive channel MSL8 to protect pollen from hypoosmotic shock during hydration. Nat Commun 13:1–14
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
UB and VS designed the whole work, UB perform the experiment and wrote the article. VS supervised the whole work and corrected the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bhatt, U., Soni, V. Study of biochemical and biophysical adjustments during transition from desiccation-to-fully-hydrated states in Riccia gangetica and Semibarbula orientalis. Vegetos 36, 550–558 (2023). https://doi.org/10.1007/s42535-022-00409-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42535-022-00409-z