Abstract
Entomopathogenic fungi (EPF) and essential oils (EOs) can show either positive or negative interactions when used for controlling insect pests. First, the insecticidal efficacy of EPF including Beauveria bassiana isolates Z1 and IRAN1395C, Lecanicillium lecanii, and Paecilomyces lilacinus was tested against adults of Tribolium castaneum using two methods (standard insect dip and wheat diet incorporation). Additionally, the toxicity of EOs from Trachyspermum ammi, Foeniculum vulgare, Eucalyptus globulus, Salvia mirzayanii, Majorana hortensis, and Thymus vulgaris was evaluated against adult T. castaneum. Thereafter, the effect of an LC25 concentration of F. vulgare (86.13 µl L−1), T. ammi (235.2 µl L−1), and E. globulus (111.33 µl L−1) EOs on mycelial growth, spore germination, and sporulation of the EPF was determined. In standard dip bioassay, the lowest LT50 of 10.4 days was induced by L. lecanii, while the wheat diet incorporation method resulted in LT50 values ranging between 13.1 and 15.2 days. The LC50 values for E. globulus, F. vulgare, and T. ammi were 162.3, 140.3, and 310 μl L−1 air against adults, respectively. The EOs examined showed strong inhibition of mycelial growth, conidial germination, and sporulation at sublethal concentrations. EOs of F. vulgare and T. ammi completely inhibited mycelial growth and sporulation of the tested EPF. Germination inhibition ranged from 100% in L. lecanii exposed to EO from F. vulgare to 52.3% in B. bassiana Z1 exposed to EO from T. ammi. Based on the results, although EOs and EPF are successful agents to control adults T. castaneum when used separately, it cannot be applied in combination because of the conflicting effect.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The red flour beetle, Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae), has a world-wide distribution and is among the most economically important pest species in stored products. T. castaneum has a broad range of food preferences and is particularly abundant in flour mills, grocery shops, and stored grains (Garcìa et al. 2005; Lu et al. 2010). Insecticides are important components of insect pest management programs for stored grains, mills, processing plants, and retail stores (Arthur and Subramanyam 2012). However, the frequent and continuous application of synthetic chemical insecticides has resulted in serious drawbacks on the environment, toxicity hazards on non-target organisms, and the development of resistance (Isman 2006; Daglish 2008; Watts and Williamson 2015). These concerns along with consumer demand for less toxic pest insect control products have pushed research toward more ecologically compatible bio-agents for insect pest management.
One possible approach for safer stored products pest insect control is the use of essential oils (EOs) that show no or minimal off target effects (Rajendran and Sriranjini 2008; Cosimi et al. 2009). Essential oils are generally complex mixtures of organic compounds rich in monoterpenes which cause insect death by suppressing acetylcholinesterase activity (Houghton et al. 2006; Bakkali et al. 2008). These botanicals may have different types of action such as fumigant activity (Ilboudo et al. 2010; Nennah and Ibrahim 2011), contact toxicity (Taghizadeh-Saroukolai 2010; Kim et al. 2011), repellency (Celar and Kos 2016; Caballero-Gallardo et al. 2012), and anti-feedant activity (Stefanazzi et al. 2011). They may also induce changes in biological parameters such as growth rate, reproduction, and lifespan (Papachristos and Stamopoulos 2002).
Insecticidal efficacy has been shown with some EOs against adult T. castaneum. Islam et al. (2009) reported that fumigation of coriander (Coriandrum sativum L.) with EO (0.08 µg ml−1) yields 100% mortality against larvae, pupae, and adults of T. castaneum at 96 h post-exposure, and 100% egg mortality with fumigation at a significantly higher level (20 µg ml−1). The fumigant and contact toxicities of EOs from 20 different Egyptian plants against adults of T. castaneum and their inhibitory effects on acetylcholinesterase and adenosine triphosphatases have also been studied by Abou-Taleb et al. (2016).
Cosmopolitan entomopathogenic fungi (EPF) with their diverse range of insecticidal activity are used for integrated pest management in agriculture (Duarte et al. 2016). The application of these pathogenic agents is one of the most promising alternatives to traditional synthetic chemical insecticides as they combine high efficacy, low mammalian toxicity, and natural biological origins (Moore et al. 2000). EPF such as Beauveria bassiana (Bals.-Criv.) Vuill. (Hypocreales: Cordycipitaceae), Lecanicillium (= Verticillium) lecanii (Zimm.) Zare & Gams (Hypocreales: Clavicipitaceae), and Purpureocillium lilacinum (Thom.) Luangsa-ard, Hou-braken, Hywel-Jones & Samson (Hypocreales: Ophiocordycipitaceae) are considered the most important EPF that have been used against a wide spectrum of insect pests (Ambethger 2009). These fungi have been shown to be pathogenic against adult T. castaneum in numerous laboratory trials. Golshan et al. (2014) tested nine isolates of B. bassiana against adult T. castaneum and showed that virulence is highly variable. Specifically, they showed that B. bassiana isolate IRAN 440C is the most virulent against T. castaneum, whereas isolate DEBI 014 showed the lowest median lethal time (LT50). In another study, Shafighi et al. (2014) tested B. bassiana and M. anisopliae alone or in combination with diatomaceous earth (DE) against T. castaneum, Rhyzopertha dominica (F.), and Oryzaephilus surinamensis (L.) They found that DE enhances the insecticidal efficacy of these EPF.
By combining plant EOs and EPF, enhanced insecticidal efficacy may be accomplished thereby minimizing reliance on synthetic pesticide control and decreasing the risks of environmental contamination. However, the use of incompatible botanical insecticides may inhibit the germination and vegetative growth of the fungal biocontrol agent and adversely affect the overall IPM program (Hirose et al. 2001). The main goal of the present study was, therefore, to evaluate the compatibility of EOs isolated from six reputed medicinal plants (Trachyspermum ammi (L.) Sprauge ex Turrill (Apiacae), Foeniculum vulgare Mill. (Apiaceae), Eucalyptus globulus Labill. (Myrtaceae), Salvia mirzayanii Rech. F. & Esfand (Lamiaceae), Majorana hortensis Moench. (Lamiaceae), and Thymus vulgaris L. (Lamiacae)) with the EPF L. lecanii, B. bassiana, and P. lilacinum. The effects of these EOs alone or in combination with EPF on the mortality of T. castaneum were determined. The findings of this study will allow more effective application of these insecticidal compounds in IPM programs for T. castaneum.
Materials and methods
Plant material and extraction of essential oils
Plant parts including leaves and twigs of S. mirzayanii, M. hortensis, T. vulgaris, and E. globulus as well as the fruit of F. vulgare and T. ammi were collected from research fields of the Faculty of Agriculture (29° 22′ N, 51° 10′ E), Persian Gulf University, Bushehr Province, Bushehr, Iran. Plant parts of S. mirzayanii, M. hortensis, T. vulgaris, F. vulgare and T. ammi were collected during April and March, and those of E. globulus were collected during August 2019. The collected species were identified by comparison with existing herbarium specimens at Persian Gulf University. Plant materials were washed with distilled water and then air dried in the shade at 27 ± 1 °C. Plant parts were subjected to hydro-distillation for 3 h using a Clevenger-type apparatus (Goldis Company, Iran). The obtained EOs were dried over anhydrous sodium sulfate and stored at 4 °C prior to use in bioassays (Sohrabi and Kohanmoo 2017).
Fungi
Four fungal species were used in this study. B. bassiana isolate Z1, P. lilacinum isolate Iran 1026, and L. lecanii isolate Iran 229 were extracted from Spodoptera exigua Hubner (Lepidoptera: Noctuidae) in Nazlu, Urmia, Iran by Dr. Youbert Ghosta (University of Urmia, Iran). B. bassiana isolate IRAN1395C, was obtained from the Institute of Iranian Plant Protection in Tehran, Iran. These fungal strains were chosen for this study on the basis of their laboratory efficacy against other stored product pests like the waxworm (Galleria mellonella L.) and date sap beetle (Carpophilus hemipterus L.) that are commonly found in Iran (Sohrabi et al. 2019; Jamali et al. 2017). The fungi were cultured on potato dextrose agar (PDA) for the mass production of the conidia. During conidial production, culture plates were incubated at 25 ± 1 °C and 16 h illumination per day. Conidia were harvested by surface scraping 14-day-old sporulating cultures using a sterile scalpel and placed in a glass bottle containing 0.02% polyoxyethylene sorbitan monolaurate (Tween 80™; Merck). Spore suspensions were stirred vigorously on a shaker at 10,000 rpm for 5 min before being filtered through one layer of sterile jaconet. The concentration of fungal conidia in the homogenous conidial suspension was determined using a Neubauer haemocytometer (Precicolor, HBG; Germany). The conidial viability of fungal isolates was determined after 24 h as described by Lane et al. (1988). For all bioassays, the average viability of the conidia was over 95%.
Insect rearing
Red flour beetle adults were obtained from a laboratory colony and reared on wheat flour and yeast (10:1; w/w), at 28 ± 1 °C in darkness. Mixed-age adults were used in the bioassays.
Pathogenicity of fungi on adults of T. castaneum
Adult T. castaneum were exposed to the fungi using one of two application methods (i) conidial suspension (i.e., standard insect dip method) and (ii) wheat inoculated with conidial suspension (i.e., wheat diet incorporation method). The initial bioassay was performed using the standard insect dip method (Anonymus 1990). In brief, fifteen adults of T. castaneum were dipped into spore suspensions of P. lilacinum, L. lecanii, B. bassiana Z1, or B. bassiana IRAN1395C (concentrations of 1.52, 2.94, 2.98, and 3.07 × 109 conidia ml−1 of water containing 0.02% (v/v) Tween 80, respectively) for l0 s. Control insects were submerged in sterile distilled water containing 0.02% (v/v) Tween 80. Following exposure to the conidia, the treated insects were placed into cylindrical plastic containers (40 mm in diameter and 52 mm in height) with one screened hole (10 mm diameter) on the top of the container for ventilation. The container contained 10 g of sterilized partially damaged wheat as a food source.
The second bioassay was performed based on Kavallieratos et al. (2006) with some modifications. In this method, 50 g of sterilized damaged wheat was sprayed with 2 ml of the above mentioned conidia concentrations using a hand sprayer of 2000 ml capacity. Spraying was performed in a tray, on which the appropriate amount of wheat grain was spread into a thin layer. The treated grain was left to dry for 24 h post-spraying at room temperature (25 ± 1 °C). After the conidia-treated grain was completely dry, 10-g aliquots were placed in the cylindrical containers. Three additional containers, containing wheat treated with distilled water containing 0.02% (v/v) Tween 80, were used as a control. Fifteen T. castaneum adults were transferred into each container. All of the containers were incubated at 26 ± 1 °C, 70 ± 5% RH and a photoperiod of 14:10 (L:D). Three replicates were used for each fungus, and each experiment was repeated three times. The number of dead and live larvae were counted for 14 d every other day. Dead adults in all treatments were removed and surface sterilized with 2.5% sodium hypochlorite for 3 min, washed twice with sterile distilled water, and then incubated into Petri dishes on moistened filter paper for 3–5 days. Adults with fungal sporulation were considered to have died from the fungal infection.
Fumigant toxicity of essential oils against T. castaneum
In order to examine the fumigant toxicity of the EOs, 15 adults of T. castaneum were released into a closed cylindrical container (40 mm diameter by 52 mm height) containing one of six EO concentrations (133.33 to 800 μl L−1) with three replicates for each concentration. In order to accomplish this, the desired concentration of each EO was applied on a 10-mm-diameter piece of Whatman No. 1 filter paper that was attached to the inner surface of the lid of the container. A filter paper disk without EO was placed in each control cylinder. The containers were sealed with parafilm, and mortality was determined after 24, 48, and 72 h from the commencement of the exposure. The bioassays were carried out at 26 ± 1 °C, 70 ± 5% RH, and a photoperiod of 14:10 (L: D).
Effect of essential oils on fungal mycelial growth, sporulation, and conidial germination of fungi
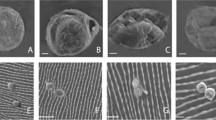
The antifungal properties of the most effective EOs against T. castaneum (determined from previous bioassay) were evaluated in terms of their volatile effects toward mycelial growth and sporulation as described by Soylu et al. (2007) and Nana et al. (2016) with some modifications. In brief, a conidial suspension (100 µl) containing 1 × 107 conidia ml−1 was spread on PDA plates (reference), and the plates were incubated at 25 ± 1 °C for 3 days in order to generate mycelial mats. The unsporulated mycelial mats were then cut into round agar plugs using a 5-mm-diameter cork borer. Subsequently, each agar plug was singly transferred onto the center of a fresh PDA agar plate. Petri plates (90 × 20 mm; Isolab, Iran) which provided 80 ml of air space after the addition of 20 ml agar medium were used for the determination of the volatile phase effect of EOs from E. globulus, T. ammi, and F. vulgare. An LC25 concentration (determined from previous experiments) was applied to a 10-mm-diameter disk of Whatman No. 1 filter paper and then placed on the inner surface of the inverted lid of the Petri dish. A filter paper disk that was not treated with EO was used as a control. Three Petri plates representing three replicates per treatment were used. The Petri dishes were sealed with parafilm and incubated in complete darkness at 25 ± 1 °C for 7 days. Radial growth of each fungus was recorded at seven days’ post-treatment. The experiment consisted of three replicates, and each experiment was repeated twice on different days.
To assess conidial production, the sporulated mycelial mats were cut from the culture plates into agar plugs using a 5-mm-diameter cork borer. Each agar plug was then transferred singly into a bottle containing 10 ml of sterile distilled water containing 0.02% sterile Tween 80. The bottle was then vortexed for 4 min, and the spore concentration was determined using a Neubauer haemocytometer. The experiment consisted of four replicates and was repeated two times on different days.
To study the effect of EOs on the conidial germination of fungi, 100 µl of conidial suspension (1 × 107 conidia ml−1) was spread on a water agar (0.9%) plate. An LC25 concentration of each EO was then added to a 10-mm-diameter disk of filter paper. Control plates were not treated with EOs. The plates were sealed with parafilm and incubated at 25 ± 1 °C in darkness. The percentage of germinated conidia was quantified at 24 h post-exposure to each EO. One hundred conidia were counted on a random basis for each Petri dish. Conidia were considered as germinated when the germ tube was longer than the conidial diameter (Marcuzzo and Eli 2016). Each treatment was replicated three times and repeated twice on different days.
Data analysis
Mortality data on the toxicity of fungi and EOs tested against T. castaneum adults were subjected to arcsin square root transformation before analysis. A 4 × 2 × 7 factorial analysis of variance (ANOVA) (SAS Institute 2003) was applied to study the possible effects of the two application methods on the percentage mortality data of T. castaneum adults exposed to EPF isolates at different times. The analysis of variance model included the main effects of each fungus, method of application, lethal time, and the interaction of the main effects. The time necessary to produce 50% mortality (LT50) was estimated by probit analysis (SAS Institute 2003). A 6 × 6 × 3 factorial ANOVA (SAS Institute 2003) was also applied to study the possible effects of the EOs on the percentage mortality data of T. castaneum adults exposed to different concentrations at various times. This analysis of variance model included the main effects of the EO, EO concentration, lethal time, and the interaction of the main effects. Lethal concentration values (LC25, LC50 and LC90) and their corresponding 95% fiducial limits (FL) for each essential oil were also estimated by probit analysis (SAS Institute 2003). The fungus–essential oil compatibility data were analyzed according to the classification scheme of Ambethgar et al. (2009). The replicated fungal radial growth, sporulation, and spore germination data were averaged and expressed as percentage of growth, percentage sporulation, and percentage conidial germination inhibition in comparison with the corresponding control. The percentage of inhibition (I) of mycelial growth/sporulation/conidial germination inhibition was determined using the formula of Hokkanen and Kotiluoto (1992):
where C and P are mycelial growth/sporulation/conidial germination of fungus in the control medium and medium with EO, respectively. Four inhibition levels were used to evaluate the effect of the EO on EPF (Ambethger et al. 2009): 1 = harmless (< 25%), 2 = slightly harmful (25–35%), 3 = moderately harmful (36—50%) and 4 = harmful (> 50%). This classification takes into account that inhibition higher than 50% is scarcely justifiable since biological control agents are generally not as effective as chemical pesticides (Celar and Kos 2016). An arcsine square root transformation was performed on the percentage of mycelial growth/sporulation/conidial germination inhibition data before analysis. A 2 × 2 factorial analysis of variance (ANOVA) (SAS Institute, 2003) was applied to study the possible effects of the EOs on the mycelial growth/sporulation/germination inhibition percentage of fungi exposed to EOs. This analysis of variance model included primary effects of the fungus, EO, and the interaction of the primary effects. Means were separated by the Duncan's Multiple Range Test (DMRT) (P = 0.05).
Results
Virulence of EPF on adult T. castaneum
Orthogonal contrasts revealed that mortality was significantly different among different times (F6, 466 = 4.26; P < 0.0003). The interactive effect between lethal time and application method used was also significant (F6, 466 = 5.14; P < 0.001).
In the first experiment using the standard dip method, the lowest LT50 value of 10.4 days was found with L. lecanii (Table 1), while in the wheat diet incorporation method the median lethal times ranged between 13 and 15 days, and no significant differences were found in lethal times between the EPF (Table 2). No significant difference in cumulative mortality was observed between treatments (Tables 1 and 2).
Fumigant toxicity bioassay
Orthogonal contrasts indicated that mortality was significantly different among the EOs (F5, 266 = 315.44; P < 0.001), concentrations applied (F5, 266 = 62.38; P < 0.001), and among different times (F2, 266 = 95.02; P < 0.001). The difference between the EOs and concentration applied for each EO was significantly different (F25, 266 = 15.51; P < 0.001). The interactive effect between each EO and lethal time was also significant (F10, 266 = 15.26; P < 0.001).
The LC25, LC50, and LC90 values of the EOs tested are summarized in Table 3. These values indicated that E. globulus and F. vulgare were approximately two times more effective than T. ammi against adult T. castaneum. Differences between the toxicity of the EOs from E. globulus and F. vulgare were not significant (Table 3). The median lethal concentrations (LC50) of EOs from E. globulus, F. vulgare, and T. ammi against adult T. castaneum at 72 h post-exposure were 162, 140, and 310 μl L−1 air, respectively (Table 3). The mortality of adult T. castaneum caused by EOs from S. mirzayanii, M. hortensis, and T. vulgaris at 72 h post-exposure was too low to compute LC50 values. Mortality rates of adults T. castaneum at the highest concentration tested (800 μl L−1) of these EOs were 13.33, 3.33, and 10.00%, respectively, at 72 h post-exposure.
Effects of EOs on inhibition of conidial germination, mycelial growth, and sporulation of EPF
Based on the results of the fumigant toxicity assays, EOs from E. globulus, T. ammi, and F. vulgare were chosen for further analysis of their effects on the germination of EPF. The germination inhibition percentage of the EPF tested in this study was significantly affected by fungal isolates (F3, 60 = 22.99; P < 0.001). The inhibitory effect of the EOs on the germination percentage of L. lecanii (96.7 ± 1.4) and B. bassiana IRAN1395C (92.9 ± 1.52) was significantly higher than that of P. lilacinum and B. bassiana Z1 (Table 4).
The growth inhibition percentage of fungal isolates was significantly affected by the EOs (F2, 60 = 84.62; P < 0.001). Mycelial growth inhibition ranged from 100% with EOs of F. vulgare and T. ammi to 82.1% with that of E. globulus (Table 4). Similarly, the sporulation of fungal isolates was significantly affected by the EOs (F2, 84 = 59.82; P < 0.001). Sporulation inhibition ranged from 100% with EOs of F. vulgare and T. ammi to 79.2% with that of E. globulus (Table 4).
Essential oils tested significantly inhibited mycelial growth, sporulation, and conidial germination of EPF, with all of them placed in the highest inhibition class 4 (Table 5). F. vulgare and T. ammi inhibited mycelial growth and sporulation entirely at concentrations equivalent to LC25 (100% inhibition), while E. globulus had a smaller effect on mycelial growth and sporulation ranging from 74.2–86.6% and 71.9–88% inhibition, respectively.
Essential oils used had more inhibitory effects on conidial germination of B. bassiana IRAN 1395C and L. lecanii (ranging from 91.3–100% inhibition) in comparison with B. bassiana Z1 and P. lilacinum (ranging from 52.3–81.7% inhibition).
Discussion
The findings of the two bioassay methods used in the current study show that the four EPF (B. bassiana isolates Z1 and IRAN1395C, L. lecanii Iran 229, and P. lilacinum Iran 1026) tested in this study have strong potential to control adults of T. castaneum. These entomopathogenic fungi can be considered as promising agents for use in biocontrol programs. The pathogenicity of EPF has been previously documented against T. castaneum and other stored product pests (Michalaki et al. 2007; Wakil et al. 2014; Storm et al. 2016; Ashraf et al. 2017; Dal Bello et al. 2018). The pathogenicity of L. lecanii and P. lilacinum against adults of T. castaneum has been evaluated for the first time in the present study, although susceptibility of other stored product pests including Sitophilus zeamais Motsch. (Coleoptera: Curculionidae) and Tribolium confusum Jacquelin du Val (Coleoptera: Tenebrionidae) to these fungi has been previously documented (Ahmed 2010; Barra et al. 2013). The efficacy of the L. lecanii IRAN 229 and B. bassiana Z1 isolates used in this study has recently been determined against several instar larvae of Galleria mellonella L. (Lepidoptera: Pyralidae) by Sohrabi et al. (2019). In the current study, we found that the LT50 value of P. lilacinum against T. castaneum was about 14 days with both bioassay methods that were tested. In another study, among 20 isolates of P. lilacinum that were tested, the shortest LT50 value was found to be 4.66 days when determined using three stored product pests including T. confusum (Barra et al. 2013). The differences in the LT50 values in our study and that of the other studies are likely due to different fungal isolates that were tested and/or differences in the virulence of the same isolates against various insect species.
In the present study, the toxicity of EOs obtained from six plant species was evaluated against T. castaneum; of these, EOs of E. globulus, F. vulgare, and T. ammi exhibited insecticidal activities. Insecticidal effects have been previously reported with EOs from various Eucalyptus spp. against major stored-grain insects including T. castaneum, Callosobruchus maculatus Fabr. (Coleoptera: Chrysomelidae), Rhyzopertha dominica (F.) (Coleoptera: Bostrychidae), and Sitophilus oryzae (L.) (Coleoptera: Curculionidae) (Lee et al. 2004; Negahban and Moharramipour 2007; Nattudurai et al. 2012; Siddique et al. 2017). The repellency and toxicity of EOs from T. ammi and F. vulgare against larvae and adults of T. castaneum have also been reported previously by other researchers (Chaubey 2007a, b; Khorrami et al. 2018). The insecticidal activity of constituents of EO of F. vulgare including (E)-anethole, estragole, and fenchone against some stored product pests is largely attributable to fumigant activity rather than contact activity as reported by Kim and Ahn (2001). Our results showing insecticidal effects of EOs from E. globules, F. vulgare, and T. ammi on T. castaneum are consistent with those reported by other researchers. The insecticidal components of many plant essential oils are mainly monoterpenoids and their toxicity on different pests has been reported in previous studies (Regnault-Roger and Hamraoui 1995; Ibrahim et al. 2001; Kim and Ahn 2001). In the current study, the efficacy of E. globulus essential oil against T. castaneum might be attributed to its major component 1, 8-cineole (78.7%) (Sohrabi et al. 2015), which exhibits insecticidal activities against several insects (Liska et al. 2011; Pant et al. 2014). The main active compounds found in the EOs of F. vulgare and T. ammi are E anethole (76.8%) (unpublished data) and thymol (38.97%) (Sohrabi and Kohanmoo 2017), respectively. The repellency and insecticidal properties of thymol and E anethole have been proved in previous studies (Kim and Ahn 2001; Pandey et al. 2009; Bedini et al. 2016).
In the current study, EOs from M. hortensis, S. mirzayanii, and T. vulgaris showed unsatisfactory toxicity against adults of T. castaneum. There was insufficient mortality even to compute LC25 values. Similarly, Mohamed et al. (2008, 2009) stated that the oil of M. hortensis displayed very strong toxic activity by contact assay, while it showed no toxic effects by fumigant assay.
The fumigant toxicity of T. vulgaris against adult T. castaneum was also investigated in this study, and the mortality rate was too low. In previous researches, T. castaneum adults showed no susceptibility to the EOs from other Thymus species (Karabörklü et al. 2010; Taghizadeh-Saroukolai et al. 2010). In the present study, the fumigant toxicity of EO of S. mirzayanii was evaluated for the first time against adults of T. castaneum, and the oil exhibited little mortal effects. Same results have also been previously observed against T. confusum even under the highest concentration applied by Nikooei and Moharramipour (2010).
Our current results indicated that EOs from E. globules, F. vulgare, and T. ammi exhibit fumigant toxicity against EPF. Sublethal concentrations of the EOs significantly inhibited all the growth parameters tested including spore germination, radial growth, and conidial yield of isolates of P. lilacinum, B. bassiana, and L. lecanii. The use of incompatible EOs may inhibit the development and reproduction of EPF resulting in negative effects on integrated pest management strategies. Since germination is the first step in the infection process, compatibility between plant EOs and fungal spore germination should be considered as the most important factor when considering the use of these compounds (Anderson and Roberts 1983). Thus, if germination inhibition occurs, the fungal control efficiency will be affected by the EOs (Hirose et al. 2001).
The negative impact of EO of E. globulus and other Eucalyptus species on EPF including B. bassiana has been previously reported by other researchers (Immediato et al. 2016; Nardoni et al. 2018). In the current study, the finding that E. globulus was toxic to B. bassiana might be attributed to its major component 1, 8-cineole (Sohrabi et al. 2015), an oxygenated monoterpene which exhibits lower antifungal properties than phenolic compounds (Safaei-Ghomi and Ahd 2010; Nardoni et al. 2018).
In vitro antifungal activities of EOs of T. ammi and F. vulgare have been reported against several non-pathogenic fungal species (Abou-Jawdah et al. 2002; Mimica-Dukić et al. 2003; Singh et al. 2004; Soylu et al. 2005, 2006, 2007; Moein et al. 2014). To the best of our knowledge, this study is the first to show susceptibility of EPF to EOs of T. ammi and F. vulgare. The antimicrobial properties of EOs of T. ammi and F. vulgare and their major constituents thymol and anethole, respectively, have been shown to be able to suppress several human and plant pathogenic fungi (Mimica-Dukić et al. 2003; Soylu et al. 2006, 2007; Kordali et al. 2008; Moein et al. 2014).
Combining EPF and plant EOs as natural biocontrol agents may lead to fewer negative side effects compared to the use of synthetic chemical insecticides. However, according to our results, the volatile phases of the EOs used in this study negatively affected all growth factors of B. bassiana, L. lecanii, and P. lilacinum even at very low concentrations. The volatile phase of EOs has been reported to possess higher antimicrobial activity against plant pathogenic fungi and bacteria (Edris and Farrag 2003; Soylu et al. 2006, 2007). This higher antimicrobial activity likely originates from the ability of the fungal mycelium to easily absorb the naturally lipophilic EOs that are found in the vapor phase (Inouye et al. 2000; Edris and Farrag 2003).
Our findings suggest that the essential oils of E. globulus, T. ammi, and F. vulgare and entomopathogenic fungi B. bassiana, L. lecanii, and P. lilacinum can be used separately as valuable tools to control adults of T. castaneum. These agents, however, were not compatible when used in combination. However, future studies need to evaluate effects of these agents in the field applications, when they are either used in a sequence or rotation, or as applied to mixed populations as would occur in nature.
Availability of data and material
The data and material will be available as needed.
References
Abou-Jawdah Y, Sobh H, Salameh A (2002) Antimycotic activities of selected plant flora, growing wild in Lebanon, against phytopathogenic fungi. J Agric Food Chem 50:3208–3213
Abou-Taleb HK, Mohamed MIE, Shawir MS, Abdelgaleil SAM (2016) Insecticidal properties of essential oils against Tribolium castaneum (Herbst) and their inhibitory effects on acetylcholinesterase and adenosine triphosphatases. Nat Prod Res 30(6):710–714
Ahmed BI (2010) Potentials of entomopathogenic fungi in controlling the menace of maize weevil Sitophilus zeamais Motsch (Coleoptera: Curculinidae) on stored maize grain. Arch Phytopathol Plant Protect 43(2):107–115
Ambethger V (2009) Potential of entomopathogenic fungi in insecticide resistance management (IRM): a review. J Biopestic 2(2):177–193
Anderson TE, Roberts DW (1983) Compatibility of Beauveria bassiana isolates with insecticide formulations used in Colorado potato beetle (Coleoptera: Chrysomelidae) control. J Econ Entomol 76:1437–1441
Anonymous (1990) EPPO Bull 20: 399–400
Arthur FH, Subramanyam Bh (2012) Chemical control in stored products. In: Hagstrum DW, Phillips TW, Cuperus G (eds) Stored Product Protection. Kansas State University, Manhattan, pp 95–100
Ashraf M, Farooq M, Shakeel M, Din N, Hussain S, Saeed N, Shakeel Q, Rajput NA (2017) Influence of entomopathogenic fungus, Metarhizium anisopliae, alone and in combination with diatomaceous earth and thiamethoxam on mortality, progeny production, mycosis, and sporulation of the stored grain insect pests. Environ Sci Pollut Res 24(36):28165–28174
Bakkali F, Averbeck S, Averbeck D, Idaomar M (2008) Biological effects of essential oils: a review. Food Chem Toxicol 46:446–475
Barra P, Rosso L, Etcheverry M (2013) Isolation and identification of entomopathogenic fungi and their evaluation against Tribolium confusum, Sitophilus zeamais, and Rhyzopertha dominica in stored maize. J Pest Sci 86:217–226
Bedini S, Bougherra HH, Flamini G, Cosci F, Belhamel K, Ascrizzi R, Conti B (2016) Repellency of anethole- and estragole-type fennel essential oils against stored grain pests: the different twins. Bull Insectol 69:149–157
Caballero-Gallardo K, Olivero- Verbel J, Stashenko EE (2012) Repellency and toxicity of essential oils from Cymbopogon martinii, Cymbopogon flexuosus and Lippia origanoides cultivated in Colombia against Tribolium castaneum. J Stored Prod Res 50:62–65
Celar FA, Kos K (2016) Effects of selected herbicides and fungicides on growth, sporulation and conidial germination of entomopathogenic fungus Beauveria bassiana. Pest Manag Sci 72(11):2110-2117
Chaubey MK (2007a) Insecticidal activity of Trachyspermum ammi (Umbelliferae), Anethum graveolens (Umbelliferae) and Nigella sativa (Ranunculaceae) essential oils against stored product beetle Tribolium castaneum Herbst (Coleoptera: Tenebrionidae). Afr J Agric Res 2(11):596–600
Chaubey MK (2007b) Toxicity of essential oils from Cuminum cyminum (Umbelliferae), Piper nigrum (Piperaceae) and Foeniculum vulgare (Umbelliferae) against stored-product beetle Tribolium castaneum Herbst (Coleoptera: Tenebrionidae). Electr J Environ Agric Food Chem 6:1719–1727
Cosimi S, Rossi E, Cioni PL, Canale A (2009) Bioactivity and qualitative analysis of some essential oils from Mediterranean plants against stored-product pests: evaluation of repellency against Sitophilus zeamais Motschulsky, Cryptolestes ferrugineus (Stephens) and Tenebrio molitor L. J Stored Prod Res 45:125–132
Daglish GJ (2008) Impact of resistance on the efficacy of binary combinations of spinosad, chlorpyrifos-methyl and s-methoprene against five stored-grain beetles. J Stored Prod Res 44:71–76
Dal Bello GM, Fuse CB, Pedrini N, Padin SB (2018) Insecticidal efficacy of Beauveria bassiana, diatomaceous earth and fenitrothion against Rhyzopertha dominica and Tribolium castaneum on stored wheat. Int J Pest Manage 64:279–286
Duarte R, Gonçalves K, Espinosa D, Moreira L, De Bortoli S, Humber R, Polanczyk R (2016) Potential of entomopathogenic fungi as biological control agents of diamondback moth (Lepidoptera: Plutellidae) and compatibility with chemical insecticides. J Econ Entomol 109:594–601
Edris AE, Farrag ES (2003) Antifungal activity of peppermint and sweet basil essential oils and their major aroma constituents on some plant pathogenic fungi from the vapour phase. Nahrung 47:117–121
Garcìa M, Donael OJ, Ardanaz CE, Tonn CE, Sosa ME (2005) Toxic and repellent effects of Baccharis salicifolia essential oil on Tribolium castaneum. Pest Manag Sci 61:612–618
Golshan H, Saber M, Majidi-Shilsar F, Karimi F, Ebadi AA (2014) Laboratory evaluation of Beauveria bassiana isolates on red flour beetle Tribolium castaneum and their characterization by random amplified polymorphic DNA. J Agr Sci Tech 16:747–758
Hirose E, Neves PMOJ, Zequi JAC, Martins LH, Peralta CH, Alcides M Jr (2001) Effect of biofertilizers and neem oil on the entomopathogenic fungi Beauveria bassiana (Bals.) vuill. and Metarhizium anisopliae (Metsch.) sorok. Braz Arch Biol Technol 44(4):419–423
Hokkanen HMT, Kotiluoto R (1992) Bioassay of the side effects of pesticides on Beauveria bassiana and Metarhizium anisopliae: standardized sequential testing procedure. IOBC-WPRS Bull 11(3):148–151
Houghton PJ, Ren Y, Howes M-J (2006) Acetylcholinesterase inhibitors from plants and fungi. Nat Prod Rep 23:181–199
Ibrahim MA, Kainulainen P, Aflatuni A, Tiilikkala K, Holopainen JK (2001) Insecticidal, repellent, antimicrobial activity and phytotoxicity of essential oils: with special reference to limonene and its suitability for control of insect pests. Agr Food Sci Finland 10:243–259
Ilboudo Z, Dabiré LC, Nébié RC, Dicko IO, Dugravot S, Cortesero AM, Sanon A (2010) Biological activity and persistence of four essential oils towards the main pest of stored cowpeas, Callosobruchus maculates (F.) (Coleoptera: Bruchidae). J Stored Prod Res 46:124–128
Immediato D, Figueredo LA, Iatta R, Camarda A, de Luna RLN, Giangaspero A, Brandão-Filho SP, Otranto D, Cafarchia C (2016) Essential oils and Beauveria bassiana against Dermanyssus gallinae (Acari: Dermanyssidae): towards new natural acaricides. Vet Parasitol 229:159–165
Inouye S, Tsuruoka T, Watanabe M, Takeo K, Akao M, Nishiyama Y, Yamaguchi H (2000) Inhibitory effect of essential oils on apical growth of Aspergillus fumigatus by vapour contact. Mycoses 43:17–23
Islam MS, MahbubHasan M, Xiong W, Zhang SC, Lei CL (2009) Fumigant and repellent activities of essential oil from Coriandrum sativum (L.) (Apiaceae) against red flour beetle Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). J Pestic Sci 82:171–177
Isman MB (2006) Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu Rev Entomol 51:45–66
Jamali F, Kohanmoo MA, Sohrabi F (2017) Lethal effect of Beauveria bassiana (Bals.) Vuill. and Metarhizium anisopliae (Metsch.) Sorokinin against larvae and adults of date sap beetle (Carpophilus hemipterus). J Appl Res Plant protect 6(2):93–105 (in Persian)
Karabörklü S, Ayvaz A, Yilmaz S (2010) Bioactivities of different essential oils against the adults of two stored product insects. Pak J Zool 42(6):679–686
Kavallieratos NG, Athanassiou CG, Michalaki MP, Batta YA, Rigatos HA, Pashalidou FG, Balotis GN, Tomanović Ž, Vayias BJ (2006) Effect of the combined use of Metarhizium anisopliae (Metschinkoff) Sorokin and diatomaceous earth for the control of three stored-product beetle species. Crop Prot 25:1087–1094
Kim D-H, Ahn Y-J (2001) Contact and fumigant activities of constituents of Foeniculum vulgare fruit against three coleopteran stored-product insects. Pest Manag Sci 57:301–306
Kim SI, Chae SH, Youn HS, Yeon SH, Ahn YJ (2011) Contact and fumigant toxicity of plant essential oils and efficacy of spray formulations containing the oils against B- and Q-biotypes of Bemisia tabaci. Pest Manag Sci 67:1093–1099
Khorrami F, Valizadegan O, Forouzan M, Soleymanzade A (2018) The antagonistic/synergistic effects of some medicinal plant essential oils, extracts and powders combined with Diatomaceous earth on red flour beetle, Tribolium castaneum Herbst (Coleoptera: Tenebrionidae). Arch Phytopathol Plant Protect 51(13–14):685–695
Kordali S, Cakir A, Ozer H, Cakmakci R, Kesdek M, Mete E (2008) Antifungal, phytotoxic and insecticidal properties of essential oil isolated from Turkish Origanum acutidens and its three components, carvacrol, thymol and p-cymene. Bioresour Technol 99:8788–8795
Lane BS, Humphreys AM, Thompson K, Trinci APJ (1988) ATP content of stored spores of Paecilomyces farinosus and the use of ATP as a criterion of spore viability. Trans Br Mycol Soc 90:109–148
Lee B-H, Annis PC, Tumaalii F, Choi W-C (2004) Fumigant toxicity of essential oils from the Myrtaceae family and 1,8-cineole against 3 major stored-grain insects. J Stored Prod Res 40:553–564
Liska A, Rozman I, Eded A, Mustac S, Perhoc B (2011) Bioactivity of 1,8-cineol against red four beetle, Tribolium castaneum (Herbst), Pupae. Poljoprivreda 17:58–63
Lu H, Zhou J, Xiong S, Zhao S (2010) Effects of low-intensity microwave radiation on Tribolium castaneum physiological and biochemical characteristics and survival. J Insect Physiol 56:1356–1361
Marcuzzo LL, Eli K (2016) Effect of temperature and photoperiod on the in vitro germination of conidia of Botrytis squamosa, the causal agent of Botrytis leaf blight of onion. Summa Phytopathol 42(3):261–263
Michalaki MP, Athanassiou CG, Steenberg T, Buchelos CTh (2007) Effect of Paecilomyces fumosoroseus (Wise) Brown and Smith (Ascomycota: Hypocreales) alone or in combination with diatomaceous earth against Tribolium confusum Jacquelin du Val (Coleoptera: Tenebrionidae) and Ephestia kuehniella Zeller (Lepidoptera: Pyralidae). Biol Control 40(2):280–286
Mimica-Dukić N, Kujundžić S, Soković M, Couladis M (2003) Essential oil composition and antifungal activity of Foeniculum vulgare Mill. obtained by different distillation conditions. Phytother Res 17:368–371
Moein MR, Zomorodian K, Pakshir K, Yavari F, Motamedi M, Zarshenas MM (2014) Trachyspermum ammi (L.) Sprague: chemical composition of essential oil and antimicrobial activities of respective fractions. Evid. based complement. Alternat Med 20(1):50–56
Mohamed MIE, Abdelgaleil SAM (2008) Chemical composition and insecticidal potential of essential oils from Egyptian plants against Sitophilus oryzae (L.) (Coleoptera: Curculionidae) and Tribolium castaneum (Herbst) (Coleoptera: Tenebrionidae). Appl Entomol Zool 43(4):599–607
Mohamed MIE, Abdelgaleil SAM, Abdel Rasoul MA (2009) Potential of essential oils to control Sitophilus oryzae (L.) and Tribolium castaneum (Herbst) on stored wheat. ASEG 30(4):419–426
Moore D, Lord JC, Smith SM (2000) Pathogens. In: Subramanyam Bh, Hagstrum DW (eds) Alternatives to pesticides in stored-product IPM. Kluwer Academic Publishers, Dordrecht, pp 193–227
Nana P, Ekesi S, Nchu F, Maniania NK (2016) Compatibility of Metarhizium anisopliae with Calpurnia aurea leaf extracts and virulence against Rhipicephalus pulchellus. J Appl Entomol 140(8):590–597
Nardoni S, Ebani VV, D’Ascenzi C, Pistelli L, Mancianti F (2018) Sensitivity of entomopathogenic fungi and bacteria to plants secondary metabolites, for an alternative control of Rhipicephalus (Boophilus) microplus in cattle. Front Pharmacol 9:937
Nattudurai G, Gabriel Paulraj M, Ignacimuthu S (2012) Fumigant toxicity of volatile synthetic compounds and natural oils against red flour beetle Tribolium castaneum (Herbst) (Coleopetera: Tenebrionidae). JKSUS 24:153–159
Negahban M, Moharramipour S (2007) Fumigant toxicity of Eucalyptus intertexta, Eucalyptus sargentii and Eucalyptus camaldulensis against stored-product beetles. J Appl Ent 131:256–261
Nenaah GE, Ibrahim SIA (2011) Chemical composition and the insecticidal activity of certain plants applied as powders and essential oils against two stored-products coleopteran beetles. J Pest Sci 84(3):393–402
Nikooei M, Moharramipour S (2010) Fumigant toxicity and repellency effects of essential oil of Salvia mirzayanii on Callosobruchus maculatus (Col.: Bruchidae) and Tribolium confusum (Col.: Tenebrionidae). J Entomol Soc Iran 30(2):17–30 (in Persian)
Pandey SK, Upadhyay S, Tripathi AK (2009) Insecticidal and repellent activities of thymol from the essential oil of Trachyspermum ammi (Linn) Sprague seeds against Anopheles stephensi. Parasitol Res 105:507–512
Pant M, Dubey S, Patanjali PK, Naik SN, Sharma S (2014) Insecticidal activity of eucalyptus oil nanoemulsion with karanja and jatropha aqueous filtrates. Int Bioiodeterior Biodegrad 91:119–127
Papachristos DP, Stamopoulos DC (2002) Repellent, toxic and reproduction inhibitory effects of essential oil vapours on Acanthoscelides obtectus (Say) Coleoptera: Bruchidae). J Stored Prod Res 38:117–128
Rajendran S, Sriranjini V (2008) Plant products as fumigants for stored-product insect control. J Stored Prod Res 44:126–135
Regnault-Roger C, Hamraoui A (1995) Fumigant toxic activity and reproductive inhibition induced by monoterpenes on Acanthoscelides obtectus (Say) (Coleoptera), a bruchid of kidney bean (Phaseolus vulgaris L). J Stored Prod Res 31:291–299
Safaei-Ghomi J, Ahd AA (2010) Antimicrobial and antifungal properties of the essential oil and methanol extracts of Eucalyptus largiflorens and Eucalyptus intertexta. Parmacogn mag 6(23):172–175
Saroukolai AT, Moharramipour S, Meshkatalsadat MH (2010) Insecticidal properties of Thymus persicus essential oil against Tribolium castaneum and Sitophilus oryzae. J Pest Sci 83:3–8
SAS Institute (2003) The SAS system for windows, Release 9.0. SAS, Institute, Cary, NC
Shafighi Y, Ziaee M, Ghosta Y (2014) Diatomaceous earth used against insect pests, applied alone or in combination with Metarhizium anisopliae and Beauveria bassiana. J Plant Prot Res 54(1):62–66
Siddique S, Parveen Z, Bareen F, Butt A, Chaudhary MN, Akram M (2017) Chemical composition and insecticidal activities of essential oils of Myrtaceae against Tribolium castaneum (Coleoptera: Tenebrionidae). Pol J Environ Stud 26(4):1653–1662
Singh G, Maurya S, Catalan C, De Lampasona MP (2004) Chemical constituents, antifungal and antioxidative effects of ajwain essential oil and its acetone extract. J Agric Food Chem 52:3292–3296
Sohrabi F, Jamali F, Morammazi S, Saber M, Kamita SG (2019) Evaluation of the compatibility of entomopathogenic fungi and two botanical insecticides tondexir and palizin for controlling Galleria mellonella L. (Lepidoptera: Pyralidae). Crop Prot 117:20–25
Sohrabi F, Kohanmoo MA (2017) Fumigant Toxicity of plant essential oils against Oligonychus afrasiaticus (MCG) (Acari: Tetranychidae) and identification of their chemical composition. J Essent Oil Bear Pl 20(11):1–7
Sohrabi F, Kohanmoo MA, Jamali F (2015) Fumigant toxicity of five medicinal plant essential oils against the date sap beetle, Carpophilus hemipterus (Linnaeus) and identification of their chemical composition. Plant Prot 39(3):13–26 (in Persian)
Soylu EM, Soylu S, Kurt Ş (2006) Antimicrobial activities of the essential oils of various plants against tomato late blight disease agent Phytophthora infestans. Mycopathologia 161:119–128
Soylu EM, Tok FM, Soylu S, Kaya AD, Evrendilek GA (2005) Antifungal activities of the essential oils on post-harvest disease agent Penicillium digitatum. Pak J Biol Sci 8:25–29
Soylu S, Yigitbas H, Kurt Ş (2007) Antifungal effects of essential oils from oregano and fennel on Sclerotinia sclerotiorum. J Appl Microbiol 103:1021–1030
Stefanazzi N, Stadler T, Ferrero A (2011) Composition and toxic, repellent and feeding deterrent activity of essential oils against the stored-grain pests Tribolium castaneum (Coleoptera: Tenebrionidae) and Sitophilus oryzae (Coleoptera: Curculionidae). Pest Manag Sci 67:639–646
Storm C, Scoates F, Nunn A, Potin O, Dillon A (2016) Improving efficacy of Beauveria bassiana against stored grain beetles with a synergistic co- formulant. Insects 7(3):42
Taghizadeh-Saroukolai A, Moharramipour S, Meshkatalsadat MH (2010) Insecticidal properties of Thymus persicus essential oil against Tribolium castaneum and Sitophilus oryzae. J Pest Sci 83:3–8
Wakil W, Ghazanfar MU, Yasin M (2014) Naturally occurring entomopathogenic fungi infecting stored grain insect species in Punjab. Pakistan J Insect Sci 14:182
Watts M, Williamson S (2015) Replacing chemicals with biology: phasing out highly hazardous pesticides with agroecology. PAN International, Fremont
Acknowledgements
The authors gratefully acknowledge Dr. Shizuo George Kamita for help with scientific editing and the college of Agriculture and Natural resources, Persian Gulf University, Bushehr, Iran, for the support in conducting the current study. The authors also acknowledge Dr. Youbert Ghosta (University of Urmia, Iran) for supplying fungal isolates.
Funding
This work was supported by Persian Gulf University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Consent to participate
Include appropriate statements.
Consent for publication
The authors are fully satisfied that the manuscript is published in the Journal of Plant Diseases and Protection.
Ethical approval
Include appropriate approvals or waivers.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jamali, F., Sohrabi, F. & Kohanmoo, M.A. Entomopathogenic fungi and plant essential oils are not compatible in controlling Tribolium castaneum (Herbst). J Plant Dis Prot 128, 799–808 (2021). https://doi.org/10.1007/s41348-021-00430-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41348-021-00430-5