Abstract
Systemic fungicide seed treatments are routinely used in conventional agriculture to control soil and seed-borne diseases, but little is known about their unintended adverse effects on non-target beneficial fungal endophytes that are known to be involved in plant growth and development. This study evaluated the seed treatment effect of a broad spectrum systemic fungicide, carbendazim (bavistin) on symbiotic association of fungal endophytes in rice and on early seedling growth of rice, green gram, soybean, and cowpea. Seeds were surface sterilized with sodium hypochlorite followed by 0.2% bavistin treatment. Growth of fungal endophytes was significantly affected by the seed treatment with fungicide in rice seedlings, while shoot and root growth was suppressed in all the crops. Quantitative real time PCR showed that the level of expression of two basal transcriptional regulator genes, OsBTF3 and OsNF-YC1 that are required for seed germination and seedling growth significantly decreased in bavistin treated rice seedlings. Re-inoculation of consortia of fungal endophytes onto bavistin treated rice seedlings significantly recovered seedling growth and development. These results suggest that fungicide treatment of seeds affects early seedling growth and has negative impact on beneficial fungal endophytes that are involved in plant growth and development. This study provides information on possible ill effects of fungicide on beneficial fungal endophytes that play key roles in early seedling growth of plants and also open up the prospect to additional research on different crops in vitro and field conditions to determine the consequences of fungicide effects and optimise fungicide application strategies to develop sustainable disease control methods.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fungicide seed treatments are commonly used to control pathogens in most of the agriculturally important crops since the 1920s (Lambert et al. 1926). Seed treatment with fungicides prior to sowing enhances seed health, plant stand and crop yield as well as control seed-borne fungi (Tanweer 1982). In general, most of the fungicides act by limiting the growth of fungi on plants, roots or seeds mostly by inhibiting the energy metabolism, interfering in signal transduction pathways, blocking biosynthetic processes or modifying cell membranes of fungus (Garcia et al. 2003; Yang et al. 2011). Carbendazim, a member of the benzimidazole group of fungicides which is a broad spectrum systemic fungicide is used for seed treatment in the concentration of 2 g/kg (0.2%) to control several diseases such as blast, powdery mildew, root rot, damping off and smut diseases in agriculturally important crops (Zhang et al. 2016).
Although fungicides are effective against targeted, soil and seed-borne pathogens, they are not species-specific and unfortunately, also effect non-target beneficial fungi. Since these fungicides are systemic in nature, it is possible for them to have adverse effects on endophytic fungi. Diverse array of endophytic fungi have been found to be associated with virtually all plant species in the natural ecosystem, where they reside in the internal tissues of their host plant and play important role in conferring fitness and in imparting tolerance to pathogens (Waqas et al. 2015; Wiewióra et al. 2015), herbivory (Finch et al. 2015), drought (Penuelas et al. 2012), heat stress (Rodriguez et al. 2004; Márquez et al. 2007), salt stress (Gahlot et al. 2015), as well as in promoting plant growth (Khan et al. 2015). However, adverse effects on beneficial microorganisms may attenuate the net benefit of fungicides. Recently, Prior et al. (2017) reported that systemic fungicide azoxystrobin affected the endophytic fungal species richness in common and broad beans. Similarly, Nettles et al. (2016) showed that treatment of seeds with systemic fungicides (mefenoxam and sedaxane) significantly affected the soybean leaf endophyte fungal community. As a result, fungicide application to control one pathogen might have significant, unintended effects on beneficial microorganisms. It is important to understand the effect of fungicides on the beneficial activities of endophytes to assess the hazards associated with fungicide use in agriculture and optimise fungicide application strategies. The present study was carried out to evaluate the effect of seed treatment with bavistin on early seedling growth and on fungal endophytes in rice and a few pulse crops (green gram, soybean, and cowpea).
Materials and methods
Fungicide seed treatment
Seeds of rice (Oryza sativa L.) cv. IR-64, green gram (Vigna radiata L.), soybean (Glycine max L.) and cowpea (Vigna unguiculata L.) were procured from All India Coordinated Research Project (AICRP), University of Agricultural Sciences, GKVK, Bengaluru, India. Prior to fungicide treatment, seeds were first surface-disinfected in 70% (v/v) ethanol for 1 min, then soaked in 4% (w/v) sodium hypochlorite for 1 min and with second immersion in 70% (v/v) ethanol for 1 min, followed by washing in sterile distilled water for several times (Arnold et al. 2000). Surface sterilized seeds were treated with 0.2% (w/v) bavistin (50WP Carbendazim) for 3 h and seeds soaked in sterile distilled water were used as control. After 3 h of fungicide treatment, seeds were washed thrice with sterile distilled water to remove the traces of bavistin. The bavistin treated and untreated seeds were placed uniformly on germination paper separately, roll towel was made and grown at room temperature for 5 days. Seed germination and growth parameters such as root and shoot lengths of 5 days old seedlings were recorded.
Effect of bavistin on early seedling growth under NaCl stress
To evaluate the effect of systemic fungicide under NaCl stress, bavistin treated and untreated green gram, soybean and cowpea seeds were placed on the sterile moistened blotter discs in petriplate and incubated at room temperature for 2 days. The pre-germinated seeds were transferred to germination paper and subjected to NaCl stress (100 mM). The growth parameters of 5 days old seedlings were recorded.
Isolation and characterization of endophytic fungi from rice seedlings
The fungal endophytes were isolated from the 11 days old bavistin treated and untreated rice seedlings. Seedlings were cut into 1 cm long segments and surface sterilized as described by Arnold et al. (2000). After blot drying, five tissue segments were transferred to petriplates containing potato dextrose agar (PDA) and incubated at 27 °C for 10 days under 12/12 h regimes of light and darkness (Suryanarayanan 1992). Imprints of sterilized cut segments were examined on PDA plates and thus checked for the effectiveness of the surface sterilization procedure (Schulz et al. 1998). Fungi emerging from the cut ends of the tissue segments were sub-cultured onto fresh PDA plates to obtain pure cultures. The purified isolates were cultured on PDA slants and stored at 4 °C. Voucher number were assigned and deposited in School of Ecology and Conservation Lab, University of Agricultural Sciences, GKVK, Bengaluru.
The purified isolates were categorized into operational taxonomic units (OTUs) based on the cultural characteristics and morphology of reproductive structures/spores/conidia (Domsch and Gams 1972; Ellis 1976; Sutton 1980; Arx 1981). For the molecular characterization, genomic DNA was isolated from the fungal mycelium using cetyl trimethyl ammonium bromide (CTAB) method. The universal ITS primers, ITS1 and ITS4 (White et al. 1990) were used to amplify fungal internal transcribed spacer regions (ITS1-5.8S- ITS2). Amplified products were purified and sequenced (SciGenom Labs Pvt. Ltd. Cochin, India). The ITS sequence was used to search the closest sequences from the GenBank database by performing blast search against nucleotide collection (nr/nt) database in NCBI blast server (http://blast.ncbi.nlm.nih.gov/). The sequences were deposited in NCBI GenBank.
Inoculation of consortia of fungal endophytes onto bavistin treated rice seedlings
To study the influence of fungal endophytes on improved seedling growth in bavistin treated IR-64 seedlings; the consortium of fungal endophytes was prepared from the isolates obtained from untreated (control) rice seedlings. Briefly, hyphal tip from the colony cultures of the 10 OTUs obtained were cultured aseptically on PDA. Five-day-old colonies were used to prepare the mycelial suspension (2 × 106 cfu ml−1) (Dhingra and Sinclair 1985) and equal volume of (10 ml) mycelial suspension of all the 10 OTUs were mixed to prepare the consortia of fungal inoculum.
Bavistin treated rice seedlings (11 days old) were treated with consortia of fungal inoculum (obtained from control seedlings) for 3 h by soaking seedlings in mycelial suspension (Zhang et al. 2014) and a control was maintained by treating with sterile water. After 3 h of treatment, seedlings were transferred to the moistened germination paper and incubated at room temperature for 3 days. Root and shoot lengths of the seedlings were recorded after 2 weeks. Seedlings were also used to isolate the endophytic fungi to evaluate the colonization by the fungal OTUs.
Expression analysis of OsBTF3 and OsNF-YC genes
To examine the expression of Osj10gBTF3 and OsNF-YC genes in control and bavistin treated 11 days old rice seedlings, total RNA was extracted from 100 mg tissue of shoot using the modified protocol by Sajeevan et al. (2014) and then subjected to DNAse treatment with DNase I (Thermo Fisher Scientific, USA). First-strand cDNA was synthesized from total RNA using the RevertAid First Strand cDNA Synthesis Kit following the manufacturer’s protocol (Thermo Fisher Scientific, USA) and quantitative real time PCR (qRT-PCR) was performed using cDNA as a template with the gene-specific primers (Osj10gBTF3 forward: 5′- GCTTGTCCCTGGTGAGACAT-3′ reverse: 5′- CAACATACCGGACCAAATCC-3′ and OsNF-YC1 forward: 5′- CAACTCCGTGAGTTCTGGGGCTAACC-3′ reverse: 5′- CTCGCATGCCTTTGCGAAGAC-3′). The rice actin gene was used as a reference gene, the primer, forward: 5′- TCCATAATGAAGTGTGATGT-3′ reverse: 5′- GGACCTGACTCGTCATACTC-3′. The qRT-PCR was performed on iQ5 real-time PCR detection system (Bio-Rad, USA) using iQ SYBR® Green Supermix (Bio-Rad, USA) at an annealing temperature of 60 °C (for both Osj10gBTF3 and OsNF-YC). The melting curve analysis was carried out from 50 to 90 °C with a hold of 1 min for every 1 °C. The threshold cycle (Ct) values of the triplicate PCR’s were averaged, and relative expression levels of the Osj10gBTF3 and OsNF-YC genes were calculated using the 2−ΔΔCT method (Livak and Schmittgen 2001).
Data analysis
All experiments were conducted in triplicates (20 seedlings per replicate) and one-way ANOVA was carried out to statistically validate the results. The means of various treatments were compared using a Tukey’s 95% confidence interval. MS-Excel was used for all statistical analysis.
Results
Seed treatment, endophytic isolation, identification, and rice plant bioassays
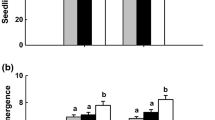
The effect of systemic fungicide, bavistin on early seedling growth and influence of the seed treatment on endophytes in rice genotype, IR-64 was evaluated. Fungicide treatment resulted in stunted growth in 11 days old seedlings, when compared to un-treated seedlings (Fig. 1). The shoot and root length of bavistin treated seedlings was significantly less (P < 0.05) compared to the control (without bavistin). The seed treatment reduced the shoot and root growth by 25 and 60%, respectively. Correspondingly, the same trend was observed when the experiment was repeated for the second time (Fig. S1).
Effect of carbendazim (bavistin) on growth of 11 days old rice (IR-64) seedlings. a IR-64 seedlings with and without bavistin (0.2%) seed treatment. b Root and Shoot growth of the seedlings with and without bavistin seed treatment (Significant difference between treated and control plants at P < 0.05.). Different letters (A, a, B, b) denote significant difference
The fungicide used in this study is a broad-spectrum systemic fungicide and not species-specific, therefore may have influence on beneficial fungal endophytes. Thus, we isolated endophytic fungi from 11 days old IR-64 control and treated seedlings. A total of 32 endophytic fungi were isolated, of which 27 were from control and five from treated seedlings. These 32 isolates were classified into 10 OTU’s (D-OTU, E-OTU, F-OTU, H-OTU, J-OTU, K-OTU, M-OTU, N-OTU, O-OTU-10a and O-OTU-10c). The fungal isolates from control seedlings were represented in all 10 OTUs, whereas isolates from bavistin-treated seedlings belonged to only three OTUs. The 10 OTU’s from control plants were further subjected to molecular characterization using universal ITS primers (ITS1 and ITS4). Following PCR amplification, only in four (F-OTU, J-OTU, M-OTU and O-OTU) out of 10 OTU’s, amplification of target region was noticed. The nucleotide sequences obtained after ITS sequencing for F-OTU, J-OTU and O-OTU revealed highest homology for Dendryphiella sp. (weed pathogenic fungus) and, M-OUT for Alternaria tenuissima (saprophytic fungus and opportunistic plant pathogen) (Table 1). The nucleotide sequences of fungal isolates (J-OTU, M-OTU, and O-OTU) submitted to NCBI GeneBank was allotted accession numbers KY399876, KY399874 and KY399875, respectively. The obtained F-OTU sequence was less than 200 bp and, therefore was not deposited in the database.
Furthermore, the fungal endophytes isolated from control seedlings were evaluated for their role in enhancing early seedling growth in bavistin treated rice seedlings. The consortia of fungal endophytes were inoculated on 11 days old seedlings and 3 days post-inoculation, these seedlings showed partial recovery in growth and development. The shoot and root length of 14 days old bavistin treated seedlings with endophytes was significantly more (P < 0.05) compared to control (without endophyte treatment, Fig. 2).
Effect of inoculation of consortia of fungal endophytes on improving early seedling growth in bavistin treated paddy seedlings. a IR-64 seedlings (14 days old) with and without bavistin (0.2%) seed treatment and bavistin treated seedlings enriched with endophytic fungal consortia. b Shoot and root growth of IR-64 seedlings (14 days old). Significant difference between treated and control plants at P < 0.05. Control: IR-64 seedlings without bavistin treatment, B +: bavistin treated IR-64 seedlings without endophytic fungal consortia and B + EF: bavistin treated IR-64 seedlings with endophytic fungal consortia. Different letters (A, a, B, b, C, c) denote significant difference
Effect of bavistin seed treatment in pulses
The effect of bavistin seed treatment on early seedling growth was evaluated under control and salt stress (100 mM NaCl) in green gram, soybean and cowpea. In all three crops, bavistin seed treatment progressively delayed the germination of seed and considerably affected the seedling growth (4 days old) under both control and salt stress (100 mM NaCl). Bavistin-treated seedlings under NaCl stress (P < 0.01) had significant reduction in shoot and root growth compared to the control seedlings in all the three crop plants studied (Figs. 3, 4 and 5). In green gram and soybean, the seed treatment reduced the shoot and root growth by nearly 45 and 60% under control, and 90 and 93% under salt stress, respectively compared to the seedlings grown without NaCl (control). However, seed treatment of cowpea decreased the shoot and root growth by about 40 and 25% under control, and 85 and 75% under salt stress, respectively. Similarly, bavistin seed treatment under control conditions in all three crop plants was repeated for second time and the results showed similar trend as observed in the first experiment except for soybean, in which bavistin seed treatment inhibited germination (Fig. S2, S3 and S4).
Effect of bavistin seed treatment on early seedling growth in green gram. Green gram seedlings (4 days old) with and without bavistin seed treatment grown under a control (without NaCl stress) and b NaCl (100 mM) stress. c Root and shoot growth of 4 days old green gram seedlings. B +: bavistin seed treatment. Different letters (A, a, B, b, C, c, D, d) denote significant difference at P < 0.01
Effect of bavistin seed treatment on early seedling growth in soybean. Soybean seedlings (4 days old) with and without bavistin seed treatment grown under a control (without NaCl stress) and b NaCl (100 mM) stress. c Root and shoot growth of 4 days old soybean seedlings. B +: bavistin seed treatment. Different letters (A, a, B, b, C, c, D) denote significant difference at P < 0.01
Effect of bavistin seed treatment on early seedling growth in cowpea. Cowpea seedlings (4 days old) with and without bavistin seed treatment grown under a control (without NaCl stress) and b NaCl (100 mM) stress. c Root and shoot growth of 4 days old cowpea seedlings. B +: bavistin seed treatment. Different letters (A, a, B, b, C, c) denote significant difference at P < 0.01
Expression analysis of Osj10gBTF3 and OsNF-YC1 genes
To understand the level of expression of basal transcription factor 3 (Osj10gBTF3) and NUCLEAR FACTOR Y (OsNF-YC1) transcription factor genes in control and bavistin treated 11 days old rice (IR-64) seedlings, qRT-PCR was carried out. The expression level of Osj10gBTF3 and OsNF-YC1 genes in rice control (without fungicide seed treatment) was set to ‘1’ as a standard to compare the relative expressions of the two genes in fungicide treated seedlings. The level of expression of Osj10gBTF3 and OsNF-YC1 genes was 0.5 and 0.35 fold less in bavistin treated IR-64 seedlings, respectively compared to control (Fig. 6). The expression level of both the genes was reduced significantly (P < 0.01) in response to the systemic fungicide seed treatment in rice.
Expression analysis of Osj10gBTF3 and OsNF-YC1 genes using qRT-PCR in 11 days old rice (IR-64) seedlings. Graph shows fold decrease in the expression level of Osj10gBTF3 and OsNF-YC1 genes under bavistin treated IR-64 seedlings. The letters ‘a’ and ‘b’ denote significant (P < 0.01) difference in level of expression of genes. IR-64 F+: bavistin seed treated IR-64 seedlings (11 days old)
Discussion
Several studies have revealed that the fungicide seed treatment controls early onset of diseases, improves seed health and plant stand in agriculturally important crops such as rice (Van Nghiep and Gaur 2005; Bagga and Sharma 2006), wheat (Sharma-Poudyal et al. 2005), maize (Munkvold and O’mara 2002), mung bean (Dubey and Singh 2010), chickpea (Gan et al. 2006), soybean (Mueller et al. 1999), etc. However, the effects of systemic fungicides on beneficial microorganisms are not well studied. Because of their systemic nature, these fungicides may have negative impacts on non-target beneficial fungi and therefore, we expected that endophyte fungal communities that promote plant growth and development would be affected by the systemic fungicide seed treatment resulting in stunted seedling growth. In this study, we selected four plant species, rice, green gram (mung bean), soybean and cowpea based on their importance in agriculture and frequent use of fungicides as seed treatment to control soil and seed-borne pathogens.
Our study showed that the application of systemic fungicide, bavistin (0.2%) as a seed treatment has a significant effect on early seedling growth in all the crops tested. The seed treatment with bavistin delayed seed germination, and significantly reduced shoot and root length in rice (25 and 60%), green gram (45 and 60%), soybean (45 and 60%) and cowpea (40 and 25%). Similar trend was observed when the experiment was repeated for the second time except for soybean in which fungicide seed treatment completely inhibited germination. These results were in contrast to the general agreement that fungicide seed treatments increase germination, seedling survival and vigor (Taylor and Harman 1990). Alternative to fungicides could be plant growth-promoting fungal association that has been gaining attention in recent years. Among fungi, endophytes are known to improve plant growth and development under stress conditions by producing phytohormones such as gibberellins and indole acetic acid (Waqas et al. 2012) and possess antimicrobial activity (Waqas et al. 2015).
Fungicide seed treatment in green gram, soybean and cowpea, and their interaction with salt stress was studied to investigate the impact of fungicides on plant growth and development under stress and assess the performance of early seedling growth. The fungicide treated seeds under salt stress were severely affected with regard to early seedling growth compared to control and bavistin treated seedlings grown without salt stress (Figs. 3, 4 and 5). This could be due to the loss of non-target beneficial fungal endophytes that are known to be involved in promoting plant growth and development during stress conditions (Khan et al. 2015). It was evident from our study on rice seedlings that the bavistin seed treatment had a significant effect on native endophytic fungal community composition. The total OTU richness was significantly lower in the fungicide-treated seedlings (3) compared to control (10), and the consortia of fungal endophytes from control seedlings when inoculated on bavistin treated seedlings restored seedling growth in rice. This restoration may be due to the deterioration of bavistin effect in 11 days old seedlings and thereby endophytes were able to colonize and aid in seedling growth. The 3 OTUs observed in fungicide-treated rice seedlings might be resistant to bavistin and further work should be carried out with regard to the molecular characterization of these bavistin tolerant OTUs and evaluate their role in plants when treated with fungicides. Also, the fungal OTU richness in agriculturally important crops with and without bavistin treatment should be examined to understand the negative impact of fungicides on beneficial endophytes. There are reports on the negative effects of fungicides on non-target beneficial microorganisms (Karlsson et al. 2014; Buysens et al. 2015; Nettles et al. 2016). While endophyte fungal communities are known to promote plant growth and development during biotic and abiotic stress (Gahlot et al. 2015; Wiewióra et al. 2015), the significant effect of fungicides on these beneficial fungi deserves further investigation.
Furthermore, the expression level of genes in response to fungicide seed treatment during seed germination and early seedling growth is not well known. In this study, we analyzed two transcription factors (TFs), OsBTF3 and OsNF-YC1 in response to the systemic fungicide seed treatment in rice seedlings. Both the TFs are known to be associated with plant developmental processes. In rice, BTF3 plays an important role in plant growth and development (Wang et al. 2012) and recently, Wang et al. (2014) demonstrated that a variant of OsBTF, Osj10gBTF3 is required during seed germination and seedling growth. Increasing evidence suggests that NF-Y subunits are involved in flowering control (Kumimoto et al. 2010), phytohormone response (Hou et al. 2014), and abiotic stress tolerance (Palmeros-Suárez et al. 2015). In a recent study, Liu et al. (2016) showed that the NF-YC members are responsible for suppression of GA-mediated seed germination by interacting with the DELLA protein RGL2 (GA signalling repressor). In the present work, we showed that the Osj10gBTF3 and OsNF-YC genes were significantly down regulated in bavistin treated rice seedlings suggesting that the fungicide directly or indirectly affected the expression of genes required for the early seedling growth. This could be also due to the removal of native endophytes that may be responsible for the expression of both genes involved in plant growth and development under stress conditions, as recently, we have observed that the salt sensitive rice genotype IR-64 treated with salt tolerant endophyte (Botryosphaeria dothidea) showed tolerance to salt stress and up-regulated both Osj10gBTF3 and OsNF-YC genes (unpublished data).
The study demonstrated that the systemic fungicide seed treatment significantly reduced early seedling growth that may be due to the negative impact on non-target beneficial fungal endophytes. Therefore, understanding the possible side effects of fungicides on endophytic fungi is necessary to use fungicide more wisely in agriculture. Further research should be carried out on different agriculturally important crops to evaluate the effect of seed treatment in vitro and field conditions, and also understand the mechanisms behind fungicide-fungal endophyte-seedling growth interactions that could have the potential to guide the development of sustainable disease control strategies.
References
Arnold, A. E., Maynard, Z., Gilbert, G. S., Coley, P. D., & Kursar, T. A. (2000). Are tropical fungal endophytes hyperdiverse? Ecology Letters, 3, 267–274.
Arx, J. (1981). The genera of fungi sporulating in pure culture. Vaduz: J Cramer.
Bagga, P. S., & Sharma, V. K. (2006). Evaluation of fungicides as seedling treatment for controlling bakanae/foot-rot (Fusarium moniliforme) disease in basmati rice. Indian Phytopathology, 59, 305–308.
Buysens, C., De Boulois, H. D., & Declerck, S. (2015). Do fungicides used to control Rhizoctonia solani impact the non-target arbuscular mycorrhizal fungus Rhizophagus irregularis? Mycorrhiza, 25, 277–288.
Dhingra, O., & Sinclair, J. (1985). Culture media and their formulas. In O. Dhingra & J. Sinclair (Eds.), Basic plant pathology methods (pp. 285–315). Boca Raton: CRC Press.
Domsch, K., & Gams, W. (1972). Fungi in agriculture soils. London: Longman Group Ltd.
Dubey, S. C., & Singh, B. (2010). Seed treatment and foliar application of insecticides and fungicides for management of cercospora leaf spots and yellow mosaic of mungbean (Vigna radiata). International Journal of Pest Management, 56, 309–314.
Ellis, M. (1976). More dematiaceous Hypomycetes. Kew Surrey: Commonwealth Mycological Institute.
Finch, S., Pennell, C., Kerby, J., & Cave, V. (2015). Mice find endophyte-infected seed of tall fescue unpalatable—implications for the aviation industry. Grass & Forage Science, 71, 659–666.
Gahlot, S., Joshi, A., Singh, P., Tuteja, R., Dua, M., Jogawat, A., et al. (2015). Isolation of genes conferring salt tolerance from Piriformospora indica by random overexpression in Escherichia coli. World Journal of Microbiology & Biotechnology, 31, 1195–1209.
Gan, Y., Siddique, K., MacLeod, W., & Jayakumar, P. (2006). Management options for minimizing the damage by ascochyta blight (Ascochyta rabiei) in chickpea (Cicer arietinum L.). Field Crops Research, 97, 121–134.
Garcia, P. C., Rivero, R. M., Ruiz, J. M., & Romero, L. (2003). The role of fungicides in the physiology of higher plants: Implications for defense responses. Botanical Review, 69, 162–172.
Hou, X., Zhou, J., Liu, C., Liu, L., Shen, L., & Yu, H. (2014). Nuclear factor Y-mediated H3K27me3 demethylation of the SOC1 locus orchestrates flowering responses of Arabidopsis. Nature Communications, 5, 4601.
Karlsson, I., Friberg, H., Steinberg, C., & Persson, P. (2014). Fungicide effects on fungal community composition in the wheat phyllosphere. PLoS ONE, 9, e111786.
Khan, A. R., Ullah, I., Waqas, M., Shahzad, R., Hong, S. J., Park, G. S., et al. (2015). Plant growth-promoting potential of endophytic fungi isolated from Solanum nigrum leaves. World Journal of Microbiology & Biotechnology, 31, 1461–1466.
Kumimoto, R. W., Zhang, Y., Siefers, N., & Holt, B. F. (2010). NF–YC3, NF–YC4 and NF–YC9 are required for CONSTANS-mediated, photoperiod-dependent flowering in Arabidopsis thaliana. Plant Journal, 63, 379–391.
Lambert, E., Rodenhiser, H., & Flor, H. (1926). The effectiveness of various fungicides in controlling the covered smuts of small grains. Phytopathology, 16, 393–411.
Liu, X., Hu, P., Huang, M., Tang, Y., Li, Y., Li, L., et al. (2016). The NF-YC–RGL2 module integrates GA and ABA signalling to regulate seed germination in Arabidopsis. Nature Communications, 7, 12768.
Livak, K. J., & Schmittgen, T. D. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods, 25, 402–408.
Márquez, L. M., Redman, R. S., Rodriguez, R. J., & Roossinck, M. J. (2007). A virus in a fungus in a plant: Three-way symbiosis required for thermal tolerance. Science, 315, 513–515.
Mueller, D., Hartman, G., & Pedersen, W. (1999). Development of sclerotia and apothecia of Sclerotinia sclerotiorum from infected soybean seed and its control by fungicide seed treatment. Plant Disease, 83, 1113–1115.
Munkvold, G., & O’mara, J. (2002). Laboratory and growth chamber evaluation of fungicidal seed treatments for maize seedling blight caused by Fusarium species. Plant Disease, 86, 143–150.
Nettles, R., Watkins, J., Ricks, K., Boyer, M., Licht, M., Atwood, L. M., et al. (2016). Influence of pesticide seed treatments on rhizosphere fungal and bacterial communities and leaf fungal endophyte communities in maize and soybean. Applied Soil Ecology, 102, 61–69.
Palmeros-Suárez, P. A., Massange-Sánchez, J. A., Martínez-Gallardo, N. A., Montero-Vargas, J. M., Gómez-Leyva, J. F., & Délano-Frier, J. P. (2015). The overexpression of an Amaranthus hypochondriacus NF-YC gene modifies growth and confers water deficit stress resistance in Arabidopsis. Plant Science, 240, 25–40.
Penuelas, J., Rico, L., Ogaya, R., Jump, A., & Terradas, J. (2012). Summer season and long-term drought increase the richness of bacteria and fungi in the foliar phyllosphere of Quercus ilex in a mixed Mediterranean forest. Plant Biology, 14, 565–575.
Prior, R., Mittelbach, M., & Begerow, D. (2017). Impact of three different fungicides on fungal epi-and endophytic communities of common bean (Phaseolus vulgaris) and broad bean (Vicia faba). Journal of Environmental Science and Health, Part B, 52, 376–386.
Rodriguez, R. J., Redman, R. S., & Henson, J. M. (2004). The role of fungal symbioses in the adaptation of plants to high stress environments. Mitigation and Adaptation Strategies for Global Change, 9, 261–272.
Sajeevan, R. S., Shivanna, M. B., & Nataraja, K. N. (2014). An efficient protocol for total RNA isolation from healthy and stressed tissues of mulberry (Morus sp.) and other species. American Journal Plant Science, 5, 2057–2065.
Schulz, B., Guske, S., Dammann, U., & Boyle, C. (1998). Endophyte-host interactions. II. Defining symbiosis of the endophyte-host interaction. Symbiosis, 25, 213–227.
Sharma-Poudyal, D., Duveiller, E., & Sharma, R. (2005). Effects of seed treatment and foliar fungicides on Helminthosporium leaf blight and on performance of wheat in warmer growing conditions. Journal of Phytopathology, 153, 401–408.
Suryanarayanan, T. (1992). Light-incubation: A neglected procedure in mycology. Mycologist, 6, 144.
Sutton, B. (1980). The Coelomycetes, Fungi Imperfecti with acervuli, pycnidia and stromata. Kew: Commonwealth Mycological Institute.
Tanweer, A. (1982). Effect of a new fungicide on the viability of rice and sorghum seeds. Pestology, 6, 9–10.
Taylor, A., & Harman, G. (1990). Concepts and technologies of selected seed treatments. Annual review of Phytopathology, 28, 321–339.
Van Nghiep, H., & Gaur, A. (2005). Efficacy of seed treatment in improving seed quality in rice (Oryza sativa L.). Omonrice, 13, 42–51.
Wang, Y., Zhang, X., Lu, S., Wang, M., Wang, L., Wang, W., et al. (2012). Inhibition of a basal transcription factor 3-like gene Osj10gBTF3 in rice results in significant plant miniaturization and typical pollen abortion. Plant and Cell Physiology, 53, 2073–2089.
Wang, W., Xu, M., Wang, Y., & Jamil, M. (2014). Basal transcription factor 3 plays an important role in seed germination and seedling growth of rice. BioMed Research International, 2014, 465739.
Waqas, M., Khan, A. L., Kamran, M., Hamayun, M., Kang, S.-M., Kim, Y.-H., et al. (2012). Endophytic fungi produce gibberellins and indoleacetic acid and promotes host-plant growth during stress. Molecules, 17, 10754–10773.
Waqas, M., Khan, A. L., Hamayun, M., Shahzad, R., Kim, Y.-H., Choi, K.-S., et al. (2015). Endophytic infection alleviates biotic stress in sunflower through regulation of defence hormones, antioxidants and functional amino acids. European Journal of Plant Pathology, 141, 803–824.
White, T. J., Bruns, T., Lee, S., & Taylor, J. (1990). Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protocols: A Guide to Methods and Applications, 18, 315–322.
Wiewióra, B., Żurek, G., & Żurek, M. (2015). Endophyte-mediated disease resistance in wild populations of perennial ryegrass (Lolium perenne). Fungal Ecology, 15, 1–8.
Yang, C., Hamel, C., Vujanovic, V., & Gan, Y. (2011). Fungicide: modes of action and possible impact on non-target microorganisms. International Scholarly Research Notices: Ecology. https://doi.org/10.5402/2011/130289.
Zhang, Q., Zhang, J., Yang, L., Zhang, L., Jiang, D., Chen, W., et al. (2014). Diversity and biocontrol potential of endophytic fungi in Brassica napus. Biological Control, 72, 98–108.
Zhang, Y., Di Wang, H., & Zhang, C. Q. (2016). Management of benzimidazole fungicide resistance in eggplant brown rot (Phomopsis vexans) with pyraclostrobin. Phytoparasitica, 44, 313–324.
Acknowledgements
The authors would like to thank DBT, India funded project “Chemical Ecology of the North East Region (NER) of the India: A collaborative programme Linking NER and Bangalore Researchers” for financial support DBT-NER/Agri/24/2013, and Poornima A, and Santhosh Kumar J U for technical assistance. This work is partially supported by Indian Council of Agricultural Research (ICAR-CAAST- F.No./NAHEP/CAAST/2018-19), Government of India, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vasanthakumari, M.M., Shridhar, J., Madhura, R.J. et al. Role of endophytes in early seedling growth of plants: a test using systemic fungicide seed treatment. Plant Physiol. Rep. 24, 86–95 (2019). https://doi.org/10.1007/s40502-018-0404-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40502-018-0404-6