Abstract
Background
Collegiate football players who started playing tackle football before age 12 years did not show worse neuropsychological test performance than those who started playing tackle football after age 12 years. It is unknown if beginning other contact sports, such as lacrosse, at a younger age is associated with worse neurocognitive performance, greater psychological distress, or worse postural stability in collegiate student athletes.
Objective
The purpose of this study was to examine the association between estimated age of first exposure (eAFE) to repetitive head impacts (RHI) and these outcome measures in collegiate student athletes.
Methods
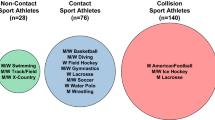
1891 female and 4448 male collision/contact (i.e., football, ice hockey, lacrosse, wrestling, soccer) and non-contact (i.e., golf, rifle, rowing/crew, swimming, tennis) sport athletes completed baseline testing, including the Immediate Post-Concussion Assessment and Cognitive Testing (ImPACT), Brief Symptom Inventory 18 (BSI-18), and Balance Error Scoring System (BESS).
Results
For women, the eAFE-by-sport interaction was associated with ImPACT Verbal Memory and Visual Memory, whereby earlier eAFE to contact sports was associated with higher composite scores (B = − 0.397, B = − 0.485, respectively). For men, the eAFE-by-sport interaction was associated with BSI-18 Depression and Global Severity Index and symptom severity scores, whereby earlier eAFE to football was associated with lower psychological distress and symptom severity [Depression, Exp(B) = 1.057; Global Severity Index, Exp(B) = 1.047; Symptom Severity, Exp(B) = 1.046]. Parameter estimates were small suggesting these results may have minimal practical relevance.
Conclusion
Findings suggest that RHI during early adolescence is unrelated to brain health as measured by these specific outcome measures in collegiate student athletes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
We observed no association between earlier estimated age of first exposure to repetitive head injury and worse neurocognitive function, greater psychological distress, or worse postural stability in male or female collision and contact sport collegiate student athletes. |
This study suggests that exposure to contact sports, such as ice hockey, lacrosse, wrestling, and soccer, during early adolescence is unrelated to baseline neurocognitive function, psychological distress, and postural stability in healthy NCAA collegiate student athletes. |
1 Introduction
Early adolescence (e.g., age 10–12 years) is a critical stage of brain development [1,2,3,4,5]. One research group has conducted several studies with former National Football League players [6,7,8] and former amateur players [9, 10] illustrating that earlier self-reported age of first exposure (AFE) to football is associated with differences in later-life cognitive functioning, altered corpus callosum white matter microstructure, decreased thalamic volume, greater behavioral and mood symptoms, and earlier age of neurobehavioral symptom onset [6,7,8,9,10]. However, other researchers did not find these associations in retired NFL players [11,12,13,14] or in current high school and collegiate football players [15,16,17,18,19] suggesting that the literature with former football players is mixed and inconclusive as to whether exposure to tackle football prior to age of 12 is associated with worse later-in-life brain health. These studies have been limited to football players; thus, the results may not be generalizable to other groups. Considering the widespread nature of repetitive head impacts (RHI) across sex and sport, it is important to study a diverse cohort to understand the association between AFE to RHI and neurocognitive function more broadly.
Although the study of former football players allows for investigation of a group with the highest exposure to RHI, the nature and number of head impacts incurred in other youth sports, including ice hockey and soccer, likely differs from those incurred in youth football [20,21,22,23,24,25]. Moreover, the timing of brain development stages and milestones differs between males and females [1,2,3,4,5]; therefore, previous findings may not apply to women exposed to RHI during early adolescence. Considering the growth of women’s sports since the passing of Title IX, it is critical to study the effects of exposure to RHI during early adolescence in both men and women. The National Collegiate Athletic Association (NCAA) and the US Department of Defense (DoD) established the Concussion Assessment, Research and Education (CARE) Consortium to study the natural history of concussion in current student athletes and military personnel [26]. This database will allow us to examine the association between AFE to RHI and neurocognitive function, psychological distress, and postural stability in a diverse cohort of collegiate men and women across a variety of collision, contact, and non-contact sports. While not addressing potential later-life impairments, this dataset currently allows for a more thorough understanding of possible short-to-medium-term effects of RHI in sport. Neurodevelopment continues throughout college, so observable neurocognitive deficits in this cohort may suggest future impairments in cognitive tasks that develop during later stages of neurodevelopment [1,2,3,4,5].
We aimed to examine the association between estimated AFE (eAFE) to RHI through contact and collision sports and neurocognitive performance, psychological distress, and postural stability in healthy collegiate student athletes, while controlling for factors known to be associated with neurocognitive function. We hypothesized that earlier eAFE to contact sport participation would not be associated with worse neurocognitive performance, greater psychological distress, or worse postural stability.
2 Methods
2.1 Participants
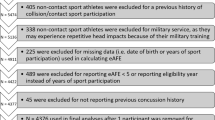
Participants were recruited through the CARE Consortium [20]. Inclusion criteria included civilian both men and women currently participating in collision/contact (i.e., football, ice hockey, lacrosse, wrestling, soccer) or non-contact (i.e., golf, rifle, rowing/crew, swimming, or tennis) sports. Exclusion criteria included serving as a service academy cadet and non-contact sport athletes with any history of collision or contact sport participation (i.e., if the participant reported years of participation for any collision or contact sport). Service academy cadets were excluded because they may experience RHI during their military training and because they are being studied separately. The University of Michigan institutional review board (IRB) and the local IRB at each of the performance sites reviewed and approved all study procedures. Participants provided written informed consent prior to participation.
2.2 Instrumentation
Baseline assessments from the Clinical Study Core were used in analyses [26,27,28], including the Brief Symptom Inventory 18 (BSI-18), the Immediate Post-Concussion Assessment and Cognitive Testing (ImPACT), and the Balance Error Scoring System (BESS). Each assessment has been described in detail elsewhere but will be described briefly below.
2.3 Brief Symptom Inventory 18 (BSI-18)
The BSI-18 is a brief symptom inventory that requires respondents to rate their level of distress over the past 7 days using a 5-point (ordinal) Likert-type scale, in which 0 is not at all and 4 is extremely often [29]. The BSI-18 provides scores on three dimensions: somatization, depression, and anxiety. Each of these clinical subscales comprises six items, so that the range of possible scores for each is 0 to 24, in which lower is better. In addition, a composite score is calculated based on all 18 items, so that the range of possible scores is 0–72. The BSI-18 has been shown to be reliable (ICC: 0.91) and valid in a brain injury cohort [29].
2.4 Immediate Post-Concussion Assessment and Cognitive Testing (ImPACT)
The ImPACT is the most widely used computerized neuropsychological test for sport-related concussion assessment [30, 31]. From six test modules, four composite scores are generated, including: (1) Verbal Memory (higher is better), (2) Visual Memory (higher is better), (3) Visual Motor Speed (higher is better), and (4) Reaction Time (lower is better). Combined test sensitivity is 0.81 and specificity is 0.89 [32]. Reliability varies (ICC: 0.34–0.72) depending on composite score measure [33]. Baseline testing group sizes were variable, often with multiple research assistants present for proctoring. Given the large number of participating sites and student athletes, sites were often not able to administer the ImPACT test one-on-one [26].
2.5 Balance Error Scoring System (BESS)
The BESS is commonly used for the sideline evaluation of postural control following concussion [34]. The BESS involves three different stances (double-limb stance, single-limb stance, and tandem stance) on two surfaces (firm and foam). Each position is held for 20 s with the participants’ eyes closed and hands on their hips. Errors are recorded by the administering clinician, including: (1) the hands coming off of the iliac crest; (2) opening the eyes; (3) step, stumble, or fall; (4) moving the hip into greater than 30° of abduction; (5) lifting the forefoot or heel; and (6) remaining out of the test position longer than 5 s. Each stance has a maximum error score of ten and thus, the total range for scoring was 0–60, where a lower score is better. Sensitivity for the BESS is low (0.34), but specificity is high (0.91–0.97) with moderate test–retest reliability (ICC: 0.41) [33]. Inter-rater reliability for the BESS is typically low (ICC: 0.57) [35], although it was not directly assessed in this study.
2.6 Demographic and Medical History
The Demographics and Personal and Family Medical History unique case report form captures demographic data (e.g., race and ethnicity), information on current and previous sport history, concussion history, and pre-existing personal and family medical history [e.g.. neurodevelopmental history—history of a learning disorder or Attention-Deficit/Hyperactivity Disorder (ADHD)]. These data were used to compute eAFE (see below). In addition, participants self-reported maternal and paternal education and occupation, which were used to classify socioeconomic status (SES). SES was determined by the Hollingshead Four-Factor Index [36]. Predefined SES groups were applied based on published cutoffs: low (0–19), middle–low (20–29), middle (30–39), middle–high (40–54), and high (55–66) [36].
2.7 Estimated Age of First Exposure (eAFE)
The eAFE was defined as the participant’s age at the time of assessment minus the number of years the participant reported playing his/her primary sport. The minimum eAFE for this study was 4 years old because this is the youngest age for many organized sport leagues and any participant indicating eAFE prior to age 4 was excluded (n = 350, 5%). If an athlete reported playing another collision/contact (i.e., football, ice hockey, lacrosse, wrestling, soccer) prior to playing his/her primary contact sport (e.g., current soccer player who played ice hockey younger), the age at which the participant reported playing the other contact sport (e.g., ice hockey) was used as eAFE.
2.8 Statistical Analyses
Generalized linear modeling was used to examine the association between eAFE and outcome measures (i.e., BSI-18 Somatization, Depression, Anxiety, Global Severity Index sub-scores, ImPACT Verbal Memory, Visual Memory, Visual Motor Speed, Reaction Time composite scores, Post-Concussion Symptom Scale (PCSS) Symptom Severity, and BESS total scores). Outcome measures for men and women were modeled independently. Predictors in the models included eAFE (continuous), sport (women: contact and non-contact; men: football, contact, and non-contact), the eAFE-by-sport interaction, concussion history (Y/N), neurodevelopmental history (Y/N), migraine history (Y/N), SES (high, middle–high, middle, middle–low, low), race (White, African American, Other), and ethnicity (Hispanic or Latino, Not Hispanic or Latino, Not Reported). We initially fit generalized linear models for each outcome measure based on a normal (Gaussian) distribution and identity link function but then considered models with alternative distributions and link functions and selected the model with the lowest Akaike information criterion value (i.e., best fit) [37]. For the BSI-18 Somatization, Depression, Anxiety, Global Severity Index sub-scores, and PCSS Symptom Severity score, which have a high zero count and extreme positive skewing, a negative binomial distribution with a log link achieved the lowest Akaike information criterion value. Reaction time scores were positively skewed; an inverse Gaussian distribution with power link function best fits these data. All analyses were conducted using SPSS (IBM Inc., Armonk, NY, USA), and significance was defined a priori as p < 0.05.
A generalized linear model based on a normal (Gaussian) distribution with identity link function is a linear regression model; so, parameter estimates (Tables 3 and 6) are the estimates of the model coefficients. Positive estimates suggest a higher response for that parameter; negative estimates suggest a lower response for that parameter. For example, in Table 3, ImPACT Verbal Memory B = 5.405 (95% confidence interval 2.693–8.117) for women involved in contact sports. Therefore, the parameter estimate suggests that women involved in contact sports score 5.405 points higher than the reference category (women involved in non-contact sports) when controlling for all other predictors in the model. For a continuous measure (i.e., eAFE), if ImPACT Visual Motor Speed B = −0.116 (95% confidence interval −0.245 to 0.013), then for every increase in eAFE by 1 year, ImPACT Visual Motor Speed would decrease by 0.116. For models described by negative binomial distribution with a log link function, Exp(B), or the exponential parameters, are included in Tables 3 and 6. The exponential parameters have an interpretation of by what percentage the psychological distress or symptom scores increase (or decrease) with each parameter. For example, in Table 3, BSI-18 somatization sub-score Exp(B) = 1.256 (95% confidence interval 1.069–1.474) for women with a positive concussion history. Therefore, the exponential parameter estimate suggests that BSI-18 somatization sub-scores are 25.6% higher in women with a positive concussion history.
3 Results
3.1 Women
There were 1891 women (Fig. 1) included in the analyses (current age 18.9 ± 1.2 years, eAFE 7.7 ± 3.2 years). Descriptive statistics are provided in Table 1. Visual inspection of the top row of Table 1 reveals that women involved in contact sports, compared to women involved in noncontact sports, (i) report comparable or fewer physical and emotional symptoms; (ii) perform better on the four neurocognitive composite scores from ImPACT; and (iii) perform comparably on the BESS.
3.1.1 Sport-by-eAFE Interaction
The sport-by-eAFE interaction was associated with ImPACT Verbal Memory and Visual Memory composite scores (Table 2), whereby later AFE to contact sports was associated with lower (worse) Verbal Memory (B = − 0.397, 95% confidence interval − 0.712 to − 0.083, Fig. 2a) and Visual Memory (B = − 0.485, 95% confidence interval − 0.897 to − 0.074, Fig. 2b) composite scores (Table 3).
Scatter plots for eAFE-by-sport interaction for a women’s ImPACT Verbal Memory composite scores, b women’s ImPACT Visual Memory composite scores, c men’s BSI-18 Depression sub-scores, d men’s BSI-18 Global Severity Index sub-scores, e men’s PCSS Symptom Severity scores, f men’s subgroup analysis for BESS total scores
3.1.2 Sport
Sport was associated with BSI-18 Somatization sub-scores, ImPACT Verbal Memory and Visual Memory composite scores, and BESS totals (Table 2), whereby contact sport athletes had lower (better) Somatization sub-scores [Exp(B) = 0.548, 0.366–0.821], higher (better) Verbal Memory (B = 5.405, 2.693–8.117) and Visual Memory (B = 8.433, 4.879–11.987) composite scores, and lower (better) BESS total scores (B = − 2.512, − 4.108 to − 0.916) (Table 3).
3.1.3 eAFE
Later eAFE was associated with lower (worse) ImPACT Visual Motor Speed (B = − 0.116, − 0.245 to 0.013) composite scores (Tables 2, 3).
3.1.4 Concussion History
Positive concussion history was associated with higher (worse) BSI-18 Somatization [Exp(B) = 1.256, 1.069–1.474] sub-scores and higher (worse) PCSS Symptom Severity [Exp(B) = 1.130, 1.001–1.274] (Tables 2, 3).
3.1.5 Neurodevelopmental History
Positive neurodevelopmental history was associated with (i) higher (worse) BSI-18 Somatization [Exp(B) = 1.318, 1.043–1.664], Depression [Exp(B) = 1.573, 1.251–1.978], Anxiety [Exp(B) = 1.702, 1.374–2.109], and Global Severity Index [Exp(B) = 1.516, 1.255–1.831] sub-scores; (ii) lower (worse) ImPACT Verbal Memory (B = − 2.682, − 4.339 to − 1.024), Visual Memory (B = − 4.252, − 6.424 to − 2.080), and Visual Motor Speed (− 2.690, − 3.714 to − 1.666), and higher (worse) Reaction Time (0.022, 0.006–0.038) composite scores; (iii) higher (worse) PCSS Symptom Severity [Exp(B) = 1.564, 1.303–1.878]; and (iv) higher (worse) BESS total scores (B = 1.287, 0.330–2.243) (Tables 2, 3).
3.1.6 Migraine History
Positive migraine history was associated with higher (worse) BSI-18 Somatization [Exp(B) = 1.866, 1.511–2.305], Depression [Exp(B) = 1.896, 1.534–2.347], Anxiety [Exp(B) = 1.537, 1.247–1.893], Global Severity Index [Exp(B) = 1.761, 1.471–2.106] sub-scores, and PCSS Symptom Severity scores [Exp(B) = 1.438, 1.207–1.713] (Tables 2, 3).
3.1.7 SES
High SES was associated with lower (better) BSI-18 Depression [Exp(B) = 0.662, 0.468–0.937] and Anxiety sub-scores [Exp(B) = 0.711, 0.511–0.990], and Low SES was associated with lower (better) BSI-18 Anxiety sub-scores [Exp(B) = 0.416, 0.242–0.714] (Tables 2, 3).
3.1.8 Race and Ethnicity
African American race was associated with higher (worse) BSI-18 Somatization [Exp(B) = 2.145, 1.517–3.031], Depression [Exp(B) = 2.123, 1.501–3.007], and Global Severity Index [Exp(B) = 1.828, 1.361–2.455] sub-scores. Other Race was associated with (i) higher (worse) BSI-18 Somatization [Exp(B) = 1.256, 1.034–1.528], Depression [Exp(B) = 1.606, 1.331–1.939], Anxiety [Exp(B) = 1.298, 1.080–1.560], and Global Severity Index [Exp(B) = 1.377, 1.177–1.611] sub-scores; (ii) higher (worse) PCSS Symptom Severity scores [Exp(B) = 1.252, 1.077–1.456]; and (iii) lower (better) BESS total scores (− 0.945, − 1.747 to − 0.143) (Tables 2, 3). Non-Hispanic or Latino ethnicity was associated with higher (better) Verbal Memory (B = 2.063, 0.427–3.699) and Visual Motor Speed (B = 1.563, 0.552–2.574).
3.2 Men
There were 4448 men (Fig. 1) included in the analyses (current age 19.3 ± 1.5 years, eAFE 8.6 ± 3.2 years). Descriptive statistics are provided in Table 4. Visual inspection of the top row of Table 4 reveals that men involved in contact sports or football, compared to men involved in noncontact sports, (i) report comparable or fewer physical and emotional symptoms, and (ii) perform comparably on the four neurocognitive composite scores from ImPACT. There were small differences in mean scores amongst the groups on the BESS.
3.2.1 Sport-by-eAFE Interaction
The sport-by-eAFE interaction was associated with BSI-18 Depression and Global Severity Index sub-scores, and the PCSS Symptom Severity scores (Table 5), whereby later AFE to football was associated with higher (worse) Depression [Exp(B) = 1.057, 95% confidence interval 1.002–1.115, Fig. 2c] and Global Severity Index [Exp(B) = 1.047, 95% confidence interval 1.002–1.093, Fig. 2d] sub-scores and PCSS Symptom Severity scores [Exp(B) = 1.046, 95% confidence interval 1.005–1.088, Fig. 2e, Table 6].
3.2.2 Sport
Sport was associated with BSI-18 Somatization, Depression, and Global Severity Index sub-scores, PCSS Symptom Severity scores, and BESS totals (Table 5), whereby football players had lower (better) Somatization [Exp(B) = 0.433, 0.258–0.728], Depression [Exp(B) = 0.383, 0.233–0.631], Global Severity Index [Exp(B) = 0.457, 0.305–0.685] sub-scores, and PCSS Symptom Severity scores [Exp(B) = 0.377, 0.262–0.541]. Contact sport athletes also had lower (better) Somatization sub-scores [Exp(B) = 0.515, 0.297–0.893], PCSS Symptom Severity scores [Exp(B) = 0.633, 0.434–0.924], and BESS total scores (B = − 3.323, − 5.447 to − 1.199, Table 6).
3.2.3 eAFE
Later eAFE was associated with lower (worse) ImPACT Verbal Memory (B = − 0.201, − 0.558 to 0.157), Visual Memory (B = − 0.277, − 0.707 to 0.152), and Visual Motor Speed (B = − 0.173, − 0.386 to 0.039) composite scores (Tables 5, 6).
3.2.4 Concussion History
Positive concussion history was associated with (i) higher (worse) BSI-18 Somatization [Exp(B) = 1.351, 1.208–1.511], Depression [Exp(B) = 1.415, 1.271–1.575], Anxiety [Exp(B) = 1.418, 1.270–1.584], and Global Severity Index [Exp(B) = 1.423, 1.310–1.547] sub-scores; (ii) higher (better) ImPACT Verbal Memory (B = 1.187, 0.466–1.908) and Visual Motor Speed (B = 1.211, 0.783–1.639) composite scores, and lower (better) Reaction Time (B = − 0.015, − 0.021 to − 0.008) composite scores; (iii) higher (worse) PCSS Symptom Severity scores [Exp(B) = 1.283, 1.188–1.384]; and (iv) lower (better) BESS total scores (B = − 0.454, − 0.866 to − 0.041) (Tables 5, 6).
3.2.5 Neurodevelopmental History
Positive neurodevelopmental history was associated with (i) higher (worse) BSI-18 Somatization [Exp(B) = 1.327, 1.126–1.564], Depression [Exp(B) = 1.376, 1.176–1.610], Anxiety [Exp(B) = 1.357, 1.154–1.594], and Global Severity Index [Exp(B) = 1.373, 1.212–1.556] sub-scores; (ii) lower (worse) ImPACT Verbal Memory (B = − 3.111, − 4.221 to − 2.000), Visual Memory (B = − 3.200, − 4.534 to − 1.866), and Visual Motor Speed (B = − 2.233, − 2.892 to − 1.574) composite scores, and higher (worse) Reaction Time (B = 0.028, 0.017–0.039) composite scores; and (iii) higher (worse) PCSS Symptom Severity (B = 1.446, 1.290–1.623) (Tables 5, 6).
3.2.6 Migraine History
Positive migraine history was associated with higher (worse) BSI-18 Somatization [Exp(B) = 1.531, 1.257–1.866], Depression [Exp(B) = 1.402, 1.155–1.702], Anxiety [Exp(B) = 1.451, 1.188–1.770], Global Severity Index [Exp(B) = 1.465, 1.255–1.713] sub-scores, and PCSS Symptom Severity scores [Exp(B) = 1.914, 1.657–2.210] (Tables 5, 6).
3.2.7 SES
High SES was associated with (i) higher (worse) BSI-18 Somatization [Exp(B) = 1.264, 1.017–1.568]; and (ii) higher (better) ImPACT Visual Memory (B = 1.969, 0.405–3.534) and Visual Motor Speed (B = 2.401, 1.628–3.174) composite scores, and lower (better) Reaction Time (B = − 0.014, − 0.026 to − 0.001) composite scores (Tables 5, 6). Middle High SES was associated with (i) higher (worse) BSI-18 Somatization [Exp(B) = 1.300, 1.053–1.605]; and (ii) higher (better) ImPACT Visual Memory (B = 1.675, 0.160–3.189) and Visual Motor Speed (B = 1.270, 0.522–2.019) composite scores (Tables 5, 6). Middle SES was also associated with higher (better) ImPACT Visual Motor Speed composite scores (1.181, 0.385–1.977). Low SES was associated with higher (worse) ImPACT Reaction Time composite scores (B = 0.027, 0.012–0.042). Middle High, Middle, and Low SES were all associated with higher (worse) PCSS Symptom Severity [Exp(B) = 1.151, 1.004–1.320; Exp(B) = 1.245, 1.078–1.439; Exp(B) = 1.218, 1.043–1.422, respectively].
3.2.8 Race and Ethnicity
African American race was associated with (i) higher (worse) BSI-18 Depression [Exp(B) = 1.213, 1.064–1.384] and lower (better) Anxiety [Exp(B) = 0.738, 0.639–0.852] sub-scores; (ii) lower (worse) ImPACT Verbal Memory (B = − 2.996, − 3.867 to − 2.125), Visual Memory (B = − 4.497, − 5.542 to − 3.452), and Visual Motor Speed (B = − 2.911, − 3.428 to − 2.393), and slower (worse) Reaction Time (B = 0.060, 0.051–0.069) composite scores; and (iii) higher (worse) PCSS Symptom Severity scores [Exp(B) = 1.182, 1.077–1.298] (Tables 5, 6). Other Race was associated with (i) higher (worse) BSI-18 Depression [Exp(B) = 1.338, 1.157–1.547] sub-scores; (ii) lower (worse) ImPACT Verbal Memory (B = − 1.811, − 2.816 to − 0.806), Visual Memory (B = − 1.984, − 3.191 to − 0.776), and slower (worse) Reaction Time (B = 0.021, 0.012–0.030) composite scores; and (iii) higher (worse) PCSS Symptom Severity scores [Exp(B) = 1.113, 1.000–1.237] (Tables 5, 6). Non-Hispanic or Latino ethnicity was associated with higher (better) Verbal Memory (B = 1.325, 0.454–2.196), Visual Memory (B = 2.087, 1.041–3.133), and Visual Motor Speed (B = 1.305, 0.788–1.822), and faster (better) Reaction Time (B = − 0.020, − 0.029 to − 0.011) composite scores.
3.2.9 Subgroup Analysis
Given the similarities between this work and our previous work [15], we performed a subgroup analysis among men, excluding football players. Results were largely unchanged in the subgroup analysis with two exceptions. First, the sport-by-eAFE interaction was associated with BESS total scores (Χ2 = 4.740, p = 0.029), whereby later AFE to contact sport participation was associated with higher (worse) BESS total scores (B = 0.254, 95% confidence interval 0.025–0.483, Fig. 2f). Second, sport was associated with BSI-18 Global Severity Index sub-scores (Χ2 = 4.380, p = 0.036), whereby contact sport athletes had lower (better) Global Severity Index [Exp(B) = 0.628, 0.406–0.971] sub-scores.
4 Discussion
The purpose of this study was to examine the association between eAFE to RHI and neurocognitive performance, psychological health, and postural stability in healthy collegiate student athletes by sex. We examined ten outcome measures, five self-report symptom measures, four neurocognitive composite scores, and one measure of postural stability. In a cohort of over 6000 current NCAA men and women student athletes from collision, contact, and non-contact sports, we observed no association between earlier eAFE to RHI exposure and worse neurocognitive performance, greater psychological distress, or worse postural stability. Men and women participating in contact sports performed similarly or better than those in non-contact sports on cognitive and balance testing.
Among the women’s cohort, the eAFE-by-sport interaction was associated with ImPACT Verbal Memory and Visual Memory composite scores, but in the opposite direction, whereby earlier eAFE to contact sports was associated with higher (better) composite scores. Among the men’s cohort, the eAFE-by-sport interaction was associated with BSI-18 Depression and Global Severity Index subscales and for PCSS symptom severity scores, also in the opposite direction, whereby earlier eAFE to football was associated with less psychological distress and symptom severity. Parameter estimates were very small (e.g., among women involved in contact sport participation, ImPACT Verbal Memory estimates decreased 0.397 points for each year increase in eAFE) suggesting these results have minimal practical or clinical relevance (Tables 3, 6).
In former professional and amateur football players, tackle football participation prior to age 12 was associated with worse later-in-life cognitive function, neuropsychiatric outcomes, and findings on neuroimaging [6,7,8,9,10], although other researchers studying retired NFL players did not find worse outcomes in association with when the person started playing football [11,12,13,14]. In current collegiate NCAA football players, exposure to tackle football prior to age 12 was not associated with behavioral, cognitive, psychological, or physical outcomes [15,16,17,18,19]. Studies examining the association between eAFE to repetitive head impacts and both short- and long-term outcomes have been limited to football players; thus, the results may not be generalizable to other groups. This is the second study (see [17]) to include sports other than football and the first to include women student athletes. The results herein extend the previous findings suggesting that among both collegiate men and women, earlier contact sport participation was not associated with worse neurocognitive performance, greater psychological distress, or worse postural stability. Overall, these findings support previous work suggesting that early exposure to RHI through collision and contact sport participation may not be associated with neurocognitive deficits in current collegiate student athletes [15,16,17,18,19]. However, later-in-life effects cannot be estimated or inferred from this study. These findings should not be used alone to inform safety and/or policy decisions regarding youth sport participation. Future longitudinal studies are needed to examine the association between contact sport participation at all levels and later-life outcomes [38]. Later-life outcomes must be interpreted with the understanding of the important biopsychosocial benefits of participating in athletics and team sports during youth and adolescence [38].
Men and women involved in contact sports, compared to men and women involved in non-contact sports, (1) report comparable or fewer physical and emotional symptoms, and (2) perform comparably or better on the four neurocognitive composite scores from ImPACT. Although these are current NCAA student athletes, several large-scale, cross-sectional and longitudinal studies have found that playing high school football is not associated with later-in-life neurodegenerative diseases, cognitive impairment, or psychological health problems [39,40,41]. However, also consistent with previous work among former high school football players[42], we reported that participants with a positive concussion history did have worse outcomes on some measures (Tables 3, 6), albeit the effects were small.
We controlled other factors that may affect neurocognitive testing, such as neurodevelopmental history, migraine history, socioeconomic status, race, and ethnicity. Although these parameters made significant contributions to the models, most were not practically or clinically meaningful (i.e., parameter estimates were small, Tables 3 and 6). Exceptions were that women and men with a neurodevelopmental history of academic difficulties obtained lower scores on ImPACT, as did African American men. Demographically adjusted normative data are often used in interpreting neurocognitive performance [43]. Despite initial evidence that race/ethnicity was not associated with baseline ImPACT composite scores [44], more recent work from a large cohort suggested that Black/African American race was associated with lower Verbal/Visual memory (β = − 0.113; p = 0.026) and Visual Motor Speed/Reaction Time performance (β = − 0.242; p < 0.001) [18]. Subsequent work suggested that academic aptitude either fully or partially mediated these race effects, further highlighting the importance of considering longstanding intellectual functioning and aptitudes and sociodemographic variables when interpreting neurocognitive performance [19].
4.1 Limitations
We used a multimodal baseline concussion assessment battery to examine the association between eAFE to RHI and neurocognitive performance, psychological distress, and postural stability in a relatively diverse cohort of men and women NCAA student athletes. While many of the outcome measures included herein are recommended as assessments in the recent NIH/NINDS Sports Concussion Common Data Elements [45], they may not be sensitive enough to identify more subtle deficits associated with RHI. Incorporating more sensitive measures of neuroradiological, neurobehavioral, and neuropsychological testing may add to this body of work. We also do not know the previous number of times each athlete completed the tests; considering practice and learning effects on the ImPACT and BESS tests, this could have created some degree of confound in the results. Furthermore, we did not draw comparisons between men and women. The sample sizes were very different between men (N = 4448) and women (N = 1891), and many of the parameters that we controlled for (e.g., ADHD) have different prevalence between men and women, so we chose to address these findings separately. Finally, many of the parameters included in the model were self-reported, which is an unavoidable limitation and self-reporting is a typical and practical method for acquiring this type of information in behavioral and medical research [46].
5 Conclusion
We observed no association between earlier eAFE to RHI exposure and worse neurocognitive function, greater psychological distress, or worse postural stability in men or women collision and contact sport collegiate student athletes. This study suggests that exposure to contact sports, such as ice hockey, lacrosse, wrestling, and soccer, during early adolescence is unrelated to baseline neurocognitive function, psychological distress, and postural stability in healthy NCAA collegiate student athletes.
Data availability statement
The CARE Consortium datasets generated and analyzed during the current study are available in the FITBIR repository (https://fitbir.nih.gov/).
References
Lebel C, Walker L, Leemans A, Phillips L, Beaulieu C. Microstructural maturation of the human brain from childhood to adulthood. Neuroimage. 2008;40(3):1044–55.
Blakemore S, Choudhury S. Development of the adolescent brain: implications for executive function and social cognition. J Child Psychol Psychiatry. 2006;47(3–4):296–312.
Snook L, Paulson L, Roy D, Phillips L, Beaulieu C. Diffusion tensor imaging of neurodevelopment in children and young adults. Neuroimage. 2005;26(4):1164–73.
Courchesne E, Chisum HJ, Townsend J, Cowles A, Covington J, Egaas B, et al. Normal brain development and aging: quantitative analysis at in vivo MR imaging in healthy volunteers. Radiology. 2000;216(3):672–82.
Shaw P, Kabani NJ, Lerch JP, Eckstrand K, Lenroot R, Gogtay N, et al. Neurodevelopmental trajectories of the human cerebral cortex. J Neurosci. 2008;28(14):3586–94.
Stamm JM, Koerte IK, Muehlmann M, Pasternak O, Bourlas AP, Baugh CM, et al. Age at first exposure to football is associated with altered corpus callosum white matter microstructure in former professional football players. J Neurotrauma. 2015;32(22):1768–76.
Schultz V, Stern RA, Tripodis Y, Stamm J, Wrobel P, Lepage C, et al. Age at first exposure to repetitive head impacts is associated with smaller thalamic volumes in former professional American football players. J Neurotrauma. 2018;35(2):278–85.
Alosco ML, Kasimis AB, Stamm JM, Chua AS, Baugh CM, Daneshvar DH, et al. Age of first exposure to American football and long-term neuropsychiatric and cognitive outcomes. Transl Psychiatry. 2017;7(9):e1236.
Stamm JM, Bourlas AP, Baugh CM, Fritts NG, Daneshvar DH, Martin BM, et al. Age of first exposure to football and later-life cognitive impairment in former NFL players. Neurology. 2015;84(11):1114–20.
Alosco ML, Mez J, Tripodis Y, Kiernan PT, Abdolmohammadi B, Murphy L, et al. Age of first exposure to tackle football and chronic traumatic encephalopathy. Ann Neurol. 2018;83(5):886–901.
Solomon GS, Kuhn AW, Zuckerman SL, Casson IR, Viano DC, Lovell MR, et al. Participation in Pre-High school football and neurological, neuroradiological, and neuropsychological findings in later life: a study of 45 retired National Football League players. Am J Sports Med. 2016;44(5):1106–15.
Casson IR, Viano DC, Haacke EM, Kou Z, LeStrange DG. Is there chronic brain damage in retired NFL players? neuroradiology, neuropsychology, and neurology examinations of 45 retired players. Sports Health. 2014;6(5):384–95.
Roberts AL, Pascual-Leone A, Speizer FE, Zafonte RD, Baggish AL, Taylor Jr H, et al. Exposure to American football and neuropsychiatric health in former National Football League players: findings from the football players health study. Am J Sports Med. 2019. (epub ahead of print).
Montenigro PH, Alosco ML, Martin BM, Daneshvar DH, Mez J, Chaisson CE, et al. Cumulative head impact exposure predicts later-life depression, apathy, executive dysfunction, and cognitive impairment in former high school and college football players. J Neurotrauma. 2017;34(2):328–40.
Caccese JB, DeWolf RM, Kaminski TW, Broglio SP, McAllister TW, McCrea M, et al. Estimated age of first exposure to american football and neurocognitive performance amongst NCAA male student-athletes: a cohort study. Sports Med. 2019;49(3):477–87.
Brett BL, Huber DL, Wild A, Nelson LD, McCrea MA. Age of first exposure to American football and behavioral, cognitive, psychological, and physical outcomes in high school and collegiate football players. Sports Health. 2019. (epub ahead of print).
Caccese JB, Iverson GL, Cameron K, Houston MN, McGinty J, Jackson JC, et al. Estimated age of first exposure to contact sports is not associated with greater symptoms or worse cognitive functioning in US service academy athletes. J Neurotrauma. 2019. (epub ahead of print).
Houck Z, Asken B, Clugston J, Perlstein W, Bauer R. Socioeconomic status and race outperform concussion history and sport participation in predicting collegiate athlete baseline neurocognitive scores. J Int Neuropsychol Soc. 2018;24(1):1–10.
Houck ZM, Asken BM, Bauer RM, Caccese JB, Buckley TA, McCrea MA, et al. Academic aptitude mediates the relationship between socioeconomic status and race in predicting ImPACT scores in college athletes. Clin Neuropsychol. 2019. (epub ahead of print).
Daniel RW, Rowson S, Duma SM. Head impact exposure in youth football. Ann Biomed Eng. 2012;40(4):976–81.
Cobb BR, Urban JE, Davenport EM, Rowson S, Duma SM, Maldjian JA, et al. Head impact exposure in youth football: elementary school ages 9–12 years and the effect of practice structure. Ann Biomed Eng. 2013;41(12):2463–73.
Mihalik JP, Guskiewicz KM, Marshall SW, Blackburn JT, Cantu RC, Greenwald RM. Head impact biomechanics in youth hockey: comparisons across playing position, event types, and impact locations. Ann Biomed Eng. 2012;40(1):141–9.
Reed N, Taha T, Keightley M, Duggan C, McAuliffe J, Cubos J, et al. Measurement of head impacts in youth ice hockey players. Int J Sports Med. 2010;31(11):826–33.
Chrisman SP, Mac Donald CL, Friedman S, Andre J, Rowhani-Rahbar A, Drescher S, et al. Head impact exposure during a weekend youth soccer tournament. J Child Neurol. 2016;31(8):971–8.
Hanlon EM, Bir CA. Real-time head acceleration measurement in girls’ youth soccer. Med Sci Sports Exerc. 2012;44(6):1102–8.
Broglio SP, McCrea M, McAllister T, Harezlak J, Katz B, Hack D, et al. A national study on the effects of concussion in collegiate athletes and US military service academy members: the NCAA–DoD concussion assessment, research and education (CARE) consortium structure and methods. Sports Med. 2017;47(7):1437–51.
Katz BP, Kudela M, Harezlak J, McCrea M, McAllister T, Broglio SP, et al. Baseline performance of NCAA athletes on a concussion assessment battery: a report from the CARE consortium. Sports Med. 2018;48(8):1971–85.
Broglio SP, Katz BP, Zhao S, McCrea M, McAllister T, CARE Consortium Investigators. Test–retest reliability and interpretation of common concussion assessment tools: Findings from the NCAA–DoD CARE consortium. Sports Med. 2017;48(5):1255–68.
Meachen S, Hanks RA, Millis SR, Rapport LJ. The reliability and validity of the brief symptom inventory-18 in persons with traumatic brain injury. Arch Phys Med Rehabil. 2008;89(5):958–65.
Kelly KC, Jordan EM, Joyner AB, Burdette GT, Buckley TA. National collegiate athletic association division I athletic trainers’ concussion-management practice patterns. J Athl Train. 2014;49(5):665–73.
Buckley TA, Burdette G, Kelly K. Concussion-management practice patterns of National Collegiate Athletic Association division II and III athletic trainers: how the other half lives. J Athl Train. 2015;50(8):879–88.
Schatz P, Pardini JE, Lovell MR, Collins MW, Podell K. Sensitivity and specificity of the ImPACT test battery for concussion in athletes. Arch Clin Neuropsychol. 2006;21(1):91–9.
Broglio SP, Katz BP, Zhao S, McCrea M, McAllister T, CARE Consortium Investigators. Test–retest reliability and interpretation of common concussion assessment tools: findings from the NCAA–DoD CARE consortium. Sports Med. 2018;48(5):1255–68.
Buckley TA, Oldham JR, Caccese JB. Postural control deficits identify lingering post-concussion neurological deficits. J Sport Health Sci. 2016;5(1):61–9.
Finnoff JT, Peterson VJ, Hollman JH, Smith J. Intrarater and interrater reliability of the balance error scoring system (BESS). PM&R. 2009;1(1):50–4.
Hollingshead AD. Four factor index of social status. New Haven, CT: Yale University; 1975. https://sociology.yale.edu/sites/default/files/files/yjs_fall_2011.pdf#page=21
Brooks BL, Silverberg N, Maxwell B, Mannix R, Zafonte R, Berkner PD, et al. Investigating effects of sex differences and prior concussions on symptom reporting and cognition among adolescent soccer players. Am J Sports Med. 2018;46(4):961–8.
Alosco ML, Stern RA. Youth exposure to repetitive head impacts from tackle football and long-term neurologic outcomes: a review of the literature, knowledge gaps and future directions, and societal and clinical implications. Semin Pediatr Neurol. 2019. (epub ahead of print).
Janssen PH, Mandrekar J, Mielke MM, Ahlskog JE, Boeve BF, Josephs K, Savica R. High school football and late-life risk of neurodegenerative syndromes, 1956–1970. Mayo Clin Proc. 2017;92(1):66–71).
Savica R, Parisi JE, Wold LE, Josephs KA, Ahlskog JE. High school football and risk of neurodegeneration: a community-based study. Mayo Clin Proc. 2012;87(4):335–40.
Deshpande SK, Hasegawa RB, Rabinowitz AR, Whyte J, Roan CL, Tabatabaei A, et al. Association of playing high school football with cognition and mental health later in life. JAMA Neurol. 2017;74(8):909–18.
Schmidt JD, Terry DP, Ko J, Newell KM, Miller LS. Balance regularity among former high school football players with or without a history of concussion. J Athl Train. 2018;53(2):109–14.
Manly JJ. Advantages and disadvantages of separate norms for African Americans. Clin Neuropsychol. 2005;19(2):270–5.
Kontos AP, Elbin RJ III, Covassin T, Larson E. Exploring differences in computerized neurocognitive concussion testing between African American and white athletes. Arch Clin Neuropsychol. 2010;25(8):734–44.
Broglio SP, Kontos AP, Levin H, Schneider K, Wilde EA, Cantu RC, et al. The National Institute of Neurological Disorders and Stroke and Department of Defense Sport-Related Concussion Common Data Elements version 1.0 recommendations. J Neurotrauma. 2018;35(23):2776–83.
Stone AA, Bachrach CA, Jobe JB, Kurtzman HS, Cain VS. The science of self-report: implications for research and practice. London: Psychology Press; 1999.
Acknowledgements
This publication was made possible, in part, with support from the Grand Alliance CARE Consortium, funded by the NCAA and the DoD. The USAMRAA, Ford Detrick, MD, USA, is the awarding and administering acquisition office. This work was supported by the Office of the Assistant Secretary of Defense for Health Affairs through the Psychological Health and Traumatic Brain Injury Program under Award no. W81XWH-14-2-0151. Opinions, interpretations, conclusions and recommendations are those of the authors and are not necessarily endorsed by the DoD (DHP funds).
Contributing CARE Consortium investigators include: Justus D. Ortega, PhD (Humboldt State University); Nicholas Port, PhD (Indiana University); Joshua T. Goldman, MD, MBA and Christopher C. Giza, MD (University of California, Los Angeles); Holly J. Benjamin MD (University of Chicago); James R. Clugston, MD, MS (University of Florida); Luis A. Feigenbaum, DPT, ATC (University of Miami); James T. Eckner, MD, MS (University of Michigan); Jason P. Mihalik, PhD, CAT(C), ATC (University of North Carolina at Chapel Hill); Christina L. Master, MD (University of Pennsylvania); Anthony P. Kontos, PhD (University of Pittsburgh Medical Center); Jonathan Jackson, MD (United States Air Force Academy); Adam Susmarski, DO (United States Naval Academy); Stefan Duma, PhD (Virginia Tech); Christopher M. Miles, MD (Wake Forest University); Laura Lintner DO (Winston-Salem University).
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Funding
This publication was made possible, in part, with support from the Grand Alliance CARE Consortium, funded by the NCAA and the DoD. The USAMRAA, Ford Detrick, MD, USA, is the awarding and administering acquisition office. This work was supported by the Office of the Assistant Secretary of Defense for Health Affairs through the Psychological Health and Traumatic Brain Injury Program under Award no. W81XWH-14-2-0151. Opinions, interpretations, conclusions and recommendations are those of the authors and are not necessarily endorsed by the DoD (DHP funds).
Conflict of interest
Grant L. Iverson serves as a scientific advisor for BioDirection, Inc., Sway Operations, LLC, and Highmark, Inc. He has a consulting practice in forensic neuropsychology, including expert testimony. He has received research funding from several test publishing companies, including ImPACT Applications, Inc., CNS Vital Signs, and Psychological Assessment Resources (PAR, Inc.). He has received research funding as a principal investigator from the National Football League, and salary support as a collaborator from the Harvard Integrated Program to Protect and Improve the Health of National Football League Players Association Members. He acknowledges unrestricted philanthropic support from ImPACT Applications, Inc., the Heinz Family Foundation, the Boston Bolts, the Mooney-Reed Charitable Foundation, and the Spaulding Research Institute. Jaclyn B. Caccese, Barry Bodt, Thomas W. Kaminski, Kelsey Bryk, Jessie Oldhamn, Steven P. Broglio, Thomas W. McAllister, Michael McCrea, and Thomas A. Buckley declare that they have no additional conflicts of interest to disclose.
Research involving human participants
The study was performed in accordance with the standards of ethics outlined in the Declaration of Helsinki.
Informed consent
All study procedures were reviewed and approved by the University of Michigan IRB, the US Army Medical Research and Materiel Command Human Research Protection Office (HRPO), as well the local IRB at each of the performance sites. Participants provided written informed consent prior to participation.
Additional information
The members of CARE Consortium Investigators are listed in Acknowledgements section.
This article is part of a Topical Collection on The NCAA-DoD Concussion Assessment, Research and Education (CARE) Consortium.
Rights and permissions
About this article
Cite this article
Caccese, J.B., Bodt, B.A., Iverson, G.L. et al. Estimated Age of First Exposure to Contact Sports and Neurocognitive, Psychological, and Physical Outcomes in Healthy NCAA Collegiate Athletes: A Cohort Study. Sports Med 50, 1377–1392 (2020). https://doi.org/10.1007/s40279-020-01261-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-020-01261-4