Abstract
Background
Maternal-perinatal interventions delivered during pregnancy or childbirth have unique characteristics that impact the health-related quality of life (HRQoL) of the mother, fetus, and newborn child. However, maternal-perinatal cost-utility analyses (CUAs) often only consider either maternal or child health outcomes. Challenges include, but are not limited to, measuring fetal, newborn, and infant health outcomes, and assessing their impact on maternal HRQoL. It is also important to recognize the impact of maternal-perinatal health on family members’ HRQoL (i.e., family spillover effects) and to incorporate these effects in maternal-perinatal CUAs.
Objective
The aim was to systematically review the methods used to include health outcomes of pregnant women, fetuses, and children and to incorporate family spillover effects in maternal-perinatal CUAs.
Methods
A literature search was conducted in Medline, Embase, EconLit, Cochrane Collection, Cumulative Index to Nursing and Allied Health Literature (CINAHL), International Network of Agencies for Health Technology Assessment (INAHTA), and the Pediatric Economic Database Evaluation (PEDE) databases from inception to 2020 to identify maternal-perinatal CUAs that included health outcomes for pregnant women, fetuses, and/or children. The search was updated to December 2022 using PEDE. Data describing how the health outcomes of mothers, fetuses, and children were measured, incorporated, and reported along with the data on family spillover effects were extracted.
Results
Out of 174 maternal-perinatal CUAs identified, 62 considered the health outcomes of pregnant women, and children. Among the 54 quality-adjusted life year (QALY)-based CUAs, 12 included fetal health outcomes, the impact of fetal loss on mothers’ HRQoL, and the impact of neonatal demise on mothers’ HRQoL. Four studies considered fetal health outcomes and the effects of fetal loss on mothers’ HRQoL. One study included fetal health outcomes and the impact of neonatal demise on maternal HRQoL. Furthermore, six studies considered the impact of neonatal demise on maternal HRQoL, while four included fetal health outcomes. One study included the impact of fetal loss on maternal HRQoL. The remaining 26 only included the health outcomes of pregnant women and children. Among the eight disability-adjusted life year (DALY)-based CUAs, two measured fetal health outcomes. Out of 174 studies, only one study included family spillover effects. The most common measurement approach was to measure the health outcomes of pregnant women and children separately. Various approaches were used to assess fetal losses in terms of QALYs or DALYs and their impact on HRQoL of mothers. The most common integration approach was to sum the QALYs or DALYs for pregnant women and children. Most studies reported combined QALYs and incremental QALYs, or DALYs and incremental DALYs, at the family level for pregnant women and children.
Conclusions
Approximately one-third of maternal-perinatal CUAs included the health outcomes of pregnant women, fetuses, and/or children. Future CUAs of maternal-perinatal interventions, conducted from a societal perspective, should aim to incorporate health outcomes for mothers, fetuses, and children when appropriate. The various approaches used within these CUAs highlight the need for standardized measurement and integration methods, potentially leading to rigorous and standardized inclusion practices, providing higher-quality evidence to better inform decision-makers about the costs and benefits of maternal-perinatal interventions. Health Technology Assessment agencies may consider providing guidance for interventions affecting future lives in future updates.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Maternal-perinatal interventions are unique because they impact the health outcomes of pregnant women, fetuses, and children, requiring the inclusion of quality-adjusted life years (QALYs) or disability-adjusted life years (DALYs) of all these individuals in cost-utility analyses (CUAs) from a societal perspective when appropriate. |
Only about one-third of the CUAs of maternal-perinatal intervention consider the health outcomes of pregnant women, fetuses, and/or children, and only one study considered family spillover effects, potentially resulting in an incomplete understanding of their benefits and inequitable policymaking. |
The methods for quantifying fetal losses in QALYs or DALYs and their effects on maternal health-related quality of life are not well-established; further conceptual and empirical work is needed. |
1 Background
Maternal-perinatal interventions are those delivered during pregnancy or childbirth. Effective maternal-perinatal interventions, whether in the form of screening, diagnostic, preventive, or therapeutic interventions, can improve the short-term and long-term health and well-being of the mother and child [1,2,3,4,5,6]. They have been shown to significantly decrease maternal mortality, fetal death, and neonatal mortality [7,8,9,10]. For example, maternal vaccination during influenza and other infectious diseases reduces morbidity and mortality for pregnant women and their newborns [2, 9,10,11,12]. Prenatal congenital heart disease (CHD) screening, followed by appropriate treatments, is associated with improved newborn survival and health outcomes [13,14,15,16,17]. Smoking cessation during pregnancy reduces congenital disabilities, preterm birth, and low birth weight, providing long-term benefits for mothers and infants [18,19,20,21]. Nutritional interventions such as folic acid and iron supplementation for pregnant women in low-income countries have been correlated with long-term health benefits for mothers and the healthier development of fetuses [4, 22, 23]. However, it is important to note that some maternal-perinatal treatments aimed at improving the fetus’s health may entail a risk to the mother’s health and well-being and vice versa [24].
Maternal-perinatal health also affects the health and well-being of fathers, partners, and other offspring [25,26,27,28,29,30,31,32]. These effects are referred to as ‘family spillover effects’ [33,34,35,36,37]. Family spillover effects can occur during pregnancy and persist after childbirth if the child and/or mother experiences illness or disability [26, 38,39,40,41,42]. Family spillover effects stem from two sources: the family effect and the caregiving effect. Family effects (‘caring about others’) arise from the direct impact of patients’ health on the physical health, psychological health, emotional well-being, quality of life, and overall well-being (including happiness and life satisfaction) of family members [33, 34, 43, 44]. These effects result when family members witness the suffering or declining health or death of their loved one (a child or mother or both). Caregiving effects (‘caring for others’) occur in those who provide care for a family member who is ill or has a disability [33, 34, 43, 44]. Participating in physically and emotionally demanding caregiving over extended durations may lead to psychological distress, depression, anxiety, and other mental health challenges for caregivers [45,46,47]. Consequently, interventions aimed at improving the health outcomes of mothers and/or children can have a ripple effect, leading to improved health and well-being for family members.
In both public and private healthcare systems, economic evaluations, often in the form of cost-utility analysis (CUA), are used to assess the value of interventions [48,49,50]. Health outcomes in CUA are expressed in quality-adjusted life years (QALYs) or disability-adjusted life years (DALYs). Current economic evaluation guidelines do not specify when the health outcomes of both pregnant women and children should be included. The guidelines recommend that CUAs from the societal perspective should consider all costs and outcomes related to the intervention [48,49,50]. For example, the second US Panel on Cost-Effectiveness in Health and Medicine recommends that the societal perspective include all costs and health benefits, regardless of who incurs the costs and to whom the benefits accrue [50]. This implies a need to consider a broader range of health outcomes in maternal-perinatal interventions, including maternal health outcomes during and after pregnancy, fetal health outcomes, child health outcomes after birth, and family spillover effects.
Most maternal-perinatal CUAs have focused on either maternal or child health outcomes. For example, Hulst et al. found that only 34% (28/82) of CUAs on obstetric care treatments considered both maternal and newborn health outcomes [51]. In another review of maternal and newborn health service use in low- and lower-middle-income countries, only three of 48 CUAs included both maternal and newborn health outcomes [52]. These reviews did not assess the inclusion of family spillover effects.
Several methodological challenges are apparent when incorporating mother and child health outcomes, including difficulties in measuring children’s health status, combining maternal and child health outcomes, and constructing decision-analytic models that capture health states of the child and mother. These challenges are not unique to maternal-perinatal CUAs – they also arise in CUAs of pediatric health interventions when considering family spillover effects [35,36,37, 53, 54]. Distinctive challenges specific to maternal-perinatal CUAs involve measuring and valuing fetal losses as part of composite health outcome measures like QALYs or DALYs and measuring the impact of fetal losses on parents’ health-related quality of life (HRQoL) [30, 51, 55]. HRQoL refers to how effectively a person functions in various aspects of life and their subjective sense of well-being across physical, mental, and social health domains [56]. These methodological challenges may have hindered the inclusion of maternal, fetal, and child health outcomes into CUAs.
Given these methodological challenges, understanding how existing maternal-perinatal CUAs have considered the health outcomes of pregnant women, fetuses, and offspring is a critical first step toward developing standardized methods. Gaining a better understanding of the strategies used will help to establish a uniform approach to ensure that future maternal-perinatal CUAs consider the health outcomes of all affected individuals. This systematic review aims to investigate the approaches used by researchers in measuring, incorporating, and reporting health outcomes of pregnant women, fetuses, and children in maternal-perinatal CUAs. For the purpose of this review, ‘children’ indicates offspring, including neonates, newborns, and/or infants, defined as those from birth to 1 year of age Additionally, this review assesses the frequency with which maternal-perinatal CUAs included family spillover effects and the approaches used to consider these effects.
2 Methods
2.1 Data Sources and Search Strategy
A literature search was conducted in seven electronic bibliographic databases: Medline, Embase, EconLit, Cochrane Collection, Cumulative Index to Nursing and Allied Health Literature (CINAHL), International Network of Agencies for Health Technology Assessment (INAHTA), and Pediatric Economic Database Evaluation (PEDE). These databases were searched from their inception to November 16, 2020. Additionally, the report repositories of two Health Technology Assessment (HTA) agencies, the Canadian Agency of Drugs and Technologies in Health (CADTH) and the National Institute for Health and Care Excellence (NICE), were also searched. The PEDE (pede.ccb.sickkids.ca/pede/index.jsp) database contains pediatric economic evaluations identified from a wide array of databases, including all of the databases originally searched and over 70 HTA academic and government websites [57]. The database is updated annually. PEDE was used to update the search through December 2022. A comprehensive search strategy was developed using search terms identified from published literature reviews and combining these search terms to identify cost-effectiveness analysis (CEA) and CUA affecting the fetus, premature birth, neonates, newborns, and pregnancy [36, 51, 52, 58, 59]. A combined search for both pediatric CUAs [44] and maternal-perinatal CUAs was conducted. The search strategy also incorporated terms to identify CEAs and CUAs in neonates, newborns, infants, children, and adolescents [44]. The search strategy for Medline is provided in Supplementary Fig. 1 (see the electronic supplementary material). The results of pediatric CUAs are reported elsewhere [44]. Articles generated by all database searches were compiled and duplicates were removed using EndNote X8.2. This review adhered to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement to the extent possible given the focus on methods [60]. The systematic review protocol was not registered.
2.2 Study Selection
Inclusion criteria were applied in two stages. Initially, a single researcher (RL) screened titles and abstracts, retaining studies that included pregnant women, mothers, fetuses, and newborns, with health outcomes measured in QALYs or DALYs. For this review, ‘fetus’ was defined as an offspring from the embryo stage until birth. The requirement for studies to be published in a peer-reviewed journal and written in English was incorporated at the first stage. After the title and abstract review, full texts of potentially eligible articles were acquired. Studies were excluded if study participants, or any subset of the sample presented by study authors, were not pregnant women, fetuses, or newborns. Next, studies were excluded if they were not original CUAs, i.e., economic evaluations that did not use QALYs or DALYs to quantify health outcomes. Lastly, studies must have included health outcomes of both pregnant women (mothers) and fetuses and/or newborns. Reference lists of included studies were manually searched for further studies. Maternal-perinatal CUAs that considered family spillover effects in addition to the health outcomes of pregnant women, fetuses, or newborns were included.
2.3 Data Extraction and Synthesis
The following descriptive data were extracted from eligible studies by a single researcher (RL) using a standardized data collection form: (1) bibliographic information; (2) country of the population; (3) disease/condition; (4) participants; (5) aim/objective of the study; (6) perspective and time horizon; and (7) intervention(s) and comparator(s). Furthermore, the following information was extracted from included studies: (1) reported health utilities or QALYs or DALYs of pregnant women and fetuses and/or newborns; (2) methods used to combine the health outcomes of pregnant women and fetuses and/or newborns; and (3) approaches for reporting the impacts of a maternal-perinatal intervention on pregnant women and children. Finally, data on family spillover effects, including (1) the family members considered, (2) the instrument or approach used to measure family spillover effects, and (3) the form in which these effects were measured and expressed (e.g., disutility or decrement in utility, QALYs, and QALY loss), were extracted. The approaches used to measure, integrate, and report the health outcomes of pregnant women and children were tabulated and described. Health conditions/diseases were categorized by the International Classification of Diseases 10th revision (ICD-10) guidelines [61]. The synthesis was carried out separately for QALY-based and DALY-based maternal-perinatal CUAs. Common methods and approaches were grouped conceptually. The lead author (RL) regularly met with co-authors to discuss the results. Any disagreements during the screening and data extraction phases were resolved through discussions with co-authors.
3 Results
3.1 Search Results
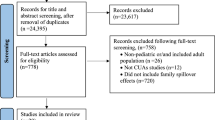
Figure 1 illustrates an overview of the search and retrieval processes. The literature search yielded 27,070 articles. Following duplicate removal, 24,395 articles were eligible for review. The full texts of 146 studies were accessed in the second stage. Among these, 39 studies met the inclusion criteria, and 107 studies were excluded for not being maternal-perinatal CUAs (n = 12), not being CUAs studies (n = 7), or not incorporating health outcomes of pregnant women and fetuses or newborns (n = 88). A search of the PEDE database between November 2020 and December 2022 yielded 41 maternal-perinatal CUAs, of which 17 met the inclusion criteria. An additional six studies were identified through manual reference searching of included studies and previous reviews. Thus, 62 studies were eligible.
3.2 Study Characteristics
Table 1 summarizes the 62 studies included in this review. Additional information on study characteristics is available in Supplementary Table 1 (see the electronic supplementary material). Fifty-four studies (87%) were QALY-based CUAs, and eight (13%) were DALY-based CUAs. The lower number of DALY-based CUAs reflected the fewer CUAs conducted in low- and middle-income countries. Delivery (n = 13, 21%) and diabetes mellitus in pregnancy (n = 7, 11%) were the most common disease or health condition (ICD-10 categories) studied. Screening or diagnostic strategy was the most common type of intervention (n = 21, 34%). Thirty-two studies (50%) were conducted from the societal perspective. Fifty-nine studies were model-based CUAs with inputs obtained from published literature, while the remaining three were trial-based CUAs [63,64,65].
3.3 Incorporating Health Outcomes of Pregnant Women, Fetuses, and Children in QALY-based CUAs
Table 2 summarizes the approaches used to measure, integrate, and report the health outcomes of pregnant women, fetuses, and newborns. Among the 54 QALY-based CUAs, 12 included fetal health outcomes, the impact of fetal loss on mothers’ HRQoL, and the impact of neonatal demise on mothers’ HRQoL. Four studies considered fetal health outcomes and the effects of fetal loss on mothers’ HRQoL. One study included fetal health outcomes and the impact of neonatal demise on maternal HRQoL. Furthermore, six studies considered the impact of neonatal demise on maternal HRQoL, while four included fetal health outcomes. One study included the impact of fetal loss on maternal HRQoL. The remaining 26 only included the health outcomes of pregnant women and children. Additional information is available in Supplementary Table 2 (see the electronic supplementary material).
3.3.1 Measuring Health Outcomes of Pregnant Women, Fetuses, and Children
In most studies (n = 50, 93%), health utilities were assessed separately for pregnant women and children; subsequently, separate QALYs were estimated. Fourteen of these studies also considered disutility and/or QALY losses due to disease(s) [64, 66,67,68,69,70,71,72,73,74,75,76,77,78]. Two studies measured health outcomes as QALY losses due to disease(s) or conditions in pregnant women and children [79, 80]. Two studies measured the QALYs of the pregnant woman–child dyad [81, 82]. This approach considers the joint QALYs of the pregnant woman and the child, rather than estimating separately and adding QALYs together. Finally, Culligan et al. measured the health utility of a pregnant woman–child dyad [83]. The health utilities of the pregnant woman–child dyad represent the combined current health states of both the pregnant woman and the child. Expert panels comprising healthcare providers assigned varying health utilities to clinical scenarios involving the mother and newborn. For instance, a utility of 0.5 was assigned for a mother with anal incontinence and a healthy child, and 0.35 for a child with severe permanent brachial plexus injury and anal incontinence in the mother. Subsequently, QALYs of the maternal–neonatal dyad were calculated.
Most QALY-based CUAs did not measure or include the value of fetal losses in their analyses. A fetal loss is defined at the loss of a fetus at any time during pregnancy [84]. Among the 21 studies (39%) that considered the value of fetus losses, researchers assigned a utility of '0' in 19 studies [63, 67, 70, 76, 85,86,87,88,89,90,91,92,93,94,95,96,97,98,99]. The remaining two studies measured the fetal losses as QALYs lost [77, 100]. The QALYs lost associated with a stillbirth was estimated at 25 QALYs, which is an approximation of the discounted QALYs at a rate of 3.5% per year expected from a full life expectancy of 80 years in perfect health.
Seventeen QALY-based CUAs (31%) considered the impact of fetal loss on the mother’s HRQoL. In 11 studies [67, 85,86,87, 89, 91, 93, 94, 96, 97, 99], a utility value of 0.92 was assigned to mothers who experienced a fetal loss, based on the research conducted by Kuppermann et al. [101]. However, the duration of the effects of fetal loss on maternal HRQoL varied widely across studies, ranging from 1 year to the remainder of the maternal life or until menopause. In some studies, the duration of the effects on maternal HRQoL varied based on the gestational age at which fetal death occurred. For example, intrauterine death was assumed to impact the mother's HRQoL for 10 years, while termination of pregnancy was assumed to impact the mother's HRQoL for 2 years [86]. In three studies [76, 98, 102], a utility value of 0.76 was assigned for mothers who experienced a fetal loss. Two studies measured the impact of fetal loss as a disutility to a mother’s utility [70, 103]. Disutility refers to the decrease in a woman’s utility caused by a fetal loss. For instance, Jones et al. applied a disutility of 0.1 to women who experienced fetal loss [70], but the authors were unclear about the duration of this disutility. Finally, Alkmark et al. conducted a CUA comparing induction of labor at 41 weeks with expectant management until 42 weeks alongside a randomized controlled trial [63]. The authors used the EQ-5D to measure mothers' HRQoL, including the impact of fetal loss.
Nineteen QALY-based studies (35%) incorporated the impact of neonatal death on the mother’s HRQoL. Neonatal demise (or death) is defined as the death of a live born infant within the first 28 completed days of life [84]. In 12 studies [75, 83, 85, 86, 88, 89, 91, 92, 94, 96, 104, 105], a utility of 0.92 was assigned to mothers who had experienced neonatal demise, while five studies used a utility of 0.76 [76, 87, 93, 97, 102]. Alkmark et al. measured the impact of neonatal death on mother’s HRQoL using the EQ-5D [63]. In the remaining study, the effects were measured using QALYs of the pregnant woman–child dyad. For instance, researchers assigned 15.3 QALYs when the mother lived without morbidity and the infant died [81]. The duration of the effect of a neonatal demise on mothers’ HRQoL varied across studies, ranging from 2 years to the remainder of the maternal life.
Twenty-one studies (39%) considered the impact on a mother’s HRQoL when her child had a disability or illnesses after birth [62,63,64, 67, 76, 77, 83, 85,86,87, 89, 92, 93, 96,97,98,99,100, 103, 105, 106]. For instance, Clennon et al. used a utility of 0.95 for mothers who had a neonate with CHD and 0.73 for mothers who had a neonate with cerebral palsy. Another example is premature birth, which was assumed to reduce maternal HRQoL by 0.04 in the first year only [67].
Only one study (2%) measured and incorporated the effects of a mother’s mortality and morbidity on a child's HRQoL [83]. Two studies explicitly assumed that the utility for neonatal outcomes was independent of maternal outcomes [73, 87], while the remaining studies did not provide justification for the exclusion.
3.3.2 Approaches for Integrating and Reporting Health Outcomes of Pregnant Women, Fetuses, and Children
The most commonly employed method (46, 85%) for integrating health outcomes of pregnant women and children was to sum the QALYs of pregnant women and children in each comparator group, subsequently estimating the combined QALYs for pregnant women and children at the family level (Table 2). Additional information is available in Supplementary Table 2 (see the electronic supplementary material). As described in Sect. 3.3.1, three studies [81, 83, 102] measured the health outcomes of a pregnant woman–child dyad; therefore, there was no need to integrate the health outcomes of pregnant women and children. Of the 54 included studies, 42 (78%) reported combined QALYs and incremental QALYs for pregnant women and children at the family level. Seven studies (13%) reported combined QALYs and incremental QALYs at the family level while also providing separate QALYs and incremental QALYs for pregnant women and children [62, 70,71,72, 74, 90, 109]. The remaining five studies (9%) only reported QALYs and incremental QALYs separately for pregnant women and children [65, 73, 85, 107, 108].
3.4 Approaches for Measuring, Integrating, and Reporting Health Outcomes of Pregnant Women, Fetuses, and Children in DALY-Based CUAs
Table 3 summarizes the approaches employed by researchers to assess, incorporate, and report the health outcomes of pregnant women, fetuses, and children in DALY-based CUAs. Additional information is available in Supplementary Table 1 (see the electronic supplementary material). All eight included studies measured the DALYs of pregnant women and children separately [65, 110,111,112,113,114,115,116]. Two studies considered the value of fetal loss in their analysis [112, 113]. For instance, in a CUA examining maternal influenza immunization in Mali, Orenstein et al. assigned a value of 57.28 DALYs for stillbirth [113]. This value was estimated based on the years of future life lost, calculated using life tables for life expectancy at birth for Mali [117].
Regarding integration, all eight studies calculated DALYs averted separately for pregnant women and children, and summed DALYs averted in each comparator group. Subsequently, the combined incremental DALYs averted of pregnant women and children were estimated at the family level. In terms of reporting, three studies reported combined DALYs averted and incremental DALYs averted for pregnant women and children at the family level [110, 111, 115], while five studies reported separate DALYs averted and incremental DALYs averted, as well as combined DALYs averted and incremental DALYs averted at the family level [65, 112,113,114, 116].
3.5 Approaches for Measuring, Incorporating, and Reporting Family Spillover Effects
Finally, among the 54 included maternal-perinatal CUAs, only one QALY-based CUA considered family spillover effects on fathers. Rowley et al. conducted a CUA of prenatal screening for cystic fibrosis carriers and utilized the time trade-off (TTO) method to measure family spillover effects on fathers [62]. Parents, including fathers, were asked about the effects of having a child with cystic fibrosis on their own quality of life. The authors separately estimated QALYs for the father, mother, and child. Subsequently, the QALYs for the father and mother were combined, and then the QALYs for the father, mother, and child were aggregated in each comparator group to calculate family QALYs. The authors reported the combined QALYs and the between-group incremental QALYs at the family level. None of the DALY-based maternal-perinatal CUAs considered family spillover effects.
4 Discussion
This review found that only about one-third of the maternal-perinatal CUAs included the health outcomes of both pregnant women and children, despite strong evidence supporting the positive impact of maternal-perinatal interventions on their health and well-being [1,2,3,4,5, 7, 10, 12, 18, 19, 22, 23, 110, 118]. Fetal losses were often excluded. Some studies considered the impact of fetal and neonatal demise on maternal HRQoL, but the methods varied widely. Only one study considered family spillover effects. The most common method of integrating the health outcomes of pregnant women and children was to sum their QALYs or DALYs, with most studies reporting combined QALYs and incremental QALYs gained, or DALYs averted and incremental DALYs averted, at the family level. The findings of the review are discussed with regard to unique features of maternal-perinatal intervention, including fetal health outcomes and the impact of fetal loss, neonatal death, and child’s illness on mothers’ well-being. It also discusses the measurement and incorporation of family spillover effects in maternal-perinatal health, in addition to integrating and reporting the child and mother health outcomes.
4.1 Unique Features of the Maternal-Perinatal Intervention
Maternal-perinatal interventions possess unique features that distinguish their CUAs from those of individual patient interventions. Firstly, maternal-perinatal health involves two distinct yet intricately related individuals—a mother and a fetus—whose health and well-being are inherently interconnected [24, 119,120,121,122]. In maternal-perinatal care, both mother and fetus are considered patients. Pregnancy is a period of rapid development, marked by significant psychological, anatomical, and physiological changes for both the mother and the fetus [123,124,125,126]. This sensitive period can be influenced by adversities, but it is also a time when positive factors can significantly impact the health and well-being of the pregnant woman and her offspring [125, 127,128,129,130,131,132,133]. These bidirectional effects continue to persist after childbirth if the child and/or mother experiences illness or disability. Secondly, maternal-perinatal interventions improve short-term health outcomes for both the mother, fetus, and child [134,135,136]. For example, hypertension in pregnancy, particularly preeclampsia, is a significant cause of maternal and perinatal morbidity and mortality [137,138,139]. Maternal-perinatal interventions such as calcium supplementation and low-dose aspirin can reduce the risk of preeclampsia in pregnant women [140,141,142,143]. Consequently, these interventions reduce the risk of preterm births and mitigate other adverse effects on newborns and mothers [140,141,142,143,144]. Finally, maternal-perinatal interventions improve the long-term health outcomes of both mothers and children. Pregnancy complications, such as gestational diabetes, pregnancy-induced hypertension, and maternal undernutrition lead to enduring consequences for mothers and their children [145,146,147,148,149,150]. For instance, preeclampsia is associated with later cardiovascular issues in mothers and neurodevelopmental delays in children, and inadequate maternal nutrition can result in intrauterine growth restriction, low birth weight, and an increased susceptibility to long-term health problems [151,152,153,154,155,156]. Consequently, effective maternal-perinatal intervention can improve long-term health outcomes of both the mother and the child [157,158,159].
Given these distinctive characteristics of maternal-perinatal interventions, it is crucial to incorporate the health outcomes of the mother, fetus, and child in CUAs conducted from a societal perspective. Excluding these outcomes undervalues the societal burden of maternal-perinatal illnesses and the effectiveness of maternal-perinatal interventions. Even in exceptional cases where the interests of the mother and fetus may conflict, necessitating medical decisions that prioritize one over the other, it is important to consider the health outcomes for both to reach impartial and informed conclusions [24, 160, 161].
There are several challenges in measuring the HRQoL of newborns and children. These challenges include, but are not limited to, the lack of validated preference-based HRQoL instruments for newborns or children under 6 years, bias from proxy assessment, and uncertainty regarding the relevance of descriptive classification systems to the experiences of children (with disabilities) [53, 162, 163]. Several new preference-based HRQoL instruments for younger children are in development, with the potential to overcome many limitations of current instruments in use [164, 165].
4.2 Incorporating Fetal Health Outcomes
This review identified 23 maternal-perinatal CUAs that included fetal health outcomes. Maternal-perinatal intervention presents a unique and often overlooked challenge in conducting economic evaluation: determining when to begin ‘counting’ a human’s life in the calculus. In public health research, including economic evaluations, fetal losses such as miscarriages and stillbirths have received less attention when compared to neonatal deaths [30, 55, 166, 167]. The effects of disease on very young infants, including those who die immediately after birth, are included in disease burden estimates. But the effects of diseases on fetuses that do not survive during pregnancy are often excluded. This, and findings from a previous review [55], highlight the infrequent incorporation of fetal losses in CUAs of maternal-perinatal interventions. When interventions impact the development of a fetus and the probability of birth (e.g., stillbirth prevention) and future life, failing to include these effects underestimates intervention benefits. Policy- or decision-makers who use the results of CUAs to guide resource allocation decisions are likely to assign a lower priority to interventions aimed at preventing stillbirths than they would if preventing a stillbirth were considered equivalent to saving a neonate’s life.
In some cases, including QALY losses or DALYs due to fetal death can be controversial. Consider prenatal screening tests identifying babies at risk of genetic or congenital disorders [168,169,170,171]. While these tests can provide expectant parents with time to prepare for a child with a clinical or genetic condition, they can also lead to expectant parents making informed decisions about terminating a pregnancy if the fetus has a harmful genetic or clinical condition. In the latter scenario, counting QALY losses or DALYs from the medical decision to terminate a fetus with a genetic condition who might have otherwise been born with the condition or disabilities becomes controversial [172]. Two women carrying fetuses with identical conditions could make different decisions based on their individual preferences and cultural, religious, and ethical beliefs [173]. Further, a termination may lead to future family planning that involves in vitro fertilization with pre-implantation genetic testing, resulting in a healthy embryo [174,175,176]. In this scenario, the consequence of prenatal genetic testing is not only the termination of an affected fetus but also the birth of a future healthy individual—‘a replacement child’ [177,178,179,180]. There is a positive utility effect of prenatal genetic screening and terminating the fetus on a future child [177, 178, 181]. As the use of emerging technologies like exome and genome sequencing for detecting fetal congenital anomalies early in pregnancy increases [171, 182,183,184,185], more expectant mothers (and parents) will make such decisions about whether or not to continue a pregnancy with fetal structural abnormalities or to have another child through in vitro fertilization. The question of whether to consider the QALY losses or DALYs associated with the fetal death in CUAs of such technologies becomes increasingly relevant.
The literature presents inconsistencies regarding the inclusion of fetal outcomes in economic evaluations. Simon et al. examined the impact of various definitions of the beginning of human life—at birth, at the biological threshold of physiological viability at 24 gestational weeks, and conception—on CUA outcomes by incorporating related life-year gained, QALYs gained, or DALYs averted [30]. However, they did not provide specific guidance on when to incorporate fetal outcomes. Phillips and Millum suggested that fetal deaths occurring after 28 weeks of gestational age, excluding those resulting from voluntary termination and miscarriage, should be considered in disease burden estimates [186]. Abel and Quaife suggested that the inclusion of fetal outcomes should depend on the purpose of the intervention and the pregnant woman’s preferences [173]. If the pregnant woman’s preference and the intervention aim to increase the likelihood of a live and healthy birth, fetal outcomes should be incorporated. On the other hand, if the pregnant woman's preference and the intervention do not involve continuing the pregnancy (e.g., termination), fetal outcomes should be excluded. The authors acknowledge that the situation becomes complicated if maternal preferences shift during pregnancy. Some studies suggest that decisions regarding the inclusion of fetal outcomes should be based on the primary aim of the intervention [172, 187]. For instance, CUAs of interventions aimed at reducing fertility (e.g., contraception or carrier screening) or those related to assisted reproductive technologies should exclude fetal outcomes [172], while interventions aimed at improving fetal and/or child health outcomes, which can potentially impact the child’s health and well-being throughout their lifetime, such as maternal vaccinations, nutritional fortification and supplementation, psychotherapy during pregnancy, or other parental interventions, should include fetal outcomes [55, 172].
Even if there is a consensus regarding the inclusion of fetal outcomes, challenges persist in determining how to quantify them within a composite health outcome measure, such as a QALY or DALY [30, 186, 188]. Some studies included in this review assigned values to fetal deaths equivalent to infant deaths following live births [67, 85,86,87,88,89,90, 112, 113]. However, whether fetal QALY losses or DALYs should carry weight equal to infant deaths in CUAs remains a subject of ongoing debate [186, 188,189,190]. There is also an ongoing discussion regarding whether to include miscarriages (the loss of a pregnancy before the 20th week of gestation) and whether to assign them QALY losses or DALYs equal to stillbirths (pregnancy loss after the 20th week of gestation) [166, 186, 188, 191]. The distinction between stillbirth and miscarriage lacks a universally accepted definition, with reporting policies varying between countries and within jurisdictions of the same country [192]. Some researchers argue that late stillbirths, occurring close to birth, should be given greater weight than earlier stillbirths or miscarriages [186]. Calculating QALYs or DALYs for fetal deaths at different stages of pregnancy poses both conceptual and empirical challenges.
The lack of standardized guidelines for including fetal outcomes and a method for measuring their value in QALYs or DALYs can introduce bias in cost-effectiveness estimates. Goldhaber-Fiebert and Brandeau examined CUAs of fertility interventions and found that fetal outcomes are often selectively chosen to demonstrate the cost-effectiveness of the related intervention and are often justified with flawed rationales [55]. A discussion of this complex, ethical and subjective matter regarding the moral status of future lives is beyond the scope of this paper. There is a need for further research, discussion, and guidance on the inclusion of fetal outcomes in CUAs of maternal-perinatal interventions. Determining where to draw the line and from which point onwards the health outcomes (health benefits) of the human life should be counted is crucial in the context of maternal-perinatal CUA.
4.3 Incorporating the Impacts of Fetal Loss, Newborn Death, and Child Illness on Maternal Health and Well-Being
This review found that 17 maternal-perinatal CUAs included the impact of fetal loss on mothers’ HRQoL and 19 considered the effects of neonatal demise on mothers’ HRQoL. Fetal losses and neonatal deaths can significantly impact maternal health and well-being, often resulting in depression, anxiety, and post-traumatic stress disorder (PTSD) [193,194,195,196,197,198]. Even when mothers (or parents) choose to terminate a pregnancy due to prenatal screening indicating fetal abnormalities, it can lead to long-lasting psychosocial sequelae for women, affecting their quality of life and their decisions about future pregnancies [199,200,201,202]. Failing to account for maternal bereavement effects (disutility experienced by mothers following the death of a fetus or newborn) can significantly underestimate the benefits of interventions. Our understanding of the bereavement effects is limited, and methods for estimating them are not well-developed. NICE recommends avoiding modelling bereavement effects in CUAs due to the underdeveloped methods [203]. The scarcity of research measuring how grief affects mothers' HRQoL may explain why bereavement effects are rarely incorporated. For example, out of 17 QALY-based studies that considered the impact of fetal loss on the mother’s HRQoL, 12 studies in this review obtained their utility estimates from a study conducted by Kuppermann and colleagues for mothers experiencing a fetal loss [101]. There is also a question of the duration of bereavement effects. For instance, it is reasonable to imagine that late pregnancy loss (e.g., stillbirths) may result in more prolonged bereavement effects compared to early pregnancy loss (e.g., miscarriage) [198]. Additionally, one could expect that the bereavement effects on mothers would be substantial around the time of the neonate’s death and then gradually decrease over time [204]. Studies included in this review used various durations, ranging from 1 year to the remainder of the maternal life or until menopause. The research on the magnitude and duration of bereavement effects on the lives of mothers is much needed.
Maternal-child health and pregnancy complications can lead to long-term effects on children related to behavioral and neurodevelopmental disorders [153, 205,206,207]. Additionally, they can place caregiving burdens on mothers, impacting their own health and well-being [37, 47, 54, 208,209,210]. Consequently, maternal-perinatal interventions directly or indirectly preventing such illnesses or disabilities in children positively impact mothers’ health and well-being. For instance, inadequate folic acid intake by prospective mothers increases the risk of spina bifida in children [211,212,213], a condition that can result in a wide range of disabilities depending on the lesion’s location, often requiring extensive caregiving [214, 215]. Caregivers of such children with disabilities, whether parents or other individuals, face various health-related problems [216, 217], and it is recommended that caregiver costs and health consequences be included in economic evaluation from a societal perspective [48, 49, 218]. Studies have demonstrated that taking folic acid supplements before conception significantly reduces the risk of spina bifida [118, 219,220,221,222,223,224], thereby preventing disabilities in children and eliminating the need for caregiving. Therefore, it is essential to consider the impacts of a child’s illnesses or disabilities on maternal HRQoL in CUAs of maternal-perinatal interventions from a societal perspective, such as folic acid fortification, to measure the full benefits.
4.4 Incorporating Family Spillover Effects of Maternal-Perinatal Health
Only one maternal-perinatal CUA considered the family spillover effects. Maternal-perinatal health also has an impact on the health and well-being of other family members, including fathers and other children. Traumatic events during pregnancy, such as adverse fetal and neonatal outcomes, can lead to depression, anxiety, and PTSD in fathers [225,226,227,228,229]. When a newborn experiences short-term or long-term disability, the father may take on a caregiving role [46, 230, 231]. Fathers often serve as the primary or sole wage earners in many low-income and middle-income countries. The responsibility predominantly falls on a father to provide adequate support and services to their children with disabilities or chronic illnesses, significantly affecting their quality of life [232]. Some conditions can be life-threatening for both mother and child [233, 234], with the potential for substantial spillover effects on fathers. Limited research exists on how maternal-perinatal health affects other children (or siblings), but studies have shown that stillbirths and the death of a twin have been associated with long-term effects on children’s physical and mental health [32]. When a child develops a disability or chronic illness after birth, it affects their unaffected siblings’ well-being [235, 236]. Additionally, some interventions could impact on future pregnancies, particularly increasing the risk of a subsequent uterine rupture or preterm birth [96].
Maternal-perinatal interventions that affect the health outcomes of both the mother and child and the extent of caregiving needed are likely to affect the health and well-being of other family members. Formally incorporating family spillover effects in economic evaluations is a relatively new concept that presents challenges in measurement, valuation, and incorporation [35, 37, 43, 53, 54, 237]. We previously reviewed the methods used for measuring, valuing, and incorporating family spillover effects in pediatric CUAs and suggested potential solutions to address some of these challenges, including collecting HRQoL data on family members alongside clinical trials, extending the time horizon of the study beyond the patient’s life expectancy to include bereavement effects on family members and considering family-level or parent–child dyad health utilities [44]. It is important to note that the total spillover QALY gains/losses for parents, family members, and others may be correlated with the size of the family and caregiving circle. Recently, an international task force was formed to provide guidance on the incorporation of family and caregiver health spillovers in cost-effectiveness and cost-utility analyses [218]. While the task force does not specifically provide recommendations for maternal-perinatal interventions, future researchers are encouraged to consult the SHEER Task Force recommendations on spillover effects when conducting CUAs for maternal-perinatal interventions.
4.5 Integrating and Reporting Health Outcomes of the Pregnant Woman and Child
This review found that the most common method of integrating mother and child health outcomes was the summation of QALYs or DALYs. Simple summation of mother and child QALYs may not always be appropriate. Applying equal weight to QALY gains in mother and child may unintentionally shift decisions toward benefiting the child because of the potential for a greater QALY gain due to a child’s longer life expectancy. Thus, assigning equal weights to QALYs gained by both the pregnant woman and child can be problematic, particularly when interventions are aimed at improving the mother’s health. Researchers have proposed applying a weighting factor to adjust for the importance of changes in health in family members relative to the primary patient [238,239,240,241]. The question of whether QALY gains in the mother be weighted equally to QALY gains in the child remains an area requiring further research.
Finally, in this review, most studies reported summed QALY and incremental QALY gains or summed DALYs and incremental DALYs averted for mothers and children at the family level. It is essential to report incremental QALY or incremental DALYs separately for both the mother and the child, as well as summed. This approach enhances transparency and provides a clearer understanding of where the benefits of the intervention accrue. Presenting both aggregated and disaggregated results for mothers and children helps inform allocation funding decision-making.
4.6 Limitations
There were four main limitations of this study. First, there was potential for missing relevant articles despite the comprehensive search strategy. This is due to the requirement for studies to be published in English and peer-reviewed journals, which may have excluded some eligible research. Second, only one reviewer conducted abstract and full-text screening, as well as data extraction. Third, the quality of the included studies was not assessed. There are no existing quality assessment tools or guidelines for evaluating methods used to incorporate health outcomes for mothers, fetuses, and children. As the aim was to systematically identify and report the diverse methods used by researchers rather than aggregating quantifiable estimates, a quality assessment of the included studies was not indicated. Finally, this review was limited by the information provided by the authors.
5 Conclusion
Maternal-perinatal interventions are unique because they influence the health and well-being of both the mother and the fetus, affecting the fetus's chances of a healthy birth, and the child’s health and well-being after birth. Only about one-third of the maternal-perinatal CUAs considered maternal and child health outcomes, potentially underestimating intervention benefits. Incorporating the health outcomes of the pregnant woman, fetus, and child presents unique methodological challenges. It is clear that further conceptual and empirical research is needed to standardize the inclusion and measurement of these health outcomes in maternal-perinatal CUAs. Future CUAs of maternal-perinatal interventions conducted from a societal perspective should also aim to incorporate family spillover effects. HTA agencies may consider providing specific guidance for interventions that impact future lives in their guideline updates.
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
References
O’Connor E, Rossom RC, Henninger M, Groom HC, Burda BU. Primary care screening for and treatment of depression in pregnant and postpartum women: evidence report and systematic review for the US preventive services task force. JAMA. 2016;315(4):388–406. https://doi.org/10.1001/jama.2015.18948.
Jarvis JR, Dorey RB, Warricker FDM, Alwan NA, Jones CE. The effectiveness of influenza vaccination in pregnancy in relation to child health outcomes: systematic review and meta-analysis. Vaccine. 2020;38(7):1601–13. https://doi.org/10.1016/j.vaccine.2019.12.056.
Baxter R, Bartlett J, Fireman B, Lewis E, Klein NP. Effectiveness of vaccination during pregnancy to prevent infant pertussis. Pediatrics. 2017. https://doi.org/10.1542/peds.2016-4091.
Bulloch RE, Lovell AL, Jordan VMB, McCowan LME, Thompson JMD, Wall CR. Maternal folic acid supplementation for the prevention of preeclampsia: a systematic review and meta-analysis. Paediatr Perinat Epidemiol. 2018;32(4):346–57. https://doi.org/10.1111/ppe.12476.
Lassi ZS, Mansoor T, Salam RA, Das JK, Bhutta ZA. Essential pre-pregnancy and pregnancy interventions for improved maternal, newborn and child health. Reprod Health. 2014;11(Suppl 1):S2. https://doi.org/10.1186/1742-4755-11-S1-S2.
Dewidar O, John J, Baqar A, Madani MT, Saad A, Riddle A, et al. Effectiveness of nutrition counseling for pregnant women in low- and middle-income countries to improve maternal and infant behavioral, nutritional, and health outcomes: a systematic review. Campbell Syst Rev. 2023;19(4): e1361. https://doi.org/10.1002/cl2.1361.
Bhutta ZA, Ahmed T, Black RE, Cousens S, Dewey K, Giugliani E, et al. What works? Interventions for maternal and child undernutrition and survival. Lancet. 2008;371(9610):417–40.
Jones G, Steketee RW, Black RE, Bhutta ZA, Morris SS, Bellagio Child Survival Study G. How many child deaths can we prevent this year? Lancet. 2003;362(9377):65–71. https://doi.org/10.1016/S0140-6736(03)13811-1.
Blencowe H, Lawn J, Vandelaer J, Roper M, Cousens S. Tetanus toxoid immunization to reduce mortality from neonatal tetanus. Int J Epidemiol. 2010;39(Suppl 1):i102–9. https://doi.org/10.1093/ije/dyq027.
Bhutta ZA, Das JK, Bahl R, Lawn JE, Salam RA, Paul VK, et al. Can available interventions end preventable deaths in mothers, newborn babies, and stillbirths, and at what cost? Lancet. 2014;384(9940):347–70.
Tamma PD, Ault KA, del Rio C, Steinhoff MC, Halsey NA, Omer SB. Safety of influenza vaccination during pregnancy. Am J Obstet Gynecol. 2009;201(6):547–52. https://doi.org/10.1016/j.ajog.2009.09.034.
Sahni LC, Olson SM, Halasa NB, Stewart LS, Michaels MG, Williams JV, et al. Maternal vaccine effectiveness against influenza-associated hospitalizations and emergency department visits in infants. JAMA Pediatr. 2024;178(2):176–84. https://doi.org/10.1001/jamapediatrics.2023.5639.
Landis BJ, Levey A, Levasseur SM, Glickstein JS, Kleinman CS, Simpson LL, Williams IA. Prenatal diagnosis of congenital heart disease and birth outcomes. Pediatr Cardiol. 2013;34(3):597–605. https://doi.org/10.1007/s00246-012-0504-4.
Mahle WT, Clancy RR, McGaurn SP, Goin JE, Clark BJ. Impact of prenatal diagnosis on survival and early neurologic morbidity in neonates with the hypoplastic left heart syndrome. Pediatrics. 2001;107(6):1277–82. https://doi.org/10.1542/peds.107.6.1277.
Bonnet D, Coltri A, Butera G, Fermont L, Le Bidois J, Kachaner J, Sidi D. Detection of transposition of the great arteries in fetuses reduces neonatal morbidity and mortality. Circulation. 1999;99(7):916–8. https://doi.org/10.1161/01.cir.99.7.916.
Donofrio MT, Moon-Grady AJ, Hornberger LK, Copel JA, Sklansky MS, Abuhamad A, et al. Diagnosis and treatment of fetal cardiac disease: a scientific statement from the American Heart Association. Circulation. 2014;129(21):2183–242. https://doi.org/10.1161/01.cir.0000437597.44550.5d.
Bonnet D. Impacts of prenatal diagnosis of congenital heart diseases on outcomes. Transl Pediatr. 2021;10(8):2241–9. https://doi.org/10.21037/tp-20-267.
Lumley J, Chamberlain C, Dowswell T, Oliver S, Oakley L, Watson L. Interventions for promoting smoking cessation during pregnancy. Cochrane Database Syst Rev. 2009;8(3):CD001055. https://doi.org/10.1002/14651858.CD001055.pub3.
Schneider S, Huy C, Schutz J, Diehl K. Smoking cessation during pregnancy: a systematic literature review. Drug Alcohol Rev. 2010;29(1):81–90. https://doi.org/10.1111/j.1465-3362.2009.00098.x.
Delcroix-Gomez C, Delcroix M-H, Jamee A, Gauthier T, Marquet P, Aubard Y. Fetal growth restriction, low birth weight, and preterm birth: effects of active or passive smoking evaluated by maternal expired CO at delivery, impacts of cessation at different trimesters. Tob Induc Dis. 2022;20(August):1–15.
Higgins ST, Nighbor TD, Kurti AN, Heil SH, Slade EP, Shepard DS, et al. Randomized controlled trial examining the efficacy of adding financial incentives to best practices for smoking cessation among pregnant and newly postpartum women. Prev Med. 2022;165: 107012.
Caffrey A, McNulty H, Rollins M, Prasad G, Gaur P, Talcott JB, et al. Effects of maternal folic acid supplementation during the second and third trimesters of pregnancy on neurocognitive development in the child: an 11-year follow-up from a randomised controlled trial. BMC Med. 2021;19(1):73. https://doi.org/10.1186/s12916-021-01914-9.
Pena-Rosas JP, De-Regil LM, Garcia-Casal MN, Dowswell T. Daily oral iron supplementation during pregnancy. Cochrane Database Syst Rev. 2015;2015(7):CD004736. https://doi.org/10.1002/14651858.CD004736.pub5.
D’Souza R, Shah PS, Sander B. Clinical decision analysis in perinatology. Acta Obstet Gynecol Scand. 2018;97(4):491–9. https://doi.org/10.1111/aogs.13264.
Philpott LF, Savage E, FitzGerald S, Leahy-Warren P. Anxiety in fathers in the perinatal period: a systematic review. Midwifery. 2019;76:54–101. https://doi.org/10.1016/j.midw.2019.05.013.
Paulson JF, Bazemore SD. Prenatal and postpartum depression in fathers and its association with maternal depression: a meta-analysis. JAMA. 2010;303(19):1961–9. https://doi.org/10.1001/jama.2010.605.
Ramchandani PG, Psychogiou L, Vlachos H, Iles J, Sethna V, Netsi E, Lodder A. Paternal depression: an examination of its links with father, child and family functioning in the postnatal period. Depress Anxiety. 2011;28(6):471–7. https://doi.org/10.1002/da.20814.
Cameron EE, Sedov ID, Tomfohr-Madsen LM. Prevalence of paternal depression in pregnancy and the postpartum: an updated meta-analysis. J Affect Disord. 2016;206:189–203. https://doi.org/10.1016/j.jad.2016.07.044.
Rogers AM, Youssef GJ, Teague S, Sunderland M, Le Bas G, Macdonald JA, et al. Association of maternal and paternal perinatal depression and anxiety with infant development: a longitudinal study. J Affect Disord. 2023;1(338):278–88. https://doi.org/10.1016/j.jad.2023.06.020.
Simon J, Petrou S, Gray A. The valuation of prenatal life in economic evaluations of perinatal interventions. Health Econ. 2009;18(4):487–94. https://doi.org/10.1002/hec.1375.
Fernandez-Sola C, Camacho-Avila M, Hernandez-Padilla JM, Fernandez-Medina IM, Jimenez-Lopez FR, Hernandez-Sanchez E, et al. Impact of perinatal death on the social and family context of the parents. Int J Environ Res Public Health. 2020;17(10):3421. https://doi.org/10.3390/ijerph17103421.
Burden C, Bradley S, Storey C, Ellis A, Heazell AE, Downe S, et al. From grief, guilt pain and stigma to hope and pride—a systematic review and meta-analysis of mixed-method research of the psychosocial impact of stillbirth. BMC Pregnancy Childbirth. 2016;16(1):1–12.
Bobinac A, van Exel NJA, Rutten FF, Brouwer WB. Caring for and caring about: disentangling the caregiver effect and the family effect. J Health Econ. 2010;29(4):549–56.
Bobinac A, van Exel NJ, Rutten FF, Brouwer WB. Health effects in significant others: separating family and care-giving effects. Med Decis Mak. 2011;31(2):292–8. https://doi.org/10.1177/0272989X10374212.
Lamsal R, Zwicker JD. Economic evaluation of interventions for children with neurodevelopmental disorders: opportunities and challenges. Appl Health Econ Health Policy. 2017;15(6):763–72. https://doi.org/10.1007/s40258-017-0343-9.
Lavelle TA, D’Cruz BN, Mohit B, Ungar WJ, Prosser LA, Tsiplova K, et al. Family spillover effects in pediatric cost-utility analyses. Appl Health Econ Health Policy. 2019;17(2):163–74. https://doi.org/10.1007/s40258-018-0436-0.
Wittenberg E, James LP, Prosser LA. Spillover effects on caregivers’ and family members’ utility: a systematic review of the literature. Pharmacoeconomics. 2019;37(4):475–99. https://doi.org/10.1007/s40273-019-00768-7.
Belza C, Avitzur Y, Ungar WJ, Stremler R, Fehlings D, Wales PW. Stress, anxiety, depression, and health-related quality of life in caregivers of children with intestinal failure receiving parenteral nutrition: a cross-sectional survey study. JPEN J Parenter Enteral Nutr. 2023;47(3):342–53. https://doi.org/10.1002/jpen.2461.
Bourke-Taylor H, Cotter C, Stephan R. Young children with cerebral palsy: families self-reported equipment needs and out-of-pocket expenditure. Child Care Health Dev. 2014;40(5):654–62. https://doi.org/10.1111/cch.12098.
Ireland MJ, Pakenham KI. The nature of youth care tasks in families experiencing chronic illness/disability: development of the Youth Activities of Caregiving Scale (YACS). Psychol Health. 2010;25(6):713–31. https://doi.org/10.1080/08870440902893724.
Pakenham KI. The nature of caregiving in multiple sclerosis: development of the caregiving tasks in multiple sclerosis scale. Mult Scler. 2007;13(7):929–38. https://doi.org/10.1177/1352458507076973.
Rogers B. Feeding method and health outcomes of children with cerebral palsy. J Pediatr. 2004;145(2 Suppl):S28-32. https://doi.org/10.1016/j.jpeds.2004.05.019.
Brouwer WBF. The inclusion of spillover effects in economic evaluations: not an optional extra. Pharmacoeconomics. 2019;37(4):451–6. https://doi.org/10.1007/s40273-018-0730-6.
Lamsal R, Yeh EA, Pullenayegum E, Ungar WJ. A systematic review of methods used by pediatric cost-utility analyses to include family spillover effects. Pharmacoeconomics. 2024;42(2):199–217. https://doi.org/10.1007/s40273-023-01331-1.
Bakula DM, Sharkey CM, Perez MN, Espeleta HC, Gamwell KL, Baudino M, et al. The relationship between parent distress and child quality of life in pediatric cancer: a meta-analysis. J Pediatr Nurs. 2020;50:14–9.
Cohn LN, Pechlivanoglou P, Lee Y, Mahant S, Orkin J, Marson A, Cohen E. Health outcomes of parents of children with chronic illness: a systematic review and meta-analysis. J Pediatr. 2020;218:166 e2-177. https://doi.org/10.1016/j.jpeds.2019.10.068.
Pinquart M. Parenting stress in caregivers of children with chronic physical condition—a meta-analysis. Stress Health. 2018;34(2):197–207. https://doi.org/10.1002/smi.2780.
Canadian Agency for Drugs and Technology in Health (CADTH). Guidelines for the economic evaluation of health technologies: Canada. 4th ed. Ottawa: CADTH; 2017.
National Institute of Health and Sciences (NICE). Guide to the methods of technology appraisal 2013; 2013 [cited 21 Feb 2022]. https://www.nice.org.uk/process/pmg9/chapter/foreword.
Sanders GD, Neumann PJ, Basu A, Brock DW, Feeny D, Krahn M, et al. Recommendations for conduct, methodological practices, and reporting of cost-effectiveness analyses: second panel on cost-effectiveness in health and medicine. JAMA. 2016;316(10):1093–103. https://doi.org/10.1001/jama.2016.12195.
Hulst SM, Brouwer W, Mol BW, van den Akker-van Marle ME. Challenges in economic evaluations in obstetric care: a scoping review and expert opinion. BJOG. 2020;127(11):1399–407. https://doi.org/10.1111/1471-0528.16243.
Mangham-Jefferies L, Pitt C, Cousens S, Mills A, Schellenberg J. Cost-effectiveness of strategies to improve the utilization and provision of maternal and newborn health care in low-income and lower-middle-income countries: a systematic review. BMC Pregnancy Childbirth. 2014;14:243. https://doi.org/10.1186/1471-2393-14-243.
Ungar WJ. Challenges in health state valuation in paediatric economic evaluation: are QALYs contraindicated? Pharmacoeconomics. 2011;29(8):641–52. https://doi.org/10.2165/11591570-000000000-00000.
Wittenberg E, Prosser LA. Disutility of illness for caregivers and families: a systematic review of the literature. Pharmacoeconomics. 2013;31(6):489–500. https://doi.org/10.1007/s40273-013-0040-y.
Goldhaber-Fiebert JD, Brandeau ML. Evaluating cost-effectiveness of interventions that affect fertility and childbearing: how health effects are measured matters. Med Decis Mak. 2015;35(7):818–46.
Karimi M, Brazier J. Health, health-related quality of life, and quality of life: what is the difference? Pharmacoeconomics. 2016;34(7):645–9. https://doi.org/10.1007/s40273-016-0389-9.
Ungar WJ, Santos MT. The Pediatric Economic Database Evaluation (PEDE) Project: establishing a database to study trends in pediatric economic evaluation. Med Care. 2003;41(10):1142–52. https://doi.org/10.1097/01.MLR.0000088451.56688.65.
Fisk NM, Atun R. Systematic analysis of research underfunding in maternal and perinatal health. BJOG. 2009;116(3):347–56. https://doi.org/10.1111/j.1471-0528.2008.02027.x.
Sacks E, Freeman PA, Sakyi K, Jennings MC, Rassekh BM, Gupta S, Perry HB. Comprehensive review of the evidence regarding the effectiveness of community-based primary health care in improving maternal, neonatal and child health: 3. Neonatal health findings. J Glob Health. 2017;7(1):1–12.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372.
World Health Organization. International statistical classification of diseases and related health problems 10th revision. 2019 [cited 2021 15 August]. https://icd.who.int/browse10/2019/en.
Rowley PT, Loader S, Kaplan RM. Prenatal screening for cystic fibrosis carriers: an economic evaluation. Am J Hum Genet. 1998;63(4):1160–74. https://doi.org/10.1086/302042.
Alkmark M, Wennerholm UB, Saltvedt S, Bergh C, Carlsson Y, Elden H, et al. Induction of labour at 41 weeks of gestation versus expectant management and induction of labour at 42 weeks of gestation: a cost-effectiveness analysis. BJOG. 2022;129(13):2157–65. https://doi.org/10.1111/1471-0528.16929.
Hunter R, Beardmore-Gray A, Greenland M, Linsell L, Juszczak E, Hardy P, et al. Cost-utility analysis of planned early delivery or expectant management for late preterm pre-eclampsia (PHOENIX). Pharmacoecon Open. 2022;6(5):723–33. https://doi.org/10.1007/s41669-022-00355-1.
Sicuri E, Bardaji A, Nhampossa T, Maixenchs M, Nhacolo A, Nhalungo D, et al. Cost-effectiveness of intermittent preventive treatment of malaria in pregnancy in southern Mozambique. PLoS ONE. 2010;5(10): e13407. https://doi.org/10.1371/journal.pone.0013407.
Chung A, Macario A, El-Sayed YY, Riley ET, Duncan B, Druzin ML. Cost-effectiveness of a trial of labor after previous cesarean. Obstet Gynecol. 2001;97(6):932–41. https://doi.org/10.1016/s0029-7844(01)01355-2.
Danyliv A, Gillespie P, O’Neill C, Tierney M, O’Dea A, McGuire BE, et al. The cost-effectiveness of screening for gestational diabetes mellitus in primary and secondary care in the Republic of Ireland. Diabetologia. 2016;59(3):436–44. https://doi.org/10.1007/s00125-015-3824-0.
Gilbert SA, Grobman WA, Landon MB, Spong CY, Rouse DJ, Leveno KJ, et al. Cost-effectiveness of trial of labor after previous cesarean in a minimally biased cohort. Am J Perinatol. 2013;30(1):11–20. https://doi.org/10.1055/s-0032-1333206.
Gilbert SA, Grobman WA, Landon MB, Varner MW, Wapner RJ, Sorokin Y, et al. Lifetime cost-effectiveness of trial of labor after cesarean in the United States. Value Health. 2013;16(6):953–64. https://doi.org/10.1016/j.jval.2013.06.014.
Jones M, Smith M, Lewis S, Parrott S, Coleman T. A dynamic, modifiable model for estimating cost-effectiveness of smoking cessation interventions in pregnancy: application to an RCT of self-help delivered by text message. Addiction. 2019;114(2):353–65. https://doi.org/10.1111/add.14476.
Mrus JM, Goldie SJ, Weinstein MC, Tsevat J. The cost-effectiveness of elective cesarean delivery for HIV-infected women with detectable HIV RNA during pregnancy. AIDS. 2000;14(16):2543–52. https://doi.org/10.1097/00002030-200011100-00017.
Schackman BR, Oneda K, Goldie SJ. The cost-effectiveness of elective Cesarean delivery to prevent hepatitis C transmission in HIV-coinfected women. AIDS. 2004;18(13):1827–34. https://doi.org/10.1097/00002030-200409030-00012.
Xu X, Ivy JS, Patel DA, Patel SN, Smith DG, Ransom SB, et al. Pelvic floor consequences of cesarean delivery on maternal request in women with a single birth: a cost-effectiveness analysis. J Womens Health (Larchmt). 2010;19(1):147–60. https://doi.org/10.1089/jwh.2009.1404.
van Hoek AJ, Campbell H, Amirthalingam G, Andrews N, Miller E. Cost-effectiveness and programmatic benefits of maternal vaccination against pertussis in England. J Infect. 2016;73(1):28–37. https://doi.org/10.1016/j.jinf.2016.04.012.
Little SE, Caughey AB. Acyclovir prophylaxis for pregnant women with a known history of herpes simplex virus: a cost-effectiveness analysis. Am J Obstet Gynecol. 2005;193(3 Pt 2):1274–9. https://doi.org/10.1016/j.ajog.2005.05.042.
Avram CM, Caughey AB, Norton ME, Sparks TN. Cost-effectiveness of exome sequencing versus targeted gene panels for prenatal diagnosis of fetal effusions and non-immune hydrops fetalis. Am J Obstet Gynecol MFM. 2022;4(6): 100724. https://doi.org/10.1016/j.ajogmf.2022.100724.
Dietz J, Plumb J, Banfield P, Soe A, Chehadah F, Chang-Douglass S, Rogers G. Immediate birth for women between 34 and 37 weeks of gestation with prolonged preterm prelabour rupture of membranes and detection of vaginal or urine group B streptococcus: an economic evaluation. BJOG. 2022;129(10):1779–89. https://doi.org/10.1111/1471-0528.17119.
Neuwahl SJ, Sharma AJ, Zhang P, Hoerger TJ. Postdelivery intervention to prevent type 2 diabetes and the cost-effectiveness of screening criteria for gestational diabetes. Prev Chronic Dis. 2022;19:E89. https://doi.org/10.5888/pcd19.220055.
Mrus JM, Tsevat J. Cost-effectiveness of interventions to reduce vertical HIV transmission from pregnant women who have not received prenatal care. Med Decis Mak. 2004;24(1):30–9.
VanDeusen A, Paintsil E, Agyarko-Poku T, Long EF. Cost effectiveness of option B plus for prevention of mother-to-child transmission of HIV in resource-limited countries: evidence from Kumasi, Ghana. BMC Infect Dis. 2015;15(1):130. https://doi.org/10.1186/s12879-015-0859-2.
Sinkey RG, Odibo AO. Vasa previa screening strategies: decision and cost-effectiveness analysis. Ultrasound Obstet Gynecol. 2018;52(4):522–9. https://doi.org/10.1002/uog.19098.
Cipriano L, Barth W Jr, Zaric G. The cost-effectiveness of targeted or universal screening for vasa praevia at 18–20 weeks of gestation in Ontario. BJOG. 2010;117(9):1108–18.
Culligan PJ, Myers JA, Goldberg RP, Blackwell L, Gohmann SF, Abell TD. Elective cesarean section to prevent anal incontinence and brachial plexus injuries associated with macrosomia—a decision analysis. Int Urogynecol J Pelvic Floor Dysfunct. 2005;16(1):19–28. https://doi.org/10.1007/s00192-004-1203-3. (discussion).
Pathirana J, Muñoz FM, Abbing-Karahagopian V, Bhat N, Harris T, Kapoor A, et al. Neonatal death: case definition and guidelines for data collection, analysis, and presentation of immunization safety data. Vaccine. 2016;34(49):6027–37.
Albright CM, Werner EF, Hughes BL. Cytomegalovirus screening in pregnancy: a cost-effectiveness and threshold analysis. Am J Perinatol. 2019;36(7):678–87. https://doi.org/10.1055/s-0038-1676495.
Bak GS, Shaffer BL, Madriago E, Allen A, Kelly B, Caughey AB, Pereira L. Impact of maternal obesity on fetal cardiac screening: which follow-up strategy is cost-effective? Ultrasound Obstet Gynecol. 2020;56(5):705–16.
Clennon EK, Pare E, Amato P, Caughey AB. Use of gestational surrogates for women with Eisenmenger syndrome: a cost-effectiveness analysis. J Matern Fetal Neonatal Med. 2021;34(4):526–31. https://doi.org/10.1080/14767058.2019.1610734.
Chen PY, Finkelstein EA, Ng MJ, Yap F, Yeo GSH, Rajadurai VS, et al. Incremental cost-effectiveness analysis of gestational diabetes mellitus screening strategies in Singapore. Asia Pac J Public Health. 2015;28(1):15–25. https://doi.org/10.1177/1010539515612908.
Powell JM, Frank ZC, Clark GV, Lo JO, Caughey AB. Expectant management of preterm premature rupture of membranes at 34 weeks: a cost effectiveness analysis. J Matern Fetal Neonatal Med. 2022;35(25):9136–44.
Wastlund D, Moraitis AA, Thornton JG, Sanders J, White IR, Brocklehurst P, et al. The cost-effectiveness of universal late-pregnancy screening for macrosomia in nulliparous women: a decision analysis. BJOG. 2019;126(10):1243–50. https://doi.org/10.1111/1471-0528.15809.
Chaiken SR, Hersh AR, Zimmermann MS, Ameel BM, Layoun VR, Caughey AB. Cost-effectiveness of influenza vaccination during pregnancy. J Matern Fetal Neonatal Med. 2022;35(25):5244–52. https://doi.org/10.1080/14767058.2021.1876654.
Chatroux IC, Hersh AR, Caughey AB. Herpes simplex virus serotyping in pregnant women with a history of genital herpes and an outbreak in the third trimester of pregnancy: a cost-effectiveness analysis. Obstet Gynecol. 2021;137(1):63–71. https://doi.org/10.1097/AOG.0000000000004181.
Hersh AR, Megli CJ, Caughey AB. Repeat screening for syphilis in the third trimester of pregnancy: a cost-effectiveness analysis. Obstet Gynecol. 2018;132(3):699–707. https://doi.org/10.1097/AOG.0000000000002795.
Kaimal AJ, Little SE, Odibo AO, Stamilio DM, Grobman WA, Long EF, et al. Cost-effectiveness of elective induction of labor at 41 weeks in nulliparous women. Am J Obstet Gynecol. 2011;204(2):137 e1-137 e9. https://doi.org/10.1016/j.ajog.2010.08.012.
Kastenberg ZJ, Hurley MP, Luan A, Vasu-Devan V, Spain DA, Owens DK, Goldhaber-Fiebert JD. The cost-effectiveness of pre-operative imaging for appendicitis after indeterminate ultrasound in the 2nd or 3rd trimester of pregnancy. Obstet Gynecol. 2013;122(4):821.
Packer CH, Hersh AR, Caughey AB. Fetoscopic compared with open repair of myelomeningocele: a 2-delivery cost-effectiveness analysis. Am J Obstet Gynecol MFM. 2021;3(6): 100434. https://doi.org/10.1016/j.ajogmf.2021.100434.
Powell JM, Hersh AR, Greiner KS, Frank ZC, Pilliod RA, Caughey AB. Obstetric management for stillbirth complicated by a prior cesarean delivery: a cost-effectiveness analysis. J Matern Fetal Neonatal Med. 2022;35(19):3684–93. https://doi.org/10.1080/14767058.2020.1837770.
Walker AR, Caughey AB. Positivity thresholds of HbA1c assay as a screening test for diabetes mellitus in the first trimester in high-risk populations. J Matern Fetal Neonatal Med. 2022;35(2):230–4. https://doi.org/10.1080/14767058.2020.1716213.
Robin AM, Hersh AR, John C, Caughey AB. Cost effectiveness of buprenorphine vs. methadone for pregnant people with opioid use disorder. J Matern Fetal Neonatal Med. 2022;35(25):4918–26. https://doi.org/10.1080/14767058.2021.1873266.
Round JA, Jacklin P, Fraser RB, Hughes RG, Mugglestone MA, Holt RI. Screening for gestational diabetes mellitus: cost-utility of different screening strategies based on a woman’s individual risk of disease. Diabetologia. 2011;54(2):256–63. https://doi.org/10.1007/s00125-010-1881-y.
Kuppermann M, Nease RF, Learman LA, Gates E, Blumberg B, Washington AE. Procedure-related miscarriages and down syndrome-affected births: implications for prenatal testing based on women’s preferences. Obstet Gynecol. 2000;96(4):511–6. https://doi.org/10.1016/s0029-7844(00)00969-8.
Cipriano LE, Barth WH Jr, Zaric GS. The cost-effectiveness of targeted or universal screening for vasa praevia at 18–20 weeks of gestation in Ontario. BJOG. 2010;117(9):1108–18. https://doi.org/10.1111/j.1471-0528.2010.02621.x.
Mistry H, Gardiner HM. The cost-effectiveness of prenatal detection for congenital heart disease using telemedicine screening. J Telemed Telecare. 2013;19(4):190–6. https://doi.org/10.1258/jtt.2012.120418.
Ohno MS, Sparks TN, Cheng YW, Caughey AB. Treating mild gestational diabetes mellitus: a cost-effectiveness analysis. Am J Obstet Gynecol. 2011;205(3):282 e1-282 e7. https://doi.org/10.1016/j.ajog.2011.06.051.
Schmidt EM, Hersh AR, Tuuli M, Cahill AG, Caughey AB. Considering criteria for active phase labor management of nulliparous women: a cost-effectiveness analysis. Am J Perinatol. 2023;40(1):99–105. https://doi.org/10.1055/s-0041-1728836.
Schmidt EM, Hersh AR, Tuuli M, Cahill AG, Caughey AB. Timing of active phase labor arrest diagnosis in nulliparous women: a cost-effectiveness analysis. J Matern Fetal Neonatal Med. 2022;35(25):6124–31. https://doi.org/10.1080/14767058.2021.1907334.
Nicholson WK, Fleisher LA, Fox HE, Powe NR. Screening for gestational diabetes mellitus: a decision and cost-effectiveness analysis of four screening strategies. Diabetes Care. 2005;28(6):1482–4. https://doi.org/10.2337/diacare.28.6.1482.
Wymer KM, Shih YC, Plunkett BA. The cost-effectiveness of a trial of labor accrues with multiple subsequent vaginal deliveries. Am J Obstet Gynecol. 2014;211(1):56 e1-56 e12. https://doi.org/10.1016/j.ajog.2014.01.033.
Xu J, Zhou F, Reed C, Chaves SS, Messonnier M, Kim IK. Cost-effectiveness of seasonal inactivated influenza vaccination among pregnant women. Vaccine. 2016;34(27):3149–55.
Adam T, Lim SS, Mehta S, Bhutta ZA, Fogstad H, Mathai M, et al. Cost effectiveness analysis of strategies for maternal and neonatal health in developing countries. BMJ. 2005;331(7525):1107. https://doi.org/10.1136/bmj.331.7525.1107.
Choi SE, Brandeau ML, Bendavid E. Cost-effectiveness of malaria preventive treatment for HIV-infected pregnant women in sub-Saharan Africa. Malar J. 2017;16(1):403. https://doi.org/10.1186/s12936-017-2047-x.
Goodman DM, Ramaswamy R, Jeuland M, Srofenyoh EK, Engmann CM, Olufolabi AJ, Owen MD. The cost effectiveness of a quality improvement program to reduce maternal and fetal mortality in a regional referral hospital in Accra, Ghana. PLoS ONE. 2017;12(7): e0180929. https://doi.org/10.1371/journal.pone.0180929.
Orenstein EW, Orenstein LA, Diarra K, Djiteye M, Sidibe D, Haidara FC, et al. Cost-effectiveness of maternal influenza immunization in Bamako, Mali: a decision analysis. PLoS ONE. 2017;12(2): e0171499. https://doi.org/10.1371/journal.pone.0171499.
Prinja S, Bahuguna P, Gupta A, Nimesh R, Gupta M, Thakur JS. Cost effectiveness of mHealth intervention by community health workers for reducing maternal and newborn mortality in rural Uttar Pradesh, India. Cost Eff Resour Alloc. 2018;16:25. https://doi.org/10.1186/s12962-018-0110-2.
Kurzawa Z, Cotton CS, Mazurkewich N, Verney A, Busch-Hallen J, Kashi B. Training healthcare workers increases IFA use and adherence: evidence and cost-effectiveness analysis from Bangladesh. Matern Child Nutr. 2021;17(2): e13124. https://doi.org/10.1111/mcn.13124.
Kim SY, Min KD, Jung SM, Russell LB, Toscano C, Minamisava R, et al. Cost-effectiveness of maternal pertussis immunization: Implications of a dynamic transmission model for low- and middle-income countries. Vaccine. 2021;39(1):147–57. https://doi.org/10.1016/j.vaccine.2020.09.012.
DALYs GBD, Collaborators H, Murray CJ, Barber RM, Foreman KJ, Abbasoglu Ozgoren A, et al. Global, regional, and national disability-adjusted life years (DALYs) for 306 diseases and injuries and healthy life expectancy (HALE) for 188 countries, 1990–2013: quantifying the epidemiological transition. Lancet. 2015;386(10009):2145–91. https://doi.org/10.1016/S0140-6736(15)61340-X.
Mai CT, Evans J, Alverson CJ, Yue X, Flood T, Arnold K, et al. Changes in spina bifida lesion level after folic acid fortification in the US. J Pediatr. 2022;249:59 e1-66 e1. https://doi.org/10.1016/j.jpeds.2022.06.023.
Cranley MS. Development of a tool for the measurement of maternal attachment during pregnancy. Nurs Res. 1981;30(5):281–4.
Muller ME. Development of the Prenatal Attachment Inventory. West J Nurs Res. 1993;15(2):199–211. https://doi.org/10.1177/019394599301500205. (discussion-5).
Ainsworth MD. Object relations, dependency, and attachment: a theoretical review of the infant-mother relationship. Child Dev. 1969;40(4):969–1025.
Federenko IS, Wadhwa PD. Women’s mental health during pregnancy influences fetal and infant developmental and health outcomes. CNS Spectr. 2004;9(3):198–206. https://doi.org/10.1017/s1092852900008993.
Moya J, Phillips L, Sanford J, Wooton M, Gregg A, Schuda L. A review of physiological and behavioral changes during pregnancy and lactation: potential exposure factors and data gaps. J Expo Sci Environ Epidemiol. 2014;24(5):449–58. https://doi.org/10.1038/jes.2013.92.
Soma-Pillay P, Nelson-Piercy C, Tolppanen H, Mebazaa A. Physiological changes in pregnancy: review articles. Cardiovasc J Afr. 2016;27(2):89–94.
Howland MA, Sandman CA, Glynn LM. Developmental origins of the human hypothalamic–pituitary–adrenal axis. Expert Rev Endocrinol Metab. 2017;12(5):321–39. https://doi.org/10.1080/17446651.2017.1356222.
Adane AA, Bailey HD, Morgan VA, Galbally M, Farrant BM, Marriott R, et al. The impact of maternal prenatal mental health disorders on stillbirth and infant mortality: a systematic review and meta-analysis. Arch Womens Ment Health. 2021;24(4):543–55. https://doi.org/10.1007/s00737-020-01099-9.
Davis EP, Narayan AJ. Pregnancy as a period of risk, adaptation, and resilience for mothers and infants. Dev Psychopathol. 2020;32(5):1625–39.
Davis EP, Glynn LM, Dunkel Schetter C, Hobel C, Chicz-Demet A, Sandman CA. Corticotropin-releasing hormone during pregnancy is associated with infant temperament. Dev Neurosci. 2005;27(5):299–305.
Burton GJ, Hempstock J, Jauniaux E. Nutrition of the human fetus during the first trimester—a review. Placenta. 2001;22(Suppl A):S70–7. https://doi.org/10.1053/plac.2001.0639.
Grigoriadis S, VonderPorten EH, Mamisashvili L, Tomlinson G, Dennis CL, Koren G, et al. The impact of maternal depression during pregnancy on perinatal outcomes: a systematic review and meta-analysis. J Clin Psychiatry. 2013;74(4):e321–41. https://doi.org/10.4088/JCP.12r07968.
Davis EP, Sandman CA. The timing of prenatal exposure to maternal cortisol and psychosocial stress is associated with human infant cognitive development. Child Dev. 2010;81(1):131–48.
Lahdepuro A, Lahti-Pulkkinen M, Pyhala R, Tuovinen S, Lahti J, Heinonen K, et al. Positive maternal mental health during pregnancy and mental and behavioral disorders in children: a prospective pregnancy cohort study. J Child Psychol Psychiatry. 2023;64(5):807–16. https://doi.org/10.1111/jcpp.13625.
Phua DY, Kee MZL, Meaney MJ. Positive maternal mental health, parenting, and child development. Biol Psychiatry. 2020;87(4):328–37. https://doi.org/10.1016/j.biopsych.2019.09.028.
World Health Organization and Aga Khan University. Essential Interventions, Commodities and Guidelines for Reproductive, Maternal, Newborn and Child Health: A global review of the key interventions related to Reproductive, Maternal, Newborn and Child Health (RMNCH). Geneva: PMNCH, World Health Organization; 2011.
Salam RA, Mansoor T, Mallick D, Lassi ZS, Das JK, Bhutta ZA. Essential childbirth and postnatal interventions for improved maternal and neonatal health. Reprod Health. 2014;11(Suppl 1):S3. https://doi.org/10.1186/1742-4755-11-S1-S3.
Bhutta ZA, Das JK, Rizvi A, Gaffey MF, Walker N, Horton S, et al. Evidence-based interventions for improvement of maternal and child nutrition: what can be done and at what cost? Lancet. 2013;382(9890):452–77.
Duley L. Pre-eclampsia and the hypertensive disorders of pregnancy. Br Med Bull. 2003;67(1):161–76. https://doi.org/10.1093/bmb/ldg005.
Davis EF, Lazdam M, Lewandowski AJ, Worton SA, Kelly B, Kenworthy Y, et al. Cardiovascular risk factors in children and young adults born to preeclamptic pregnancies: a systematic review. Pediatrics. 2012;129(6):e1552–61. https://doi.org/10.1542/peds.2011-3093.
Dimitriadis E, Rolnik DL, Zhou W, Estrada-Gutierrez G, Koga K, Francisco RPV, et al. Pre-eclampsia. Nat Rev Dis Primers. 2023;9(1):8. https://doi.org/10.1038/s41572-023-00417-6.
Askie LM, Duley L, Henderson-Smart DJ, Stewart LA, Group PC. Antiplatelet agents for prevention of pre-eclampsia: a meta-analysis of individual patient data. Lancet. 2007;369(9575):1791–8. https://doi.org/10.1016/S0140-6736(07)60712-0.
Duley L, Meher S, Hunter KE, Seidler AL, Askie LM. Antiplatelet agents for preventing pre-eclampsia and its complications. Cochrane Database Syst Rev. 2019. https://doi.org/10.1002/14651858.CD004659.pub3.
Imdad A, Jabeen A, Bhutta ZA. Role of calcium supplementation during pregnancy in reducing risk of developing gestational hypertensive disorders: a meta-analysis of studies from developing countries. BMC Public Health. 2011;11(Suppl 3):S18. https://doi.org/10.1186/1471-2458-11-S3-S18.
Roberge S, Bujold E, Nicolaides KH. Meta-analysis on the effect of aspirin use for prevention of preeclampsia on placental abruption and antepartum hemorrhage. Am J Obstet Gynecol. 2018;218(5):483–9. https://doi.org/10.1016/j.ajog.2017.12.238.
Jabeen M, Yakoob MY, Imdad A, Bhutta ZA. Impact of interventions to prevent and manage preeclampsia and eclampsia on stillbirths. BMC Public Health. 2011;11(Suppl 3):S6. https://doi.org/10.1186/1471-2458-11-S3-S6.
Black RE, Victora CG, Walker SP, Bhutta ZA, Christian P, de Onis M, et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet. 2013;382(9890):427–51. https://doi.org/10.1016/S0140-6736(13)60937-X.
Widen EM, Kahn LG, Cirillo P, Cohn B, Kezios KL, Factor-Litvak P. Prepregnancy overweight and obesity are associated with impaired child neurodevelopment. Matern Child Nutr. 2018;14(1): e12481. https://doi.org/10.1111/mcn.12481.
Martins VJ, Toledo Florencio TM, Grillo LP, do Carmo PFM, Martins PA, Clemente AP, et al. Long-lasting effects of undernutrition. Int J Environ Res Public Health. 2011;8(6):1817–46. https://doi.org/10.3390/ijerph8061817.
Li J, Song C, Li C, Liu P, Sun Z, Yang X. Increased risk of cardiovascular disease in women with prior gestational diabetes: a systematic review and meta-analysis. Diabetes Res Clin Pract. 2018;140:324–38. https://doi.org/10.1016/j.diabres.2018.03.054.
Camprubi Robles M, Campoy C, Garcia Fernandez L, Lopez-Pedrosa JM, Rueda R, Martin MJ. Maternal diabetes and cognitive performance in the offspring: a systematic review and meta-analysis. PLoS ONE. 2015;10(11): e0142583. https://doi.org/10.1371/journal.pone.0142583.
Turbeville HR, Sasser JM. Preeclampsia beyond pregnancy: long-term consequences for mother and child. Am J Physiol Renal Physiol. 2020;318(6):F1315–26. https://doi.org/10.1152/ajprenal.00071.2020.
Pittara T, Vyrides A, Lamnisos D, Giannakou K. Pre-eclampsia and long-term health outcomes for mother and infant: an umbrella review. BJOG. 2021;128(9):1421–30. https://doi.org/10.1111/1471-0528.16683.
Poon LC, Shennan A, Hyett JA, Kapur A, Hadar E, Divakar H, et al. The International Federation of Gynecology and Obstetrics (FIGO) initiative on preeclampsia (PE): a pragmatic guide for first trimester screening and prevention. Int J Gynaecol Obstet. 2019;145(Suppl 1):1.
Cortes-Albornoz MC, Garcia-Guaqueta DP, Velez-van-Meerbeke A, Talero-Gutierrez C. Maternal nutrition and neurodevelopment: a scoping review. Nutrients. 2021;13(10):3530. https://doi.org/10.3390/nu13103530.
Young MF, Ramakrishnan U. Maternal undernutrition before and during pregnancy and offspring health and development. Ann Nutr Metab. 2020;76(3):41–53.
Imdad A, Lassi Z, Salaam R, Bhutta ZA. Prenatal nutrition and nutrition in pregnancy: effects on long-term growth and development. Early nutrition and long-term health. Amsterdam: Elsevier; 2017. p. 3–24.
Horton R. Maternal and child undernutrition: an urgent opportunity. Lancet. 2008;371(9608):179. https://doi.org/10.1016/S0140-6736(07)61869-8.
Marshall NE, Abrams B, Barbour LA, Catalano P, Christian P, Friedman JE, et al. The importance of nutrition in pregnancy and lactation: lifelong consequences. Am J Obstet Gynecol. 2022;226(5):607–32. https://doi.org/10.1016/j.ajog.2021.12.035.
Borge TC, Aase H, Brantsaeter AL, Biele G. The importance of maternal diet quality during pregnancy on cognitive and behavioural outcomes in children: a systematic review and meta-analysis. BMJ Open. 2017;7(9): e016777. https://doi.org/10.1136/bmjopen-2017-016777.
Koletzko B, Godfrey KM, Poston L, Szajewska H, van Goudoever JB, de Waard M, et al. Nutrition during pregnancy, lactation and early childhood and its implications for maternal and long-term child health: the early nutrition project recommendations. Ann Nutr Metab. 2019;74(2):93–106. https://doi.org/10.1159/000496471.
Christian NT, Borges VF. What Dobbs means for patients with breast cancer. N Engl J Med. 2022;387(9):765–7.
Suran M. Treating cancer in pregnant patients after Roe v Wade overturned. JAMA. 2022;328(17):1674–6. https://doi.org/10.1001/jama.2022.13668.
Kwon J, Kim SW, Ungar WJ, Tsiplova K, Madan J, Petrou S. A systematic review and meta-analysis of childhood health utilities. Med Decis Mak. 2018;38(3):277–305. https://doi.org/10.1177/0272989X17732990.
Lamsal R, Finlay B, Whitehurst DGT, Zwicker JD. Generic preference-based health-related quality of life in children with neurodevelopmental disorders: a scoping review. Dev Med Child Neurol. 2020;62(2):169–77. https://doi.org/10.1111/dmcn.14301.
Furlong W, Rae C, Feeny D, Ghotra S, Breakey VR, Carter T, et al. Generic health-related quality of life utility measure for preschool children (health utilities preschool): design, development, and properties. Value Health. 2023;26(2):251–60. https://doi.org/10.1016/j.jval.2022.07.015.
Jabrayilov R, van Asselt ADI, Vermeulen KM, Volger S, Detzel P, Dainelli L, et al. A descriptive system for the Infant health-related quality of life instrument (IQI): Measuring health with a mobile app. PLoS ONE. 2018;13(8): e0203276. https://doi.org/10.1371/journal.pone.0203276.
Frøen JF, Cacciatore J, McClure EM, Kuti O, Jokhio AH, Islam M, Shiffman J. Stillbirths: why they matter. Lancet. 2011;377(9774):1353–66.
Michalski ST, Porter J, Pauli RM. Costs and consequences of comprehensive stillbirth assessment. Am J Obstet Gynecol. 2002;186(5):1027–34. https://doi.org/10.1067/mob.2002.122450.
de Jong A, Dondorp WJ, Frints SG, de Die-Smulders CE, de Wert GM. Advances in prenatal screening: the ethical dimension. Nat Rev Genet. 2011;12(9):657–63. https://doi.org/10.1038/nrg3036.
van den Berg M, Timmermans DR, ten Kate LP, van Vugt JM, van der Wal G. Informed decision making in the context of prenatal screening. Patient Educ Couns. 2006;63(1–2):110–7. https://doi.org/10.1016/j.pec.2005.09.007.
Santalahti P, Hemminki E, Latikka AM, Ryynanen M. Women’s decision-making in prenatal screening. Soc Sci Med. 1998;46(8):1067–76. https://doi.org/10.1016/s0277-9536(97)10038-7.
Monaghan KG, Leach NT, Pekarek D, Prasad P, Rose NC, Practice AP, Guidelines C. The use of fetal exome sequencing in prenatal diagnosis: a points to consider document of the American College of Medical Genetics and Genomics (ACMG). Genet Med. 2020;22(4):675–80. https://doi.org/10.1038/s41436-019-0731-7.
Luyten J, Verbeke E, Schokkaert E. To be or not to be: future lives in economic evaluation. Health Econ. 2022;31(1):258–65.
Abel L, Quaife M. A pregnant pause: rethinking economic evaluation in contraception and pregnancy. Value Health. 2022;25(1):32–5. https://doi.org/10.1016/j.jval.2021.07.009.
Mastenbroek S, Twisk M, van Echten-Arends J, Sikkema-Raddatz B, Korevaar JC, Verhoeve HR, et al. In vitro fertilization with preimplantation genetic screening. N Engl J Med. 2007;357(1):9–17. https://doi.org/10.1056/NEJMoa067744.
Vernimmen V, Paulussen AD, Dreesen JC, van Golde RJ, Zamani Esteki M, Coonen E, et al. Preimplantation genetic testing for neurofibromatosis type 1: more than 20 years of clinical experience. Eur J Hum Genet. 2023;31(8):918–24.
Mirzayeva D. Features of the course of pregnancy and childbirth after in vitro fertilization (IVF), taking into account the factor of infertility. J Educ Sci Med. 2023;1(1):12–7.
Garber AM, Fenerty JP. Costs and benefits of prenatal screening for cystic fibrosis. Med Care. 1991;29(5):473–89. https://doi.org/10.1097/00005650-199105000-00007.
Nielsen R, Gyrd-Hansen D. Prenatal screening for cystic fibrosis: an economic analysis. Health Econ. 2002;11(4):285–99. https://doi.org/10.1002/hec.652.
Shorey S, Lalor J, Pereira TL, Jarasiunaite-Fedosejeva G, Downe S. Decision-making and future pregnancies after a positive fetal anomaly screen: a scoping review. J Clin Nurs. 2023;32(17–18):5534–49. https://doi.org/10.1111/jocn.16628.
Lilford R. The health economics of infertility treatment. Glob Reprod Health. 2018;3(4): e23.
Petrou S. Methodological limitations of economic evaluations of antenatal screening. Health Econ. 2001;10(8):775–8. https://doi.org/10.1002/hec.636.
de Koning MA, Hoffer MJV, Nibbeling EAR, Bijlsma EK, Toirkens MJP, Adama-Scheltema PN, et al. Prenatal exome sequencing: a useful tool for the fetal neurologist. Clin Genet. 2022;101(1):65–77. https://doi.org/10.1111/cge.14070.
Fu F, Li R, Yu Q, Wang D, Deng Q, Li L, et al. Application of exome sequencing for prenatal diagnosis of fetal structural anomalies: clinical experience and lessons learned from a cohort of 1618 fetuses. Genome Med. 2022;14(1):123. https://doi.org/10.1186/s13073-022-01130-x.
Lord J, McMullan DJ, Eberhardt RY, Rinck G, Hamilton SJ, Quinlan-Jones E, et al. Prenatal exome sequencing analysis in fetal structural anomalies detected by ultrasonography (PAGE): a cohort study. Lancet. 2019;393(10173):747–57. https://doi.org/10.1016/S0140-6736(18)31940-8.
Chung WK, Berg JS, Botkin JR, Brenner SE, Brosco JP, Brothers KB, et al. Newborn screening for neurodevelopmental diseases: are we there yet? Am J Med Genet Part C Semin Med Genet. 2022:222–30.
Phillips J, Millum J. Valuing stillbirths. Bioethics. 2015;29(6):413–23. https://doi.org/10.1111/bioe.12120.
Chambers GM, Adamson GD, Eijkemans MJ. Acceptable cost for the patient and society. Fertil Steril. 2013;100(2):319–27. https://doi.org/10.1016/j.fertnstert.2013.06.017.
Kant C. Stillbirths: how should its rate be reported, its disability-adjusted-life-years (DALY), and stillbirths adjusted life expectancy. BMC Med Inform Decis Mak. 2019;19(1):133. https://doi.org/10.1186/s12911-019-0850-8.
Qureshi ZU, Millum J, Blencowe H, Kelley M, Fottrell E, Lawn JE, et al. Stillbirth should be given greater priority on the global health agenda. BMJ. 2015;351.
Jamison DT, Shahid-Salles SA, Jamison J, Lawn JE, Zupan J. Incorporating deaths near the time of birth into estimates of the global burden of disease. 2011.
Benatar D, Benatar M. A pain in the fetus: toward ending confusion about fetal pain. Bioethics. 2001;15(1):57–76. https://doi.org/10.1111/1467-8519.00212.
Tavares Da Silva F, Gonik B, McMillan M, Keech C, Dellicour S, Bhange S, et al. Stillbirth: case definition and guidelines for data collection, analysis, and presentation of maternal immunization safety data. Vaccine. 2016;34(49):6057–68. https://doi.org/10.1016/j.vaccine.2016.03.044.
Herbert D, Young K, Pietrusinska M, MacBeth A. The mental health impact of perinatal loss: a systematic review and meta-analysis. J Affect Disord. 2022;15(297):118–29. https://doi.org/10.1016/j.jad.2021.10.026.
Heazell AEP, Siassakos D, Blencowe H, Burden C, Bhutta ZA, Cacciatore J, et al. Stillbirths: economic and psychosocial consequences. Lancet. 2016;387(10018):604–16. https://doi.org/10.1016/S0140-6736(15)00836-3.
Kreicbergs U, Valdimarsdottir U, Onelov E, Henter JI, Steineck G. Anxiety and depression in parents 4–9 years after the loss of a child owing to a malignancy: a population-based follow-up. Psychol Med. 2004;34(8):1431–41. https://doi.org/10.1017/s0033291704002740.
Dias N, Friebert S, Donelan J, Schoemann AM, Morris A, Guard K, Grossoehme DH. Bereaved parents’ health outcomes following the death of their child. Clin Pract Pediatr Psychol. 2021;9(3):272.
Djelantik A, Smid GE, Mroz A, Kleber RJ, Boelen PA. The prevalence of prolonged grief disorder in bereaved individuals following unnatural losses: systematic review and meta regression analysis. J Affect Disord. 2020;15(265):146–56. https://doi.org/10.1016/j.jad.2020.01.034.
Mergl R, Quaatz SM, Edeler LM, Allgaier AK. Grief in women with previous miscarriage or stillbirth: a systematic review of cross-sectional and longitudinal prospective studies. Eur J Psychotraumatol. 2022;13(2):2108578. https://doi.org/10.1080/20008066.2022.2108578.
Davies V, Gledhill J, McFadyen A, Whitlow B, Economides D. Psychological outcome in women undergoing termination of pregnancy for ultrasound-detected fetal anomaly in the first and second trimesters: a pilot study. Ultrasound Obstet Gynecol. 2005;25(4):389–92. https://doi.org/10.1002/uog.1854.
Fuller AE, Horváth-Puhó E, Ray JG, Ehrenstein V, Sørensen HT, Cohen E. Mortality among parents of children with major congenital anomalies. Pediatrics. 2021;147(5).
Woolf-King SE, Anger A, Arnold EA, Weiss SJ, Teitel D. Mental health among parents of children with critical congenital heart defects: a systematic review. J Am Heart Assoc. 2017;6(2): e004862.
Png ME, Yang M, Taylor-Phillips S, Ratushnyak S, Roberts N, White A, et al. Benefits and harms adopted by health economic assessments evaluating antenatal and newborn screening programmes in OECD countries: a systematic review of 336 articles and reports. Soc Sci Med. 2022;314: 115428. https://doi.org/10.1016/j.socscimed.2022.115428.
National Institue for Health and Care Excellence. CHTE methods review. 2020. https://rees-france.com/wp-content/uploads/2020/12/2020-CHTE-2020-Health-related-quality-of-life-.pdf. Accessed 13 Aug 2023
Rogers CH, Floyd FJ, Seltzer MM, Greenberg J, Hong J. Long-term effects of the death of a child on parents’ adjustment in midlife. J Fam Psychol. 2008;22(2):203–11. https://doi.org/10.1037/0893-3200.22.2.203.
Smith B, Reyes T. Offspring neuroimmune consequences of maternal malnutrition: potential mechanism for behavioral impairments that underlie metabolic and neurodevelopmental disorders. Front Neuroendocrinol. 2017;47:109–22.
Bale TL, Baram TZ, Brown AS, Goldstein JM, Insel TR, McCarthy MM, et al. Early life programming and neurodevelopmental disorders. Biol Psychiatry. 2010;68(4):314–9. https://doi.org/10.1016/j.biopsych.2010.05.028.
Poston L, Caleyachetty R, Cnattingius S, Corvalan C, Uauy R, Herring S, Gillman MW. Preconceptional and maternal obesity: epidemiology and health consequences. Lancet Diabetes Endocrinol. 2016;4(12):1025–36. https://doi.org/10.1016/S2213-8587(16)30217-0.
Schieve LA, Gonzalez V, Boulet SL, Visser SN, Rice CE, Braun KVN, Boyle CA. Concurrent medical conditions and health care use and needs among children with learning and behavioral developmental disabilities, National Health Interview Survey, 2006–2010. Res Dev Disabil. 2012;33(2):467–76.
Martin CA, Papadopoulos N, Chellew T, Rinehart NJ, Sciberras E. Associations between parenting stress, parent mental health and child sleep problems for children with ADHD and ASD: systematic review. Res Dev Disabil. 2019;93: 103463.
Scherer N, Verhey I, Kuper H. Depression and anxiety in parents of children with intellectual and developmental disabilities: a systematic review and meta-analysis. PLoS ONE. 2019;14(7): e0219888. https://doi.org/10.1371/journal.pone.0219888.
Imbard A, Benoist JF, Blom HJ. Neural tube defects, folic acid and methylation. Int J Environ Res Public Health. 2013;10(9):4352–89. https://doi.org/10.3390/ijerph10094352.
Blom HJ. Folic acid, methylation and neural tube closure in humans. Birth Defects Res A Clin Mol Teratol. 2009;85(4):295–302. https://doi.org/10.1002/bdra.20581.
Pitkin RM. Folate and neural tube defects. Am J Clin Nutr. 2007;85(1):285S-S288. https://doi.org/10.1093/ajcn/85.1.285S.
Sawin KJ, Margolis RHF, Ridosh MM, Bellin MH, Woodward J, Brei TJ, Logan LR. Self-management and spina bifida: a systematic review of the literature. Disabil Health J. 2021;14(1): 100940. https://doi.org/10.1016/j.dhjo.2020.100940.
Simeonsson RJ, McMillen JS, Huntington GS. Secondary conditions in children with disabilities: spina bifida as a case example. Ment Retard Dev Disabil Res Rev. 2002;8(3):198–205. https://doi.org/10.1002/mrdd.10038.
Tilford JM, Grosse SD, Robbins JM, Pyne JM, Cleves MA, Hobbs CA. Health state preference scores of children with spina bifida and their caregivers. Qual Life Res. 2005;14(4):1087–98. https://doi.org/10.1007/s11136-004-3305-2.
Grosse SD, Flores AL, Ouyang L, Robbins JM, Tilford JM. Impact of spina bifida on parental caregivers: findings from a survey of Arkansas families. J Child Fam Stud. 2009;18(5):574–81. https://doi.org/10.1007/s10826-009-9260-3.
Henry E, Al-Janabi H, Brouwer W, Cullinan J, Engel L, Griffin S, et al. Recommendations for emerging good practice and future research in relation to family and caregiver health spillovers in health economic evaluations: a report of the SHEER task force. Pharmacoeconomics. 2024;42(3):343–62. https://doi.org/10.1007/s40273-023-01321-3.
Odewole OA, Williamson RS, Zakai NA, Berry RJ, Judd SE, Qi YP, et al. Near-elimination of folate-deficiency anemia by mandatory folic acid fortification in older US adults: reasons for Geographic and Racial Differences in Stroke study 2003–2007. Am J Clin Nutr. 2013;98(4):1042–7. https://doi.org/10.3945/ajcn.113.059683.
Atta CA, Fiest KM, Frolkis AD, Jette N, Pringsheim T, St Germaine-Smith C, et al. Global birth prevalence of spina bifida by folic acid fortification status: a systematic review and meta-analysis. Am J Public Health. 2016;106(1):e24-34. https://doi.org/10.2105/AJPH.2015.302902.
Castillo-Lancellotti C, Tur JA, Uauy R. Impact of folic acid fortification of flour on neural tube defects: a systematic review. Public Health Nutr. 2013;16(5):901–11. https://doi.org/10.1017/S1368980012003576.
Force USPST, Barry MJ, Nicholson WK, Silverstein M, Chelmow D, Coker TR, et al. Folic acid supplementation to prevent neural tube defects: US preventive services task force reaffirmation recommendation statement. JAMA. 2023;330(5):454–9. https://doi.org/10.1001/jama.2023.12876.
Kancherla V, Wagh K, Priyadarshini P, Pachón H, Oakley GP Jr. A global update on the status of prevention of folic acid-preventable spina bifida and anencephaly in year 2020: 30-year anniversary of gaining knowledge about folic acid’s prevention potential for neural tube defects. Birth Defects Res. 2022;114(20):1392–403.
Bergman JE, Otten E, Verheij JB, de Walle HE. Folic acid supplementation influences the distribution of neural tube defect subtypes: a registry-based study. Reprod Toxicol. 2016;59:96–100. https://doi.org/10.1016/j.reprotox.2015.11.007.
Turton P, Badenhorst W, Hughes P, Ward J, Riches S, White S. Psychological impact of stillbirth on fathers in the subsequent pregnancy and puerperium. Br J Psychiatry. 2006;188(2):165–72. https://doi.org/10.1192/bjp.188.2.165.
Campbell-Jackson L, Bezance J, Horsch A. “A renewed sense of purpose”: mothers’ and fathers’ experience of having a child following a recent stillbirth. BMC Pregnancy Childbirth. 2014;14:1–12.
Kothari A, Bruxner G, Callaway L, Dulhunty J. “It’s a lot of pain you’ve got to hide”: a qualitative study of the journey of fathers facing traumatic pregnancy and childbirth. BMC Pregnancy Childbirth. 2022;22(1):1–15.
Helle N, Barkmann C, Ehrhardt S, Bindt C. Postpartum posttraumatic and acute stress in mothers and fathers of infants with very low birth weight: cross-sectional results from a controlled multicenter cohort study. J Affect Disord. 2018;235:467–73.
Spurr S, Danford CA, Roberts KJ, Sheppard-LeMoine D, Machado Silva-Rodrigues F, Darezzo Rodrigues Nunes M, et al. Fathers’ experiences of caring for a child with a chronic illness: a systematic review. Children. 2023;10(2):197.
Cardinali P, Migliorini L, Rania N. The caregiving experiences of fathers and mothers of children with rare diseases in Italy: challenges and social support perceptions. Front Psychol. 2019;10:1780. https://doi.org/10.3389/fpsyg.2019.01780.
Pisula E, Porebowicz-Dorsmann A. Family functioning, parenting stress and quality of life in mothers and fathers of Polish children with high functioning autism or Asperger syndrome. PLoS ONE. 2017;12(10): e0186536. https://doi.org/10.1371/journal.pone.0186536.
Ogwulu CB, Jackson LJ, Heazell AE, Roberts TE. Exploring the intangible economic costs of stillbirth. BMC Pregnancy Childbirth. 2015;15:188. https://doi.org/10.1186/s12884-015-0617-x.
Bramham K, Parnell B, Nelson-Piercy C, Seed PT, Poston L, Chappell LC. Chronic hypertension and pregnancy outcomes: systematic review and meta-analysis. BMJ. 2014;348: g2301. https://doi.org/10.1136/bmj.g2301.
Podymow T, August P. Hypertension in pregnancy. Adv Chronic Kidney Dis. 2007;14(2):178–90. https://doi.org/10.1053/j.ackd.2007.01.008.
Lamsal R, Ungar WJ. Impact of growing up with a sibling with a neurodevelopmental disorder on the quality of life of an unaffected sibling: a scoping review. Disabil Rehabil. 2021;43(4):586–94. https://doi.org/10.1080/09638288.2019.1615563.
Sharpe D, Rossiter L. Siblings of children with a chronic illness: a meta-analysis. J Pediatr Psychol. 2002;27(8):699–710. https://doi.org/10.1093/jpepsy/27.8.699.
Mott DJ, Schirrmacher H, Al-Janabi H, Guest S, Pennington B, Scheuer N, et al. Modelling spillover effects on informal carers: the carer QALY trap. Pharmacoeconomics. 2023;41(12):1557–61. https://doi.org/10.1007/s40273-023-01316-0.
Al-Janabi H, Flynn TN, Coast J. QALYs and carers. Pharmacoeconomics. 2011;29(12):1015–23. https://doi.org/10.2165/11593940-000000000-00000.
Dixon P, Round J. Caring for carers: positive and normative challenges for future research on carer spillover effects in economic evaluation. Value Health. 2019;22(5):549–54. https://doi.org/10.1016/j.jval.2018.10.010.
Dhanji N, Brouwer W, Donaldson C, Wittenberg E, Al-Janabi H. Estimating an exchange-rate between care-related and health-related quality of life outcomes for economic evaluation: an application of the wellbeing valuation method. Health Econ. 2021;30(11):2847–57. https://doi.org/10.1002/hec.4411.
Al-Janabi H, Wittenberg E, Donaldson C, Brouwer W. The relative value of carer and patient quality of life: a person trade-off (PTO) study. Soc Sci Med. 2022;292: 114556. https://doi.org/10.1016/j.socscimed.2021.114556.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
RL was supported by the Hospital for Sick Children through a Restracomp Doctoral Scholarship and by the Institute of Health Policy, Management and Evaluation at the University of Toronto. WJU is supported by the Canada Research Chair in Economic Evaluation and Technology Assessment in Child Health, Canada Excellence Research Chairs, Government of Canada, Sickkids Research Institute.
Conflict of interest
E.A. Yeh has received research funding from NMSS, CMSC, CIHR, NIH, OIRM, SCN, CBMH Chase an Idea, SickKids Foundation, Rare Diseases Foundation, MS Scientific Foundation, McLaughlin Centre, Leong Center, and Peterson Foundation; investigator- initiated research funding from Biogen; scientific advisory funding from Hoffman-LaRoche; and speaker honoraria from Biogen, Saudi Epilepsy Society, NYU, MS-ATL, ACRS, PRIME, and CNPS. WJU has received funding from Broadstreet HE Inc. RL and EP declare that they have no conflict of interest.
Author contributions
All authors contributed to the study’s conception and design. RL designed and executed the search strategy and conducted the analysis and wrote the first draft of the manuscript. All authors critically revised the manuscript and approved the final version for publication.
Ethical approval
Institutional research ethics board approval was not required as no human or animal data were collected or analyzed.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Lamsal, R., Yeh, E.A., Pullenayegum, E. et al. A Systematic Review of Methods and Practice for Integrating Maternal, Fetal, and Child Health Outcomes, and Family Spillover Effects into Cost-Utility Analyses. PharmacoEconomics 42, 843–863 (2024). https://doi.org/10.1007/s40273-024-01397-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40273-024-01397-5