Abstract
Evidence about the association between maternal mental health disorders and stillbirth and infant mortality is limited and conflicting. We aimed to examine whether maternal prenatal mental health disorders are associated with stillbirth and/or infant mortality. MEDLINE, Embase, PsycINFO, and Scopus were searched for studies examining the association of any maternal prenatal (occurring before or during pregnancy) mental health disorder(s) and stillbirth or infant mortality. A random-effects meta-analysis was used to calculate pooled odds ratios (ORs) with 95% confidence intervals (CIs). The between-study heterogeneity was quantified using the I2 statistic. Subgroup analyses were performed to identify the source of heterogeneity. Of 4487 records identified, 28 met our inclusion criteria with 27 contributing to the meta-analyses. Over 60% of studies examined stillbirth and 54% of them evaluated neonatal or infant mortality. Thirteen studies investigated the association between maternal depression and anxiety and stillbirth/infant mortality, pooled OR, 1.42 (95% CI, 1.16–1.73; I2, 76.7%). Another 13 studies evaluated the association between severe maternal mental illness and stillbirth/infant mortality, pooled OR, 1.47 (95% CI, 1.28–1.68; I2, 62.3%). We found similar results for the association of any maternal mental health disorders and stillbirth/infant mortality (OR, 1.59; 95% CI, 1.43–1.77) and in subgroup analyses according to types of fetal/infant mortality. We found no significant evidence of publication bias. Maternal prenatal mental health disorders appear to be associated with a moderate increase in the risk of stillbirth and infant mortality, although the mechanisms are unclear. Efforts to prevent and treat these disorders may reduce the scale of stillbirth/infant deaths.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Maternal mental health disorders are common problems worldwide (Gold and Marcus 2008) and include anxiety and depression, psychotic disorders, and a range of other mental disorders. These disorders constitute a prominent public health issue given their scale; for instance, depression alone affects up to 25% of pregnancies (Gelaye et al. 2016) and their association with myriad adverse outcomes for mother and offspring (Pierce et al. 2019; Zhong et al. 2018). A large body of evidence highlights that women with a range of different mental disorders are at distinctly higher risk of delivering a preterm or low birthweight baby (Ding et al. 2014; Grigoriadis et al. 2018; Grote et al. 2010) and of having children with poorer developmental outcomes (Reupert et al. 2013). In addition, the results of a limited set of studies suggest that poor maternal mental health poses an increased risk for early life mortality (Webb et al. 2005; Zhong et al. 2018).
The mechanisms whereby maternal mental health impact on adverse fetal and infant outcomes are complex, not fully understood, and likely include alterations to the intrauterine environment as well as behavioural pathways. Depression and anxiety, for example, may alter the cortisol response to stress, which can affect fetal growth (Field et al. 2006; Zorn et al. 2017). For some women, mental health disorders are also associated with inadequate antenatal care, smoking, and misuse of illicit substances and drugs—all common risk factors of perinatal mortality (Judd et al. 2014; Weaver et al. 2003). However, only a limited number of studies have evaluated the association of maternal mental health disorders and stillbirth and infant mortality outcomes, with inconsistent findings (Di Prinzio et al. 2020; Heun-Johnson et al. 2018; Pavlov et al. 2014; Surkan et al. 2016; Vigod et al. 2014; Zhong et al. 2018).
To our knowledge, only two systematic reviews (Jacques et al. 2019; Webb et al. 2005) have focused on the risks for mortality, and none has comprehensively investigated the link between maternal pre-conception and prenatal (occurring before or during pregnancy) mental health disorders and perinatal and infant mortality. A 2005 review focused exclusively on the association between schizophrenia and stillbirth but did not include any other mental health disorders, and identified only six studies, all with important methodological limitations (Webb et al. 2005). A 2019 review examining maternal depression and infant hospitalization and mortality identified only three in-scope studies that examined infant mortality outcomes (Jacques et al. 2019). Notably, most of the included studies in these reviews (three in the first and two in the second) included maternal mental health disorders that occurred in the postnatal period or after the fetal/infant outcome, exposing the possibility of reverse causality bias.
The current systematic review and meta-analysis aimed to provide a comprehensive evaluation of published research that has examined the association between maternal prenatal mental health disorders and stillbirth and infant mortality. The resulting evidence can support prevention and early interventions that target the high proportion of women with mental health disorders.
Material and methods
Protocol and registration
We followed the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) (Moher et al. 2009) and the Meta-analysis of Observational Studies in Epidemiology (MOOSE) (Stroup et al. 2000) guidelines. The review protocol has been published (Adane et al. 2020) and registered with the PROSPERO (# CRD42020159834).
Search strategy
The details of the search terms and strategies are available in the published protocol (Adane et al. 2020) and as a supplementary file (Supplementary Box 1) but, in summary, we searched MEDLINE, Embase, PsycINFO, and Scopus electronic databases using keywords tailored to each database, from their inception to December 2019. These searches were limited to studies published in English language and on humans only, with no restriction on the year of publication. The reference lists and citations of relevant articles were also checked manually for additional studies. Three authors were contacted for additional information, but only one provided further details (Di Prinzio et al. 2020).
Inclusion and exclusion criteria
Original studies examining the association of any prenatal mental health disorder (occurring at any time before or during pregnancy) and stillbirth (death of a fetus at 20 or more weeks of gestation), neonatal (the first 28 days of life), post-neonatal (the period after 28 days of life and before 1 year), or infant (under 1 year) mortality were eligible. The use of either a diagnostic measure of mental health or symptom screening measure formed part of the eligibility criteria. However, correspondence, theses, reviews, editorials, case-only studies, and conference abstracts were excluded. We also excluded studies where the onset of maternal mental health disorders in relation to the date of childbirth was unclear and/or when postnatal mental health disorders were combined with prenatal disorders.
Study selection
First, all citations identified through database searches were imported into an Endnote library, and duplicates were removed. The titles of the remaining records were screened, with eligible studies uploaded to Rayyan (Ouzzani et al. 2016) and abstracts and full-texts independently evaluated by two of the authors (AAA and HDB). Disagreements between assessors were resolved by face-to-face discussion.
Data extraction
Using a standardized excel sheet, one of the researchers (AAA) extracted the data from articles that passed the full-text evaluation, and the second author (HDB) verified the results. Extracted data included the first author’s last name, country and year of publication, study population and design, exposure and outcome assessment, and association/s as well as confounders adjusted for. When available, adjusted odds ratios (ORs) with 95% confidence intervals (CIs) were extracted. For studies which did not report ORs, crude measures were manually calculated when enough data were available. Some studies provided relative risk (RR) estimates, which were converted to ORs using a previously developed package (Barendregt 2016).
Assessment of methodological quality and risk of bias
We used the Newcastle-Ottawa quality assessment scale (Wells 2001), which includes the following: the selection of the study groups, the comparability of the groups, and the ascertainment of either the exposure (for case-control studies) or outcome of interest (for cohort studies). We also employed the GRADE (Grading of Recommendations Assessment, Development and Evaluation) approach (Guyatt et al. 2008) to assess the degree of risk of bias for each included study (Higgins et al. 2011). Two of the authors (AAA and HDB) independently performed the quality and risk of bias assessments. When the involved authors differ in the results of the quality assessments, an agreement was reached during a face-to-face discussion.
Data analysis
We have included studies with several differences between them, such as in the type of maternal mental disorders investigated, diagnostic classification system, mortality outcome measure, geographical location, and time period. As such, we do not expect them to share a common effect (fixed effect). Therefore, we employed a random-effects model (Harris et al. 2008), which accounts for between-study heterogeneity and provides an estimate of the average effect, to calculate pooled ORs with 95% CIs for the overall and subgroup meta-analyses of the associations between maternal mental health disorders and stillbirth/infant mortality outcomes. The maternal mental health disorders were grouped into depression and anxiety (including major depressive disorder) and severe mental illness (including schizophrenia, bipolar, schizoaffective/affective psychosis, and psychotic disorders not otherwise specified). We undertook further subgroup analyses for each outcome (stillbirth, perinatal, and infant mortality) separately and collectively as a ‘stillbirth/infant mortality’ variable within each maternal mental health disorder group. Eight studies only reported perinatal mortality (includes stillbirth and neonatal death) as an aggregate outcome, so the perinatal mortality subgroup includes these studies and those studies that separately reported stillbirth and neonatal mortality. Additional sensitivity and subgroup analyses were conducted by excluding each study and also based on whether an adjustment was made for smoking (yes/no), the number of offspring mortality in women with mental health disorders (< 20 vs. ≥ 20 cases), year of publication (≤ 2010 vs. > 2010), and risk of bias based on the GRADE approach (low vs. moderate). We also ran a sensitivity analysis by excluding studies using symptom only screening measures for depression and anxiety. A few of the included studies contributed two or more estimates for stillbirth, neonatal, post-neonatal, and infant death as they assessed different mental disorders. So to check the effect of non-independent effect sizes, we repeated the primary analyses by including an average measure (obtained via a fixed effect model) for studies with two or more estimates. Analyses were conducted using STATA 15 (StataCorp, College Station).
Assessment of heterogeneity and publication bias
Between-study heterogeneity was examined using the I2 statistic (Higgins et al. 2003) and was considered low (I2 < 50%), moderate (I2, 50–74.9%), or high (I2 ≥ 75%). The risk of publication bias was graphically examined using funnel plot asymmetry and further quantified using the Egger’s test (p value ≤ 0.05 suggests significant publication bias) (Harbord et al. 2009).
Results
Search results
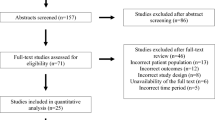
A total of 4487 records were identified through the database and manual searches. After duplicates were removed, 3756 were screened by title and abstract. Full-text assessments were made on 78 articles with 50 of these excluded for reasons such as no assessment (n = 22) or separate reporting (n = 6) of early mortality, and no assessment of prenatal mental health (n = 6) or inclusion of postnatal mental health cases (n = 7). Finally, 28 studies (Ban et al. 2012; Bansil et al. 2010; Bitew et al. 2017; De Lange et al. 2008; Di Prinzio et al. 2020; Gardosi et al. 2013; Hanlon et al. 2009; Heun-Johnson et al. 2018; Hizkiyahu et al. 2010; Howard et al. 2003; Howard et al. 2007; Huang et al. 2017; MacCabe et al. 2007; Mei-Dan et al. 2015; Navaratne et al. 2016; Nilsson et al. 2002; Pare-Miron et al. 2016; Pavlov et al. 2014; Raisanen et al. 2014; Simoila et al. 2018; Vigod et al. 2014; Webb et al. 2006; Webb et al. 2010; Weobong et al. 2014; Wrede et al. 1980; Yedid Sion et al. 2016; Zax et al. 1977; Zhong et al. 2018) were included in this systematic review (n = 78,492,940), with all but one (Hanlon et al. 2009) contributing at least one estimate for the meta-analyses (Fig. 1).
Characteristics of included studies
The included studies are described in Table 1 and summarized in Box 1. The majority of studies were from European countries (n = 12) and the USA (n = 5). The most common maternal mental health disorders examined were depression (n = 8) and schizophrenia and related disorders (n = 5). The most common outcome was stillbirth (n = 17). The vast majority (n = 24) of studies were cohort studies, while two each were case-control and analytical cross-sectional studies. The sample size (births) of included studies ranged from 56 to 32,156,438 (Table 1).
Included studies varied considerably in terms of confounder or covariate adjustments. While most studies adjusted for maternal age, very few studies fully adjusted for other maternal sociodemographic and lifestyle factors (Supplementary Table 1).
Methodological quality and risk of bias
Based on the Newcastle-Ottawa quality assessment scale, 17 studies scored eight or nine, and the remaining 11 studies received scores of six or seven out of nine (Supplementary Tables 2 and 3). Using the GRADE approach, the risk of bias in most studies was low (n = 15) or moderate (n = 12) with only one study having a high risk of bias (Supplementary Table 4).
Results of the meta-analyses
Depression and anxiety
Using 16 estimates (i.e., ORs) from 13 unique studies (a study can assess more than one child outcome; so it can contribute more than one estimate) including 39,526,270 births, the pooled OR for the association between maternal depression and anxiety and stillbirth/infant mortality was 1.42 (95% CI, 1.16–1.73; I2, 76.7%). There were similar associations for stillbirth and perinatal mortality, but the association for infant mortality was marginally lower and spanned the null value (Fig. 2). The pooled ORs (95% CI) were 1.46 (1.17–1.82: I2, 75.1%) for the association between maternal depression and stillbirth/infant mortality, 1.51 (1.17–1.96: I2, 81.3%) for the association between maternal depression and stillbirth, 1.46 (1.16–1.84: I2, 76.4%) for the association between maternal depression and perinatal mortality, and 1.36 (0.90–2.05: I2, 54.6%) for the association between maternal depression and infant mortality.
Severe mental illness
Using 27 estimates from 13 unique studies which included 36,038,106 births, the pooled OR for the association between severe maternal mental illness and stillbirth or infant mortality was 1.47 (95% CI, 1.28–1.68; I2, 62.3%). We found similar associations with stillbirth (OR, 1.42; 95% CI, 1.20–1.69; 12 estimates from 10 unique studies and 33,365,387 births; I2, 68.5%), perinatal mortality (OR, 1.51; 95% CI, 1.28–1.79; 19 estimates from 12 unique studies and 33,557,786 births; I2, 72.6%), and infant mortality (OR, 1.49; 95% CI, 1.20–1.85; 13 estimates from seven unique studies and 6,812,821 births; I2, 46.2%) (Fig. 2).
Any maternal prenatal mental health disorders
Overall, using 52 estimates from 27 unique studies including 78,316,822 births, the pooled OR for the association between any maternal prenatal mental health disorders (listed in Table 1) and stillbirth/infant mortality was 1.59 (95% CI, 1.43–1.77; I2, 75.5%). We found similar associations for stillbirth and perinatal mortality and a stronger association for infant mortality, particularly for post-neonatal mortality and sudden infant death syndrome (Fig. 2 and Fig. 3).
Hanlon et al. (Hanlon et al. 2009), which is not included in the meta-analyses, assessed common mental health disorders using the Self-Reporting Questionnaire (SRQ-20). The study did not find a statistically significant association between maternal antenatal SRQ score (≥ 6) and stillbirth (RR, 1.7; 95% CI, 0.6–5.5) or neonatal mortality (RR, 0.8; 95% CI, 0.2–3.0), perhaps because of low statistical power.
Heterogeneity and publication bias
The between-study heterogeneity was high for the associations of maternal depression and anxiety and stillbirth/infant mortality, and any maternal mental health disorders and stillbirth/infant mortality. A high degree of between-study heterogeneity remained in subgroup analyses according to the types of mortality outcomes, particularly for stillbirth and perinatal mortality. These could be partly due to differences in the types of maternal mental health disorders, confounders adjusted for, sample size, and year of publication, as shown in the stratified and sensitivity analyses (Supplementary Table 5). We found no evidence of publication bias for studies evaluating the associations of maternal depression and anxiety and stillbirth/infant mortality; severe maternal mental illness and stillbirth/infant mortality, and studies assessing any maternal prenatal mental health disorders and stillbirth (Supplementary Fig. 1).
Sensitivity and subgroup analysis
We did not find any significant changes for the associations between maternal depression and anxiety and severe mental illness with stillbirth/infant mortality outcomes when conducting sensitivity analyses that removed each study one by one (Supplementary Fig. 2). However, we found considerable differences in pooled ORs in the stratified analyses based on whether the adjustment was made for smoking, year of publication, and degree of risk of bias using the GRADE approach. While the pooled effect estimates for maternal depression and anxiety were stronger for studies adjusted for smoking and those published after 2010, those estimates for severe maternal mental illness were stronger if studies did not adjust for smoking, published in 2010 or earlier, and had a moderate risk of bias (Supplementary Table 5). Overall, we found slightly stronger associations and lower between-study heterogeneity in the sensitivity analyses excluding studies that used symptom only screening measures for depression and anxiety: stillbirth/infant mortality (OR, 1.47; 95% CI, 1.18–1.82; I2, 74.0%), stillbirth (OR, 1.67; 95% CI, 1.35–2.08; I2, 65.1%), perinatal mortality (OR, 1.48; 95% CI, 1.19–1.85; I2, 75.5%), and infant mortality (OR, 1.22; 95% CI, 0.59–2.53; I2, 55.3%). We found similar results to the primary analyses in the sensitivity analyses using the average measure for studies with two or more estimates (Supplementary Fig. 3 and Fig. 4). However, the between-study heterogeneity slightly increased (Supplementary Fig. 3), and the pooled estimate for neonatal death slightly attenuated and spanned the null value (Supplementary Fig. 4).
Discussion
Summary of main findings
In this systematic review and meta-analysis, which included 28 studies comprising over 78 million births, we found a moderate and statistically significant higher risk of stillbirth and infant mortality for women with prenatal mental health disorders (including high and low prevalence disorders), compared with women without prenatal mental health disorders. These findings persisted in sensitivity and subgroup analyses according to the types of maternal mental health disorders and fetal or infant mortality outcomes, with results typically indicating a 40–50% increase in the risk of stillbirth and infant mortality. However, considerable heterogeneity was observed in effect sizes across studies included in the meta-analysis, and very few studies evaluated the impact of common maternal mental health disorders, particularly anxiety disorders.
Research in context and potential biological mechanisms
The previous meta-analyses found that women with depression and anxiety disorders were 40 to 80% more likely to have preterm birth and low birthweight (Ding et al. 2014; Grigoriadis et al. 2018; Grote et al. 2010), the leading causes of perinatal mortality. The findings of this systematic review and meta-analysis extend the evidence base by including a broader range of mental health disorders and further corroborated the detrimental impact of maternal prenatal mental health disorders. The underlying reasons for the higher risk of adverse perinatal outcomes among women with prenatal mental health disorders are not yet fully understood, but they are likely to include a complex array of mechanisms that include direct disease effects, behavioural factors and comorbidities, and increased risk of pregnancy complications.
There are several hypothesised pathways through which fetal health may be compromised by intrauterine exposure to psychological distress, including pathways that impact fetal neurodevelopment, placental function, and physical growth (Kinsella and Monk 2009; Lewis et al. 2016). Recent studies, for example, have linked maternal prenatal anxiety and depression with disruptions in the placental 11β-hydroxysteroid dehydrogenase type 2 (11B-HSD2) enzyme and cortisol transfer to the fetus (Cottrell et al. 2014; Duthie and Reynolds 2013; O’Donnell et al. 2012), inflammatory pathways, and altered physiological responses (Christian 2014; Seth et al. 2016). While these effects have generally been associated with greater morbidity, a link with mortality risks is less clear (Ding et al. 2014; Field and Diego 2008). Unhealthy lifestyles and behaviours are highly correlated with psychiatric disorders, and their interplay plausibly accounts for part of the association between maternal mental health disorders and perinatal morbidities and mortality. Women with mental health disorders tend to have poor health-seeking behaviour (late and less frequent antenatal care) (Ben-Sheetrit et al. 2018; Lin et al. 2009) and are more likely to misuse illicit substances, alcohol, and drugs, smoke tobacco, and have poor nutrition (Barker et al. 2013; Frayne et al. 2019; Judd et al. 2014; Teasdale et al. 2019; Weaver et al. 2003), all of which can have profound consequences for fetal development. Most prenatal mental disorders are likely to persist in the postnatal period and directly compromise the quality of essential parenting, which may contribute to the increased infant mortality risk (Chen et al. 2010). Recent evidence also indicates that some mental disorders and/or their medications increase the risk of pregnancy complications such as gestational diabetes and pre-eclampsia (Galbally et al. 2020; Qiu et al. 2009; Uguz 2017), which are key risk factors for perinatal morbidity and mortality (Hutcheon et al. 2011; Schmidt et al. 2001). However, the evidence about the association of mental disorders or antipsychotic treatments and pregnancy complications is inconsistent and likely to include complex pathways (Vigod et al. 2015; Vigod et al. 2014). Collectively, these underscore the need for comprehensive support, care, and careful treatment for women with a mental health disorder.
Strengths and limitations
The strengths of this systematic review and meta-analysis are that it is based on a pre-published protocol and employed independent reviews and quality assessments on records obtained via electronic searches in multiple databases (supplemented with a manual search). To the best of our knowledge, this is the first systematic review and meta-analysis that has comprehensively evaluated the association between maternal prenatal mental health disorders and stillbirth or infant mortality—providing the most robust evidence to date on this topic.
The review does, however, have some limitations. Although a restriction was not imposed on the year of publication, electronic search results were limited to English language and hence relevant non-English studies could have been excluded. Relatively fewer studies were available with different maternal mental health disorders and assessments. This precluded us from a comprehensive investigation of the source of heterogeneity and publication bias. Most studies included in the systematic review did not adequately control for potential socioeconomic and lifestyle factors and, among those that did, there was variation in the choice of modelled covariates (Supplementary Table 1). In addition, there is always a possibility of residual confounding. However, a statistically significant association remained (albeit attenuated for severe maternal mental illness) in the meta-analysis, which included studies that adjusted for smoking. Additionally, one of the largest studies (Zhong et al. 2018) included in this systematic review demonstrated a significant association between maternal psychosis and stillbirth (OR, 1.37; 95% CI, 1.23–1.53) after adjustment for several factors including sociodemographic (age, race, and income) and lifestyle (smoking and alcohol/substance abuse) factors. The other limitation is that the studies included in the review did not examine or provide separate data for mentally ill mothers with and without psychotropic medications, so we could not evaluate the effect of the use of psychotropic drugs.
Implications
The findings of this review have important implications for public health, particularly given the high prevalence of maternal mental health problems (especially anxiety and depression) in populations worldwide. Efforts to prevent chronic and acute prenatal mental health disorders and treat those suffering from mental health disorders are likely to be critical in reducing the scale of stillbirth and infant mortality and the consequent grief experienced by families and communities. Focusing interventions on the earliest stages of family planning and prenatal period is important as many mental health problems are chronic at the time of conception (O'Hara and Wisner 2014). In terms of policy, holistic healthcare models, with a multidisciplinary approach, are preferable (Frayne et al. 2019; Galbally et al. 2013; Tachibana et al. 2019). Evidence-based treatments for women with severe mental illness, particularly depression, may reduce the rate of adverse birth outcomes, including perinatal and infant mortality, but this requires further investigation as the evidence in this area is conflicting (Ban et al. 2012; Jarde et al. 2016; Ross and Grigoriadis 2014). The fact that most of the studies included in this review were not designed to assess the associations of maternal prenatal mental health disorders and perinatal or infant mortality highlights the need for high-quality prospective cohort studies to better understand the issue.
Conclusions
The findings of this comprehensive systematic review and meta-analysis have demonstrated that women with prenatal mental health disorders are at a higher risk of perinatal and infant mortality, compared with women without prenatal mental health disorders, with consistent effects across a range of mental health disorders. Prenatal screening and access to treatment for mental health disorders with better and co-ordinated antenatal, obstetric, and psychiatric care may reduce the risk of perinatal and infant mortality. Further well-designed studies are required to better understand the nature of the problem, underlying mechanisms, and to confirm whether medical treatments for women with prenatal mental illness reduce perinatal and infant mortality.
Data availability
Not applicable.
References
Adane AA, Bailey HD, Marriott R, Farrant BM, White SW, Morgan VA, Shepherd CCJ (2020) Role of maternal mental health disorders on stillbirth and infant mortality risk: a protocol for a systematic review and meta-analysis. BMJ Open 10:e036280. https://doi.org/10.1136/bmjopen-2019-036280
Ban L, Tata LJ, West J, Fiaschi L, Gibson JE (2012) Live and non-live pregnancy outcomes among women with depression and anxiety: a population-based study. PLoS One 7:e43462. https://doi.org/10.1371/journal.pone.0043462
Bansil P, Kuklina EV, Meikle SF, Posner SF, Kourtis AP, Ellington SR, Jamieson DJ (2010) Maternal and fetal outcomes among women with depression. J Women's Health 1:329–334
Barendregt J (2016) EpiGear International. EpiGear http://www.epigear.com/index_files/or2rr.html. Accessed 6 Apr 2020
Barker ED, Kirkham N, Ng J, Jensen SK (2013) Prenatal maternal depression symptoms and nutrition, and child cognitive function. Br J Psychiatry 203:417–421. https://doi.org/10.1192/bjp.bp.113.129486
Ben-Sheetrit J, Huller-Harari L, Rasner M, Magen N, Nacasch N, Toren P (2018) Psychiatric disorders and compliance with prenatal care: A 10-year retrospective cohort compared to controls. Eur Psychiatry 49:23–29. https://doi.org/10.1016/j.eurpsy.2017.11.011
Bitew T, Hanlon C, Kebede E, Honikman S, Fekadu A (2017) Antenatal depressive symptoms and perinatal complications: a prospective study in rural Ethiopia. BMC Psychiatry 1:301
Chen YH, Chiou HY, Tang CH, Lin HC (2010) Risk of death by unnatural causes during early childhood in offspring of parents with mental illness. Am J Psychiatry 167:198–205. https://doi.org/10.1176/appi.ajp.2009.09070979
Christian LM (2014) Effects of stress and depression on inflammatory immune parameters in pregnancy. Am J Obstet Gynecol 211:275–277. https://doi.org/10.1016/j.ajog.2014.06.042
Cottrell EC, Seckl JR, Holmes MC, Wyrwoll CS (2014) Foetal and placental 11β-HSD2: a hub for developmental programming. Acta Physiol (Oxford) 210:288–295. https://doi.org/10.1111/apha.12187
De Lange TE, Budde MP, Heard AR, Tucker G, Kennare R, Dekker GA (2008) Avoidable risk factors in perinatal deaths: a perinatal audit in South Australia. Aust N Z J Obstet Gynaecol 48:50–57. https://doi.org/10.1111/j.1479-828X.2007.00801.x
Di Prinzio P et al (2020) Parsing components of risk of premature mortality in the children of mothers with severe mental illness. Schizophr Res 218:180–187. https://doi.org/10.1016/j.schres.2020.01.006
Ding XX et al (2014) Maternal anxiety during pregnancy and adverse birth outcomes: a systematic review and meta-analysis of prospective cohort studies. J Affect Disord 159:103–110. https://doi.org/10.1016/j.jad.2014.02.027
Duthie L, Reynolds RM (2013) Changes in the maternal hypothalamic-pituitary-adrenal axis in pregnancy and postpartum: influences on maternal and fetal outcomes. Neuroendocrinology 98:106–115. https://doi.org/10.1159/000354702
Field T, Diego M (2008) Cortisol: the culprit prenatal stress variable. Int J Neurosci 118:1181–1205. https://doi.org/10.1080/00207450701820944
Field T, Hernandez-Reif M, Diego M, Figueiredo B, Schanberg S, Kuhn C (2006) Prenatal cortisol, prematurity and low birthweight. Infant Behav Dev 29:268–275. https://doi.org/10.1016/j.infbeh.2005.12.010
Frayne J, Nguyen T, Allen S, Hauck Y, Liira H, Vickery A (2019) Obstetric outcomes for women with severe mental illness: 10 years of experience in a tertiary multidisciplinary antenatal clinic. Arch Gynecol Obstet 300:889–896. https://doi.org/10.1007/s00404-019-05258-x
Galbally M, Blankley G, Power J, Snellen M (2013) Perinatal mental health services: what are they and why do we need them? Austr Psychiatry 21:165–170. https://doi.org/10.1177/1039856213476924
Galbally M, Frayne J, Watson SJ, Morgan V, Snellen M (2020) The association between gestational diabetes mellitus, antipsychotics and severe mental illness in pregnancy: a multicentre study. Aust N Z J Obstet Gynaecol 60:63–69. https://doi.org/10.1111/ajo.12986
Gardosi J, Madurasinghe V, Williams M, Malik A, Francis A (2013) Maternal and fetal risk factors for stillbirth: population based study. BMJ 1:f108
Gelaye B, Rondon MB, Araya R, Williams MA (2016) Epidemiology of maternal depression, risk factors, and child outcomes in low-income and middle-income countries. Lancet Psychiatry 3:973–982. https://doi.org/10.1016/S2215-0366(16)30284-X
Gold KJ, Marcus SM (2008) Effect of maternal mental illness on pregnancy outcomes. Expert Rev Obstet Gynecol 3:391–401. https://doi.org/10.1586/17474108.3.3.391
Grigoriadis S, Graves L, Peer M, Mamisashvili L, Tomlinson G, Vigod SN, Dennis CL, Steiner M, Brown C, Cheung A, Dawson H, Rector NA, Guenette M, Richter M (2018) Maternal anxiety during pregnancy and the association with adverse perinatal outcomes: systematic review and meta-analysis. J Clin Psychiatry 79. https://doi.org/10.4088/JCP.17r12011
Grote NK, Bridge JA, Gavin AR, Melville JL, Iyengar S, Katon WJ (2010) A meta-analysis of depression during pregnancy and the risk of preterm birth, low birth weight, and intrauterine growth restriction. JAMA Psychiatry 67:1012–1024. https://doi.org/10.1001/archgenpsychiatry.2010.111
Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, Schünemann HJ (2008) GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 336:924–926
Hanlon C et al (2009) Impact of antenatal common mental disorders upon perinatal outcomes in Ethiopia: the P-MaMiE population-based cohort study. Tropical Med Int Health 1:156–166
Harbord RM, Harris RJ, Sterne JA (2009) Updated tests for small-study effects in meta-analyses. Stata J 9:197–210
Harris R, Bradburn M, Deeks J, Harbord R, Altman D, Sterne J (2008) Metan: fixed- and random-effects meta-analysis. Stata J 8:3–28
Heun-Johnson H, Seabury SA, Menchine M, Claudius I, Axeen S, Lakshmanan A (2018) Association between maternal serious mental illness and adverse birth outcomes. J Investig Med 66(1):193–194. https://doi.org/10.1136/jim-2017-000663.305
Higgins JPT, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JAC, Cochrane Bias Methods Group, Cochrane Statistical Methods Group (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928. https://doi.org/10.1136/bmj.d5928
Higgins JPT, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557–560. https://doi.org/10.1136/bmj.327.7414.557
Hizkiyahu R, Levy A, Sheiner E (2010) Pregnancy outcome of patients with schizophrenia. Am J Perinatol 1:19–23
Howard LM, Goss C, Leese M, Thornicroft G (2003) Medical outcome of pregnancy in women with psychotic disorders and their infants in the first year after birth. Br J Psychiatry 182:63–67. https://doi.org/10.1192/bjp.182.1.63
Howard LM, Kirkwood G, Latinovic R (2007) Sudden infant death syndrome and maternal depression. J Clin Psychiatry 1:1279–1283
Huang HC, Sung FC, Chen PC, Chang CYY, Muo CH, Shiue HS, Huang JP, Li TC, Tzeng YL, Wu SI (2017) Obstetric outcomes in pregnant women with and without depression: population-based comparison. Sci Rep 7:13937. https://doi.org/10.1038/s41598-017-14266-3
Hutcheon JA, Lisonkova S, Joseph KS (2011) Epidemiology of pre-eclampsia and the other hypertensive disorders of pregnancy. Best Pract Res Clin Obstet Gynaecol 25:391–403. https://doi.org/10.1016/j.bpobgyn.2011.01.006
Jacques N, de Mola CL, Joseph G, Mesenburg MA, da Silveira MF (2019) Prenatal and postnatal maternal depression and infant hospitalization and mortality in the first year of life: a systematic review and meta-analysis. J Affect Disord 243:201–208. https://doi.org/10.1016/j.jad.2018.09.055
Jarde A et al (2016) Neonatal outcomes in women with untreated antenatal depression compared with women without depression: a systematic review and meta-analysis. JAMA Psychiatry 73:826–837. https://doi.org/10.1001/jamapsychiatry.2016.0934
Judd F, Komiti A, Sheehan P, Newman L, Castle D, Everall I (2014) Adverse obstetric and neonatal outcomes in women with severe mental illness: to what extent can they be prevented? Schizophr Res 157:305–309. https://doi.org/10.1016/j.schres.2014.05.030
Kinsella MT, Monk C (2009) Impact of maternal stress, depression and anxiety on fetal neurobehavioral development. Clin Obstet Gynecol 52:425–440. https://doi.org/10.1097/GRF.0b013e3181b52df1
Lewis AJ, Austin E, Galbally M (2016) Prenatal maternal mental health and fetal growth restriction: a systematic review. J Dev Orig Health Dis 7:416–428. https://doi.org/10.1017/S2040174416000076
Lin HC, Chen YH, Lee HC (2009) Prenatal care and adverse pregnancy outcomes among women with schizophrenia: a nationwide population-based study in Taiwan. J Clin Psychiatry 70:1297–1303. https://doi.org/10.4088/JCP.09m05087
MacCabe JH, Martinsson L, Lichtenstein P, Nilsson E, Cnattingius S, Murray RM, Hultman CM (2007) Adverse pregnancy outcomes in mothers with affective psychosis. Bipolar Disord 1:305–309
Mei-Dan E, Ray JG, Vigod SN (2015) Perinatal outcomes among women with bipolar disorder: a population-based cohort study. Am J Obstet Gynecol 212:367.e361–367.e368. https://doi.org/10.1016/j.ajog.2014.10.020
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097. https://doi.org/10.1371/journal.pmed.1000097
Navaratne P, Foo XY, Kumar S (2016) Impact of a high Edinburgh Postnatal Depression Scale score on obstetric and perinatal outcomes. Sci Rep 6:33544. https://doi.org/10.1038/srep33544
Nilsson E, Lichtenstein P, Cnattingius S, Murray RM, Hultman CM (2002) Women with schizophrenia: pregnancy outcome and infant death among their offspring. Schizophr Res 1:221–229
O'Hara MW, Wisner KL (2014) Perinatal mental illness: definition, description and aetiology. Best Pract Res Clin Obstet Gynaecol 28:3–12. https://doi.org/10.1016/j.bpobgyn.2013.09.002
O’Donnell KJ, Bugge Jensen A, Freeman L, Khalife N, O’Connor TG, Glover V (2012) Maternal prenatal anxiety and downregulation of placental 11β-HSD2. Psychoneuroendocrinology 37:818–826. https://doi.org/10.1016/j.psyneuen.2011.09.014
Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A (2016) Rayyan—a web and mobile app for systematic reviews. Syst Rev 5(210):210
Pare-Miron V, Czuzoj-Shulman N, Oddy L, Spence AR, Abenhaim HA (2016) Effect of Borderline personality disorder on obstetrical and neonatal outcomes. Womens Health Issues 26:190–195. https://doi.org/10.1016/j.whi.2015.11.001
Pavlov M, Steiner N, Kessous R, Weintraub AY, Sheiner E (2014) Obstetric and neonatal outcome in patients with anxiety disorders. J Matern Fetal Neonatal Med 1:1339–1342
Pierce M et al (2019) Effects of parental mental illness on children’s physical health: systematic review and meta-analysis. Br J Psychiatry:1–10. https://doi.org/10.1192/bjp.2019.216
Qiu C, Williams MA, Calderon-Margalit R, Cripe SM, Sorensen TK (2009) Preeclampsia risk in relation to maternal mood and anxiety disorders diagnosed before or during early pregnancy. Am J Hypertens 22:397–402. https://doi.org/10.1038/ajh.2008.366
Raisanen S, Lehto SM, Nielsen HS, Gissler M, Kramer MR, Heinonen S (2014) Risk factors for and perinatal outcomes of major depression during pregnancy: a population-based analysis during 2002-2010 in Finland. BMJ Open 4(11) (no pagination). https://doi.org/10.1136/bmjopen-2014-004883
Reupert AE, Maybery JD, Kowalenko NM (2013) Children whose parents have a mental illness: prevalence, need and treatment. Med J Aust 199:S7–S9. https://doi.org/10.5694/mja11.11200
Ross LE, Grigoriadis S (2014) Selected pregnancy and delivery outcomes after exposure to antidepressant medication. JAMA Psychiatry 71:716–717. https://doi.org/10.1001/jamapsychiatry.2014.59
Schmidt MI et al (2001) Gestational diabetes mellitus diagnosed with a 2-h 75-g oral glucose tolerance test and adverse pregnancy outcomes. Diabetes Care 24:1151. https://doi.org/10.2337/diacare.24.7.1151
Seth S, Lewis AJ, Galbally M (2016) Perinatal maternal depression and cortisol function in pregnancy and the postpartum period: a systematic literature review. BMC Pregnancy Childbirth 16:124. https://doi.org/10.1186/s12884-016-0915-y
Simoila L, Isometsä E, Gissler M, Suvisaari J, Halmesmäki E, Lindberg N (2018) Obstetric and perinatal health outcomes related to schizophrenia: a national register-based follow-up study among Finnish women born between 1965 and 1980 and their offspring. Eur Psychiatry 52:68–75. https://doi.org/10.1016/j.eurpsy.2018.04.001
Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, Moher D, Becker BJ, Sipe TA, Thacker SB (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. JAMA 283:2008–2012
Surkan PJ, Patel SA, Rahman A (2016) Preventing infant and child morbidity and mortality due to maternal depression. Best Pract Res Clin Obst Gynaecol 36:156–168
Tachibana Y, Koizumi N, Akanuma C, Tarui H, Ishii E, Hoshina T, Suzuki A, Asano A, Sekino S, Ito H (2019) Integrated mental health care in a multidisciplinary maternal and child health service in the community: the findings from the Suzaka trial. BMC Pregnancy Childbirth 19:58. https://doi.org/10.1186/s12884-019-2179-9
Teasdale SB, Ward PB, Samaras K, Firth J, Stubbs B, Tripodi E, Burrows TL (2019) Dietary intake of people with severe mental illness: systematic review and meta-analysis. Br J Psychiatry 214:251–259. https://doi.org/10.1192/bjp.2019.20
Uguz F (2017) Is there any association between use of antidepressants and preeclampsia or gestational hypertension?: a systematic review of current studies. J Clin Psychopharmacol 37:72–77. https://doi.org/10.1097/jcp.0000000000000618
Vigod SN, Gomes T, Wilton AS, Taylor VH, Ray JG (2015) Antipsychotic drug use in pregnancy: high dimensional, propensity matched, population based cohort study. BMJ 350:h2298. https://doi.org/10.1136/bmj.h2298
Vigod SN et al (2014) Maternal and newborn outcomes among women with schizophrenia: a retrospective population-based cohort study. BJOG 1:566–574
Weaver T, Madden P, Charles V, Stimson G, Renton A, Tyrer P, Barnes T, Bench C, Middleton H, Wright N, Paterson S, Shanahan W, Seivewright N, Ford C, Comorbidity of Substance Misuse and Mental Illness Collaborative (Cosmic) Study Team (2003) Comorbidity of substance misuse and mental illness in community mental health and substance misuse services. Br J Psychiatry 183:304–313. https://doi.org/10.1192/bjp.183.4.304
Webb R, Abel K, Pickles A, Appleby L (2005) Mortality in offspring of parents with psychotic disorders: a critical review and meta-analysis. Am J Psychiatry 162:1045–1056
Webb RT, Abel KM, Pickles AR, Appleby L, King-Hele SA, Mortensen PB (2006) Mortality risk among offspring of psychiatric inpatients: a population-based follow-up to early adulthood. Am J Psychiatry 1:2170–2177
Webb RT et al (2010) Influence of environmental factors in higher risk of sudden infant death syndrome linked with parental mental illness. Arch Gen Psychiatry 1:69–77
Wells G (2001) The Newcastle-Ottawa Scale (NOS) for assessing the quality of non randomised studies in meta-analyses http://www.ohrica/programs/clinical_epidemiology/oxford.asp. Accessed 6 Apr 2020
Weobong B, Ten Asbroek AHA, Soremekun S, Manu AA, Owusu-Agyei S, Prince M, Kirkwood BR (2014) Association of antenatal depression with adverse consequences for the mother and newborn in rural Ghana: findings from the DON population-based cohort study. PLoS One 9(12) (no pagination). https://doi.org/10.1371/journal.pone.0116333
Wrede G, Mednick SA, Huttunen M, Nilsson C (1980) Pregnancy and delivery complications in the births of unselected series of Finnish children with schizophrenic mothers. Acta Psychiatr Scand 62:369–381. https://doi.org/10.1111/j.1600-0447.1980.tb00623.x
Yedid Sion M, Harlev A, Weintraub AY, Sergienko R, Sheiner E (2016) Is antenatal depression associated with adverse obstetric and perinatal outcomes? J Matern Fetal Neonatal Med 1:863–867
Zax M, Sameroff AJ, Babigian HM (1977) Birth outcomes in the offspring of mentally disordered women. Am J Orthop 1:218–230
Zhong Q-Y, Gelaye B, Fricchione GL, Avillach P, Karlson EW, Williams MA (2018) Adverse obstetric and neonatal outcomes complicated by psychosis among pregnant women in the United States. BMC Pregnancy Childbirth 18:120
Zorn JV, Schür RR, Boks MP, Kahn RS, Joëls M, Vinkers CH (2017) Cortisol stress reactivity across psychiatric disorders: A systematic review and meta-analysis. Psychoneuroendocrinology 77:25–36. https://doi.org/10.1016/j.psyneuen.2016.11.036
Funding
This research is supported by funding from an Australian National Health and Medical Research Council (NHMRC) Project Grant (APP1127265) which funds AAA, CCJS, and HDB. BMF is funded by a NHRMC Project Grant (APP1098844).
Author information
Authors and Affiliations
Contributions
AAA, HDB, and CCJS conceived and designed the study. AAA conducted the statistical analyses and drafted the manuscript. HDB, VAM, MG, BMF, RM, SWW, and CCJS participated in the analysis or interpretation of data. All authors critically revised the manuscript for methodological and intellectual content and have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Adane, A.A., Bailey, H.D., Morgan, V.A. et al. The impact of maternal prenatal mental health disorders on stillbirth and infant mortality: a systematic review and meta-analysis. Arch Womens Ment Health 24, 543–555 (2021). https://doi.org/10.1007/s00737-020-01099-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00737-020-01099-9