Abstract
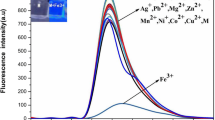
Two new compounds, 4-(2-bromophenyl-4,5-diphenyl-imidazol-1-yl)aniline(probe 1) and 4-[2,4,5-tris(4-bromophenyl)-1H-imidazol-1-yl]aniline(probe 2), were synthesized via a soft and high-efficiency one-pot microwave- assisted method under solvent-free conditions. Their sensing to different metal ions was detected by UV spectrophotometry and fluorescence spectrometry. Probe 2 revealed highly selective and sensitive UV and fluorescence response to Fe3+ ion. Upon the addition of Fe3+ ion, probe 2 showed obvious color change of the solution, conspicuous absorbance enhancement and relatively quick fluorescence quenching. The detection limit for Fe3+ ion was respectively calculated to be 0.72 μmol/L(fluorescent detection) and 0.48 μmol/L(UV-spectrum detection). Also, probe 2 was bound by Fe3+ ion to form a 1:1 complex. Moreover, preliminary application of probe 2 for detecting Fe3+ ion in aqueous solution was attempted, and satisfying results were obtained.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Aisen P., Wessling-Resnick M., Leibold E. A., Curr. Opin. Cjem. Biol. USA Universities, 1999, 3(2), 200
Touati D., Arch. Biochem. Biophys., 2000, 373(1), 1
Bonda D. J., Lee H. G., Blair J. A., Zhu X., Perry G., Smith M. A., Metallomics. Western Reserve University, 2011, 3(3), 267
Han J., Zhou Z., Bu X., Zhu S., Zhang H., Sun H., Yang B., Analyst Chinese Universities, 2013, 138(12), 3402
Suryawanshi S. B., Mahajan P. G., Bodake A. J., Spectrochim. Acta. A., Kolekar. G. B., Patil. S. R., Spectrochim. Acta A, 2017, 183, 232
Mohammed A. J., World Journal of Pharmace utical Research, 2015, 5(3), 129
Hao X., Wang D., Wang P., Wang Y., Zhou D., Environ. Monit. Assess. Chinese Universities, 2016, 188(1), 1
Luo A., Wang H. Q., Wang Y. Y., Huang Q., Zhang Q., Spectrochim. Acta A, 2016, 168, 37
Wang K. P., Lei Y., Zhang S. J., Zheng W. J., Chen J. P., Zhang Q., Zhang Y. B., Hu Z. Q., Sensor Actuat B: Chem., 2017, 252, 1140
Halliwell B., Gutteridge J. M., Method Enzymol., 1990, 186(186), 1
Halliwell B., Gutteridge J. M., Febs. Lett., 1992, 307(1), 108
Gaeta A., Hider R. C., Brit. J. Pharmacol., 2010, 146(8), 1041
Molina-Holgado F., Hider R. C., Gaeta A., Williams R., Francis P., Biometals, 2007, 20(3/4), 639
Bacon B. R., Britton R. S., Hepatology, 1990, 11(1), 127
Kehrer J. P., Toxicolog., 2000, 149(1), 43
Kowdley K. V., Gastroenterology, 2004, 127(Suppl. 1), S79
Valko M., Rhodes C. J., Moncol J., Izakovic M., Mazur. M., Chem-Biol Interact., 2006, 160(1), 1
Valko M., Leibfritz D., Moncol J., Int. J. Biochem. Cell B, 2007, 39(1), 44
Nayab P. S., Shkir M., Sensor Actuat. B: Chem., 2017, 245
Zhou Y., Yoon J., Chem. Soc. Rev., 2011, 40(7), 3416
Gao Y., Liu H., Liu Q., Wang W., Tetrahedron Lett., 2016, 57(17), 1852
Kumar N., Bhalla V., Kumar M., Analyst, 2014, 139(3), 543
Saleem M., Lee K. H., RSC Adv., 2015, 5(88), 72150
Hu Z. Q., Feng Y. C., Huang H. Q., Ding L., Wang X. M., Lin C. S., Sensor Actuat. B: Chem., 2011, 156(1), 54
Gupta V. K., Mergu N., Singh A. K., Sensor Actuat. B: Chem., 2015, 220, 420
Luxami V., Renukamal., Paul K., Kumar S., RSC Adv., 2013, 3(24), 9189
Fan S. M., Yang W. G., Hao J. F., Li H. J., Zhao W. D., Zhang J., Hu Y. H., J. Photoch. Photobio. A, 2016, 328, 129
Zhao J. X., Gao Q, Zhang F. F., Sun W., Bai Y. J., Polycycl. Aromat. Comp., 2018, 38(1), 13
Yan F. Y, Zheng T. C., Guo S S., Shi D. C., Han Z. Y., Zhou S. Y. S., Chen L., Spectrochim Acta B, 2015, 151, 881
Balalaie S., Hashemi M. M., Akhbari M., Tetrahedron Letters, 2003, 44(8), 1709
Katsuhiro S., Yoriko M., Seiji N., Kazunari N., Tomoki K.,Yoshiyuki O., Aryl or Heteroaryl Fused Imidazole Compounds as Indlammatory and Analgesic, US7141580, 2006
Bereket G., Hür E., Öğretir C., J. Mol. Struc.-Theochem., 2002, 578(1), 79
Mi Z. M., Chen Y., Chen X. D., Yan L. Q., Gu Q., Zhang H. Q., Chen C. H., Zhang Y. M., Chem. Res. Chinese Universities, 2018, 34(3), 369
Li Z. A., Lou X. D., Yu H. B., Li Z., Qin J. G., Macromolecules, 2008, 41(20), 7433
Gao D. W., Yu H. F., Jia J. L., Hua S. Y., Chen X. D., Acta Scientiarum Naturalium University Jilinensis, 1998, 1, 107
Zhou F., Leng T. H., Liu Y. J., Wang C. Y., Shi P., Zhu W. H., Dyes Pigments, 2017, 142, 429
Yan L. Q., Chen Y., Sun X. F., Yo M. J., Chen X. D., Gu Q., Zhang Y. M., Chem. Pap., 2016, 71(3), 1
Li J., Liu H. W., Meng F. Y., Yan L. Q., Shi Y. P., Zhang Y. M., Gu Q., Chem. Res. Chinese Universities, 2018, 34(2), 197
Wang L., Ye D., Li W., Liu Y., Li L., Zhang W., Ni L., Spectrochim. Acta A, 2017, 183, 291
Yan L. Q., Ma Y., Cui M. F., Anal. Methods, 2015, 7(15), 6133
Valeur B., Pouget J., Bourson J., Kachke M., Ernsting N. P., J. Phys. Chem., 1992, 96, 6545
Silva A. P. D., Gunaratne H. Q. N., Gunnlaugsson T., Huxley A. J. M., Mccoy C. P., Rademacher J. T., Chem. Rev., 1997, 97, 1515
Szacillowski K., Macyk H., Drzewiecka-Matuszek A., Brindell M., Stochel G., Chem. Rev., 2005, 105, 2647
Fagade U., Singh A., Chaitanya G. K., Singh N., Attarde S., Kuwar A., Spectrochim. Acta A, 2014, 121, 569
Li P., Zhao Y., Yao L., Nie H., Zhang M., Sensor Actuat. B: Chem, 2014, 191, 332
Bishnoi S., Milton M. D., J. Photoch. Photobio. A, 2017, 335, 52
Musa K., Kaya I., J. Ind. Eng. Chem., 2017, 46, 234
Chen J., Li Y., Lv K., Zhong W. B., Wang H., Wu Z., Yi P. G., Jiang J. H., Sensor Actuat. B: Chem., 2016, 224, 298
Frisch M. J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Petersson G. A., Nakatsuji H., Li X., Caricato M., Marenich A. V., Bloino J., Janesko B. G., Gomperts R., Mennucci B., Hratchian H. P., Ortiz J. V., Izmaylov A. F., Sonnenberg J. L., Williams-Young D., Ding F., Lipparini F., Egidi F., Goings J., Peng B., Petrone A., Henderson T., Ranasinghe D., Zakrzewski V. G., Gao J., Rega N., Zheng G., Liang W., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Throssell K., Montgomery J. A., Peralta J. E., Ogliaro F., Bearpark M. J., Heyd J. J., Brothers E. N., Kudin K. N., Staroverov V. N., Keith T. A., Kobayashi R., Normand J., Raghavachari K., Rendell A. P., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Millam J. M., Klene M., Adamo C., Cammi R., Ochterski J. W., Martin R. L., Morokuma K., Farkas O., Foresman J. B., Fox D. J., Gaussian 16, Revision A.03, Gaussian, Inc., Wallingford CT, 2016
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
40242_2019_8244_MOESM1_ESM.pdf
A New Tetrasubstituted Imidazole Based Difunctional Probe for UV-spectrophotometric and Fluorometric Detecting of Fe3+ Ion in Aqueous Solution
Rights and permissions
About this article
Cite this article
Shi, Y., Chen, X., Mi, Z. et al. A New Tetrasubstituted Imidazole Based Difunctional Probe for UV-spectrophotometric and Fluorometric Detecting of Fe3+ Ion in Aqueous Solution. Chem. Res. Chin. Univ. 35, 200–208 (2019). https://doi.org/10.1007/s40242-019-8244-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-019-8244-0