Abstract
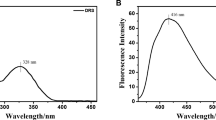
A small molecule fluorescent probe, 4-[2-(4-chlorophenyl)-4,5-diphenyl-1H-imidazol-1-yl]aniline(L) for detecting Ag+ ion was gently synthesized via one-pot multi-component reaction catalyzed by H3[PW12O40] under solvent-free microwave irradiation. When the concentration of Ag+ ion changed from 0 to 8.0×10−6 mol/L in the solution of H2O/CH3OH(19:1, volume ratio), the fluorescence emission spectrum was blue-shifted and accompanied by a gradual increase in fluorescence intensity with a low detection limit of 3.0×10−11 mol/L. Moreover, UV-Vis absorption titration experiment demonstrated a 1:1 stoichiometric ratio and an association constant of (9.95±0.44)×105 L/mol between probe L and Ag+ ion, and thus their complexation mechanism was also proposed and verified. More importantly, this fluorescent probe was remarkably specific for Ag+ ion under the interference of other metal ions and exhibited a wide pH application range of 4.0―8.0. Additionally, preliminary application of this probe was also carried out and satisfactory results were shown.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ye Z., Duan C., Sheng R., Xu J., Wang H., Zeng L., Talanta 2018, 176, 389
Zhao Q., Kang J., Wen Y., Huo F., Zhang Y., Yin C., Spectrochim. Acta A 2018, 189, 8
Melaiye A., Sun Z., Hindi K., Milsted A., Ely D., Reneker D. H., Tessier C. A., Youngs W. J., J. Am. Chem. Soc., 2005, 127, 2285
Yang Y., Li W., Qi H., Zhang Q., Chen J., Wang Y., Wang B., Wang S., Yu C., Anal. Biochem. 2012, 430, 48
Xiang G., Cui W., Lin S., Wang L., Meier H., Li L., Cao D., Sensor Actuat. B: Chem. 2013, 186, 741
Wang H., Zhu X., Xu H., Yuan Q., Sensor Actuat. B: Chem. 2017, 238, 985
Li H. W., Yue Y., Liu T. Y., Li D., Wu Y., J. Phys. Chem. C, 2013, 117, 16159
Lou X., Qiang L., Qin J., Li Z., ACS Appl. Mater. Inter., 2009, 1, 2529
Wang H., Wang D., Wang Q., Li X., Schalley C. A., Org. Biomol. Chem. 2010, 8, 1017
Huo F. J., Yin C. X., Yang Y. T., Su J., Chao J. B., Liu D. S., Anal. Chem. 2012, 84, 2219
Shin D. H., Ko Y. G., Choi U. S., Kim W. N., Ind. Eng. Chem. Res. 2006, 45, 656
Liu J., Lu Y., J. Am. Chem. Soc., 2007, 129, 9838
Mao J., Wang L., Dou W., Tang X., Yan Y., Liu W., Org. Lett. 2007, 9, 4567
Zhao Y., Zhang X. B., Han Z. X., Qiao L., Li C. Y., Jian L. X., Shen G. L., Yu R. Q., Anal. Chem. 2009, 81, 7022
Chen Y. Y., Lin H. C., Polymer 2007, 48, 5268
Tan J. L., Zhang M. X., Zhang F., Yang T. T., Liu Y., Li Z. B., Zuo H., Spectrochim. Acta A 2015, 140, 489
Bao Y., Li Q., Liu B., Du F., Tian J., Wang H., Wang Y., Bai R., Chem. Commun. 2012, 48, 118
Dierschke F., Müllen K., Macromol. Chem. Phys. 2007, 208, 37
Chen Y., Gu Q., Li B., Chen Q., Chen X., Zhang Y., Liu J., CR. Chim., 2013, 16, 1103
Baghbanzadeh M., Carbone L., Cozzoli P. D., Kappe C. O., Angew. Chem. Int. Ed. 2011, 50, 11312
Chen S. S., Cryst. Eng. Comm. 2016, 18, 6543
Faridbod F., Sedaghat M., Hosseini M., Ganjali M. R., Khoobi M., Shafiee A., Norouzi P., Spectrochim. Acta A 2015, 137, 1231.
Valeur B., Leray I., Coordin. Chem. Rev. 2000, 205, 3
Liu B., Baudrey S., Jaeger L., Bazan G. C., J. Am. Chem. Soc., 2004, 126, 4076
Ho H. A., Leclerc M., J. Am. Chem. Soc., 2004, 126, 1384
Wen Y., Xing F., He S., Song S., Wang L., Long Y., Li D., Fan C., Chem. Commun. 2010, 46, 2596
Askari M. S., Girard B., Murugesu M., Ottenwaelder X., Chem. Commun. 2011, 47, 8055
Zhang X., Wang G., Spectrochim. Acta A 2009, 72, 1071
Yang X. B., Yang B. X., Ge J. F., Xu Y. J., Xu Q. F., Liang J., Lu J. M., Org. Lett. 2011, 13, 2710
Benesi H. A., Hildebrand J. H., J. Am. Chem. Soc., 1949, 71, 2703
Zhu M., Yuan M., Liu X., Xu J., Lv J., Huang C., Liu H., Li Y., Wang S., Zhu D., Org. Lett. 2008, 10, 1481
Mirzaei M., Saeed J., Spectrochim. Acta A 2011, 82, 351
Li C. Y., Xu F., Li Y. F., Spectrochim. Acta A 2010, 76, 197
Mu H., Gong R., Ren L., Zhong C., Sun Y., Fu E., Spectrochim. Acta A 2008, 70, 923
Acknowledgements
We are grateful to Dr. WANG Chunyu(Jilin University) for NMR spectra test, Dr. WEI Zhonglin(Jilin University) for MS spectra test and Dr. DAO Jungang(Jilin University) for fluorescent emission spectra test.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
40242_2018_7426_MOESM1_ESM.pdf
Synthesis of highly sensitive fluorescent probe based on tetrasubstituted imidazole and its application for selective detection of Ag+ ion in aqueous media
Rights and permissions
About this article
Cite this article
Mi, Z., Chen, Y., Chen, X. et al. Synthesis of Highly Sensitive Fluorescent Probe Based on Tetrasubstituted Imidazole and Its Application for Selective Detection of Ag+ Ion in Aqueous Media. Chem. Res. Chin. Univ. 34, 369–374 (2018). https://doi.org/10.1007/s40242-018-7426-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-018-7426-5