Abstract
Congenital heart disease affects 1–2 % of the world’s population and is the leading cause of mortality among infants in the US. The diagnosis and management of congenital heart disease are largely driven by review of two-dimensional (2D) images derived from echocardiography, cardiac magnetic resonance, and cardiac computed tomography. However, congenital heart disease is a three-dimensional (3D) problem, and 2D display methods often lack critical spatial information. Cardiologists and cardiovascular surgeons rely on mental conversion of 2D data into a 3D understanding of the spatial relationships of intracardiac structures. Over the last 10 years, significant advances in 3D printing technology have made it possible to create life-like, printed models of any part of the human anatomy, including congenital heart defects. These printed models, placed in an operator’s hands, have the potential to assist in communication of the size, location, and degree of defect and aid in procedural planning. The use of 3D models has the potential to decrease operative procedure times, decrease radiation exposure in the cardiac catheterization laboratory, and overall sedation and anesthetic requirement. In addition, they have considerable educational value wherein defects can be examined from every angle, and the complex 3D relationships of cardiac structures can be displayed in three dimensions and held in the hand.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Congenital heart disease affects 1–2 % of the world’s population and is the leading cause of mortality among infants in the US [1••, 2]. The diagnosis and management of congenital heart disease are largely driven by review of two-dimensional (2D) images derived from echocardiography, cardiac magnetic resonance (CMR), and cardiac computed tomography (CT). However, congenital heart disease is a three-dimensional (3D) problem, and 2D display methods often lack critical spatial information. Cardiologists and cardiovascular surgeons rely on mental conversion of 2D data into a 3D understanding of the spatial relationships of intracardiac structures. Although 3D images of the heart represent a significant advancement, this still falls short of providing a precise roadmap for cardiovascular surgery or other type of intervention.
Over the last 10 years, significant advances in 3D printing technology have made it possible to create life-like, printed models of any part of the human anatomy [3], including congenital heart defects [4, 5]. These printed models, placed in an operator’s hands, have the potential to assist in communication of the size, location, and degree of defect and aid in procedural planning. In short, 3D models have the potential to decrease operative procedure times, decrease radiation exposure in the cardiac catheterization laboratory, and overall sedation and anesthetic requirement [6]. In addition, they have considerable educational value wherein defects can be examined from every angle. With 3D modes, the complex 3D relationships of cardiac structures no longer need to be inferred from 2D imaging, and they can be displayed in three dimensions and held in the hand [7•].
In this review, the current clinical cardiovascular imaging standards will be discussed, along with the process of creation of a 3D model from imaging. The utility of these models will be reviewed, both in terms of clinical utility and educational utility. Finally, future therapies involved with 3D printing of cardiac structures will be discussed.
Current Cardiovascular Imaging
Echocardiography
Echocardiography is the main diagnostic imaging technique of the heart, largely owing to its portability, safety, excellent spatial and temporal resolution, and ease of use. This is particularly true for infants and children with favorable ultrasound windows who are being imaged to understand their congenital heart defect. A single acquisition in 2D echocardiography requires specific placement of the ultrasound probe to display heart anatomy in standardized cross-sectional views (subcostal, parasternal, apical and suprasternal with their respective long axis and short axis planes), and then recording the image over several heart beats. The 3D orientation of a heart lesion can be obtained from a single acquisition using special 3D probes and software to process and display the acquired data [8]. Occasionally, an acquisition performed over longer recording (more heart beats) can be “stitched” together for increased temporal and spatial resolution with acquisition over more heart beats, ideally with respiratory suspension. Experienced clinicians are required to then mentally piece together the heart anatomy to fully understand the defect and guide therapy.
This mental reconstructive process can be difficult for 3D interpretation of atypical structures in congenital heart disease—for example, atrioventricular orientation in a crisscross heart, systemic and pulmonary venous blood flow in an inter-atrial baffle, or ventricular septal defects in double-outlet right ventricle. With the advancement of 3D ultrasound probes and software, 3D images can also be obtained in one acquisition [9], although they are still limited by the acoustic quality of transthoracic imaging. Echocardiography is also often limited in evaluating extracardiac vessels such as the aorta and the pulmonary arteries due to acoustic shadowing.
Cross-Sectional Imaging (CMR and CT)
The images provided by CMR and CT allow for effective 3D interpretation of cardiovascular anatomy, as the images are displayed in a consistent cross-sectional plane. Additionally, CMR and CT allow for image acquisition of extracardiac vessel anomalies not well visualized by echocardiography. Vascular rings, pulmonary slings, and coarctation of the aorta are appreciated well with contrast-enhanced CMR or CT angiography [10••]. The distal pulmonary vasculature in congenital heart disease can also be well defined with cardiac imaging angiograms, such as the major aortopulmonary collateral arteries (MAPCAs) seen in Tetralogy of Fallot with pulmonary atresia [11]. However, traditional cross-sectional imaging without post-processing still requires a mental reconstruction of anatomy.
Creating 3D Heart Models
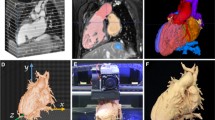
The creation of 3D models is a collaborative process that requires coordination between imaging personnel and clinicians to properly represent complex heart structures. As shown in Fig. 1, the relevant cardiac structures should be carefully identified and preserved during each step of creation, from data acquisition to image segmentation and subsequent post-processing.
Creation of a cardiac model. CMR images were obtained from an adult patient with transposition of great arteries status post Mustard atrial switch procedure, followed by image segmentation, creation of a digital model and, finally, a printed model. a Three orthogonal views (axial, sagittal, and coronal) of the 3D dataset with segmentation masks overlaid in color. Noted are the systemic venous baffle (blue mask), left ventricle (orange mask), pulmonary arteries (purple mask), pulmonary venous baffle (red mask), right ventricle (yellow mask), and aorta (green mask). b The segmentation software reconstructs a 3D digital object from each respective segmentation mask. c A combination of Boolean functions, smoothing, and wrapping functions are used to finalize the 3D digital model. Cuts are made onto the model to display internal anatomy. d The 3D digital models are transferred to a 3D printer and physical components are printed as shown. Each piece is held together by magnets. The internal chambers are also painted with color to represent the flow of saturated and desaturated blood (Color figure online)
Data Acquisition
Creation of a 3D heart model begins with acquisition of a 3D image dataset from CT angiography, cardiac MRI, or 3D echocardiogram. The optimal dataset for 3D imaging should have adequate spatial resolution such that the blood pool and myocardium are contrasted and defined clearly. As with all cardiac imaging modalities, ECG gating is necessary to ensure that the images acquired are in the same cardiac phase (typically end of diastole to maximize volume). For 3D echocardiograms, probe technique and ultrasound settings are particularly important to minimize stitch artifact, and the image datasets may require post-acquisition processing such as using filters to remove ultrasound speckle and other image noise [12••].
Image Segmentation
The conversion of a 2D image dataset into a digital 3D model is typically performed by a process known as “segmentation,” where each 2D image in the dataset is partitioned into boundaries that define regions or masks (Fig. 1a). By stacking adjacent 2D images together, the defined contours across each respective image form a 3D reconstruction (Fig. 1b). This technique is different from other conventional 3D visualization methods such as volume rendering, which have limited capability in 3D editing. Segmentations can be performed manually by drawing contours between the endocardium and blood pool, or automated via thresholding, edge detection, region growing, and other functions that select image voxels of similar intensity and other characteristics (in this case, the blood pool), or by a combination of both [13]. Numerous freeware and commercial software are currently available for image segmentation; two examples are given in these references [14•: http://www.itksnap.org/pmwiki/pmwiki.php; http://biomedical.materialise.com/mimics].
While image segmentations of static organs can easily be performed automatically, the complexity and large patient-to-patient variation of congenital heart disease often demands some degree of manual segmentation. Segmentation should also be performed in a systematic and meticulous manner. Thin-walled cardiac structures such as the atrial septum are often misrepresented by the automated thresholding function. Separate masks should be created to represent respective atria, ventricles, and great vessels. This limits the potential for overlap artifact that would inadvertently represent an inter-chamber communication that did not exist. Segmentations should be reviewed by overlaying the masks with the original images to ensure proper representation of all cardiac structures.
Preparation and Display of 3D Printed Model
The 3D reconstructions generated by image segmentation can then be exported as digital 3D triangulated surface models [typically stored in a STL (STereoLithography) file format]. The 3D digital models often require further editing by software, such as removing small artifacts, smoothing the wall of the models, or adding thickness to external features (Fig. 1c). Further preparation of the model can be made based on the intended viewing method. 3D models can be viewed digitally via software; in those cases, simple cuts to the model can be made to display internal anatomy. Ideally, the cuts should be made such that the relevant anatomy is in full view (for example, removing the right ventricular-free wall in order to visualize a ventricular septal defect). For 3D models that are expected to be printed into physical objects (Fig. 1d), the cuts should also be oriented properly to allow for adequate connections to hold the respective pieces together. At our institution, small circular holes are cut onto the articulating surfaces of each piece for placement of magnets to hold the pieces together with anatomically correct alignment. Using newest printing and post-processing techniques enables the creation of soft and multi-colored models that closely match the look and feel of a heart during surgery.
Preparation and Display of 3D Digital Model
Software applications are available for display of 3D digital models of the heart, using the STL file that would be sent to the printer, and loading it into an application designed for 3D display. There are many types of software packages available with user interfaces with varying degrees of “user-friendliness” and ability to modify and manipulate the file [https://indicated.co/cardiac-review-3d-printing-surgical-planning/]. The advantages of displaying the 3D model in a digital environment (as opposed to slicing the file and printing physical models) are that the user is not constrained by predefined cut planes which section the model to view the intracardiac defect. There are advantages to viewing an intracardiac defect from every angle and magnification. Another advantage of digital models is the ability to easily share and archive the models, while printed display of these defects conveys a sense of reality that is difficult to match with digital displays. Coming closest to reality, novel holographic display of the 3D cardiac imaging data [15] holds promise for future display of cardiac defects.
Clinical Value of 3D Heart Models
Digital 3D models are useful in improving the understanding of congenital heart disease without requiring a mastery of the reconstructive visualization process. In complex septal defects, or septal defects associated with other lesions, 3D models can help with procedural planning [16–18•]. Integration of 3D modeling can enhance the cardiac surgeon’s understanding of heart defects [19, 20], thus increasing efficiency of surgical interventions and reducing procedural time. This has been shown in particular, in Tetralogy of Fallot with MAPCAs, where there is a high degree of variability among patients with this disease [21•]. In fluoroscopic-based cardiac catheterizations, 3D models can help the interventionist with planning approach, such as planning out catheter course and appropriate device placement, thereby reducing radiation exposure and contrast dose required for the procedure [22, 23]. Finally, 3D models have been utilized in understanding aortic stenosis in the setting of decreased left ventricular systolic function to gain a deeper understanding of the aortic valve dysfunction through 3D display [24].
Patient-specific care team briefings can also be performed with 3D models, to enhance bedside comprehension in the postoperative care of a congenital heart disease patient after surgical repair [25•].
Educational Value of 3D Heart Models
Utilization of 3D printed models of the human anatomy has long been shown to be excellent adjunctive teaching aids in various parts of the human anatomy [26, 27]. This is particularly true in congenital heart disease, where a wide spectrum of severity is observed within each type of cardiac defect. This wide spectrum of severity makes learning about these lesions complex to trainees, and it is encouraging that evidence suggests 3D models aid in learning about congenital heart defects and their clinical sequelae for trainees of various levels [28, 29]. Complex anatomical concepts in congenital heart disease such as malalignment-type ventricular septal defects, double-outlet right ventricle, and the spectrum of heterotaxy syndromes can be demonstrated in a hands-on, interactive environment. Depth and spatial relationships can be expressed and elaborated on. The learner can have a primary, tangible experience with a particular type of heart defect. With pediatric residents, 3D printed models of tetralogy of Fallot used during a standard teaching session were more highly rated than sessions without the models, and learner satisfaction scores were higher, which correlated with subject matter retention in adult learners [30].
Work is ongoing to test the ability of an interactive model displaying hypoplastic left heart syndrome with moveable components that can be magnetically attached to simulate creation and takedown of a Sano shunt, creation, and takedown of a Blalock-Taussig shunt, creation of a Glenn anastomosis and creation of a lateral tunnel intracardiac Fontan. This model is being used with trainees in a “Build-A-Fontan” type session directed toward understanding the physiology of hypoplastic left heart syndrome and the need for three-staged palliative repair. Results from this trial should be available soon.
Finally, patient-specific 3D models created from patient data may also be beneficial during consultations with family members regarding a patient’s diagnosis of congenital heart disease. The models make the details of a particular congenital heart defect more readily attainable, regardless of educational background.
Future Therapeutic Utility of 3D Printed Cardiac Devices
In addition to the use of 3D printing for improved diagnosis, education, and interventional planning, research is also underway to utilize 3D printing to create surgical implants for patient-specific vascular grafts, heart valves, and eventually an entire heart. This is either achieved by printing a scaffold (either acellular or seeded with cells) that provides a 3D template for tissue growth or direct bioprinting of cellular structures. Researchers have explored the use of fabricating grafts with solvent-cast molding processes, but 3D printing the graft directly allows for creating more complex 3D designs of the grafts to best match the patient’s anatomy including bifurcations [31, 32•].
Thus far, researchers have created 3D printed polyurethane scaffolds using an inkjet technique potentially suitable for vascular materials [33]. Another group created bio-tubular scaffolds combining 3D printing and electrospinning techniques [34]. Researchers also developed 3D printing of vascular tissue engineering scaffolds utilizing poly(propylene fumarate) (PPF) and demonstrated its efficacy in the mouse venous system [35].
Current work in bioprinting using hyaluronan hydrogels and tubular channels encapsulating cells in alginate has accomplished the fabrication of biologically functional blood vessels [36]. Another group has implemented 3D bioprinting to fabricate living alginate/gelatin hydrogel valve conduits with anatomical architecture [37].
Conclusion
3D printed models of congenital heart disease are an innovative method of displaying 3D cardiovascular imaging. These models can be made using a roadmap from 3D echocardiograms, or angiograms from cardiac CT or CMR images. Models have enormous potential for improving clinical care, and have already gained an important place in education of medical trainees and allied health professionals. Work is ongoing to print exciting therapeutics, such as pulmonary artery conduits, heart valve tissues, and other potentially implantable devices. Innovative imaging display techniques like 3D printing and 3D digital display of cardiovascular imaging are fast becoming a mainstay of diagnosis and treatment in congenital heart disease.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
•• Marelli AJ, Mackie AS, Ionescu-Ittu R, Rahme E, Pilote L. Congenital heart disease in the general population: changing prevalence and age distribution. Circulation. 2007;115(2):163–72. This reference is required for all pediatricians to understand the impact of congenital heart disease and the extent of complex congenital heart disease that requires intervention.
Ferencz C, Rubin JD, McCarter RJ, Brenner JI, Neill CA, Perry LW, Hepner SI, Downing JW. Congenital heart disease: prevalence at livebirth. The Baltimore-Washington Infant Study. Am J Epidemiol. 1985;121(1):31–6.
Rengier F, Mehndiratta A, von Tengg-Kobligk H, Zechmann CM, Unterhinninghofen R, Kauczor H-U, Giesel FL. 3D printing based on imaging data: review of medical applications. Int J Comput Assist Radiol Surg. 2010;5(4):335–41.
Noecker AM, Chen J-F, Zhou Q, White RD, Kopcak MW, Arruda MJ, Duncan BW. Development of patient-specific three-dimensional pediatric cardiac models. ASAIO J. 2006;52(3):349–53.
Greil GF, Wolf I, Kuettner A, Fenchel M, Miller S, Martirosian P, Schick F, Oppitz M, Meinzer H-P, Sieverding L. Stereolithographic reproduction of complex cardiac morphology based on high spatial resolution imaging. Clin Res Cardiol. 2007;96(3):176–85.
Kurup HK, Samuel BP, Vettukattil JJ. Hybrid 3D printing: a game-changer in personalized cardiac medicine? Expert Rev Cardiovasc Ther. 2015;13(12):1281–4.
• Ejaz F, Ryan J, Henriksen M, Pophal S, Richardson R, Frakes D. Color-coded patient-specific physical models of congenital heart disease. Rapid Prototyp J. 2014;20:336–43. This reference gives extensive description of process of making congenital heart disease models with 3D printing technology.
Lang RM, Badano LP, Tsang W, Adams DH, Agricola E, Buck T, Faletra FF, Franke A, Hung J, de Isla LP, Kamp O, Kasprzak JD, Lancellotti P, Marwick TH, McCulloch ML, Monaghan MJ, Nihoyannopoulos P, Pandian NG, Pellikka PA, Pepi M, Roberson DA, Shernan SK, Shirali GS, Sugeng L, Ten Cate FJ, Vannan MA, Zamorano JL, Zoghbi WA, American Society of Echocardiography, European Association of Echocardiography. EAE/ASE recommendations for image acquisition and display using three-dimensional echocardiography. J Am Soc Echocardiogr. 2012;25(1):3–46.
Cossor W, Cui VW, Roberson DA. Three-dimensional echocardiographic en face views of ventricular septal defects: feasibility, accuracy, imaging protocols and reference image collection. J Am Soc Echocardiogr. 2015;28(9):1020–9.
•• Gerrah R, Bardo DM, Reed RD, Sunstrom RE, Langley SM. Adjustment of the surgical plan in repair of congenital heart disease: the power of cross-sectional imaging and three-dimensional visualization. Congenit Heart Dis. 2014;9(1):E31–6. This reference helps the reader to understand the power of 3D display.
Nallamshetty L, Dadlani GH, Berlowitz MS. Unrepaired tetralogy of fallot with major aortopulmonary. Congenit Heart Dis. 2011;8:24–30.
•• Olivieri LJ, Krieger A, Loke Y-H, Nath DS, Kim PCW, Sable CA. Three-dimensional printing of intracardiac defects from three-dimensional echocardiographic images: feasibility and relative accuracy. J Am Soc Echocardiogr. 2015;28(4):392–7. This is the first paper publishing the process and validation technique for 3D printing heart models from 3D echo, the most versatile of all of the cardiac imaging modalities.
Riesenkampff E, Rietdorf U, Wolf I, Schnackenburg B, Ewert P, Huebler M, Alexi-Meskishvili V, Anderson RH, Engel N, Meinzer H-P, Hetzer R, Berger F, Kuehne T. The practical clinical value of three-dimensional models of complex congenitally malformed hearts. J Thorac Cardiovasc Surg. 2009;138(3):571–80.
• http://mitk.org/wiki/Interactive_segmentation. Accessed 16 April 2016. This is an interactive guide on the image segmentation process.
Hunziker PR, Smith S, Scherrer-Crosbie M, Liel-Cohen N, Levine RA, Nesbitt R, Benton SA, Picard MG. Dynamic holographic imaging of the beating human heart. Circulation. 1999;99(5):1–6.
Jacobs S, Grunert R, Mohr FW, Falk V. 3D-Imaging of cardiac structures using 3D heart models for planning in heart surgery: a preliminary study. Interact CardioVasc Thorac Surg. 2008;7(1):6–9.
Mottl-Link S, Hübler M, Kühne T, Rietdorf U, Krueger JJ, Schnackenburg B, De Simone R, Berger F, Juraszek A, Meinzer H-P, Karck M, Hetzer R, Wolf I. Physical models aiding in complex congenital heart surgery. Ann Thorac Surg. 2008;86(1):273–7.
• Schmauss D, Haeberle S, Hagl C, Sodian R. Three-dimensional printing in cardiac surgery and interventional cardiology: a single-centre experience. Eur J Cardiothorac Surg. 2015;47(6):1044–52. This is a robust experience from one clinical center and reviews all previous reports of 3D model utility in cardiology.
Kim MS, Hansgen AR, Wink O, Quaife RA, Carroll JD. Rapid prototyping: a new tool in understanding and treating structural heart disease. Circulation. 2008;117(18):2388–94.
Sodian R, Weber S, Markert M, Rassoulian D, Kaczmarek I, Lueth TC, Reichart B, Daebritz S. Stereolithographic models for surgical planning in congenital heart surgery. Ann Thorac Surg. 2007;83(5):1854–7.
• Ryan JR, Moe TG, Richardson R, Frakes DH, Nigro JJ, Pophal S. A novel approach to neonatal management of tetralogy of Fallot, with pulmonary atresia, and multiple aortopulmonary collaterals. JACC Cardiovasc Imaging. 2015;8(1):103–4. This is a case where 3d models are particularly helpful in a specific disease process and are used in an ongoing manner.
Olivieri L, Krieger A, Chen MY, Kim P, Kanter JP. 3D heart model guides complex stent angioplasty of pulmonary venous baffle obstruction in a Mustard repair of D-TGA. Int J Cardiol. 2014;172(2):e297–8.
Lazkani M, Bashir F, Brady K, Pophal S, Morris M, Pershad A. Postinfarct VSD management using 3D computer printing assisted percutaneous closure. Indian Heart J. 2015;67(6):581–5.
Maragiannis D, Jackson MS, Igo SR, Schutt RC, Connell P, Grande-Allen J, Barker CM, Chang SM, Reardon MJ, Zoghbi WA, Little SH. Replicating patient-specific severe aortic valve stenosis with functional 3D modeling. Circ Cardiovasc Imaging. 2015;8(10):e003626.
• Olivieri L, Su L, Hynes C, Krieger A, Alfares F, Ramakrishnan K, Zurakowski D, Marshall M, Kim P, Jonas R, Nath D. Just-in-time training using 3D printed cardiac models after congenital cardiac surgery. World J Pediatr Congenit Heart Surg. 2016;7(2):164–8. This is a description of utility of 3D models in preparation of bedside caregivers following heart surgery.
Hu A, Wilson TD, Ladak H, Haase P, Fung K. Three-dimensional educational computer model of the larynx. Head Neck. 2011;135(7):677–81.
Knobe M, Carow JB, Ruesseler M, Leu BM, Simon M, Beckers SK, Ghassemi A, Sönmez TT, Pape H-C. Arthroscopy or ultrasound in undergraduate anatomy education: a randomized cross-over controlled trial. BMC Med Educ. 2012;12(1):85.
Costello J, Olivieri L, Su L, Krieger A, Alfares F, Thabit O, Marshall MB, Yoo S, Kim P, Jonas RA, Nath DS. Incorporating three-dimensional printing into a simulation-based congenital heart disease and critical care training curriculum for resident physicians. Congenit Heart Dis. 2015;10:185–90.
Costello JP, Olivieri LJ, Krieger A, Thabit O, Marshall MB, Yoo S-J, Kim PC, Jonas RA, Nath DS. Utilizing three-dimensional printing technology to assess the feasibility of high-fidelity synthetic ventricular septal defect models for simulation in medical education. World J Pediatr Congenit Heart Surg. 2014;5(3):421–6.
Loke Y-H, Krieger A, Harahsheh A, and Olivieri L. A randomized controlled trial of 3D models in pediatric cardiology resident education. Pediatr Acad Soc Meet
Nelson GN, Mirensky T, Brennan MP, Roh JD, Yi T, Wang Y, Breuer CK. Functional small-diameter human tissue-engineered arterial grafts in an immunodeficient mouse model: preliminary findings. Arch Surg. 2008;143(5):488–94.
• Melchiorri AJ, Hibino N, Brandes ZR, Jonas RA, Fisher JP. Development and assessment of a biodegradable solvent cast polyester fabric small-diameter vascular graft. J Biomed Mater Res A. 2014;102(6):1972–81. This is an important report of 3D printed material which is implantable and may one day be the basis for heart valves, heart conduits and patch materials used in congenital heart surgery.
Zhang C, Wen X, Vyavahare NR, Boland T. Synthesis and characterization of biodegradable elastomeric polyurethane scaffolds fabricated by the inkjet technique. Biomaterials. 2008;29(28):3781–91.
Lee SJ, Heo DN, Park JS, Kwon SK, Lee JH, Lee JH, Kim WD, Kwon IK, Park SA. Characterization and preparation of bio-tubular scaffolds for fabricating artificial vascular grafts by combining electrospinning and a 3D printing system. Phys Chem Chem Phys. 2015;17(5):2996–9.
Melchiorri AJ, Hibino N, Best CA, Yi T, Lee YU, Kraynak CA, Kimerer LK, Krieger A, Kim P, Breuer CK, Fisher JP. 3D-printed biodegradable polymeric vascular grafts. Adv Healthc Mater. 2015;5:309–25.
Yu Y, Zhang Y, Martin JA, Ozbolat IT. Evaluation of cell viability and functionality in vessel-like bioprintable cell-laden tubular channels. J Biomech Eng. 2013;135(9):91011.
Duan B, Hockaday LA, Kang KH, Butcher JT. 3D Bioprinting of heterogeneous aortic valve conduits with alginate/gelatin hydrogels. J Biomed Mater Res A. 2013;101A(5):1255–64.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
Tom Loke, Craig Sable, Axel Krieger, and Laura Olivieri have a pending US Patent App 14/333,234 for three-dimensional printed replicas of patient’s anatomy for medical applications.
Human and Animal Rights and Informed Consent
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional review board and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
This article is part of the Topical Collection on Cardiology.
Rights and permissions
About this article
Cite this article
Loke, T., Krieger, A., Sable, C. et al. Novel Uses for Three-Dimensional Printing in Congenital Heart Disease. Curr Pediatr Rep 4, 28–34 (2016). https://doi.org/10.1007/s40124-016-0099-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40124-016-0099-y