Abstract
Micronutrients are essential factors for human health and integral for plant growth and development. Among the micronutrients, zinc (Zn) and iron (Fe) deficiency in dietary food are associated with malnutrition symptoms (hidden hunger), which can be overcome through biofortification. Different strategies, such as traditional and molecular plant breeding or application of chemical supplements along with fertilizers, have been employed to develop biofortified crop varieties with enhanced bioavailability of micronutrients. The use of microorganisms to help the crop plant in more efficient and effective uptake and translocation of Zn and Fe is a promising option that needs to be effectively integrated into agronomic or breeding approaches. However, this is less documented and forms the subject of our review. The major findings related to the mobilization of micronutrients by microorganisms highlighted the significance of (1) acidification of rhizospheric soil and (2) stimulation of secretion of phenolics. Plant–microbe interaction studies illustrated novel inferences related to the (3) modifications in the root morphology and architecture, (4) reduction of phytic acid in food grains, and (5) upregulation of Zn/Fe transporters. For the biofortification of Zn and Fe, formulation(s) of such microbes (bacteria or fungi) can be explored as seed priming or soil dressing options. Using the modern tools of transcriptomics, metaproteomics, and genomics, the genes/proteins involved in their translocation within the plants of major crops can be identified and engineered for improving the efficacy of plant–microbe interactions. With micronutrient nutrition being of global concern, it is imperative that the synergies of scientists, policy makers, and educationists focus toward developing multipronged approaches that are environmentally sustainable, and integrating such microbial options into the mainframe of integrated farming practices in agriculture. This can lead to better quality and yields of produce, and innovative approaches in food processing can deliver cost-effective nutritious food for the undernourished populations.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
In the pursuit of increasing food grain production and feeding the ever increasing global population, agricultural research in the last few decades mainly focused on cultivation of high yielding varieties of crops and intensive cropping systems, mainly involving the imbalanced use of chemical fertilizers (Elkoca et al. 2010; Foley et al. 2005; Gliessman 2014; Singh 2000). It is well-documented in literature that the intensive use of chemical fertilizers has a negative impact on soil ecology, as it disrupts nutrient equilibrium in the soil, leading to impaired soil structure and functioning. Imbalances in fertility lead to harmful impacts on the proliferation of communities and health of macro/micro fauna, flora, and human beings (Lockhart et al. 2013; Wu and Ma 2015). In order to understand the relationship between micronutrient supply and human health, there is an urgent need to understand the level of micronutrient deficiencies in soils and their influence of crop physiology. Millions of hectares of land in the world, including India, have low availability of micronutrients. In Indian soils, the deficiency of Zn has gone as high as 47% and that of Fe to 13% (Singh 2009). Micronutrient deficiency in soil is mainly attributed to high levels of more than the recommended dose of fertilizers (RDF), soil erosion, and other agronomic practices that interfere with the translocation of micronutrients. This causes imbalances in the amounts of organic matter and phosphorus in soil, which is particularly accentuated in soils such as calcareous type, water-logged, peat, arid, alkaline, sandy, and saline soils, which also limit Fe mobilization (Alloway 2008). The mobilization of Fe in plants often occurs via chelating with phytosiderophores, citrate, nicotianamine, and mugineic acid (strategy II), or in the form of free iron ions–Fe2+ (both strategy I and strategy II plants) (Zhang et al. 2019). Fe2+ is the free form of iron in the soil which is limited in soil. Fe2+ is a more soluble form and can be taken up by the roots of all types of plants. But it is readily oxidized into Fe3+ due to chemical reactions. Fe3+ is the most dominant form of iron in soil but it is insoluble, therefore not available to plants (Mahender et al. 2019). In this regard, plants have developed different mechanisms to acquire sufficient iron under deficient conditions. Nongraminaceous plants, known as strategy I plants, use a reduction-based strategy to reduce the ferric ions (Fe3+) to free form of Fe ions (Fe2+). Graminaceous plants, known as strategy II plants, have a chelation-based strategy for acquisition of Fe3+. Strategy II plants secrete phytosiderophore (PS) in the rhizosphere which form PS–Fe3+ soluble complex, taken up by plant roots (Kobayashi and Nishizawa 2012; Römheld and Marschner 1986). Micronutrients are essential for plant growth and development, and their presence in sufficient amounts is important for proper human and animal health (Welch and Graham 2004). In the last two decades, the concept of hidden hunger (deficiency of certain vitamins and micronutrient) has been well-documented (Nilson and Piza 1998). Low availability of micronutrients in soils not only reduces crop yields, but also leads to poor nutritional quality of the edible parts of crops, resulting in malnutrition in human populations, particularly in developing and underdeveloped countries (Hurst et al. 2013; Kumssa et al. 2015). The micronutrients Fe and Zn are important for all organisms. Fe is an important cofactor for various enzymes involved in plant and human metabolism; its deficiency causes stunted growth and anemia (Hentze et al. 2004). About 25% of the world’s population suffers from anemia (Ministry of Health and Family Welfare 2016; World Health Organization 2008), and the Global Burden of Disease Study (2015) reported that Fe-deficiency anemia led to 54,000 deaths in 2015 (Forouzanfar et al. 2016). Low Zn levels lead to stunted growth and development of neonatal, immune dysfunction, hypogonadism, and impairment in cognition (Sauer et al. 2016). An average of 17.3% of the world population is affected by Zn deficiency (Wessells and Brown 2012) and about 433,000 children perish due to Zn malnutrition (World Health Organization 2009). Iron deficiency in humans is relatively easy to quantify, as hemoglobin is the most commonly used biochemical indicator of population response, which is reliable. On the other hand, zinc deficiency is difficult to quantify, as no reliable biochemical indicator is currently available to denote zinc status (Mei et al. 2005; Wieringa et al. 2015).
Biofortification is a promising and sustainable agriculture-based strategy to minimize Zn and Fe deficiency in dietary food substances (Garcia et al. 2016; Petry et al. 2016; Vasconcelos et al. 2017). Among the different strategies deployed, the plant breeding approach to develop biofortified crops and agronomic supplementation of micronutrients, such as foliar/soil application along with chemical fertilizers, have received maximum attention (Cakmak et al. 2010; Di Tomaso 1995; Rengel 2001).
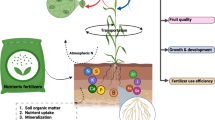
A less investigated approach, which is promising, involves exploring plant–microbe interactions, which are known to have a crucial role in improving the nutritional status of soil and enriching micronutrients through metal solubilization, mobilization, and translocation to different parts of the plant (Chen et al. 2014a, 2014b; Kothari et al. 1990; Rana et al. 2012) (Fig. 1). Therefore, microorganisms can be used to enhance the accumulation of micronutrients in the grains of staple cereal crops; this has been successful in rice and wheat (Mader et al. 2011; Prasanna et al. 2016; Rana et al. 2012, 2015; Singh et al. 2017a, b, Singh et al. 2018, Singh et al. 2020; Vaid et al. 2014; Zhang et al. 2012a, b). However, its potential is still to be explored across other crops, ecologies, and farming systems.
In this review, an attempt has been made to collate and critically analyze the available information on the major mechanisms employed by microorganisms for the enrichment of micronutrients in the plant, and in particular, their prospects for the biofortification of Fe and Zn.
2 Zn and Fe deficiency in soil—a global concern
Zn deficiency is one of the important nutritional constraints in cereal crops, especially rice and wheat, and Zn-deficient cereal-based diets create serious problems for human health. Zn deficiency was found to be prevalent in 50% of the soil samples collected from different countries (Dharejo et al. 2011; Hansen et al. 1996; Manyevere et al. 2017). Fageria et al. (2002) observed that under such deficiencies, cereal crop yields can exhibit growth and yield a reduction, up to 80%, along with a reduced grain Zn concentration. Mark et al. (2016) reported that micronutrient deficiencies are predominant in the low income South Asian countries, including India.
Deficiency of Zn is the most widespread among the micronutrients, in almost 50% of soils surveyed from India (Reza et al. 2017; Shukla et al. 2017, 2018). Analyses of 0.25 million surface soil samples collected from different parts of the country revealed the predominance of Zn deficiency in divergent soils (Singh et al. 2005). The magnitude of Zn deficiency varies widely among soil types and within the various states in India. In India, the extent of deficiency of Zn was to the tune of 86% in Maharashtra, 72.8% in Karnataka, 60.5% in Haryana, 58.4% in Tamil Nadu, 57% in Meghalaya, 54% in Bihar and Orissa, 49.4% in Andhra Pradesh, and 48.1% in Punjab (Shukla et al. 2015, 2016, 2017, 2018; Singh 2009). Such an alarming prevalence is a crucial issue.
Low availability of DTPA (diethylenetriamine pentaacetic acid) extractable Fe in soils is also of global concern. Fe deficiency-mediated interveinal chlorosis in crops is a widespread phenomenon in arid and semiarid soils or calcareous soils worldwide, including India, which causes a significant loss in yield (Mortvedt 1991). In India, Fe is mainly deficient in the soils of Karnataka (35%), H.P. (27%), Maharashtra (24%), Haryana (20%), Tamilnadu (17%), and Punjab (14%) (Shukla et al. 2015, 2016, 2017, 2018; Singh 2009).
3 Causes of micronutrient deficiency in soil
Different environmental and edaphic factors, such as organic matter content in soil, soil pH, cation exchange capacity, clay content etc., and soils more prone to water logging, or peat/calcareous soils are characteristics that affect the bioavailability of micronutrients (Ibrahim et al. 2011; Lindsay 1984; Ramzan et al. 2014). The critical limit of DTPA-extractable Fe and Zn in soil are 4.5 and 0.6 mg kg−1, respectively (Alloway 2009; Sillanpaa 1982). The critical limit for the soil is defined as minimum soil test value which is associated with maximum crop yield. It represents the concentration below which deficiency manifests as it designates the lower end of the sufficiency range.
A major percentage of Fe on the earth’s crust is present as Fe3+ which is not readily accessible to plants. Fe2+ is the more soluble form of iron but readily oxidized to ferric form (Fe3+), which is precipitated in oxide/hydroxide, phosphate, carbonate, and other unavailable complex forms in the soil (Lindsay and Schwab 1982). A large amount of Fe is present in soils, but its bioavailability is very low. Chirwa and Yerokun (2012) and Harter (1983) suggested that Zn bioavailability decreased in soil, with increasing soil pH due to precipitation or adsorption of Zn on the surface of CaCO3 and Fe oxides. There is a negative correlation between available Zn or Fe and cation exchange capacity (CEC) of soil (Yoo and James 2002). Sidhu and Sharma (2010) reported that with increasing clay content in the soils, there was a lower availability of Zn. The available Zn was negatively correlated with electrical conductivity (EC) (Chattopadhyay et al. 1996). Gao et al. (2011) also stated that the availability of Zn is negatively correlated with phosphorus content in soil.
4 Role of Zn and Fe in plant growth and development
Among the micronutrients essential for the proper growth and development of plants, animals, and human beings, Zn and Fe are the most important elements. Even though they are needed in trace amounts, they are involved in critical functions and play an important role in the maintenance of the structural integrity of biological membranes, gene expression, and regulation (Hänsch and Mendel 2009; Mahender et al. 2019; Mousavi et al. 2013). Additionally, they are also involved in mediating several processes such as carbohydrate metabolism, protein synthesis, nucleic acid synthesis, phytohormone levels, photosynthesis, fertility, seed production, and defense against abiotic/biotic stresses (Brown et al. 1993; Kobayashi and Nishizawa 2012; Rout and Sahoo 2015). Romheld and Marschner (1991) illustrated that Zn is essential for carbohydrate metabolism, protein synthesis, nucleic acid synthesis, and phytohormone synthesis and also as a cofactor for all six classes of enzymes. Fe is an important constituent of enzymes like peroxidase, catalase, and nitrogenase (Kerkeb and Connoly 2006), and along with Mo, plays an important role in nitrogen fixation (Kim and Rees 1992). Fe deficiency decreases chlorophyll production, leading to interveinal chlorosis, which is exhibited as sharp distinctions between veins and chlorotic areas in young leaves (Kobayashi et al. 2003), followed progressively by the entire leaf becoming whitish-yellow and necrosis, along with slower plant growth (Follett and Westfall 1992). White and Broadley (2009) recommended a serious concern towards the alleviation of micronutrient deficiency in soil, as it not only leads to declining crop yields but also contributes to poor quality of produce, leading to dietary micronutrient deficiencies in human beings.
5 Role of Zn and Fe in human health
Zn has an important structural and functional role in biological systems (Parkin 2004), as it plays a significant role in reproductive, catalytic, and anabolic processes in human beings (Bonaventuraa et al. 2015; Corvol et al. 2004; Cummings and Kovacic 2009). Zn deficiency causes stunted growth of human beings, immaturity of sexual organs, and distortions of the immune system and central nervous system (Welch 2001). Ross et al. (1985) reported that women who receive Zn supplements of 4–13 mg day−1 delivered babies with low birth weight compared to the control group. Shankar and Prasad (1998) also reported infertility in Zn-deficient men. Zn deficiency may cause congenital diseases like Acrodermatitis enteropathica (Moynahan 1974). The recommended daily allowance (RDA) for Zn is 15 mg day−1 for adults (Food and Nutrition Board 2001).
Fe is a crucial element for human fitness, as it is involved in a wide variety of metabolic processes, including deoxyribonucleic acid (DNA) synthesis, catalase and peroxidase enzyme synthesis, oxygen and electron transport, etc. (Abbaspour et al. 2014). The recommended dietary allowance (RDA) for Fe for adults of 19–50 years age is 8 mg day−1 for males and 18 mg day−1 for females, but during the pregnancy period, it is 27 mg day−1 (Food and Nutrition Board 2001). In the 2013 Global Burden of Diseases Study, Fe deficiency anemia was predominantly implicated in nearly 200,000 deaths and 45 million disability-adjusted life-years (DALYs) lost annually (4.5% of all risk-attributable DALYs; Forouzanfar et al. 2016). Fe biofortification in India is essential, where over 50% of women and 74% of children are anemic (International Institute for Population Sciences and ORC Macro 2000), attributed largely to the insufficient intake or bioavailability of Fe. Stevens et al. (2013) showed that anemic prevalence was highest in pre-school age children, pregnant, and nonpregnant women in Western Pacific, Southeast Asia, and Africa. In a nationwide study in India, Chellan and Paul (2010) found a moderate to severe Fe deficiency anemia in 47.9% pre-school children (below 6 years), 74.8% in adolescent girls (10–19 years), and 41.5% in pregnant women (15–44 years).
6 Strategy of Zn and Fe biofortification in food grains
Biofortification is a bouquet of approaches that focus on improving the availability of micronutrients biologically in staple food products like wheat, maize, pearl millet, rice, and others. This could be achieved genetically or through soil management practices, agronomic approaches, or by using microbiological interventions or a combination of these. Although plants can take up higher amounts of micronutrients from soil, their availability in edible parts may be low, because of anti-nutritional factors, thus having no influence in alleviating human malnutrition (Frossard et al. 2000). Some of the possible interventions include the subsections that follow.
6.1 Agronomic interventions
Applying mineral fertilizers to the soil for maintaining soil health and improving plant quality is an age old practice (Rengel et al. 1999). In general, it is observed that the response to an applied nutrient, in terms of its translocation to grains, is more pronounced when there is a deficiency of that particular element in the soil or the characteristics of the particular element allow its rapid mobilization. Micronutrient supplementation with chemical fertilizers has been an effective measure employed by farmers to gain maximize crop yield. However, micronutrient-use efficiency in crops is low and only 2–5% of total applied fertilizer dose is utilized (Tian et al. 2008). Some of the methodologies for improving use efficiency of micronutrients, such as foliar application or granular/dust-type formulations of Zn or modulating dose and frequency of applications or enrichment of urea and other fertilizers, have shown promising results in several crops, particularly cereals (Prasad et al. 2013; Shivay et al. 2008, 2015; Wissuwa et al. 2008).
6.2 Genetic approaches
Genetic biofortification involves classical breeding approaches to characterize and exploit the genetic variation for mineral content, as well as new approaches involving gene discovery and marker-assisted breeding (Grusak 2002). Hindu et al. (2018) used genome-wide association studies (GWAS) for identification of different genomic regions in maize for kernel Zn and Fe biofortification. Velu et al. (2016) suggested that genomic selection (GS) may be a potential breeding method for Fe and Zn biofortification in wheat. In terms of sustainability, the nutrition-oriented breeding of crop plants has several advantages. Recent advances empower us to modulate signaling pathways, although breeding, by and large, relies on long and repetitive cycle of hybridization and selection, which are time-consuming and labor-intensive. Nevertheless, in recent years, modern molecular tools like DNA markers and marker-assisted selection (MAS) technologies are expediting the development of nutrient-rich genotypes. Kumar et al. (2018) reported quantitative trait loci (QTLs) for Fe and Zn biofortification in pearl millet using diversity array technology (DArT) and simple sequence repeat (SSR) markers. Literature reported that many Fe- and/or Zn-biofortified varieties of rice, wheat, and maize have been released in the world, including in India, to alleviate malnutrition (Table 1). Besides the genetic or plant breeding approach, several transgenic interventions have been applied for successful biofortification of food crops. Transgenic techniques permit the exchange of genes between totally irrelevant species or bring new genes into food or cash crops. Ramesh et al. (2004) developed a novel approach for increased seed zinc and iron content, through the overexpression of a zinc transporter in Hordeum vulgare cv. Golden Promise, facilitated by a ubiquitin promoter. Goto et al. (1999) were able to enhance the iron content in rice grains 3-fold using an Agrobacterium-mediated transfer of complete coding sequence of the ferritin gene from soybean plants. Lucca et al. (2002) developed transgenic rice plants, with higher iron content, which was rich in phytase and cysteine-peptides; this can facilitate better iron intake and bioavailability. Vasconcelos et al. (2003) were able to engineer the expression of the soybean ferritin gene, under the control of the glutelin promoter in an elite Indica rice line that has highly desirable agronomic and field performance traits; enhanced grain nutritional levels were recorded not only in brown grains but also in polished grains. Liu et al. (2004) developed ferritin-incorporated rice varieties that showed 64% higher iron content in the milling, and this ferritin gene could be specifically expressed in the endosperm of transgenic rice with a high level.
Genetic approaches can be a challenging task for breeders in soils inherently low in Fe and Zn micronutrients. To realize the full potential of biofortified varieties, there is a need to also give simultaneous attention toward other factors, like soil pH and organic matter, which influence root exudation and enzyme activities in the rhizosphere, and thereby micronutrient uptake and accumulation (Cakmak 2008).
6.3 Microbe-based approaches
The mechanisms of Fe acquisition by higher plants under Fe deficiency have been categorized into two groups (Kobayashi and Nishizawa 2012; Römheld and Marschner 1986): strategy I in nongraminaceous plants and strategy II in graminaceous plants. The two main processes in the strategy I response are (1) the reduction of ferric chelates at the root surface with the help of Ferric reduction oxidase gene (FRO2) and (2) the absorption of the generated ferrous ions across the root plasma membrane by the Iron-regulated transporter gene (IRT1). Other processes involved in strategy I include extrusion of proton and phenolics compounds from the roots to the rhizosphere, which increases the solubility of ferric ions or supports the reduction capacity of ferric Fe on the root surface. Strategy II plants take up Fe under Fe deficiency by the excretion of phytosiderophores (PSs), which are low molecular weight Fe chelating compounds, i.e., mugineic acids (MA) and nicotianamine (NA) have strong affinity for Fe (III) and form an Fe–phytosiderophore soluble complex. The Fe–phytosiderophore complex is then transported into root cells through a high affinity uptake system. Suzuki et al. (2006) reported that barley plants secreted mugineic acid (MA) phytosiderophore under Zn deficiency and the form Zn (II)–mugineic acid complex and Zn (II)–mugineic acid complex absorbed more than Zn2+ by the roots of a Zn-deficient plant. The amounts and kinds of phytosiderophores secreted by plants into the rhizosphere vary from species to species (Mori 1999).
There are large amounts of Fe and Zn present in the earth’s crust but unavailable to plants, as they are present in the form of insoluble salts. Plant-based intrinsic strategies like phytosiderophore or organic acid production or secretions of chelators are not always sufficient to make micronutrients available in micronutrient-deficient soils. With our improved understanding of crosstalk between soils, plants, and microorganisms, greater insights into the rhizosphere environment have been gained (De Santiago et al. 2011; Mishra et al. 2011; Pii et al. 2015; Zaidi et al. 2003). Plant growth-promoting microorganisms play a crucial role in the fortification of macronutrients and micronutrients in food crops, through various mechanisms such as siderophore production, transformations, nitrogen fixation, and phosphorus mobilization (Khan et al. 2019; Singh et al. 2018). Figure 1 illustrates the diverse microbe-mediated mechanisms involved in the biofortification of Fe.
Microorganisms are known to have crucial roles in the biofortification of Zn and Fe in cereal grains (Gosal et al. 2010; Rana et al. 2012; Sharma et al. 2012). Both rhizospheric and endophytic microorganisms have significant impacts on micronutrient bioavailability to plants. However, endophytic microorganisms are considered more promising agents to enhance Fe and Zn uptake and translocation, because endophytic microorganisms can indirectly influence the regulation of metal transporters (Reiter et al. 2002; Weyens et al. 2013). Bacterial and fungal endophytes have been implicated in the biofortification of grains of wheat and rice with Fe and Zn (Abaid-Ullah et al. 2015; Ramesh et al. 2014). Balakrishnan and Subramanian (2012) revealed that the inoculation of mycorrhizal fungi (arbuscular mycorrhiza) improves the availability of micronutrients, particularly Zn in soils, as a consequence of rhizospheric acidification and siderophore production besides hyphal transport of nutrients through the external mycelium. Gosal et al. (2010) reported that an endophytic fungus Piriformaspora indica had a significant impact on plant growth, biomass, and micronutrients uptake. Wang et al. (2014) also found a positive influence of endophyte inoculation on Zn accumulation in rice grains. In earlier studies, Bacillus subtilis (DS-178) and Arthrobacter sp. (DS-179) were able to increase the Zn content by an average of 75% over the control in Zn-deficient soils. Similar results were also found with Arthrobacter sulfonivorans (DS-68) and Enterococcus hirae (DS-163), with respect to Fe availability in wheat grains (Singh et al. 2017a, 2018). Utilization of PGP rhizobacterium Pseudomonas fluorescens was found to be suitable towards Zn biofortification in wheat grains (Sirohi et al. 2015). Rana et al. (2012) reported that Fe content in wheat grains increased significantly due to inoculation of Providencia sp. PW5. Similarly, Prasanna et al. (2015) and Tariq et al. (2007) also found a significant effect of rhizospheric microorganisms on Zn biofortification in maize and rice, respectively.
Both rhizospheric and endophytic microorganisms play crucial roles in metal solubilization in soil and redistribution in the plant parts. Bacteria capable of solubilizing insoluble sources of Zn can enhance uptake of Zn by 21% in soybean plants (Sharma et al. 2012). There are different mechanisms through which microbes increase the availability of Zn and Fe in soil and enhance their mobilization in plant parts or increase the bioavailability of Fe and Zn in food grains. These include the following:
-
6.3.1 Production of siderophores and other chelating substances
-
6.3.2 Organic acid secretion and proton extrusion
-
6.3.3 Modification in root morphology and anatomy
-
6.3.4 Upregulation of Zn and Fe transporters
-
6.3.5 Reduction of phytic acids or anti-nutritional factors in food grains
-
6.3.6 Secretion of phenolics and related reducing moieties
-
6.3.7 Secretion of phytohormones like signaling molecules
6.3.1 Production of siderophore and other chelating substances
Siderophores are low molecular weight Fe chelating compounds that have high affinity toward Fe (III) (Ganz 2018). The literature suggests that many microorganisms secrete siderophores to overcome the Fe deficiency in soil (Schalk et al. 2011). Fe (III) is insoluble in soil, but siderophores form siderophore–Fe (III) complexes and increase the availability of Fe in the environment (Saha et al. 2012). Because of their solubilizing effect on Fe hydroxides, the production of siderophores in the rhizosphere is the key microbial activity that benefits plant Fe acquisition (Desai and Archana 2011; Hayat et al. 2012). Khalid et al. (2015) reported that the inoculation of siderophore-producing fluorescent Pseudomonas was effective in enhancing Fe content in chickpea grains. In our earlier field experiments, inoculation with siderophore-producing endophytes (Arthrobacter sulfonivorans DS-68 and Enterococcus hirae DS-163) enhanced the Fe concentration in grains of low and high Fe accumulating wheat genotypes by 67 and 46%, respectively, over the uninoculated control, as compared to the application of RDF (recommended doses of fertilizers) + FeSO4, in which 63 and 28% enhancement was recorded over the uninoculated control, respectively. The percent increase of Fe was more pronounced with low Fe accumulating wheat cultivars due to application of siderophore-producing endophytes or FeSO4, as compared to high Fe accumulating wheat genotypes. It was interesting to note that microbial treatment was statistically on par with chemical fertilizer (FeSO4) application, in terms of Fe content in wheat plant tissues (Singh et al. 2018).
Zn cations, being more reactive species in soil, are present in low amounts in soil solution (Alloway 2009); hence, the bioavailability of Zn ions in soil is meager. However, Zn chelating compounds (synthetic or plant and microbial derived) can enhance the mobilization and solubilization of Zn fractions in soil (Obrador et al. 2003). Such chelators form soluble complexes with Zn2+ (Tarkalson et al. 1998) and decrease their interactions with soil constituents. Whiting et al. (2001) observed the significant impact of Zn chelator metallophores, produced by bacteria (Microbacterium saperdae, Pseudomonas monteilii, and Enterobacter cancerogenes), on the bioavailability of Zn in soil and uptake by plant roots. Tariq et al. (2007) also reported that the Azospirillum lipoferum (JCM-1270, ER-20), Pseudomonas sp. (96-51), and Agrobacterium sp. (Ca-18) bioinoculants were able to solubilize the zinc hydroxide and zinc phosphate or other insoluble Zn salts and increase the availability of Zn to rice plants for a longer time in soil. This was found to be mediated by the production of natural chelating agents (Tariq et al. 2007). According to the reports of Kucey (1988), inoculation of Penicillium bilaji increased Zn solubilization and uptake in plants to a greater extent as compared to that achieved through ethylene diamine tetra acetate (EDTA) chelating mechanisms. Singh et al. (2018) reported a positive correlation between siderophore production and Fe accumulation in wheat grains by endophytes (Fig. 2a), which illustrates the important role of siderophores in iron acquisition and transport into the plant; however, its translocation within the plant, particularly to grains, involves several steps and mechanisms—transport, remobilization, and storage processes, mediated by membrane transporters, chelators, and regulatory proteins.
Relationship between endophytic bacterial activities (siderophore secretion, organic acid production, and Zn solubilization) and Fe/Zn accumulation in wheat tissues. a Siderophore production and Fe acquisition in grains. b Zn solubilization and Zn acquisition in grains. c Organic acid secretion and Fe accumulation in root and shoot. d Organic acid secretion and Zn accumulation in root and shoot
6.3.2 Organic acid secretion and proton extrusion
The plant root exudates significantly increase the soluble proportion of Zn in soil solution through different biochemical processes (Zhang et al. 2010); however, microorganisms can also modify the root exudation patterns and influence the activities in the rhizosphere (da Silva et al. 2014; Malinowski et al. 2004; Singh et al. 2017b; Subramanian et al. 2009). Plants mediate through mobilization and solubilization of metal cations through root exudates by several biochemical routes: (i) acidification of rhizosphere through proton ions or organic acids in root exudates; (ii) formation of soluble complex of metal ions with amino acids or organic acids and other chelators; (iii) enzymatic redox reaction reactions; or indirectly through (iv) biostimulation effect of root exudates on beneficial microbes in the rhizosphere (Pérez-Esteban et al. 2013; Sessitsch et al. 2013; Ström et al. 2002).
Organic acids represent the dominant moiety in the root exudates, particularly, in relation to metal solubilization in the rhizosphere region (Chiang et al. 2011; Luo et al. 2014). Kim et al. (2010) reported that oxalic and citric acids secreted by Echinochloa crusgalli significantly increased the micronutrient uptake and translocation in plant tissues. The nature of soil affects the availability of micronutrients to plants. When chemical fertilizers are applied to soil, most of the phosphorus, potassium, Zn, or Fe precipitates in soil, making them unavailable for use by plants. Micronutrient availability is sensitive to soil pH; it has been reported that minimal changes in pH have drastic effects on the solubility of micronutrients in soil. Havlin et al. (2007) suggested that the availability of Zn decreases 100 times with one unit increase in pH. Similarly, Guerinot and Yi (1994) found that the availability of Fe decreased up to 1000-fold, when pH was increased by one unit. Several reports have been published on the positive contribution of organic acid-producing microorganisms in increasing the availability of nutrients (P, K, Zn) in the rhizosphere (Goswami et al. 2014; Meena et al. 2015; Sirohi et al. 2015). Chen et al. (2014a, 2014b) and Raja et al. (2006) observed significant changes in the root exudate pattern and solubilization of precipitated metals due to inoculation of plant growth-promoting rhizobacteria. Singh et al. (2018) also reported that the Zn solubilization activity of endophytic bacteria positively correlates with the accumulation of Zn in wheat grains (Fig. 2b).
In addition to organic acids, proton excretion by microorganisms may lead to acidification of the rhizosphere and an increase in the availability of nutrients. Asea et al. (1988) reported that the phosphorus solubilizing fungi—Penicillium bilaji and Penicillium cf. fuscum—significantly increase the phosphorus availability in soil by lowering the soil pH, as a result of secretion of protons. Besides phosphate-solubilizing bacteria (PSB), mycorrhizae also excrete H+. Thus, the presence of protons in micronutrient-deficient soils may help the crop plant in more efficient uptake of micronutrients.
In our earlier investigation, the inoculation of Arthrobacter sulfonivorans (DS-68) and Arthrobacter sp. DS-179 in wheat plants led to both qualitative and quantitative changes in organic acid profiles over the control, with several fold changes in the quantity of organic acids in inoculated treatments. This also positively correlated with increasing amounts of Zn and Fe in wheat root and shoot tissues (Singh et al. 2017b) (Fig. 2c and d).
6.3.3 Modification in root morphology and anatomy
Zn or Fe hyperaccumulator plants have various morphological, anatomical, and physiological adaptations, such as elaborative root hairs or root surface area, and metal-mobilizing root exudates (Dong et al. 1995; Genc et al. 2007; Lynch 2007). Singh et al. (2005) also reported that a well-developed root system of plants can be an important strategy to enhance the uptake of micronutrients. Zn-efficient plants appear to employ a plethora of physiological mechanisms that allow them to withstand Zn deficiency stress better than Zn-inefficient plants. Zn-efficient plants have a greater proportion and longer length of fine roots (≤ 0.2 μM), and this plays an important role in the differential Zn efficiency observed among various genotypes (Rengel and Wheal 1997). In general, Zn-efficient plants have thinner roots with increased surface area, which increases the availability of Zn along with other nutrients, due to a more thorough exploration of the soil (Singh et al. 2005). Chen et al. (2009) for rice and Genc et al. (2006) for wheat showed that the Zn-efficient genotype developed longer and thinner roots (≤ 0.2 mm) than a less Zn-efficient genotype. Available reports also illustrate that the inoculation of plant growth-promoting rhizobacteria and endophytic bacteria has notable effects on root morphology and architecture (Delaplace et al. 2015; Vacheron et al. 2013; Wang et al. 2014). Our earlier published work (Singh et al. 2017a) also supports this hypothesis that inoculation with a plant growth-promoting, Zn-solubilizing endophyte (Bacillus subtilis DS-178 and Arthrobacter sp. DS-179) enhances the root volume, surface area, root length, root diameter, and average number of root tips in a wheat crop. Inoculation of these Zn-solubilizing endophytes also enhanced Zn accumulation in wheat grains by 2-fold over the control, which, in turn, was better or equal to the application of 40 kg ZnSO4 ha−1.
Investigations undertaken in low available Fe content soil with inoculation of siderophore-producing Arthrobacter sulfonivorans DS-68 and Enterococcus hirae DS-163 endophytes led to increases in the root surface area and average number of root tips by 2-fold and 1.6-fold, respectively, over the control (RDF). However, in high available Fe content soil, root surface area and average number of root tips increased by 1.5-fold and 1.2-fold, respectively, over the control (RDF). These increased root parameters directly facilitated Fe fortification in wheat grains (Singh 2016). Chen et al. (2014a, 2014b) reported that inoculation with endophytic bacterium Sphingomonas SaMR12 significantly improved the root length, root surface area, and average number of root tips in Sedum alfredii plant, as compared to uninoculated treatment. Batista et al. (2016) suggested that root morphological parameters, such as total root length and root surface area, play an important role in nutrient uptake, particularly under micronutrient-limiting conditions.
Besides root morphology, the internal structure of roots is also modified by plant growth-promoting microbes, as a consequence of which a higher amount of nutrients is taken up by roots from the soil (López-Bucio et al. 2007; Ortíz-Castro et al. 2008). Rêgo et al. (2014) conducted an experiment with rice plants to understand the effect of the bioinoculant (bacteria and a fungus) on root anatomical features. They observed that inoculation of Trichoderma asperellum, Burkholderia pyrrocinia, and Pseudomonas fluorescens had a significant impact on root anatomy, particularly in terms of the diameter of the root cortex, dimension of vascular bundles, and numerical changes in the protoxylem and metaxylem vessels. Several reports suggest that well-developed root anatomical features, such as expansion of the root cortex and volume of xylem vessels, more elaborative root hairs, thickening of the endodermis, and vascular bundles, were positively correlated with nutrient uptake (Kotula et al. 2009; Kramer et al. 1980). In our earlier investigation, inoculation of both Arthrobacter sulfonivorans DS-68 and Arthrobacter sp. DS-179 individually enhanced the volume of xylem vessels, thickness of the root cortex, and diameter of vascular bundles and pericycle (Fig. 3). Inoculation of these microorganisms also enhanced the Fe or Zn uptake by 60 to 75% (Singh et al. 2017b). Thus, the modification of internal structure of roots, which leads to better anchoring and uptake system as a result of inoculation, can be one of the strategies that also results in biofortification of micronutrients in crop plants.
6.3.4 Upregulation of Zn and Fe transporters
Micronutrient uptake and translocation are both different processes; in some crop genotypes, micronutrient uptake efficiency is very high, but the translocation of micronutrients from root to shoot and shoot to grains is very poor (Singh et al. 2018). Therefore, nutrient translocation or redistribution in plant parts is an important phenomenon that needs to be modulated to increase the micronutrients in the edible parts of plant. Tables 2 and 3 provide details of Fe and Zn transporters identified in various crops.
Arbuscular mycorrhiza (AM) fungal inoculants were found to bring about significant variations in the uptake of nutrients and their accumulation in roots, shoots, and grains (Chatzistathis et al. 2009). This indicated the significant role of metal transporters in the translocation of nutrients from shoots to grains. There are several metal transporters in plants, including a zinc-regulated transporter (ZRT)/iron-regulated transporter (IRT)-like protein (ZIP) family, which is involved in the translocation of Zn and Fe, a cation diffusion facilitator (CDF) family, and a P-type ATPase family involved in xylem loading of Zn as well as other heavy metals (Colangelo and Guerinot 2006; Eide 2006). The ZIP family proteins have been reported in rice, wheat, maize, and Arabidopsis thaliana (Evens et al. 2017; Grotz et al. 1998; Ishimaru et al. 2005; Krämer et al. 2007; Xu et al. 2010). The overexpression of these proteins led to an accumulation of excess amounts of Zn in the cells of wild emmer wheat (Durmaz et al. 2011). The Zn-solubilizing Enterobacter cloacae strain ZSB14 has been reported to upregulate OsZIP1 and OsZIP5 expression and downregulate OsZIP4 expression in rice genotypes (Krithika and Balachandar 2016). In Hordeum vulgare, the colonization of Rhizophagus irregularis improved grain Zn concentrations under Zn-deficient conditions through upregulation of HvZIP13 (Watts-Williams and Cavagnaro 2018).
Our recent published reports also support this hypothesis that microbes enhance the expression of ZIP genes. In the shoots, inoculation of Zn-solubilizing Arthrobacter sp. DS-179 endophyte led to 1.9- and 4.0-fold increase in TaZIP3 and TaZIP7 transcripts, respectively. The expression levels of TaZIP7 in shoots due to siderophore-producing Arthrobacter sulfonivorans DS-68 endophyte inoculation were 2.6-fold higher than uninoculated control, and TaZIP3 was not influenced by endophyte inoculation, whereas in roots, inoculation of Arthrobacter sp. DS-179 endophyte led to 1.7-fold increase in TaZIP3 gene and 40% downregulation in TaZIP7 gene with respect to uninoculated control. The expression level of TaZIP3 and TaZIP7 genes in roots due to Arthrobacter sulfonivorans DS-68 inoculation was 1.5- and 2.2-fold higher than uninoculated control, respectively (Fig. 4) (Singh et al. 2017b).
Relative changes in TaZIP3 and TaZIP7 gene expression levels (expressed in fold increase) after 30 days of sowing, in low Fe and Zn accumulating wheat genotype-4HPYT-414, due to endophyte inoculation (Arthrobacter sulfonivorans DS-68 or Arthrobacter sp. DS-179), over control. Data represents the average of three replicates; error bars depict standard deviation (SD)
6.3.5 Reduction of phytic acids or anti-nutritional factors in food grains
Another aspect relevant to biofortification strategies is the bioavailability of micronutrients in cereal and legume grains, which is often low because it is affected by anti-nutritional factors such as phytic acid (Liang et al. 2008). Phytic acid forms chelation complexes with metals (Cu, Fe, Mn, Zn, etc.) and decreases the bioavailability of these micronutrients in dietary food, thus acting as an anti-nutritional factor (Hunt 2003; Kumssa et al. 2015). Vaid et al. (2014) reported that the inoculation of Burkholderia sp. SG1 + Acinetobacter sp. SG3 led to a reduction in phytate:Zn ratio in the grains of rice.
In our recent report, inoculation of the promising two Zn-solubilizing (Bacillus subtilis DS-178 and Arthrobacter sp. DS-179) and two siderophore-producing endophytes (Arthrobacter sulfonivorans DS-68 and Enterococcus hirae DS-163) brought about significant enhancement in wheat plant growth, biomass, yield, and micronutrient uptake. Interestingly, endophyte inoculation decreased phytic acid concentration in wheat grains by approximately 26% over the RDF. This reduction of phytic acid concentration in grains may be correlated with increasing Fe or Zn concentration in grains (r = − 0.825 for phytic acid vs Fe content in grains; r = − 0.660 between phytic acid and Zn content in grains), as depicted by the positive and significant correlations between phytic acid content in wheat grains of genotypes classified as low/high accumulators (Singh et al. 2018). However, more research needs to be undertaken to decipher the exact mechanisms involved.
6.3.6 Stimulation of secretion of phenolics like reducing substances
In nongraminaceous monocots and dicots (strategy I plants), phenolic compounds are the most frequently reported as a component of the root exudates produced in response to Fe deficiency (Curie and Briat 2003; Römheld and Marschner 1986; Susin et al. 1996). Compared to other compounds in the root exudates, phenolics are particularly interesting because of their multiple chemical and biological functions, such as chelating, reducing, radical scavenging, antimicrobial activity, and serving as a carbon source for microbial growth (Blum et al. 2000; Cao et al. 1997; Jin et al. 2007; Rice-Evans and Miller 1996). It has been suggested that the released phenolics function to enhance Fe availability in the rhizosphere soil, as an alternative or supplement to the plasma membrane-bound ferric reductase, through chelating and reducing insoluble Fe (Dakora and Phillips 2002). Recently, it has been reported that the removal of the secreted phenolics from a hydroponic culture solution significantly enhances Fe accumulation and Fe deficiency responses in roots; this is attributed to the inhibition of solubilization and utilization of apoplasmic Fe (Jin et al. 2007). Moreover, phenolics such as protocatechuic acid (PCA) are reported to chelate Fe (III) and solubilize and reduce it to Fe (II) in vitro (Yoshino and Murakami 1998).
Moreover, many rhizosphere beneficial microbes that elicit the induced systemic resistance (ISR) improve Fe and perhaps Zn acquisition in strategy I plants (in strategy II plants, this has been less studied), through the induction of Fe deficiency responses (Romera et al. 2019; Verbon et al. 2017). The ISR and the Fe uptake signaling pathways interact in plant roots via the transcription factor MYB72, which controls the biosynthesis of Fe-mobilizing phenolics. MYB72-dependent BGLU42 activity is required for the secretion of Fe-mobilizing phenolics into the rhizosphere and the onset of ISR, as outlined by Verbon et al. (2017). They illustrated that the colonization of Arabidopsis plant roots (strategy I plants) by ISR eliciting beneficial microbes, e.g., Pseudomonas spp. and Trichoderma spp., activates the FIT (Fer-like Fe-deficiency-induced transcription factor) regulated transcription factor gene MYB72 and the Fe uptake genes FRO2 (Ferric reduction oxidase 2) and IRT1 (Iron-regulated transporter 1). Volatile organic compounds secreted by ISR inducing microbes coordinate MYB72 expression in Arabidopsis roots during the onset of induced systemic resistance and Fe deficiency responses through the activation of the FIT transcription factor gene (Zamioudis et al. 2015). Transcription factor MYB72 controls the biosynthesis of phenolic compounds and the expression of the glucose hydrolase gene BGLU42 (β-glucosidase 42) and the ABC (ATP-binding cassette) transporter gene PDR9 (Pleiotropic Drug Resistance transporter 9). Glucose hydrolase activity of BGLU42 (β-glucosidase 42) is involved in the processing of phenolic compounds to enable their secretion into the rhizosphere. Induced ABC (ATP-binding cassette) transporter gene PDR9 (Pleiotropic Drug Resistance 9) is involved in the secretion of processed phenolic compounds from plant roots to the rhizosphere, and the phenolic compounds chelate and mobilize Fe3+, making it available for the reduction by FRO2 and IRT1 genes and uptake by the roots. The antimicrobial activity of some phenolic compounds may play a role in shaping the rhizosphere microbial community. BGLU42 is required for rhizobacteria-mediated ISR and, when overexpressed, confers resistance against a broad spectrum of plant pathogens (Zamioudis et al. 2014).
Neotyphodium coenophialum can stimulate the exudation of phenolic compounds in the rhizosphere of continental tall fescue (Lolium arundinaceum) with chelating characteristics; this was implicated in directly improving Fe uptake (Malinowski et al. 2004; Malinowski and Belesky 2000).
6.3.7 Secretion of phytohormones like signaling molecules
Several phytohormones, such as gibberellic acid and cytokinins, are key players in metal stress mitigation (Al-Hakimi 2007; Gangwar et al. 2010; Masood et al. 2016; Zhu et al. 2012). There are several reports on the effect of various phytohormones on Fe uptake gene expression—IRT1 (Iron-regulated transporter 1) and FRO2 (Ferric reduction oxidase 2). Auxin is known to act positively in FRO2 induction under Fe deficiency (Chen et al. 2010); ethylene is also a positive regulator of IRT1 and FRO2 in Arabidopsis and cucumber plants (Lucena et al. 2006).
Enhancing Fe-deficiency-inducible responses can facilitate an increase in the plant acquisition of Fe from Fe-limited soils. Xie et al. (2009) reported that the soil bacterium Bacillus subtilis GB03 could enhance Fe acquisition of Arabidopsis plants by activating the Fe-deficiency-inducible responses, suggesting that soil microorganisms could regulate plant Fe acquisition via signaling processes. In the last decade, plant physiologists have made efforts to uncover the signals responsible for triggering Fe deficiency responses in plant roots, and several hormonal compounds have been identified as signaling elements (Hindt and Guerinot 2012; Ivanov et al. 2012; Kobayashi and Nishizawa 2012). These include auxins (Chen et al. 2010), nitric oxide (NO) (Graziano and Lamattina 2007), ethylene (García et al. 2011), cytokinin (Seguela et al. 2008), and brassinosteroids (Wang et al. 2012). Among these, auxins, NO, and ethylene are particularly interesting, as several soil microorganisms can produce these compounds. This helps to emphasize the significant and potential interactions between soil microorganisms and Fe uptake of plants. Auxins have been demonstrated to be an important chemical signal, enhancing Fe-deficiency-inducible responses. Exogenous addition of synthetic auxin, either IAA or α-naphthaleneacetic acid, enhances Fe-deficiency-induced reduction of ferric Fe, expression of FRO2 and IRT1, and development of root hairs and lateral roots to increase the surface area for Fe uptake (Chen et al. 2010; Wu et al. 2012). Production of auxin-like compounds by soil microorganisms can play a similar role and prove beneficial for plant Fe uptake under Fe-limited conditions. In support of this interaction, auxins produced by a microbe, isolated from soil mixed with phenolics, and secreted from Fe-deficient red clover plants markedly enhanced the activity of ferric chelate reductase in roots of Fe-deficient plants (Jin et al. 2006).
7 Conclusions and future outlook
The availability of low amounts of micronutrients is generally known as “hidden hunger” and draws less consideration than the conspicuous starvation of individuals. As an example, micronutrient deficiency refers to a situation in which a family can just earn enough to eat rice but not any of the natural products, including vegetables and meat that would make a nutritious diet. There is a need to understand, as biologists, that there is an intricate networking between soil, plants, and microbiomes of soil and plants that is responsible for crop productivity and soil fertility. Hence, along with plant breeding and agronomic fortification, significant efforts need to be undertaken to include microbes as partners in such approaches. Therefore, focused research needs to be undertaken toward the following:
-
Priming seeds with microbes or their products to improve bioavailability of micronutrients or reduce phytic acid in grains
-
Understanding the interactions of the microbiome of plants and gut microflora for improving absorption, including prebiotics and Fe absorption in the human gut
-
Conserving and exploring our native germplasm of wild cereals to identify the novel plant–microbiome combinations responsible for micronutrient enrichment
-
Development of food grain mixes, with probiotic or prebiotic supplements
Future challenges include a synergism of technologies toward providing nutritious food for the growing populations, using sustainable and environment-friendly technologies.
References
Abaid-Ullah M, Hassan MN, Jamil M, Brader G, Shah MKN, Sessitsch A, Hafeez FY (2015) Plant growth promoting rhizobacteria: an alternate way to improve yield and quality of wheat (Triticum aestivum). Int J Agric Biol 17:51–60
Abbaspour N, Hurrell R, Kelishadi R (2014) Review on iron and its importance for human health. J Res Med Sci 19(2):64
Al-Hakimi AMA (2007) Modification of cadmium toxicity in pea seedlings by kinetin. Plant Soil Environ 53(3):129
Alloway BJ (2008) Micronutrients and Crop Production: An Introduction. In: Micronutrient Deficiencies in Global Crop Production. Springer, Dordrecht, pp. 1–39. https://doi.org/10.1007/978-1-4020-6860-7_1
Alloway BJ (2009) Soil factors associated with zinc deficiency in crops and humans. Environ Geochem Health 31:537–548
Andersson MS, Saltzman A, Virk PS, Pfeiffer WH (2017) Progress update: crop development of biofortified staple food crops under HarvestPlus. Afr J Food Agric Nutr Dev 17(2):11905–11935
Aoyama T, Kobayashi T, Takahashi M, Nagasaka S, Usuda K, Kakei Y, Ishimaru Y, Nakanishi H, Mori S, Nishizawa NK (2009) OsYSL18 is a rice iron (III)–deoxymugineic acid transporter specifically expressed in reproductive organs and phloem of lamina joints. Plant Mol Biol 70:681–692
Asea PEA, Kucey RMN, Stewart JWB (1988) Inorganic phosphate solubilization by two Penicillium species in solution culture and soil. Soil Biol Biochem 20(4):459–464. https://doi.org/10.1016/0038-0717(88)90058-2
Axelsen KB, Palmgren MG (2001) Inventory of the super family of P-type ion pumps in Arabidopsis. Plant Physiol 126:696–706. https://doi.org/10.1104/pp.126.2.696
Balakrishnan N, Subramanian KS (2012) Mycorrhizal symbiosis and bioavailability of micronutrients in maize grain. Maydica 57(2):129–138
Batista RO, Furtini Neto AE, Deccetti SFC, Viana CS (2016) Root morphology and nutrient uptake kinetics by Australian cedar clones. Revista Caatinga 29(1):153–162. https://doi.org/10.1590/1983-21252016v29n118rc
Bereczky Z, Wang HY, Schubert V, Ganal M, Bauer P (2003) Differential regulation of NRAMP and IRT metal transporter genes in wild type and iron uptake mutants of tomato. J Biol Chem 278:24697–24704. https://doi.org/10.1074/jbc.M301365200
Bernard DG, Cheng YF, Zhao YD, Balk J (2009) An allelic mutant series of ATM3 reveals its key role in the biogenesis of cytosolic iron-sulfur proteins in Arabidopsis. Plant Physiol 151:590–602. https://doi.org/10.1104/pp.109.143651
Blum U, Staman KL, Flint LJ, Shafer SR (2000) Induction and/or selection of phenolic acid-utilizing bulk-soil and rhizosphere bacteria and their influence on phenolic acid phytotoxicity. J Chem Ecol 26(9):2059–2078
Bonaventuraa P, Benedettia G, Albarèdeb F, Miossec P (2015) Zinc and its role in immunity and inflammation. Autoimmun Rev 14:277–285. https://doi.org/10.1016/j.autrev.2014.11.008
Brown PH, Cakmak I, Zhang Q (1993) Form and function of zinc plants. In: Zinc in soils and plants. Springer, Dordrecht, pp 93–106. https://doi.org/10.1007/978-94-011-0878
Cakmak I (2008) Enrichment of cereal grains with zinc: agronomic or genetic biofortification? Plant Soil 302(1–2):1–17
Cakmak I, Pfeiffer WH, McClafferty B (2010) Review: Biofortification of durum wheat with zinc and iron. Cereal Chem 87(1):10–20. https://doi.org/10.1094/CCHEM-87-1-0010
Cao G, Sofic E, Prior RL (1997) Antioxidant and prooxidant behavior of flavonoids: structure-activity relationships. Free Radic Biol Med 22(5):749–760. https://doi.org/10.1016/S0891-5849(96)00351-6
Chattopadhyay T, Sahoo AK, Singh RS, Shyampura RL (1996) Available micronutrient status in the soils of Vindhyan scarplands of Rajasthan in relation to soil characteristics. J Indian Soc Soil Sci 44(4):678–681
Chatzistathis T, Therios I, Alifragis D (2009) Differential uptake, distribution within tissues and use efficiency of manganese, iron and zinc by olive cultivars kothreiki and koroneiki. Hortic Sci 44(7):1994–1999. https://doi.org/10.21273/HORTSCI.44.7.1994
Chellan R, Paul L (2010) Prevalence of iron-deficiency anaemia in India: results from a large nationwide survey. J Popul Soc Stud 19(1):59–80
Chen B, Shen J, Zhang X, Pan F, Yang X, Feng Y (2014a) The endophytic bacterium, Sphingomonas SaMR12, improves the potential for zinc phytoremediation by its host, Sedum alfredii. PLoS One 9(9):e106826. https://doi.org/10.1371/journal.pone.0106826
Chen B, Zhang Y, Rafiq MT, Khan KY, Pan F, Yang X, Feng Y (2014b) Improvement of cadmium uptake and accumulation in Sedum alfredii by endophytic bacteria Sphingomonas SaMR12: effects on plant growth and root exudates. Chemosphere 117:367–373. https://doi.org/10.1016/j.chemosphere.2014.07.078
Chen S, Sanchez-Fernandez R, Lyver E, Dancis A, Rea P (2007) Functional characterization of AtATM1, AtATM2 and AtATM3, a subfamily of Arabidopsis half-molecule ATP-binding cassette transporters implicated in iron homeostasis. J Biol Chem 282:21561–21571. https://doi.org/10.1074/jbc.M702383200
Chen WR, He ZL, Yang XE, Feng Y (2009) Zinc efficiency is correlated with root morphology, ultrastructure, and antioxidative enzymes in rice. J Plant Nutr 32(2):287–305. https://doi.org/10.1080/01904160802608627
Chen WW, Yang JL, Qin C, Jin CW, Mo JH, Ye T, Zheng SJ (2010) Nitric oxide acts downstream of auxin to trigger root ferric-chelate reductase activity in response to iron deficiency in Arabidopsis. Plant Physiol 154(2):810–819. https://doi.org/10.1104/pp.110.161109
Chiang PN, Chiu CY, Wang MK, Chen BT (2011) Low-molecular-weight organic acids exuded by millet (Setaria italica (L.) Beauv.) roots and their effect on the remediation of cadmium-contaminated soil. Soil Sci 176(1):33–38. https://doi.org/10.1097/SS.0b013e318202fdc910.1097/SS.0b013e318202fdc9
Chirwa M, Yerokun OA (2012) The distribution of zinc fractions in surface samples of selected agricultural soils of Zambia. Int J Soil Sci 7:51–60. https://doi.org/10.3923/ijss.2012.51.60
Chu HH, Chiecko J, Punshon T, Lanzirotti A, Lahner B, Salt DE, Walker EL (2010) Successful reproduction requires the function of Arabidopsis Yellow Stripe-Like1 and Yellow Stripe Like 3 metal–nicotianamine transporters in both vegetative and reproductive structures. Plant Physiol 154:197–210. https://doi.org/10.1104/pp.110.159103
Colangelo EP, Guerinot ML (2006) Put the metal to the petal: metal uptake and transport throughout plants. Curr Opin Plant Biol 9(3):322–330. https://doi.org/10.1016/j.pbi.2006.03.015
Connorton JM, Jones ER, Rodríguez-Ramiro I, Fairweather-Tait S, Uauy C, Balk J (2017) Wheat vacuolar iron transporter TaVIT2 transports Fe and Mn and is effective for biofortification. Plant Physiol 174(4):2434–2444. https://doi.org/10.1104/pp.17.00672
Corvol P, Eyries M, Soubrier F (2004) Peptidyl-dipeptidase A/angiotensin I-converting enzyme. In: Handbook of proteolytic enzymes Barret AA, Rawlings ND, Woessner JF, Elsevier Academic, pp.332–349. https://doi.org/10.1016/B978-0-12-079611-3.50090-2
Cummings JE, Kovacic JP (2009) The ubiquitous role of zinc in health and disease. J Vet Emerg Crit Care 19(3):215–240. https://doi.org/10.1111/j.1476-4431.2009.00418.x
Curie C, Alonso JM, Le Jean M, Ecker JR, Briat JF (2000) Involvement of NRAMP1 from Arabidopsis thaliana in iron transport. Biochem J 347:749–755. https://doi.org/10.1042/bj3470749
Curie C, Briat JF (2003) Iron transport and signaling in plants. Annu Rev Plant Biol 54(1):183–206. https://doi.org/10.1146/annurev.arplant.54.031902.135018
Curie C, Panaviene Z, Loulergue C, Dellaporta SL, Briat JF, Walker EL (2001) Maize yellow stripe1 encodes a membrane protein directly involved in Fe(III) uptake. Nature 409:346–349
da Silva LL, Olivares FL, De Oliveira RR, Vega MRG, Aguiar NO, Canellas LP (2014) Root exudate profiling of maize seedlings inoculated with Herbaspirillum seropedicae and humic acids. Chem Biol Tech Agric 1(1):1. https://doi.org/10.1186/s40538-014-0023
Dakora FD, Phillips DA (2002) Root exudates as mediators of mineral acquisition in low-nutrient environments. In: Food security in nutrient-stressed environments: exploiting plants’ genetic capabilities. Springer, Dordrecht, pp 201–213. https://doi.org/10.1007/978-94-017-1570-6_23
de Santiago A, Quintero JM, Avilés M, Delgado A (2011) Effect of Trichoderma asperellum strain T34 on iron, copper, manganese, and zinc uptake by wheat grown on a calcareous medium. Plant Soil 342(1–2):97–104. https://doi.org/10.1007/s11104-010-0670-1
Delaplace P, Delory BM, Baudson C, de Cazenave MMS, Spaepen S, Varin S, Brostaux Y, du Jardin P (2015) Influence of rhizobacterial volatiles on the root system architecture and the production and allocation of biomass in the model grass Brachypodium distachyon (L.) P. Beauv. BMC Plant Biol 15(1):195. https://doi.org/10.1186/s12870-015-0585-3
Desai A, Archana G (2011) Role of siderophores in crop improvement. Bacteria in agrobiology: plant nutrient management. Springer, pp. 109–139. https://doi.org/10.1007/978-3-642-21061-7_6
Desbrosses-Fonrouge AG, Voigt K, Schroder A, Arrivault S, Thomine S, Kramer U (2005) Arabidopsis thaliana MTP1 is a Zn transporter in the vacuolar membrane which mediates Zn detoxification and drives leaf Zn accumulation. FEBS Lett 579:4165–4174. https://doi.org/10.1016/j.febslet.2005.06.046
Dharejo KA, Anuar AR, Khanif YM, Samsuri AW, Junejo N (2011) Spatial variability of Cu, Mn and Zn in marginal sandy beach ridges soil. Afr J Agric Res 6(15):3493–3498. https://doi.org/10.5897/AJAR11.753
Di Donato RJ, Roberts LA, Sanderson T, Eisley RB, Walker EL (2004) Arabidopsis Yellow Stripe-Like2 (YSL2): a metal-regulated gene encoding a plasma membrane transporter of nicotianamine–metal complexes. Plant J 39(3):403–414. https://doi.org/10.1111/j.1365-313X.2004.02128.x
Di Tomaso JM (1995) Approaches for improving crop competitiveness through the manipulation of fertilization strategies. Weed Sci 43:491–497. https://doi.org/10.1017/S0043174500081522
Dong B, Rengel Z, Graham RD (1995) Root morphology of wheat genotypes differing in zinc efficiency. J Plant Nutr 18(12):2761–2773. https://doi.org/10.1080/01904169509365098
Durmaz E, Coruh C, Dinler G, Grusak MA, Peleg Z, Saranga Y, Fahima T, Yazici A, Ozturk L, Cakmak I (2011) Expression and cellular localization of ZIP1 transporter under zinc deficiency in wild emmer wheat. Plant Mol Biol Report 29(3):582–596
Eide DJ (2006) Zinc transporters and the cellular trafficking of zinc. Biochim Biophys Acta, Mol Cell Res 1763(7):711–722. https://doi.org/10.1016/j.bbamcr.2006.03.005
Elkoca E, Turan M, Donmez MF (2010) Effects of single, dual and triple inoculation with Bacillus subtilis, Bacillus megaterium and Rhizobium leguminosarum bv. phaseoil on nodulation, nutrient uptake, yield and yield parameters of common bean (Phaseolus vulgaris L. cv. ‘Elkoca-05’). J Plant Nutr 33:2104–2119. https://doi.org/10.1080/01904167.2010.519084
Evens NP, Buchner P, Williams LE, Hawkesford MJ (2017) The role of ZIP transporters and group F bZIP transcription factors in the Zn-deficiency response of wheat (Triticum aestivum). Plant J 92(2):291–304. https://doi.org/10.1111/tpj.13655
Fageria NK, Baligar VC, Clark RB (2002) Micronutrients in crop production. Adv Agron 77:185–250. https://doi.org/10.1016/S0065-2113(02)77015-6
Foley JA, DeFries R, Asner GP, Barford C, Bonan G, Carpenter SR, Helkowski JH (2005) Global consequences of land use. Science 309(5734):570–574
Follett RH, Westfall DG (1992) Identifying and correcting zinc and iron deficiency in field crops. Colorado State University Cooperative Extension: service in action. No. 545. http://cospl.coalliance.org/fez/eserv/co:6978. Accessed Dec 2017
Food and Nutrition Board (2001) Dietary reference intakes for vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium and zinc. Institute of Medicine, National Academies, National Academies Press, Washington
Forouzanfar MH, Ashkan A, Lily TA, Anderson HR, Zulfiqar AB, Stan B, Michael B et al (2016) Global, regional, and national comparative risk assessment of 79 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet 388(10053):1659–1724. https://doi.org/10.1016/S0140-6736(17)32154-2
Frossard E, Bucher M, Mächler FMA, Hurrell R (2000) Potential for increasing the content and bioavailability of Fe, Zn and Ca in plants for human nutrition. J Sci Food Agric 80:861–879. https://doi.org/10.1002/(SICI)1097-0010(20000515)80:7%3C861::AID-SFA601%3E3.0.CO;2-P
Gangwar S, Singh VP, Prasad SM, Maurya JN (2010) Modulation of manganese toxicity in Pisum sativum L. seedlings by kinetin. Sci Hortic 126(4):467–474. https://doi.org/10.1016/j.scienta.2010.08.013
Ganz T (2018) Iron and infection. Int J Hematol 107(1):7–15. https://doi.org/10.1007/s12185-017-2366-2
Gao X, Flaten DN, Tenuta M, Grimmett MG, Gawalko EJ, Grant CA (2011) Soil solution dynamics and plant uptake of cadmium and zinc by durum wheat following phosphate fertilization. Plant Soil 338(1–2):423–434. https://doi.org/10.1007/s11104-010-0556-2
Garcia C, Maria N, Juan PPR, Helena P, Luz MDR, Elizabeth CT, Monica CFU (2016) Staple crops biofortified with increased micronutrient content: effects on vitamin and mineral status, as well as health and cognitive function in the general population. Cochrane Database Syst Rev 8. https://doi.org/10.1002/14651858.CD012311
García MJ, Suárez V, Romera FJ, Alcántara E, Pérez-Vicente R (2011) A new model involving ethylene, nitric oxide and Fe to explain the regulation of Fe-acquisition genes in strategy I plants. Plant Physiol Biochem 49(5):537–544. https://doi.org/10.1016/j.plaphy.2011.01.019
Genc Y, Huang CY, Langridge P (2007) A study of the role of root morphological traits in growth of barley in zinc-deficient soil. J Exp Bot 58(11):2775–2784. https://doi.org/10.1093/jxb/erm142
Genc Y, McDonald GK, Graham RD (2006) Contribution of different mechanisms to zinc efficiency in bread wheat during early vegetative stage. Plant Soil 281(1–2):353–367
Gliessman SR (2014) Agroecology: the ecology of sustainable food systems. CRC
Gosal S, Karlupia A, Gosal S, Chhibba I, Varma A (2010) Biotization with Piriformospora indica and Pseudomonas fluorescens improves survival rate, nutrient acquisition, field performance and saponin content of micropropagated Chlorophytum sp. Indian J Biotechnol 9:289–297
Goswami D, Dhandhukia P, Patel P, Thakker JN (2014) Screening of PGPR from saline desert of Kutch: growth promotion in Arachis hypogea by Bacillus licheniformis A2. Microbiol Res 169(1):66–75. https://doi.org/10.1016/j.micres.2013.07.004
Goto F, Yoshihara T, Shigemoto N, Toki S, Takaiwa F (1999) Iron fortification of rice seed by the soybean ferritin gene. Nat Biotechnol 17:282–286
Govindaraj M, Rai KN, Shanmugasundaram P (2016) Intra-population genetic variance for grain iron and zinc contents and agronomic traits in pearl millet. Crop J 4(1):48–54. https://doi.org/10.1016/j.cj.2015.11.002
Graziano M, Lamattina L (2007) Nitric oxide accumulation is required for molecular and physiological responses to iron deficiency in tomato roots. Plant J 52(5):949–960. https://doi.org/10.1111/j.1365-313X.2007.03283.x
Grotz N, Fox T, Connolly E, Park W, Guerinot ML, Eide D (1998) Identification of a family of zinc transporter genes from Arabidopsis that respond to zinc deficiency. Proc Natl Acad Sci U S A 95(12):7220–7224. https://doi.org/10.1073/pnas.95.12.7220
Grusak MA (2002) Enhancing mineral content in plant food products. J Am Coll Nutr 21(sup3):178S–183S. https://doi.org/10.1080/07315724.2002.10719263
Guerinot ML, Yi Y (1994) Iron: nutritious, noxious, and not readily available. Plant Physiol 104(3):815. https://doi.org/10.1104/pp.104.3.815
Hänsch R, Mendel RR (2009) Physiological functions of mineral micronutrients (Cu, Zn, Mn, Fe, Ni, Mo, B, Cl). Curr Opin Plant Biol 12(3):259–266. https://doi.org/10.1016/j.pbi.2009.05.006
Hansen NC, Jolley VD, Berg WA, Hodges ME, Krenzer EG (1996) Phytosiderophore release related to susceptibility of wheat to iron deficiency. Crop Sci 36(6):1473–1476. https://doi.org/10.2135/cropsci1996.0011183X003600060009x
Harter RD (1983) Effect of soil pH on adsorption of lead, copper, zinc, and nickel. Soil Sci Soc Am J 47:47–51. https://doi.org/10.2136/sssaj1983.03615995004700010009x
Havlin JL, Beaton JD, Tisdale SL, Nelson WL (2007) Soil fertility and fertilizers: an introduction to nutrient management. Prentice Hall, Upper Saddle River, New Jersey, pp 244–289
Hayat R, Ahmed I, Sheirdil RA (2012) An overview of plant growth promoting rhizobacteria (PGPR) for sustainable agriculture. Crop production for agricultural improvement. Springer, 557–579. https://doi.org/10.1007/978-94-007-4116-4_22
Hentze MW, Muckenthaler MU, Andrews NC (2004) Balancing acts: molecular control of mammalian iron metabolism. Cell 117(3):285–297. https://doi.org/10.1016/S0092-8674(04)00343-5
Hindt MN, Guerinot ML (2012) Getting a sense for signals: regulation of the plant iron deficiency response. Biochim Biophys Acta (BBA)-Mol Cell Res 1823(9):1521–1530. https://doi.org/10.1016/j.bbamcr.2012.03.010
Hindu V, Palacios-Rojas N, Babu R, Suwarno WB, Rashid Z, Usha R, Saykhedkar GR, Nair SK (2018) Identification and validation of genomic regions influencing kernel zinc and iron in maize. Theor Appl Genet 131:1443–1457. https://doi.org/10.1007/s00122-018-3089-3
Hunt JR (2003) Bioavailability of iron, zinc, and other trace minerals from vegetarian diets. Am J Clin Nutr 78(3):633S–639S. https://doi.org/10.1093/ajcn/78.3.633S
Hurst R, Siyame EW, Young SD, Chilimba AD, Joy EJ, Black CR, Ander EL, Watts MJ, Chilima B, Gondwe J, Kang’ombe D (2013) Soil-type influences human selenium status and underlies widespread selenium deficiency risks in Malawi. Sci Rep 3:1425
Ibrahim AK, Usman A, Abubakar B, Aminu UH (2011) Extractable micronutrients status in relation to other soil properties in Billiri local government area. J Soil Sci Environ Mgt 3(10):282–285
Inoue H, Kobayashi T, Nozoye T, Takahashi M, Kakei Y, Suzuki K, Nakazono M, Nakanishi H, Mori S, Nishizawa NK (2009) Rice OsYSL15 is an iron-regulated iron(III)-deoxymugineic acid transporter expressed in the roots and is essential for iron uptake in early growth of the seedlings. J Biol Chem 284:3470–3479. https://doi.org/10.1074/jbc.M806042200
International Institute for Population Sciences and ORC Macro. MEASURE/DHS+ (Programme), (2000) India National Family Health Survey (NFHS-2), 1998–99 (Vol. 1). International Institute for Population Sciences, Mumbai, India
Ishimaru Y, Masuda H, Bashir K, Inoue H, Tsukamoto T, Takahashi M, Nakanishi H, Aoki N, Hirose T, Ohsugi R, Nishizawa NK (2010) Rice metal–nicotianamine transporter, OsYSL2, is required for the long-distance transport of iron and manganese. Plant J 62:379–390. https://doi.org/10.1111/j.1365-313X.2010.04158.x
Ishimaru Y, Suzuki M, Kobayashi T, Takahashi M, Nakanishi H, Mori S, Nishizawa NK (2005) OsZIP4, a novel zinc-regulated zinc transporter in rice. J Exp Bot 56(422):3207–3214. https://doi.org/10.1093/jxb/eri317
Ishimaru Y, Suzuki M, Tsukamoto T, Suzuki K, Nakazono M, Kobayashi T, Wada Y, Watanabe S, Matsuhashi S, Takahashi M, Nakanishi H (2006) Rice plants take up iron as an Fe3+-phytosiderophore and as Fe2+. Plant J 45(3):335–346. https://doi.org/10.1111/j.1365-313X.2005.02624.x
Ishimaru Y, Takahashi R, Bashir K, Shimo H, Senoura T, Sugimoto K, Ono K, Yano M, Ishikawa S, Arao T, Nakanishi H, Nishizawa NK (2012) Characterizing the role of rice NRAMP5 in manganese, iron and cadmium transport. Sci Rep 2:286
Ivanov R, Bauer P (2017) Sequence and co-expression analysis of iron-regulated ZIP transporter genes reveals crossing points between iron acquisition strategies in green algae and land plants. Plant Soil 418(1–2):61–73. https://doi.org/10.1007/s11104-016-3128-2
Ivanov R, Brumbarova T, Bauer P (2012) Fitting into the harsh reality: regulation of iron-deficiency responses in dicotyledonous plants. Mol Plant 5(1):27–42. https://doi.org/10.1093/mp/ssr065
Jin CW, He YF, Tang CX, Wu P, Zheng SJ (2006) Mechanisms of microbially enhanced iron uptake in red clover. Plant Cell Environ 29:888–897. https://doi.org/10.1111/j.1365-3040.2005.01468.x
Jin CW, You GY, He YF, Tang C, Wu P, Zheng SJ (2007) Iron deficiency-induced secretion of phenolics facilitates the reutilization of root apoplastic iron in red clover. Plant Physiol 144(1):278–285. https://doi.org/10.1104/pp.107.095794
Kerkeb L, Connoly E (2006) Iron transport and metabolism in plants. Genet Eng 27:119–140
Khalid S, Asghar HN, Akhtar MJ, Aslam A, Zahir ZA (2015) Biofortification of iron in chickpea by plant growth-promoting rhizobacteria. Pak J Bot 47:1191–1194
Khan A, Singh J, Upadhayay VK, Singh AV, Shah S (2019) Microbial biofortification: a green technology through plant growth promoting microorganisms. In: Sustainable green technologies for environmental management. Springer, Singapore, pp 255–269
Kim J, Rees DC (1992) Structural models for the metal centers in the nitrogenase molybdenum-iron protein. Science 257(5077):1677–1682. https://doi.org/10.1126/science.1529354
Kim S, Lim H, Lee I (2010) Enhanced heavy metal phytoextraction by Echinochloa crus-galli using root exudates. J Biosci Bioeng 109(1):47–50. https://doi.org/10.1016/j.jbiosc.2009.06.018
Kobae Y, Uemura T, Sato MH, Ohnishi M, Mimura T, Nakagawa T, Maeshima M (2004) Zinc transporter of Arabidopsis thaliana AtMTP1 is localized to vacuolar membranes and implicated in zinc homeostasis. Plant Cell Physiol 45:1749–1758. https://doi.org/10.1093/pcp/pci015
Kobayashi T, Nishizawa NK (2012) Iron uptake, translocation, and regulation in higher plants. Annu Rev Plant Biol 63:131–152. https://doi.org/10.1146/annurev-arplant-042811-105522
Kobayashi T, Yuko N, Reiko NI, Hiromi N, Toshihiro Y, Satoshi M, Naoko KN (2003) Identification of novel cis-acting elements, IDE1 and IDE2, of the barley IDS2 gene promoter conferring iron-deficiency-inducible, root-specific expression in heterogeneous tobacco plants. Plant J 36(6):780–793. https://doi.org/10.1046/j.1365-313X.2003.01920.x
Koike S, Inoue H, Mizuno D, Takahashi M, Nakanishi H, Mori S, Nishizawa NK (2004) OsYSL2 is a rice metal nicotianamine transporter that is regulated by iron and expressed in the phloem. Plant J 39:415–424. https://doi.org/10.1111/j.1365-313X.2004.02146.x
Kothari SK, Marschner H, Romheld V (1990) Direct and indirect effects of vam mycorrhizal fungi and rhizosphere microorganisms on acquisition of mineral nutrients by maize (Zea mays) in a calcareous soil. New Phytol 116:637–645. https://doi.org/10.1111/j.1469-8137.1990.tb00549.x
Kotula L, Ranathunge K, Steudle E (2009) Apoplastic barriers effectively block oxygen permeability across outer cell layers of rice roots under deoxygenated conditions: roles of apoplastic pores and of respiration. New Phytol 184(4):909–917. https://doi.org/10.1111/j.1469-8137.2009.03021.x
Kramer D, Römheld V, Landsberg E, Marschner H (1980) Induction of transfer-cell formation by iron deficiency in the root epidermis of Helianthus annuus L. Planta 147(4):335–339. https://doi.org/10.1007/BF00379842
Krämer U, Talke IN, Hanikenne M (2007) Transition metal transport. FEBS Lett 581(12):2263–2272. https://doi.org/10.1016/j.febslet.2007.04.010
Krithika S, Balachandar D (2016) Expression of zinc transporter genes in rice as influenced by zinc-solubilizing Enterobacter cloacae strain ZSB14. Front Plant Sci 7:446. https://doi.org/10.3389/fpls.2016.00446
Kucey RMN (1988) Effect of Penicillium bilaji on the solubility and uptake of P and micronutrients from soil by wheat. Can J Soil Sci 68:261–270. https://doi.org/10.4141/cjss88-026
Kumar S, Hash C, Nepolean T, Mahendrakar M, Satyavathi C, Singh G, Rathore A, Yadav R, Gupta R, Srivastava R (2018) Mapping grain iron and zinc content quantitative trait loci in an Iniadi-derived immortal population of pearl millet. Genes 9(5):248. https://doi.org/10.3390/genes9050248
Kumssa DB, Joy EJ, Ander EL, Watts MJ, Young SD, Walker S, Broadley MR (2015) Dietary calcium and zinc deficiency risks are decreasing but remain prevalent. Sci Rep 5:10974
Lanquar V, Lelievre F, Bolte S, Hames C, Alcon C, Neumann D, Vansuyt G, Curie C, Schroder A, Kramer U, Brygoo HB, Thomine S (2005) Mobilization of vacuolar iron by AtNRAMP3 and AtNRAMP4 is essential for seed germination on low iron. EMBO J 24:4041–4051. https://doi.org/10.1038/sj.emboj.7600864
Lanquar V, Ramos MS, Lelievre F, Barbier-Brygoo H, Krieger-Liszkay A, Kramer U, Thomine S (2010) Export of vacuolar manganese by AtNRAMP3 and AtNRAMP4 is required for optimal photosynthesis and growth under manganese deficiency. Plant Physiol 152:1986–1999. https://doi.org/10.1104/pp.109.150946
Li P, Qi JL, Wang L, Huang QN, Han ZH, Yin LP (2006) Functional expression of MxIRT1, from Malus xiaojinensis, complements an iron uptake deficient yeast mutant for plasma membrane targeting via membrane vesicles trafficking process. Plant Sci 171:52–59. https://doi.org/10.1016/j.plantsci.2006.02.012
Li S, Zhou X, Huang Y, Zhu L, Zhang S, Zhao Y, Guo J, Chen J, Chen R (2013) Identification and characterization of the zinc-regulated transporters, iron-regulated transporter-like protein (ZIP) gene family in maize. BMC Plant Biol 13(1):114. https://doi.org/10.1186/1471-2229-13-114
Liang J, Han BZ, Nout MJR, Hamer RJ (2008) Effect of soaking, germination and fermentation on phytic acid, total and in vitro soluble zinc brown rice. Food Chem 110(4):821–828. https://doi.org/10.1016/j.foodchem.2008.02.064
Lin YF, Liang HM, Yang SY, Boch A, Clemens S, Chen CC, Wu JF, Huang JL, Yeh KC (2009) Arabidopsis IRT3 is a zinc-regulated and plasma membrane localized zinc/iron transporter. New Phytol 182:392–404. https://doi.org/10.1111/j.1469-8137.2009.02766.x
Lindsay WL (1984) Soil and plant relationships associated with iron deficiency with emphasis on nutrient interactions. J Plant Nutr 7(1–5):489–500. https://doi.org/10.1080/01904168409363215
Lindsay WL, Schwab AP (1982) The chemistry of iron in soils and its availability to plants. J Plant Nutr 5:821–840. https://doi.org/10.1080/01904168209363012
Liu QQ, Yao QH, Wang HM, Gu MH (2004) Endosperm-specific expression of the ferritin gene in transgenic rice (Oryza sativa L.) results in increased iron content of milling rice. Yi Chuan Xue Bao 31:518–524
Lockhart K, King A, Harter T (2013) Identifying sources of groundwater nitrate contamination in a large alluvial groundwater basin with highly diversified intensive agricultural production. J Contam Hydrol 151:140–154. https://doi.org/10.1016/j.jconhyd.2013.05.008
López-Bucio J, Campos-Cuevas JC, Hernández-Calderón E, Velásquez-Becerra C, Farías-Rodríguez R, Macías-Rodríguez LI, Valencia-Cantero E (2007) Bacillus megaterium rhizobacteria promote growth and alter root-system architecture through an auxin- and ethylene-independent signaling mechanism in Arabidopsis thaliana. Mol Plant-Microbe Interact 20(2):207–217. https://doi.org/10.1094/MPMI-20-2-0207
Lucca P, Hurrel R, Potrykus I (2002) Fighting iron deficiency anemia with iron-rich rice. J Am Coll Nutr 3:184S–190S. https://doi.org/10.1080/07315724.2002.10719264
Lucena C, Waters BM, Romera FJ, García MJ, Morales M, Alcántara E, Pérez-Vicente R (2006) Ethylene could influence ferric reductase, iron transporter, and H+-ATPase gene expression by affecting FER (or FER-like) gene activity. J Exp Bot 57(15):4145–4154. https://doi.org/10.1093/jxb/erl189
Luo Q, Sun L, Hu X, Zhou R (2014) The variation of root exudates from the hyperaccumulator Sedum alfredii under cadmium stress: metabonomics analysis. PLoS One 9(12):e115581. https://doi.org/10.1371/journal.pone.0115581
Lynch JP (2007) Turner review no. 14. Roots of the second green revolution. Am. J. Bot. 55(5):493–512. https://doi.org/10.1071/BT06118
Mader P, Kaiser F, Adholeya A, Singh R, Uppal HS, Sharma AK, Srivastava R, Sahai V, Aragno M, Wiemken A (2011) Inoculation of root microorganisms for sustainable wheat–rice and wheat–black gram rotations in India. Soil Biol Biochem 43(3):609–619. https://doi.org/10.1016/j.soilbio.2010.11.031
Mahender A, Swamy BP, Anandan A, Ali J (2019) Tolerance of iron-deficient and -toxic soil conditions in rice. Plants 8(2):31. https://doi.org/10.3390/plants8020031
Malinowski D, Zuo H, Belesky D, Alloush G (2004) Evidence for copper binding by extracellular root exudates of tall fescue but not perennial ryegrass infected with Neotyphodium spp. endophytes. Plant Soil 267:1–12. https://doi.org/10.1007/s11104-005-2575-y
Malinowski DP, Belesky DP (2000) Adaptations of endophyte-infected cool-season grasses to environmental stresses: mechanisms of drought and mineral stress tolerance. Crop Sci 40:923–940. https://doi.org/10.2135/cropsci2000.404923x
Manyevere A, Muchaonyerwa P, Mnkeni PN, Dhau I (2017) Spatial variability of selected soil micronutrients under smallholder crop production in Zanyokwe, Eastern Cape, South Africa. S Afr J Plant Soil 34(5):339–349. https://doi.org/10.1080/02571862.2016.1266399
Mark HE, Houghton LA, Gibson RS, Monterrosa E, Kraemer K (2016) Estimating dietary micronutrient supply and the prevalence of inadequate intakes from national food balance sheets in the South Asia region. Asia Pac J Clin Nutr 25:368–376
Masood A, Khan MIR, Fatma M, Asgher M, Per TS, Khan NA (2016) Involvement of ethylene in gibberellic acid-induced sulfur assimilation, photosynthetic responses, and alleviation of cadmium stress in mustard. Plant Physiol Biochem 104:1–10. https://doi.org/10.1016/j.plaphy.2016.03.017
Meena VS, Maurya BR, Verma JP, Aeron A, Kumar A, Kim K, Bajpai VK (2015) Potassium solubilizing rhizobacteria (KSR): isolation, identification and K-release dynamics from waste mica. Ecol Eng 81:340–347. https://doi.org/10.1016/j.ecoleng.2015.04.065
Mei Z, Cogswell ME, Parvanta I, Lynch S, Beard JL, Stoltzfus RJ, Grummer-Strawn LM (2005) Hemoglobin and ferritin are currently the most efficient indicators of population response to iron interventions: an analysis of nine randomized controlled trials. J Nutr 135(8):1974–1980. https://doi.org/10.1093/jn/135.8.1974
Milner MJ, Seamon J, Craft E, Kochian LV (2013) Transport properties of members of the ZIP family in plants and their role in Zn and Mn homeostasis. J Exp Bot 64(1):369–381. https://doi.org/10.1093/jxb/ers315
Ministry of Health and Family Welfare (2016) National Family Health Survey; 4, 2015–16. New Delhi
Mishra PK, Bisht SC, Ruwari P, Joshi GK, Singh G, Bisht JK, Bhatt JC (2011) Bioassociative effect of cold tolerant Pseudomonas spp. and Rhizobium leguminosarum-PR1 on iron acquisition, nutrient uptake and growth of lentil (Lens culinaris L.). Eur J Soil Biol 47:35–43. https://doi.org/10.1016/j.ejsobi.2010.11.005
Moreau S, Thomson RM, Kaiser BN, Trevaskis B, Guerinot ML, Udvardi MK, Puppo A, Day DA (2002) GmZIP1 encodes a symbiosis specific zinc transporter in soybean. J Biol Chem 277(7):4738–4746. https://doi.org/10.1074/jbc.M106754200
Mori S (1999) Iron acquisition by plants. Curr Opin Plant Biol 2(3):250–253. https://doi.org/10.1016/S1369-5266(99)80043-0
Morrissey J, Baxter IR, Lee J, Li L, Lahner B, Grotz N, Kaplan J, Salt DE, Guerinot ML (2009) The ferroportin metal efflux proteins function in iron and cobalt homeostasis in Arabidopsis. Plant Cell 21:3326–3338. https://doi.org/10.1105/tpc.109.069401
Mortvedt JJ (1991) Correcting iron deficiencies in annual and perennial plants. Present technologies and future prospects. Plant Soil 130:273–279
Mousavi SR, Galavi M, Rezaei M (2013) Zinc (Zn) importance for crop production—a review. Int J Agron Plant Prod 4(1):64–68
Moynahan EJ (1974) Acrodermatitis enteropathica: a lethal inherited human zinc-deficiency disorder. Lancet 304(7877):399–400. https://doi.org/10.1016/s0140-6736(74)91772-3
Nilson A, Piza J (1998) Food fortification: a tool for fighting hidden hunger. Food Nutr Bull 19(1):49–60. https://doi.org/10.1177/156482659801900109
Obrador A, Novillo J, Alvarez JM (2003) Mobility and availability to plants of two zinc sources applied to a calcareous soil. Soil Science Soc Am J 67:564–572. https://doi.org/10.1177/156482659801900109
Ortíz-Castro R, Valencia-Cantero E, López-Bucio J (2008) Plant growth promotion by Bacillus megaterium involves cytokinin signaling. Plant Signal Behav 3(4):263–265. https://doi.org/10.4161/psb.3.4.5204
Parkin G (2004) Zinc-zinc bonds: a new frontier. Science 305:1117–1118. https://doi.org/10.1126/science.1102500
Pérez-Esteban J, Escolástico C, Moliner A, Masaguer A (2013) Chemical speciation and mobilization of copper and zinc in naturally contaminated mine soils with citric and tartaric acids. Chemosphere 90(2):276–283. https://doi.org/10.1016/j.chemosphere.2012.06.065
Petry N, Fabian R, Jean BG, Bruno C, Erick B, Pierrot LT, Michael BZ, Christian Z, James PW, Diego M (2016) In Rwandese women with low iron status, iron absorption from low-phytic acid beans and biofortified beans is comparable, but low-phytic acid beans cause adverse gastrointestinal symptoms. J Nutr 146(5):970–975. https://doi.org/10.3945/jn.115.223693
Pii Y, Mimmo T, Tomasi N, Terzano R, Cesco S, Crecchio C (2015) Microbial interactions in the rhizosphere: beneficial influences of plant growth-promoting rhizobacteria on nutrient acquisition process. A review. Biol Fertil Soils 51(4):403–415. https://doi.org/10.3945/jn.115.223693
Prasad R, Shivay YS, Kumar D (2013) Zinc fertilization of cereals for increased production and alleviation of zinc malnutrition in India. Agric Res 2:111. https://doi.org/10.1007/s40003-013-0064-8
Prasanna R, Bidyarani N, Babu S, Hossain F, Shivay YS, Nain L (2015) Cyanobacterial inoculation elicits plant defence response and enhanced Zn mobilization in maize hybrids. Cogent Food Agric 1(1):998507. https://doi.org/10.1080/23311932.2014.998507
Prasanna R, Nain L, Rana A, Shivay YS (2016) Biofortification of crop plants using microorganisms: present status and challenges. In: Singh U et al (eds) Biofortification of food crops. Springer, New Delhi, pp 249–262. https://doi.org/10.1007/978-81-322-2716-8_19
Rai KN, Patil HT, Yadav OP, Govindaraj M, Khairwal IS, Cherian B, Rajpurohit BS, Rao AS, Shivade H, Kulkarni MP (2014) Notification of crop varieties and registration of germplasm: pearl millet variety ‘Dhanashakti’. Indian J Genet Plant Breed 74(3):405–406
Raja P, Uma S, Gopal H, Govindarajan K (2006) Impact of bio inoculants consortium on rice root exudates, biological nitrogen fixation and plant growth. J Biol Sci 6:815–823
Ramesh A, Sharma SK, Sharma MP, Yadav N, Joshi OP (2014) Inoculation of zinc solubilizing Bacillus aryabhattai strains for improved growth, mobilization and biofortification of zinc in soybean and wheat cultivated in Vertisols of central India. Appl Soil Ecol 73:87–96. https://doi.org/10.1016/j.apsoil.2013.08.009
Ramesh SA, Choimes S, Schachtman DP (2004) Over-expression of an Arabidopsis zinc transporter in Hordeum vulgare increases short term zinc uptake after zinc deprivation and seed zinc content. Plant Mol Biol 54:373–385. https://doi.org/10.1023/B:PLAN.0000036370.70912.34
Ramesh SA, Shin R, Eide DJ, Schachtman DP (2003) Differential metal selectivity and gene expression of two zinc transporters from rice. Plant Physiol 133:126–134. https://doi.org/10.1104/pp.103.026815
Ramzan S, Bhat MA, Kirmani NA, Rasool R (2014) Fractionation of zinc and their association with soil properties in soils of Kashmir Himalayas. Ind J Agric Soil Sci 2:132–142
Rana A, Joshi M, Prasanna R, Shivay YS, Nain L (2012) Biofortification of wheat through inoculation of plant growth promoting rhizobacteria and cyanobacteria. Eur J Soil Biol 50:118–126. https://doi.org/10.1016/j.ejsobi.2012.01.005
Rana A, Kabi SR, Verma S, Adak A, Pal M, Shivay YS, Prasanna R, Nain L (2015) Prospecting plant growth promoting bacteria and cyanobacteria as options for enrichment of macro and micronutrients in grains in rice - wheat cropping sequence. Cogent Food and Agric 1:1037379. https://doi.org/10.1080/23311932.2015.1037379
Rêgo MCF, Ilkiu-Borges F, de Filippi MCC, Gonçalves LA, da Silva GB (2014) Morphoanatomical and biochemical changes in the roots of rice plants induced by plant growth-promoting microorganisms. Aust J Bot 10:818797. https://doi.org/10.1155/2014/818797
Reiter B, Pfeifer U, Schwab H, Sessitsch A (2002) Response of endophytic bacterial communities in potato plants to infection with Erwinia carotovora subsp. atroseptica. Appl Environ Microbiol 68:2261–2268. https://doi.org/10.1128/AEM.68.5.2261-2268.2002
Rengel Z (2001) Genotypic differences in micronutrient use efficiency in crops. Commun Soil Sci Plant Anal 32(7–8):1163–1186. https://doi.org/10.1081/CSS-100104107
Rengel Z, Batten GD, Crowley DE (1999) Agronomic approaches for improving the micronutrient density in edible portions of field crops. Field Crop Res 60:27–40. https://doi.org/10.1016/S0378-4290(98)00131-2
Rengel Z, Wheal MS (1997) Kinetic parameters of Zn uptake by wheat are affected by the herbicide chlorsulfuron. J Exp Bot 48(4):935–941. https://doi.org/10.1093/jxb/48.4.935
Reza SK, Nayak DC, Mukhopadhyay S, Chattopadhyay T, Singh SK (2017) Characterizing spatial variability of soil properties in alluvial soils of India using geostatistics and geographical information system. Arch Agron Soil Sci 63(11):1489–1498. https://doi.org/10.1080/03650340.2017.1296134
Rice-Evans CA, Miller NJ (1996) Antioxidant activities of flavonoids as bioactive components of food. Biochem Soc Trans 24(3):790–795. https://doi.org/10.1042/bst0240790
Romera FJ, García MJ, Lucena C, Martinez Medina A, Aparicio MÁ, Ramos J, Alcántara E, Angulo M, Pérez-Vicente R (2019) Induced systemic resistance (ISR) and Fe deficiency responses in dicot plants. Front Plant Sci 10:287. https://doi.org/10.3389/fpls.2019.00287
Römheld V, Marschner H (1986) Evidence for a specific uptake system for iron phytosiderophores in roots of grasses. Plant Physiol 80(1):175–180. https://doi.org/10.1104/pp.80.1.175
Romheld V, Marschner H (1991) Function of micronutrients in plants. In: Micronutrients in Agriculture (Mortvedt JJ, Cox FR, Shuman LM, Welch RM, Eds), 2nd ed. Soil Science Society of America, Madison, WI. pp. 297–328
Ross SM, Nel E, Naeye R (1985) Differing low and high bulk maternal dietary supplements during pregnancy. Early Hum Dev:295–302. https://doi.org/10.1016/0378-3782(85)90061-1
Rout GR, Sahoo S (2015) Role of iron in plant growth and metabolism. Rev Agric Sci 3:1–24. https://doi.org/10.7831/ras.3.1
Saha R, Saha N, Donofrio RS, Bestervelt LL (2012) Microbial siderophores: a mini review. J Basic Microbiol 52:1–15. https://doi.org/10.1002/jobm.201100552
Saltzman A, Birol E, Bouis HE, Boy E, De Moura FF, Islam Y, Pfeiffer WH (2013) Biofortification: progress toward a more nourishing future. Glob Food Secur 2(1):9–17. https://doi.org/10.1016/j.gfs.2012.12.003
Sauer AK, Hagmeyer S, Grabrucker AM (2016) Zinc deficiency. In: Nutritional deficiency. Intech Open
Schaaf G, Honsbein A, Meda AR, Kirchner S, Wipf D, von Wiren N (2006) AtIREG2 encodes a tonoplast transport protein involved in iron dependent nickel detoxification in Arabidopsis thaliana roots. J Biol Chem 281:25532–25540. https://doi.org/10.1074/jbc.M601062200
Schaaf G, Ludewig U, Erenoglu BE, Mori S, Kitahara T, von Wiren N (2004) ZmYS1 functions as a proton coupled symporter for phytosiderophore and nicotianamine-chelated metals. J Biol Chem 5:9091–9096. https://doi.org/10.1074/jbc.M311799200
Schalk IJ, Hannauer M, Braud A (2011) New roles for bacterial siderophores in metal transport and tolerance. Environ Microbiol 13:2844–2854. https://doi.org/10.1111/j.1462-2920.2011.02556.x
Seguela M, Briat JF, Vert G, Curie C (2008) Cytokinins negatively regulate the root iron uptake machinery in Arabidopsis through a growth dependent pathway. Plant J 55:289–300. https://doi.org/10.1111/j.1365-313X.2008.03502.x
Senoura T, Sakashita E, Kobayashi T, Takahashi M, Aung MS, Masuda H, Nakanishi H, Nishizawa NK (2017) The iron-chelate transporter OsYSL9 plays a role in iron distribution in developing rice grains. Plant Mol Biol 95(4–5):375–387. https://doi.org/10.1007/s11103-017-0656-y
Sessitsch A, Kuffner M, Kidd P, Vangronsveld J, Wenzel WW, Fallmann K, Puschenreiter M (2013) The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol Biochem 60:182–194. https://doi.org/10.1016/j.soilbio.2013.01.012
Shankar AH, Prasad AS (1998) Zinc and immune function: the biological basis of altered resistance to infection. Am J Clin Nutr 68(2 Suppl):447S–463S. https://doi.org/10.1093/ajcn/68.2.447S
Sharma SK, Sharma MP, Ramesh A, Joshi OP (2012) Characterization of zinc-solubilizing Bacillus isolates and their potential to influence zinc assimilation in soybean seeds. J Microbiol Biotechnol 22:352–359. https://doi.org/10.4014/jmb.1106.05063
Shimoni-Shor E, Hassidim M, Yuval-Naeh N, Keren N (2010) Disruption of Nap14, a plastid-localized non-intrinsic ABC protein in Arabidopsis thaliana results in the over-accumulation of transition metals and in aberrant chloroplast structures. Plant Cell Environ 33:1029–1038. https://doi.org/10.1111/j.1365-3040.2010.02124.x
Shivay YS, Kumar D, Prasad R (2008) Relative efficiency of zinc sulfate and zinc oxide–coated urea in rice–wheat cropping system. Commun Soil Sci Plant Anal 39(7–8):1154–1167. https://doi.org/10.1080/00103620801925869
Shivay YS, Prasad R, Pal M (2015) Effects of source and method of zinc application on yield, zinc biofortification of grain, and Zn uptake and use efficiency in chickpea (Cicer arietinum L.). Commun Soil Sci Plant Anal 46(17):2191–2200. https://doi.org/10.1080/00103624.2015.1069320
Shukla AK, Sinha NK, Tiwari PK, Prakash C, Behera SK, Lenka NK, Singh VK, Dwivedi BS, Majumdar K, Kumar A, Srivastava PC, Pachauri SP, Meena MC, Lakaria BL, Siddiqui S (2017) Spatial distribution and management zones for sulfur and micronutrients in Shiwalik Himalayan region of India. Land Degrad Dev 28(3):959–969. https://doi.org/10.1002/ldr.2673
Shukla AK, Babu PS, Tiwari PK, Prakash C, Patra AK, Patnaik MC (2015) Current micronutrient deficiencies in soils of Telengana for their precise management. Indian J Ferti 11(8):33–43
Shukla AK, Behera SK, Lenka NK, Tiwari PK, Prakash C, Malik RS, Sinha NK, Singh VK, Patra AK, Chaudhary SK (2016) Spatial variability of soil micronutrients in the intensively cultivated trans-Gangetic Plains of India. Soil Tillage Res 163:282–289. https://doi.org/10.1016/j.still.2016.07.004
Shukla AK, Sinha NK, Tiwari PK, Prakash C, Behera SK, Surendra Babu P, Patnaik MC, Somasundaram J, Singh P, Dwivedi BS, Datta SP (2018) Evaluation of spatial distribution and regional zone delineation for micronutrients in a semiarid Deccan Plateau Region of India. Land Degrad Dev 29(8):2449–2459. https://doi.org/10.1002/ldr.2992
Sidhu GS, Sharma BD (2010) Diethylene triamine penta acetic acid–extractable micronutrients status in soil under a rice–wheat system and their relationship with soil properties in different agroclimatic zones of indo-Gangetic plains of India. Commun Soil Sci Plant Anal 41:29–51. https://doi.org/10.1080/00103620903360262
Sillanpaa M (1982) Micronutrients and the nutrient status of soils: a global study, Food Agricultural Organization
Singh B, Natesan SKA, Singh B, Usha K (2005) Improving zinc efficiency of cereals under zinc deficiency. Curr Sci 88:36–44
Singh D (2016) Enhancement of uptake and translocation of micronutrients in wheat by using endophytes. IARI Post Graduate School, New Delhi. Ph.D. thesis
Singh D, Geat N, Rajawat MV, Prasanna R, Saxena AK (2020) Performance of low and high Fe accumulator wheat genotypes grown on soils with low or high available Fe and endophyte inoculation. Acta Physiol Plant 42(2):24. https://doi.org/10.1007/s11738-019-2997-4
Singh D, Geat N, Rajawat MVS, Mahajan MM, Prasanna R, Singh S, Kaushik R, Singh RN, Kumar K, Saxena AK (2017b) Deciphering the mechanisms of endophyte-mediated biofortification of Fe and Zn in wheat. J Plant Growth Regul 37(1):174–182. https://doi.org/10.1007/s00344-017-9716-4
Singh D, Geat N, Rajawat MVS, Prasanna R, Kar A, Singh AM, Saxena AK (2018) Prospecting endophytes from different Fe or Zn accumulating wheat genotypes for their influence as inoculants on plant growth, yield, and micronutrient content. Ann Microbiol 68(12):815–833. https://doi.org/10.1007/s13213-018-1388-1
Singh D, Rajawat MVS, Kaushik R, Prasanna R, Saxena AK (2017a) Beneficial role of endophytes in biofortification of Zn in wheat genotypes varying in nutrient use efficiency grown in soils sufficient and deficient in Zn. Plant Soil 416(1–2):107–116. https://doi.org/10.1007/s11104-017-3189-x
Singh M (2009) Micronutrient nutritional problems in soils of India and improvement for human and animal health. Indian J Fertil 5(4):11–56
Singh RB (2000) Environmental consequences of agricultural development: a case study from the green revolution state of Haryana, India. Agric Ecosyst Environ 82(1):97–103. https://doi.org/10.1016/S0167-8809(00)00219-X
Sirohi G, Upadhyay A, Srivastava PS, Srivastava S (2015) PGPR mediated zinc biofertilization of soil and its impact on growth and productivity of wheat. J Soil Sci Plant Nutr 15(1):202–216. https://doi.org/10.4067/S0718-95162015005000017
Stevens GA, Finucane MM, De-Regil LM, Paciorek CJ, Flaxman SR, Branca F, Pena-Rosas JP, Bhutta ZA, Ezzati M (2013) Global, regional and national trends in haemoglobin concentration and prevalence of severe anaemia in children and pregnant and non-pregnant women for 1995-2011: a systematic analysis of population representative data. Lancet Glob Health 1(1):e16–e25. https://doi.org/10.1016/S2214-109X(13)70001-9
Ström L, Owen AG, Godbold DL, Jones DL (2002) Organic acid mediated P mobilization in the rhizosphere and uptake by maize roots. Soil Biol Biochem 34(5):703–710. https://doi.org/10.1016/S0038-0717(01)00235-8
Subramanian K, Tenshia V, Jayalakshmi K, Ramachandran V (2009) Role of arbuscular mycorrhizal fungus (Glomus intraradices)-(fungus aided) in zinc nutrition of maize. J Agric Biotech Sust Dev 1(1):29
Susin S, Abadia A, González-Reyes JA, Lucena JJ, Abadia J (1996) The pH requirement for in vivo activity of the iron-deficiency-induced “Turbo” ferric chelate reductase (a comparison of the iron-deficiency-induced iron reductase activities of intact plants and isolated plasma membrane fractions in sugar beet). Plant Physiol 110(1):111–123. https://doi.org/10.1104/pp.110.1.111
Suzuki M, Takahashi M, Tsukamoto T, Watanabe S, Matsuhashi S, Yazaki J, Kishimoto N, Kikuchi S, Nakanishi H, Mori S, Nishizawa NK (2006) Biosynthesis and secretion of mugineic acid family phytosiderophores in zinc-deficient barley. Plant J 48(1):85–97. https://doi.org/10.1111/j.1365-313X.2006.02853.x
Tariq M, Hameed S, Malik KA, Hafeez FY (2007) Plant root associated bacteria for zinc mobilization in rice. Pak J Bot 39(1):245
Tarkalson DD, Jolley VD, Robbins CW, Terry RE (1998) Mycorrhizal colonization and nutrient uptake of dry bean in manure and composted manure treated subsoil and untreated topsoil and subsoil. J Plant Nutr 21:1867–1878. https://doi.org/10.1080/01904169809365529
Thomine S, Wang R, Ward JM, Crawford NM, Schroeder JI (2000) Cadmium and iron transport by members of a plant metal transporter family in Arabidopsis with homology to Nramp genes. PNAS, USA 97:4991–4996. https://doi.org/10.1073/pnas.97.9.4991
Tian X, Lu X, Mai W, Yang X, Li S (2008) Effect of calcium carbonate content on availability of zinc in soil and zinc and iron uptake by wheat plants. Soils 40(3):425–431
Tiong J, McDonald G, Genc Y, Shirley N, Langridge P, Huang CY (2015) Increased expression of six ZIP family genes by zinc (Zn) deficiency is associated with enhanced uptake and root to shoot translocation of Zn in barley (Hordeum vulgare). New Phytol 207(4):1097–1109. https://doi.org/10.1111/nph.13413
Vacheron J, Desbrosses G, Bouffaud ML, Touraine B, Moënne-Loccoz Y, Muller D, Legendre L, Wisniewski-Dyé F, Prigent-Combaret C (2013) Plant growth-promoting rhizobacteria and root system functioning. Front Plant Sci 4:356. https://doi.org/10.3389/fpls.2013.00356
Vaid SK, Kumar B, Sharma A, Shukla AK, Srivastava PC (2014) Effect of Zn solubilizing bacteria on growth promotion and Zn nutrition of rice. J Soil Sci Plant Nutr 14(4):889–910. https://doi.org/10.4067/S0718-95162014005000071
Varotto C, Maiwald D, Pesaresi P, Jahns P, Salamini F, Leister D (2002) The metal ion transporter IRT1 is necessary for iron homeostasis and efficient photosynthesis in Arabidopsis thaliana. Plant J 31:589–599. https://doi.org/10.1046/j.1365313X.2002.01381.X
Vasconcelos M, Datta K, Oliva N, Khalekuzzaman M, Torrizo L, Krishnan S, Oliveira M, Goto F, Datta SK (2003) Enhanced iron and zinc accumulation in transgenic rice with the ferritin gene. Plant Sci 164:71–378. https://doi.org/10.1016/S0168-9452(02)00421-1
Vasconcelos MW, Gruissem W, Bhullar NK (2017) Iron biofortification in the 21st century: setting realistic targets, overcoming obstacles, and new strategies for healthy nutrition. Curr Opin Biotechnol 44:8–15. https://doi.org/10.1016/j.copbio.2016.10.001
Velu G, Crossa J, Singh RP, Hao Y, Dreisigacker S, Perez-Rodriguez P, Joshi AK, Chatrath R, Gupta V, Balasubramaniam A, Tiwari C (2016) Genomic prediction for grain zinc and iron concentrations in spring wheat. Theor Appl Genet 129(8):1595–1605. https://doi.org/10.1007/s00122-016-2726-y
Verbon EH, Trapet PL, Stringlis IA, Kruijs S, Bakker PA, Pieterse CM (2017) Iron and immunity. Annu Rev Phytopathol 55:355–375. https://doi.org/10.1146/annurev-phyto-080516-035537
Vert G, Barberon M, Zelazny E, Seguela M, Briat JF, Curie C (2009) Arabidopsis IRT2 cooperates with the high affinity iron uptake system to maintain iron homeostasis in root epidermal cells. Planta 229(6):1171–1179. https://doi.org/10.1007/s00425-009-0904-8
Vert G, Grotz N, Dedaldechamp F, Gaymard F, Guerinot ML, Briat JF, Curie C (2002) IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell 14:1223–1233. https://doi.org/10.1105/tpc.001388
Wang N, Qiu W, Dai J, Guo X, Lu Q, Wang T, Li S, Liu T, Zuo YM (2019) AhNRAMP1 enhances manganese and zinc uptake in plants. Front Plant Sci 10:415. https://doi.org/10.3389/fpls.2019.00415
Wang Y, Yang X, Zhang X, Dong L, Zhang J, Wei Y, Feng Y, Lu L (2014) Improved plant growth and Zn accumulation in grains of rice (Oryza sativa L.) by inoculation of endophytic microbes isolated from a Zn hyperaccumulator, Sedum alfredii H. J Agric Food Chem 62:1783–1791. https://doi.org/10.1021/jf404152u
Wang ZY, Bai MY, Oh E, Zhu JY (2012) Brassinosteroid signaling network and regulation of photomorphogenesis. Annu Rev Genet 46:701–724. https://doi.org/10.1146/annurev-genet-102209-163450
Waters BM, Chu H, DiDonato RJ, Roberts LA, Eisley RB, Lahner B, Salt DE, Walker EL (2006) Mutations in Arabidopsis yellow stripelike1 and yellow stripe-like3 reveal their roles in metal ion homeostasis and loading of metal ions in seeds. Plant Physiol 141:1446–1458. https://doi.org/10.1104/pp.106.082586
Watts-Williams SJ, Cavagnaro TR (2018) Arbuscular mycorrhizal fungi increase grain zinc concentration and modify the expression of root ZIP transporter genes in a modern barley (Hordeum vulgare) cultivar. Plant Sci 274:163–170. https://doi.org/10.1016/j.plantsci.2018.05.015
Welch RM (2001) Micronutrients, agriculture and nutrition; linkages for improved health and well being. Perspectives on the micronutrient nutrition of crops. Jodhpur, India: Scientific Publishers, pp. 247–289
Welch RM, Graham RD (2004) Breeding for micronutrients in staple food crops from a human nutrition perspective. J Exp Bot 55(396):353–364. https://doi.org/10.1093/jxb/erh064
Wessells KR, Brown KH (2012) Estimating the global prevalence of zinc deficiency: results based on zinc availability in national food supplies and the prevalence of stunting. PLoS One 7(11):e50568. https://doi.org/10.1371/journal.pone.0050568
Weyens N, Beckers B, Schellingen K, Ceulemans R, Croes S, Janssen J, Haenen S, Witters N, Vangronsveld J (2013) Plant associated bacteria and their role in the success or failure of metal phytoextraction projects: first observations of a field related experiment. Microbial Biotech 6:288–299. https://doi.org/10.1111/17517915.12038
White PJ, Broadley MR (2009) Biofortification of crops with seven mineral elements often lacking in human diets—iron, zinc, copper, calcium, magnesium, selenium and iodine. New Phytol 182:49–84. https://doi.org/10.1111/j.1469-8137.2008.02738.x
Whiting SN, De Souza M, Terry N (2001) Rhizosphere bacteria mobilize Zn for hyper accumulator by Thlaspi caerulescens. Environ Sci Technol 35:3144–3150. https://doi.org/10.1021/es001938v
Wieringa F, Dijkhuizen M, Fiorentino M, Laillou A, Berger J (2015) Determination of zinc status in humans: which indicator should we use? Nutr 7(5):3252–3263. https://doi.org/10.3390/nu7053252
Wissuwa M, Ismail AM, Graha RD (2008) Rice grain zinc concentrations as affected by genotypes, native soil-Zn availability and zinc fertilization. Plant Soil 306:37–48. https://doi.org/10.1007/s11104-007-9368-4
World Health Organization (2009) Global health risks: mortality and burden of disease attributable to selected major risks. World Health Organization, Geneva
World Health Organization (2008) The global burden of disease: 2004 update. World Health Organization, Geneva, Switzerland
Wu T, Zhang HT, Wang Y, Jia WS, Xu XF, Zhang XZ, Han ZH (2012) Induction of root Fe (III) reductase activity and proton extrusion by iron deficiency is mediated by auxin-based systemic signalling in Malus xiaojinensis. J Exp Bot 63(2):859–870. https://doi.org/10.1093/jxb/err314
Wu W, Ma B (2015) Integrated nutrient management (INM) for sustaining crop productivity and reducing environmental impact: a review. Sci Total Environ 512:415–427. https://doi.org/10.1016/j.scitotenv.2014.12.101
Xiao H, Yin L, Xu X, Li T, Han Z (2008) The iron-regulated transporter, MbNRAMP1, isolated from Malus baccata is involved in Fe, Mn and Cd trafficking. Ann Bot 102:881–889. https://doi.org/10.1093/aob/mcn178
Xie X, Zhang H, Pare P (2009) Sustained growth promotion in Arabidopsis with long-term exposure to the beneficial soil bacterium Bacillus subtilis (GB03). Plant Signal Behav 4(10):948–953. https://doi.org/10.4161/psb.4.10.9709
Xiong H, Kobayashi T, Kakei Y, Senoura T, Nakazono M, Takahashi H, Nakanishi H, Shen H, Duan P, Guo X, Nishizawa NK, Zuo Y (2012) AhNRAMP1 iron transporter is involved in iron acquisition in peanut. J Exp Bot 63(2):695–709. https://doi.org/10.1093/jxb/ers117
Xu YG, Wang BS, Yu JJ, Ao GM, Zhao Q (2010) Cloning and characterisation of ZmZLP1, a gene encoding an endoplasmic reticulum-localised zinc transporter in Zea mays. Funct Plant Biol 37(3):194–205. https://doi.org/10.1071/FP09045
Yadava DK, Choudhury PR, Hossain F, Kumar D (2017) Biofortified varieties: sustainable way to alleviate malnutrition. Indian Council of Agricultural Research, New Delhi
Yoo SM, James BR (2002) Zinc extractability as a function of pH in organic waste amended soils. Soil Sci 167:246–259. https://doi.org/10.1097/00010694-200204000-00002
Yoshino M, Murakami K (1998) Interaction of iron with polyphenolic compounds: application to antioxidant characterization. Anal Biochem 257(1):40–44. https://doi.org/10.1006/abio.1997.2522
Zaidi A, Khan MS, Amil MD (2003) Interactive effect of rhizotrophic microorganisms on yield and nutrient uptake of chickpea (Cicer arietinum L.). Eur J Agron 19(1):15–21. https://doi.org/10.1016/S1161-0301(02)00015-1
Zamioudis C, Hanson J, Pieterse CMJ (2014) β-Glucosidase BGLU42 is a MYB72-dependent key regulator of rhizobacteria-induced systemic resistance and modulates iron deficiency responses in Arabidopsis roots. New Phytol 204:368–379. https://doi.org/10.1111/nph.12980
Zamioudis C, Korteland J, Van Pelt JA, Van Hamersveld M, Dombrowski N et al (2015) Rhizobacterial volatiles and photosynthesis-related signals coordinate MYB72 expression in Arabidopsis roots during onset of induced systemic resistance and iron-deficiency responses. Plant J 84:309–322. https://doi.org/10.1111/tpj.12995
Zhang C, Shinwari K, Luo L, Zheng L (2018) OsYSL13 is involved in iron distribution in rice. Int J Mol Sci 19(11):3537. https://doi.org/10.3390/ijms19113537
Zhang F, Shen J, Jing J, Li L, Chen X (2010) Rhizosphere processes and management for improving nutrient use efficiency and crop productivity. Molecular Environmental Soil Science at the Interfaces in the Earth’s Critical Zone: Springer pp 52–54. https://doi.org/10.1007/978-3-642-05297-2-16
Zhang X, Lin L, Chen M, Zhu Z, Yang W, Chen B, Yang X, An Q (2012a) A nonpathogenic Fusarium oxysporum strain enhances phytoextraction of heavy metals by the hyperaccumulator Sedum alfredii Hance. J Hazard Mater 229:361–370. https://doi.org/10.1016/j.jhazmat.2012.06.013
Zhang X, Zhang D, Sun W, Wang T (2019) The adaptive mechanism of plants to iron deficiency via iron uptake, transport, and homeostasis. Int J Mol Sci 20(10):2424. https://doi.org/10.3390/ijms20102424
Zhang Y, Xu YH, Yi HY, Gong JM (2012b) Vacuolar membrane transporters OsVIT1 and OsVIT2 modulate iron translocation between flag leaves and seeds in rice. Plant J 72(3):400–410. https://doi.org/10.1111/j.1365-313X.2012.05088.x
Zheng X, Chen L, Li X (2018) Arabidopsis and rice showed a distinct pattern in ZIPs genes expression profile in response to Cd stress. Bot Stud 59(1):22. https://doi.org/10.1186/s40529-018-0238-6
Zhu XF, Jiang T, Wang ZW, Lei GJ, Shi YZ, Li GX, Zheng SJ (2012) Gibberellic acid alleviates cadmium toxicity by reducing nitric oxide accumulation and expression of IRT1 in Arabidopsis thaliana. J Hazard Mater 239:302–307. https://doi.org/10.1016/j.jhazmat.2012.08.077
Acknowledgments
The Division of Microbiology, ICAR-IARI, New Delhi is gratefully acknowledged for the facilities provided, during the present study.
Funding
The authors are thankful to the ICAR-Indian Agricultural Research Institute and Indian Council of Agricultural Research (ICAR), New Delhi for providing financial support.
Author information
Authors and Affiliations
Contributions
Devendra Singh collected the information, wrote the first draft, and prepared the figures and tables. Both the authors conceptualized the topic and its subdivisions. Radha Prasanna edited the draft and revised versions and provided inputs for the improvement of figures and tables.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Singh, D., Prasanna, R. Potential of microbes in the biofortification of Zn and Fe in dietary food grains. A review. Agron. Sustain. Dev. 40, 15 (2020). https://doi.org/10.1007/s13593-020-00619-2
Accepted:
Published:
DOI: https://doi.org/10.1007/s13593-020-00619-2