Abstract
To compare the oncological survival outcome between extended resections (ER) and segmental resection (SR) for non-metastatic splenic flexure tumors. A total of 10,063 splenic flexure colon cancers patients who underwent ER (n = 5546) or SR (n = 4517) from 2010 to 2018 were included from the Surveillance, Epidemiology, and End Results (SEER)-registered database. Additionally, we included 135 patients from our center who underwent ER (n = 54) or SR (n = 81) between 2011 and 2021. Survival rates were compared between groups. To reduce the inherent bias of retrospective studies, propensity score matching (PSM) analysis was performed. In the SEER database, patients in the ER group exhibited higher pT stage, pN stage, larger tumor size, and elevated rates of CEA level, perineural invasion, and tumor deposits compared to those in the SR group (each P < 0.05). The 5-year cancer-specific survival (CSS) rate was slightly lower in the ER group than in the SR group (79.2% vs. 81.6%, P = 0.002), while the 5-year overall survival (OS) rates were comparable between the two groups (66.2% vs. 66.9%, P = 0.513). After performing PSM, both the 5-year CSS and 5-year OS rates were comparable between the ER and SR groups (5-year CSS: 84.9% vs. 83.0%, P = 0.577; 5-year OS: 70.6% vs. 66.0%, P = 0.415). These findings were consistent in the subgroup analysis that included only patients with stage III disease or tumor size ≥ 7 cm. Furthermore, although the number of harvested lymph nodes was higher in the ER group compared to the SR group (14.4 vs. 12.7, P < 0.001), the number of invaded lymph nodes remained similar between the two groups (0.5 vs. 0.5, P = 0.90). Similarly, our center's data revealed comparable 3-year OS and 3-year disease-free survival (DFS) rates between the two groups. ER have no significant oncological benefits over SR in the treatment of non-metastatic splenic flexure colon cancer, even for locally advanced cases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Experience with splenic flexure colon cancers is very limited because it is a relatively rare disease that represents only less than 10% of all colon cancers [1]. Although left-sided colon cancer was generally associated with better survival compared to right-sided colon cancer [2], splenic flexure tumors had a higher risk of obstruction and had a more advanced stage and lower cure rate among left-sided colon cancers [3].
Since anatomical variations of the vasculature at splenic flexure by left colic artery (LCA) and middle colic artery (MCA) remain ambiguous [4], surgery for splenic flexure cancers is one of the most challenging operations among other colorectal surgical procedures [5]. Splenic flexure colon, located between the right and left colon, received a dual vascular supply of both superior mesenteric vessels and inferior mesenteric vessels. Moreover, due to the complexity of the lymphatic drainage in this site, a consensus is lacking regarding the optimal extent of surgical resection for splenic flexure tumors. The AJCC and College of American Pathologists recommend the examination of a minimum of 12 lymph nodes to accurately stage colon cancers [6]. However, since right colon cancer and left colon cancer have different characteristics, different thresholds for the total number of lymph nodes retrieved have been proposed. Yang et al. [7] proposed a threshold total number of lymph nodes of 11 for left colon cancer while the 12-node standard might only be applicable in right-sided colon cancer. In addition, since splenic colon cancers exhibited exclusively specific characteristics as compared to colon cancers at other sites [8], the optimum number of lymph nodes harvested remains ill-defined. Various extent of conventional techniques has been advocated, going from more aggressive extended resection (ER) to segmental resection (SR) [9]. However, there is a scarcity of publications regarding its surgical outcomes, especially the long-term oncological survival [10]. Some researchers supported ER due to the highly heterogeneous lymphatic drainage of the splenic flexure tumors [11], whereas their opponents advocated for colon-sparing options to avoid an unnecessary resection to preserve colonic length [8]. In our updated meta-analysis with the latest evidence, ER, including subtotal colectomy (STC), extended right hemicolectomy (ERHC), and standard left hemicolectomy (LHC), provided similar survival compared to SR of splenic flexure colectomy (SFC) [12]. However, it is worth noting that only retrospective studies with small sample sizes were included and proper confounder analysis was impossible due to methodological limitations. In addition, although cases from 1996 to 2018 were included and the extent of resection varied according to diagnosis year across centers, it was impossible to evaluate their impact on the oncological survival of splenic flexure colon cancer surgery.
The primary aim of this study was to assess the oncological survival rates of patients with non-metastatic splenic flexure tumors who underwent either ER or SR. This analysis was conducted by examining data from the Surveillance, Epidemiology, and End Results (SEER)-registered database, as well as our institution's own experience over the course of a decade. Additionally, the study aimed to determine the optimal number of lymph nodes that should be removed during surgeries for splenic flexure tumors. Finally, the changes in surgical extent for splenic flexure tumors over time in this global series and their impact on oncological outcome were also analyzed.
Materials and methods
Patients
This was a retrospective cohort study. This study was approved for exemption by the Institutional Review Board, identification number 2023KY221.
SEER program
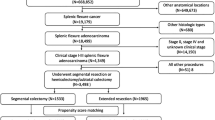
The SEER program includes population-based tumor registries that cover 28% of the U.S. population. A total of 13,682 patients diagnosed with splenic flexure colon cancer from 2010 to 2018 were initially included. Patients were excluded according to the following criteria: 1) patients had other histology (n = 506); 2) patients had stage 0 (n = 263) and stage IV diseases (n = 1782); 3) patients had received radiotherapy (n = 128); 4) patients had no surgery of primary site (n = 181) or unknown if surgery performed (n = 3), had local tumor destruction or excision (n = 200), or had total colectomy or total proctocolectomy (n = 556). Finally, a total of 10,063 patients were included in the analysis (Fig. S1).
According to the SEER Program Coding and Staging Manual 2018, SR was defined as partial colectomy [but less than hemicolectomy] segmental resection. ER was defined as subtotal colectomy/hemicolectomy (total right or left colon and a portion of transverse colon).
Our center's data
In order to comprehensively analyze the specific details of the surgical extent, a total of 135 patients from our institution who underwent ER (n = 54) or SR (n = 81) between 2011 and 2021 were also included. SR was defined as resection of the distal part of the transverse colon and the descending colon by ligating the LCA with or without ligating the left branch of the MCA or accessory middle colic artery (aMCA). ER encompassed two distinct surgical procedures, namely left hemicolectomy (LHC) and extended left hemicolectomy (ELHC). LHC involved the removal of the distal part of the transverse colon, the descending colon, and the sigmoid colon by ligating the MCA and LCA. ELHC was a more extensive surgery that involved the removal of the distal part of the transverse colon, descending and sigmoid colon down to the rectosigmoid union by ligating the inferior mesenteric artery (IMA) with or without ligating other arteries. D3 lymphadenectomy was routinely performed. The resection procedure entailed the excision of a segment of the colon measuring at least 10 cm in length on both sides of the tumor. However, it is customary to consider wider margins due to the mandatory ligation of the arterial blood supply.
Statistical analysis
The primary endpoint was the oncological survival rates, including cancer-specific survival (CSS), overall survival (OS), and disease-free survival (DFS).
Categorical variables were compared using Fisher's exact tests, while the T-test or Wilcoxon rank-sum test was applied for continuous variables. The Kaplan–Meier method with a Log-rank test was used for survival analysis and comparison of CSS, OS and DFS rates. X-tile (ver. 3.6.1) was used to determine the thresholds for tumor size, harvested lymph nodes, and diagnosis year, by selecting the maximal chi-square values of the log-rank test for CSS between 2 groups. In the SEER database, multivariate analyses of CSS were performed using Cox proportional regression models to explore the independent prognostic factors. To reduce the imbalance in the distribution of the baseline data between ER and SR, propensity score matching (PSM) was performed by logistic regression for each patient using the covariates of age race, grade, pT stage, pN stage, CEA status, perineural invasion, tumor deposits, tumor size and diagnosis year. One-to-one matching without replacement was performed using a 2e−14 caliper width. The score-matched pairs were used in subsequent survival analyses. P values of less than 0.05 were considered to indicate statistical significance. All statistical analyses were performed using the SPSS software (ver. 22) and R (ver. 3.4.1).
Results
SEER program
A total of 10,063 splenic flexure colon cancers patients who underwent ER (n = 5546) or SR (n = 4517) were included in this study. Overall, 3447 (34.5%) patients had stage III disease. Baseline characteristics of patients between the ER group and SR group were reported in Table 1. Patients from the ER group were younger than those from the SR group (P < 0.001). The patients in the ER group exhibited more advanced tumor stages, including higher pT stage and pN stage, compared to those in the SR group. Additionally, the ER group had higher rates of elevated carcinoembryonic antigen (CEA) levels, perineural invasion, and tumor deposits, all of which were statistically significant (P < 0.05) when compared to the SR group. The size of the tumor was larger in the ER group than in the SR group (P < 0.001). In addition, these two groups had imbalanced distributions regarding race, grade, and diagnosis year (each P < 0.05).
The median follow-up period was 58 months (interquartile range: 22–114). The 5-year CSS rate was slightly lower in ER patients than that in SR patients (79.2% vs. 81.6%, respectively, P = 0.002) (Fig. 1A), while the 5-year OS was comparable in the ER group and SR group (66.2% vs. 66.9%, respectively, P = 0.513) (Fig. 1C). However, after the PSM performed, both the 5-year CSS and 5-year OS rates in ER group and SR group were comparable (5-year CSS: 84.9% vs. 83.0%, P = 0.577; 5-year OS: 70.6% vs. 66.0%, P = 0.415, Fig. 1B, D). In addition, after the PSM performed, although the numbers of harvested lymph nodes were larger in the ER group compared to the SR group (14.4 vs. 12.7, P < 0.001) the numbers of invaded lymph nodes remained similar between groups (0.5 vs. 0.5, P = 0.90) (Table 1).
Survival between the segmental resection group and extended resection group in the Surveillance, Epidemiology, and End Results database. A CSS of unmatched patients, B CSS of propensity-matched patients, C OS of unmatched patients, and D OS of propensity-matched patients. CSS, cancer specific survival, OS, overall survival
X-tile analysis determined the optimum threshold of 11 for the harvested lymph node count during non-metastatic splenic flexure colon cancer surgeries (Fig. S2A). Further survival analysis showed that patients with ≥ 11 lymph nodes harvested had better 5-year CSS than patients with less than 11 lymph nodes harvested (81.3% vs. 78.2%, respectively, P < 0.001, Fig. S2B). For standard threshold of 12 lymph nodes, patients with ≥ 12 lymph nodes harvested also experienced better 5-year CSS than patients with less than 12 lymph nodes harvested (81.4% vs. 78.7%, respectively, P < 0.001, Fig. S2C). However, it is worth noting that there was no difference regarding 5-year CSS between patients with < 11 lymph nodes harvested and patients with 11 lymph nodes harvested (P = 0.937), while significant CSS difference was found between patients with 11 lymph nodes harvested and patients with ≥ 12 lymph nodes harvested (P < 0.001, Fig. S2D). Thus, twelve appeared a superior threshold to 11 regarding the harvested lymph node count during non-metastatic splenic flexure colon cancer surgeries, as it better distinguished patients with a poor prognosis from those with a good prognosis.
Using the optimum threshold of 2002 and 2013 obtained with X-tile, there was a significant trend toward better CSS and OS over 2000–2018 (P < 0.001, Fig. 2A, C). Regarding the indicator of the quality of surgery, an increased rate of harvested lymph node count ≥ 12 was observed over these 18 years (P < 0.001, Fig. 2D). Interestingly, we found an increased adoption of SR for the treatment of non-metastatic splenic flexure colon cancer as the year progresses (P = 0.005, Fig. 2B).
The impact of diagnosis year on survival in the Surveillance, Epidemiology, and End Results database. A OS among ~ 2002 group, 2002–2013 group and 2013 ~ group; B adoption of segmental resection among diagnosis year groups; C CSS among ~ 2002 group, 2002–2013 group and 2013 ~ group; D rate of harvested lymph node count ≥ 12 among diagnosis year groups. CSS, cancer specific survival, OS, overall survival
Multivariate analysis
To identify prognostic factors of CSS, univariate and multivariate analysis were performed. Among these factors, older age at diagnosis (P < 0.001), poorly differentiation (P = 0.018), higher pT stages (P < 0.001) and pN stages (P < 0.001), elevated CEA level (P < 0.001), the presence of perineural invasion (P = 0.005), a smaller number of harvested lymph nodes (P < 0.001), a larger number of invaded lymph nodes (P < 0.001) and larger tumor size (≥ 7 cm, threshold obtained with X-tile) (P < 0.001) were found to be independently and significantly correlated with worse CSS (Table 2). In addition, diagnosis year, marital status, and race were independent prognostic factors of CSS, while the procedure of SR was slightly and independently associated with improved CSS (P = 0.035).
Subgroup analysis
Since ER was more frequently applied in advance stage tumors as shown in the analysis of baseline characteristics, we performed a subgroup analysis that included only stage III cancer cases or cases with a tumor larger than ≥ 7 cm. The results revealed that both OS and CSS in the ER and the SR groups were similar across all subgroups (each P > 0.05, Fig. 3).
Our center's data
A total of 135 patients from our center who underwent ER (n = 54) or SR (n = 81) between 2011 and 2021 were included. Baseline characteristics of patients between the ER group and SR group were reported in Table S1. The variables of sex, age at diagnosis, pT stage, pN stage, histopathology, grade, and presence of lymphatic or vascular invasion are well-balanced and exhibit comparability between the ER group and the SR group (P > 0.05). With a median follow-up duration of 56 months, the survival analysis revealed that the 3-year OS and 3-year DFS rates were similar between the ER and SR groups (3-year OS: 91.0% vs. 91.6%, P = 0.715; 3-year DFS: 86.7% vs. 86.3%, P = 0.935, Fig. 4).
Discussion
The extent of resection for non-metastatic splenic flexure colon cancer chosen by the surgeon is often empirically supported rather than based on evidence [13]. Extended procedures are the most common surgical treatments so far for splenic flexure colon cancer [14], and have been recommended to overcome the poor oncologic outcomes observed in patients with splenic flexure colon cancers [15]. One survey fielded on this topic showed that ERHC was the most preferred procedure by 63% of those surveyed followed by LHC (23%) and SR (14%) in the United Kingdom [16]. In China, our recent survey revealed that the most preferred procedure for splenic flexure colon cancer was LHC or ELHC (97.4%), followed by SR (1.8%) and ERHC (0.9%) (manuscript not published). Interestingly, increased adoption of SR for the treatment of non-metastatic splenic flexure colon cancer as the year progresses was observed in the present study.
For short-term outcomes, compared to a formal LHC, a SR was reported to be associated with a shorter operative time with equivalent post-operative morbidity based on the analysis of the ACS-NSQIP colectomy-targeted database [17]. Our previous network meta-analysis revealed that ERHC was associated with a higher risk of postoperative ileus but a non-significant trend for lower anastomotic dehiscence when compared with more restricted resections [12]. However, the evidence in the literature regarding the long-term oncological advantages of ER over SR is sparse. A nationwide retrospective cohort study including 1304 cases showed that SR and extended procedures (extended right and left colectomies) had comparable survival rates (5-year OS 84% vs. 83%, 5-year progression-free survival 85% vs. 84%) [18, 19]. In fact, our study is the largest study to date to compare survival between SR and extended procedure. Our initial analysis of baseline characteristics showed that ER was more frequently applied in advance-stage tumors, which was probably the reason why ER was associated with a slightly lower 5-year CSS rate. This hypothesis was confirmed by the re-comparison of the survival after the PSM or subgroups analysis of locally advanced stage tumors (stage III or tumor size ≥ 7 cm). The results of the re-comparison revealed that both the 5-year CSS and 5-year OS rates in the ER group and SR group were comparable. Furthermore, the robustness of this hypothesis was strengthened by the inclusion of data from our center, which spanned over a decade. Moreover, while the numbers of harvested lymph nodes increased in the ER group the numbers of invaded lymph nodes remained relatively constant between groups. This fact together with the previous indications suggested that extended procedures have no statistically significant oncological benefits over a less aggressive approach of SR. SR seems an effective alternative for splenic flexure tumors, even for non-metastatic locally advanced cases.
Interestingly, in the present study, we found a potential link between a later year of diagnosis and improved CSS over 2000–2018. From a population-based perspective, an increased rate of harvested lymph node count ≥ 12 or ≥ 11 observed over this 18-year period might be the underlying reason. Stocchi et al. [20] found that harvest of at least 12 nodes was related to surgery after 1991 (85% vs. 69%, P < 0.001) in stage II colon carcinoma. Since the 12-node minimum rule was first proposed in 1990 [21], this fact together with the positive link indicated that it is of great importance to understand and determine the optimal cutoff for the number of lymph nodes needed for surgery of non-metastatic splenic flexure colon cancer.
Besides the detection of involvement, the number of harvested lymph nodes itself has prognostic relevance in stage colorectal II/III cancers [22]. However, the exact relationship between the examined lymph node count and CSS at the time of resection for non-metastatic splenic flexure colon cancer is still ill-defined. In addition, although the 12-node minimum has been recognized as a consensus standard for surgical performance for colorectal cancer [23], this threshold was met only in about three-quarters of cases (85% for right colon carcinomas and 72% for left colon carcinomas [20]). It is known that the number of harvested lymph nodes was related to surgery over time, colon tumor location (right colon or left colon), individual surgeon, and length of specimen [20]. Thus, the 12-node standard for the total number of lymph nodes may not be equally applicable in all cancer locations. At least two previous studies fielded on this topic had proposed a lower threshold of 11 to be the optimum number of harvested lymph nodes for left-sided colon cancer [7, 24]. In the present study, the analysis of population-level data showed that no difference regarding 5-year CSS between patients with < 11 lymph nodes harvested and patients with 11 lymph nodes harvested were found, while significant CSS difference was observed between patients with 11 lymph nodes harvested and patients with ≥ 12 lymph nodes harvested. Thus, we confirmed the optimum threshold of 12 for the harvested lymph node count during non-metastatic splenic flexure colon cancer surgeries. On the other hand, larger numbers of harvested lymph nodes in the ER did not confer any additional benefit relative to SR with regard to CSS, probably due to no increase in the resected invaded lymph node numbers in the present study. Thus, understanding the distribution of metastatic lymph nodes and the different anatomies for non-metastatic splenic flexure colon cancer might be essential. For instance, the inferior mesenteric artery lymph node was routinely resected during LHC while might normally have been left behind in ERHC and SR in China. Our recently published study analyzed a single-center experience of 117 consecutive non-metastatic splenic flexure colon cancer surgeries performed over a period of 7 years and observed no metastatic lymph nodes at the root of the inferior mesenteric artery [25], which is similar to a previous study observed by Nakagoe et al. [26] Consequently, It might not be necessary to resect the inferior mesenteric artery lymph node during non-metastatic splenic flexure colon cancer surgery.
The strength of this study lies in the large patient numbers, even for relatively rare colon cancer of splenic flexure, and long follow-up time, which indicates the almost real-world outcomes and might be a powerful resource for identifying gross trends. However, there are several limitations of this study. First, confusing nomenclature as well as the uncertainty in exact surgical extent are common in the literature regarding colectomy for splenic flexure colon cancer [27]. Like all registry studies based on the SEER database, more detailed information about surgical extent was not provided. However, to provide a more comprehensive elucidation of the surgical extent, we have integrated a consecutive case series encompassing a decade from our center, delineating the intricate details of surgical extent in terms of both SR and ER. Secondly, our study was a retrospective data analysis and therefore subject to the limitation common to this research. However, the PSM was performed to adjust for confounding factors and to overcome potential selection bias.
Conclusion
ER have no significant oncological benefits over SR in the treatment of non-metastatic splenic flexure colon cancer, even for locally advanced cases.
Data availability
The data used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Shaikh IA, Suttie SA, Urquhart M, Amin AI, Daniel T, Yalamarthi S (2012) Does the outcome of colonic flexure cancers differ from the other colonic sites? Int J Colorectal Dis 27(1):89–93
Azar I, Al Masalmeh N, Esfandiarifard S, Virk G, Kiwan W, Frank Shields A et al (2021) The impact of primary tumor sidedness on survival in early-onset colorectal cancer by stage: a national veterans affairs retrospective analysis. Cancer Med 10(9):2987–2995
Nakagoe T, Sawa T, Tsuji T, Jibiki M, Nanashima A, Yamaguchi H et al (2000) Carcinoma of the splenic flexure: multivariate analysis of predictive factors for clinicopathological characteristics and outcome after surgery. J Gastroenterol 35(7):528–535
Zou J, Jiang X, Feng J, Cai J, Kong D, Cao W et al (2022) Anatomical variations of the branches from left colic artery and middle colic artery at splenic flexure. Surg Radiol Anatomy SRA 44(3):467–473
Goksoy B (2022) Techniques and feasibility of the cranial-to-caudal approach for laparoscopic left colectomy in obstructive splenic flexure cancers: a consecutive case series (with video). Colorectal Dis 24:1430
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) (2019) Colon Cancer Version 2.2019—May 15
Yang L, Xiong Z, Xie Q, He W, Liu S, Kong P et al (2018) Prognostic value of total number of lymph nodes retrieved differs between left-sided colon cancer and right-sided colon cancer in stage III patients with colon cancer. BMC Cancer 18(1):558
Kim CW, Shin US, Yu CS, Kim JC (2010) Clinicopathologic characteristics, surgical treatment and outcomes for splenic flexure colon cancer. Cancer Res Treat 42(2):69–76
Bademci R, Bollo J, Martinez Sanchez C, Hernadez P, Targarona EM (2019) Is segmental colon resection an alternative treatment for splenic flexure cancer? J Laparoendosc Adv Surg Tech A 29(5):621–626
de’Angelis N, Martínez-Pérez A, Winter DC, Landi F, Vitali GC, Le Roy B et al (2021) Extended right colectomy, left colectomy, or segmental left colectomy for splenic flexure carcinomas: a European multicenter propensity score matching analysis. Surg Endosc 35(2):661–672
Kim S, Gendia A, Ur Rehman M, Malik K, Ahmed J (2022) Laparoscopic extended right hemicolectomy for locally advanced splenic flexure tumour—a video vignette. Colorectal Dis 24(6):802
Wang X, Zheng Z, Chen M, Lu X, Huang S, Huang Y et al (2021) Subtotal colectomy, extended right hemicolectomy, left hemicolectomy, or splenic flexure colectomy for splenic flexure tumors: a network meta-analysis. Int J Colorectal Dis 36(2):311–322
Odermatt M, Siddiqi N, Johns R, Miskovic D, Khan O, Khan J et al (2014) Short- and long-term outcomes for patients with splenic flexure tumours treated by left versus extended right colectomy are comparable: a retrospective analysis. Surg Today 44(11):2045–2051
Rega D, Pace U, Scala D, Chiodini P, Granata V, Fares Bucci A et al (2019) Treatment of splenic flexure colon cancer: a comparison of three different surgical procedures: experience of a high volume cancer center. Sci Rep 9(1):10953
Sadler GP, Gupta R, Foster ME (1992) Carcinoma of the splenic flexure–a case for extended right hemicolectomy? Postgrad Med J 68(800):487
Chan D, Shah P, Saklani A (2013) Current trends and controversies in the management of patients with splenic flexure tumours. J Cancer Res Therapy 1:8–10
Pang AJ, Marinescu D, Morin N, Vasilevsky CA, Boutros M (2022) Segmental resection of splenic flexure colon cancers provides an adequate lymph node harvest and is a safe operative approach—an analysis of the ACS-NSQIP database. Surg Endosc 36(8):5652–5659
Degiuli M, Reddavid R, Ricceri F, Di Candido F, Ortenzi M, Elmore U et al (2020) Segmental colonic resection is a safe and effective treatment option for colon cancer of the splenic flexure: a nationwide retrospective study of the Italian society of surgical oncology-colorectal cancer network collaborative group. Dis Colon Rectum 63(10):1372–1382
Martín Arévalo J, Moro-Valdezate D, García-Botello SA, Pla-Martí V, Garcés-Albir M, Pérez Santiago L et al (2018) Propensity score analysis of postoperative and oncological outcomes after surgical treatment for splenic flexure colon cancer. Int J Colorectal Dis 33(9):1201–1213
Stocchi L, Fazio VW, Lavery I, Hammel J (2011) Individual surgeon, pathologist, and other factors affecting lymph node harvest in stage II colon carcinoma. is a minimum of 12 examined lymph nodes sufficient? Ann Surg Oncol 18(2):405–412
Fielding LP, Arsenault PA, Chapuis PH, Dent O, Gathright B, Hardcastle JD et al (1991) Clinicopathological staging for colorectal cancer: an international documentation system (IDS) and an international comprehensive anatomical terminology (ICAT). J Gastroenterol Hepatol 6(4):325–344
Märkl B (2015) Stage migration vs immunology: the lymph node count story in colon cancer. World J Gastroenterol 21(43):12218–12233
Wong SL (2009) Lymph node counts and survival rates after resection for colon and rectal cancer. Gastrointest Cancer Res GCR 3:S33-35
Guan X, Chen W, Liu Z, Jiang Z, Hu H, Zhao Z et al (2016) Whether regional lymph nodes evaluation should be equally required for both right and left colon cancer. Oncotarget 7(37):59945–59956
Huang M, Wang X, Shao Y, Huang S, Huang Y, Chi P (2022) Surgical treatment of splenicflexure colon cancer: analysisof short-term and long-term outcomes of three differentsurgical procedures. Front Oncol 12:884484
Nakagoe T, Sawai T, Tsuji T, Jibiki M, Ohbatake M, Nanashima A et al (2001) Surgical treatment and subsequent outcome of patients with carcinoma of the splenic flexure. Surg Today 31(3):204–209
Wang X, Zheng Z, Yu Q, Chi P (2020) Comment on “comparison of extended right hemicolectomy, left hemicolectomy, and segmental colectomy for splenic flexure colon cancer (SFC): a systematic review and meta-analysis.” Colorectal Dis 22(12):2332–2333
Funding
This study was financially supported by Fujian provincial health technology project (2021GGA013), Natural Science Foundation of Fujian Province (2023J01099).
Author information
Authors and Affiliations
Contributions
Xiaojie Wang: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Validation, Visualization, Roles, Writing original draft. Yangyang Wang: Data curation, Formal analysis, Writing original draft. Zhifang Zheng: Data curation, Review & editing. Zhiping Chen: Data curation, Review & editing. Zhongdong Xie: Data curation, Review & editing. Xingrong Lu: Methodology. Shenghui Huang: Supervision. Ying Huang: Supervision, Funding acquisition. Pan Chi: Supervision, Funding acquisition, Review & editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Wang, Y., Zheng, Z. et al. Extended procedure has no oncological benefits over segmental resection in the treatment of non-metastatic splenic flexure colon cancer, a population-based cohort study and a single center’s decade-long experience. Updates Surg 76, 1289–1299 (2024). https://doi.org/10.1007/s13304-024-01897-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13304-024-01897-1