Abstract
Since the roles of autophagy in gastric cancer remain unclear, we aim to investigate the expression of autophagy-related proteins MAP1LC3B and Beclin-1 in human gastric cancer and discuss their clinical significance and correlation with prognosis of patients with gastric cancer. A total of 160 consecutive patients with gastric cancer who had undergone gastrectomy were enrolled in this study. The expressions of MAP1LC3B and Beclin-1 were assessed by immunohistochemistry. The protein expression rates were analyzed with χ 2 and Fisher’s exact tests. Survival analysis (overall survival (OS) and relapse-free survival (RFS)) was determined using the Kaplan–Meier method and Cox’s proportional hazard regression model. Both the expressions of MAP1LC3B and Beclin-1 were lower in gastric cancer tissues than adjacent normal tissues (57 vs. 82 %, p = 0.007; 72 vs. 88 %, p = 0.046, respectively). Relativity analysis indicated MAP1LC3B expression was positively correlated with Beclin-1 expression (r = 0.424, p < 0.001). Both the MAP1LC3B-high-expression patients and Beclin-1-high-expression patients have longer OS time and RFS time than MAP1LC3B-low-expression patients and Beclin-1-low-expression patients (MAP1LC3B: both p < 0.001; Beclin-1: p = 0.014, p = 0.015, respectively). High simultaneous MAP1LC3B and Beclin-1 expressions were associated with longer OS and RFS compared with low simultaneous MAP1LC3B and Beclin-1 expressions (56.77 vs. 24.42 months, p < 0.001; 53.56 vs. 22.33 months, p < 0.001, respectively). Multivariate survival analysis showed both MAP1LC3B and Beclin-1 were independent prognostic factors for OS time (p = 0.016, p = 0.041, respectively). However, MAP1LC3B (p = 0.022) was an independent prognostic factor for RFS. Moreover, low expressions of MAP1LC3B and Beclin-1 were significantly associated with lymph node metastasis (p = 0.007, p = 0.030, respectively). The loss of MAP1LC3B, correlated with loss of Beclin-1, was observed in gastric cancer and correlated with poor prognosis and lymph node metastasis of gastric cancer patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer is the fifth most common cancer over the world, and the fatality rate is 75 %. It is also the third leading cause of death in both sexes (723,000 deaths), accounting for 8.8 % of the total deaths from cancer [1]. Gastric cancer incidence rates vary wildly between men and women and across different countries. Rates are twofold to threefold higher in men than women. Comparing nations, the highest incidence rates are observed in East Asia, East Europe, and South America, while the lowest rates are observed in North America and most parts of Africa [2]. Since the 1970s, there have been notable improvements in the relative 5-year survival rates for gastric cancer [3], however, survival rates remain dismal [4]. The overall 5-year relative survival rate is about 20 % in most areas of the world, except in Japan, where 5-year survival rates of above 70 % for stages I and II of gastric cancer have been reported [5]. As measured by disability-adjusted life years lost, gastric cancer also causes one of the highest cancer burdens [6].
Autophagy is a process of self-cannibalization. Cells capture their own cytoplasm and organelles and consume them in lysosomes. The resulting breakdown products are inputs for cellular metabolism, through which they are used to generate energy and to build new proteins and membranes [7]. Autophagy is thought to play a dual role in cancer, where it can prevent tumor initiation by suppressing chronic tissue damage, inflammation, and genome instability via its quality control function or can sustain tumor metabolism, growth, and survival via nutrient recycling [8]. The autophagy gene Beclin-1 (the mammalian counterpart of the yeast ATG6 gene), which is part of a type III phosphatidylinositol 3-kinase complex required for autophagic vesicle formation, is a haploinsufficient tumor suppressor in mice [9, 10] and is monoallelically lost in human breast, ovarian, and other tumors [11]. MAP1LC3B is produced in a pro-form (pro-MAP1LC3B), which is cleaved by ATG4 into a cytosolic form referred to as MAP1LC3B-I. During autophagy, MAP1LC3B-I is conjugated to the lipid phosphatidylethanolamine (referred to as MAP1LC3B-II), which is then inserted into both inner and outer membranes of the growing autophagic vesicle [12, 13]. Cells deficient in MAP1LC3B or ATG5 are defective in formation of autophagic vesicles and reveal numerous phenotypes related to the role of autophagy in energy homeostasis and removal of protein aggregates [14, 15].
Recently, the role of autophagy in cancer development and cancer treatment has been given great concern. Several researches showed that Beclin-1 was an independent prognostic factor for gastric cancer patients [16–18]. However, there are still relatively few studies that have investigated the association between MAP1LC3B and gastric cancer. In addition, it has been reported that the expression of LC3 was positively correlated with Beclin-1 in colorectal cancer [19], which is still unclear in gastric cancer.
In this study, we investigated the expression of MAP1LC3B and Beclin-1 in gastric cancer and discuss their prognostic value as well as clinicopathologic features in gastric cancer patients. Furthermore, correlation analysis was conducted to explore the potential association between the expressions of MAP1LC3B and Beclin-1.
Materials and methods
Patients and tissue specimens
In this study, a series of 160 gastric cancer patients who had undergone gastrectomy in the Gastrointestinal Surgery Department of the First Affiliated Hospital of Sun Yat-sen University from 2007 to 2008 were recruited according to the following inclusion criteria: (1) had undergone gastrectomy and histologically proven adenocarcinoma of the stomach, (2) no preoperative chemotherapy and/or radiotherapy, and (3) no synchronous or metachronous cancers. Of the 160 patients with gastric cancer, there were 91 men and 69 women. The median age was 60 years (range 25–82). The tumor location was as follows: lower third in 47 (29.4 %) cases, middle third in 57 (35.6 %) cases, and upper third in 56 (35 %) cases. There were 105 (65.6 %) cases with lymph node metastasis (LNM) and 55 (34.4 %) cases without LMN. Among 160 patients, 61 (38.1 %) cases received standard postoperative chemotherapy according to the NCCN Gastric Cancer Guidelines. TNM stages were classified according to the American Joint Committee on Cancer guidelines and the criteria defined by the WHO International Histological Classification of Tumors [20]. Differentiated type was divided into two groups: (1) well-differentiated type, including papillary adenocarcinoma, well-differentiated tubular adenocarcinoma, and moderately differentiated tubular adenocarcinoma, and (2) poorly differentiated type, including solid-type poorly differentiated adenocarcinoma, nonsolid-type poorly differentiated adenocarcinoma, signet ring cell carcinoma, and mucinous adenocarcinoma [21]. Patient characteristics are provided in Table 1.
Follow-up
Follow-up data were obtained by phone, letter, and the outpatient clinical database. The median follow-up time was 54 months (range 2–80). All patients were subjected to close follow-up observation every 3 months for the first year, every 6 months for the second to fifth year, and every 12 months after 5 years. Demographic and clinical data were collected respectively.
Immunohistochemical staining of MAP1LC3B and Beclin-1
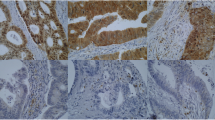
The expressions of MAP1LC3B and Beclin-1 in primary tumors (n = 160) were detected through immunohistochemistry (IHC) and then compared with that in adjacent noncancerous tissues (n = 34). A total of 160 primary gastric cancer tissues and 34 paraneoplastic normal tissues, which were more than 5 cm from the primary tumor sites, were obtained from the Pathology Department of the First Affiliated Hospital of Sun Yat-sen University. Then consecutive sections (4 mm thick) were cut from each block and used for immunohistochemical staining. The tissue sections were deparaffinized and rehydrated. Then, the sections were subjected to high temperature and high pressure in citrate buffer (pH 6.0) to retrieve antigenicity before being immersed in 3 % hydrogen peroxide to block endogenous peroxidase. The immunohistochemistry procedure was performed according to the manufacturer’s instructions. The sections were then incubated with a polyclonal primary antibody at 4 °C overnight. The primary detection antibodies, MAP1LC3B antibody (AP1802a, Abgent, Atlanta, USA) and anti-Beclin-1 (Sigma, USA), were used at a dilution of 1:200. After incubation with a HRP-conjugated sheep anti-rabbit secondary antibody (GTVision; Shanghai, China) and DAB, the slides were counterstained with Mayer’s hematoxylin. A known positive tissue sample (normal colonic mucosa slide) was used as a positive control. And PBS buffer was used to replace the primary antibodies in negative control staining.
Assessment of immunostaining
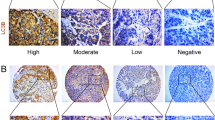
Immunostaining was evaluated twice by two pathologists who were blinded to patient outcomes and other clinical findings. The immunoreactivity of MAP1LC3B and Beclin-1 was evaluated according to the intensity and percentage of positively stained cells. At least five of the largest immunostained areas for each antibody were selected. Immunostaining intensity was rated as follows: 0, negative; 1, weak; 2, moderate; and 3, strong. The percentage of positively stained cells was graded as follows: grade 0, 0–5 %; grade 1, 6–25 %; grade 2, 26–50 %; grade 3, 51–75 %; and grade 4, 76–100 %. Immunoreactive score was calculated by adding the score of staining intensity and the percentage score of positively stained cells (0–7). Cases with an immunoreactive score of 0–3 were designated as low expression, and those with 4–7 were classified as high expression [22].
Statistical analysis
The statistical analysis was performed with the Statistical software package SPSS 13.0. To investigate, the associations between two categorical variables were evaluated using Fisher’s exact test or the chi-square test. Chi-square test was also used to evaluate the correlation of MAP1LC3B and Beclin-1. Overall survival (OS) was defined as the interval between surgery and death or the last follow-up. Relapse-free survival (RFS) time was defined as the time from curative surgery to recurrence, the date of last follow-up, or death. Receiver operating characteristic (ROC) curve analysis was conducted to compare the predictive value of MAP1LC3B staining with that of Beclin-1 staining for 5-year OS and 5-year RFS. Cumulative survival time was calculated using the Kaplan–Meier method and analyzed using the log-rank test. Cox’s proportional hazard regression model was used for analysis of independent prognostic factors.
Results
Expression and correlation of MAP1LC3B and Beclin-1 in gastric cancer tissues
The expressions of MAP1LC3B and Beclin-1 in gastric cancer tissues and adjacent tissues are shown in Figs. 1 and 2. Statistic results showed that the high expression rates of MAP1LC3B and Beclin-1 in cancer tissues were 57 % (92/160) and 72 % (115/160), respectively, which were lower than those in adjacent tissues (MAP1LC3B, 82 %, p = 0.007; Beclin-1, 88 %, p = 0.046; Table 2). Chi-square test showed that MAP1LC3B was positively associated with Beclin-1 in gastric cancer with a correlation coefficient of 0.446 (P < 0.001, Fig. 4d).
Association of MAP1LC3B and Beclin-1 expressions with clinicopathological parameters of gastric cancer
Table 3 shows the correlations between MAP1LC3B and Beclin-1 expressions and clinicopathologic characteristics, including age, gender, differentiation, LNM, tumor invasion, distant metastasis, tumor stage, tumor size, surgery type, tumor location, preoperative serum carcinoembryonic antigen (CEA), surgery, and adjuvant chemotherapy. As shown in Table 3, the expression of MAP1LC3B was associated with LNM, tumor invasion, and tumor stage (Table 3, all P < 0.05). The expression of Beclin-1 was also associated with LNM (Table 3, P = 0.03). From Table 4, we can see the high expression rates of MAP1LC3B and Beclin-1 in different tumor stages. A stepwise decrease trend in MAP1LC3B expression exists in tumor progression. Likewise, the expression of Beclin-1 gradually decreases in stage II, stage III, and stage IV.
Survival analysis
The total median OS time of 160 patients was 54 months. The median OS time of patients with high expression of MAP1LC3B in tumor tissues was 70 months, and with high expression of Beclin-1, it was 60 months, while the median OS time of patients with low expression of MAP1LC3B was only 23 months, and with low expression of Beclin-1, it was only 23 months. The 5-year OS rates of MAP1LC3B-high-expression patients were significantly higher than those of MAP1LC3B-low expression patients (60.9 vs. 30.9 %, p < 0.05). Similarly, the Beclin-1-high-expression patients have higher 5-year OS rates than Beclin-1-low expression patients (52.2 vs. 22.2 %, p < 0.05). Figure 3 shows that both the areas under ROC curve (AUC) of MAP1LC3B staining were bigger than those of Beclin-1 staining (Fig. 3a, 5-year OS 0.72 vs. 0.63; Fig. 3b, 5-year RFS 0.71 vs. 0.64), which indicated that MAP1LC3B staining has better predictive value for 5-year OS and 5-year RFS in gastric cancer patients than Beclin-1 staining.
ROC curve analysis of the expressions of MAP1LC3B and Beclin-1 for 5-year OS and 5-year RFS in gastric cancer patients. Both the areas under ROC curve (AUC) of MAP1LC3B staining were bigger than those of Beclin-1 staining, which indicated that MAP1LC3B staining has better predictive value for 5-year OS (a) and 5-year RFS (b) in gastric cancer patients
As shown in the Figs. 4 and 5, Kaplan–Meier survival analysis showed that patients with high expression of MAP1LC3B in primary tumor had significantly longer OS and RFS time than patients with low MAP1LC3B expression (Figs. 4a and 5a, P = 0.013, p < 0.01, respectively). Likewise, high Beclin-1 expression patients had a longer OS and RFS time (Figs. 4b and 5b, p = 0.015, p = 0.016, respectively) than patients with low expression of Beclin-1. High simultaneous MAP1LC3B and Beclin-1 expressions were associated with longer OS and RFS compared with low simultaneous MAP1LC3B and Beclin-1 expressions (Figs. 4c and 5c 56.77 vs. 24.42 months, p < 0.001; 53.56 vs. 22.33 months, p < 0.001, respectively).
Kaplan–Meier estimates of the probability of overall survival (OS) in gastric cancer patients. Patients with high expression of MAP1LC3B showed significantly longer OS than those with MAP1LC3B low expression (a). Patients with high expression of Beclin-1 showed significantly longer OS than those with Beclin-1 low expression (b). High simultaneous MAP1LC3B and Beclin-1 expression patients showed longer OS than low simultaneous MAP1LC3B and Beclin-1 expression patients (c). MAP1LC3B was positively associated with Beclin-1 in gastric cancer (d)
Kaplan–Meier estimates of the probability of relapse-free survival (RFS) in gastric cancer patients. Patients with high expression of MAP1LC3B showed significantly longer RFS than those with MAP1LC3B low expression (a). Patients with high expression of Beclin-1 showed significantly longer RFS than those with Beclin-1 low expression (b). High simultaneous MAP1LC3B and Beclin-1 expression patients showed longer RFS compared with low simultaneous MAP1LC3B and Beclin-1 expression patients (c)
In addition, univariate analyses revealed that OS significantly correlated with LMN, tumor invasion, distant metastasis, tumor stage, tumor size, surgery type, CEA, MAP1LC3B, and Beclin-1. RFS also significantly correlated with the factors mentioned above except surgery type (Table 5, all p < 0.05). Furthermore, multivariate analysis was used to evaluate all the statistically significant variables revealed in univariate analysis. The result displayed that MAP1LC3B, Beclin-1, and distant metastasis were independent prognostic factors for OS of gastric cancer patients (Table 5, p = 0.016, p = 0.041, p = 0.021, respectively), while MAP1LC3B, distant metastasis, and tumor invasion were independent prognostic factors for RFS of gastric cancer patients (Table 5, p = 0.022, p = 0.004, p = 0.042, respectively). Beclin-1 was not an independent factor for RFS of gastric cancer patients.
Discussion
Autophagy is an intracellular catabolic system in which excessive cytoplasmic organelles or cytosolic components reach lysosomes for elimination and recycling. An isolation membrane encapsulates minor portions of the cytoplasm, including soluble materials and organelles, to form the autophagosome. The autophagosome coalesces with the lysosome to become an autolysosome and resolve the internal substance [23]. Autophagy is thought to play a dual role in cancer, and its role in cancer is thought to be double edged. On one hand, autophagy system prohibits cell carcinogenesis in the initiation phase of tumorigenesis. On the other hand, autophagy could be used by cancer cells to counteract numerous chemotherapeutic interventions [24, 25]. Beclin-1 and LC3 genes play a crucial role in mammalian autophagy. Beclin-1 is involved in the signaling pathway that activates autophagy and in the initial step of autophagosome formation. MAP1LC3B comprises a soluble MAP1LC3B-I and a lapidated form called MAP1LC3B-II. MAP1LC3B-II is recruited into autophagosomes, which are considered to be a reliable marker of autophagy [26, 27]. On the basis of these results, we propose that MAP1LC3B and Beclin-1 play important roles in the development of gastric cancer and are prognostic factors for patients with gastric cancer.
In the present study, we investigated the expression and significance of MAP1LC3B and Beclin-1 in the tumorigenesis and development of gastric cancer. By using IHC, we found that their expression was significantly lower in the gastric cancer tissues than that in the counterpart normal tissues. Chi-square test showed that the expression of MAP1LC3B was positively correlated with the expression of Beclin-1. We also analyzed the association between protein expression and clinicopathological characteristics, including age, gender, differentiation, LNM, tumor invasion, distant metastasis, tumor stage, tumor size, surgery type, tumor location, preoperative serum CEA, surgery, and adjuvant chemotherapy. We found that both the expressions of MAP1LC3B and Beclin-1 were associated with LNM. Furthermore, the expression of MAP1LC3B was also associated with tumor invasion and tumor stage (Table 3). We also analyzed the expression level of MAP1LC3B and Beclin-1 in different stages (Table 4) and found a stepwise decrease trend in MAP1LC3B expression in tumor progression. Likewise, the expression of Beclin-1 gradually decreases in stage II, stage III, and stage IV. It means that the early-stage (stage I and stage II) tumor tissues show a higher autophagy activity than advanced stage tumor tissues, which indicates that autophagy plays different roles during tumor progression. In addition, we conducted the Kaplan–Meier method and Cox proportional hazard models for survival analysis, which revealed that MAP1LC3B-high-expression patients and Beclin-1-high-expression patients had significantly longer OS time and RFS time than patients with low expression of MAP1LC3B and patients with low expression of Beclin-1. High simultaneous MAP1LC3B and Beclin-1 expressions were associated with longer OS and RFS compared with low simultaneous MAP1LC3B and Beclin-1 expressions. Both MAP1LC3B and Beclin-1 were independent prognostic factors for OS of gastric cancer patients. Furthermore, the ROC curve analysis showed that the expression of MAP1LC3B has better predictive value for 5-year OS and 5-year RFS in gastric cancer patients than the expression of Beclin-1.
Currently, there are few well-documented reports about the levels of autophagy-related protein LC3 in gastric cancer, while LC3 had been widely studied in other cancers and the roles of LC3 in other cancers were proved to be varied [28–32]. Researches showed that LC3 expression was increased in hepatocellular carcinoma and colorectal cancer but decreased in prostate cancer, papillary thyroid cancer, and cervical squamous cell carcinoma. Patients with high expression of LC3 were proved to have worse survival in hepatocellular carcinoma and esophageal squamous cell carcinoma. Inversely, patients with high expression of LC3 were proved to have better survival in prostate cancer, ovarian cancer, colorectal cancer, and cervical squamous cell carcinoma [28–32]. These results indicated that the roles of LC3 in different cancers were complicated and varied. Our research showed that the expression of MAP1LC3B was significantly lower in the gastric cancer tissues than that in the counterpart normal tissues and low expression of MAP1LC3B predicted worse prognosis in gastric cancer patients. This indicated that autophagy may be downregulated in gastric cancer tissues, which contributed to poor prognosis. This finding may be explained by the basic function of autophagy in inhibiting cancer development by which damaged organelles are eradicated, thereby maintaining cell homeostasis by protecting normal cell growth or inducing caspase-independent autophagic cell death. In addition, we found that the expression of MAP1LC3B was inversely associated with LNM, tumor invasion, and tumor stage. These results indicated that autophagy may play important roles in the progress of gastric cancer. Inhibiting autophagy activity by downregulated MAP1LC3B in gastric cancer may promote LNM, tumor invasion, and tumor advancing. The underlying mechanisms are complex and remain to be fully characterized.
We proposed that the expression of Beclin-1 may correlate with MAP1LC3B in gastric cancer as both of them were reported to play crucial role in mammalian autophagy. Consisted with the previous studies, we found that the gastric cancer patients with high expression of Beclin-1 had a significant longer OS and RFS than patients with low expression of Beclin-1. However, our study found that Beclin-1 was downregulated in cancer tissues, which is different with other researches in gastric cancer [16–18]. Nevertheless, other studies have also demonstrated that Beclin-1 expression is frequently decreased in tumor cells, such as breast cancer, hepatocellular carcinoma, and cervical cancer, compared with that in normal cells [33–35]. This finding may be explained by the biphasic function of autophagy in cancer development. Moreover, we found that the expression of Beclin-1 was positively correlated with MAP1LC3B and both of them were adversely associated with LNM in gastric cancer. Survival analysis showed that simultaneous MAP1LC3B and Beclin-1 expressions were associated with longer OS and RFS compared with low simultaneous MAP1LC3B and Beclin-1 expressions. These can be explained by that Beclin-1 is an autophagy-related gene that can activate autophagy, whereas MAP1LC3B protein is incorporated into autophagosomes, and the protein expression level serves as a good indicator of autophagic activation. These results indicated that the prognostic value and clinical significance of MAP1LC3B in gastric cancer are associated with the expression of Beclin-1.
There are various regulators of autophagy, and the roles of autophagy in cancer are complicated and variety. Only assessing the expressions of MAP1LC3B and Beclin-1 cannot accurately reflect the autophagy. Meanwhile, simply by IHC, our study was not able to explore the mechanisms of MAP1LC3B and Beclin-1 in detail; further studies should be carried out to precisely understand the potential function of MAP1LC3B and Beclin-1 in human gastric cancer pathogenesis and which signaling pathway is involved in the tumorigenesis.
Conclusion
In the present study, we found that the expressions of MAP1LC3B and Beclin-1 were correlated and both of them were downregulated in gastric cancer. Gastric cancer patients with loss of MAP1LC3B and Beclin-1 were more likely to suffer LNM. Survival analysis indicated that both MAP1LC3B and Beclin-1 were independent prognostic factors for gastric cancer patients. Gastric cancer patients with lower expressions of MAP1LC3B and Beclin-1 were proved to have poor prognosis. Our findings suggested that the expressions of MAP1LC3B and Beclin-1 can be used as prognostic markers of patients with gastric cancer.
References
Fock KM. Review article: the epidemiology and prevention of gastric cancer. Aliment Pharmacol Ther. 40:250–60.
Karimi P, Islami F, Anandasabapathy S, et al. Gastric cancer: descriptive epidemiology, risk factors, screening, and prevention. Cancer Epidemiol Biomark Prev. 2014;23(5):700–13.
Siegel R, Ma J, Zou Z, Jemal A. Cancer statistics, 2014. CA Cancer J Clin. 2014;64:9–29.
Cunningham SC, Kamangar F, Kim MP, et al. Survival after gastric adenocarcinoma resection: eighteen-year experience at a single institution. J Gastrointest Surg. 2005;9:718–25.
Isobe Y, Nashimoto A, Akazawa K, et al. Gastric cancer treatment in Japan: 2008 annual report of the JGCA nationwide registry. Gastric Cancer. 2011;14:301–16.
Soerjomataram I, Lortet-Tieulent J, Parkin DM, et al. Global burden of cancer in 2008: a systematic analysis of disability-adjusted life-years in 12 world regions. Lancet. 2012;380:1840–50.
Rabinowitz JD, White E. Autophagy and metabolism. Science. 2010;330(6009):1344–8.
Guo JY, Xia B, White E. Autophagy-mediated tumor promotion. Cell. 2013;155(6):1216–9.
Yue Z, Jin S, Yang C, et al. Beclin 1, an autophagy gene essential for early embryonic development, is a haploinsufficient tumor suppressor. Proc Natl Acad Sci U S A. 2003;100:15077–82.
Qu X, Yu J, Bhagat G, et al. Promotion of tumorigenesis by heterozygous disruption of the beclin 1 autophagy gene. J Clin Invest. 2003;112:1809–20.
Kondo Y, Kanzawa T, Sawaya R, Kondo S. The role of autophagy in cancer development and response to therapy. Nat Rev Cancer. 2005;5:726–34.
Kuma A et al. The role of autophagy during the early neonatal starvation period. Nature. 2004;432(7020):1032–6.
Mizushima N, Ohsumi Y, Yoshimori T. Autophagosome formation in mammalian cells. Cell Struct Funct. 2002;27(6):421–9.
Ding WX et al. Linking of autophagy to ubiquitin-proteasome system is important for the regulation of endoplasmic reticulum stress and cell viability. Am J Pathol. 2007;171(2):513–24.
Iwata A et al. Increased susceptibility of cytoplasmic over nuclear polyglutamine aggregates to autophagic degradation. Proc Natl Acad Sci U S A. 2005;102(37):13135–40.
Yu M, Gou W-F, Zhao S, et al. Beclin 1 expression is an independent prognostic factor for gastric carcinomas. Tumor Biol. 2013;34:1071–83.
Chen Y-B, Hou J-H, Feng X-Y, et al. Decreased expression of Beclin 1 correlates with a metastatic phenotypic feature and adverse prognosis of gastric carcinomas. J Surg Oncol. 2012;105:542–7.
Zhou W-H, Tang F, Xu J, et al. Low expression of Beclin 1, associated with high Bcl-xL, predicts a malignant phenotype and poor prognosis of gastric cancer. Autophagy. 2012;8(3):389–400.
Wu S, Sun C, D T, et al. Expression and clinical significances of Beclin1, LC3 and mTOR in colorectal cancer. Int J Clin Exp Pathol. 2015;8(4):3882–91.
Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17:1471–4.
Wang Z, Cai SR, He YL, et al. Elevated PRL-3 expression was more frequently detected in the large primary gastric cancer and exhibits a poor prognostic impact on the patients. J Cancer Res Clin Oncol. 2009;135:1041–6.
Li G, Xu J, Wang Z, et al. Low expression of SOCS-1 and SOCS-3 is a poor prognostic indicator for gastric cancer patients. J Cancer Res Clin Oncol. 2015;141(3):443–52.
Tooze SA, Yoshimori T. The origin of the autophagosomal membrane. Nat Cell Biol. 2010;12:831–5.
Rouschop KM, Wouters BG. Regulation of autophagy through multiple independent hypoxic signaling pathways. Curr Mol Med. 2009;9:417–24.
Chen N, Karantza-Wadsworth V. Role and regulation of autophagy in cancer. Biochim Biophys Acta. 1793;2009:1516–23.
Dalby KN, Tekedereli I, Lopez-Berestein G, Ozpolat B. Targeting the prodeath and prosurvival functions of autophagy as novel therapeutic strategies in cancer. Autophagy. 2010;6(3):322–9.
Kimmelman AC. The dynamic nature of autophagy in cancer. Genes Dev. 2011;25(19):1999–2010.
Wu W-Y, Kim H, Zhang C-L, et al. Clinical significance of autophagic protein LC3 levels and its correlation with XIAP expression in hepatocellular carcinoma. Med Oncol. 2014;31:108.
Liu C, Xu P, Chen D, et al. Roles of autophagy-related genes Beclin-1 and LC3 in the development and progression of prostate cancer and benign prostatic hyperplasia. Biomedical Reports. 2013;1:855–60.
Chen Y, Li X, Wu X, et al. Autophagy-related proteins LC3 and Beclin-1 impact the efficacy of chemoradiation on esophageal squamous cell carcinoma. Pathology – Research and Practice. 2013;209:562–7.
Valente G, Morani F, Nicotra G, et al. Expression and clinical significance of the autophagy proteins BECLIN 1 and LC3 in ovarian cancer. Biomed Res Int. 2014;462658.
Hu Y-F, Lei X, Zhang H-Y, et al. Expressions and clinical significance of autophagy-related markers Beclin1, LC3, and EGFR in human cervical squamous cell carcinoma. OncoTargets and Therapy. 2015;8:2243–9.
Daniel F, Legrand A, Pessayre D, et al. Beclin-1 mRNA strongly correlates with Bcl-XL mRNA expression in human hepatocellular carcinoma. Cancer Investig. 2007;25(4):226–31.
Wang ZH, Xu L, Duan ZL, et al. Beclin 1-mediated macroautophagy involves regulation of caspase-9 expression in cervical cancer HeLa cells. Gynecol Oncol. 2007;107(1):107–13.
Dong M, Wan XB, Yuan ZY, et al. Low expression of Beclin 1 and elevated expression of HIF-1α refine distant metastasis risk and predict poor prognosis of ER-positive, HRE2-negative breast cancer. Med Oncol. 2013;30(1):355.
Acknowledgments
This work was supported by Doctoral Fund of Ministry of Education of China (Grant No. 20100171120065).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Additional information
Shuangjin Yu and Guanghua Li have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Yu, S., Li, G., Wang, Z. et al. Low expression of MAP1LC3B, associated with low Beclin-1, predicts lymph node metastasis and poor prognosis of gastric cancer. Tumor Biol. 37, 15007–15017 (2016). https://doi.org/10.1007/s13277-016-5383-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-016-5383-5