Abstract
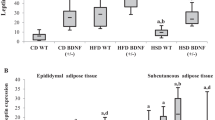
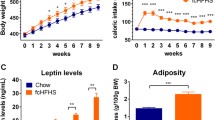
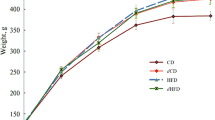
We investigated leptin expression in four fat tissues located in different body parts of male mice. In the absence of caloric restriction (CR), the highest relative levels of leptin mRNA were detected in epididymal fat (EF). Lateral abdominal fat (LAF) and subcutaneous fat (SF) had intermediate leptin expression levels, while the lowest leptin mRNA content was revealed in brown adipose tissue (BAT). We also investigated differential regulation of leptin expression in these adipose tissues following diets that were based on various levels of CR for 10 weeks. The largest reduction of leptin mRNA levels by CR was observed in adipose tissue where maximal leptin mRNA expression was detected in the absence of CR. Leptin protein expression was also decreased by CR as revealed by immunohistochemistry. The changes in leptin mRNA and protein expression positively correlated with adipocyte size that was also diminished with the increase in the strength of CR.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Fain, J. N., Madan, A. K., Hiler, M. L., Cheema, P. & Bahouth, S. W. Comparison of the release of adipokines by adipose tissue, adipose tissue matrix, and adipocytes from visceral and subcutaneous abdominal adipose tissues of obese humans. Endocrinology 145: 2273–2282 (2004).
Zhang, Y. et al. Positional cloning of the mouse obese gene and its human homologue. Nature 372: 425–432 (1994).
Halaas, J. L. et al. Weight-reducing effects of the plasma protein encoded by the obese gene. Science 269: 543–546 (1995).
Singh, P. et al. Leptin signaling in adipose tissue: Role in lipid accumulation and weight gain. Circ Res 111: 599–603 (2012).
Wang, M. Y., Lee, Y. & Unger, R. H. Novel form of lipolysis induced by leptin. J Biol Chem 274: 17541–17544 (1999).
Shimomura, I., Hammer, R. E., Ikemoto, S., Brown, M. S. & Goldstein, J. L. Leptin reversesinsulin resistance and diabetes mellitus in mice with congenital lipodystrophy. Nature 401: 73–76 (1999).
da Silva, A. A., Kuo, J. J. & Hall, J. E. Role of hypothalamic melanocortin 3/4-receptors in mediating chronic cardiovascular, renal, and metabolic actions of leptin. Hypertension 43: 1312–1317 (2004).
Weindruch, R. The retardation of aging by caloric restriction: studies in rodents and primates. Toxicol Pathol 24: 742–745 (1996).
Okita, N. et al. Differential responses of white adipose tissue and brown adipose tissue to caloric restriction in rats. Mech Ageing Dev 133: 255–266 (2012).
Shimokawa, I. & Higami, Y. Leptin signaling and aging: insight from caloric restriction. Mech Ageing Dev 122: 1511–1519 (2001).
Cinti, S. The adipose organ at a glance. Dis Model Mech 5: 588–594 (2012).
Fox, C. S. et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation 116: 39–48 (2007).
Hansen, I. R., Jansson, K. M., Cannon, B. & Nedergaard, J. Contrasting effects of cold acclimation versus obesogenic diets on chemerin gene expression in brown and brite adipose tissues. Biochim Biophys Acta 1841: 1691–1699 (2014).
Bjorndal, B., Burri, L., Staalesen, V., Skorve, J. & Berge, R. K. Different adipose depots: Their role in the development of metabolic syndrome and mitochondrial response to hypolipidemic agents. J Obes 2011:490650, 15 pages (2011).
Kim, S. S. et al. Whole-transcriptome analysis of mouse adipose tissue in response to short-term caloric restriction. Mol Genet Genomics 291: 831–847 (2016).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Hong, SJ., Kim, SS., Lim, W. et al. Leptin is a dose-dependent marker of caloric restriction in adipose tissues located in different parts of the mouse body. Mol. Cell. Toxicol. 14, 53–59 (2018). https://doi.org/10.1007/s13273-018-0007-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-018-0007-3