Abstract
Oligosaccharides are low molecular weight carbohydrates with a wide range of health benefits due to their excellent bio-preservative and prebiotic properties. The popularity of functional oligosaccharides among modern consumers has resulted in impressive market demand. Organoleptic and prebiotic properties of starch-derived oligosaccharides are advantageous to food quality and health. The extensive health benefits of oligosaccharides offered their applications in the food, pharmaceuticals, and cosmetic industry. Maltooligosaccharides and isomaltooligosaccharides comprise 2–10 glucose units linked by α-1-4 and α-1-6 glycoside bonds, respectively. Conventional biocatalyst-based oligosaccharides processes are often multi-steps, consisting of starch gelatinization, hydrolysis and transglycosylation. With higher production costs and processing times, the current demand cannot meet on a large-scale production. As a result, innovative and efficient production technology for oligosaccharides synthesis holds paramount importance. Malto-oligosaccharide forming amylase (EC 3.2.1.133) is one of the key enzymes with a dual catalytic function used to produce oligosaccharides. Interestingly, Malto-oligosaccharide forming amylase catalyzes glycosidic bond for its transglycosylation to its inheritance hydrolysis and alternative biocatalyst to the multistep technology. Genetic engineering and reaction optimization enhances the production of oligosaccharides. The development of innovative and cost-effective technologies at competitive prices becomes a national priority.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Oligosaccharides (OS) are low molecular weight carbohydrates with a degree of polymerization (DP) ranges of 2–10 sugar moieties. Among other classes of OS such as xylooligosaccharide (XOS), fructooligosaccharides (FOS), and galactooligosaccharides (GOS), starch-related OS is one of the important class functional OS. They offer multiple advantages to the food product in light of their bio-preservative and prebiotic characteristic. Starch-related OS enhance rheological characteristics including the taste and texture of various food items (Qiang et al. 2009). Further, they have been classified into Maltooligosacharide (MOS) and Isomaltooligosaccharides (IMOS) depending on the type of bond linkage. MOS are comprised of α- D-1-4 glycosidic linkage, whereas IMOS contain additional α- D-1-6 or D-1-2 or D-1-3 to existing D-1-4 linkage. Starch related OS have been acknowledged as Generally Recognized As Safe (GRAS) (Ibrahim 2018). Investigations on animal models have claimed the ability of OS to lessen the event of life-threatening diseases such as irritable bowel syndrome (IBS), diabetes, obesity, and colon cancer (Kootte et al. 2012; Chong et al. 2019). As a result, they have earned a lot of interest owing to their health-promoting effects in human, poultry, and ruminant animals. Additionally, they provide benefits to the consumers by improving the fibre content in the diet. Likewise, starch related OS represents a potential and attractive alternative to antibiotics to promote growth and feed efficacy use in poultry animals, and weaned pigs (Gadde et al. 2017).
Consequently, increased demand for OS globally expanded the market size. Functional food ingredients are projected to reach USD 94.21 billion by 2023.Similarly for prebiotics it is likely to grow at a CAGR of 8.5% with an estimated USD 8.04 Billion by 2026. Approximately 20.3% of yogurts manufactured contain prebiotic ingredients in the Asia Pacific region. About 10.2% highest growth rates in in the Asia Pacific region, for the healthcare and pharmaceutical industries. Consequently, OS become a prominent market in the pharma and food industries. However, the challenges associated with the purification of OS using chemical synthesis lead to exceedingly high production costs to obtain pure OS in bulk amounts. Thus, significant research is being extended to discover and characterize potent biocatalysts for OS production on an industrial scale.
The synthesis of OS is important and can be achieved by extraction, chemical, and enzymatic synthesis. However, the first two methods are limited by lower yield, and demands for heavy metal catalyst (Ibrahim 2018). In contrast, enzymatic amalgamation is a mild, cost-effective, and eco-friendly. Then, the identification of an efficient biocatalyst at industrial scale is till challenging.
In this context, MFAse is a special class of enzyme that hydrolyzes the starch and produces MOS with different DP depending on its type and source. Besides, α-glucosidase and other member of GH family with transglycosylation catalysis are involved in the OS production (Abdul Manas et al. 2018). Progressively increased demands have led to the search of novel, efficient and cost-effective biocatalysts. The present review is focused on MFAses, with highlighting on different strategies to enhance transglycosylation. Furthermore, future potential alternatives and efficient technology for synthesizing the complex OS have been discussed in the review.
Physicochemical and biological properties of OS
Maltooligosaccharide
Maltooligosaccharide (MOS) comprise 2–10 glucose moiety linked via α-D-1-4 glycoside bond (Fig. 1), including maltose, maltotriose, maltotetrose, maltopentose, maltohexose, and maltoheptaose. All types of MOS share common properties such as mild sweetness (60% compared sweet to sucrose), heat resistance, low calorie, low osmolality, high solubility, non-hygroscopic, and moderate viscosity than mono and disaccharides (Ji et al. 2019). Furthermore, heat-processed food prefers heat resistance MOS to avoid browning by Maillard reactions (Kroh et al. 2002). MOS determines the sensory characteristics of beer and other beverages as their contents are responsible for beer viscosity and stability (Li et al. 2020). Low osmolality of MOS makes it suitable for consumptions as it does not create any discomfort. They prevent microbial contaminants, due to their lower water activity in food items. Above and beyond, MOS with high DP act as substrate to intestinal α-glucosidase and provide steady and continuous energy. Especially, MOS act a potential laxative to relieve constipation as it improves mucosal thickness, mucus secretion and Cajal cell (Jang et al. 2020). OS such as maltoheptaose have a strong film-forming ability and are used as saccharide based representative for block polymers (Ji et al. 2019). Different types of OS are commercially available in the form of syrup and powder, and exhibited various physicochemical and biological properties (Table 1).
Isomaltooligosaccharide
Isomaltooligosaccharide (IMOS) are non-digestible carbohydrates that consist of additional α-D-1–6 linkage. It includes isomaltose, isomaltotriose, isomaltotetrose, panose, isopanose (Fig. 1). Moreover, the definition of IMSO has been extended to α-D (1-3), nigero-oligosaccharides, or α-D (1-2), kojo-oligosaccharides. Structurally IMOS is characterized by the DP, types of linkage, and monosaccharide, and disaccharide sugar donor (Obama 2018). MOS and IMOS escape the digestive process in the gut and metabolized by intestinal microbes, thereby; promote the growth of beneficial bacteria and suppressing pathogen. The degradation of OS by microbes produces short-chain fatty acid (SCFA) which, improves intestinal function such as mineral absorption, pathogen exclusion, improved epithelial barrier function, glucose homeostasis, immune-modulation, and results in systemic effects through interaction with the specific receptor proteins (de Paulo et al. 2019; Jang et al. 2020). Similarly, a study conducted on rats showed isomaltodextrin increase colonic H2 production, protecting inflammation induced oxidative stress. Oxidative stress further cause progression of metabolic syndrome (Nishimura et al. 2016).
Malto-oligosaccharide forming amylases (MFAses)
The glycoside hydrolase (GH), one of the largest enzyme families involved in the synthesizing OS. It consists of 80,000 sequences and approximately 30 diverse enzyme specificities. Further the GHs family characterizes a clan GH-H of three GH families GH13, GH70, and GH77. Hydrolysis, transglycosylation and, isomerization types of mechanism have been reported among them. MFAses is also one of the member of GH family, comprises maltotriose forming amylase (EC 3.2.1.116), maltotetrose forming amylase (EC 3.2.1.60), maltopentose forming amylase (EC 3.2.1.-), and maltohexose forming amylase (EC 3.2.1.98) etc., collectively known as MFAses. They have been reported to produce MOS ranges 2–10 DP. Also, they are of two types, one type producing a single MOS as a prominent product, while the other type forms a mixture of MOS (Pan et al. 2017). However, MFAse has been reported for its endo-, exo-hydrolytic and multiple attack patterns containing both endo- and exo-type mode of action on a substrate including starch and maltodextrins. Diverse groups of bacteria and fungi were reported to produce a variety of MFAse, across the globe (Table 2). Till today, few advanced studies on structure-functional relations are available. Henceforth, researchers need to explore novel MFAse and its in depth understanding for its applications.
Structure of MFAse
As a member of the GH family, MFAse also has similar kinds of active centres, catalytic, and substrate binding residues. Most of MFAses show similarity with N-terminus amino acids compare to C-terminal with α amylases. Besides, they have been reported to show relatively low homology with some exceptions. In addition to three A, B and C domain α- amylases and related member, MFAses are reported for the presence of CBM 20 at C-terminal of a polypeptide (Pan et al. 2017).
Several studies have investigated on 3-Dstructure of MFAses to investigate thir function. However, in depth information is not available on product specificity. Moreover, it further limits the preparation of desired MOS at large scale. Thus, MFAse has raised the need of understanding of how structure contributes to the specificity (Ali et al. 2001). X-ray crystal structure of G4-forming amylase and its interaction with maltotetrose reported by Pseudomonas saccharophila STB0 revealed a comparatively distinct difference in loop structure of active cleft at non reducing end among other amylases and MFAse of P. saccharophila STB0. Besides, it showed that substrate occupies −4 and −1 subsite at non reducing end governs maltotetrose specificity for the MFAse (Zhang et al. 2020). However, very few reported MFAse to have commercial applications as they shown low thermosatbility. Nowadays, starch processing industry demands for the thermotolerant enzymes. The bioconversion reactions at higher temperature reduce contaminations and accelerate the reaction. Consequently, the exploitation of thermostable enzymes from extreme habitat is gaining a lot of attention (Aquino et al. 2003; Ji et al. 2019). Streptomyces species are one of the significant saprophytic bacteria, is known for industrially important hydrolytic enzymes, and also reported for different types of MFAse from extreme habitat with thermostability, alkali tolerant, and Ca-independent etc. Besides, they have been shown for the presence of beneficial CBM 20 domain, it acts as ancillary modules, to degrade raw starch and thus play as advantageous property in reduction of cost in the biotechnological industry (Shivlata et al. 2017).
MFAse produced by Streptomyces has been reported for the transglycosylation and the hydrolytic mode of action and attracted wide range of applications and could be consider a potential alternative biocatalyst for the production of OS (Lakshmi et al. 2020; Sidar et al. 2020).
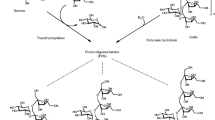
Recently, very few MFAse have been reported for their specific transglycosylation activity. Transglycosylation is a reverse mechanism to hydrolysis, where other than water molecules (alcohol, phenol) donate hydroxyl group to the acceptor molecule (Abdul Manas et al. 2018) (Fig. 2). Currently, this phenomenon is used by the GH family members. Some MFAses have been reported for G3 amylase activity among Microbacterium imperial, B. lehensis, and S. griseus for its transglycosylation to produce long MOS with their own hydrolyzed product (Pan et al. 2019) (Table 3). In this review, we have focused on enzyme based technology, transglycosylation mechanism of MFAse and its future potential use in the commercial production of the complex OS. Hence, development of such innovative enzymes based technology at competitive prices assumes national importance.
Enzyme based technology for the synthesis of MOS and IMOS
Glycosyltransferase (EC. 2.4) and GH (EC. 3.2) have been reported for transglycosylation mechanisms (Díez-Municio et al. 2014). However, the former limits its applications as it lacks flexibility for a variety of substrates and also demands expensive activated sugar nucleotides. Whereas GHs has offer characteristics like, dual hydrolysis, and transglycosylation activity that are attractive on the industrial front. Several enzymes including α amylases α-β glucosidase, dextransucrase and 4, 6- α -Glucotransferase from the GH family’s including GH 31, GH 13, GH 70, and GH57 with transglycosylation activity have been reported for IMOS synthesis. Therefore, GH is currently used preferred biocatalyst at the commercial point (Table 2). The composition of MOS through conventional starch hydrolysis is entirely dependent on reaction conditions (pH, temp), substrate specificity and hydrolytic enzyme. However, discovery of MFAses produce specific MOS in the range of 2–10 DP opened an avenue manufacture particular type of saccharides with high yield. Commercial production of MOS is a dual-step process that involves, starch liquefaction and saccharification followed by purifications. Traditional processes takes place in amylases and high temperature, MFAse further specifically hydrolyzes liquefied starch slurry partially to produce MOS.
An additional debranching enzyme, pullulanase, is used to break down α 1–6 glycosidic linkage of amylopectin to increase the efficiency and yield. MFAses are a prerequisite for the production; therefore significant efforts have been made towards discovery, and characterizations. Apart from MFAse, other starch-related OS such as trehalose and cyclodextrin are synthesized by Maltooligosyltrehalose synthase (MTSase) or Maltooligosyltrehalose trehalohydrolase (MTHase) and cyclodextrin glycosyltransferase (CGTase) (Ibrahim 2018). IMOS synthesis or complex MOS at a commercial scale requires, additional steps i.e. transglycosylation. It involves sequential multi-step enzymatic processes. The liquefaction of starch is followed by saccharification with α-amylase, maltogenic amylase, pullulanase, and further transglycosidase (Niu et al. 2017; Sorndech et al. 2018). Various researchers have recently applied an alternative approach consisting of simultaneous saccharification and transglycosylation mechanism to shorten process time and avoid the inhibitory effect of glucose (Basu et al. 2016).
Strategies involved in transglycosylation activity
Simultaneous hydrolysis, and transglycosylation at the same active site, is relatively challenging. Moreover, higher-concentration of acceptor shifts the reaction into transglycosylation regardless of thermodynamically more ideal hydrolysis. Maximum yield could be achievable with optimal experiment conditions. Various strategies have been reported to improve the transglycosylation such as a) elevation of substrate molecule b) the reduction of water availability, with replacement of organic co-solvents c) use of an acceptor molecule other than water in the reaction medium, and d) physical parameter such as high temperatures, etc. (Bissaro et al. 2015).
Reaction equilibrium
The accessibility of water highly influences the equilibrium shift toward hydrolysis of transglycosylation. Therefore, under ideal circumstances, a presence of nucleophiles (other than water) shifts the reaction towards transglycosylation and thus forms a new glycosyl linkage. Removal of water can be achieved by reverse hydrolysis in two ways; the adding a high concentration of an acceptor molecule and incorporating the organic solvent in the reaction mixture. Few studies showed improved production of OS with higher substrate concentration by overturning hydrolytic activity at low water availability. As a consequence, longer OS accumulated as a product and prevent re-hydrolysis (Manas et al. 2014).
Protein engineering
Protein engineering is a valuable tool to improve the yield and specificity of the desired product. Protein engineering ranges from rational modifications to specifically targeted designing to produce a precise and high yield of OS. Therefore, protein engineering of the MFAse is considered a potential tool to enhance the product specificity. These strategies are based on loop engineering, targeting conserved residues and rational designing, which further includes modification of subsite, acceptor binding site and destabilising catalytic binding of water molecule (Zeuner et al. 2019). These improvements can be accomplished by site-directed mutagenesis with in-depth understanding of the structural–functional relationship of enzymes. Several efforts have been made for the development of modified MFAse and related enzyme.
This has prompted researchers to reveal X-ray crystal structures of available MFAse to better understand the enzyme’s mechanism. Interactions of MFAse with its substrate in its 3-D structure reveal the nature and involvement of amino acids in the product specificity for OS synthesis. Furthermore, tailored approaches aid in reducing the synthesis of the mixed OS into desired OS. This lowers the cost of purification and the overall cost of production. As a result, site directed mutagenesis has the potential to be cost effective. MFAse of B. stearothermophilus STB04 drastically boosted production of maltopentose (G5) over maltohexose (G6) when compared to wild MFAse by replacing Gly109 with aspartic acid or phenylalanine (Xie et al. 2019).
Several studies have documented on substitution of aromatic amino residue at active sites among starch and other polysaccharide degrading enzymes includes, α amylase, α –β glucosidase, dextransucrase, and maltogenic amylases. Site directed mutagenesis study revealed that Trp324 and Asn 694 of Schwannionmyces occidentalis and A. niger involved in the transglycosylation.
The product profile of isomaltose was modified to isomaltotriose after site directed mutagenesis of Asn 694 of A. niger with alanine, leucine, and phenylalanine (Casa-villegas. 2018).
Applications
Oligosaccharides has received a wide range of deliberation, due to their functional dietary properties. As a result, they are highly recommended in various food formulations. Other applications of MOS include the measurement the glucose level in clinical chemistry (Ibrahim 2018). High purity and higher DP of OS such as maltoheptaose has specific applications in the diagnostics to recognize protein and cell marker. OS has been employed in the healthcare products such as dental caries, relieves fatigue, alleviate stress, and boosts the peristalsis function of the intestine, and avoids constipation (Abou Hachem et al. 2013). Also have been used in cosmetics, lipsticks, oral refreshing agents toiletries.
Bio preservative and functional food
Oligosaccharides are used as a preservative, cryoprotec-tants, stabilizer and a variety of other properties. Negative effect of retrogradation gives firmness and staling of starchy baked food items. Consequently, anti-staling agents are a pre-requisite to prevent losses in the food industry. Several MFAse used for inhibition of bread firming via the production of MOS viz., maltotetrose (G4), G5, and other dextrins they lower the water activity. Commercially available anti-stealing agent Powerfresh of DuPont Nutrition and Health and optimal 4G was reported to improve dough rheological properties, food quality, and baking performance of whole-grain flour. Therefore, OS have been used in confectionery, bread, and bakery, beverages, infant milk powders, maltose syrup, and brown rice syrup, sports drinks for athletes, jam, jelly, wines, lower mellow beers and various other applications of OS worldwide (Fig. 3).
Prebiotics and dietary fibers
Prebiotics are non-digestible food components, which confer a medical advantage to the host by modulating “gut micro-flora”. Dietary fibres and carbohydrate with 3–9 DP have a positive impact on health. Prebiotic food ingredients are important and play a crucial role as they are non-digestive to gut enzymes and resist gastrointestinal assimilation while modulating the microbiota. Species of Bifidobacterium, Lactobacillus and Faecalibacterium further stimulate the production of bacteriocins and lactic acid, aid in the digestion of lactose, produce B vitamins synthesis (Fig. 4). The fermentation of prebiotics by micro-flora produces SCFAs, which act as immune-modulator (Lawrence et al. 2017).
Alternative sweetener
Sweetness is an important feature of food and it imparts rheological properties. Glucose and sucrose are leading to obesity and linked to diabetes complications due to its high GI. The added sugar content in ccommercially available foods including rice pudding, cake, fortified yogurt, and fruit juices added sugar content in the range 13–50%. Sugar consumption is contraindicated in type II diabetes and metabolically challenged populations. The FDA recently mandated the addition of “added sugars” in the commercial products.
Sugar should account for 5% of total energy intake, according to the WHO (Erickson et al. 2000). Alcohol or polyols are the alternative sugars that are limited for use in patients with IBS due to its gastrointestinal intolerance. Similarly, in comparison to IMOS, FOS is unsuitable since it produces digestive intolerance and acidic instability in the gut (Gourineni et al. 2018). The foremost study demonstrated gastrointestinal tolerance and the low post-prandial glycaemic response of IMOS and approval of partial or full replacement of sugar with IMOS in food products. A significant study in high-fat diet-induced rats showed prevention of systemic and tissue inflammation on co-supplementation of cranberry products with IMOS (Singh et al. 2018).
Anti-diabetes, anti-obese and anti-cancerous role
The global prevalence of type 2 diabetes, cardiovascular illnesses, and other diseases is due to lifestyle and nutritional habits. Owing to the side effects of anti-diabetic medications, there is the quest for an alternative. Animal studies showed IMOS reduced the postprandial blood glucose and insulin responses in diabetes mellitus patients. OS has involved in an ant-diabetic activity such as α‐glucosidase inhibition, enhancement of pancreatic function relieve insulin and resistance and anti‐inflammatory effects though SCFA mediated mechanisms regulation of gut microbiota and hormones (Zhu et al. 2019). SCFA regulate the expression of fasting-induced adipose factor (FIAF) known as a satiety hormone. Consequently, the gut microbiota is one of the most effective therapies in weight treatment (Andersson et al., 2010). The high fat-instigated diet has been accounted for to build LPS containing gut microbiota and consequently down-regulate the Bifidobacteria which results in metabolic endotoxemia achieves insulin obstruction and weight gain (Kootte et al. 2012). IBS is also an emerging functional disorder, and patients with this disorder showed a decline production of butyrate-producing bacteria, a diminished amount of Bifidobacterium and other beneficial bacteria. As a result, developing prebiotic and or probiotic to the IBS targeted micro-biome is one of the therapeutic approach (Chong et al. 2019). The predictable association between prebiotics, gut microbiota, colonocytes has been shown in Fig. 5.
Alternatives to antibiotics
Antimicrobial resistance (AMR) is rapidly becoming one of the world's most critical public health challenges. The poultry industry is one of the important public food sectors dealing with the detrimental impacts of AMR. To combat this, situation prebiotics has been showcased as a promising alternative to antibiotics. It offers multiple health benefits, including immune-modulatory effects, positive changes in intestine morphology, improvement in productivity parameters (egg production, feed conversion ratio, increased body weight), and prevention of pathogenic bacteria include as Clostridium perfringens, Campylobacter spp, Escherichia coli, Staphylococcus aureus or Salmonella spp. (Mookiah et al. 2014). Thitaram et al. (2005) conducted a study on 3-week old chicken, fed with 1% IMOS in presence of antibiotic-resistant Salmonella typhimurium Nalr strain. The chicken showed good growth and health via enrichment of the cecal population with beneficial Bifidobacteria.
Future trends
Besides its nutraceuticals and pharmaceuticals applications, OS with prebiotic properties has been implemented in cosmetics and agriculture sector. Undesirable skin conditions i.e. inflammation caused by skin bacteria have reported to controlled by prebiotics applications with a 91% success rate in the human trial (Patel et al. 2012). Similarly, their incorporation range can be extended into other tropical products such as toilet paper etc. Prebiotic is conceptualized to modulate microbial communities for their health-promoting effects. This further leads to its wide scope on mass applications as microbiome targeted intervention for significant change in a public area (Cunningham et al. 2021). In combination with probiotics, OS have been reported for clinical trials concerning aging, bariatric surgery, obesity, cancer, and other conditions and have shown substantial results. This indicates future scope in investing in a variety of novel prebiotics for their wider benefits. Additionally, this has resulted in an increased patent and huge investment in the market sector as well. The mode of actions of prebiotic is complex and diverse and the action of prebiotics is structure specific. Therefore it demands a deeper understanding of structure and function mechanism. This can be further achieved through genetic modification and in silico docking. Furthermore, glycol-conjugates are a modern prerequisite for nano-carrier design. Therefore, investing in a novel biocatalyst and newer technology holds the potential in the future. An efficient technology can be applied to the synthesis of a related class of OS such as FOS, XOS, and GOS and among others. In the future, OS will likely be isolated from novel sources, and more advances and inventions in techniques to reduce the cost of current OS for its large-scale applications. Moreover, unique and novel combinations of OS of the optimized mixture may offer an interesting profile of benefits among consumers.
Conclusion
With remarkable physicochemical properties and post-prebiotic impact, starch-related OS have widespread use in food and pharmaceutical formulations. As well, these fermentable fibres help to preserve intestinal homeostasis and reduce metabolic disorders. Despite its significant and future potential, OS production is challenging due to higher production costs and increased demand. However, present OS production technology involves a multistep process which has led to high cost and time. Few reported on MFAses offer a dual mechanism of hydrolysis and transglycosylation in the reaction cascade. Henceforth, MFAse can be exploited for single pot biocatalyst reaction rather than the existing multi-sequential method for OS production. Numerous studies have suggested, addition linkage to OS may contribute to extra health benefits. Hence, it is important to synthesize wide range of OS with novel structures, and to study their biocompatibility and potential. Furthermore, it holds the potential for an alternative and viable technology to ensure the national economy.
Abbreviations
- MOS:
-
Malto-oligosaccharides
- IMOS:
-
Isomalto-oligosaccharides
- MFAse:
-
Malto-oligosaccharide forming amylases
- GH:
-
Glycosyl hydrolase
- DP:
-
Degree of polymerisation
- FOX:
-
Fructo-oligosaccharide
- XOS:
-
Xylo-oligosaccharides
- GOS:
-
Galacto-oligosaccharides
- G3:
-
Maltotriose
- G4:
-
Maltotetrose
- G5:
-
Maltopentose
- G6:
-
Maltohexose
References
Abdul Manas NH, Md. Illias R Mahadi NM (2018) Strategy in manipulating transglycosylation activity of glycosyl hydrolase for oligosaccharide production. Crit Rev Biotechnol 38(2):272–293. https://doi.org/10.1080/07388551.2017.1339664
Abou Hachem M, Andersen JM, Barrangou R, Møller MS, Fredslund F, Majumder A, Ejby M, Lahtinen SJ, Jacobsen S, Lo Leggio L, Goh YJ (2013) Recent insight into oligosaccharide uptake and metabolism in probiotic bacteria. Biocatal Biotransformation 31(4):226–235. https://doi.org/10.3109/10242422.2013.828048
Ali MB, Mhiri S, Mezghani M, Bejar S (2001). Purification and sequence analysis of the atypical maltohexaose-forming α-amylase of the B. stearothermophilus US100. Enzyme Microb Technol 28(6): 537–542. https://doi.org/10.1016/S0141-0229(01)00294-0
Andersson U, Bränning C, Ahrné S, Molin G, Alenfall J, Önning G, Nyman M, Holm CJ (2010) Probiotics lower plasma glucose in the high-fat fed C57BL/6J mouse. Benef Microbes 1(2):189–196. https://doi.org/10.3920/BM2009.0036
Aquino AC, Jorge JA, Terenzi HF, Polizeli ML (2003) Studies on a thermostable α-amylase from the thermophilic fungus Scytalidium thermophilum. Appl Microbiol Biotechnol 61(4):323–328. https://doi.org/10.1007/s00253-003-1290-y
Bai Y, Gangoiti J, Dijkstra BW, Dijkhuizen L, Pijning T (2017) Crystal structure of 4, 6-α-glucanotransferases supports diet-driven evolution of GH70 enzymes from α-amylases in oral bacteria. Structure 25(2):231–242. https://doi.org/10.1016/j.str.2016.11.023
Basu A, Mutturi S, Prapulla SG (2016) Production of isomaltooligosaccharides (IMO) using simultaneous saccharification and transglycosylation from starch and sustainable sources. Proc Biochem 51(10):1464–1471. https://doi.org/10.1016/j.procbio.2016.06.008
Bissaro B, Monsan P, Fauré R, O’Donohue MJ (2015) Glycosynthesis in a waterworld: new insight into the molecular basis of transglycosylation in retaining glycoside hydrolases. Biochem J 467(1):17–35. https://doi.org/10.1042/BJ20141412
Casa-Villegas M, Marín-Navarro J, Polaina J (2018) Amylases and related glycoside hydrolases with transglycosylation activity used for the production of isomaltooligosaccharides. Amylase 2(1):17–29. https://doi.org/10.1515/amylase-2018-0003
Champreda V, Kanokratana P, Sriprang R, Tanapongpipat S, & Eurwilaichitr L (2007). Purification, biochemical characterization, and gene cloning of a new extracellular thermotolerant and glucose tolerant maltooligosaccharide-forming α-amylase from an endophytic ascomycete Fusicoccum sp. BCC4124. Biosci Biotechnol Biochem 71(8), 2010–2020. https://doi.org/10.1271/bbb.70198
Chong PP, Chin VK, Looi CY, Wong WF, Madhavan P, Yong VC (2019) The microbiome and irritable bowel syndrome–a review on the pathophysiology, current research and future therapy. Front Microbio 10:1136. https://doi.org/10.3389/fmicb.2019.01136
Cunningham M, Azcarate-Peril MA, Barnard A, Benoit V, Grimaldi R, Guyonnet D, Gibson GR (2021) Shaping the future of probiotics and prebiotics. Trends Microbio 29(8):667–685. https://doi.org/10.1016/j.tim.2021.01.003
de Paulo FD, de Araújo FF, Neri-Numa IA, Pastore GM (2019) Prebiotics: trends in food, health and technological applications. Trends Food Sci Technol 93:23–35. https://doi.org/10.1016/j.tifs.2019.09.004
Díez-Municio M, Herrero M, Olano A, Moreno FJ (2014) Synthesis of novel bioactive lactose-derived oligosaccharides by microbial glycoside hydrolases. Microb Biotechnol 7(4):315–331. https://doi.org/10.1111/1751-7915.12124
Erickson KL, Hubbard NE (2000) Probiotic immunomodulation in health and disease. J Nutr 130(2):403S-409S. https://doi.org/10.1093/jn/130.2.403S
Gadde U, Kim WH, Oh ST, Lillehoj HS (2017) Alternatives to antibiotics for maximizing growth performance and feed efficiency in poultry: a review. Anim Health Res Rev 18(1):26–45. https://doi.org/10.1017/S1466252316000207
Gourineni V, Stewart ML, Icoz D, Zimmer JP (2018) Gastrointestinal tolerance and glycaemic response of isomaltooligosaccharides in healthy adults. Nutrients 10(3):301. https://doi.org/10.3390/nu10030301
Ibrahim OO (2018) Functional oligosaccharides: chemicals structure, manufacturing, health benefits, applications and regulations. J Food Chem Nanotechnol 4(4):65–76. https://doi.org/10.17756/jfcn.2018-060
Ichinose H, Suzuki R, Miyazaki T, Kimura K, Momma M, Suzuki N, Fujimoto Z, Kimura A, Funane K (2017). Paenibacillus sp. 598K 6-α-glucosyltransferase is essential for cycloisomaltooligosaccharide synthesis from α-(1→ 4)-glucan. Appl Microbial Biotechnol 101(10): 4115–4128. https://doi.org/10.1007/s00253-017-8174.z
Jana M, Maity C, Samanta S, Pati BR, Islam SS, Mohapatra PK, Mondal KC (2013) Salt independent thermophilic α-amylase from Bacillus megaterium VUMB109: an efficacy testing for preparation of maltooligosacharide. Ind Crops Prod 41:386–391. https://doi.org/10.1016/j.indcrop.2012.04.048
Jang EY, Hong KB, Chang YB, Shin J, Jung EY, Jo K, Suh HJ (2020) In vitro prebiotic effects of malto-oligosaccharides containing water-soluble dietary fiber. Molecules 25(21):5201. https://doi.org/10.3390/molecules25215201
Ji H, Bai Y, Li X, Wang J, Xu X, Jin Z (2019) Preparation of malto-oligosaccharides with specific degree of polymerization by a novel cyclodextrinase from Palaeococcus pacificus. Carbohydr Polym 210:64–72. https://doi.org/10.1016/j.carbpol.2019.01.041
Kootte RS, Vrieze A, Holleman F, Dallinga-Thie GM, Zoetendal EG, de Vos WM, Groen AK, Hoekstra JB, Stroes ES, Nieuwdorp M (2012) The therapeutic potential of manipulating gut microbiota in obesity and type 2 diabetes mellitus. Diabet Obes Metabol 14(2):112–120. https://doi.org/10.1111/j.1463-1326.2011.01483.x
Kroh LW, Hollnagel A (2002) News on the Maillard reaction of oligomeric carbohydrates. Int Congr Ser 1245:369–371. https://doi.org/10.1016/s0531-5131(02)00956-1
Kumar S, Khare SK (2012) Purification and characterization of maltooligosacharide-forming α-amylase from moderately halophilic Marinobacter sp. EMB8. Bioresour Technol 116:247–251. https://doi.org/10.1016/j.biortech.2011.11.109
Lakshmi SA, Shafreen RB, Priyanga A, Shiburaj S, Pandian SK (2020) A highly divergent α-amylase from Streptomyces spp.: An evolutionary perspective. Int J Biol Macromol 163:2415–2428. https://doi.org/10.1016/j.ijbiomac.2020.09.103
Lawrence K, Hyde J (2017) Microbiome restoration diet improves digestion, cognition and physical and emotional wellbeing. PLoS ONE 12(6):e0179017. https://doi.org/10.1371/journal.pone.0179017
Li W, Chen G, Gu L, Zeng W, Liang Z (2014) Genome shuffling of Aspergillus niger for improving transglycosylation activity. Appl Biochem Biotechnol 172(1):50–61. https://doi.org/10.1128/AEM.03934-14
Li M, Du J, Zhang K (2020) Profiling of carbohydrates in commercial beers and their influence on beer quality. J Sci Food Agri 100(7):3062–3070. https://doi.org/10.1002/jsfa.10337
Maalej H, Ben Ayed H, Ghorbel-Bellaaj O, Nasri M, Hmidet N (2014) Production and biochemical characterization of a high maltotetraose (G4) producing amylase from Pseudomonas stutzeri AS22. BioMed Res Int 20140:1–11. https://doi.org/10.1155/2014/156438
Manas NH, Pachelles S, Mahadi NM, Illias RM (2014) The characterisation of an alkali-stable maltogenic amylase from Bacillus lehensis G1 and improved malto-oligosaccharide production by hydrolysis suppression. PLoS ONE 9(9):e106481. https://doi.org/10.1371/journal.pone.0106481
Mookiah S, Sieo CC, Ramasamy K, Abdullah N, Ho YW (2014) Effects of dietary prebiotics, probiotic and synbiotics on performance, caecal bacterial populations and caecal fermentation concentrations of broiler chickens. J Sci Food Agric 94(2):341–348. https://doi.org/10.1002/jsfa.6365
Nishimura N, Tanabe H, Yamamoto T (2016) Isomaltodextrin, a highly branched α-glucan, increases rat colonic H2 production as well as indigestible dextrin. Biosci Biotechnol Biochem 80(3):554–563. https://doi.org/10.1080/09168451.2015.1104237
Niu D, Qiao J, Li P, Tian K, Liu X, Singh S, Lu F (2017) Highly efficient enzymatic preparation of isomalto-oligosaccharides from starch using an enzyme cocktail. Electr J Biotechnol 26:46–51. https://doi.org/10.1016/j.ejbt.2016.12.002
Pan S, Ding N, Ren J, Gu Z, Li C, Hong Y, Cheng L, Holler TP, Li Z (2017) Maltooligosacharide-forming amylase: Characteristics, preparation, and application. Biotechnol Adv 35(5):619–632. https://doi.org/10.1016/j.biotechadv.2017.04.004
Pan S, Gu Z, Ding N, Zhang Z, Chen D, Li C, Hong Y, Cheng L, Li Z (2019) Calcium and sodium ions synergistically enhance the thermostability of a maltooligosacharide-forming amylase from Bacillus stearothermophilus STB04. Food Chem 283:170–176. https://doi.org/10.1016/j.foodchem.2019.01.023
Patel S, Goyal A (2012) The current trends and future perspectives of prebiotics research: a review. 3 Biotech 2 (2):115–125. https://doi.org/10.1007/s13205-012-0044-x
Qiang X, YongLie C, QianBing W (2009) Health benefits application of functional oligosaccharides. Carbohyd Polym 77(3):435–441. https://doi.org/10.1016/j.carbpol.2009.03.016
Shi Q, Hou Y, Juvonen M, Tuomainen P, Kajala I, Shukla S, Goyal A, Maaheimo H, Katina K, Tenkanen M (2016) Optimization of isomaltooligosaccharide size distribution by acceptor reaction of Weissella confusa dextransucrase and characterization of novel α-(1→ 2)-branched isomaltooligosaccharides. J Agric Food Chem 64(16):3276–3286. https://doi.org/10.1021/acs.jafc.6b01356
Shivlata L, Satyanarayana T (2017) Characteristics of raw starch-digesting α-amylase of Streptomyces badius DB-1 with transglycosylation activity and its applications. Appl Biochemi Biotechnol 181(4):1283–1303. https://doi.org/10.1007/s12010-016-2284-4
Sidar A, Albuquerque ED, Voshol GP, Ram AF, Vijgenboom E, Punt PJ (2020) Carbohydrate binding modules: diversity of domain architecture in amylases and cellulases from filamentous microorganisms. Front Bioeng Biotechnol. https://doi.org/10.3389/2Ffbioe.2020.00871
Singh DP, Singh S, Bijalwan V, Kumar V, Khare P, Baboota RK, Singh P, Boparai RK, Singh J, Kondepudi KK, Chopra K (2018) Co-supplementation of isomalto-oligosaccharides potentiates metabolic health benefits of polyphenol-rich cranberry extract in high fat diet-fed mice via enhanced gut butyrate production. European J Nutr 57(8):2897–2911. https://doi.org/10.1007/s00394-017-1561-5
Sorndech W, Nakorn KN, Tongta S, Blennow A (2018) Isomalto-oligosaccharides: recent insights in production technology and their use for food and medical applications. LWT 95:135–142. https://doi.org/10.1016/j.lwt.2018.04.098
Subramanian G, Ayyadurai S, Sharma T, Singh SA, Rele M, Kumar LS (2012) Studies on maltoheaose (G6) producing alkaline amylase from a novel alkaliphilic Streptomyces species. IIOAB J 3(3):15–30
Thitaram SN, Chung CH, Day DF, Hinton A, Bailey JS, Siragusa GR (2005) Isomaltooligosaccharide increases cecal Bifidobacterium population in young broiler chickens. Poult Sci 84(7):998–1003. https://doi.org/10.1093/ps/84.7.998
Xie X, Li Y, Ban X, Zhang Z, Gu Z, Li C, Li Z (2019) Crystal structure of a maltooligosaccharide-forming amylase from Bacillus stearothermophilus STB04. Int J Biol Macromo 138:394–402. https://doi.org/10.1016/j.ijbiomac.2019.07.104
Yang CH, Liu WH (2007) Cloning and characterization of a maltotriose-producing α-amylase gene from Thermobifida fusca. J Ind Microbiol Biotechnol 34(4):325–330. https://doi.org/10.1007/s10295-006-0200-6
Zeuner B, Teze D, Muschiol J, Meyer AS (2019) Synthesis of human milk oligosaccharides: protein engineering strategies for improved enzymatic transglycosylation. Molecules 24(11):2033. https://doi.org/10.3390/molecules24112033
Zhang Z, Jin T, Xie X, Ban X, Li C, Hong Y, Cheng L, Gu Z, Li Z (2020) Structure of maltotetraose-forming amylase from Pseudomonas saccharophila STB07 provides insights into its product specificity. Int J Biol Macromol 154:1303–1313. https://doi.org/10.1016/j.ijbiomac.2019.11.006
Zhu D, Yan Q, Liu J, Wu X, Jiang Z (2019) Can functional oligosaccharides reduce the risk of diabetes mellitus? FASEB J 33(11):11655–11667. https://doi.org/10.1096/fj.201802802RRR
Acknowledgements
We would like to acknowledge both the Authors and funding agency under Women Scientists Scheme (WOS-A), Department of Science and technology (DST), Delhi, India and CSIR- National Chemical Laboratory, Pune, India for necessary facilities. The work described has not been published before, it is not under consideration for publication elsewhere, its submission to JFST publication has been approved by all authors as well as the responsible authorities tacitly or explicitly at the institute where the work has been carried out, if accepted, it will not be published elsewhere in the same form, in English or in any other language, including electronically without the written consent of the copyright holder, and JFST will not be held legally responsible should there be any claims for compensation or dispute on authorship.
Funding
Present research study is financially supported by Women Scientists Scheme (WOS-A), Department of Sciences and Technology (DST) and CSIR- National Chemical Laboratory, Pune, India for necessary facilities.
Author information
Authors and Affiliations
Contributions
VS (Vidhya Shinde) persuaded, and he concept and drafted the manuscript. VKR (Dr.V. Koteswara Rao) scientific correction, and English editing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The Author’s declares that they don’t have no competing Interests for the present research study.
Ethics Approval
Present study does not involve any ethical experiment.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Shinde, V.K., Vamkudoth, K.R. Maltooligosaccharide forming amylases and their applications in food and pharma industry. J Food Sci Technol 59, 3733–3744 (2022). https://doi.org/10.1007/s13197-021-05262-7
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-021-05262-7