Abstract
Processing methods involved in value addition to pigmented broken rice, a milling by-product may affect the nutraceutical qualities. Hence the effect of pulverisation and toasting on nutraceutical content in brown rice flours of red and black rice varieties were studied. Plate milling reduced soluble, bound and total polyphenols in red, but increased in black; increased insoluble polyphenols and oryzanol, and reduced anthocyanin in both varieties; antioxidant activity is reduced in red, but increased in black. Toasting caused reduction of soluble, insoluble and total polyphenol, anthocyanin and antioxidant activity in both varieties with an increase in bound polyphenols, and retention of flavonoids and oryzanol. HPLC characterization of phenolics showed changes on processing in content of catechin, caffeic, vanillic, protocatechuic and ferulic acid. It can be concluded that processing impacts the phenolic components of pigmented broken brown rice flour and this information is useful for functional food industry.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rice (Oryza sativa L.) is majorly consumed cereal by more than half of the world population as a fundamental source of energy (Monks et al. 2013). In recent times, pigmented rice varieties such as red, black, and wild rice, have received attention of health-conscious consumers. Pigmented rice varieties possess phenolic compounds which exert various health benefits such as antioxidant, anti-inflammatory, anti-atherosclerotic, anticarcinogenic, anti-allergic and hypoglycemic activities (Deng et al. 2013). Rice is generally consumed by de-husking the paddy and polishing to remove the bran. Broken rice generation is high (30%) during husk removal and polishing of pigment rice and is sold as low cost milling by-product. Brown (unpolished) broken generated during husk removal can be value added to develop nutraceutical and functional foods.
Development of convenience products such as rice flour, string hopper, roti etc., involves preprocessing such as size reduction and heat-processing which alters the phyto components. Research studies have shown that heat processing affects the level of phytochemicals (Xu et al. 2007); antioxidant activity (Queiroz et al. 2009; Xu et al. 2007), functional and nutritional properties of foods (Rehman and Shah 2005). At present attention is being given to the anti-oxidative and radical-scavenging properties of coloured rice cultivars and information on the effect of processing on the phenolic compounds and the antioxidant properties of pigmented rice are rather scarce. Hence, the objective of the study is to investigate the impact of different processing on the phenolic components and antioxidant properties of brown rice flours of red and black rice varieties.
Materials and methods
Materials
Jyothi red rice and Burma black rice paddies stored for 6 months after harvest were purchased from a local vendor in Mysore district Karnataka, India. Chemicals and reagents used were-Folin–Ciocalteu reagent and HPLC solvents from Spectochem, Mumbai, India. 6-hydroxy-2,5,7,8-tetramethyl chroman-2-carboxylic acid (Trolox), trifluoroacetic acid (TFA), Vanillin, Catechin hydrate, Gallic acid, Protocatechuic acid (PCA), caffeic acid, ferulic acid and p-coumaric acid were purchased from Sigma-Aldrich, Inc. (St. Louis, MO). Other chemicals were of analytical grade.
Sample preparation
Paddies were de-husked using centrifugal disc sheller at 3600 rpm, (Kisan Krishi Yantra Udyog, Kanpur, India) followed by broken rice separation using a paddy separator. To study the effect of pulverisation, unpolished broken rice of red and black varieties were milled thrice in the hammer (Natrajatta maker, India) and plate mill (Madras standard plate mill, India). To study the effect of toasting, plate milled flours of both verities toasted at 90–100 °C for 30 min in an Uruli roaster at 20 rpm (Model No. CD-10, Pilot Smith (India) Pvt. Ltd, Kerala, India). Processed samples were brought to room temperature (RT) and stored at either RT or − 20 °C depending on usage for further analysis.
Phenolic compounds
Extraction of soluble, bound and insoluble polyphenols compounds
Soluble/free, bound and insoluble phenolics were extracted according to the methods described by Tian et al. (2004) with minor modifications. Briefly, to extract soluble phenolic compounds, 2 g of rice flour was extracted with 16 ml of methanol for 1 h (hr) at RT by mixing in a tube mixer. Supernatants were collected after centrifugation at 6000 rpm for 7 min. For bound phenolic compounds extraction, the above-obtained residues were mixed with 16 ml of 1% HCl methanol and collected the supernatant as explained above. Insoluble phenolic components extracted by combining above obtained residue with 16 ml methanol with 4 M sodium hydroxide (NaOH). Samples were vortex mixed for 4 h and allowed to stand at RT overnight. The supernatant collected by centrifuging at 6000 rpm for 7 min. The supernatant pH is adjusted to 2 with 4 M HCl and extracted separately three times with ethyl acetate. To the combined ethyl acetate mixture one spatula of sodium sulfate was added and allowed to stand at RT overnight. After filtering the mixture in Whatman no. 1 filter paper, it was evaporated to dryness using a rotary evaporator at 60 °C (BUCHI, Rotavapor R-215). Residues dissolved in 10 ml methanol, the supernatant collected by centrifuging at 6000 rpm for 7 min. Extracts of soluble, bound and insoluble phenolics stored at − 4 °C until analysis and filtered using a disc filter (Agilent 0.45micron) before use.
Phenolic contents
Phenolic contents were determined using the Folin–Ciocalteu reagent (Singleton et al. 1999). To a known volume of extract (250 µl), freshly prepared Folin–Ciocalteu reagent (1:2 diluted) and 500 µl of sodium carbonate solution (Na2CO3) (20%) added. After vortexing for a minute, 4 ml of distilled water was added to the above mixture, mixed well and kept in the dark for 30 min. Sample mixture centrifuged at 2000 rpm for 5 min. The absorbance of the blue coloured supernatant was measured at 760 nm. Ferulic acid was used as standard and values are expressed as mg ferulic acid (FA) equivalent per 100 g flour. Total polyphenol content was a sum of all the three phenolic components.
HPLC characterisation of phenolics
Phenolic components of extract were measured using an analytical HPLC system (Waters 2545, U.S.A.) according to the method of Alu’datt et al. (2013). A Supelco analytical C18 column (15 cm × 4.6 mm, 2.7 µm Supelco Co., PA, U.S.A) was used. The phenolics components detected at a wavelength of 280 nm with photodiode array detector (PDA, Waters Co). Gradient mobile phase consisted of 0.2% trifluoroacetic acid (solvent A) and 100% methanol (solvent B). The flow rate was adjusted to 0.75 ml/min at 25 °C for the total running time of 65 min. The gradient flow set as follows; 0–5 min 95% solvent A to 5% solvent B at 0–50 min, 80% B at 51–60 min, 95% A to 5% B at 61–65 min. Based on the retention time of standards, peaks were identified and areas used for all calculations. Linearity for each standard was found to be between 4 and 9 ng whereas Protocatechuic acid was in the range of 20–40 ng.
Total anthocyanin content
Total anthocyanin content (TAC) determined through the pH differential method described by Giusti and Wrolstad (2001). Anthocyanin content was extracted from 2 g of rice flour with 16 ml of methanol for 1 h at room temperature (RT) by mixing in a tube mixer. Supernatants collected after centrifugation at 6000 rpm for 7 min. Total volume in the tubes was made up to 1.5 ml using KCl–HCl buffer of pH 1 and citrate buffer of pH 4.6. Above mixtures were vortexed and kept in the dark for 20 min. Absorbance measured at two different wavelengths 515 nm and 700 nm against a reagent blank. Anthocyanin content was expressed as mg cyanidin 3-glucoside equivalent/100 g flour.
Flavonoid content
Flavonoids content was assayed as described by Bao et al. (2005), with slight modification. 1 g of flour de-fated at RT by mixing with hexane and decanted flour was allowed to dry overnight, followed by addition of 1% HCl methanol and extraction at RT for 1 h. Supernatant collected after centrifuging at 2000 rpm for 10 min. A known volume of extract was taken and made up to 500 μl using distilled water. To this mixture 75 μl of sodium nitrite (NaNO2) (1:20), followed by 150 μl of aluminium chloride hexahydrate (AlCl3·6H2O) (1:10) added and allowed to stand at RT for 6 min. After adding 500 μl of 1 N sodium hydroxide (NaOH), sample mixture was vortexed and kept in the dark for 15 min. The supernatant was collected by centrifuging at 2500 rpm for 5 min, and the absorbance was measured at 512 nm using catechin as standard. Flavonoid content expressed as mg of catechin equivalent (CE)/100 g of the sample.
Tannin content
Tannin content was determined using the method described by Earp et al. (1981). 0.1 g rice flour mixed with 4 ml of 1% HCl-methanol and extracted by using tube mixer for 1 h at RT followed by collecting the supernatant after centrifuging at 2500 rpm for 10 min. A known volume of sample extract was made up to 250 μl by adding 1% HCl-methanol, and 1.25 ml of freshly prepared vanillin-HCl reagent added as well. Sample blank was prepared by adding 4% HCl methanol. The samples were allowed to rest for 20 min at RT, and OD was read at 500 nm against sample blank, using catechin as standard. Tannin content expressed as mg catechin equivalent (CE)/100 g of the sample.
Oryzanol content
The oryzanol content was determined based on the method described by Seetharamaiah and Prabhakar (1986). 0.2 g of rice flour was extracted with 5 ml petroleum ether twice by heating at 65 °C for 7 min. Supernatants collected and volume was made up to 10 ml using petroleum ether. Further, centrifuged at 2000 rpm for 7 min and the absorbance of the supernatant recorded at 314 nm. Oryzanol content was calculated by using absorbance of 1% standard Oryzanol in petroleum ether at 314 nm and expressed as mg/100 g of the sample.
Bioactivity assays
Ferric reducing power (FRP)
The FRP test was carried out using soluble extract (methanolic extracts) based on the reduction of the Ferric (Fe3+) complex to the ferrous (Fe2+) form at low pH. Briefly, different volumes of the soluble extract were mixed with 0.2 M phosphate buffer of pH 6.6 to make up the volume to 500 µl followed by 500 µl of 1% potassium ferricyanide. Sample mixture incubated at 50 °C for 20 min. 500 µl of 10% trichloroacetic acid (TCA) was added, mixed well followed by centrifugation at 1000 rpm for 10 min. 300 µl of supernatant mixed with 300 µl distilled water and 100 µl of 0.1% ferric chloride (FeCl3). Absorption measured at 700 nm. Results were calculated with percentage reduction relative to the control and expressed as mg Trolox equivalent/100 g sample.
DPPH+ radical scavenging assay
Soluble extracts tested for their ability to scavenge the stable radical of DPPH (2, 2′-diphenyl-1-picrylhydrazyl) as described by Bondet et al. (1997). Different volumes of soluble extracts mixed with 0.08 mg/ml DPPH in methanol. After 30 min incubation at RT, the absorbance was measured at 517 nm against methanol as blank and the percentage reduction calculated. Catechin used as standard and values expressed as mg catechin equivalent (CE)/100 g sample.
Statistical analysis
All statistical analysis conducted in (MS Office Excel 2016)—one-way ANOVA to study the effect of milling on untoasted red rice flours, and two-way ANOVA to examine the varietal (red and black) and treatment effects (untoasted and toasted). The values in the tables are mean ± SD (standard deviation) of three independent replicates. Phenolics value by HPLC is an average of two separate observations.
Results and discussion
Effect of milling and thermal treatment on polyphenols
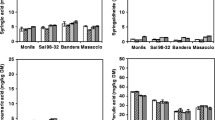
Polyphenols contents of the flours after different processing is shown in Table 1. In red rice flour obtained by hammer milling, soluble polyphenol content was 279 mg FAE/100 g which was reduced by 19% and 25% on plate milling and toasting respectively. Bound polyphenol was about 227 mg FAE/100 g which reduced by 23% on plate milling and increased by 23% on toasting. Insoluble polyphenol content was about 85 mg FAE/100 g which increased by 47% on plate milling and reduced by 50% on toasting. Total polyphenol content was about 592 mg FAE/100 g which reduced by 11% and 16% on plate milling and toasting respectively and end product retained about 73–89% of total polyphenol content compared to control.
In the case of black rice soluble polyphenol content was about 121 mg FAE/100 g, and plate milling and toasting did not affect the content. Bound polyphenol content was about 186 mg FAE/100 g which increased by 18% on plate milling with an insignificant effect on toasting. Insoluble polyphenol was about 105 mg FAE/100 g; which increased slightly on plate milling, but reduced by 16% on toasting. Total polyphenol content was about 412 mg FAE/100 g which increased by 12% on plate milling and toasting had an insignificant effect. Results show retention of total polyphenol content in the range of 105–112% compared to control.
The two-way ANOVA showed significant (p < 0.05) interactions between the rice varieties and treatments in the polyphenol fractions indicating the response of two rice varieties to treatment is not the same.
Simultaneous attrition and shearing between the plates during plate milling causes rupturing of cell wall (Smitha et al. 2008) which improves the accessibility of the solvent for an efficient extraction increasing polyphenol content. Haridas Rao et al. (1989) reported that heat generated (up to 95 °C) on the clearance between the plates and severity of grinding attributes to the reduction in the polyphenol content.
As reported by Goufo and Trindade (2014), roasting causes degradation of phenolic compounds, polymerisation, oxidation and the Maillard reaction which reduces the solubility of the phenolic compounds thereby decreases polyphenol content. Heat transfer also causes the structural and morphological changes in the matrix providing easy accessibility of the solvent to extract more polyphenol content.
HPLC characterisation of phenolics
Effects on phenolics were investigated through HPLC analysis as shown in Tables 2, 3 and 4. Plate milling and toasting showed varying results in the content of phenolics in both red and black rice flours.
HPLC characterisation of soluble phenolics revealed that red rice flour extract contained health beneficial phenolics like catechin and caffeic, and black rice had ferulic and p-coumaric as the major phenolic components. Table 2 shows that in red rice, plate milling increased the quantity of catechin, caffeic, and ferulic acid by 23%, 42% and 379% respectively, whereas vanillic acid and protocatechuic acid (PCA) decreased by 72% and 63% respectively compared to control. Further toasting caused an increase of PCA and ferulic acid by 95% and 7% respectively whereas catechin, vanillic and caffeic acid decreased by 42%, 26%, and 48% respectively.
In black rice, plate milling reduced the quantity of catechin, and caffeic acid by 14% and 33% respectively but increased the amount of vanillic by 31% and ferulic acid by 15% compared to control. Further toasting increased vanillic, caffeic acid and ferulic acid by 108%, 105%, and 72% respectively and decreased catechin by 19%. Both the processing caused an increase in ferulic acid content.
HPLC characterisation of bound phenolics showed (Table 3) that red rice flour extracts contained catechin, vanillic, ferulic and caffeic, and black rice had ferulic and p-coumaric as the major components. In red rice, plate milling decreased the quantity of catechin, vanillic, caffeic and ferulic acid by 47%, 56%, 77% and 16% compared to control. Further toasting increased catechin and vanillic by 14% and 52% respectively and reduced caffeic, ferulic and protocatechuic acid by 12%, 76%, and 81% respectively.
In black rice, plate milling reduced ferulic acid by 16% and increased catechin by 15% with 100% decrease in PCA, vanillic and caffeic acid. Further toasting reduced catechin and ferulic acid by 77% and 8% respectively.
HPLC characterization of insoluble phenolics showed (supplementary figure 1) that red rice flour extracts contained ferulic, catechin and caffeic, and black rice had ferulic, vanillic and caffeic acids as the major components. In red rice, plate milling increased the quantity of catechin, caffeic acid and ferulic acid by 12%, 144%, and 191% respectively compared to control (Table 4). Further toasting decreased catechin and caffeic acid by 66% and 64% respectively and did not affect the ferulic acid content. In black rice (Supplementary figure 2), plate milling reduced vanillic, caffeic and ferulic acid by 46%, 84%, and 73% respectively compared to control. Further toasting increased vanillic by 101%. It did not affect caffeic acid but reduced ferulic acid content by 73%.
Present study results are in agreement with the research study of Kaur et al. (2018) who have reported that soluble/free fraction of pigmented rice had of catechin, caffeic and vanillic acid and bound fractions had ferulic and p-coumaric acid in higher concentration.
However various studies showed different results under different processing conditions. Finocchiaro et al. (2007) observed that ferulic and sinapic acid contents reduced by grinding. Moore et al. (2009) and Abdel-Aal and Rabalski (2013) stated that cooking increase free ferulic acid quantity and reduces bound ferulic acid amount in bakery products. Marinova and Yanishlieva (1992) observed that ferulic acid antioxidant activity at 25–100 °C did not change in purified lard triacylglycerols.
Effect of processing on phytonutrients
The content of other phytonutrients in different processed rice flours shown in Table 5. The two way ANOVA showed significant (p < 0.05) interactions between the rice varieties and treatments.
Flavonoid content in red and black rice flour was about 67 and 65 mg CE/100 g respectively. Both plate milling and toasting had an insignificant effect on both the verities.
Anthocyanin content in red rice flour was about 6.5 mg cynd. 3 glu.Eq/100 g and on plate milling, it reduced by 14%, but toasting did not effect. In black rice flour, anthocyanin content was found to be 23.2 mg cynd. 3 glu.Eq/100 g, on plate milling it reduced by 10%, and further toasting decreased it by 17% with retention in the range of 73–90%.
Reduction of anthocyanin content may be due to the degradation by heating as observed by Hiemori et al. (2009) in black rice cooked by different methods.
HPLC characterisation showed the presence of Cyanidin 3-glucoside and peonidin- 3-glucoside as significant anthocyanin components in black rice (supplementary figure 3) and processing had an insignificant effect (supplementary figure 4). This indicates that the marginal reduction in total anthocyanin content is due to the loss in minor components.
Oryzanol content in red rice flour was found to be 20 mg/100 g of the sample with 22% increase on plate milling and 12% reduction on toasting. In black rice flour, it was found to be 41 mg/100 g of the sample which was increased by 12% on plate milling, and toasting did not effect. Results indicate that plate milling causes an increase in oryzanol, whereas toasting has an insignificant impact. Finally, the retention compared to control was ~ 122–112% in both red and black. As reported by Wanyo et al. (2016) the increase in oryzanol content on plate milling can be attributed to the smaller particle size of the flour which increases the extraction of oryzanol. Studies on thermal processing of brown rice by Pascual et al. (2013) showed 20% loss after parboiling in 27 brown rice samples. In contrast, Khatoon and Gopalakrishna (2004) reported no change in gamma oryzanol after parboiling. Pradeep et al. (2014) also reported that direct steam exposure of bran increased oryzanol compared to native.
Tannin content in red rice flour was about 300 mg CE/100 g, and in black it was below the level of detection. It was reduced by 40% and 19% on plate milling and toasting respectively. The decrease in tannins contents is because of the loss of compounds (Nithya et al. 2007), and also degradation or interaction with other components of seeds, such as proteins, to form insoluble complexes (Embaby 2010) on exposure to heat or temperature. Reduction in tannin on milling and toasting is beneficial as tannin is considered an anti-nutrient.
Effect on bioactivity
Bioactivity of the different flour samples reported in Table 6. The two-way ANOVA showed significant interactions (p < 0.05) between varieties and treatments.
FRP assay showed that the reducing power of red rice flour was 545 mg TE/100 g which was reduced by 37% and 19% on plate milling and toasting respectively. Black rice showed the reducing power of 210 mg TE/100 g which increased by 35% on milling and reduced by 14% on toasting.
DPPH assay revealed that scavenging ability of the red rice flour was about 267 mg CE/100 g which was reduced by 33% and 27% on plate milling and toasting respectively. Black rice showed the scavenging ability of 84.2 mg CE/100 g, plate milling had an insignificant effect, but toasting caused 15% reduction.
The study results are in agreement with the research work carried out by Ktenioudaki et al. (2014) which showed that the antioxidant capacity of grain bioactive components is affected by the grinding of grains. Walter et al. (2013) demonstrated that thermal processing causes a reduction in polyphenol content due to thermal decomposition and also reduced polyphenol concentration lowers antioxidant activity in rice.
Conclusion
Effect of pulverisation and toasting on polyphenols, flavonoids, anthocyanin, tannin, oryzanol contents and bioactivity of red and black broken rice were studied. Plate milling caused an increase in insoluble polyphenols, oryzanol, reduction in tannin and antioxidant activity and retention of flavonoids of red rice flour. On the other hand, in black rice oryzanol and antioxidant activities increased with retention of flavonoid and decrease in anthocyanin. Phenolics characterization revealed an increase in the contents of ferulic acid in the soluble fraction, catechin in soluble and insoluble fractions; decrease in ferulic acid and catechin in the bound fraction in red rice flour. In black rice flour, ferulic acid in the soluble fraction, catechin in insoluble and bound fractions increased whereas catechin in soluble fraction and ferulic acid in bound and insoluble fractions decreased on plate milling. Toasting caused the reduction in tannins, anthocyanin and antioxidant activity in both varieties and retention of flavonoids and oryzanol. Polyphenol fractions revealed reduction in catechin in all fractions except bound fraction in the red and insoluble fraction in black followed by ferulic acid in all fractions except soluble fraction for black observed on toasting. Though processing caused changes, there is retention of ~ 73–105% polyphenols, ~ 73–90% of anthocyanin and ~ 112–122% of oryzanol in both red and black with ~ 44–100% retention of antioxidant activities. It can be concluded that brown rice flours obtained through different pulverizers as well as toasting of unpolished rice brokens of pigmented rice varieties are potential sources of nutraceuticals like phenolic compounds, and can be used for preparation of convenience food products.
References
Abdel-Aal ESM, Rabalski I (2013) Effect of baking on free and bound phenolic acids in whole grain bakery products. J Cereal Sci 57:312–318
Alu’datt MH, Rababah T, Ereifej K, Alli I (2013) Distribution, antioxidant and characterization of phenolic compounds in soybeans, flaxseed, and olives. Food Chem 139:93–99
Bao JS, Cai Y, Sun M, Wang GY, Corke H (2005) Anthocyanins, flavonols, and free radical scavenging activity of Chinese bayberry (Myrica rubra) extracts and their color properties and stability. J Agric Food Chem 53:2327–2332
Bondet V, Brand-Williams W, Berset C (1997) Kinetics and mechanisms of antioxidant activity using the DPPH-free radical method. LWT Food Sci Technol 30:609–615
Deng GF, Xu XR, Zhang Y, Li D, Gan RY, Li HB (2013) Phenolic compounds and bioactivities of pigmented rice. Crit Rev Food Sci Nutr 53:296–306
Earp CF, Akingbala JO, Ring SH, Rooney LW (1981) Evaluation of several methods to determine tannins in sorghums with varying kernel characteristics. Cereal Chem 58:234–238
Embaby HES (2010) Effect of soaking, dehulling, and cooking methods on certain anti-nutrients and in vitro protein digestibility of bitter and sweet lupin seeds. Food Sci Biotechnol 19:1055–1062
Finocchiaro F, Ferrari B, Gianinetti A, Dall’asta C, Galaverna G, Scazzina F, Pellegrini N (2007) Characterization of antioxidant compounds of red and white rice and changes in total antioxidant capacity during processing. Mol Nutr Food Res 51:1006–1019
Giusti MM, Wrolstad RE (2001) Characterization and measurement of anthocyanins by UV–Visible spectroscopy. Curr Protoc Food Anal Chem. https://doi.org/10.1002/0471142913.faf0102s00
Goufo P, Trindade H (2014) Rice antioxidants: phenolic acids, flavonoids, anthocyanins, proanthocyanidins, tocopherols, tocotrienols, c-oryzanol, and phytic acid. Food Sci Nutr 2(2):75–104
Haridas Rao P, Leelavathi K, Shurpalekar SR (1989) Effect of damaged starch on the chapati-making quality of whole wheat flour. Cereal Chem 66(4):329–333
Hiemori M, Koh E, Mitchell AE (2009) Influence of cooking on anthocyanins in black rice (Oryza sativa L. japonicavar. SBR). J Agric Food Chem 57:1908–1914
Kaur P, Singh N, Pal P, Kaur A (2018) Variation in composition, protein and pasting characteristics of different pigmented and non-pigmented rice (Oryza sativa L.) grown in Indian Himalayan region. J Food Sci Technol 55(9):3809–3820
Khatoon S, Gopalakrishna AG (2004) Fat-soluble nutraceuticals and fatty acid composition of selected Indian rice varieties. J Am Oil Chem Soc 81:939–943
Ktenioudaki A, Alverez-Jubate L, Gallagher E (2014) A review of the process-induced changes in the phytochemical content of cereal grains: the bread making process. Crit Rev Food Sci Nutr 55:611–619
Marinova EM, Yanishlieva NV (1992) Effect of temperature on the antioxidative action of inhibitors in lipid autoxidation. J Sci Food Agric 60:313–318
Monks JLF, Vanier NL, Casaril J, Berto RM, de Oliveira M, Gomes CB, de Carvalho MP, Dias ARG, Elias MC (2013) Effects of milling on proximate composition, folic acid, fatty acids and technological properties of rice. J Food Compost Anal 30:73–79
Moore J, Luther M, Cheng Z, Yu LL (2009) Effects of baking conditions, dough fermentation, and bran particle size on antioxidant properties of whole-wheat pizza crusts. J Agric Food Chem 57:832–839
Nithya KS, Ramachandramurty B, Krishnamoorthy VV (2007) Effect of processing methods on nutritional and anti-nutritional qualities of hybrid (COHCU-8) and traditional (CO7) pearl millet varieties of India. J Biol Sci 7:643–647
Pascual CDCI, Massaretto IL, Kawassaki F, Barros RMC, Noldin JA, Marquez UML (2013) Effects of parboiling, storage and cooking on the levels of tocopherols, tocotrienols, and γ-Oryzanol in brown rice (Oryza sativa L.). Food Res Int 50:676–681
Pradeep PM, Jayadeep A, Guha M, Singh V (2014) Hydrothermal and biotechnological treatments on nutraceutical content and antioxidant activity of rice bran. J Cereal Sci 60:187–192
Queiroz YS, Manolio SRA, Capriles VDC, Toress EA, Areas JA (2009) Effect of processing on the antioxidant activity of amaranth grain. Arch Latinoam Nutr 59:419–424
Rehman ZU, Shah WH (2005) Thermal heat processing effects on anti-nutrients, protein and starch digestibility of food legumes. Food Chem 91(2):327–331
Seetharamaiah GS, Prabhakar JV (1986) Oryzan ol content of Indian rice bran oil and its extraction from soap stock. J Food Sci Technol 23:270–274
Singleton VL, Orthofer R, Lamuela-Raventos RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteu reagent. Methods Enzymol 299:152–178
Smitha VK, Kumar Sourav, Malleshi NG (2008) Studies on the effect of milling finger millet in different pulverisers on physico-chemical properties of the flour. J Food Sci Technol 45(5):398–405
Tian S, Nakamura K, Kayahara H (2004) Analysis of phenolic compounds in white rice, brown rice, and germinated brown rice. J Agric Food Chem 52:4808–4813
Walter M, Marchesan E, Massoni PFS, Silva LP, Sartori GMS, Ferreira RB (2013) Antioxidant properties of rice grains with light brown, red and black pericarp colors and the effect of processing. Food Res Int 50:698–703
Wanyo P, Kaewseejan N, Meeso N, Siriamornpun S (2016) Bioactive compounds and antioxidant properties of different solvent extracts derived from Thai rice by-products. Appl Biol Chem 59:373–384
Xu G, Ye X, Chen J, Liu D (2007) Effect of heat treatment on the phenolic compounds and antioxidant capacity of citrus peel extract. J Agric Food Chem 55:330–335
Acknowledgements
Project funded by Govt. of India XII plan Project AGROPATHY (BSC0105).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sapna, I., Kamaljit, M., Priya, R. et al. Milling and thermal treatment induced changes on phenolic components and antioxidant activities of pigmented rice flours. J Food Sci Technol 56, 273–280 (2019). https://doi.org/10.1007/s13197-018-3487-1
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-018-3487-1