Abstract
Residual thyroid tissue after total thyroidectomy in differentiated thyroid cancers is considered an independent risk factor for recurrence. Guidelines recommend following up patients after surgery with thyroglobulin (Tg), neck ultrasonography, and occasionally whole-body radioactive scan. However, the results of serum thyroglobulin and whole-body radioiodine scan are often discordant. The present study was undertaken to determine the levels of serum-stimulated thyroglobulin to complement the findings of residual thyroid tissue in the radioactive whole-body scan. One hundred twenty-six patients had undergone a radioiodine (131 I) whole-body scan (WBS) during the study duration, and 121 were available for analysis. The thyroglobulin level (measured by the CLIA method) was recorded at the time of these scans. The data was analysed to determine the level of stimulated thyroglobulin correlating with residual thyroid tissue, locoregional, and distant metastasis as assessed by WBS. The presence of residual thyroid tissue was noted in an overwhelmingly high 94% of cases. Twenty-four of the 28 patients with stimulated Tg < 2 ng/dl had residual thyroid tissue on a WBS. The discordancy rate (positive moderate — large WBS and negative serum thyroglobulin) of 64.28% was seen. Using ROC the serum thyroglobulin cut-offs levels for the loco-regional disease were found to be 27.705 ng/dl and 94.770 ng/dl for distant metastasis. The results highlight the fact that serum Tg levels cannot be used as an accurate predictor of the extent of the remnant thyroid tissue. Irrespective of the quality of surgery, which was analysed based on the centre and surgical specialty, over 90% of cases had residual thyroid tissue on WBS. The use of only stimulated Tg levels for follow-up may be inaccurate. Serum Tg is a useful test along with radioactive whole-body scans to distinguish local disease, loco-regional disease, and distant metastasis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The residual thyroid tissue after total thyroidectomy in cases of differentiated thyroid cancers can harbour a high risk of malignancy. This residual tissue is an independent factor for disease recurrence. The residual thyroid tissue is commonly detected on a postoperative diagnostic 131 I radioactive iodine whole-body scans (RAI WBSs). Once detected it has a significant impact on the treatment outcomes that can alter the decision making [1].
The surveillance after a total thyroidectomy today is based on stimulated thyroglobulin (Tg), neck ultrasonography (USG), and WBS. The postoperative risk stratification is based on a dynamic model dependent on stimulated Tg [2]. Serum Tg is a large (660 kDa) dimeric glycoprotein produced by the follicular cells of the thyroid gland and provides an estimation of functional thyroid tissue. It is anticipated that the larger the residual functional thyroid tissue, the higher should be the Tg levels. In practice, this relationship is not linear and often there is a variation [3].
There are instances of missed diagnosis of recurrence in low-risk patients during follow-up when only serum Tg levels. This undermines the reliability of using stimulated Tg or serum anti-Tg as a sole marker for disease risk stratification and monitoring. The variability also needs to be assessed with relevance in the Indian context as other factors may contribute to different results from western countries.
The current study evaluated and compared the relationship between stimulated serum Tg after total thyroidectomy in differentiated thyroid cancer with the extent of functional residual thyroid tissue on the whole-body radioactive scan.
Materials and Methods
The present study was approved by the Institutional Review Board at our institution. This was a prospective single-arm study. Patients with a histopathological diagnosis of DTC who had undergone total thyroidectomy at our centre or referred to our centre after total thyroidectomy for a whole-body iodine scan from April 2017 to April 2018 were included in the study.
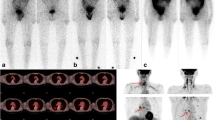
All the patients underwent a WBI scan only if the TSH was more than 30 mIU/ml. All the patients underwent (131I) whole-body radioactive scan (planar imaging) 24 h after oral administration of 111 MBQ of I-131. The images were categorised based on the extent and intensity of residual thyroid tissue by two experienced nuclear medicine physicians and results derived by consensus between the two into a visual tissue burden scale. The scale was based on visual evaluation of the intensity of residual thyroid tissue and ranged from tissue burden 0–2 {tissue burden 0, no residual thyroid tissue; 1, small and minimal intensity, minimal thyroid tissue; 2, moderate or large thyroid tissue} (Fig. 1) [4].
The serum Tg levels were recorded at the time of the RAI scan and analysed. The measurements were done by the electrochemiluminescence method (ECLIA) calibrated against the international standard CRM 457. The functional sensitivity of the assay is 0.04 ng/ml. The same assay has been used for all the patients included in the study. The dose of I 131 for therapeutic purpose was variable depending on the extent of residual tissue.
Statistical Analysis
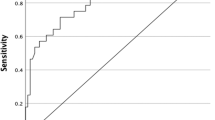
The data was analysed using IBM SPSS 20.0 and Microsoft Office 2010 software. The cut-off values were found using ROC curves. Frequencies and proportions were used to describe categorical variables (sex, histology type) and metastasis (locoregional and distant).
Results
A total of 126 patients underwent whole-body radioiodine scan during the period from 1st April 2017 to 30th April 2018. Out of these patients, serum Tg levels before whole-body radioiodine scans were not available for 5 patients. The total number of patients for the final analysis is 121 patients. The average age was 44.15 years ranging from 16 to 68 years.
Fifty-one patients had undergone a central compartment clearance or lateral neck dissection or both in addition to a total thyroidectomy/completion thyroidectomy (Table 1). The tissue located in the thyroid bed was considered to be thyroid remnant. The tissue in the lateral aspect of the neck was considered lymph node metastasis. Residual thyroid tissue was present in the WBS scan in 113 patients (94%). Fifteen patients had minimal residual tissue (12.4%), 98 had moderate to the large residue (81%) (Fig. 2). Six patients had distant metastasis (lung/bone) on WBS, and 17 of the 121 patients had regional metastasis (Table 2). All patients with residual thyroid tissue (113 of the 121) underwent ablation for the residual disease.
A total of 28 patients in our study had residual thyroid tissue despite having serum Tg levels < 2 ng/dl. Eighteen of them had moderate to large residual thyroid tissue, 6 had minimal residual thyroid tissue, and 4 had no residual tissue in the thyroid bed (Table 3 and Fig. 3). Based on the ROC curve, the cut-off values of serum Tg for loco-regional metastasis and distant metastasis are 27.705 ng/dl and 94.77 ng/dl, respectively. Fifty-eight cases were operated in tertiary care private hospitals (47.9%), 27 in medical colleges (22.4%), and 36 in tertiary cancer centres (29.7%). Fifty-nine patients were primarily operated by surgical oncologists (48.7%) and rest by head and neck surgical oncologist, otolaryngologists, and general surgeons. The patients who had residual thyroid tissue despite having serum Tg levels < 2 ng/dl were operated by surgical oncologists in 50% of cases.
Discussion
The incidence of differentiated thyroid cancer has increased in the last decades. The majority of these are small tumours, and the trend has been attributed to rampant use of USG [5, 6]. The majority of these incidentally detected asymptomatic patients are low risk. And according to the American Thyroid Association (ATA) guidelines [2], these patients are managed with surgery and use of radioiodine ablation when indicated. Recommendations are to follow them up with serum Tg levels and neck sonograms. Radioactive iodine is not recommended in this group.
Serum Tg is an indicator of the amount of functional thyroid tissue present in the body. However, several studies have shown wide variability in the values. This variation can be attributed to the presence of residual thyroid tissue even after the patient has undergone a total thyroidectomy [3].
The completeness of total thyroidectomy has been controversial. Despite the removal of all macroscopic gross thyroid tissue, the radioiodine uptake may be evident in the thyroid bed and estimated to be as high as 52% [2]. The high rate of RAI uptake following thyroidectomy could be because the surgeon missed to include the superior and pyramidal poles [7]. The area of the posterolateral suspensory ligament of berry may be another problematic area [8]. The patient population in our study was operated upon by well-trained surgeons and at tertiary care centres. Despite this residual thyroid tissue as detected on the RAI scan was overwhelmingly high at 94%. On subset analysis, the distribution of the patients based on the operating surgeon and the treating hospital showed no such correlation with the presence or absence of the residual thyroid tissue. It is prudent to understand if this uptake is of clinical significance. In our patient population, this was in fact due to the residual thyroid tissue in most cases. The presence of this residual thyroid tissue may interfere with the measurements of serum Tg during follow-up. This raises the question of whether only serum Tg and sonogram be sufficient in low-risk patients who have undergone total thyroidectomy without radioiodine ablation.
The false-negative rates of Tg assays have been attributed to technical problems, heterogeneity of circulating Tg, inadequate TSH stimulation, interference with anti-Tg antibodies, small tumour mass-producing low Tg, and “hook effect” which implies falsely low response in presence of a high concentration of Tg [8, 9]. In our study, out of the 28 patients who had simulated serum Tg < 2 ng/dl, 18 of them showed residual thyroid tissue of moderate to large extent in the thyroid bed. This suggests that serum Tg may not be a reliable estimate of the amount of residual thyroid tissue in the body.
The level of circulating anti-Tg antibodies ranges from 10 to 25%. Anti-Tg levels were not measured in all cases and are one of the limitations of our study. However, this alone cannot explain the high levels of discordance seen between the level of serum Tg and the presence of residual thyroid tissue. We suggest future studies should include anti-Tg as a standard parameter to draw better conclusions.
Wide variability exists in the values of serum Tg measured post total thyroidectomy [10]. In our study, the cut-off values of serum Tg ascertained for locoregional and distant metastasis were 27.7 ng/dl and 94.8 ng/dl, respectively. There has, however, been no consensus regarding the cut-offs for locoregional and distant metastasis. These cut-off values may assist in planning surveillance strategies.
Studies have shown a discordancy rate from 4 to 47% (positive WBS and negative Tg levels) between serum Tg and WBS scan [4,5,6]. In our study, the discordance rate was much higher at 64.28% (positive moderate — large WBS and negative serum Tg). Phan et al. [11] had shown that serum Tg levels may not be the only factor to be determined to assess for the disease status of the patient. This incongruence has been well highlighted in our study.
Like most global health systems, even in India surgeons from different surgical specialties treat thyroid cancers. Our study included patients who were treated at different tertiary care centres and were operated upon by qualified and well-trained surgeons. Yet, residual thyroid tissue was picked up in the WBS in over 90% of patients. This finding is cardinal and may reflect the true picture globally and designates the need for a WBS in all patients despite the risk status.
There are several limitations to this study. The patients have not been followed up to detect recurrences, and hence, the clinical significance of the findings needs to be further investigated. The measurement of serum anti-Tg antibodies has not been done in all cases. All the patients have not been operated by a single surgeon. This may also add to the variation seen in the residual thyroid tissue.
Conclusion
Serum Tg values with the heightened functional sensitivity available at present times may not serve as a marker of the amount of remnant thyroid tissue as shown in our study. This finding is crucial considering the current recommendations, which suggest follow-up with serum Tg and sonographic examination. This can lead to a missed diagnosis of residual thyroid tissue. The other pertinent issue is whether the removal of all residual thyroid tissue can be achieved or not. A combination of imaging studies like USG and WBS scan may be of value while following up patients of differentiated thyroid cancer.
References
Bal CS, Padhy AK (2015) Radioiodine remnant ablation: a critical review. World J Nucl Med 14(3):144–155. https://doi.org/10.4103/1450-1147.163240
Haugen BR, Alexander EK, Bible KC et al (2016) 2015 American Thyroid Association Management guidelines for adult patients with thyroid nodules and differentiated thyroid cancer: the American Thyroid Association Guidelines Task Force on Thyroid Nodules and Differentiated Thyroid Cancer. Thyroid 26(1):1–133. https://doi.org/10.1089/thy.2015.0020
Indrasena BS (2017) Use of thyroglobulin as a tumour marker. World J Biol Chem 8(1):81–85. https://doi.org/10.4331/wjbc.v8.i1.81
Silva F, Martin RJ, Figueroa J, Rincón F, Román D (2016) Variability of serum thyroglobulin levels in post- thyroidectomy patients with well-differentiated thyroid cancer: the ATA guidelines. P R Health Sci J 35(3):142–146
Vishal Rao US, Koya S, Gandla S et al (2018) Small-sized thyroid cancers-a single institutional experience in India. Indian J Surg Oncol 9(1):35–38. https://doi.org/10.1007/s13193-017-0673-y
Mathew IE, Mathew A (2017) Rising thyroid cancer incidence in Southern India: an epidemic of overdiagnosis? J Endocrine Soc 1(5):480–487
Fratkin MJ, Newsome HH, Sharpe AR, Tatum JL (1983) Cervical distribution of iodine 131 following total thyroidectomy for thyroid cancer. Arch Surg 118(7):864–867
Salvatori MA, Raffaelli MA, Castaldi PA, Treglia GI, Rufini V, Perotti GE, Lombardi CP, Rubello D, Ardito GU, Bellantone R (2007) Evaluation of the surgical completeness after total thyroidectomy for differentiated thyroid carcinoma. Eur J Surgical Oncol 33(5):648–654
Zucchelli G, Iervasi A, Ferdeghini M, Iervasi G (2009) Serum thyroglobulin measurement in the follow-up of patients treated for differentiated thyroid cancer. Q J Nucl Med Mol Imaging 53(5):482
Silva F, Martin RJ, Figueroa J, Rincón F, Román D (2016) Variability of serum thyroglobulin levels in post thyroidectomy patients with well differentiated thyroid cancer and the ATA guidelines. P R Health Sci J 35(3):142–146
Phan HT, Jager PL, van der Wal JE, Sluiter WJ, Plukker JT, Dierckx RA, Wolffenbuttel BH, Links TP (2008) The follow-up of patients with differentiated thyroid cancer and undetectable thyroglobulin (Tg) and Tg antibodies during ablation. Eur J Endocrinol 158(1):77–83
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
IRB ethical clearance has been obtained for the study (ECR/386/Inst/KA/2013/RR-19).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Prasad, R., Rao, V., Subash, A. et al. Residual Thyroid Tissue on Postoperative Diagnostic 131 I Radioactive Whole-Body Scan After Surgery in Differentiated Thyroid Cancer: A Tertiary Referral Centre Experience. Indian J Surg Oncol 13, 109–114 (2022). https://doi.org/10.1007/s13193-021-01357-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13193-021-01357-x