Abstract
Chronic brain injury following cerebral ischemia is a severe debilitating neurological condition, where clinical intervention is well known to decrease morbidity and mortality. Despite the development of several therapeutic strategies, clinical outcome in the majority of patients could be better improved, since many still face life-long neurological deficits. Among the several strategic options that are currently being pursued, tissue engineering provides much promise for neural tissue salvage and regeneration in brain ischemia. Specifically, hydrogel biomaterials have been utilized to docket biomolecules, adhesion motifs, growth factors, and other proneural cues for stable stem cell encapsulation. Here, we provide an overview of therapeutic applications of hydrogels in stroke treatment. Special focus is given to design considerations for generation of efficient hydrogel systems for neurological applications. Therapeutic applications of hydrogels in stroke as conducive microenvironments for stem cell transplantation and drug delivery have been discussed. Finally, we present our perspectives on clinical translation of hydrogels for neural tissue regeneration.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Ischemic stroke is a major cause of death and morbidity worldwide [1], with cerebral ischemic events accounting for 85% of all stroke conditions [2]. It is characterized by vascular occlusion that causes inadequate perfusion of oxygenated blood and depleted supply of nutrients to the brain [3]. The hypoxic/ischemic condition initiates a cascade of neuropathological pathways in hypoperfused tissue that results in an extensive loss of cerebral parenchyma, which in turn causes impairment of brain functions and severe neurological disabilities [4]. Considering the multitude of pathways involved in tissue damage, the clinical management of stroke addresses strategies that primarily involve early restoration of focal blood flow (recanalization), neuroprotection, and stimulation of neuronal regeneration [5, 6]. Even though there has been a strong push in research, focusing on signaling pathways of angiogenesis, neuronal regeneration, and repair following cerebral ischemia, the complexity of brain physiology and the hostile environment of the ischemic penumbra pose potential impediment in successful clinical translation [7].
Ongoing research on combinatorial neuroregenerative strategies based on stem cell therapy and delivery of biomolecules have shown improvement in neural regeneration following stroke [8, 9]. Exogenous stem cell transplantation restores or repairs damaged brain tissue by providing a conducive environment for regeneration [10], while the delivery of growth factors was found to further enhance neurogenesis and angiogenesis by facilitating endogenous brain restorative mechanisms [11, 12]. However, conventional oral and intravenous delivery routes for the administration of these molecules are hampered by their limited diffusion across the blood-brain barrier (BBB) [7]. Furthermore, the efficacy of stem cell therapy in neural tissue regeneration is limited by low survival rates of transplanted stem cells in the infarct cavity, inadequate migration of stem cells into the lesion site, differentiation of stem cells into glial lineage [13], and poor integration of transplanted cells with the injured brain tissue [14, 15]. Combining controlled delivery of drug or biomolecules, and stem cell transplantation within an engineered matrix that supports cellular interactions, while mimicking the native neural tissue environment would be a significant development in the management of stroke.
Recent advance in tissue engineering addresses challenges in clinical translation of stem cell- and biomolecule-based stroke therapy. Tissue engineering combines stem cells and biomolecules with biomaterials to facilitate restoration of tissue integrity, by attempting to produce fully functional three-dimensional (3D) tissues. It is now becoming clear that for restoration of functional tissue, three principal components need to be involved, viz., (a) implantation of cell/stem cell-laden biomaterial construct into the site of injury/repair within the host organism, (b) sustained delivery of tissue-specific growth factors to the site of implantation, and (c) provision of physiochemical cues of the integration of tissue construct [16]. Along these lines, various protein-, peptide-, polysaccharide-, polymer-, and ceramic-based microporous, nanofibrous, and hydrogel biomaterial scaffolds have been investigated as substrates for developing functional tissues [17,18,19,20,21,22,23,24,25]. Engineered tissues such as autologous bladders, skin grafts, fat grafts, bone and cartilage grafts, heart valves as well as a whole trachea have been implanted in patients [26,27,28,29,30]. So far, nanoparticle-based drug/stem cell delivery or hydrogel microencapsulation of neurotrophic factors and small molecules has shown much promise as potential therapeutic option for ischemic stroke [31,32,33,34]. Development of recent technologies of decellularized extracellular matrix (ECM) scaffolds [35, 36] and 3D bioprinting of hydrogels [37,38,39] with a focus on mimicking the physicochemical properties of the in vivo architecture also holds promise of generating functional scaffolds for central nervous system (CNS) applications.
Tissue-engineered biopolymer-based scaffolds recreate the native tissue microenvironment to support stem cell transplantation, integration, and survival in vivo [40]. The high biocompatibility, ease of functionalization, and precise control over physical properties make biopolymer hydrogels excellent candidates for tissue engineering [41]. They provide tunable substrates for cell encapsulation and site-specific spatiotemporal sustained release of therapeutic molecules, thus facilitating tissue regeneration [42]. This review will focus fundamentally on tissue engineering strategies using hydrogels in stroke therapy with special emphasis on the criteria for designing hydrogels for neurological applications and hydrogels as scaffolds for stem cell transplantation and biomolecule delivery.
Hydrogels: Extracellular Matrix (ECM) Mimicking Cross-Linked Networks
Hydrogels are scaffolds formed by a network of cross-linked polymeric units with high water content (> 90%) and exhibiting a 3D structure that is analogous to native tissue ECM. These gel systems support cell survival and growth by facilitating efficient transfer of oxygen, nutrients, and other soluble factors by diffusion, while providing a microenvironment with characteristic architecture and mechanics of ECM [43]. Hydrogels can be broadly classified as natural and synthetic hydrogels. Natural hydrogels, as the name suggests, are derived from natural polymers such as hyaluronic acid (HA), collagen, fibrin, gelatin, and alginate, while synthetic hydrogels are those derived from synthetic polymers or chemically modified natural biopolymers [44]. When compared to naturally derived hydrogels, synthetic polymers facilitate precise control over synthesis, i.e., polymerization and gelation parameters, bulk properties, microarchitecture, and degradation rates of the hydrogel [45]. To this end, synthetic hydrogels have been efficiently tailored with incorporation of biological signals to support cell growth and proliferation [46]. These characteristics are highly advantageous while designing scaffold systems to assist cell transplantation in biomaterial-based stem cell therapy.
Owing to their ability to recapitulate the native tissue microenvironment and thus instruct stem cell fate, over the past several decades, hydrogels have been extensively applied in stroke treatment for neural tissue regeneration [47]. Peptide molecules, growth factors, and glycosaminoglycans (GAGs) can be used to functionalize hydrogels to support growth and survival of transplanted stem cells or stimulate neural repair by augmentation of endogenous stem cells [48]. Hydrogels having mechanical properties tailored to encompass the range of compressive moduli of brain tissues can be efficiently implanted into the injury site without much damage to the surrounding soft tissues [49]. Although preformed implantable hydrogels have been used as permissive scaffolds for neural regeneration [50, 51], the lack of flexibility to integrate into the injury site upon delivery might further aggravate tissue damage [52]. Minimally invasive in situ forming injectable hydrogels present a promising platform for localized therapeutic application in stroke injury [53]. They find application in stem cell delivery and drug delivery to stimulate neurogenesis and angiogenesis and enhance endogenous repair. Injectable hydrogels usually have low elastic moduli comparable to that of native brain tissue [54], that enhances neural differentiation of stem cells.
Designing Hydrogels for Neurological Applications
The complex extracellular matrix of the brain is a milieu of interacting molecules and cells including neurons, astrocytes, microglia, and oligodendrocytes that pose a formidable challenge for the in vitro reconstruction of the native tissue microenvironment [55]. Scaffold must be engineered to comprehend this complexity while maintaining the physicochemical properties of the native tissue. Hydrogels can be tailored to emulate characteristics of the brain ECM and thus promote stem cell adhesion, proliferation, and differentiation. Hence, it is important to understand how certain attributes of hydrogel facilitate stem cell survival and regulate its fate in vitro [56,57,58,59]. In the following section we highlight the impact of material and scaffold properties of hydrogels such as biocompatibility, biodegradability, functionalization, elasticity, conductivity, porosity, and pore size as well as response to stimuli, in designing effective substrates for neural tissue regeneration (Table 1).
Biocompatibility and Biodegradability
For successful delivery of stem cells or biomolecules to the injured brain, hydrogel should be biocompatible at two levels: cytocompatible with the encapsulated cells and histocompatible with the host tissue [85]. Long-term biocompatibility correlates with the efficiency of implantation. Brain tissue damage triggers inflammatory response that adversely affects the survival of transplanted cells. Hydrogels being biologically inert evade the immune system and enhance graft survival during postimplantation neuroimmune response [86, 87]. Implantation of chemically biocompatible and mechanically stable hydrogels with nontoxic degradation products reduces infiltration of activated macrophages and microglia into the site of injury [88].
Polymerization and gelation methods employed in the synthesis of hydrogels also affect its biocompatibility. Temperature-dependent and pH-dependent polymerization under physiological conditions are ideal for in situ forming injectable hydrogels that can be delivered by minimally invasive methods [89]. Injectable hydrogels with elastic moduli comparable to that of the brain tissue evade the BBB and efficiently fill the infarct cavity. In addition, for the injected hydrogel to fill the lesion defect and stay loculated within the site, the gel must be designed such that it undergoes moderate to rapid gelation as soon as it comes in contact with the surrounding tissue. Moreover, gelation has also been shown to be affected by the presence of extracellular fluid (ECF) within the lesion site. ECF is known to interfere with physicochemical properties of the hydrogel, and hence, it is considered ideal to drain the ECF prior to injection of the hydrogel to prevent change in its bulk properties [90].
Biodegradable hydrogels degrade slowly over time within the host tissue without causing tissue scarring and glia formation [85]. For neural applications, hydrogels are often functionalized with peptides for cell-mediated enzymatic degradation for site-specific delivery of encapsulated cells and growth factors [91]. Degradation kinetics of a hydrogel is crucial in determining its functionality and host tissue response. A slow degrading hydrogel supports transplanted cell survival, ECM development, and host tissue integration, while a fast degrading hydrogel reduces the risks associated with immune response. Hence, it is essential to balance the cell supportive nature of the hydrogel while evading the immune response [90].
Functionalization
The brain has a specialized ECM rich in proteoglycans, glycosaminoglycans, and proteins such as laminin, collagen, and fibronectin, that regulate cell adhesion, migration, and neurite process extension and, thus, contributes to brain tissue homeostasis [92]. Many of the biopolymer hydrogels used in neural tissue engineering (e.g., alginate, agarose, poly(ethylene glycol) (PEG)) require functionalization to make them conducive for cell growth, proliferation, and differentiation [93, 94]. Hence, neural ECM components are potential candidates for scaffold functionalization as they are biocompatible, contain adhesion moieties, and also support cell growth and differentiation [95] (Table 1). Laminin and its derivatives, peptide molecules containing sequences such as YIGSR and IKVAV, and neural cell adhesion molecule (N-CAM) are few examples of neural ECM components that are known to stimulate neurite outgrowth [96], enhance neuronal survival [97], and promote cell adhesion [98].
Such peptide moieties have been previously incorporated into hydrogels in ischemic stroke models. Multifunctionalized HA hydrogel was designed for transplantation of human neural progenitor cells (hNPCs) into peri-infarct and infarct stroke regions of mice brain [60]. The hydrogel was modified with IKVAV, YIGSR, and RGD adhesion moieties; cross-linked with matrix metalloproteinase (MMP)-sensitive motifs; and incorporated with heparin-bound brain-derived neurotrophic factor (BDNF) and bone morphogenic protein-4 (BMP-4). The complexly designed hydrogel supported hNPC survival, transplantation, and differentiation in vivo, which could be tracked using magnetic resonance imaging (MRI). Similarly, simple RGD-modified injectable HA hydrogels have been used to deliver induced pluripotent stem cell (iPSC)-derived NPCs (iPS-NPCs) into stroke cavity [61]. Though the hydrogel did not support cell survival 1 week postimplantation, it facilitated differentiation of functionally viable cells. Another variant, HA hydrogel, with optimized concentrations of RGD, YIGSR, and IKVAV ECM peptide facilitated survival and differentiation of iPS-NPCs [62]. HA hydrogel surface functionalized with a peptide containing the IKVAV sequence, upon implantation in rat brain, repaired the tissue defect and integrated with the host tissue [63] by stimulating cell infiltration and vascularization. In another study, alginate-poly(γ-glutamic acid) composite hydrogel was functionalized with TATVHL peptide for iPSC adhesion, and the composite hydrogel promoted differentiation of iPSC into neuronal lineage [64]. Similarly, arginine-glycine-aspartic acid (RGD)-modified elastin-like peptides (ELPs) containing lysine enhanced neurite growth and provided tunable 3D microenvironment for neurite extension [65].
Biosynthetic designer peptide sequences are capable of self-assembling to form scaffolds to facilitate neural stem cell (NSC) attachment and differentiation. A self-assembling IKVAV peptide-based gel has been shown to support long-term survival and functional maturation of human pluripotent stem cell (hPSC)-derived cortical neurons that were implanted into the site of ischemia injury [21]. The peptide-based scaffold reduced host tissue atrophy and improved motor functions. Similarly, RADA16-IKVAV self-assembling peptide hydrogels increased the survival of encapsulated NSCs and improved brain tissue regeneration while reducing the formation of glial astrocytes [66]. Moreover, modified RADA16 hydrogels have also been shown to be angiogenic in nature. RADA16 functionalized with self-assembling peptides, PRG and KLT, enhanced tubulogenesis in human umbilical vein endothelial cells (hUVECs) [67] and stimulated endothelial cell migration, sprouting, and vessel formation in chicken embryo chorioallantoic membrane (CAM) [68]. Thus, these angiogenic hydrogels might have the potential to induce endogenous repair of ischemic penumbra.
These studies establish the fact that there is a constant crosstalk between the cells and the ECM molecules which governs cell fate. The presence of ECM molecules that facilitate cell adhesion, growth, and differentiation is an important factor that needs to be considered while designing hydrogels for cell transplantation into the brain.
Elasticity
Elasticity is an essential parameter to be considered while designing hydrogels for neurological applications. It contributes significantly not only in regulating the fate of encapsulated cells and the host integration but also in the distribution of hydrogel at the injury site [85] (Table 1).
Elastic modulus (E) of brain tissue ranges from 0.5 to 10 kPa observed within mechanical microenvironments from NSCs to blood vessels, glial scar, and gray and white matter [69]. The elasticity of cell-culture substrate is well known to regulate stem cell differentiation into neural lineage [99]. In vitro studies have shown that softer substrates best support NSCs, neurons, and glia. Pliable and compliant hydrogel matrices (E = 0.5–1.5 kPa) favored neuronal attachment and growth, while stiffer hydrogels (E = 7.2 kPa) supported astrocyte survival [49]. Similarly, adult neural stem and progenitor cells (NSPCs) responded to hydrogels with elastic moduli ranging from E = 0.01 to 10 kPa by differentiating into neurons on softer surfaces and into glial cells on harder substrates [69]. Polyacrylamide and fibrin hydrogels of stiffness 250 Pa supported neuronal growth, while stiffer hydrogels of 9100 Pa supported astrocyte growth and distribution [70]. Similarly, the stiffness of methacrylamide chitosan (MAC) hydrogel has been shown to have varying effects on differentiation and proliferation of NSPCs [71]. Neuronal differentiation of NSPCs was better on softer surface (E < 1 kPa), while oligodendrocytes were favored on stiffer hydrogels (E = 7 kPa). Additionally, NSPCs displayed maximum proliferation when cultured on gels with an intermediate stiffness (E = 3.5 kPa). Thus, subtle variations in the mechanical properties of the substrate might translate significantly with respect to stem cell fate, which needs to be taken into account when designing hydrogels for specific neurological applications.
Since ischemic stroke can lead to the formation of lesion sites with large volume and irregular shape, it is essential to design in situ forming injectable hydrogels that efficiently fill lesion cavity [100]. Recently, rheological properties and in situ gelation of urinary bladder matrix (UBM)-ECM hydrogel were analyzed for application in stroke treatment [100]. Gelation and retention of the ECM hydrogel within the stroke cavity was dependent on its concentration. At higher ECM concentrations (> 3 mg/mL), hydrogel was retained efficiently within the stroke cavity, and at 8 mg/mL concentration, the elastic moduli of the hydrogel were found to be 500–1000 Pa, which is similar to that of brain tissue.
Porosity and Pore Size
Hydrogels are complex macromolecular systems with finite pore structures tailored to enhance tissue regeneration by augmenting cell infiltration and local angiogenesis, by establishing tissue ingrowth, and by stimulating cellular interactions with the host system. Tissue engineering strategy in ischemic stroke treatment fundamentally aims at healing the injured tissue by promoting cell growth and axonal regeneration within an engineered 3D tissue construct. The pores within the hydrogel create a permissive interface for cell migration toward and within the 3D network, thus enabling tissue regeneration and integration with the host system [101]. Hydrogel matrices can be designed with various pore microgeometries found within the biological tissue structures such as macroporosity for tissue expansion and micro- and mesoporosity for tissue interface at cellular and molecular levels (Table 1). Not only do these parameters influence the neural ingrowth and homogeneity of cell distribution within the hydrogel matrix but also enable efficient oxygen and mass transfer in the absence of functional vasculature [102].
Open porosity defines the ratio of accessible pores to the total gel volume [103], which is the effective porosity available for cell migration and infiltration to form an interface with the host tissue. An early study on the effect of porosity of poly(glycol monomethacrylate) gels on healing-in of implants revealed that gels with higher porosity had broader zone of cellularization and neovascularization at the interface of the implant [104]. Similarly, varying the concentration of the monomer and crosslinker to monomer ratio had an effect on the porosity within a narrow range of crosslinker concentration in poly(glyceryl methacrylate) (p(GMA)) hydrogels [72]. It was also shown that by increasing crosslinker concentration, the porosity of the macroporous p(GMA) gel increased greater than 50% of gel volume. Furthermore, p(GMA) hydrogels with pore size greater than 10 μm enabled tissue ingrowth, while poly(2-hydroxyethylmethacrylate) (p(HEMA)) hydrogels with pore size less than 10 μm induced capsule formation and isolation of the implant from the host neural tissue.
Furthermore, other attributes such as pore diameter, pore surface area, and void volume are crucial in sustaining cell migration and attachment as well as tissue formation and tissue expansion within the 3D matrix of the hydrogels [101]. Various studies on the effect of scaffold microarchitecture on tissue regeneration has validated the optimal pore size of 5 μm for neovascularization, 20–125 μm for mammalian skin and 100–350 μm for bone regeneration, 5–15 μm for fibroblast, 40–100 μm for osteoid, 100 μm for neuronal, and 20 μm for hepatocyte ingrowths [105,106,107,108,109,110,111]. The degree of porosity also affects the mechanical strength of the hydrogel, i.e., the gel strength decreases as porosity increases, making them more susceptible to deformation caused by fluid flux [112].
Posttraumatic neural tissue regeneration requires intimate cellular interactions. Macroporous poly(lactide-co-glycolide) hydrogels were developed with the aim to enhance the hydrogel pore interconnectivity [73]. Highly interconnected pores (pore size 78 μm) were formed by fusion with NaCl porogen prior to hydrogel matrix formation and subsequent removal of porogen upon formation of 3D matrix. Such tissue-engineered porous scaffolds with controlled pore interconnectivity might help permit vascular and tissue ingrowth and prevent pruning of regenerating axonal fibers. Porous network structures of poly(N-2-hydroxypropyl methacrylamide) (PHPMA) hydrogels were characterized and analyzed for their ability to induce tissue growth and axonal regeneration in spinal cord injury [74]. The developed PHPMA hydrogel showed neuroinductive and neuroconductive properties similar to that of native neural tissue. Characterization of pore structure with mercury porosimetry and diffusion analysis revealed that the PHPMA hydrogel comprised of an extensive network of interconnected open pores of the size 10–30 μm in diameter. The porous structure enabled the gel to form a permeable interface with the host tissue and allowed spatial orientation and expansion of ingrown tissue. The hydrogel system was able to bridge the defective tissues and supported angiogenesis and axonogenesis, in addition to cellular growth within the polymer matrix. HA hydrogels with IKVAV peptides formed porous and permeable substrate (pores ranging from 10 to 100 μm) for glial tissue formation and enabled lesion cavity bridging by inducing neuronal ingrowth within the porous matrix [63]. An interconnected macroporous conductive PANI/poly(ethyleneglycol diacrylate) (PEGDA) hydrogel was designed using UV photocrosslinking and salt leaching to recapitulate the microenvironment necessary for neural regeneration. The addition of PANI slightly increased the porosity, while there was a marked decrease in the pore size from (158 ± 9) to (136 ± 13) μm. The porous scaffold facilitated efficient charge transfer, protected the cells from mechanical stress, and enabled homogenous tissue formation as well as transportation of nutrients and molecular signals.
The precise control of gel porosity is essential for regulating the development of functional tissues. Generation of porous hydrogels depends primarily on the methods of gel preparation. Techniques such as introduction of porogen agents, solvent casting, electrospinning, and gas foaming are commonly used for the preparation of macroporous hydrogels for [113]. Novel methods of micromolding and micropatterning not only offer well-defined control over pore features and overall gel microarchitecture but also create microvascular features for better gel integration with the host tissue [114,115,116,117,118,119]. Dynamic mask projection photolithography was used to generate PEG hydrogels of void size ranging from 62 to 206 μm, as biomimetic models for neurite outgrowths [120, 121].
Porosity and other parameters of pore microgeometry combine to create a 3D construct that allows cell permeation, orientation, and tissue development. Thus, the control of porosity and pore size is important for optimal neural tissue regeneration.
Stimuli-Responsive Hydrogels
Though hydrogels have been designed to mimic stem cell niche, there is a need for stimuli-responsive hydrogels that can better imitate the native tissue microenvironment. Drawing inspiration from nature, scientists have been designing stimuli-responsive or ‘smart’ hydrogels that perceive and respond to external stimuli, which includes physical stimuli such as temperature, light, and electric field; chemical stimuli such as pH, ionic strength, glucose, etc.; and biological stimuli such as enzymes, DNA, glutathione, etc. [122] (Table 1). In response to environmental stimuli, these polymeric systems undergo drastic change in physical configuration or chemical behavior [123]. Stimuli-responsive change in shape, surface characteristics, solubility, sol-gel transition, and formation of molecular self-assembly of hydrogels has enabled their application in tissue engineering, drug delivery, gene therapy, bioseparators, sensors, and actuator systems [124].
Thermosensitivity
Thermosensitive hydrogels respond to change in temperature by exhibiting a phenomenon called sol-gel transition, where the polymer switches from a solution form to a gel form [125]. Above a particular temperature, some polymers become hydrophobic and insoluble and separate out forming a hydrogel. This threshold temperature is called lower critical solution temperature (LCST). Below LCST, strong hydrogen bonding between the polymeric chain and water leads to solubility of polymer in water [123]. Above LCST, hydrophobic interactions are predominant, leading to gelation of the polymer. In contrast, polymeric solutions having upper critical solution temperature (UCST) form a gel upon cooling [126]. Poly(N-isopropylacrylamide) (pNIPAAm), methylcellulose, PEG-poly-l-(lactic acid) (PLLA-PEG) tri-block polymer, and poly(ethylene oxide)-b-poly(propylene oxide)-b-poly(ethylene oxide) (PEO-PPO-PEO) (Pluronics) are polymers that have been investigated for minimally invasive delivery of cells and biomolecules, as they exhibit in situ gelation in response to difference in ambient and physiological temperature [125, 127, 128]. Thermoresponsive pNIPAAm-grafted PEG (PNIPAAm-PEG) was used as injectable scaffold for the treatment of spinal cord injury [75] (Table 1). Below its LCST, at around 29–32 °C, the polymer was miscible in water, but above its LCST, it became hydrophobic, separating out from water and forming a semiporous gel. The porous nature of the scaffold enabled the incorporation of stem cells and sustained the release of neurotrophic factors. The swelling kinetics of the gel revealed that upon gelation, the hydrogel was able to retain 90% of its volume for a long period of time (14 days) without swelling or shrinkage. Thus, the polymeric system might be useful for delivering stem cells and growth factors, as it can form a multifunctional scaffold upon injection into the site of injury. Similarly, laminin-functionalized methylcellulose thermoresposive hydrogel was developed for neural tissue engineering [76]. The functionalized methylcellulose hydrogel was stabilized by the secondary inter- and intramolecular hydrophobic interactions, that increased with the increase in temperature. It was observed that there was a decrease in LCST with the increase in polymer concentration. The swelling and degradation studies along with cell viability analysis demonstrate that this thermoresponsive hydrogel might be a robust cell delivery system for CNS applications.
pH Sensitivity
Polymers containing ionizable functional groups such as carboxyl and amino, that respond to change in pH, form pH-sensitive hydrogels [122]. The mechanism of pH-responsive hydrogels is based on swelling or contraction of polymeric network depending upon the pH of the surrounding environment [129]. At acidic pH, amino/imine groups on the polymer chains of cationic hydrogels like chitosan, polyacrylamide, and poly(ethylene imine) [130,131,132] get protonated and cause repulsion, resulting in swelling of the polymeric system. In contrast, at basic pH, ionization of acidic groups on the polymer chains of anionic hydrogels such as carrageenan, carboxymethyl chitosan, and poly(acrylic acid) causes repulsion which leads to swelling of the hydrogel [122].
Following an ischemic cascade, CO2 accumulates within the intercellular space resulting in reduction of pH in the affected area [133]. Furthermore, reduced oxygen and glucose supply leads to depletion of glycogen and phosphocreatine, which increase H+ ion production, thus creating an intracellular acidic pH of 6.3–6.4. In a recent study, the reduced pH with the ischemic area was utilized to deliver IgG antibody, that was organically conjugated to HA using hydrazone linkage to form a pH-sensitive hydrogel [77]. It was observed that at lower pH, acidic cleavage of hydrazone bond between the IgG and HA resulted in the rapid release of IgG when compared to neutral and alkaline pH. Within the in vivo model, IgG release was observed to be much rapid at lower pH, which then slowed down when restored to physiological pH. Likewise, a dual ionic pH-sensitive copolymer was designed for the delivery of stromal cell-derived factor 1α (SDF-1α) to the ischemic site [78]. At alkaline pH, SDF-1α binds to pH-sensitive poly(urethane amino sulfamethazine) (PUASM), and at pH 7.4, the anionic sulfonamide groups of PUASM deionize while the cationic tertiary amine groups are unionized. Consequently, the PUASM form the hydrophobic core encapsulating SDF-1α, while PEG forms the hydrophilic shell, protecting the complex from proteolytic digestion. At pH below 5.5 and above 8, the amine groups of PUASM get protonated and release SDF-1α by electrostatic repulsion. Such pH-sensitive hydrogel might be useful in modifying the tissue microenvironment postischemic injury so as to induce innate regeneration.
Electrical Conductivity
Studies have shown that electrical stimulation can augment endogenous brain repair following an ischemic stroke insult, mainly by inducing the proliferation of hNPCs and stimulating the production of trophic factors such as EGF and FGF within the infarct cavity [134]. Emerging technologies in tissue engineering have incorporated electrical conductivity to polymeric systems to form conductive hydrogels for CNS applications [135] (Table 1). Recently, mechanically stable polypyrrole (PPy) conductive hydrogels were developed for electrical preconditioning of hNPCs to enhance their efficiency of implantation within the peri-infarct area of the cortex into the peri-infarct cortex of stroke-injured rat brain [79]. Electrical stimulation not only enhanced stroke recovery by upregulating vascular endothelial growth factor (VEGF) expression but also induced cortical remodeling and endogenous vascularization via secreted factors. To address the poor mechanical stability of conventional conductive hydrogels, heparin methacrylate (HepMA) was doped within poly(vinyl alcohol) (PVA) hydrogel and electrodeposited with poly(3,4-ethylenedioxythiophene)-doped paratoluenesulfonate (PEDOT/pTS) to form an interpenetrating network (IPN) conducting system [136]. The hybrid system had enhanced mechanical properties comparable to native neural tissues and also supported growth of PC12 cell line. Similarly, PVA macromers were modified with methacrylate and taurine to form pliable IPN conducting hydrogels [137]. Conducting polymers such as polyaniline (PANI) and PEDOT have been utilized in hydrogels and have been found to modulate stem cell fate toward neural lineages [80,81,82], thereby strongly supporting their potential beneficial role in stroke. However, more studies are warranted to validate these compounds, since many of the conducting polymers have been shown to have some degree of cellular toxicity [138,139,140].
Light Sensitivity
Light-responsive hydrogels comprise of photochromes such as azobenzenes and spiropyrans incorporated within the polymeric backbone that induces volume change in response to light [141]. Spiropyran derivaties can be used to create a significant change in gel dimension [142]. Using light stimuli, stable hydrophilic merocyanine-H+ isomer formed in acidic environment can be switched to hydrophobic spiropyran form, thus changing the overall property of the hydrogel, allowing swelling and shrinkage of the material [141]. Agarose hydrogels modified with photolabile S-(2-nitrobenzyl)cysteine (S-NBC), upon irradiation, exposed sulfhydryl groups which when activated allowed immobilization of GRGDS (Gly-Arg-Gly-Asp-Ser) peptide for adhesion of and neurite outgrowth from embryonic chick dorsal root ganglion neurons [83]. Light-responsive hydrogels have the potential for being biosensors and microfluidic valves, as well as in photoregulated drug delivery [143]. Photolithographic techniques can be used to micropattern photoresponsive hydrogels [144]. Cell adhesive photocaged RGDS (Arg-Gly-Asp-Ser) peptide were micropatterned on HA hydrogel using near UV light through a patterned photomask [84]. Fibroblasts adhered and proliferated along the micropatterns, thus demonstrating control of spatial localization of cells using patterned protein gradients.
Given the possibility that a single factor does not contribute to the hydrogel’s functionality, rational designing of stimuli-responsive, porous, and biocompatible hydrogels with efficient degradation kinetics and mechanics comparable to that of the host brain tissue is of utmost importance.
Therapeutic Potential of Hydrogels
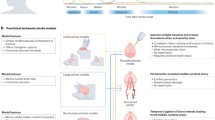
The following section discusses the application of hydrogels in the delivery of growth factors and stem cells, to promote tissue regeneration following ischemic stroke injury (Fig. 1; Table 2).
Hydrogel Mediated Growth Factor Release for Endogenous Repair
Though the innate mechanisms in the brain trigger restorative pathways, endogenous neurorestoration is inadequate to compensate for the deleterious stroke condition. Therapeutic applications targeting growth factors may provide a prolonged therapeutic window to restore the stroke-damaged tissue endogenously. Till date, erythropoietin (EPO) and granulocyte colony-stimulating factor (G-CSF) are the only trophic factors applied clinically [11], since systemic administration of most growth factors requires high concentrations in order to cross the BBB and may cause systemic toxicity due to their off-target distribution [7]. Thus, there is a need for directed delivery of growth factors to the injury site while maintaining their sustained release. The tunable mechanical properties, porosity, and functionality of tissue-engineered hydrogels make them excellent scaffolds for site-specific delivery of neurotrophic factors. Since stroke injury results in irregular lesion cavities, injectable hydrogels are often desirable for minimally invasive delivery of growth factors and cells to the stroke cavity [165] (Fig. 1).
Growth factor-incorporated injectable hydrogels have been extensively studied at the Shoichet Lab as an alternative to conventional bolus delivery for endogenous tissue regeneration (Table 2). In several studies, minimally invasive epicortical delivery of therapeutic factors has proven to be efficacious. Recently, epicortically injectable hyaluronan/methyl cellulose (HAMC) hydrogel was used to deliver cyclosporin A (CsA) to stroke-injured cortical region of adult mice [145, 146]. Sustained release of CsA from poly(lactic-co-glycolic acid) (PLGA) microspheres along with HAMC significantly reduced the infarct volume and enhanced the proliferation of NSPCs within the NSC niche, when compared to minipump delivery of CsA. Similarly, HAMC hydrogel was employed for EPO delivery to attenuate tissue inflammation, stimulate migration of NSPCs and mature neuroblasts, and reduce apoptosis in the subventricular zone and injured cortex region [147]. Sequential combinatorial application of EPO and EGF using HAMC hydrogels composed of EPO-loaded PLGA microparticles and EGF-PEG nanoparticles stimulated endogenous stem cells and facilitated neural regeneration [12, 166]. In the same study, PEGylation enhanced the protein stability, bioactivity, and tissue penetration rates, while EGF stimulated neurogenesis in mice models. Taken together, these results suggest that injectable hydrogels can be excellent carrier systems for bioactive molecule delivery in stroke treatment.
BDNF has shown to enhance sensory motor recovery, reduce infarct volume, and stimulate NSC migration in several in vivo preclinical studies [167, 168]. However, its low diffusion potential across the BBB and its limited tissue distribution upon delivery make intravenous administration of BDNF unfeasible [169]. Depot release of BDNF from hyaluronic acid hydrogel enhanced tissue repair and recovery of motor function in primate and mice stroke models within 3 weeks after injection into cortical and subcortical infarct cavity [170]. Thiolation of hyaluronic acid facilitated sustained release of BDNF, which slowly diffused into the peri-infarct tissue and induced infiltration of immature neurons. VEGF is another growth factor that has proven to increase BBB permeability, enhance angiogenesis, and reduce neurological defects in ischemic brain [171]. Similar to BDNF, the short half-life and high dosage concentrations limit the practical application of VEGF, leading to a need for hydrogel-based systems that facilitate their site-specific and controlled delivery. VEGF was incorporated in injectable alginate hydrogels to ameliorate neuronal degeneration following cerebral ischemia [172]. Alginate-VEGF hydrogel stereotaxically injected into the striatum of adult mice brain reduced the lesion size and attenuated behavioral defects. Similarly, implantation of polydimethysiloxane-tetraethoxysilane (PDMS-TEOS)-VEGF hybrid hydrogel increased the number of astrocytes and endothelial cell infiltrated into the stroke cavity, suggesting that PDMS-TEOS-VEGF hydrogels might have therapeutic potential in treating cerebral infarction [173]. Likewise, VEGF and angiopoietin-1 (Ang1) releasing HA-PLGA hydrogels promoted neural regeneration following ischemic injury [174]. The novel composite reduced the inflammation, enhanced angiogenesis, and facilitated behavioral improvements in vivo.
Stroke is a complex neurological condition wherein a combinatorial growth factor therapy is essential to enhance angiogenesis, neurogenesis, and axonal sprouting and to restore neuronal functions. Development of hydrogel scaffolds that conforms to the infarct region and facilitates spatiotemporal sustained release of multiple growth factors would be a vital step in stroke therapy.
Hydrogels as Conducive Microenvironment for Stem Cell Transplantation
During an ischemic stroke, the central area of the lesion that experiences severe hypoxia is rapidly and irrevocably damaged with necrotic cell death [175]. Parallelly, ischemia-related cascade of cellular events is initiated in the structurally intact penumbra, which becomes the site for stroke progression and therapeutic salvage [176]. Stimulating neuronal regeneration within the salvageable penumbra that is highly complex in space and time is a major challenge in tissue engineering. While stem cell therapy is still in its infancy, it holds promise as a means to restore brain function after the stroke episode [177] (Table 2). Engineering hydrogel-based biomaterial interface that supports stem cell engraftment, viability, proliferation, and functionality in vivo is cardinal in the translation of cell-based therapy (Fig. 1). NSPCs encapsulated in HA/heparin/collagen hydrogel supported the survival of NSPCs in vivo and further reduced the infiltration of inflammatory cells when transplanted into the infarct cavity [88]. Recently, self-healing injectable carboxymethyl chitosan and oxidized alginate hydrogel with elastic moduli comparable to brain tissue were demonstrated to be viable 3D carriers for NSC transplantation [178]. Likewise, human embryonic stem cell (ESC)-derived NPCs when injected with Matrigel in vivo decreased the volume of the necrotic infarct cavity and enhanced neuronal differentiation and behavioral outcomes [179].
Recent studies have shown that substrates that enhance neuroblast migration and differentiation within stroke lesion can improve neuronal regeneration in poststroke brain injuries. Injectable self-assembling laminin hydrogels promoted vasculature-mediated neuronal migration toward injured tissue lesions by activating the β1 integrin signaling pathway [180]. Similarly, 3D HA hydrogels embedded with aligned nanofibers composed of polycaprolactone (PCL), PCL-gelatin, and laminin-coated PCL have shown to support encapsulated neuronal cell survival, neurite alignment, and extension [181].
Protein-based hydrogels have also found application in cell-based stroke therapies. Genipin cross-linked sericin hydrogel (GSH) was found to be a potential cell carrier for tissue regeneration after ischemic injury [148]. The porous structure of GSH was conducive for neuron attachment and growth, while the neurotropic and neuroprotective sericin facilitated axonal branching and extension while rescuing immature neurons from hypoxic cell death. Fibrin is an important ECM protein and has shown to provide permissive 3D scaffolds for human endometrial stem cells (hEnSCs)-derived neuron-like cell survival and tissue regeneration [182]. A recent study reported the development of salmon fibrin/HA/laminin composite scaffold with prolonged degradation kinetics and higher biocompatibility and that maintained hNPSC function compared to fibrin [95]. Similarly, iPSCs mixed with fibrin glue upon subdural transplantation into focal ischemic infarct region in vivo reduced infarct volume, recovered neurological and behavioral functions, and was neuroprotective in nature [183]. To protect Schwann cells (SCs) from hypoxia-induced cell death during ischemia, fibrin gel containing perfluorotributylamine (PFTBA) was used as oxygen carrier [184]. SCs cultured in PFTBA-hydrogels overexpressed neurotrophic factors such as BDNF, GDNF, N-CAM, and VEGF. The hydrogel demonstrated its potential for neural tissue regeneration by supporting cell adhesion, migration, and proliferation.
ESCs and iPSCs are potential cell sources in neural tissue engineering. Nonetheless, scalability and dearth of well-defined culture strategies for PSC expansion pose major challenge to the in vitro and clinical application of PSCs. Application of hydrogel-based technologies for directed cell differentiation would be useful in addressing these predicaments. In a recent study, glycosaminoglycan ligand-bound polyacrylamide hydrogel of elastic modulus 10 kPa enhanced hESC adhesion and colony formation and maintained the state of pluripotency [185], while cell culture on hydrogels of 0.7 kPa resulted in robust neuronal differentiation with > 80% expression of neuronal markers. Similarly, PEG hydrogels of gradient of 1.4 kPa or less were able to differentiate hiPSC-derived neural stem cells to neuronal cells. Also gels with 970 Pa helped in the formation of significantly longer neurites [186]. These studies also highlight the effect of biomechanical cues of the microenvironment on stem cell fate.
Since stem cell-based therapy may potentially prove to be one of the best options for stroke treatment, it is essential to design hydrogel scaffolds that facilitate efficient delivery, in vivo survival, and differentiation of stem cells for successful clinical translations.
Imaging of Hydrogels
Evidently, real-time monitoring of stroke tissue infarct and its response to biomaterial-based regenerative therapy in vivo would be ideal to evaluate the therapeutic efficacy. Brain tissue imaging using MRI, positron emission tomography (PET), or bioluminescence imaging (BLI) helps in identifying salvageable ischemic penumbra and provides treatment for a larger proportion of stroke patients. PET enables the evaluation of stroke progression in vivo by delineating the ischemic penumbra [187] and facilitating targeted stem cell/drug delivery [188]. Experimental trials have been conducted using MRI to track the infarct region and volume of tissue loss, to monitor biomaterial grafted cells, and to assess their infiltration into stroke lesion [189]. Diamagnetic chemical exchange saturation transfer (CEST) MRI was used to track the ECM hydrogel injected into the stroke cavity [190]. Concentration-dependent CEST signals for chondroitin sulfate and fibronectin ECM hydrogel molecules were detected in vitro. In vivo imaging of the implant detected specific CEST signals corresponding to endogenous ECM molecules from the peri-infarct cavity. Thus, this technique can be used to selectively identify ECM hydrogel within the host tissue and monitor its in vivo distribution. Similarly, MRI was used to detect the oxygen levels in acute stroke model to identify ischemic core and evaluate the extent of necrosis [191] following which a HA-based hydrogel was used for transplantation of MSCs into the infarct cavity. A diffusion MRI tracking of transplanted hydrogel revealed that the hydrogel remained stable and supported MSC differentiation and angiogenesis. In vivo BLI has been used as a noninvasive imaging modality to detect cellular migration, proliferation, and survival of HA-xenografted hNPCs [192] within the stroke cavity. Recently, FITC-conjugated albumin containing injectable gelatin hydrogel with 3D solvent clearing and light sheet microscopy was tested on mice brain to visualize the microvessels within the ischemic regions [193]. Images of the brain vasculature demarcated the healthy tissue from the ischemic tissue. Analysis of vascular networks within the infarct identified small microvessels to be highly damaged by focal ischemia. This approach might have possible application in quantifying the extent of ischemic injury and analyzing the efficiency of angiogenic treatments. These studies clearly elucidate the importance of noninvasive imaging in validating the therapeutic efficiency of hydrogel biomaterial-based applications.
Conclusion and Perspectives
Ischemic stroke is a life-threatening neurological condition with limited therapeutic interventions. Given that stroke injury compromises neuronal regeneration and repair, tissue engineering strategies have focused on designing neuroprotective and neurorestorative hydrogel systems for targeted stem cell and bioactive molecule delivery. To this end, several studies have proven that in situ forming injectable hydrogels are optimal for therapeutic applications in stroke-injured brain. Designer-injectable gels in combination with stem cells and a range of pro-angiogenic and neurogenic biomolecules have neuroregenerative capabilities. Stem cells in combination with hydrogel scaffolds have potential therapeutic significance for the structural remodeling and functional recovery following stroke and other CNS diseases (Table 2).
Regardless of the fact that a gamut of tissue-engineered hydrogels have been studied for their remedial application in stroke, clinical translation of hydrogels in stroke treatment has a long way to go. The human brain is extremely complex and dynamic with varying tissue mechanical strength, multiple cell types, and ECM molecules. These factors need to be addressed systematically while designing hydrogels for neural applications for better clinical outcomes. The lack of spatiotemporal control within engineered hydrogel constructs results in nonuniform cell distribution and low cell penetration capacity along with poor vascularization and mass transfer leading to loss of cell viability and functionality upon transplantation [41]. Translation from small animal models to the human biological system, with its complex vasculature, innervation, and other micro- and macroarchitecture, poses a major challenge in hydrogel engineering. Furthermore, drug loading efficiency of hydrogel systems is often limited by their poor tensile strength, which also affects the hydrogel retention within the site of application [194]. Also, high porosity and limited control over the material properties in many hydrogels often leads to rapid initial burst release, which might deter the clinical application of hydrogels as sustained drug delivery systems [195]. Synthetic hydrogels tailored for specific applications are often associated with challenges such as the presence of toxic crosslinkers, unreacted monomers, high crystallinity, etc. [194]. A fine balance needs to be maintained between scaffold efficiency and safety without compromising on its functionality. While hydrogels can be engineered complying with the design constraints for neurological applications, its safety and feasibility are also of paramount significance. The scalability of the developed hydrogels for clinical translation also needs to be considered. There is a need to develop novel methods to harness the potential of biomaterials and stem cells to fabricate cytocompatible and efficacious tissue constructs. Engineering biomaterial constructs with sound knowledge about their chemical and mechanical properties, interactions at the cellular interface, immunogenicity, mass transfer efficiency as well as scalability and reproducibility is cardinal for clinical translation of tissue engineering technologies [196].
Despite the proven efficacy of stem cells, there are still ethical issues regarding their clinical applications. While hiPSCs are an answer to the ethical concerns and lack of patient specificity of stem cells, hiPSC generation and expansion is cumbersome and needs optimization [197]. Moreover, inefficient reprogramming efficiency of iPSCs, immunogenicity, and teratoma formation upon transplantation pose challenges to the clinical application of hiPSCs [198]. Overcoming these limitations depends on designing well-defined biomaterial-based cell culture strategies for iPSC derivation, expansion, and differentiation. Tissue-engineered scaffolds should not only be able to modulate the presentation and kinetics of reprogramming factors but also provide a conducive microenvironment to facilitate iPSC differentiation.
Although there are several technical and clinical issues to be addressed for the application of hydrogels in clinics, the future of these promising biomaterials appears bright. The brain being a challenging target for tissue engineering, successful translation of these biomaterials from bench to bedside requires their site-specific delivery, monitored by noninvasive high-resolution imaging systems. On the whole, tissue-engineered hydrogels have the potential to surmount the present therapeutic challenges that limit restitution of the stroke-injured brain.
Abbreviations
- BBB :
-
Blood blood-brain barrier
- 3D :
-
Three-dimensional
- CNS :
-
Central nervous system
- ECM :
-
Extracellular matrix
- HA :
-
Hyaluronic acid
- GAG :
-
Glycoseaminoglycans
- ECF :
-
Extracellular fluid
- PEG :
-
poly(ethylene glycol)
- N-CAM :
-
Neural cell adhesion molecule
- hNPCs :
-
Human neural progenitor cells
- MMP :
-
Matrix metalloproteinase
- BMP-4 :
-
Bone morphogenic protein-4
- BDNF :
-
Brain-derived neurotrophic factor
- iPSCs :
-
Induced pluripotent stem cells
- iPSC-NPCs :
-
iPSC-derived NPCs
- RGD :
-
Arginine-glycine-aspartic acid
- ELPs :
-
Elastin-like peptides
- NSC :
-
Neural stem cell
- hPSC :
-
Human pluripotent stem cell
- hUVECs :
-
Human umbilical vein endothelial cells
- CAM :
-
Chorioallantoic membrane
- E :
-
Elastic modulus
- NSPCs :
-
Neural stem and progenitor cells
- MAC :
-
Methacrylamide chitosan
- UBM :
-
Urinary bladder matrix
- EGF :
-
Epidermal growth factor
- PPy :
-
polypyrrole
- VEGF :
-
Vascular endothelial growth factor
- HepMA :
-
Heparin methacrylate
- PVA :
-
poly(vinyl alcohol)
- PEDOT :
-
poly(3,4-ethylenedioxythiophene)
- pTS :
-
paratoluenesulfonate
- PANI :
-
polyaniline
- p(GMA) :
-
poly(glyceryl methacrylate)
- PHPMA :
-
poly(N-2-hydroxypropyl methacrylamide)
- PEGDA :
-
poly(ethyleneglycol diacrylate)
- LCST :
-
Lower critical solution temperature
- UCST :
-
Upper critical solution temperature
- pNIPAAm :
-
poly(N-isopropylacrylamide)
- PLLA :
-
poly-l-(lactic acid)
- PEO-PPO-PEO :
-
poly(ethylene oxide)-b-poly(propylene oxide)-b-poly(ethylene oxide)
- pNIPAAm-PEG :
-
pNIPAAm grafted PEG
- PUASM :
-
poly(urethane amino sulfamethazine)
- S-NBC :
-
S-(2-nitrobenzyl)cysteine)
- GRGDS :
-
Gly-Arg-Gly-Asp-Ser
- EPO :
-
Erythropoietin
- G-CSF :
-
Granulocyte colony-stimulating factor
- HAMC :
-
Hyaluronan/methyl cellulose
- PLGA :
-
poly(lactic-co-glycolic acid)
- PDMS-TEOS :
-
polydimethysiloxane-tetraethoxysilane
- Ang-1 :
-
Angiopoietin-1
- ESC :
-
Embryonic stem cell
- PCL :
-
polycaprolactone
- GSH :
-
Genipin cross-linked sericin hydrogel
- hEnSCs :
-
Human endometrial stem cells
- SCs :
-
Schwann cells
- PFTBA :
-
perfluorotributylamine
- MRI :
-
Magnetic resonance imaging
- PET :
-
Positron emission tomography
- BLI :
-
Bioluminescence imaging
- CEST:
-
Chemical exchange saturation transfer
- GLP-1 :
-
Glucagon-like peptide-1
- STEP :
-
Striatal enriched tyrosine phosphatase
- TBI :
-
Traumatic brain injury
- hUC-MSCs :
-
Human umbilical cord mesenchymal stem cells
- SDF-1α :
-
Stromal cell-derived factor 1α
- CS-GAG :
-
Chondroitin sulfate glycosaminoglycan
- SCI :
-
Spinal cord injury
- NGF :
-
Nerve growth factor
- hUTCs :
-
Human umbilical tissue-derived cells
- VM :
-
Ventral midbrain
- DA :
-
Dopamine
- BDNF-His :
-
Hexahistidine peptide-linked recombinant BDNF
- PD :
-
Parkinson’s disease
- PAA :
-
poly(amidoamine)
- TH :
-
Tyrosine hydroxylase
- GBM :
-
Glioblastoma
- GemC12 :
-
Gemcitabine
- ICG :
-
Indocyanine green
- PEG-DMA :
-
polyethylene glycol dimethacrylate
- TMZ :
-
Temozolomide
- PC gel :
-
poly(ethylene glycol)-g-chitosan hydrogel
References
Benjamin EJ, Blaha MJ, Chiuve SE, Cushman M, Das SR, Deo R, et al. Heart disease and stroke statistics-2017 update: a report from the American Heart Association. Circulation. 2017;135:146–603.
Feigin VL, Norrving B, Mensah GA. Global burden of stroke. Circ Res. 2017;120:439–48.
Donnan GA, Fisher M, Macleod M, Davis SM. Stroke. Lancet. 2008;371:1612–23.
Brouns R, De Deyn PP. The complexity of neurobiological processes in acute ischemic stroke. Clin Neurol Neurosurg. 2009;111:483–95.
Moskowitz MA, Lo EH, Iadecola C. The science of stroke: mechanisms in search of treatments. Neuron. 2010;67:181–98.
Thomas B, Julien B. Treatment of acute ischemic stroke. N Engl J Med. 2000;343:710–22.
Tam RY, Fuehrmann T, Mitrousis N, Shoichet MS. Regenerative therapies for central nervous system diseases: a biomaterials approach. Neuropsychopharmacology. 2014;39:169–88.
Nakagomi N, Nakagomi T, Kubo S, Nakano-Doi A, Saino O, Takata M, et al. Endothelial cells support survival, proliferation, and neuronal differentiation of transplanted adult ischemia-induced neural stem/progenitor cells after cerebral infarction. Stem Cells. 2009;27:2185–95.
Zhang P, Lei X, Sun Y, Zhang H, Chang L, Li C, et al. Regenerative repair of pifithrin-α in cerebral ischemia via VEGF dependent manner. Sci Rep. 2016;6:1–10.
Abe K, Yamashita T, Takizawa S, Kuroda S, Kinouchi H, Kawahara N. Stem cell therapy for cerebral ischemia: from basic science to clinical applications. J Cereb Blood Flow Metab. 2012;32:1317–31.
Chan SJ, Love C, Spector M, Cool SM, Nurcombe V, Lo EH. Endogenous regeneration: engineering growth factors for stroke. Neurochem Int. 2017;107:57–65.
Cooke MJ, Wang Y, Morshead CM, Shoichet MS. Controlled epi-cortical delivery of epidermal growth factor for the stimulation of endogenous neural stem cell proliferation in stroke-injured brain. Biomaterials. 2011;32:5688–97.
Kriegstein A, Alvarez-Buylla A. The glial nature of embryonic and adult neural stem cells. Annu Rev Neurosci. 2009;32:149–84.
Tang YH, Ma YY, Zhang ZJ, Wang YT, Yang GY. Opportunities and challenges: stem cell-based therapy for the treatment of ischemic stroke. CNS Neurosci Ther. 2015;21:337–47.
Wang J, Yang W, Xie H, Song Y, Li Y, Wang L. Ischemic stroke and repair: current trends in research and tissue engineering treatments. Regen Med Res. 2014;2:1–10.
Babita Mahanta RN. An overview of various biomimetic scaffolds: challenges and applications in tissue engineering. J Tissue Sci Eng. 2014;5:1–5.
Osathanon T, Linnes ML, Rajachar RM, Ratner BD, Somerman MJ, Giachelli CM. Microporous nanofibrous fibrin-based scaffolds for bone tissue engineering. Biomaterials. 2008;29:4091–9.
Kumbar SG, Toti US, Deng M, James R, Laurencin CT, Aravamudhan A, et al. Novel mechanically competent polysaccharide scaffolds for bone tissue engineering. Biomed Mater. 2011;6:1–13.
Florine EM, Miller RE, Liebesny PH, Mroszczyk KA, Lee RT, Patwari P, et al. Delivering heparin-binding insulin-like growth factor 1 with self-assembling peptide hydrogels. Tissue Eng A. 2015;21:637–46.
Shin YC, Kim J, Kim SE, Song SJ, Hong SW, Oh JW, et al. RGD peptide and graphene oxide co-functionalized PLGA nanofiber scaffolds for vascular tissue engineering. Regen Biomater. 2017;4:159–66.
Somaa FA, Wang TY, Niclis JC, Bruggeman KF, Kauhausen JA, Guo H, et al. Peptide-based scaffolds support human cortical progenitor graft integration to reduce atrophy and promote functional repair in a model of stroke. Cell Rep. 2017;20:1964–77.
Wang J, Chen F, Liu L, Qi C, Wang B, Yan X, et al. Engineering EMT using 3D micro-scaffold to promote hepatic functions for drug hepatotoxicity evaluation. Biomaterials. 2016;91:11–22.
Srinivasan S, Jayasree R, Chennazhi KP, Nair SV, Jayakumar R. Biocompatible alginate/nano bioactive glass ceramic composite scaffolds for periodontal tissue regeneration. Carbohydr Polym. 2012;87:274–83.
Mohtaram NK, Karamzadeh V, Shafieyan Y, Willerth SM. Commercializing electrospun scaffolds for pluripotent stem cell-based tissue engineering applications. Gruyter Open. 2017;2:62–72.
Qi C, Sun T. Comparative study of porous hydroxyapatite / chitosan and whitlockite / chitosan scaffolds for bone regeneration in calvarial defects. Int J Nanomedicine. 2017;12:2673–87.
Atala A, Bauer SB, Soker S, Yoo JJ, Retik AB. Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet. 2006;367:1241–6.
Garfein ES, Orgill DP, Pribaz JJ. Clinical applications of tissue engineered constructs. Clin Plast Surg. 2003;30:485–98.
Dohmen PM, Lembcke A, Holinski S, Pruss A, Konertz W. Ten years of clinical results with a tissue-engineered pulmonary valve. Ann Thorac Surg. 2011;92:1308–14.
Khouri RK, Rigotti G, Cardoso E, Marchi A, Rotemberg SC, Baker TJ, et al. Tissue-engineered breast reconstruction with brava-assisted fat grafting: a 7-year, 488-patient, multicenter experience. Plast Reconstr Surg. 2015;135:643–58.
Macchiarini P, Jungebluth P, Go T, Asnaghi MA, Rees LE, Cogan A, et al. Clinical transplantation of a tissue-engineered airway. Lancet. 2008;372:2023–30.
Saraiva C, Praça C, Ferreira R, Santos T, Ferreira L, Bernardino L. Nanoparticle-mediated brain drug delivery: overcoming blood-brain barrier to treat neurodegenerative diseases. J Control Release. 2016;235:34–47.
Mdzinarishvili A, Sutariya V, Talasila PK, Geldenhuys WJ, Sadana P. Engineering triiodothyronine (T3) nanoparticle for use in ischemic brain stroke. Drug Deliv Transl Res. 2013;3:309–17.
Wong Po Foo CTS, Lee JS, Mulyasasmita W, Parisi-Amon A, Heilshorn SC. Two-component protein-engineered physical hydrogels for cell encapsulation. Proc Natl Acad Sci. 2009;106:22067–72.
Franco CL, Price J, West JL. Development and optimization of a dual-photoinitiator, emulsion-based technique for rapid generation of cell-laden hydrogel microspheres. Acta Biomater. 2011;7:3267–76.
Baiguera S, Del Gaudio C, Lucatelli E, Kuevda E, Boieri M, Mazzanti B, et al. Electrospun gelatin scaffolds incorporating rat decellularized brain extracellular matrix for neural tissue engineering. Biomaterials. 2014;35:1205–14.
Harris GM, Madigan NN, Lancaster KZ, Enquist LW, Windebank AJ, Schwartz J, et al. Nerve guidance by a decellularized fibroblast extracellular matrix. Matrix Biol. 2017;61:176–89.
Lee SJ, Zhu W, Nowicki M, Lee G, Heo DN, Kim J, et al. 3D printing nano conductive multi-walled carbon nanotube scaffolds for nerve regeneration. J Neural Eng. 2017;15:1–29.
Zhu W, George JK, Sorger VJ, Zhang LG. 3D printing scaffold coupled with low level light therapy for neural tissue regeneration. Biofabrication. 2017;9:0–17.
Zhu W, Harris BT, Zhang LG. Gelatin methacrylamide hydrogel with graphene nanoplatelets for neural cell-laden 3D bioprinting. Proc Annu Int Conf IEEE Eng Med Biol Soc EMBS. 2016;2016:4185–8.
Bhatia SK. Tissue engineering for clinical applications. Biotechnol J. 2010;5:1309–23.
El-Sherbiny I, Yacoub M. Hydrogel scaffolds for tissue engineering: progress and challenges. Glob Cardiol Sci Pract. 2013;2013:316–42.
Carballo-Molina OA, Velasco I. Hydrogels as scaffolds and delivery systems to enhance axonal regeneration after injuries. Front Cell Neurosci. 2015;9:1–12.
Tibbitt MW, Anseth KS. Hydrogels as extracellular matrix mimics for 3D cell culture. Biotechnol Bioeng. 2009;103:655–63.
Zhu J, Marchant RE. Design properties of hydrogel tissue-engineering scaffolds. Expert Rev Med Devices. 2011;8:607–26.
Nicodemus GD, Bryant SJ. Cell encapsulation in biodegradable hydrogels for tissue engineering applications. Tissue Eng B Rev. 2008;14:149–65.
Barros D, Amaral IF, Pêgo AP. Biomimetic synthetic self-assembled hydrogels for cell transplantation. Curr Top Med Chem. 2015;15:1209–26.
Nih LR, Carmichael ST, Segura T. Hydrogels for brain repair after stroke: an emerging treatment option. Curr Opin Biotechnol. 2016;40:155–63.
Skop NB, Calderon F, Cho CH, Gandhi CD, Levison SW. Improvements in biomaterial matrices for neural precursor cell transplantation. Mol Cell Ther. 2014;2:1–19.
Seidlits SK, Khaing ZZ, Petersen RR, Nickels JD, Vanscoy JE, Shear JB, et al. The effects of hyaluronic acid hydrogels with tunable mechanical properties on neural progenitor cell differentiation. Biomaterials. 2010;31:3930–40.
Woerly S, Laroche G, Marchand R, Pato J, Subr V, Ulbrich K. Intracerebral implantation of hydrogel-coupled adhesion peptides tissue reaction. J Neural Transplant Plast. 1995;5:245–55.
Guan J, Zhu Z, Zhao RC, Xiao Z, Wu C, Han Q, et al. Transplantation of human mesenchymal stem cells loaded on collagen scaffolds for the treatment of traumatic brain injury in rats. Biomaterials. 2013;34:5937–46.
Nomura H, Katayama Y, Shoichet MS, Tator CH. Complete spinal cord transection treated by implantation of a reinforced synthetic hydrogel channel results in syringomyelia and caudal migration of the rostral stump. Neurosurgery. 2006;59:183–92.
Pakulska MM, Ballios BG, Shoichet MS. Injectable hydrogels for central nervous system therapy. Biomed Mater. 2012;7:1–13.
Boisserand LSB, Kodama T, Papassin J, Auzely R, Moisan A, Rome C, et al. Biomaterial applications in cell-based therapy in experimental stroke. Stem Cells Int. 2016;2016:1–14.
James DT, Kyle JL. From de novo peptides to native proteins: advancements in biomaterial scaffolds for acute ischemic stroke repair. Biomed Mater. 2018;13:1–41.
Lutolf M. Designing materials to direct stem cell fate. Eur Cells Mater. 2011;462:433–41.
Lin X, Shi Y, Cao Y, Liu W. Recent progress in stem cell differentiation directed by material and mechanical cues. Biomed Mater. 2016;11:1–23.
Griffin MF. Control of stem cell fate by engineering their micro and nanoenvironment. World J Stem Cells. 2015;7:37–50.
Akhmanova M, Osidak E, Domogatsky S, Rodin S, Domogatskaya A. Physical, spatial, and molecular aspects of extracellular matrix of in vivo niches and artificial scaffolds relevant to stem cells research. Stem Cells Int. 2015;2015:1–35.
Moshayedi P, Nih LR, Llorente IL, Berg AR, Cinkornpumin J, Lowry WE, et al. Systematic optimization of an engineered hydrogel allows for selective control of human neural stem cell survival and differentiation after transplantation in the stroke brain. Biomaterials. 2016;105:145–55.
Lam J, Lowry WE, Carmichael ST, Segura T. Delivery of iPS-NPCs to the stroke cavity within a hyaluronic acid matrix promotes the differentiation of transplanted cells. Adv Funct Mater. 2014;24:7053–62.
Lam J, Carmichael ST, Lowry WE, Segura T. Hydrogel design of experiments methodology to optimize hydrogel for iPSC-NPC culture. Adv Healthc Mater. 2015;4:534–9.
Wei YT, Tian WM, Yu X, Cui FZ, Hou SP, Xu QY, et al. Hyaluronic acid hydrogels with IKVAV peptides for tissue repair and axonal regeneration in an injured rat brain. Biomed Mater. 2007;2:142–6.
Kuo Y-C. Chung C-Y. TATVHL peptide-grafted alginate/poly(γ-glutamic acid) scaffolds with inverted colloidal crystal topology for neuronal differentiation of iPS cells. Biomaterials. 2012;33:8955–66.
Kyle JL, Antaris AL, Heilshorn SC. Design of three-dimensional engineered protein hydrogels for tailored control of neurite growth. Acta Biomater. 2013;9:5590–9.
Cheng T, Chen M, Chang W, Huang M, Wang T. Biomaterials neural stem cells encapsulated in a functionalized self-assembling peptide hydrogel for brain tissue engineering. Biomaterials. 2016;34:2005–16.
Wang X, Horii A, Zhang S. Designer functionalized self-assembling peptide nanofiber scaffolds for growth, migration, and tubulogenesis of human umbilical vein endothelial cells. Soft Matter. 2008;4:2388–95.
Liu X, Wang X, Horii A, Wang X, Qiao L, Zhang S, et al. In vivo studies on angiogenic activity of two designer self-assembling peptide scaffold hydrogels in the chicken embryo chorioallantoic membrane. Nanoscale. 2012;4:2720–7.
Saha K, Keung AJ, Irwin EF, Li Y, Little L, Schaffer DV, et al. Substrate modulus directs neural stem cell behavior. Biophys J. 2008;95:4426–38.
Georges PC, Miller WJ, Meaney DF, Sawyer ES, Janmey PA. Matrices with compliance comparable to that of brain tissue select neuronal over glial growth in mixed cortical cultures. Biophys J. 2006;90:3012–8.
Leipzig ND, Shoichet MS. The effect of substrate stiffness on adult neural stem cell behavior. Biomaterials. 2009;30:6867–78.
Woerly S, Marchand R, Lavallée G. Intracerebral implantation of synthetic polymer/biopolymer matrix: a new perspective for brain repair. Biomaterials. 1990;11:97–107.
Murphy WL, Dennis RG, Kileny JL, Mooney DJ. Salt fusion: an approach to improve pore interconnectivity within tissue engineering scaffolds. Tissue Eng. 2002;8:43–52.
Woerly S, Petrov P, Syková E, Roitbak T, Simonová Z, Harvey AR. Neural tissue formation within porous hydrogels implanted in brain and spinal cord lesions: ultrastructural, immunohistochemical, and diffusion studies. Tissue Eng. 1999;5:467–88.
Comolli N, Neuhuber B, Fischer I, Lowman A. In vitro analysis of PNIPAAm-PEG, a novel, injectable scaffold for spinal cord repair. Acta Biomater. 2009;5:1046–55.
Stabenfeldt SE, García AJ, LaPlaca MC. Thermoreversible laminin-functionalized hydrogel for neural tissue engineering. J Biomed Mater Res A. 2006;77:718–25.
Tian WM, Zhang CL, Hou SP, Yu X, Cui FZ, Xu QY, et al. Hyaluronic acid hydrogel as Nogo-66 receptor antibody delivery system for the repairing of injured rat brain: in vitro. J Control Release. 2005;102:13–22.
Kim DH, Seo YK, Thambi T, Moon GJ, Son JP, Li G, et al. Enhancing neurogenesis and angiogenesis with target delivery of stromal cell derived factor-1α using a dual ionic pH-sensitive copolymer. Biomaterials. 2015;61:115–25.
George PM, Bliss TM, Hua T, Lee A, Oh B, Levinson A, et al. Electrical preconditioning of stem cells with a conductive polymer scaffold enhances stroke recovery. Biomaterials. 2017;142:31–40.
Ghasemi-Mobarakeh L, Prabhakaran MP, Morshed M, Nasr-Esfahani MH, Ramakrishna S. Electrical stimulation of nerve cells using conductive nanofibrous scaffolds for nerve tissue engineering. Tissue Eng A. 2009;15:3605–18.
Thrivikraman G, Madras G, Basu B. Intermittent electrical stimuli for guidance of human mesenchymal stem cell lineage commitment towards neural-like cells on electroconductive substrates. Biomaterials. 2014;35:1–17.
Pires F, Ferreira Q, Rodrigues CAV, Morgado J, Ferreira FC. Neural stem cell differentiation by electrical stimulation using a cross-linked PEDOT substrate: expanding the use of biocompatible conjugated conductive polymers for neural tissue engineering. Biochim Biophys Acta Gen Subj. 1850;2015:1158–68.
Luo Y, Shoichet MS. Light-activated immobilization of biomolecules to agarose hydrogels for controlled cellular response. Biomacromolecules. 2004;5:2315–23.
Goubko CA, Majumdar S, Basak A, Cao X. Hydrogel cell patterning incorporating photocaged RGDS peptides. Biomed Microdevices. 2010;12:555–68.
Emily RA, Kyle JL, Kimberly BB. Defining and designing polymers and hydrogels for neural tissue engineering. Neurosci Res. 2013;72:199–213.
Lin C-C, Metters AT, Anseth KS. Functional PEG-peptide hydrogels to modulate local inflammation induced by the pro-inflammatory cytokine TNFalpha. Biomaterials. 2009;30:4907–14.
Zhang L, Cao Z, Bai T, Carr L, Ella-Menye J-R, Irvin C, et al. Zwitterionic hydrogels implanted in mice resist the foreign-body reaction. Nat Biotechnol. 2013;31:553–6.
Zhong J, Chan A, Morad L, Kornblum HI, Fan G, Carmichael ST. Hydrogel matrix to support stem cell survival after brain transplantation in stroke. Neurorehabil Neural Repair. 2010;24:636–44.
Van Tomme SR, Storm G, Hennink WE. In situ gelling hydrogels for pharmaceutical and biomedical applications. Int J Pharm. 2008;355:1–18.
Ghuman H, Modo M. Biomaterial applications in neural therapy and repair. Chin Neurosurg J. 2016;2:1–8.
Park J, Lim E, Back S, Na H, Park Y, Sun K. Nerve regeneration following spinal cord injury using matrix metalloproteinase-sensitive, hyaluronic acid-based biomimetic hydrogel scaffold containing brain-derived neurotrophic factor. J Biomed Mater Res A. 2010;93:1091–9.
Zimmermann DR, Dours-Zimmermann MT. Extracellular matrix of the central nervous system: from neglect to challenge. Histochem Cell Biol. 2008;130:635–53.
Haile Y, Berski S, Dräger G, Nobre A, Stummeyer K, Gerardy-Schahn R, et al. The effect of modified polysialic acid based hydrogels on the adhesion and viability of primary neurons and glial cells. Biomaterials. 2008;29:1880–91.
Krsko P, McCann TE, Thach TT, Laabs TL, Geller HM. Libera MR. Length-scale mediated adhesion and directed growth of neural cells by surface-patterned poly(ethylene glycol) hydrogels. Biomaterials. 2009;30:721–9.
Arulmoli J, Wright HJ, Phan DTT, Sheth U, Que RA, Botten GA, et al. Combination scaffolds of salmon fibrin, hyaluronic acid, and laminin for human neural stem cell and vascular tissue engineering. Acta Biomater. 2016;43:122–38.
Graf J, Iwamoto Y, Sasaki M, Martin GR, Kleinman HK, Robey FA, et al. Identification of an amino acid sequence in laminin mediating cell attachment, chemotaxis, and receptor binding. Cell. 1987;48:989–96.
Bellamkonda RV, Ranieri JP, Aebischer P. Laminin oligopeptide derivatized agarose gels allow 3-dimensional neurite extension in vitro. J Neurosci Res. 1995;41:501–9.
Neiiendam JL, Køhler LB, Christensen C, Li S, Pedersen MV, Ditlevsen DK, et al. An NCAM-derived FGF-receptor agonist, the FGL-peptide, induces neurite outgrowth and neuronal survival in primary rat neurons. J Neurochem. 2004;91:920–35.
Engler AJ, Sen S, Sweeney HL, Discher DE. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126:677–89.
Massensini AR, Ghuman H, Lindsey TS, Christopher JM, Timothy JK, Francesca JN, et al. Concentration-dependent rheological properties of ECM hydrogel for intracerebral delivery to a stroke cavity. Acta Biomater. 2015;27:116–30.
Woerly S. Porous hydrogels for neural tissue engineering. Mater Sci Forum. 1997;250:53–68.
Khademhosseini A, Langer R. Microengineered hydrogels for tissue engineering. Biomaterials. 2007;28:5087–92.
Hilfer R. Transport and relaxation phenomena in porous media. Adv Chem Phys. 1995;92:299–424.
Šprincl L, Kopeček J, Lím D. Effect of porosity of heterogeneous poly(glycol monomethacrylate) gels on the healing-in of test implants. J Biomed Mater Res. 1971;5:447–58.
Bružauskaitė I, Bironaitė D, Bagdonas E, Bernotienė E. Scaffolds and cells for tissue regeneration: different scaffold pore sizes—different cell effects. Cytotechnology. 2016;68:355–69.
Whang K, Healy KE, Elenz DR, Nam EK, Tsai DC, Thomas CH, et al. Engineering bone regeneration with bioabsorbable scaffolds with novel microarchitecture. Tissue Eng. 1999;5:35–51.
Brauker JH, Carr-Brendel VE, Martinson LA, Crudele J, Johnston WD, Johnson RC. Neovascularization of synthetic membranes directed by membrane microarchitecture. J Biomed Mater Res. 1995;29:1517–24.
Klawitter JJ, Hulbert SF. Application of porous ceramics for the attachment of load bearing internal orthopedic applications. J Biomed Mater Res. 1971;5:161–229.
Gogolewski S, Pennings AJ. An artificial skin based on biodegradable mixtures of polylactides and polyurethanes for full-thickness skin wound covering. Macromol Rapid Commun. 1983;4:675–80.
Yannas IV, Lee E, Orgill DP, Skrabut EM, Murphy GF. Synthesis and characterization of a model extracellular matrix that induces partial regeneration of adult mammalian skin. Proc Natl Acad Sci. 1989;86:933–7.
Schwartz I, Robinson BP, Hollinger JO, Szachowicz EH, Brekke J. Calvarial bone repair with porous D, L-polylactide. Otolaryngol Head Neck Surg. 1995;112:707–13.
Gerecht S, Townsend SA, Pressler H, Zhu H, Nijst CLE, Bruggeman JP, et al. A porous photocurable elastomer for cell encapsulation and culture. Biomaterials. 2007;28:4826–35.
Lu L, Mikos AG. The importance of new processing techniques in tissue engineering. MRS Bull. 1996;21:28–32.
Annabi N, Nichol JW, Zhong X, Ji C, Koshy S, Khademhosseini A, et al. Controlling the porosity and microarchitecture of hydrogels for tissue engineering. Tissue Eng B Rev. 2010;16:371–83.
Yeh J, Ling Y, Karp JM, Gantz J, Chandawarkar A, Eng G, et al. Micromolding of shape-controlled, harvestable cell-laden hydrogels. Biomaterials. 2006;27:5391–8.
Fukuda J, Khademhosseini A, Yeo Y, Yang X, Yeh J, Eng G, et al. Micromolding of photocrosslinkable chitosan hydrogel for spheroid microarray and co-cultures. Biomaterials. 2006;27:5259–67.
Bae H, Nichol J, Foudeh A, Zamanian B, Kwon CH, Khadenhosseini A. Microengineering approach for directing embryonic stem cell differentiation. Stud Mechanobiol Tissue Eng Biomater. 2010;2:153–71.
Albert PJ, Schwarz US. Modeling cell shape and dynamics on micropatterns. Cell Adhes Migr. 2016;10:516–28.
Théry M. Micropatterning as a tool to decipher cell morphogenesis and functions. J Cell Sci. 2010;123:4201–13.
Curley JL, Jennings SR, Moore MJ. Fabrication of micropatterned hydrogels for neural culture systems using dynamic mask projection photolithography. J Vis Exp. 2011;48:1–6.
Curley JL, Moore MJ. Facile micropatterning of dual hydrogel systems for 3D models of neurite outgrowth. J Biomed Mater Res A. 2011;99:532–43.
Rizwan M, Yahya R, Hassan A, Yar M, Azzahari AD, Selvanathan V, et al. pH sensitive hydrogels in drug delivery: brief history, properties, swelling, and release mechanism, material selection and applications. Polymers. 2017;9:1–37.
Meng H, Hu J. A brief review of stimulus-active polymers responsive to thermal, light, magnetic, electric, and water/solvent stimuli. J Intell Mater Syst Struct. 2010;21:859–85.
Jeong B, Gutowska A. Lessons from nature: stimuli-responsive polymers and their biomedical applications. Trends Biotechnol. 2002;20:305–11.
Klouda L, Mikos AG. Thermoresponsive hydrogels in biomedical applications—a review. Eur J Pharm Biopharm. 2008;68:34–45.
Peppas NA, Bures P, Leobandung W, Ichikawa H. Hydrogels in pharmaceutical formulations. Eur J Pharm Biopharm. 2000;50:27–46.
Sá-Lima H, Tuzlakoglu K, Mano JF, Reis RL. Thermoresponsive poly(N-isopropylacrylamide)-g-methylcellulose hydrogel as a three-dimensional extracellular matrix for cartilage-engineered applications. J Biomed Mater Res A. 2011;98(A):596–603.
Kabanov AV, Batrakova EV, Alakhov VY. Pluronic® block copolymers as novel polymer therapeutics for drug and gene delivery. J Control Release. 2002;82:189–212.
Peterson DS. pH-sensitive hydrogel. Encycl Microfluid Nanofluidics. 2015:2726–9.
Kumar Dutta P, Dutta J, Tripathi VS. Chitin and chitosan: chemistry, properties and applications. J Sci Ind Res. 2004;63:20–31.
Mukhopadhyay P, Sarkar K, Bhattacharya S, Bhattacharyya A, Mishra R, Kundu PP. pH sensitive N-succinyl chitosan grafted polyacrylamide hydrogel for oral insulin delivery. Carbohydr Polym. 2014;112:627–37.
Atta S, Khaliq S, Islam A, Javeria I, Jamil T, Athar MM, et al. Injectable biopolymer based hydrogels for drug delivery applications. Int J Biol Macromol. 2015;80:240–5.
Wang C, Javadi A, Ghaffari M, Gong S. A pH-sensitive molecularly imprinted nanospheres/hydrogel composite as a coating for implantable biosensors. Biomaterials. 2010;31:4944–51.
Xiang Y, Liu HH, Bin YT, Zhuang ZQ, Jin DM, Peng Y. Functional electrical stimulation-facilitated proliferation and regeneration of neural precursor cells in the brains of rats with cerebral infarction. Neural Regen Res. 2014;9:243–51.
Brett Runge M, Mahrokh D, Jonas B, Terry R, Lichun L, Anthony JW, et al. Development of electrically conductive oligo (polyethylene glycol) fumarate-polypyrrole hydrogels for nerve regeneration. Biomacromolecules. 2010;11:2845–53.
Green RA, Hassarati RT, Goding JA, Baek S, Lovell NH, Martens PJ, et al. Conductive hydrogels: mechanically robust hybrids for use as biomaterials. Macromol Biosci. 2012;12:494–501.
Goding J, Gilmour A, Martens P, Poole-Warren L, Green R. Interpenetrating conducting hydrogel materials for neural interfacing electrodes. Adv Healthc Mater. 2017;6:1–13.
Ferraz N, Straømme M, Fellström B, Pradhan S, Nyholm L, Mihranyan A. In vitro and in vivo toxicity of rinsed and aged nanocellulose-polypyrrole composites. J Biomed Mater Res A. 2012;100 A:2128–38.
Humpolicek P, Kasparkova V, Saha P, Stejskal J. Biocompatibility of polyaniline. Synth Met. 2012;162:722–7.
Green RA, Lovell NH, Wallace GG, Poole-Warren LA. Conducting polymers for neural interfaces: challenges in developing an effective long-term implant. Biomaterials. 2008;29:3393–9.
Ter Schiphorst J, Coleman S, Stumpel JE, Ben Azouz A, Diamond D, Schenning APHJ. Molecular design of light-responsive hydrogels, for in situ generation of fast and reversible valves for microfluidic applications. Chem Mater. 2015;27:5925–31.
Tomatsu I, Peng K, Kros A. Photoresponsive hydrogels for biomedical applications. Adv Drug Deliv Rev. 2011;63:1257–66.
Alvarez-Lorenzo C, Bromberg L, Concheiro A. Light-sensitive intelligent drug delivery systems. Photochem Photobiol. 2009;85:848–60.
Lee SH, Moon JJ, West JL. Three-dimensional micropatterning of bioactive hydrogels via two-photon laser scanning photolithography for guided 3D cell migration. Biomaterials. 2008;29:2962–8.
Tuladhar A, Morshead CM, Shoichet MS. Circumventing the blood-brain barrier: local delivery of cyclosporin A stimulates stem cells in stroke-injured rat brain. J Control Release. 2015;215:1–11.
Caicco MJ, Cooke MJ, Wang Y, Tuladhar A, Morshead CM, Shoichet MS. A hydrogel composite system for sustained epi-cortical delivery of cyclosporin A to the brain for treatment of stroke. J Control Release. 2013;166:197–202.
Wang Y, Cooke MJ, Morshead CM, Shoichet MS. Hydrogel delivery of erythropoietin to the brain for endogenous stem cell stimulation after stroke injury. Biomaterials. 2012;33:2681–92.
Wang Z, Wang J, Jin Y, Luo Z, Yang W, Xie H, et al. A neuroprotective sericin hydrogel as an effective neuronal cell carrier for the repair of ischemic stroke. ACS Appl Mater Interfaces. 2015;7:24629–40.
Shi W, Nie D, Jin G, Chen W, Xia L, Wu X, et al. BDNF blended chitosan scaffolds for human umbilical cord MSC transplants in traumatic brain injury therapy. Biomaterials. 2012;33:3119–26.
Shi W, Huang CJ, Xu XD, Jin GH, Huang RQ, Huang JF, et al. Transplantation of RADA16-BDNF peptide scaffold with human umbilical cord mesenchymal stem cells forced with CXCR4 and activated astrocytes for repair of traumatic brain injury. Acta Biomater. 2016;45:247–61.
Addington CP, Heffernan JM, Millar-Haskell CS, Tucker EW, Sirianni RW, Stabenfeldt SE. Enhancing neural stem cell response to SDF-1α gradients through hyaluronic acid-laminin hydrogels. Biomaterials. 2015;72:11–9.
Betancur MI, Mason HD, Alvarado-Velez M, Holmes PV, Bellamkonda RV, Karumbaiah L. Chondroitin sulfate glycosaminoglycan matrices promote neural stem cell maintenance and neuroprotection post-traumatic brain injury. ACS Biomater Sci Eng. 2017;3:420–30.
Führmann T, Obermeyer J, Tator CH, Shoichet MS. Click-crosslinked injectable hyaluronic acid hydrogel is safe and biocompatible in the intrathecal space for ultimate use in regenerative strategies of the injured spinal cord. Methods. 2015;84:60–9.
Caron I, Rossi F, Papa S, Aloe R, Sculco M, Mauri E, et al. A new three dimensional biomimetic hydrogel to deliver factors secreted by human mesenchymal stem cells in spinal cord injury. Biomaterials. 2016;75:135–47.
Zhao YZ, Jiang X, Xiao J, Lin Q, Yu WZ, Tian FR, et al. Using NGF heparin-poloxamer thermosensitive hydrogels to enhance the nerve regeneration for spinal cord injury. Acta Biomater. 2016;29:71–80.
Caicco MJ, Zahir T, Mothe AJ, Ballios BG, Kihm AJ, Tator CH, et al. Characterization of hyaluronan-methylcellulose hydrogels for cell delivery to the injured spinal cord. J Biomed Mater Res A. 2013;101:1472–7.
Lonardo E, Parish CL, Ponticelli S, Marasco D, Ribeiro D, Ruvo M, et al. A small synthetic cripto blocking peptide improves neural induction, dopaminergic differentiation, and functional integration of mouse embryonic stem cells in a rat model of Parkinson’s disease. Stem Cells. 2010;28:1326–37.
Nakaji-Hirabayashi T, Kato K, Iwata H. Hyaluronic acid hydrogel loaded with genetically-engineered brain-derived neurotrophic factor as a neural cell carrier. Biomaterials. 2009;30:4581–9.
Li J, Darabi M, Gu J, Shi J, Xue J, Huang L, et al. A drug delivery hydrogel system based on activin B for Parkinson’s disease. Biomaterials. 2016;102:72–86.
Komatsu M, Konagaya S, Egawa EY, Iwata H. Maturation of human iPS cells-derived dopamine neuron precursors in alginate-Ca(2+) hydrogel. Biochim Biophys Acta. 1850;2015:1669–75.
Bastiancich C, Vanvarenberg K, Ucakar B, Pitorre M, Bastiat G, Lagarce F, et al. Lauroyl-gemcitabine-loaded lipid nanocapsule hydrogel for the treatment of glioblastoma. J Control Release. 2016;225:283–93.
Jin H, Zhao G, Hu J, Ren Q, Yang K, Wan C, et al. Melittin containing hybrid peptide hydrogels for enhanced photothermal therapy of glioblastoma. ACS Appl Mater Interfaces. 2017;9:25755–66.
Fourniols T, Randolph LD, Staub A, Vanvarenberg K, Leprince JG, Préat V, et al. Temozolomide-loaded photopolymerizable PEG-DMA-based hydrogel for the treatment of glioblastoma. J Control Release. 2015;210:95–104.
Tsao CT, Kievit FM, Ravanpay A, Erickson AE, Jensen MC, Ellenbogen RG, et al. Thermoreversible poly(ethylene glycol)-g-chitosan hydrogel. Biomacromolecules. 2014;15:2656–62.
Katz JS, Burdick JA. Hydrogel mediated delivery of trophic factors for neural repair. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2009;1:128–39.
Wang Y, Cooke MJ, Sachewsky N, Morshead CM, Shoichet MS. Bioengineered sequential growth factor delivery stimulates brain tissue regeneration after stroke. J Control Release. 2013;172:1–11.
Schäbitz WR, Steigleder T, Cooper-Kuhn CM, Schwab S, Sommer C, Schneider A, et al. Intravenous brain-derived neurotrophic factor enhances post stroke sensorimotor recovery and stimulates neurogenesis. Stroke. 2007;38:2165–72.
Schabitz WR, Schwab S, Spranger M, Hacke W. Intraventricular brain-derived neurotrophic factor reduces infarct size after focal cerebral ischemia in rats. J Cereb Blood Flow Metab. 1997;17:500–6.
Ren JM, Finklestein SP. Growth factor treatment of stroke. Curr Drug Targets CNS Neurol Disord. 2005;4:121–5.
Cook DJ, Nguyen C, Chun HN, L Llorente I, Chiu AS, Machnicki M, et al. Hydrogel-delivered brain-derived neurotrophic factor promotes tissue repair and recovery after stroke. J Cereb Blood Flow Metab. 2017;37:1030–45.
Zhang ZG, Zhang L, Jiang Q, Zhang R, Davies K, Powers C, et al. VEGF enhances angiogenesis and promotes blood-brain barrier leakage in the ischemic brain. J Clin Invest. 2000;106:829–38.
Emerich DF, Silva E, Ali O, Mooney D, Bell W, Yu SJ, et al. Injectable VEGF hydrogels produce near complete neurological and anatomical protection following cerebral ischemia in rats. Cell Transplant. 2010;19:1063–71.
Yamashita T, Deguchi K, Nagotani S, Abe K. Vascular protection and restorative therapy in ischemic stroke. Cell Transplant. 2011;20:95–7.
Ju R, Wen Y, Gou R, Wang Y, Xu Q. The experimental therapy on brain ischemia by improvement of local angiogenesis with tissue engineering in the mouse. Cell Transplant. 2014;23:1–38.
Bath PMW, Lees KR. Acute stroke. West J Med. 2000;173:209–12.
Bernstock JD, Peruzzotti-Jametti L, Ye D, Gessler FA, Maric D, Vicario N, et al. Neural stem cell transplantation in ischemic stroke: a role for preconditioning and cellular engineering. J Cereb Blood Flow Metab. 2017;37:2314–9.
Wechsler LR. Stem cell therapies as an emerging paradigm in stroke (STEPS) bridging basic and clinical science for cellular and neurogenic factor therapy in treating stroke. Stroke. 2009;40:510–5.
Wei Z, Zhao J, Chen YM, Zhang P, Zhang Q. Self-healing polysaccharide-based hydrogels as injectable carriers for neural stem cells. Sci Rep. 2016;6:1–12.
Jin K, Mao X, Xie L, Galvan V, Lai B, Wang Y, et al. Transplantation of human neural precursor cells in Matrigel scaffolding improves outcome from focal cerebral ischemia after delayed post ischemic treatment in rats. J Cereb Blood Flow Metab. 2010;30:534–44.
Fujioka T, Kaneko N, Ajioka I, Nakaguchi K, Omata T, Ohba H, et al. β1 integrin signaling promotes neuronal migration along vascular scaffolds in the post-stroke brain. EBioMedicine. 2017;16:195–203.
McMurtrey RJ. Patterned and functionalized nanofiber scaffolds in three-dimensional hydrogel constructs enhance neurite outgrowth and directional control. J Neural Eng. 2014;11:1–15.
Navaei-Nigjeh M, Amoabedini G, Noroozi A, Azami M, Asmani MN, Ebrahimi-Barough S, et al. Enhancing neuronal growth from human endometrial stem cells derived neuron-like cells in three-dimensional fibrin gel for nerve tissue engineering. J Biomed Mater Res A. 2014;102:2533–43.
Chen SJ, Chang CM, Tsai SK, Chang YL, Chou SJ, Huang SS, et al. Functional improvement of focal cerebral ischemia injury by subdural transplantation of induced pluripotent stem cells with fibrin glue. Stem Cells Dev. 2010;19:1757–67.
Ma T, Wang Y, Qi F, Zhu S, Huang L, Liu Z, et al. The effect of synthetic oxygen carrier-enriched fibrin hydrogel on Schwann cells under hypoxia condition in vitro. Biomaterials. 2013;34:10016–27.
Musah S, Wrighton PJ, Zaltsman Y, Zhong X, Zorn S, Parlato MB, et al. Substratum-induced differentiation of human pluripotent stem cells reveals the coactivator YAP is a potent regulator of neuronal specification. Proc Natl Acad Sci U S A. 2014;111:13805–10.
Mosley MC, Lim HJ, Chen J, Yang YH, Li S, Liu Y, et al. Neurite extension and neuronal differentiation of human induced pluripotent stem cell derived neural stem cells on polyethylene glycol hydrogels containing a continuous Young’s modulus gradient. J Biomed Mater Res A. 2017;105:824–33.
Heiss WD. Ischemic penumbra: evidence from functional imaging in man. J Cereb Blood Flow Metab. 2000;20:1276–93.
Jendelová P, Kubinová Š, Sandvig I, Erceg S, Sandvig A, Syková E. Current developments in cell- and biomaterial-based approaches for stroke repair. Expert Opin Biol Ther. 2016;16:43–56.
Ghumana H, Massensini AR, Julia D, Kim SM, Christopher JM, Stephen FB, et al. ECM hydrogel for the treatment of stroke: characterization of the host cell infiltrate. Biomaterials. 2016;14:166–81.
Jin T, Nicholls FJ, Crum WR, Ghuman H, Badylak SF, Modo M. Diamagnetic chemical exchange saturation transfer (diaCEST) affords magnetic resonance imaging of extracellular matrix hydrogel implantation in a rat model of stroke. Biomaterials. 2017;113:176–90.
Boisserand LSB, Lemasson B, Hirschler L, Moisan A, Hubert V, Barbier EL, et al. Multiparametric magnetic resonance imaging including oxygenation mapping of experimental ischaemic stroke. J Cereb Blood Flow Metab. 2017;37:2196–207.
Liang Y, Walczak P, Bulte JWM. The survival of engrafted neural stem cells within hyaluronic acid hydrogels. Biomaterials. 2013;34:5521–9.
Lugo-Hernandez E, Squire A, Hagemann N, Brenzel A, Sardari M, Schlechter J, et al. 3D visualization and quantification of microvessels in the whole ischemic mouse brain using solvent-based clearing and light sheet microscopy. J Cereb Blood Flow Metab. 2017;37:3355–67.
Raza F, Zafar H, Zhu Y, Ren Y, Ullah A, Khan AU, et al. A review on recent advances in stabilizing peptides/proteins upon fabrication in hydrogels from biodegradable polymers. Pharmaceutics. 2018;10:1–21.
Tronci G, Ajiro H, Russell SJ, Wood DJ, Akashi M. Tunable drug-loading capability of chitosan hydrogels with varied network architectures. Acta Biomater. 2014;10:821–30.
Webber MJ, Khan OF, Sydlik SA, Tang BC, Avenue MA. Perspective on the clinical translation of scaffolds for tissue engineering. Ann Biomed Eng. 2016;43:641–56.
Ohnuki M, Takahashi K. Present and future challenges of induced pluripotent stem cells. Philos Trans R Soc Lond B Biol Sci. 2015;370:1–8.
Saha K, Jaenisch R. Technical challenges in using human induced pluripotent stem cells to model disease. Cell Stem Cell. 2009;5:584–95.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Gopalakrishnan, A., Shankarappa, S.A. & Rajanikant, G.K. Hydrogel Scaffolds: Towards Restitution of Ischemic Stroke-Injured Brain. Transl. Stroke Res. 10, 1–18 (2019). https://doi.org/10.1007/s12975-018-0655-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-018-0655-6