Abstract
Disposal of saline subsurface drainage waters from croplands into evaporation basins (or ponds) in the San Joaquin Valley of California causes excessive accumulation of salts and elevated concentrations of arsenic (As), a potentially high risk element with little information about its fate, in the agricultural evaporation ponds. We examined dissolved As concentration, speciation, and distribution in waters as well as As fractionation in sediments in the 10-cell South Evaporation Basin for better understanding of processes and conditions affecting As transformations and fate in a specific drainage disposal facility. The increase of total dissolved As concentrations were observed with higher Cl− and electric conductivity along flow path indicating that evaporation was an important factor regulating total dissolved As concentration. The increases of reduced As species such as arsenite [As(III)] and organic As (monomethylarsonic acid and dimethylarsinic acid) were found towards the terminal flow pathway. However, arsenate [As(V)], the oxidized species remained greater than 67% of total dissolved As in all cell waters. Sequential extractions of sediments indicated that reducing conditions may influence As behavior in sediments to be more soluble and exchangeable. Arsenic association with oxides was appreciable only under oxidizing condition. Carbonate minerals played an important role in immobilizing As into the sediments under alkaline condition and a broad range of redox conditions. However, these sink mechanisms did not significantly reduce As concentrations in the cell waters. The reducing condition facilitated by high concentration of organic matter might be a major factor for the increase in As mobility.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Elevated arsenic (As) concentrations have been found in shallow groundwater and drainage water in the Tulare Basin and the west side of San Joaquin Valley (SJV) of California. The sources of As are the weathering of alluvial–lacustrine origin and marine sedimentary deposits, that are also high in trace elements such as B, Se, Mo and U, from the Coast Range in the west side of SJV (Welch et al. 1988; Fujii and Swain 1995). Irrigation and drainage of croplands in this region have mobilized As into the subsurface and collected in subsurface drainage systems. Evaporation basins (or ponds) are commonly used for disposal of agricultural drainage waters in the southern portion of SJV which is a closed basin. However, trace elements such as As and salts are increasingly accumulated in the evaporation ponds and approaches toxic levels to waterbirds (Ong et al. 1995). The concentrations of As were observed to increase up to 1,000 μg L−1 as electric conductivity (EC) increased in the Tulare Basin (Gao et al. 2007), which is much higher than the 10 μg L−1, US-EPA drinking water limit for As. According to the National Recommended Freshwater Water Quality Criteria, the Criteria Maximum Concentration (CMC) for arsenic is 340 μg L−1 and the Criterion Continuous Concentration (CCC) for arsenic is 150 μg L−1 (USEPA 1999). These recommended criteria were derived from data for As(III), but is applied to total arsenic assuming that As(III) and As(V) are equally toxic to aquatic life. Elevated As concentration levels in evaporation basins pose a concern about potential ecological and health impacts and can be an important factor in developing sustainable agricultural systems in regions where drainage waters are disposed inland.
In order to assess the behavior and potential impacts of As on environments, a good understanding of speciation of As is required because the toxicity, mobility, and bioavailability of As in the environment are largely controlled by its geochemical transformation. Arsenic is found in oxidation states of −3, 0, +3, and +5 in nature and the various species of As exhibit different mobility and toxicity. In reducing environments, arsenite [As(III)] is the dominant species while arsenate [As(V)] is the dominant species in oxic environments (Masscheleyn et al. 1991). Various organic forms of As, generally in minor amounts, also have been found, primarily as methylated species formed through microbial processes (Andreae 1977; Cullen and Reimer 1989). As(III) is of a greater concern due to its greater toxicity and potential for mobility in environment as compared to As(V) (Smith et al. 1998). The methylated arsenic species as monomethylarsonic acid (MMAA) and dimethylarsinic acid (DMAA), are less toxic than As(III) (Cullen and Reimer 1989). In addition, the mobility and availability of As to biological organisms are controlled by the degree and strength of As retention on soil solids (Smith et al. 1998). Thus, it is also important to determine the environmental availability of As within soils or sediments for evaluating its potential risk. To evaluate availability of As, it is critical to determine and quantify As associated with different solid phases in sediments.
The use of sequential extraction techniques has been widely used and recognized for its role in evaluation of potential contaminant effects of metals (Tessier et al. 1979). Since the fractionation is based on operationally-defined extraction procedures, limitation on interpretations of the results applies. Sequential extractions of As are relatively simple means to obtain information on As chemical status on solid-phases and useful to evaluate As mobility in natural systems (Jay et al. 2005). For example, arsenic in the more labile phases can be readily mobilized by changing redox conditions while As that has been sequestered in mineral phases is unlikely to be mobilized by typical geochemical and biological conditions in sediments.
The goal of this study was to acquire essential data to fully understand the processes and conditions of arsenic accumulation and mobility in evaporation ponds. We hypothesized that As mobility in evaporation pond waters is influenced by redox chemistry that affect As speciation and its status in the water column and underlying sediments. The specific objective of this study was to analyze biogeochemical conditions as well as acquire chemical data on As along the flow path as well as within two cells that showed contrasting conditions in a drainage disposal facility. This study provides important information to understand the fate of As in agricultural production systems that utilizes evaporation basins for safe disposal or remediation of drainage waters.
Field methods
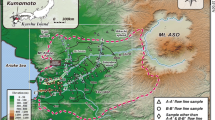
The study site was South Evaporation Basin (SEB) in Tulare Lake Drainage District (TLDD) located in the SJV, which consisted of ten cells and covered a total surface area of 726 ha (7.26 km2) (Fig. 1). Subsurface drainage waters from the main drain of TLDD were pumped into the central inlet channel (CI) and flowed by gravity flow into Cell 1 and subsequently in series from Cells 2 to 10. When the amount of drainage water become limiting as was the case during this study period, the flow of drainage water was directed from Cell 1 into Cell 6 and serially to Cell 10, bypassing Cells 3 to 5 which were kept dry. Artemia (brine shrimp) production was active and harvested in saline Cells 9 and 10 during this study period. To avoid terminal Cell 10 from drying up during limited drainage water production, fresh drainage water has been occasionally introduced into Cell 10 from the CI to sustain brine shrimp production. Occasional diversion of water from Cells 2 to 9 also occurred during the study period. To evaluate biogeochemical condition along the water flow path, water samples were collected monthly at the CI, and at the inlets and outlets of Cells 1, 2, 7, 8, 9 and 10 (Fig. 1) from March to November 2004. Arsenic analysis and speciation along the flow path were performed for water samples taken at the CI and Cells 1, 8, 9, and 10. To observe As biogeochemistry within the cells, a comprehensive sampling within Cells 1 and 9 was conducted in the July 2005 and the sampling locations are shown in Fig. 2. These two cells were chosen due to contrasting conditions in water electrical conductivity (EC) as well as redox condition. Water samples were taken from the top (~10 cm below water surface) and the bottom (near sediment) of the water columns. Water samples were stored in ice coolers (3°C) in the field and transferred to the laboratory. The samples were analyzed within 48 h and kept frozen for further analyses. Surface sediments samples in the bottom of water column were also collected with a sediment scoop attached to a sampling stick from Cells 1 and 9 at selective locations as indicated in Fig. 2. The sediment samples were sealed in a polyethylene bag, transported to the laboratory in ice-coolers, and then frozen for later analyses.
Field measurements including EC, temperature, dissolved oxygen (DO) and sulfide concentration were determined immediately in the field. DO was measured using a YSI Model 54A oxygen meter with a DO probe (Yellow Spring Instrument Inc., Yellow Springs, OH). Temperature and EC were measured with an YSI Model 32 conductance meter. Sulfide concentration was determined by ion selective electrode (Model 9416; Orion Research Inc., Beverly, MA, USA). Sulfide antioxidant was added immediately to water samples upon collection in the ratio of 1:1 to preserve the sample. The electrode was calibrated by serial standards and used to determine concentrations of samples. Ionic strength of the standards was adjusted with 5 M NaNO3 to be similar to the samples. The detection limit of the sulfide measurement was 0.3 mg L−1.
Chemical analysis
Water characterization
After measuring pH, the water samples were passed through a 0.45 μm pore-size membrane filter. The filtrates were then analyzed for major cations (Na+, Mg2+, K+, Ca2+) by atomic absorption spectrophotometry (AAS) and anions (Cl−, SO4 2−, PO4 3−) by ion chromatography (IC). The water samples for major cations and metal ions were acidified by concentrated nitric acid (1% v/v). Alkalinity was determined by titration to an end-point of pH 4.5 using an autotitrator and corrected for ions that consume protons. Boron was analyzed using azomethine-H method (John et al. 1975; Bingham 1982). Dissolved organic carbon (DOC) was determined using a Shimadzu carbon/nitrogen analyzer. Chlorophyll-a samples were collected by filtering an aliquot of a sample through a pre-ashed Gelman A/E glass fiber filter (0.45 μm). The filters were placed in a vial, immediately frozen, and stored in darkness until analyzed. Chlorophyll-a was extracted in 90% reagent-grade ethanol and determined by a fluorometric method (American Public Health Association 1992). To evaluate redox conditions in pond waters, DO was measured in the field. Iron(II) and total Fe were determined by the ferrozine method (Stookey 1970). Total Mn was determined by atomic absorption spectrophotometry. For Fe and Mn analysis, subsamples were filtered through a 0.2-μm membrane filter and preserved with HCl (1% v/v). Ammonium and nitrate were quantified using a Carlson conductometric autoanalyzer (Carlson 1986). Concentrations of solid organic matter in the sediments were determined by potassium dichromate oxidation of organic carbon and subsequent spectrophotometric measurement (modified Walkley–Black method, Nelson and Sommers 1982).
Total arsenic and arsenic speciation for As(V), As(III), MMAA, and DMAA
Total As concentration in water samples was determined using Cutter’s procedure (Cutter 1982) modified by Yoshimoto (1992). This method used a combination of heat, acid (HCl and HNO3), and oxidizer (persulfate) to oxidize all As species to As(V), which was then reduced to As(III) by KI following the procedures in Glaubig and Goldberg (1988). Arsenic was then quantified by hydride generation atomic absorption spectrophotometry (HGAAS) technique. To determine As(III), samples were adjusted to pH 6 because of the high pH and alkalinity of the samples. The hydride was generated using 0.20 mol L−1 MES in 0.033 mol L−1 NaOH and NaBH4/NaOH (0.6%/0.5%) and arsine was detected by AAS at a wavelength of 197.3 nm. For total As(III + V) analysis, As(V) in pretreated solutions (10.0 mL of sample, 4.0 mL of concentrated HCl and 4.0 mL of urea reagent 12.5%) was reduced to As(III) using KI reagent (12.5%) before HGAAS analysis. The hydride was generated using 8 mol L−1 HCl and NaBH4/NaOH (0.6%/0.5%) and detected by AAS. As(V) was obtained as the difference between total As(III + V) and As(III) concentrations. The detection limits were 3.0 ng L−1 for As(V + III) and 1.0 ng L−1 for As(III).
Arsenic speciation including inorganic As [As(V) and As(III)] and organic As [mainly as monomethyl arsine [MMAA, CH3AsO(OH)2] and dimethyl arsine [DMAA or CA, (CH3)2AsOOH] was performed on samples collected along the flow path using a modified hydride generation with cold trapping and atomic absorption spectrophotometry (HGCT-AAS) technique from Andreae (1977), Crecelius et al. (1986) and Masscheleyn et al. (1991). The detailed procedure used in this study was described in Gao and Burau (1997). Because As(V) and As(III) form the same volatile compound (AsH3), As(III) was analyzed by forming arsine at pH 6 when other forms of As could not react in the formation of arsines. The concentration of As(V) was obtained from the difference between total inorganic As(V + III) and As(III). The detection limits were 0.1 ng L−1 for MMAA and CA.
Water samples including the comprehensive sampling conducted in Cells 1 and 9 in 2005 were determined for only As(III) species and total As using the method by Glaubig and Goldberg (1988), because the minor quantity of organic As species along the flow path and the amount of work involved in its analysis. In this case, the difference between the total As and As(III) was the sum of As(V) plus org-As where org-As was considered a minor constituent.
Total arsenic in sediments
Total As in the sediment was determined using a modified procedure using a nitric-percholric (HNO3–HClO4) acid digestion in conjunction with external heating (Zasoski and Burau 1977). Digest analyte concentrations can be determined by HGAAS described above.
Sequential extraction methods
A modified sequential extraction procedure (Lipton 1991; Huang and Fujii 1996) was applied to sediment samples from Cells 1 and 9. For the sequential extraction procedure, wet sediments preserved in a freezer were used and the concentration of As were corrected for water content. To estimate the fractions in the soluble, adsorbed and carbonate-associated As in sediments, a sequential extraction procedure was performed using 0.25 M KCl, 0.1 M K2HPO4 (pH 8) and 1 M NaOAc (pH 5), respectively. Continuous sequential extractions were performed using NaOCl (pH 9.5) and 0.25 M NH2OH–HCl to determine organic material- and amorphous oxide-associated As in sediments, respectively. For the final step, the residual As in sediments was determined using a modified procedure from the HClO4–HNO3 digestion method of Zasoski and Burau (1977) followed by HGAAS analysis. For more details on the extraction procedures, readers are referred to Zhang and Moore (1996) and Gao et al. (2000).
Speciation model
SOLMINEQ. 88, a geochemical equilibrium model, was used to determine dissolved species in high ionic strength solutions using the Pitzer equation for ion activity coefficient determination (Kharaka et al. 1988). Solute activities were obtained from the model computations and solubility data for minerals of interest were acquired from thermodynamic data available from the literature. Mineral saturation indices (SI) were calculated from the ion activity product (IAP) and the equilibrium constant (K eq) for the given mineral: SI = log [IAP/K eq]. Positive, negative and zero SI values suggest that a solution is over-saturated, under-saturated or in equilibrium, respectively, with the mineral phase of interest.
Results and discussion
Water chemistry in SEB
The results of the average monthly measurement of major solutes in drainage water collected from March through November 2004 are given in Table 1. The drainwater disposed into SEB had an initial EC of about 12 dS m−1. With serial evapoconcentration along the flow path, EC rose to extremely high values in the hypersaline range (up to 140 dS m−1, Fig. 3). Inlet water pH averaged 8.3–8.4 and water pH along the flow path increased to more strongly alkaline conditions of pH 8.9–9.4 from evapoconcentration-induced carbonate mineral precipitation.
Major ion chemical composition of drainage waters was dominated by Na+, Cl− and SO4 2−. The dominance of Na+ and Cl− in drainage waters indicates that the highly soluble Na+ and Cl− were readily leachable from the irrigated agricultural field and comprise the dominant ions in the drainage waters (Eghbal et al. 1989). In addition, the high concentration of Na+ and SO4 2− reflect the drainage water passed though gypsiferous soils derived from marine sedimentary deposits (Tanji et al. 1972; Biggar et al. 1984). The concentrations of Na+, Cl− and SO4 2− were significantly correlated with EC values (Fig. 3), both increasing from CI toward terminal Cell 10. There were times when Cells 8 or 9 had higher ECs than Cell 10 due to the input of low EC waters, i.e., fresh drainage water from the CI into Cell 10 or diversion of water flow from Cells 2 to 9. Evapoconcentration of water results in increases in EC but may not follow strictly linear relationship because the solubility product constants (K sp) of minerals can be exceeded and certain minerals (e.g., calcite and gypsum) precipitate out from evapoconcentration (Smith et al. 1995). In this study, evaporation ponds represent sequentially linked cells that were differentiated by the salinity of the water contained in each cell. The behavior of a solute can be evaluated by presuming that chloride ion was the most conservative element in the waters. The evapoconcentration factor (ECF) is simply used to differentiate the level of evapoconcentration in each cell, which is defined as the ratio of [Cl−]pond water/[Cl−]inlet (Tanji 1990). The ECF varied greatly for a cell from season to season and the observed average values ranged from 1.6 in Cell 1 to greater than 10 in Cells 8, 9 or 10 observed in various months (Table 1). This indicates that as water flows from inlet to the terminal cell, water became significantly concentrated due to evaporative losses. The data showed a linear relationship between EC and Cl−: Cl− (mg L−1) = 262 × EC in dS m−1 − 1,552 (R 2 = 0.99). This indicates that changes in EC along the flow path could also represent the effects of evapoconcentration from the data collected in this study.
While EC and most solute concentrations increased from evapoconcentration along the flow path, electron acceptors such as O2, and NO3 − were consumed (Fig. 4). The data indicate that redox environments changed from oxidizing to reducing conditions as EC increased along the water flow path. The low levels of DO show that aerobic respiration was negligible in water towards the end of flow path with higher EC values with decay of euryhaline algae. Nitrate concentrations also decreased gradually along the flow path with increasing EC. The depletions of electron acceptors allowed for the development of reducing conditions, resulting in elevated NH4 and Fe(II) concentrations in drainage waters along the flow path (Table 1).
Organic matter is the primary electron donor driving microbial activity and is present as both solid (e.g., decaying algae) and dissolved forms. The organic matter in the surface sediments averaged 32 (±16), and 36 (±23) g kg−1 for Cells 1 and 9, respectively, indicating that there was no apparent differences in the solid organic matter content for oxidized and reduced conditions. However, DOC in water samples increased exponentially from 22 to 600 mg kg−1 as EC increased along the flow path (Fig. 5), similar to most solutes (Fig. 3). This increase in DOC with EC was somewhat surprising because DOC was not as a considered conservative parameter being an electron donor and energy source for microbial activity. Apparently, the transport of DOC with soluble salts and the evapoconcentration process resulted in the accumulation of DOC at much higher levels than its consumption by microorganisms. Secondly, addition of chicken manure for enhancing production of brine-shrimp production in Cells 9 and 10 can be another source of organic carbon. Both factors may be responsible for the increase of DOC with EC in water shown in Fig. 5. Nonetheless, high levels of DOC provide favorable condition for reducing processes serving as the principle reason for reduction of As species as observed in high EC waters.
Aqueous arsenic speciation along the water flow path
Figure 6 shows that both total As and As(III) accumulated in pond waters with increase in EC. It indicates that the impact of evapoconcentration on accumulation of As along the flow path through the cells from the lowest total As concentration (108–132 μg L−1) detected in the CI to higher As concentration (up to 2,099 μg L−1) towards the terminal cell. However, the concentrations of total dissolved As in pond waters are increased with higher EC value but somewhat poorly correlated with EC values (Fig. 6a). The results indicate that the evapoconcentration has an important role in regulating arsenic concentrations in the evaporation ponds, but may not be the only factor controlling As biogeochemistry.
Figure 6b shows that the higher concentration of As(III) was detected in waters with higher EC values along the flow path although As(III) generally accounted for less than 25% of total As concentration (Figs. 6, 7). Arsenic speciation was affected by transport of drainage water from the CI to the terminal cell. Because of inputs of fresh waters, the drainage water from the CI was more oxidized with negligible amount of reduced As(III) species. Reducing conditions developed due to increased hydrologic residence time toward the terminal cell, which allows bacteria to successively consume O2, NO3 −, Mn(IV), Fe(III) and SO4 2− as electron acceptors.
Redox conditions in each cell directly affected As transformation as reflected by the speciation data. Figure 7 shows total As concentrations as well as speciation in waters along the flow path. Total As concentrations in Cells 9 and 10 appeared lower than in Cell 8, most likely due to the dilution from the introduction of low EC drainage water to the cells (i.e., CI to Cell 10 and Cell 2 to Cell 9). In the CI, DO ranged from 8 to 9 mg L−1 indicating aerobic conditions where As was predominantly found as As(V), accounting for greater than 95% of the total dissolved As. Decreases in the percentage of As(V) were observed along the flow path accompanied by increases in reduced As species [As(III) and org-As]. As shown in Fig. 7, the average As(III) concentrations accounted for 11, 16, and 22% of total dissolved As in the waters collected from C8–9, C9–10, and C10, respectively. Organic-As species (MMAA and DMAA) were detected in a similar manner as As(III) with higher DMAA than MMAA (Fig. 7). Total org-As (MMAA + DMAA) accounted for 2, 6, and 14% of total dissolved As in Cells 8, 9 and 10, respectively. However, temporal changes in total dissolved As concentrations as well as the percentage of As speciation in water samples were observed during the sampling period as indicated by the large standard deviations, particularly in concentration changes. This indicates that there might be temporal changes in hydrologic residence time due to the nature of pond operation (not strictly following serial transfers of water) influencing As speciation in water samples. Arsenate remained the dominant As form along the flow path and even in the terminal cell (>67% of total dissolved As). This shows that reducing conditions did transform As(V) to reduced forms such as As(III) and org-As but not extensively.
Redox conditions and arsenic speciation in Cells 1 and 9
For the examination of As biogeochemistry, water samples within Cells 1 and 9 were collected at various locations in the top and bottom of the water column in July 2005 (Fig. 2). While no significant differences in EC and major solutes were found between the depths in the water columns (top vs. bottom layers), the electron acceptors such as DO, and nitrate decreased with depths in Cell 1 (Table 2). The DO concentration decreased from an average of 14.2 mg L−1 in the top layer to average of 3.7 mg L−1 in the bottom layer with 1 m average depth. It indicates that reducing environments were possibly developed in the bottom layer of Cell 1. It is also supported by the higher nitrate levels in the top layer (average of 5.5 mg L−1) than the bottom layer (average of 2.2 mg L−1). The abundant nitrogen and adequate phosphate in Cell 1 allowed rapid growth of euryhaline algae. As a result, chlorophyll-a concentration in the top layer of water columns in Cell 1 reached an average of 463 μg L−1 and decreased rapidly to 66 μg L−1 in the bottom layer. Some nitrate was consumed by algae in the water and denitrifying bacteria used the algal biomass as a carbon and energy source to reduce nitrate in water (Oswald et al. 1989). In addition, some dead algae were incorporated in the sediments as depositional organic matter and promoted reducing conditions in the bottom of Cell 1 and some were transported to other cells through the flow path.
Cell 9 showed more reducing conditions than Cell 1 as indicated by the lower concentrations of oxidized species and higher concentrations of reduced species (Table 2). The DO and nitrate concentrations in the bottom layer of water columns (average depth = 1 m) in Cell 9 were the lowest (<1 mg L−1 DO and ~1 mg L−1 NO3). In addition, dissolved Fe(II) and sulfide concentrations were detected at the highest level in the bottom layer of Cell 9 (Table 2).
Water samples within Cells 1 and 9 collected in July 2005 were determined for only As(III) and total As with As(V) plus org-As obtained as the difference between these two values (Table 3). Analysis of org-As as MMAA and DMAA in samples along flow path described above indicated that no org-As species were detected in Cell 1 pond waters and 2–6% of org-As were determined in Cell 9 pond waters (Fig. 7). Dissolved As(III) remained as a minor constituent of total dissolved As species (average of 5% in the top layer and 10% in the bottom layer of water columns) in Cell 1 (Table 3). The sum of dissolved As(V) and org-As were up to an average of 95% of the total As concentration in the top layer and 90% in the bottom layer of water columns in Cell 1. The As(III) concentration in Cell 9 increased up to 19% of total dissolved As in both the top and bottom layers of water columns. However, the sum of As(V) and org-As was still high, up to 81% in Cell 9 in both the top and bottom layers of water columns where As(V) was dominant (>77%), based on speciation data from Cells 9 to 10 flow data (Fig. 7).
Distribution and fractions of arsenic in sediments
The results from sequential extraction of sediment samples are shown in Table 4. The As fractions in sediments of Cells 1 and 9 indicate different dominant processes affecting As partitioning in sediments. There was a good agreement between the total As concentration determined by the HClO4–HNO3 digestion method of Zasoski and Burau (1977) applied to separate sediment samples and the sum of the sequential extraction fractions. The percentage of recovery, expressed as the sum of As fractions divided by the total As in sediments, ranged from 90 to 109% of the digestion method indicating a good recovery from the sequential extraction procedures.
The total As concentrations in the KCl extracts, which represent soluble As fraction in sediments, averaged 1.9 and 3.9 mg kg−1, accounting for average 13 and 19% of total As in sediments from Cells 1 and 9, respectively. The higher soluble portion of As in sediments from Cell 9 than Cell 1 may have resulted from higher reduced As species in Cell 9.
The total As concentrations in K2HPO4 extracts, representing adsorbed or easily exchangeable As in sediments, averaged 1.8 and 5.2 mg kg−1, accounting for 12 and 27% in Cells 1 and 9, respectively. It indicates higher adsorbed As in sediments in Cell 9 than Cell 1. In general, the more reducing condition such as in Cell 9 may result in the dissolution of adsorbing phases such as iron oxyhydroxides. However, other solid phases such as organic matters or carbonate minerals besides iron and manganese oxyhydroxides may contribute to As adsorption in Cell 9. The higher As concentration in K2HPO4 extracts might also result from the actual increases in sorbates, both dissolved As(V) and As(III) in Cell 9 compared to Cell 1 (Table 3). In general, As(V) has a greater tendency to be adsorbed on solid phases. As(III) also has a tendency to be absorbed at higher pHs (pH 8–9) compared to As(V) (Manning and Goldberg 1997; Sun and Doner 1998; Jain and Loeppert 2000), which had a similar pH condition in Cell 9.
The total As concentrations in NaOAc extracts, representing As associated with carbonates, were 5.2 and 6.0 mg kg−1, averaging 35 and 26% of total As in sediments from Cells 1 and 9, respectively. This fraction was the highest among all extractions, indicating the importance of carbonates serving as one of the major sink mechanisms.
Arsenic associated with calcite is potentially important since the arsenic-affected pond water bodies were at slightly supersaturated with respect to calcite in both Cells 1 and 9 (median calcite SI = 1.5 and 1.1 in Cells 1 and 9, respectively) according to the results from SOLMINEQ. 88. Goldberg and Glaubig (1988) shows that a maximum adsorption of As(V) on calcite at around pH 10 in their experimental settings. Romero et al. (2004) also indicates that the sorption of As(V) on calcite or carbonate-rich material is a major process in the pH range of 7–9, while the sorption of As(III) on carbonate-rich material is less important as As(V). It indicates that under alkaline condition, carbonates can play an important role in As retention at a broad range of redox conditions.
The NaOAc extract might also include As associated with calcium arsenates (Mihaljevič et al. 2003). The reactive calcium in calcareous conditions has known to be associated with As and the formation of such minerals has been found to be important process to control As concentration in heavily contaminated sites (Bothe and Brown 1999). There exist several calcium arsenates that can be precipitated from solutions containing As(V) (Nishimura et al. 1988; Nishimura and Robins 1998; Zhu et al. 2006), although some of the composition and thermodynamic data for these precipitates are still in debate. Bothe and Brown (1999) identified several precipitates, including Ca4(OH)2(AsO4)2·4H2O, Ca5(AsO4)3OH and Ca3(AsO4)2·3²/3H2O at ambient temperature, which might be possible to be precipitated due to the dominance of As(V) in the ponds. The results from SOLMINEQ. 88, however, indicate that the drainage waters in Cells 1 and 9 were undersaturated with various calcium arsenates using the thermodynamic data were adopted from Nishimura et al. (1988) and Zhu et al. (2006) (Table 5). The saturation index calculations might confirm the study indicating the calcium arsenates becomes unstable for condition of pH >8.3 (Clara and Magalhães 2002), which was the pH condition of most drainage water in the evaporation ponds.
The arsenic associated with carbonates might also include magnesium arsenates. Magnesium arsenates are considered extremely insoluble solids, although their solubility is poorly known (Clara and Magalhães 2002). Voigt et al. (1996) indicate that the crystallization of magnesium arsenates must occur in arsenic and magnesium rich environments, which is the water condition of the evaporation ponds.
The total As concentrations associated with organic matter (OM) were 1.6 mg kg−1 for both cells, accounting for 10 and 6% of total As in sediments from Cells 1 and 9, respectively. These results indicate that the As associated with OM was not a major sink mechanism to immobilize As in sediments and this is very different from Se, which showed a strong association with OM (Gao et al. 2000). Arsenic associated with OM did not show any clear relation with the total organic matter content in sediments, which did not differ much (32 and 36 g kg−1 for Cells 1 and 9, respectively). The processes of As association with organic matter are not well understood. Buschmann et al. (2006) show that As(V) was bound to humic acids more strongly than As(III). In addition, As retention process to humic acid can be more effective under acidic environments than alkaline conditions (Thanabalasingam and Pickering 1986). Thanabalasingam and Pickering (1986) also found that phosphate as well as other anions such as sulfate and carbonate significantly competes with both As(V) and As(III) in adsorption studies using humic acids. Redman et al. (2002) indicates that cations may promote the complexation of As with humic and fulvic acids. Overall, there exists great variability in complexation behavior of various organic matter samples toward As because the extent of specific adsorption of As in the organic matter was found to depend on the surface functional groups present (Thanabalasingam and Pickering 1986; Redman et al. 2002).
The total As concentrations in NH2OH–HCl extracts, representing As associated with amorphous oxyhyroxides, were 4.1 and 2.0 mg kg−1, accounting for 27 and 9% of total As in sediments at Cells 1 and 9, respectively. This indicates that As retention by amorphous oxyhydroxides was one of major processes to control total As concentration in sediments of Cell 1 but not in Cell 9. Under oxic conditions such as in Cell 1, oxides and hydroxides of Fe (Pierce and Moore 1982), Mn (Oscarson et al. 1983) and Al (Huang 1975) sorb As(V) and As(III) depending upon pH. Iron oxide and hydroxide mineral phases particularly play an important role in the biogeochemical cycle of As, because both As(V) and As(III) are strongly sorbed by iron oxides (Pierce and Moore 1982; Raven et al. 1998). The development of reducing condition can mobilize sorbed As to water during reductive dissolution of the ferric oxide (Raven et al. 1998). The released As in water can be evapoconcentrated along flow paths. In addition, iron was actively precipitated under oxic condition in Cell 1 compared to reducing condition in Cell 9. Thus, the oxic condition in Cell 1 resulted in the higher fraction of As associated with amorphous oxyhydroxides, while the development of reducing condition in Cell 9 resulted in a smaller portion of this fraction.
The last step of the sequential extractions, the total As concentrations in HClO4–HNO3 digest representing As associated with recalcitrant minerals, were found to be 0.4 and 3.0 mg kg−1, accounting for an average of 3 and 12% of total As in sediments at Cells 1 and 9, respectively. This indicates that a small amount of As in sediments was associated with recalcitrant minerals such as crystalline Fe and S in Cell 1. The relatively higher fraction of As associated with recalcitrant minerals and detection of sulfide at the bottom layer of water columns in Cell 9 as compared to Cell 1 suggests the possibility of As associated with Fe- and S-bearing recalcitrant mineral phases such as arsenic-sulfide minerals and/or Fe–As–S minerals. Nonetheless, the results indicate that most likely the sulfate reducing conditions were not extensive to influence As mobility in sediments as a major process to lead significant immobilization in sediments in Cell 9. As sulfate reducing conditions develop, reduced As forms may react with sulfides resulting in the precipitation of arsenic-sulfide minerals such as realgar (AsS) and orpiment (As2S3), adsorption and/or co-precipitation with Fe-sulfide minerals such as acid volatile sulfides and pyrite (Morse and Luther 1999). In addition, sorption of As to Fe-sulfide minerals can further transform As to recalcitrant Fe–As–S minerals (Bostick et al. 2004).
The results from sequential extraction methods indicate that As may be bound to different solid phases in the sediments at various geochemical conditions. The evaporation ponds were characterized by alkaline condition, where Fe and Mn oxyhyroxides were less effective to react with As compared to acidic condition. Consequently, carbonates became one of important phases to immobilize As under both oxidizing (Cell 1) and reducing (Cell 9) conditions. Association of As with amorphous Fe and Mn oxyhydroxides was found important only under oxidizing condition, and with iron and sulfur bearing recalcitrant minerals under reducing condition. Further, as reducing condition developed in Cell 9, in addition to the higher concentrations in surface waters, substantially higher fractions of As (46%) were found in the soluble and adsorbed phases in sediments compared to Cell 1. This illustrates that As was more mobile under the reducing condition in Cell 9 in general.
Elevated As concentrations in sediments were observed for some sampling locations (Table 4), which indicates sediments as a sink in the evaporation ponds. However, based on sediment analytical data, estimates of total As in sediment samples from Cells 1 and 9 showed no notable trend, while significant increase of dissolved As concentration with more saline water as well as more reducing environment compared to the initial receiving cell. It indicates that arsenic mobility is affected by evapoconcentration as well as redox chemistry. Evaporation of drainage water at the terminal cell produces water with elevated EC values with higher concentrations of all major elements including arsenic.
The mobilization of As in pond water along the flow path are also thought to result from bacterially-driven reducing environment and decrease in influence of solid phases available to sorb As. Redman et al. (2002) indicated that organic matter could play a significant role in arsenic mobility in natural systems. Microbial degradation of organic matter can lead to reductive dissolution of solid phases such as iron hydroxides releasing associated As on the surface of the solid phases into water columns. In addition, dissolved organic material competes with As for binding onto solid surfaces such as iron hydroxides (Redman et al. 2002; Bauer and Blodau 2006). It is difficult to determine the quantity of bioavailable organic matter in the ponds. However, the high input of organic matter from irrigation water and, particularly, the dramatic increase in DOC concentrations in the terminal cell might be one of the major factors causing arsenic accumulation in water and increase of mobility due to development of reducing conditions in the evaporation ponds.
Conclusions
This study demonstrates that arsenic accumulation, distribution, and speciation in the agricultural evaporation ponds were strongly affected by redox conditions and evaporation. Total dissolved As concentration appeared to increase along the flow path though cells as evaporation concentrates solutes in evaporation ponds. Inorganic arsenic species represent greater than 86% of total dissolved As and As(V) remained the dominant arsenic species over the flow path, while As(III) was detected in terminal cells reflecting development of reducing condition. The results from sequential extraction methods showed that appreciable level of As associated with iron oxyhydroxides under oxidizing condition. Arsenic associated with carbonate minerals was a major sink mechanism under alkaline condition for both oxidizing and reducing conditions while recalcitrant As increased under reducing conditions. Under reducing condition, higher fraction of As became soluble and exchangeable phases. The increase in As mobility was significantly affected by reducing condition facilitated by high concentration of organic carbon in the evaporation ponds.
References
American Public Health Association (1992) Standard methods for examination of water and wastewater, 18th edn. American Public Health Association, Washington, DC
Andreae MO (1977) Determination of arsenic species in sea water. Anal Chem 49:820–823
Bauer M, Blodau C (2006) Mobilization of arsenic by dissolved organic matter from iron oxides, soils and sediments. Sci Total Environ 354:179–190
Biggar JW, Rolston DE, Nielsen DR (1984) Transport of salts by water. Calif Agric 38:10–11
Bingham FT (1982) Boron. Azomethaine-H method. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis, Part 2. Chemical and microbiological properties. Agronomy monograph no. 9, 2nd edn. American Society of Agronomy, Madison, pp 436–535
Bostick B, Chen C, Fendorf S (2004) Arsenite retention mechanisms within estuarine sediments of Pescadero, CA. Environ Sci Technol 38:3299–3304
Bothe JV, Brown PW (1999) Arsenic immobilization by calcium arsenate formation. Environ Sci Technol 33:3806–3811
Buschmann J, Kappeler A, Lindauer U, Kistler D, Berg M, Sigg L (2006) Arsenite and arsenate binding to dissolved humic acids: influence of pH, type of humic acid, and aluminum. Environ Sci Technol 40:6015–6020
Carlson RM (1986) Continuous flow reduction of nitrate to ammonia with granular zinc. Anal Chem 58:1590–1591
Clara M, Magalhães F (2002) Arsenic. An environmental problem limited by solubility. Pure Appl Chem 74:1843–1850
Crecelius EA, Bloom NS, Cowan CE, Jenne EA (1986) Speciation of selenium and arsenic in natural waters and sediments, vol 2. Arsenic speciation. EPRI EA-4641. Final report, Research project 2020-2. Electric Power Research Institute, Palo Alto
Cullen WR, Reimer KJ (1989) Arsenic speciation in the environment. Chem Rev 89:713–764
Cutter GA (1982) Selenium in reducing waters. Science 217:829–831
Eghbal MK, Southard RJ, Whittig LD (1989) Dynamics of evaporate distribution in soils on a fan-playa transect in the Carrizo Plain, California. Soil Sci Soc Am J 53:898–903
Fujii R, Swain WC (1995) Areal distribution of selected trace elements, salinity, and major ions in shallow ground water, Tulare Basin, Southern San Joaquin Valley, California. US geological survey water-resources investigation report 9504048
Gao S, Burau RG (1997) Environmental factors affecting rates of arsine evolution from and mineralization of arsenicals in soil. J Environ Qual 26:753–763
Gao S, Tanji KK, Peters DW, Herbel MJ (2000) Water selenium speciation and sediment fractionation in a California flow-through wetland system. J Environ Qual 29:1275–1283
Gao S, Tanji KK, Bañuelos GS (2007) Processes and conditions affecting elevated arsenic concentrations in groundwaters of Tulare Basin, California, USA. In: Bhattacharya P, Mukherjee AB, Zevenhoven R, Loeppert RH (eds) Arsenic in soil and groundwater environment. Elsevier, New York, pp 383–410
Glaubig RA, Goldberg S (1988) Determination of inorganic arsenic (III) and arsenic (III plus V) using automated hydride-generation atomic absorption spectrometry. Soil Sci Soc Am J 52:536–537
Goldberg S, Glaubig RA (1988) Anion sorption on a calcareous, montmorillonitic soil-Arsenic. Soil Sci Soc Am J 52:1297–1300
Huang PM (1975) Retention of arsenic by hydroxyl-aluminum on surfaces of micaceous mineral colloids. Soil Sci Soc Am J 39:271–274
Huang PM, Fujii R (1996) Selenium and arsenic. In: Sparks DL (ed) Methods of soil analysis. Part 3. Chemical methods. SSSA Book Series No. 5. Soil Science Society of America, Madison, pp 793–831
Jain A, Loeppert RH (2000) Effect of competing anions on the adsorption of arsenate and arsenite by ferrihydrite. J Environ Qual 29:1422–1430
Jay JA, Blute NK, Lin K, Senn D, Hemond HF, Durant JL (2005) Controls on arsenic speciation and solid-phase partitioning in the sediments of a two-basin lake. Environ Sci Technol 39:9174–9181
John MK, Chuah HH, Neufeld JH (1975) Application of improved azomethine-H method to the determination of boron in soils and plants. Anal Lett 8:559–568
Kharaka YK, Gunter WD, Aggarwal PK, Perkins EH, DeBraal JD (1988) SOLMINEQ. 88: A computer program for geochemical modeling of water–rock interactions. US geological survey water-resources investigation report 88–4227. US Geological Survey, Menlo Park
Lipton DL (1991) Associations of selenium in inorganic and organic constituents of soils from a semi-arid region. PhD dissertation, University of California, Berkeley
Manning BA, Goldberg S (1997) Arsenic (III) and arsenic (V) adsorption on three California soils. Soil Sci 162:886–895
Masscheleyn PH, Delaune RD, Patrick WH (1991) A hydride generation atomic absorption technique for arsenic speciation. J Environ Qual 20:96–100
Mihaljevič M, Poňavič M, Ettler V, Šebek O (2003) A comparison of sequential extraction techniques for determining arsenic fractionation in synthetic mineral mixtures. Anal Bioanal Chem 377:723–729
Morse JW, Luther GW (1999) Chemical influences on trace metal-sulfide interactions in anoxic sediments. Geochim Cosmochim Acta 63:3373–3378
Nelson DW, Sommers LE (1982) Total carbon, organic carbon, and organic matter. In: Page AL, Miller RH, Keeney DR (eds) Methods of soil analysis, Part 2. Chemical and microbiological properties. Agronomy monograph no. 9, 2nd edn. American Society of Agronomy, Madison, pp 539–580
Nishimura T, Robins RG (1998) A Re-evaluation of the solubility and stability regions of calcium arsenites and calcium arsenates in aqueous solutions at 25°C. Mineral Proc Extr Met Rev 18:283–308
Nishimura T, Ito CT, Tozawa K (1988) Stabilities and solubilities of metal arsenites and arsenates in water and effect of sulfate and carbonate ions on their solubilities. In: Reddy RG, Hendrix JL, Queneau PB (eds) Arsenic metallurgy fundamentals and applications. Metallurgical Society, Warrendale, pp 77–89
Ong CG, Tanji KK, Dahlgren RA, Smith GR, Quek AF (1995) Water quality and trace element evapoconcentration in evaporation ponds for agricultural waste water disposal. J Agric Food Chem 43:1941–1947
Oscarson DW, Huang PM, Liaw WK, Hammer UT (1983) Kinetics of oxidation of arsenite by various manganese dioxides. Soil Sci Soc Am J 47:644–648
Oswald WJ, Chen PH, Gerhardt MB, Green BF, Nurdogan Y, Von Hippel DF, Newman RD, Chown L, Tam CS (1989) The role of microalgae in removal of selenate from subsurface tile drainage. In: Huntley ME (ed) Biotreatment of agricultural wastewater. CRC Press, Boca Raton, pp 131–141
Pierce M, Moore C (1982) Adsorption of arsenite and arsenate on amorphous iron hydroxide. Water Res 16:1247–1253
Raven CP, Jain A, Loeppert RH (1998) Arsenite and arsenate adsorption on ferrihydrite, kinetics, equilibrium and adsorption envelopes. Environ Sci Technol 32:344–349
Redman AD, Macalady DL, Ahmann D (2002) Natural organic matter affects arsenic speciation and sorption onto hematite. Environ Sci Technol 36:2889–2896
Romero RM, Armienta MA, Carrillo-Chavez A (2004) Arsenic sorption by carbonate-rich aquifer material, a control on arsenic mobility at Zimapan, Mexico. Arch Environ Con Tox 47:1–13
Smith GR, Tanji KK, Jurinak JJ, Burau RG (1995) Applications of a pitzer equations-based model for hypersaline solutions. In: Loeppert RH, Schwab P, Goldberg S (eds) Chemical equilibrium and reaction models. SSS Special publ. No. 42. Soil Science Society of America, Madison, pp 113–141
Smith E, Naidu R, Alston AM (1998) Arsenic in the soil environment: a review. Adv Agron 64:149–195
Stookey LL (1970) Ferrozine, a new spectrophotometric reagent for iron. Anal Chem 42:779–781
Sun X, Doner HE (1998) Adsorption and oxidation of arsenite on goethite. Soil Sci 163:278–287
Tanji KK (1990) Accumulation of salts and trace elements in agricultural evaporation ponds. In: Transaction, 14th international congress of soil science, vol 7, pp 180–185
Tanji KK, Doneen LD, Ferry GV, Ayers RS (1972) Computer simulation analysis on the reclamation of salt-affected soils in San Joaquin Valley, California. Soil Sci Soc Am Pro 36:127–133
Tessier A, Campbell PGC, Bisson M (1979) Sequential extraction procedure for the speciation of particulate trace metals. Anal Chem 51:844–851
Thanabalasingam P, Pickering WF (1986) Arsenic sorption by humic acids. Environ Pollut B 12:233–246
US Environmental Protection Agency (1999) National recommendation water quality criteria—correction. EPA 822-Z-99-001. National Center for Environmental Publications and Information, Ohio. Available at http://epa.gov/waterscience/pc/1999table.pdf (Verified 14 July 2007)
Voigt DE, Brantley SL, Hennet RJ-C (1996) Chemical fixation of arsenic in contaminated soils. Appl Geochem 11:633–643
Welch AH, Lico MS, Hughes JA (1988) Arsenic in groundwater of the western United States. Ground Water 26:333–347
Yoshimoto JT (1992) Potential mechanisms controlling soluble selenite in Sierran sands. Master of Science thesis, University of California, Davis
Zasoski RJ, Burau RG (1977) A rapid nitric-perchloric acid digestion procedure for multi-element tissue analysis. Commun Soil Sci Plan 8:425–436
Zhang Y, Moore JN (1996) Selenium fractionation and speciation in a wetland system. Environ Sci Technol 30:2613–2619
Zhu YN, Zhang XH, Xie QL, Wang DQ, Cheng GW (2006) Solubility and stability of calcium arsenates at 25°C. Water Air Soil Pollut 169:221–238
Acknowledgments
Financial support for this research was received from University of California Salinity/Drainage Program. Strong support was also received from Mr. Douglas Davis, manager of Tulare Lake Drainage District. Mr. Mike Almeda (Novalek, Inc.) and Mr. Larry Davis (TLDD) helped us sample water and sediment within Cells 1 and 9 in the evaporation basins.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ryu, JH., Gao, S. & Tanji, K.K. Speciation and behavior of arsenic in evaporation basins, California, USA. Environ Earth Sci 61, 1599–1612 (2010). https://doi.org/10.1007/s12665-010-0473-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12665-010-0473-y