Abstract
The precise mechanisms involved in the pathophysiology of acute pancreatitis (AP) are still far from clear. Several earlier studies have focused mainly on pancreatic enzyme activation as the key intracellular perturbation in the pancreatic acinar cells. For decades, the trypsin-centered hypothesis has remained the focus of the intra-acinar events in acute pancreatitis. Recent advances in basic science research have lead to the better understanding of various other mechanisms such as oxidative and endoplasmic stress, impaired autophagy, mitochondrial dysfunction, etc. in causing acinar cell injury. Despite all efforts, the clinical outcome of patients with AP has not changed significantly over the years. This suggests that the knowledge of the critical molecular pathways in the pathophysiology of AP is still limited. The mechanisms through which the acinar cell injury leads to local and systemic inflammation are not well understood. The role of inflammatory markers and immune system activation is an area of much relevance from the point of view of finding a target for therapeutic intervention. Some data are available from experimental animal models but not much is known in human pancreatitis. This review intends to highlight the current understanding in this area.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute pancreatitis (AP) is an acute inflammatory disease of the pancreas, characterized clinically by sudden-onset upper abdominal pain and biochemically by an increase in pancreatic enzymes in the blood [1]. About 70 % to 80 % of patients with acute pancreatitis have a mild disease, but in 20 % to 30 % of patients, the disease runs a severe course with a substantial mortality of up to 40 % [2]. The pathogenesis of acute pancreatitis has continued to be an enigma to the clinical researchers and basic scientists. In 1896, Chiari first proposed the concept of autodigestion of the pancreas due to activated pancreatic enzymes. Since then, trypsin-centered hypothesis has remained the focus of the intra-acinar events in acute pancreatitis [3]. With significant advances in basic science research, our understanding has increased significantly in the last couple of decades [4, 5]. Several recent studies have identified various mechanisms such as oxidative stress, endoplasmic stress, impaired autophagy, mitochondrial dysfunction, etc. in the pathogenesis of AP [6, 7]. However, the translation of basic science knowledge into clinical practice has been lagging behind and the outcome of patients with AP has not changed significantly over the years. This suggests that precise and critical molecular pathways in the pathophysiology of AP are still unknown. It remains elusive how the acinar cell injury from the initial inciting event(s) leads to local inflammation and its escalation into systemic inflammation. Variability in immune responses could be a key determinant of the severity in AP. The most severe pancreatitis ensues when local inflammation escalates into systemic inflammation leading to tissue injury and organ dysfunction [8]. Hence, the role of immune system activation and inflammatory mediators is an area of immense interest from the point of finding potential targets for therapeutic intervention. The objective of the present review is to highlight the existing knowledge of the intra-acinar mechanisms and the inflammatory pathways involved in the initiation and progression of acute pancreatitis.
The pathogenetic mechanisms in acute pancreatitis are discussed under following headings:
-
1.
Intra-acinar events
-
a.
Zymogen activation
-
b.
Autophagy
-
c.
Oxidative stress and redox signaling
-
d.
Mitochondrial dysfunction
-
e.
Endoplasmic stress
-
a.
-
2.
Inflammatory responses in acute pancreatitis
-
a.
NF-κB activation
-
b.
Local inflammation: role of damage-associated molecular patterns (DAMPs) and inflammasomes
-
c.
Systemic inflammation: role of immune cells and inflammatory cytokines
-
a.
-
3.
Multi-organ dysfunction syndrome (MODS) in acute pancreatitis
-
a.
Microcirculatory abnormalities
-
b.
Coagulation disturbances
-
c.
Bacterial translocation and secondary infection
-
d.
Role of adipose tissue
-
a.
Intra-acinar events
Zymogen activation
The premature activation of intracellular pancreatic enzymes is an important early step during in the pathogenesis of AP. The pathological conversion of inactive trypsinogen into trypsin has long been thought as the primary mechanism in initiating acute pancreatitis. It has been demonstrated that expression of trypsin in acinar cells is enough to induce cell death and inflammation in pancreatic tissue [9]. In order to precisely demonstrate the role of trypsinogen in AP, a genetically engineered mouse lacking trypsinogen 7 gene (T−/− mice), a novel knockout mice lacking mouse trypsinogen isoform-7, a paralog of human cationic trypsinogen (PRSS1) was created and used for experimental pancreatitis. Absence of trypsinogen activation in T−/− mice led to near complete inhibition of acinar cell death in vitro and a 50 % reduction in acinar necrosis, but similar degrees of local and systemic inflammation were seen during AP progression when compared with wild-type acini in caerulein model of in vitro pancreatitis [10]. This novel knockout mice study showed that while trypsinogen activation is important in causing cell injury early during pancreatitis, the progression of inflammation, both local and systemic, during acute pancreatitis is a trypsin-independent mechanism. The trypsin-independent local inflammatory reaction causes pancreatic damage during later stages, whereas there is direct trypsin-mediated pancreatic cell injury early during the induction of acute pancreatitis.
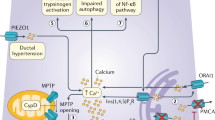
Various cellular mechanisms that are important for the premature activation of trypsinogen include alteration of calcium homeostasis, co-localization of lysosomes, and zymogens and pH alteration (Figs. 1a-c).
Intra-acinar events in the pathophysiology of acute pancreatitis: inciting events like gallstones, endoscopic retrograde cholangiopancreatography, ductal ligation causes ductal obstruction which may block the usual acinar exocytosis of trypsinogen, and increased intraductal pressure resulting in intra-acinar activation of zymogens possibly by co-localization of zymogens and lysosomal hydrolases such as cathepsin B which activates trypsinogen to trypsin. This activated enzymes may undergo basolateral secretion into the interstitium leading to disruption of the paracellular barrier (a, b). Disruption in calcium homeostasis from either pathologic intra-cytoplasmic calcium signals or inadequate calcium clearance through ATP-dependent mechanisms are the key pathogenetic mechanism in acute pancreatitis (c)
Calcium signaling
Cytosolic calcium influx plays a dominant role in both physiological and pathological responses in the acinar cell. The physiological response involves transient spikes in Ca2+ localized to the apical area, whereas sustained global rise denotes a pathological response [11, 12]. Trypsinogen activation and trypsin inactivation are primarily controlled by trypsin (autoactivation and autolysis), and the intracellular calcium concentration affects trypsin activation [13]. The abnormal increase in calcium may be either due to excess influx of Ca2+ or pathology in intracellular calcium clearance mechanisms.
The important sources of pathological calcium response are as follows:
-
1.
IP3R-mediated endoplasmic reticulum (ER) projections and ER membrane ryanodine receptors (RyR) [14].
-
2.
Plasma membrane store operated calcium channels (SOCs) [15]. TRPC3 (transient receptor potential channel 3) and Orai channels have recently been identified as important SOCs.
-
3.
Two-pore channels (TPCs) and mitochondria are other recently recognized sources of calcium.
RyRs are calcium-sensitive channels that open in response to mild rise in Ca2+ and mediate release of Ca2+ from ER. Orai1, a transmembrane spanning protein with four domains, resides at the plasma membrane. STIM1 is postulated to be an important regulator of Orai1 and is located on the ER membrane. It senses ER Ca2+ depletion and translocates towards plasma membrane, where it interacts with Orai1 to open the SOCs [16]. Whether STIM1 regulates other SOCs like TRPC3 is not clear. Dantrolene, a pharmacologic antagonist of RyR, and genetic and pharmacologic inhibition of (TRPC3) have shown that inhibition of intracellular calcium influx attenuates zymogen activation as well as pancreatic damage [17, 18].
Inadequate clearance of Ca2+ is another factor leading to increased intracellular calcium [19, 20]. The clearance of Ca2+ is an ATP-requiring process and ATP depletion or direct inhibition of smooth endoplasmic reticulum calcium ATPase (SERCA) increases intracellular Ca2+. This has been recognized as an important mechanism of pancreatic injury by bile acids and ethanol metabolites [21].
Co-localization of lysosomes and zymogens
Pancreatic enzymes exist as inactive zymogen granules in the acinar cytoplasm. During early stages of AP, the pancreatic zymogens become co-localized with lysosomal hydrolases such as cathepsin B resulting in premature trypsinogen activation [22]. Missorting of cathepsin B into the secretory compartment alone failed to activate trypsinogen but only enhanced trypsin activity during AP [23]. This suggests that co-localization is not a sufficient process; it needs another condition, most likely low vacuolar pH, to activate trypsinogen. The acidic pH enhances the catalytic activity of cathepsin B to activate trypsinogen. Vacuolar ATPase (vATPase) which pumps protons into these vacuoles is recently identified to lower pH in these vacuoles.
Alteration in pH
Behrendorff et al. [24] showed that physiological secretion of zymogen granules from the acinar cells leads to acidification of pancreatic lumen. But how low extracellular pH affects the acinar cell is not clear. The injurious effect of low pH is most likely mediated through RyR-mediated pathological increase in calcium, vATPase-mediated pumping of protons into the vacuoles, and disruption of intercellular junctions leading to zymogen activation and spread into intercellular spaces [25, 26]. Acidic luminal pH also maintains a negative feedback loop regulating acinar secretion. Inhibition of bicarbonate secretion by duct cells appears to be another mechanism leading to low pH.
Autophagy in acute pancreatitis
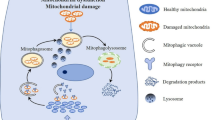
Autophagy is a conserved biological process that has evolved as a complex physiological response involving lysosome-mediated processing and elimination of damaged protein aggregates, organelles, or microorganisms. The role of autophagy in AP has recently been investigated. Grasso et al. [27] described autophagy as protective zymophagy, a response to sequester and degrade potentially deleterious activated zymogens during early stages of pancreatitis. Others suggested that autophagy exerts damaging effects in acinar cells during the onset of AP by delivering trypsinogen to the lysosomes which converts trypsinogen to trypsin [7]. Marerinova et al. [28] showed that impaired autophagy is associated with an imbalance between cathepsin L, which degrades trypsinogen and trypsin, and cathepsin B, which converts trypsinogen into trypsin, resulting in intra-acinar accumulation of active trypsin (Fig. 2a). Mitophagy, a process of degrading damaged mitochondria, has also been shown as a part of general autophagy (macroautophay) in acute pancreatitis [29].
a Impaired autophagy leads to accumulation of autophagic vacuoles, a process that exerts imbalance between the protective cathepsin L and the destructive cathepsin B form leading to intra-acinar accumulation of active trypsin. b Reactive oxygen species (ROS) in acute pancreatitis is generated primarily from NADPH oxidase and mitochondrial dysfunction. Oxidative stress can affect acinar cell at different levels: gene expression, epigenetic modification, immune interaction, and cell signals leading to cellular injury and death
Oxidative stress and redox signaling in acute pancreatitis
Sanfey et al. [30] first reported the beneficial effects of antioxidants superoxide dismutase (SOD) and catalase (CAT) in AP, supporting the role of oxidative stress in the pathogenesis of AP. Oxidative stress is now recognized as a key mediator not only in the early intra-acinar events but also the systemic inflammatory responses [31, 32] (Fig. 2b). The sources of reactive oxygen species (ROS) vary with the experimental models. In caerulein-induced mild acute pancreatitis, ROS is generated mainly from activated neutrophils, whereas in taurocholate-induced necrotizing AP, xanthine oxidase (XO) is the predominant source. XO has a potential to release large amount of free radicals when activated from xanthin dehydrogenase (XDH). XO inhibition with allopurinol or derivatives has been associated with beneficial effects in AP [30]. Oxidative stress is generated primarily from NADPH oxidase [33] and mitochondrial dysfunction during pancreatitis [34]. H2O2, a second messenger of NADPH oxidases, is a major source of ROS in inflammation [35]. Deficient production of NADPH oxidase was shown to reduce trypsin activation in mice with caerulein-induced pancreatitis [36].
The oxidative stress appears to have a dual role in pancreatic injury. ROS induction in the acinar cells promotes apoptosis, a less severe form of cellular injury as it does not induce inflammatory response. On the other hand, inhibition of ROS generation is accompanied by reduced ATP leading to necrosis, which incites an intense inflammatory response. The oxidative stress in the neutrophils activated during inflammatory response to acinar injury may be responsible for further propagation of local and systemic inflammation [37]. The levels of free radicals such as malondialdehyde and xanthine oxidase, and markers of oxidative stress such as myeloperoxidase and thiobarbituric acid reactive substances are increased in the blood early during the course of acute pancreatitis [38, 39]. The ratio between GSSG (oxidized glutathione) and GSH (reduced glutathione) is another reliable indicator of oxidative stress in cells [40]. GSH depletion is thought to allow premature activation of digestive enzymes inside acinar cells triggering the inflammatory process [41–43].
Redox signaling
Unbalanced cellular redox status not only causes oxidative damage but also acts as intracellular signal in up-regulating pro-inflammatory genes by NF-κB activation leading to expression of interleukin-1β (IL-1β), IL-6, and TNF-α. Calcium homeostasis is also sensitive to cellular redox status [44, 45]. Thiol oxidation of redox-sensitive cysteine residues increases the activity of endoplasmic reticulum Ca2+ channels, increasing the cytosolic calcium concentration [14]. Glutathione depletion and ROS generation in macrophages may lower histone deacetylase (HDAC) activity leading to increased IL-8 expression thus enhancing inflammatory response [46].
Mitochondrial dysfunction
Mitochondria utilize approximately 98 % of oxygen consumed in the body and generate ROS. Mitochondria produce most of the cell energy in the form of ATP. Critical loss of ATP generation below a threshold induces necrotic form of cell injury. Mitochondria can also initiate apoptosis through release of mitochondrial cytochrome C into the cytoplasm. Mitochondrial dysfunction is now increasingly recognized as an important cellular event in the pathogenesis of AP (Fig. 3). Excessive production of ROS, NO, and other compounds causes direct damage to mitochondria leading to mitochondrial permeability transition pore (PTP) opening and loss of mitochondrial membrane potential by proton pumping. This results in release of mitochondrial contents into cytosol, ATP depletion, and hence cellular injury [47]. Fatty acid ethyl esters may cause mitochondrial dysfunction in the pancreatic acinar cells in alcoholic pancreatitis. Mitochondrial ROS is also a well-characterized trigger for inflammasome-dependent activation of inflammatory pathway.
The ER-mitochondria membrane microdomains known as mitochondrial-associated membrane (MAM) complex (IP3R–GRP75–VDAC complex) have been described recently [48]. MAM mediates the transfer of Ca2+ from ER to mitochondria required for constitutive mitochondrial function like ATP generation, oxidative phosphorylation, and regulating apoptosis [49, 50]. Apical Ca2+ in a physiologic response is absorbed by the mitochondrial buffering layer but in a pathologic response the mitochondrial buffer capacity fails to overcome this excess Ca2+, leading to mitochondrial dysfunction and abnormal calcium propagation.
ER stress
ER stress can induce both cell death pathway and the inflammatory pathway of its own. ER stress is found to be an early event. However, whether it is dependent or independent of trypsinogen activation is not known. A study by Szmola and Sahin-Toth [51] showed that accumulation of misfolded chymotrypsin (CTRC) in the ER led to ER stress in the acinar cells and cell death by apoptosis. ER stress is now recognized as an important event in alcohol-induced pancreatitis. The unfolded protein response (UPR)/ER stress is thought to be protective against ethanol-induced oxidative stress and provides resistance to ethanol-induced pancreatic damage [52]. Reducing ER stress using tauroursodeoxycholic acid, a chemical chaperone, or by using genetic manipulation of GRP78 showed protective effect in AP [53, 54].
Inflammatory response in acute pancreatitis
NF-κB activation
Acinar cell functions as an inflammatory cell once the initiating process of AP has started. In 1998, Gukovsky et al. [55] first reported activation of NF-κB in acinar cells during acute pancreatitis. It has recently been shown in a knockout mouse model lacking trypsinogen gene that absence of pathologic trypsinogen activation did not affect intra-acinar NF-κB activation [10]. This suggests that NF-κB activation is an early event independent of trypsinogen activation and may be sufficient in inducing acute pancreatitis. However, it is not yet clear if both these events are required for developing AP.
NF-κB activation has been implicated as a key inflammatory pathway in the pathogenesis of acute pancreatitis [56, 57]. It involves degradation of inhibitory κB (IκB) through its phosphorylation at the serine residua mediated by IκB kinase (IKK) [58]. The degradation of IκB releases the nuclear translocation signals (NLS) of the NF-κB resulting in nuclear translocation of NF-κB dimer p50/p65 to the nucleus where it binds to the DNA regulatory binding site and upregulates pro-inflammatory cytokines genes. The intracellular signaling pathways leading to NF-κB activation act via increased cytosolic Ca++ concentration and protein kinase C (PKC) activation [59, 60]. The strategies to decrease NF-κB activation have resulted in attenuated severity of experimental acute pancreatitis. The inflammatory cascade is initiated in the acinar cells by activation of NF-κB before the innate immune response sets in. TNF-α has been shown to be secreted by the acinar cells in caerulein model of acute pancreatitis [61]. Among the chemokines, rodent CXCL10 (human IL-8) and CCL2 (human MCP-1) were secreted by the acinar cells within 60 min of hyperstimulation with caerulein [62].
Local inflammation: role of innate immune response
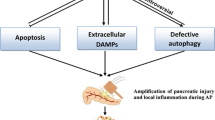
During the process of necrotic form of cell death, many self-antigens are released from the dying cells which are known as “damage-associated molecular pattern” (DAMPs). The DAMPs are recognized by the cells involved in innate immunity through pattern recognition receptors (PRRs) present on their surface. The PRRs are primarily meant to recognize what are termed as “pathogen-associated molecular patterns” (PAMPs) present on the microbes but may also treat DAMPs as foreign antigens and activate the innate immunity. PRRs are of four major types: Toll-like receptors (TLRs), nucleotide-binding oligomerization domain (NOD)-like receptors, retinoic acid-inducible gene 1-like receptors, and C-type lectin receptors. The NOD-like receptors (NLR) act in concert with cytoplasmic protein complexes called inflammasomes. Molecules that act as DAMPs include high-mobility group box protein 1 (HMGB1), DNA, ATP, heat shock protein 70 (hsp70), etc. Following cell death, these molecules are released into extracellular space and activate PRRs. The inflammatory response thus induced is sterile termed as “sterile inflammation.” A full sterile inflammatory response requires at least two distinct signals to an inflammatory cell such as macrophage (Fig. 4).
Damage-associated molecular pattern (DAMPs) released particularly from the necrotic acinar cells activate certain specific receptors known as pattern recognition receptors (PRRs) such as the Toll-like receptors (TLRs) and the nucleotide-binding oligomerization domain (NOD)-like receptor NLRP3 on the host innate immune cells to release activated pro-inflammatory cytokines and induction of sterile inflammation
-
Signal 1 is through activation of surface receptor on immune cells such as TLRs, e.g. TLR4 by HMGB1 and TLR9 by dsDNA. Downstream signaling involves NF-κB pathway to upregulate gene transcription for release of pro-inflammatory cytokines such as pro-IL-1 and pro-IL-18. A meta-analysis of 12 studies showed that there was a significant correlation between serum levels of HMGB1 and severity of acute pancreatitis in humans [63]. Interestingly, TLR-4 and TLR-9 are expressed on pancreatic ductal cells and resident macrophages but not on acinar cells suggesting that it is the immune cells which are the producers of cytokines in response to DAMPs.
-
Signal 2 is provided by DAMPs (e.g. ATP) signaling through plasma membrane purinergic receptor (P2X7) and cytosolic receptors of the Nod-like receptor family (NLRP3) activating a cytosolic complex called inflammasome. Activation of NLRP3 inflammasome regulates proteolytic maturation of caspase 1, a cytosolic cysteine protease which tightly regulates the conversion of the inflammatory cytokines pro-IL-1β and pro-IL-18 to active IL-1 and IL-18. Inhibition of caspase-1 may protect against tissue injury, e.g. acute renal failure induced by endotoxin and acute lung injury in acute pancreatitis [64, 65].
Systemic inflammation: role of immune cells and the inflammatory cytokines
Initial inflammatory response mediated by DAMPs stimulates the immune cells to release 2 major cytokines: TNF-α and IL-1. This initial response is not strong enough to cause major clinical consequences on its own. However, this leads to activation of leukocytes and up-regulation of vascular adhesion molecules resulting in infiltration by the activated leukocytes into the pancreas.
Leukocytes and their role in inflammation
In experimental acute pancreatitis, neutrophils infiltrate the pancreas within 3 h of initiation of AP [66]. The neutrophil response is the first line of defense within 24 h in any inflammation. Chemokines such as CXCL8 (IL-8 in humans) are secreted by the acinar cells, which mediate neutrophil recruitment to the site of inflammation [67]. Their presence is best demonstrated by measuring in the pancreas the activity of enzymes myeloperoxidase (MPO) and elastase, specific to neutrophils. Monocytes also infiltrate the pancreas along with neutrophils and usually take over neutrophils after 24 h. Monocytes secrete pro-inflammatory cytokines such as IL-1, IL-6, and TNF-α. Lymphocytes, mediator of the adaptive immune response, have also been shown to be involved in acute pancreatitis and are found in the pancreas within 6 h of caerulein-induced acute pancreatitis [68]. However, the role of lymphocyte activation and its relation to the severity of AP in humans is still poorly understood [69]. Abnormal phagocytic leukocyte hyperstimulation due to dysregulation in T and B lymphocyte activation is thought to play an important role in severe AP.
The migration of leukocytes requires up-regulation of vascular endothelial adhesion molecules and their interaction with activated leukocytes. P-selectin and E-selectin are endothelial receptors that bind to complementary adhesion molecules on the leukocyte namely L-selectin and integrins [70]. Mac-1(CD11bCD18) and LFA-1(CD11aCD18) integrins act as intercellular ligands for the endothelial adhesion molecules facilitating leukocyte migration [71, 72]. Activated leukocytes release their proteolytic enzymes and oxygen radicals, which damage vascular endothelial cells and pancreatic parenchymal cells. Blood levels of P-selectin and E-selectin were up-regulated in experimental pancreatitis, and their levels correlated with the severity of pancreatitis and lung injury, respectively, in a human study [73]. Endothelial cell adhesion molecule ICAM-1 has also been well studied in experimental acute pancreatitis [74].
Inflammatory mediators in AP
Inflammatory cytokines (IL-1, IL-6, TNF-α) are considered to play a central role in the progression of acute pancreatitis [75, 76]. These are produced predominantly by activated macrophages, lymphocytes, and fibroblasts. TNF-α and IL-1β are the first-order cytokines, which augment the inflammatory response by activating macrophages and regulate the release of other inflammatory mediators (IL-6, IL-8, MIF, etc.).
-
IL-1: This cytokine is produced by activated macrophages as a proprotein, which is converted to its active form by caspase-1. This cytokine is involved in a variety of cellular activities, including cell proliferation, differentiation, and apoptosis; hence, it is an important mediator of the inflammatory response.
-
TNF-α: It is a pro-inflammatory cytokine that is released as early as 30 min after the induction of AP in animal models of AP. It can cause up-regulation of other cytokines, synthesis of free radical species, cell death, and endothelial activation.
-
IL-6: IL-6 levels rise subsequent to TNF-α and IL-1 release. IL-6 activates B and T lymphocytes and the coagulation system. It leads to a sepsis-like state characterized by fever, leukocytosis, and the release of acute phase proteins such as C-reactive protein, complement components, and ferritin. In a prospective study including 108 patients, severe AP was associated with overexpression of pro-inflammatory cytokines and IL-6 was one of the best discriminators between mild and severe acute pancreatitis [77]. Another study showed that blood levels of IL-6 correlated with organ failure and mortality. At a cutoff value of 122 pg/mL on day 3, IL-6 predicted organ failure and severe pancreatitis with a sensitivity and specificity of 81.8 % and 77.7 %, respectively [78]. A recent study by Zhang et al. [79] used a mouse model of acute pancreatitis-associated acute lung injury (ALI) to determine the role of IL-6 in ALI. This study demonstrated the role of IL-6 trans-signaling-dependent STAT3/CXCL1 pathway as central to severe AP-associated lethal ALI. Inflammation-associated NF-κB induced myeloid cell secretion of IL-6, the effects of which were mediated by complexation with soluble IL-6 receptor, a process known as trans-signaling. IL-6 trans-signaling stimulated phosphorylation of STAT3 and production of the neutrophil attractant CXCL1 in pancreatic acinar cells which mediated granulocyte infiltration into the lung hence promoting ALI. Examination of human samples revealed expression of IL-6 in combination with soluble IL-6 receptor as a reliable predictor of ALI in severe AP suggesting that IL-6 was not merely a marker, but a relevant pathophysiological mediator in the disease and that therapeutic inhibition of IL-6 might prevent severe AP-associated ALI.
-
Compensatory anti-inflammatory response syndrome: With the release of pro-inflammatory mediators, anti-inflammatory cytokines are concomitantly produced leading to a compensatory anti-inflammatory response syndrome (CARS). High circulating concentrations of the anti-inflammatory mediators such as soluble TNF receptors (sTNFR), IL-10, IL-11, and IL-1ra have been documented in AP.
The summary of key inflammatory mediators involved in AP is given in Table 1.
Multi-organ dysfunction syndrome (MODS) in AP
The clinical correlates of systemic inflammation are systemic inflammatory response syndrome (SIRS) and organ dysfunction. SIRS is a clinical manifestation of intense systemic inflammation due to release of cytokines and inflammatory mediators. The end result of the severe systemic inflammation is organ dysfunction. Immune and inflammatory reactions are protective in nature and intend to contain infection in case of an invading pathogen. However, an out of proportion or dysregulated immune response may culminate into MODS. The mechanism of organ dysfunction in AP has not been well studied. Organ failure that develops early within a few days of onset of AP (early severe acute pancreatitis) is mainly due to sterile inflammation and carries a high mortality. Organ failure can also develop late during the course of the disease due to septic complications secondary to infected pancreatic necrosis. The early and late organ failures have different pathophysiological mechanisms (Fig. 5).
The mechanism of organ dysfunction, however, is not entirely clear. The key perturbations in causing organ dysfunction are (i) microcirculatory abnormalities leading to vasodilation, capillary leakage, and edema; (ii) coagulation abnormality causing tissue hypoxia and arterial hypotension; and (iii) inflammation and mitochondrial injury leading to cellular injury and tissue damage. In addition, bacterial translocation leading to infected necrosis and sepsis may lead to organ failure like in any other septic condition. A role of adipose tissue and the adipo-cytokines has also been suggested in organ failure.
Circulatory disturbances
Both pancreatic microcirculation and systemic circulation are affected in acute pancreatitis [98]. The mechanisms for the circulatory disturbances are multi-factorial:
-
1.
Leukocyte adhesion, platelet aggregation, hemoconcentration, and vasoconstriction lead to reduced pancreatic blood flow, pancreatic ischemia, and increased capillary permeability [99]. Increased vascular permeability is an important consequence of acute pancreatitis that results in pancreatic edema and exacerbates systemic hemodynamic disturbances leading to pleural effusion and ascites [100].
-
2.
Endothelial cell dysfunction in acute pancreatitis as a result of hemodynamic changes also contributes to loss of barrier function, capillary leak, edema, and sequestration of activated immune cells to the site of inflammation.
-
3.
Another contributory event to the circulatory changes is suppressed myocardial contractility by high levels of inflammatory mediators including NO [101].
The end result of these microcirculatory changes is defective tissue oxygenation resulting in circulatory failure and renal dysfunction.
Coagulation abnormalities
Coagulation abnormalities in AP are probably a result of systemic inflammatory processes that stimulate the coagulation pathway [102]. Coagulation is primarily activated as a protective response to prevent outflow of blood from the injured areas to prevent spread of toxins, bacteria, and DAMPs [103]. But exaggerated coagulation may contribute to tissue ischemia and injury. Raised D-dimer level on admission has been shown to predict development of organ failure with a sensitivity, specificity, positive, and negative predictive values of 90 %, 89 %, 75 %, and 96 %, respectively [104]. Both antithrombin III and heparin have been shown to reduce the severity of acute pancreatitis in animal models [105, 106].
Inflammation and mitochondrial injury
Neutrophil activation results in an oxidative burst and generation of reactive oxygen species. Increased oxidative stress may cause increased mitochondrial permeability and decreased ATP generation. Mitochondria release DAMPs, which can activate neutrophils and cause further tissue injury. Such marked reduction in cellular activity due to mitochondrial injury may translate into organ dysfunction. Recovery of organs within days to weeks after controlling the infection or resolution of acute pancreatitis suggests that it is the cellular inactivity rather than cell death as the main driver of organ dysfunction.
Bacterial translocation and secondary infection
Profound vascular and coagulation changes lead to ischemia of the bowel followed by reperfusion [107, 108]. The major effect of such perturbations is gut barrier dysfunction with increased intestinal permeability [109]. A meta-analysis of 18 studies showed a pooled prevalence of gut barrier dysfunction in 59 % of patients with AP [110]. The consequence of increased intestinal permeability is translocation of bacteria across the gut wall into the necrotic areas and fluid collections, which are susceptible to superinfection by microorganisms [111]. It has been recently shown that it is the small intestine rather than colon that is the source of bacterial transmigration [112]. The common organisms causing infected pancreatic necrosis are Escherichia coli, Pseudomonas, and Klebsiella [113].
Another reason for infection is a state of relative immunosuppression due to compensatory anti-inflammatory response syndrome (CARS) that also makes the patient susceptible to secondary infection.
Compensatory anti-inflammatory response syndrome and immunosuppression
While the pro-inflammatory response is beneficial to a certain extent, an out of proportion inflammation may cause deleterious systemic effects. In order to balance inflammation, a compensatory response is initiated known as CARS [114]. If the anti-inflammatory response is adequate, the patient recovers but at times a profound anti-inflammatory response develops that inhibits the immune response excessively rendering the patient susceptible to immunosuppression and infectious complications. The infectious complications as a result of impaired cellular immunity may occur in the later stage of the disease [115]. In immunosuppression, there is functional defect in monocytes characterized by reduced human leukocyte antigen (HLA)-DR expression and a diminished synthesis of pro-inflammatory cytokines, apoptosis of T and B cells, and downregulation of pro-inflammatory gene transcription [116]. Loss of monocyte HLA-DR expression predicts development of organ failure [117], development of secondary infections, and adverse outcome in AP [118].
Infection of the pancreatic necrotic tissue usually occurs after the first week of illness and results in secondary escalation of the local and systemic inflammation with release of cytokines and chemokines thus aggravating the process set in motion by the underlying pancreatitis pathology. Infected pancreatic necrosis carries a high mortality of up to 40 % [119]. The extent and infection of the pancreatic necrosis correlate with organ failure and mortality. Clinical efforts to prevent the development of pancreatic infection with the use of prophylactic antibiotics have largely failed [120]. Enteral nutrition has been found to be associated with a reduced risk of pancreatic infection by preventing increase in intestinal permeability [121]. Maintenance of the levels of intestinal alkaline phosphatase might be the likely mechanism by which early enteral nutrition preserves the intestinal integrity [122].
Role of adipose tissue in MODS
The adipose tissue itself acts an endocrine organ and releases many pro-inflammatory cytokines like IL-6, IL-1β, and TNF-α and adipokines like adiponectin and leptin. This might be the reason why obesity has been found to be associated with an enhanced inflammatory response and worse prognosis of AP [123]. Visceral fat is considered to be the major culprit [124]. Resistin and visfatin are hormones produced by neutrophils, macrophages, etc. that act on adipose tissue. In addition, lipolysis of fat by activated pancreatic enzymes in the peri-pancreatic fat tissue releases unsaturated fatty acids and other lipids that act as pro-inflammatory mediators and has been recently reported as an independent predictor of worse outcome in acute pancreatitis [125]. In experimental AP in obese mice, obesity converted mild AP to severe AP with increased levels of cytokines, unsaturated fatty acids (UFAs), and multisystem organ failure. Increased pancreatic lipase activity was seen in visceral fat necrosis of obese mice. Lipase inhibition reduced fat necrosis, UFAs, organ failure, and mortality but not the induction of AP [126]. Recent data have shown that UFAs due to lipolysis play an important role in the pathogenesis of systemic injury in AP.
Future direction for research:
Despite recent advances in the field of molecular biology and immunogenetics, the understanding of the pathophysiology of AP is still far from satisfactory. The severity of pancreatitis is model specific in experimental acute pancreatitis, and the injury is predictable and measurable. But this is not the case in humans in whom the cellular injury may vary and the inflammatory responses are unpredictable. The clinical spectrum of acute pancreatitis varies from mild to severe to even life-threatening fulminant disease. The reason why some patients develop severe or fulminant disease whereas the majority of patients have mild disease is unknown. Preliminary data suggest that genetic polymorphisms of certain pro-inflammatory genes such as TNF-α, HSP, and MCP-1 genes may confer additional risk of developing severe pancreatitis [127, 128]. But more work needs to be done in this regard.
Another important dimension is the need for developing specific therapy for acute pancreatitis. In experimental models, a variety of agents have been shown to reduce the severity of acute pancreatitis when used prophylactically. However, such experimental therapies failed to deliver in a clinical setting [129, 130]. The reasons for such a failure are (i) blockage of one pathological process or cytokine is probably not enough because of the redundancy, i.e. multiple pathways being involved in the pathophysiology of AP, and (ii) it is not possible to give these agents prophylactically in humans and by the time a patients presents with acute pancreatitis, it may be too late to reverse the pathophysiologic perturbations.
Summary
The sequence of events in the pathophysiology of acute pancreatitis can be divided into three major phases—(i) initiation phase, (ii) perpetuation phase, and (iii) secondary escalation phase (Fig. 6). The initiation phase includes the intra-acinar events that leads to cellular injury and start of local inflammation within the pancreas primarily through NF-κB. The perpetuating phase involves the progression of initial inflammatory response into systemic inflammation, which involves recruitment of immune cells mainly leukocytes and release of various inflammatory cytokines and chemokines. An out of proportion or dysregulated immune response may result in early onset organ failure with a high mortality. The other important events in this phase are microcirculatory and coagulation disturbances that also contribute to severity of AP and organ dysfunction. In the secondary escalation phase, superadded infection of the necrotic pancreatic/peri-pancreatic tissue/fluid collections leads to further worsening of the pre-existing local and systemic inflammation. Organ failure may also develop late during the course of the disease due to septic complications. There are three major drivers of systemic inflammation in acute pancreatitis: pancreas itself, intestine, and adipose tissue. While the injury starts in the pancreas, intestine and adipose tissue get affected as bystanders and contribute significantly to systemic injury.
Schematic representation of the pathophysiologic events in the evolution of acute pancreatitis: Intra-acinar trypsinogen and NF-κB activation act as independent and parallel events during early acute pancreatitis leading to acinar cell injury and local inflammation. Role of immune cell activation, intestine, and adipose tissue as drivers of systemic inflammation and organ failure in acute pancreatitis
A great deal of advances has taken place in our understanding of the pathophysiology of acute pancreatitis and systemic inflammation leading to SIRS and organ dysfunction but still a lot more needs to be understood. There is a need to decipher precise molecular mechanisms from the acinar cell to distant tissue injury in order to develop targeted therapies and make a real dent on the outcome of clinical acute pancreatitis.
References
Isenmann R, Beger HG. Natural history of acute pancreatitis and the role of infection. Best Pract Res Clin Gastroenterol. 1999;13:291–301.
Forsmark CE, Baillie J. AGA Institute technical review on acute pancreatitis. Gastroenterology. 2007;132:2022–44.
Chiari H. About the digestion of the human pancreas (in German). ZeitschriftfuHeilkunde. 1896;17:69–96.
Lerch MM, Gorelick FS. Early trypsinogen activation in acute pancreatitis. Med Clin North Am. 2000;84:549–63.
Saluja AK, Lerch MM, Phillips PA, Dudeja V. Why does pancreatic overstimulation cause pancreatitis? Annu Rev Physiol. 2007;69:249–69.
Cosen-Binker LI, Gaisano HY. Recent insights into the cellular mechanisms of acute pancreatitis. Can J Gastroenterol. 2007;21:19–24.
Hashimoto D, Ohmuraya M, Hirota M, et al. Involvement of autophagy in trypsinogen activation within the pancreatic acinar cells. J Cell Biol. 2008;181:1065–72.
Sharma M, Banerjee D, Garg PK. Characterization of newer subgroups of fulminant and subfulminant pancreatitis associated with a high early mortality. Am J Gastroenterol. 2007;102:2688–95.
Gaiser S, Daniluk J, Liu Y, et al. Intracellular activation of trypsinogen in transgenic mice induces acute but not chronic pancreatitis. Gut. 2011;60:1379–88.
Dawra R, Sah RP, Dudeja V, et al. Intra-acinar trypsinogen activation mediates early stages of pancreatic injury but not inflammation in mice with acute pancreatitis. Gastroenterology. 2011;141:2210–7. e2.
Criddle DN, Gerasimenko JV, Baumgartner HK, et al. Calcium signalling and pancreatic cell death: apoptosis or necrosis? Cell Death Differ. 2007;14:1285–94.
Petersen OH. Ca2+ induced pancreatic cell death: roles of the endoplasmic reticulum, zymogen granules, lysosomes and endosomes. J Gastroenterol Hepatol. 2008;23 Suppl 1:S31–6.
Sutton R, Criddle D, Raraty MG, Tepikin A, Neoptolemos JP, Petersen OH. Signal transduction, calcium and acute pancreatitis. Pancreatology. 2003;3:497–505.
Husain SZ, Prasad P, Grant WM, Kolodecik TR, Nathanson MH, Gorelick FS. The ryanodine receptor mediates early zymogen activation in pancreatitis. Proc Natl Acad Sci U S A. 2005;102:14386–91.
Cahalan MD. STIMulating store-operated Ca(2+) entry. Nat Cell Biol. 2009;11:669–77.
Lee KP, Yuan JP, Hong JH, So I, Worley PF, Muallem S. An endoplasmic reticulum/plasma membrane junction: STIM1/Orai1/TRPCs. FEBS Lett. 2010;584:2022–27.
Orabi AI, Shah AU, Ahmad MU, et al. Dantrolene mitigates caerulein-induced pancreatitis in vivo in mice. Am J Physiol Gastrointest Liver Physiol. 2010;299:G196–204.
Kim MS, Lee KP, Yang D, et al. Genetic and pharmacologic inhibitionof the Ca2+ influx channel TRPC3 protects secretory epithelia from Ca2+ dependent toxicity. Gastroenterology. 2011;140:2107–15.
Calcraft PJ, Ruas M, Pan Z, et al. NAADP mobilizes calcium from acidic organelles through two-pore channels. Nature. 2009;459:596–600.
Ogunbayo OA, Zhu Y, Rossi D, et al. Cyclic adenosine diphosphate ribose activates ryanodine receptors, whereas NAADP activates two-pore domain channels. J Biol Chem. 2011;286:9136–40.
Voronina SG, Barrow SL, Simpson AW, et al. Dynamic changes in cytosolic and mitochondrial ATP levels in pancreatic acinar cells. Gastroenterology. 2010;138:1976–87.
Saluja AK, Donovan EA, Yamanaka K, Yamaguchi Y, Hofbauer B, Steer ML. Caerulein induced in vitro activation of trypsinogen in rat pancreatic aciniis mediated by cathepsin B. Gastroenterology. 1997;113:304–10.
Meister T, Niehues R, Hahn D, et al. Missorting of cathepsin B into the secretory compartment of CI-MPR/IGFII-deficient mice does not induce spontaneous trypsinogen activation but leads to enhanced trypsin activity during experimental pancreatitis without affecting disease severity. J Physiol Pharmacol. 2010;61:565–75.
Behrendorff N, Floetenmeyer M, Schwiening C, Thorn P. Protons released during pancreatic acinar cell secretion acidify the lumen and contribute to pancreatitis in mice. Gastroenterology. 2010;139:1711–20.
Waterford SD, Kolodecik TR, Thrower EC, Gorelick FS. Vacuolar ATPase regulates zymogen activation in pancreatic acini. J Biol Chem. 2005;280:5430–4.
Reed AM, Husain SZ, Thrower E, et al. Low extracellular pH induces damage in the pancreatic acinar cell by enhancing calcium signaling. J Biol Chem. 2010;286:1919–26.
Grasso D, Ropolo A, Lo Re A, et al. Zymophagy, a novel selective autophagy pathway mediated by VMP1-USP9x-p62, prevents pancreatic cell death. J Biol Chem. 2011;286:8308–24.
Mareninova OA, Hermann K, French SW, et al. Impaired autophagic flux mediates acinar cell vacuole formation and trypsinogen activation in rodent models of acute pancreatitis. J Clin Invest. 2009;119:3340–55.
Jacob TG, Vipin IS, Roy TS, Garg PK. Electron-microscopic evidence of mitochondriae containing macroautophagy in experimental acute pancreatitis: implications for cell death. Pancreatology. 2014;14:433–5.
Sanfey H, Bulkley GB, Cameron JL. The role of oxygen-derived free radicals in the pathogenesis of acute pancreatitis. Ann Surg. 1984;200:405–13.
Sweiry JH, Mann GE. Role of oxidative stress in the pathogenesis of acute pancreatitis. Scand J Gastroenterol Suppl. 1996;219:10–5.
Tsai K, Wang SS, Chen TS, et al. Oxidative stress: an important phenomenon with pathogenetic significance in the progression of acute pancreatitis. Gut. 1998;42:850–5.
Chan YC, Leung PS. Angiotensin-II type 1 receptor-dependent nuclear factor-kβ activation mediated proinflammatory actions in a rat model of obstructive acute pancreatitis. J Pharmacol Exp Ther. 2007;323:10–8.
Escobar J, Pereda J, Lopez-Rodas G, Sastre J. Redox signaling and histone acetylation in acute pancreatitis. Free Radic Biol Med. 2012;52:819–37.
Ushio-Fukai M. Compartmentalization of redox signalling through NADPH oxidase-derived ROS. Antioxid Redox Signal. 2009;11:1289–99.
Gukovskaya AS, Vaquero E, Zaninovic V, et al. Neutrophils and NADPH oxidase mediate intrapancreatic trypsin activation in murine experimental acute pancreatitis. Gastroenterology. 2002;122:974–84.
Booth DM, Murphy JA, Mukherjee R, et al. Reactive oxygen species induced by bile acid induce apoptosis and protect against necrosis in pancreatic acinar cells. Gastroenterology. 2011;140:2116–25.
Schoenberg MH, Buchler M, Gaspar M, et al. Oxygen free radicals in acute pancreatitis of the rat. Gut. 1990;31:1138–43.
Abu-Zidan FM, Bonham MJ, Windsor JA. Severity of acute pancreatitis: a multivariate analysis of oxidative stress markers and modified Glasgow criteria. Br J Surg. 2000;87:1019–23.
Jones DP. Redefining oxidative stress. Antioxid Redox Signal. 2006;8:1865–79.
Alsfasser G, Gock M, Herzog L, et al. Glutathione depletion with L-buthionine-(S, R)-sulfoximine demonstrates deleterious effects in acute pancreatitis of the rat. Dig Dis Sci. 2002;47:1793–9.
Neuschwander-Tetri BA, Ferrell LD, Sukhabote RJ, Grendell JH. Glutathione monoethyl ester ameliorates caerulein-induced pancreatitis in the mouse. J Clin Invest. 1992;89:109–16.
Schulz HU, Niederau C, Klonowski-Stumpe H, Halangk W, Luthen R, Lippert H. Oxidative stress in acute pancreatitis. Hepatol Gastroenterol. 1999;46:2736–50.
Brookes PS, Yoon Y, Robotham JL, Anders MW, Sheu SS. Calcium, ATP and ROS: a mitochondrial love-hate triangle. Am J Physiol-Cell Physiol. 2004;287:C817–33.
Pantano C, Reynaert NL, van der Vliet A, Janssen-Heininger YM. Redox-sensitive kinases of the nuclear factor-kappa β signalling pathway. Antioxid Redox Signal. 2006;8:1791–806.
Yang SR, Chida AS, Bauter MR, et al. Cigarette smoke induces proinflammatory cytokine release by activation of NF-kappa β and posttranslational modifications of histone deacetylase in macrophages. Am J Physiol Lung Cell Mol Physiol. 2006;291:L46–57.
Mukherjee R, Criddle DN, Gukovskaya A, Pandol S, Petersen OH, Sutton R. Mitochondrial injury in pancreatitis. Cell Calcium. 2008;44:14–23.
de Brito OM, Scorrano L. An intimate liaison: spatial organization of the endoplasmic reticulum-mitochondria relationship. EMBO J. 2010;29:2715–23.
Cardenas C, Miller RA, Smith I, et al. Essential regulation of cell bioenergetics by constitutive Ins P3 receptor Ca2+ transfer to mitochondria. Cell. 2010;142:270–83.
Pinton P, Giorgi C, Siviero R, ecchini E, Rizzuto R. Calcium and apoptosis: ER-mitochondria Ca2+ transfer in the control of apoptosis. Oncogene. 2008;27:6407–18.
Szmola R, Sahin-Toth M. Pancreatitis-associated chymotrypsinogen C (CTRC) mutant elicits endoplasmic reticulum stress in pancreatic acinar cells. Gut. 2009;59:365–72.
Lugea A, Tischler D, Nguyen J, et al. Adaptive unfolded protein response attenuates alcohol-induced pancreatic damage. Gastroenterology. 2011;140:987–97.
Malo A, Kruger B, Seyhun E, et al. Tauro ursodeoxycholic acid reduces endoplasmic reticulum stress, trypsin activation, and acinar cell apoptosis while increasing secretion in rat pancreatic acini. Am J Physiol Gastrointest Liver Physiol. 2010;299:G877–86.
Ye R, Mareninova OA, Barron E, et al. Grp78 heterozygosity regulates chaperone balance in exocrine pancreas with differential response to caerulein-induced acute pancreatitis. Am J Pathol. 2010;177:2827–36.
Gukovsky I, Gukovskaya AS, Blinman TA, Zaninovic V, Pandol SJ. Early NF-kappaβ activation is associated with hormone-induced pancreatitis. Am J Physiol. 1998;275:G1402–14.
Rakonczay Z Jr, Hegyi P, Takacs T, Saluja AK. The role of NF-kβ activation in the pathogenesis of acute pancreatitis. Gut. 2008;57:259–67.
Hietaranta AJ, Saluja AK, Bhagat L, Singh VP, Song AM, Steer ML. Relationship between NF-kappaβ and trypsinogen activation in rat pancreas after supra maximal caerulein stimulation. Biochem Biophys Res Commun. 2001;280:388–95.
Barnes PJ, Karin M. Nuclear factor-kappa β: a pivotal transcription factor in chronic inflammatory diseases. N Engl J Med. 1997;336:1066–71.
Satoh A, Gukovskaya AS, Nieto JMC, et al. PKC-delta and epsilon regulate NF-kappaβ activation induced by cholecystokinin and TNF alpha in pancreatic acinar cells. Am J Physiol Gastrointest Liver Physiol. 2004;287:G582–91.
Hietaranta AJ, Singh VP, Bhagat L, et al. Water immersion stress prevents caerulein-induced pancreatic acinar cell NF-kappa β activation by attenuating caerulein-induced intracellular Ca2+ changes. J Biol Chem. 2001;276:18742–7.
Gukovskaya AS, Mouria M, Gukovsky I, et al. Ethanol metabolism and transcription factor activation in pancreatic acinar cells in rats. Gastroenterology. 2002;122:106–18.
Bhatia M, Brady M, Shokuhi S, Christmas S, Neoptolemos JP, Slavin J. Inflammatory mediators in acute pancreatitis. J Pathol. 2000;190:117–25.
Lin Y, Lin LJ, Jin Y, et al. Correlation between serum levels of high mobility group box-1 protein and pancreatitis: a meta-analysis. Biomed Res Int. 2015;2015:430185.
Wang W, Faubel S, Ljubanovic D, et al. Endotoxemic acute renal failure is attenuated in caspase-1-deficient mice. Am J Physiol Renal Physiol. 2005;288:F997–1004.
Zhang XH, Zhu RM, Xu WA, Wan HJ, Lu H. Therapeutic effects of caspase-1 inhibitors on acute lung injury in experimental severe acute pancreatitis. World J Gastroenterol. 2007;13:623–7.
Lampel M, Kern HF. Acute interstitial pancreatitis in the rat induced by excessive doses of a pancreatic secretagogue. Virchows Arch A Pathol Anat Histol. 1977;2:97–117.
Grady T, Liang P, Ernst SA, Logsdon CD. Chemokine gene expression in rat pancreatic acinar cells is an early event associated with acute pancreatitis. Gastroenterology. 1997;6:1966–75.
Demols A, Le Moine O, Desalle F, Quertinmont E, Van Laethem JL, Devière J. CD4+ T cells play an important role in acute experimental pancreatitis in mice. Gastroenterology. 2000;3:582–90.
Pietruczuk M, Dabrowska MI, Wereszczynska-Siemiatkowska U, Dabrowski A. Alteration of peripheral blood lymphocyte subsets in acute pancreatitis. World J Gastroenterol. 2006;12:5344–51.
Telek G, Ducroc R, Scoazec JY, Pasquier C, Feldmann G, Roze C. Differential up-regulation of cellular adhesion molecules at the sites of oxidative stress in experimental acute pancreatitis. J Surg Res. 2001;1:56–67.
Ostermann G, Weber KS, Zernecke A, Schroder A, Weber C. JAM-1 is a ligand of the β2-integrin LFA-1 involved in transendothelial migration of leukocytes. Nat Immunol. 2002;2:151–8.
Santoso S, Sachs UJ, Kroll H, et al. The junctional adhesion molecule 3 (JAM-3) on human platelets is a counterreceptor for the leukocyte integrin Mac-1. J Exp Med. 2002;5:679–91.
Powell JJ, Siriwardena AK, Fearon KC, Ross JA. Endothelial derived selectins in the development of organ dysfunction in acute pancreatitis. Crit Care Med. 2001;3:567–72.
Hartwig W, Werner J, Warshaw AL, et al. Membrane-bound ICAM-1 is up-regulated by trypsin and contributes to leukocyte migration in acute pancreatitis. Am J Physiol Gastrointest Liver Physiol. 2004;287:G1194–9.
Zhang XP, Wang L, Zhou YF. The pathogenic mechanism of severe acute pancreatitis complicated with renal injury: a review of current knowledge. Dig Dis Sci. 2008;53:297–306.
Bhatia M, Neoptolemos JP, Slavin J. Inflammatory mediators as therapeutic targets in acute pancreatitis. Curr Opin Investig Drugs. 2001;2:496–501.
Dambrauskas Z, Giese N, Gulbinas A, et al. Different profiles of cytokine expression during mild and severe acute pancreatitis. World J Gastroenterol. 2010;16:1845–53.
Sathyanarayan G, Garg PK, Prasad HK, Tandon RK. Elevated level of interleukin-6 predicts organ failure and severe disease in patients with acute pancreatitis. J Gastroenterol Hepatol. 2007;22:550–4.
Zhang H, Neuhöfer P, Song L, et al. IL-6 trans-signaling promotes pancreatitis-associated lung injury and lethality. J Clin Invest. 2013;123:1019–31.
Bhatia M, Moochhala S. Role of inflammatory mediators in the pathophysiology of acute respiratory distress syndrome. J Pathol. 2004;202:145–56.
Bhatia M, Wong FL, Cao Y, et al. Pathophysiology of acute pancreatitis. Pancreatology. 2005;5:132–44.
Norman JG, Fink GW, Franz MG. Acute pancreatitis induces intrapancreatic tumor necrosis factor gene expression. Arch Surg. 1995;130:966–70.
Fink GW, Norman JG. Specific changes in the pancreatic expression of the interleukin 1 family of genes during experimental acute pancreatitis. Cytokine. 1997;9:1023–7.
Suzuki S, Miyasaka K, Jimi A, Funakoshi A. Induction of acute pancreatitis by cerulein in human IL-6 gene transgenic mice. Pancreas. 2000;21:86–92.
Botoi G, Andercou A. Interleukin 17-prognostic marker of severe acute pancreatitis. Chirurgia (Bucur). 2009;104:431–8.
Perejaslov A, Chooklin S, Bihalskyy I. Implication of interleukin 18 and intercellular adhesion molecule (ICAM)-1 in acute pancreatitis. Hepatogastroenterology. 2008;55:1806–13.
Calandra T, Echtenacher B, Le Roy D, et al. Protection from septic shock by neutralization of macrophage migration inhibitory factor. Nat Med. 2000;6:164–70.
Rau B, Steinbach G, Gansauge F, Mayer JM, Grunert A, Beger HG. The potential role of procalcitonin and interleukin 8 in the prediction of infected necrosis in acute pancreatitis. Gut. 1997;41:832–40.
Shokuhi S, Bhatia M, Christmas S, Sutton R, Neoptolemos JP, Slavin J. Levels of the chemokines growth-related oncogene alpha and epithelial neutrophil-activating protein 78 are raised in patients with severe acute pancreatitis. Br J Surg. 2002;89:566–72.
Rau B, Baumgart K, Kruger CM, Schilling M, Beger HG. CC-chemokine activation in acute pancreatitis: enhanced release of monocyte chemoattractant protein-1 in patients with local and systemic complications. Intensive Care Med. 2003;29:622–9.
Gerard C, Frossard JL, Bhatia M, Saluja A, Lu B, Steer ML. Targeted disruption of the beta-chemokine receptor CCR1 protects against pancreatitis-associated lung injury. J Clin Invest. 1997;100:2022–7.
Konturek SJ, Dembinski A, Konturek PJ, Warzecha Z, Jaworek J, Gustaw P. Role of platelet activating factor in pathogenesis of acute pancreatitis in rats. Gut. 1992;33:1268–74.
Bhatia M, Saluja AK, Hofbauer B, et al. Role of substance P and the neurokinin 1 receptor in acute pancreatitis and pancreatitis associated lung injury. Proc Natl Acad Sci U S A. 1998;95:4760–5.
Bhatia M, Wong FL, Fu D, Lau HY, Moochhala SM, Moore PK. Role of hydrogen sulfide in acute pancreatitis and associated lung injury. FASEB J. 2005;19:623–5.
Bhatia M, Saluja AK, Hofbauer B, Steer ML. Neutral endopeptidase (NEP) plays an anti-inflammatory role in acute pancreatitis and pancreatitis-associated lung injury. Pancreas. 1997;15:428.
Simovic MO, Bonham MJD, Abu-Zidan FM, Windsor JA. Anti-inflammatory cytokine response and clinical outcome in acute pancreatitis. Crit Care Med. 1999;27:2662–5.
Bhatia M, Singh VP, Frossard JL, Lee HS, Gerard C, Steer ML. Complement factor C5a exerts an anti-inflammatory effect in acute pancreatitis and associated lung injury. Am J Physiol Gastrointest Liver Physiol. 2001;280:G974–8.
Cuthbertson CM, Christophi C. Disturbances of the microcirculation in acute pancreatitis. Br J Surg. 2006;93:518–30.
Pitkaranta P, Kivisaari L, Nordling S, Nuutinen P, Schroder T. Vascular changes of pancreatic ducts and vessels in acute necrotizing, and in chronic pancreatitis in humans. Int J Pancreatol. 1991;8:13–22.
Foitzik T, Eibl G, Hotz HG, Faulhaber J, Kirchengast M, Buhr HJ. Endothelin receptor blockade in severe acute pancreatitis leads to systemic enhancement of microcirculation, stabilization of capillary permeability, and improved survival rates. Surgery. 2000;128:399–407.
Rudiger A, Singer M. Mechanisms of sepsis-induced cardiac dysfunction. Crit Care Med. 2007;35:1599–608.
Salomone T, Tosi P, Palareti G, et al. Coagulative disorders in human acute pancreatitis: role for the D-dimer. Pancreas. 2003;26:111–6.
Dixon B. The role of microvascular thrombosis in sepsis. Anaesth Intensive Care. 2004;32:619–29.
Radenkovic D, Bajec D, Ivancevic N, et al. D-dimer in acute pancreatitis: a new approach for an early assessment of organ failure. Pancreas. 2009;38:655–60.
Bleeker WK, Agterberg J, Rigter G, Hack CE, Gool JV. Protective effect of antithrombin III in acute experimental pancreatitis in rats. Dig Dis Sci. 1992;37:280–5.
Hackert T, Werner J, Gebhard MM, Klar E. Effects of heparin in experimental models of acute pancreatitis and post-ERCP pancreatitis. Surgery. 2004;135:131–8.
Farrant GJ, Abu-Zidan FM, Liu X, Delahunt B, Zwi LJ, Windsor JA. The impact of intestinal ischaemia-reperfusion on caerulein induced edematous experimental pancreatitis. Eur Surg Res. 2003;35:395–400.
Juvonen PO, Tenhunen JJ, Heino AA, et al. Splanchnic tissue perfusion in acute experimental pancreatitis. Scand J Gastroenterol. 1999;34:308–14.
Flint RS, Windsor JA. The role of the intestine in the pathophysiology and management of severe acute pancreatitis. HPB (Oxford). 2003;5:69–85.
Wu LM, Sankaran SJ, Plank LD, Windsor JA, Petrov MS. Meta-analysis of gut barrier dysfunction in patients with acute pancreatitis. Br J Surg. 2014;101:1644–56.
Ammori BJ. Role of the gut in the course of severe acute pancreatitis. Pancreas. 2003;26:122–9.
Fritz S, Hackert T, Hartwig W, et al. Bacterial translocation and infected pancreatic necrosis in acute necrotizing pancreatitis derives from small bowel rather than from colon. Am J Surg. 2010;200:111–7.
Garg PK, Khanna S, Bohidar NP, Kapil A, Tandon RK. Incidence, spectrum, and antibiotic sensitivity pattern of bacterial infections among patients with acute pancreatitis. J Gastroenterol Hepatol. 2001;16:1055–9.
Mentula P, Kylänpää ML, Kemppainen E, et al. Plasma anti-inflammatory cytokines and monocyte human leucocyte antigen-DR expression in patients with acute pancreatitis. Scand J Gastroenterol. 2004;39:178–87.
Beger HG, Bittner R, Block S, Buchler M. Bacterial contamination of pancreatic necrosis. A prospective clinical study. Gastroenterology. 1986;91:433–8.
Finfer SR, Vincent JL. Severe sepsis and septic shock. N Engl J Med. 2013;369:840–51.
Mentula P, Kylänpää-Bäck ML, Kemppainen E, et al. Decreased HLA (human leucocyte antigen)-DR expression on peripheral blood monocytes predicts the development of organ failure in patients with acute pancreatitis. Clin Sci (Lond). 2003;105:409–17.
Lindström O, Kylänpää ML, Mentula P, et al. Upregulated but insufficient generation of activated protein C is associated with development of multiorgan failure in severe acute pancreatitis. Crit Care. 2006;10:R16.
Gotzinger P, Sautner T, Kriwanek S, et al. Surgical treatment for severe acute pancreatitis: extent and surgical control of necrosis determine outcome. World J Surg. 2002;26:474–8.
Bai Y, Gao J, Zou DW, Li ZS. Prophylactic antibiotics cannot reduce infected pancreatic necrosis and mortality in acute necrotizing pancreatitis: evidence from a meta-analysis of randomized controlled trials. Am J Gastroenterol. 2008;103:104–10.
Li JY, Yu T, Chen GC, et al. Enteral nutrition within 48 hours of admission improves clinical outcomes of acute pancreatitis by reducing complications: a meta-analysis. PLoS One. 2013;8, e64926.
Goldberg RF, Austen WG Jr, Zhang X, et al. Intestinal alkaline phosphatase is a gut mucosal defense factor maintained by enteral nutrition. Proc Natl Acad Sci U S A. 2008;105:3551–6.
Premkumar R, Phillips AR, Petrov MS, Windsor JA. The clinical relevance of obesity in acute pancreatitis: targeted systematic reviews. Pancreatology. 2015;15:25–33.
Yashima Y, Isayama H, Tsujino T, et al. A large volume of visceral adipose tissue leads to severe acute pancreatitis. J Gastroenterol. 2011;46:1213–8.
Noel P, Patel K, Durgampudi C, et al. Peripancreatic fat necrosis worsens acute pancreatitis independent of pancreatic necrosis via unsaturated fatty acids increased in human pancreatic necrosis collections. Gut. 2016;65:100–11.
Patel K, Trivedi RN, Durgampudi C, et al. Lipolysis of visceral adipocyte triglyceride by pancreatic lipases converts mild acute pancreatitis to severe pancreatitis independent of necrosis and inflammation. Am J Pathol. 2015;185:808–19.
Balog A, Gyulai Z, Boros LG, et al. Polymorphism of the TNF-alpha, HSP70-2, and CD14 genes increases susceptibility to severe acute pancreatitis. Pancreas. 2005;30:e46–50.
Papachristou GI, Sass DA, Avula H, et al. Is the monocyte chemotactic protein-1 -2518 G allele a risk factor for severe acute pancreatitis? Clin Gastroenterol Hepatol. 2005;3:475–81.
Johnson CD, Kingsnorth AN, Imrie CW, et al. Double blind, randomised, placebo controlled study of a platelet activating factor antagonist, lexipafant, in the treatment and prevention of organ failure in predicted severe acute pancreatitis. Gut. 2001;48:62–9.
Besselink MG, van Santvoort HC, Buskens E, et al. Probiotic prophylaxis in predicted severe acute pancreatitis: a randomised, double-blind, placebo-controlled trial. Lancet. 2008;371:651–9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
PS, and PKG declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Singh, P., Garg, P.K. Pathophysiological mechanisms in acute pancreatitis: Current understanding. Indian J Gastroenterol 35, 153–166 (2016). https://doi.org/10.1007/s12664-016-0647-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-016-0647-y