Abstract
Nerve agents pose a real threat to both the military and civil populations, but the current treatment of the poisoning is unsatisfactory. Thus, we studied the efficacy of prophylactic use of memantine alone or in combination with clinically used reversible acetylcholinesterase inhibitors (pyridostigmine, donepezil, rivastigmine) against soman. In addition, we tested their influence on post-exposure therapy consisting of atropine and asoxime. Pyridostigmine alone failed to decrease the acute toxicity of soman. But all clinically used acetylcholinesterase inhibitors administered alone reduced the acute toxicity, with donepezil showing the best efficacy. The combination of memantine with reversible acetylcholinesterase inhibitors attenuated soman acute toxicity significantly. The pretreatment administered alone or in combinations influenced the efficacy of post-exposure treatment in a similar fashion: (i) pyridostigmine or memantine alone did not affect the antidotal treatment, (ii) centrally acting reversible acetylcholinesterase inhibitors alone increased the antidotal treatment slightly, (iii) combination of memantine with reversible acetylcholinesterase inhibitors increased the antidotal treatment more markedly. In conclusion, memantine alone failed to decrease the acute toxicity of soman or increase post-exposure antidotal treatment efficacy. The combination of memantine with donepezil significantly increased post-exposure effectiveness (together 5.12, pretreatment alone 1.72). Both drugs, when applied together, mitigate soman toxicity and boost post-exposure treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Nerve agents or other highly toxic organophosphorus compounds (OPs) pose a real threat to both the military and civil populations since they can be employed in wartime or terrorist attacks. After exposure, OPs irreversibly bind to acetylcholinesterase (AChE, EC 3.1.1.7) and disrupt its physiological function: degradation of acetylcholine in the synaptic clefts. The accumulated neurotransmitter overstimulates both types of cholinergic receptors, leading to headaches, glandular hyperexcretion, urinary and fecal incontinence, and seizures; death commonly occurs due to acute respiratory insufficiency (Bajgar 2004; Colovic et al. 2013).
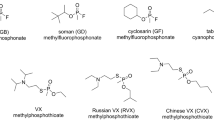
Antidotal therapy against nerve agents comprises mostly anticholinergics (atropine) and anticonvulsive drugs (diazepam). In addition, AChE reactivators (briefly oximes) can restore the physiological function of AChE (Bajgar 2004; Colovic et al. 2013). Despite all efforts, the effectiveness of oxime therapy is still limited by (i) aging of the enzyme-inhibitor complex, which impedes AChE reactivation, and (ii) poor distribution of oximes into tissues such as the brain (Chambers et al. 2020; Kassa 2019; Lorke et al. 2008). This especially hampers counteracting the centrally acting agents like soman (pinacolyl methylfluorophosphonate). This makes soman of particular interest for testing (i) new treatment strategies focused on central nervous system (CNS) protection and (ii) pretreatment strategies.
Pretreatment strategies represent the medical countermeasures administered relatively shortly before the actual exposure to nerve agents. Its administration should increase (i) resistance of humans against OPs and (ii) effectiveness of post-exposure therapy (Bajgar et al. 2009). Prophylactic AChE reversible inhibitors (AChEIs) can protect AChE against nerve agent–induced irreversible damage (Bajgar et al. 2009; Layish et al. 2005; Patocka et al. 2006). The armed forces generally accept pyridostigmine bromide as a pretreatment agent of choice (Karasova et al. 2020a, b). Based on its physico-chemical properties, we assume it acts mostly in the periphery. However, the protection of cholinergic and glutamate receptors in the brain is paramount. The prevention of nerve agent–induced excitotoxicity and dysfunction ameliorates (i) the signs and symptoms of the acute cholinergic crisis, (ii) development of secondary neuronal damage, and (iii) long-term neuropsychiatric and neurological disorders (Chen 2012; Shih and McDonough 1997). Figure 1 depicts a simplified scheme of the pathological cascade in the brain after OP exposure (inspired by Chen 2012).
To augment the pretreatment strategies, centrally acting reversible AChEIs, commonly used in Alzheimer’s disease (AD), such as donepezil or rivastigmine, should replace pyridostigmine. In addition, other centrally acting reversible AChEIs such as physostigmine were evaluated as promising prophylactic drugs against nerve agent poisoning (Leadbeater et al. 1985; Miller et al. 1993; Myhrer and Aas, 2016). The glutamate neurotoxicity associated with the cholinergic crisis could be mitigated by another anti-AD drug — memantine (non-competitive N-methyl-d-aspartate receptor antagonist). Stojiljkovic et al. (2019) demonstrated the prophylactic potential of memantine in soman-poisoned rats; memantine also alleviates dichlorvos toxicity in the same species (Zhou et al. 2005). To our knowledge, the efficacy or potential risk of using memantine with commonly used centrally acting AChEI drugs (donepezil and rivastigmine) has not been evaluated.
This study aims to evaluate the influence of memantine alone or in combination with reversible AChEI (pyridostigmine, donepezil, or rivastigmine) on the resistance against soman acute toxicity and the therapeutic efficacy of the currently used antidotal treatment (asoxime in combination with atropine) in soman-poisoned mice.
Material and Methods
Animals
Male NMRI mice weighing 18–22 g were purchased from VELAZ (Prague, Czech Republic). They were kept in an air-conditioned room (22 ± 2 °C and 50 ± 10% relative humidity), with lights from 7:00 a.m. to 7:00 p.m. and ad libitum access to standard food and tap water. The mice were divided into groups of six animals (n = 6). Mice are used in our studies for many years. Therefore, mice were used in this study to compare our new results with previously published results.
Chemicals
Soman was obtained from the Military Technical Institute in Brno (Czech Republic) and was 90.0% pure. Its purity was assayed by acidimetric titration. All other drugs and chemicals of analytical grade were obtained commercially and used without further purification. All substances were administered intramuscularly (i.m.) at a volume of 10 mL/kg body weight (b.w.).
Evaluation of Prophylactic Efficacy of Pyridostigmine and Anti-AD Drugs
Memantine was administered at a dose of 20 mg/kg based on literature (Jackson et al. 2019). Pyridostigmine was administered at a dose of 0.162 mg/kg, donepezil at a dose of 2.65 mg/kg, and rivastigmine at a dose of 1.2 mg/kg. Doses of reversible AChEIs were established based on previous studies (Bruins Slot et al. 2003; Kassa et al. 2012; Kosasa et al. 1999) and were attributed to approximately 40% brain AChE inhibition (Misik and Kassa 2014). The doses of reversible AChEIs were chosen to be sufficiently safe to avoid potential adverse drug reactions in both peripheral and central compartments.
Pyridostigmine and anti-AD drugs, or their respective combinations, were administered i.m. 30 min before i.m. soman injection. Soman-induced toxicity was evaluated by assessing its LD50 value and its 95% confidence interval using probit-logarithmical analysis of death occurring within 24 h after administering soman at five different doses with six animals per dose (Tallarida and Murray 1987). The efficacy of tested prophylactic drugs was expressed as the protective ratio (LD50 value of soman in pretreated mice/LD50 value of soman in non-pretreated mice). The differences between LD50 values were significant when p < 0.05 (Tallarida and Murray 1987).
Evaluation of the Influence of Pyridostigmine and Anti-AD Drugs on the Therapeutic Efficacy of Antidotal Treatment
The pretreatment (memantine, reversible AChEIs, and their combinations) was administered i.m. 30 min before soman to evaluate the impact of pyridostigmine and anti-AD drugs on antidotal efficacy. All doses corresponded with the previously described part of our study. Antidotal (post-exposure) treatment consists of asoxime in a dose corresponding to 5% of its LD50 (33.6 mg/kg) and atropine (10 mg/kg). Both were administered together via i.m. injection 1 min after soman intoxication. Soman-induced toxicity was evaluated as described previously (Tallarida and Murray 1987). Two protective ratios were calculated to evaluate the potential influence of pretreatment on overall antidotal efficacy: (i) protective ratio A (LD50 value of soman in pretreated mice with antidotal treatment/LD50 value of soman in non-pretreated mice without antidotal treatment), (ii) protective ratio B (LD50 value of soman in pretreated mice with antidotal treatment/LD50 value of soman in non-pretreated mice with antidotal treatment). The differences between LD50 values were significant when p < 0.05 (Tallarida and Murray 1987).
Results
The behavioral changes in animals exposed to various treatment regimens are summarized in Table 1. The mice suffered from salivation, respiratory difficulties, muscular twitching and fibrillation, and ultimately tonic–clonic convulsions. The pretreatment of soman poisoning was able to slightly postpone the onset of soman-induced toxic signs and symptoms and slightly diminish their intensity (see Table 1). To compare the time of onset of soman-induced toxic signs and symptoms and the intensity of toxic signs and symptoms, the behavioral changes in mice were observed after administration of soman at the dose corresponding to its LD50 value in each group.
Table 2 shows the overall prophylactic efficacy of pyridostigmine, anti-AD drugs, and their combinations. While peripherally acting pyridostigmine failed to decrease acute soman toxicity, centrally acting rivastigmine and memantine were slightly effective. On the other hand, donepezil decreased the acute toxicity of soman significantly. Combined pretreatment with memantine and AChEIs increased pretreatment efficacy regardless of the AChEI used (see Table 2).
Table 3 gives data on the effect of prophylactic pyridostigmine, anti-AD drugs, and their combinations on the post-exposure treatment of soman toxicity. Pyridostigmine and memantine alone failed to improve the overall therapeutic outcome. Centrally acting reversible AChEIs ameliorated the antidotal treatment only slightly with the best results in donepezil. The pretreatment combinations boosted the overall therapeutic efficacy against soman markedly; memantine and donepezil proved the most effective combination.
Discussion
Effective treatment of acute soman intoxication still proves difficult. The causal antidotes (oximes) often fail because of the rapid aging of the AChE-soman complex (Alozi and Rawas-Qalaji 2020; Antonijevic and Stojiljkovic 2007; Marrs et al. 2006; Mercey et al. 2012). Therefore, appropriate pretreatment regimes may improve the overall treatment efficacy against soman as well as other highly toxic OPs. The commonly used reversible AChEIs (e.g., pyridostigmine) can inhibit/protect some of the AChE with the subsequent spontaneous recovery of AChE activity, boosting the available pool of active enzyme (Bajgar et al. 2009; Lorke and Petroianu 2019).
The potential benefits or risks of long-term pyridostigmine therapy remain unknown. The relatively small therapeutic window also limits its practical use. The safe dose for humans is ineffective against centrally acting OPs in most cases (Dunn et al. 1997). Our study confirms the negligible prophylactic value of pyridostigmine: the protective ratio was 0.92 as pretreatment and 1.02 with post-exposure therapy. Previously published studies bring similar results (Bajgar et al. 2019; Kassa et al. 2017). This limitation may be related to insufficient brain penetration. Numerous alternative AChEIs have also been investigated for possible anti-OP effectiveness (Bajgar et al. 2019; Kassa et al. 2012; Lorke et al. 2011; Lorke and Petroianu 2019). Physostigmine is one of the promising centrally acting AChEIs. It can protect nerve agent–poisoned animals against neurological symptoms and severe behavioral incapacitation (Myhrer and Aas, 2016). Many investigators have found physostigmine to be superior to pyridostigmine in protecting against nerve agents (Leadbeater et al. 1985; Miller et al. 1993). However, it was described that physostigmine can cause undesirable behavioral side effects in high doses; for example, administration of physostigmine results in impaired shuttle-box performance and increased acoustic startle response (Philippens et al. 1996).
The commonly used anti-AD drugs (donepezil and rivastigmine) seem as viable alternatives (Karasova et al. 2020a, b). Their toxicity, brain distribution, and adverse effects have been thoroughly explored (Bures et al. 2020, 2021; Korabecny et al. 2019; Nguyen et al. 2021; Rong et al. 2021; Valis et al. 2017; Zemek et al. 2014). While pyridostigmine administered alone failed, donepezil decreased soman’s acute toxicity significantly (the protective ratio 1.35).
The acute cholinergic crisis, which springs from cholinergic receptor overstimulation, predominates in the first minutes after intoxication. This impairment induces high glutamate release leading to widespread hyper-excitation that causes generalized seizure activity (McDonough and Shih 1997). As it was previously described, administration of soman “convulsive” dose leads to brain lesions observed in the hippocampus, amygdala, and thalamus and consisted of neuron necrosis, dropout, gliosis, astrocytosis, and vascularizations (Britt et al. 2000). Combining compounds with different neuroprotective mechanisms might reduce secondary neuronal damage. Memantine mitigates glutamate excitotoxicity via NMDA receptor antagonism (Marotta et al. 2020; Stojiljkovic et al. 2019). Memantine alone fails to terminate seizure activity because of cholinergic overstimulation (Jackson et al. 2019; Shih et al. 1999). However, it was shown that memantine could attenuate AChE inhibition and prevent myonecrosis and muscle fasciculation and other signs of cholinergic toxicity in rats when administered before soman, sarin, tabun, or VX exposure (Gupta and Dettbarn 1992; McLean et al. 1992). Of note, memantine reduces OP-induced cell death in various vulnerable brain regions such as the amygdala, thalamus, piriform cortex, hippocampus, and parietal cortex (Jackson et al. 2019). Moreover, memantine lowers levels of oxidative stress markers in the cerebrospinal fluid, especially non-protein thiols and 3-nitrotyrosine (Valis et al. 2019). Although we used the maximal recommended dose (20 mg/kg; Stojiljkovic et al. 2019), memantine alone as pretreatment decreased the toxicity of soman only slightly (the protective ratio 1.18) and grants little to no effect in the efficacy of post-exposure treatment (see Tables 2 and 3). A higher dose can induce severe adverse effects; the most common are motor hyperactivity, ataxia, and behavioral impairments (hyperexcitability, stereotypic movements, and convulsions) (Stojiljkovic et al. 2019). This narrow therapeutic window seriously limits its pretreatment utilization, especially when administered alone.
In the AD treatment, a connection between glutamatergic and cholinergic systems was observed. Both possess some implications in cognitive function (Parsons et al. 2013). In the moderate or severe stages of AD, the AChEI effect is supported by memantine. Together, they can synergistically and effectively tackle the AD pathological cascade (Marotta et al. 2020; Tariot and Federoff 2003). This approach may also be beneficial for the pretreatment of highly toxic OPs. Our study demonstrated the benefit of combining memantine with AChEI due to complementary effects (reversible inhibition of AChE and elimination of glutamate release). In all cases, this combination decreased acute soman toxicity significantly (protective ratio 1.42–1.59). The experiments dealing with the influence of prophylactic drugs on the efficacy of post-exposure antidotal treatment brought similar results. Contrary to Stojiljkovic et al. (2019), who described a non-significant improvement of similar post-exposure treatment by memantine, we proved that memantine did not affect the post-exposure treatment of soman poisoning (see Table 3).
On the other hand, the combination of memantine with AChEI increased the effectiveness of post-exposure treatment. Among them, the combination with donepezil gave the best results (protective ratio of 5.12). The explanation may relate to its pharmacodynamic properties: donepezil acts rapidly as reversible, mixed competitive, and non-competitive selective AChEI; the AChE/BChE ratio in humans is 405:1 (Zeb et al. 2017). Donepezil interacts with both the catalytic and peripheral binding sites, resulting in enhanced AChE inhibition (Cheung et al. 2012). Moreover, it also protects against (i) glutamate excitotoxicity via interaction with nicotinic ACh receptors (Takada-Takatori et al. 2006) — possible potentiation of memantine effect; (ii) neural damage via increase of AChE-R expression and inhibition of AChE-S expression (Nordberg 2006); (iii) oxidative stress via alteration of free radical output (Tsukada et al. 2000); and (iv) cerebral ischemia (Chen et al. 2006).
Conclusion
The hypothetical memantine protection is based on (i) reduction of centrally mediated seizures due to non-competitive NMDA receptor antagonism with subsequent decrease of the glutamate-induced release of ACh (Lupp et al. 1992), (ii) protection of neuromuscular transmission against ACh-induced depolarization block following AChE inhibition due to its ability to inhibit the nicotinic receptor-sodium ionophore complex (Tsai et al. 1989), and (iii) partial protection of AChE against soman-induced irreversible inhibition (Stojiljkovic et al. 2019). Based on our results, the benefit of memantine administered alone was insufficient but, combined with AChEI, it is probably able to improve overall resistance against soman. Donepezil seems to be the best choice among selected AChEI. Its pharmacodynamic effects suitably complement the memantine central effect.
Availability of Data and Material
The data and material associated with this article can be found in the online version.
References
Alozi M, Rawas-Qalaji M (2020) Treating organophosphates poisoning: management challenges and potential solutions. Crit Rev Toxicol 50(9):764–779. https://doi.org/10.1080/10408444.2020.1837069
Antonijevic B, Stojiljkovic MP (2007) Unequal efficacy of pyridinium oximes in acute organophosphate poisoning. Clin Med Res 5:71–82
Bajgar J (2004) Organophosphate/nerve agent poisoning: mechanism of action, diagnosis, prophylaxis and treatment. Adv Clin Chem 38:151–216. https://doi.org/10.1016/S0065-2423(04)38006-6
Bajgar J, Fusek J, Kassa J, Kuca K, Jun D (2009) Chemical aspects of pharmacological prophylaxis against nerve agent poisoning. Curr Med Chem 16:2977–2986. https://doi.org/10.2174/092986709788803088
Bajgar J, Kassa J, Kucera T, Musilek K, Jun D, Kuca K (2019) Some possibilities to study new prophylactics against nerve agents. Mini-Rev Med Chem 19(12):970–979. https://doi.org/10.2174/1389557519666190301112530
Britt JO, Martin JL, Okerberg CV, Dick EJ (2000) Histopathologic changes in the brain, heart and skeletal muscle of rhesus macaque, ten days after exposure to soman (an organophosphorus nerve agent). Comp Med 50(2):133–139
Bruins Slot LA, Chopin P, Colpaert FC (2003) Tacrine-Scopolamine Interactions on State-Dependent Retrieval Psychopharmacology (berl) 166:33–39. https://doi.org/10.1007/s00213-002-1292-y
Bures J, Kvetina J, Radochova V, Tacheci I, Peterova E, Herman D, Dolezal R, Kopacova M, Rejchrt S, Douda T, Sestak V, Douda L, Karasova JZ (2020) The pharmacokinetic parameters and the effect of a single and repeated doses of memantine on gastric myoelectric activity in experimental pigs. Plos One 15(1): Article No. e0227781. https://doi.org/10.1371/journal.pone.0227781
Bures J, Tacheci I, Kvetina J, Radochova V, Prchal L, Kohoutova D, Valis M, Novak M, Dolezal R, Kopacova M, Rejchrt S, Sestak V, Knoblochova V, Peterova E, Zdarova Karasova J (2021) The impact of dextran sodium sulfate-induced gastrointestinal injury on the pharmacokinetic parameters of donepezil and its active metabolite 6-O-desmethyldonepezil, and gastric myoelectric activity in experimental pigs. Molecules 26(8):2160. https://doi.org/10.3390/molecules26082160
Colovic MB, Krstic DZ, Lazarevic-Pasti TD, Bondzic AM, Vasic VM (2013) Acetylcholinesterase inhibitors: pharmacology and toxicology. Curr Neuropharmacol 11:315–335. https://doi.org/10.2174/1570159X11311030006
Chambers JE, Dail MB, Meek EC (2020) Oxime-mediated reactivation of organophosphate-inhibited acetylcholinesterase with emphasis on centrally-active oximes. Neuropharmacology 175:No.108201. https://doi.org/10.1016/j.neuropharm.2020.108201
Chen XY, Magnotta VA, Duff K, Boles Ponto LL, Schultz SK (2006) Donepezil effects on cerebral blood flow in older adults with mild cognitive deficits. J Neuropsychiatr Clin Neurosci 18:178–185. https://doi.org/10.1176/appi.neuropsych.18.2.178
Chen Y (2012) Organophosphate-induced brain damage: mechanisms, neuropsychiatric and neurological consequences, and potential therapeutic strategies. Neurotoxicology 33(3):391–400. https://doi.org/10.1016/j.neuro.2012.03.011
Cheung J, Rudolph MJ, Burshteyn F, Cassidy MS, Gary EN, Love J, Franklin MC, Height JJ (2012) Structures of human acetylcholinesterase in complex with pharmacologically important ligands. J Med Chem 55:10282–10286. https://doi.org/10.1021/jm300871x
Dunn MA, Hackley BE, Sidell FR (1997) Pretreatment for nerve agent exposure. In: Zajtchuk R, Bellamy RF (eds.) Textbook of military medicine: medical aspects of chemical & biological warfare. Washington DC: Office of the Surgeon General, Department of the Army, pp 181–196
Gupta RC, Dettbarn WD (1992) Potential of memantine, D-tubocurarine and atropine in preventing acute toxic myopathy induced by organophosphate nerve agents: soman, sarin, tabun and VX. Neurotoxicology 13:649–661
Jackson C, Ardinger Ch, Winter KM, McDonough JH, McCarren HS (2019) Validating a model of benzodiazepine refractory nerve agent-induced status epilepticus by evaluating the anticonvulsant and neuroprotective effects of scopolamine, memantine and phenobarbital. J Pharmacol Toxicol Methods 97:1–12. https://doi.org/10.1016/j.vascn.2019.02.006
Karasova JZ, Hroch M, Pohanka M, Hepnarova V, Pejchal J, Kuca K (2020) Pyridostigmine bromide and its relation to Gulf War illness. Toxin Rev 39(2):138–146. https://doi.org/10.1080/15569543.2018.1480496
Karasova JZ, Hrabinova M, Krejciova M, Jun D, Kuca K (2020) Donepezil and rivastigmine: pharmacokinetic profile and brain-targeting after intramuscular administration in rats. Iran. J Pharm Res 19(3):95–102. https://doi.org/10.22037/ijpr.2019.1100723
Kassa J (2019) Oxime Research. In: Lukey BJ, Romano JA Jr, Salem H (eds.) Chemical warfare agents − biomedical and psychological effects, medical countermeasures and emergency response. Boca Raton, USA, CRC Press pp 681–695
Kassa J, Korabecny J, Nepovimova E (2017) The evaluation of benefit of newly prepared reversible inhibitors of acetylcholinesterase and commonly used pyridostigmine as pharmacological pretreatment of soman-poisoned mice. ACTA MEDICA (HK) 60(1):37–43. https://doi.org/10.14712/18059694.2017.45
Kassa J, Musilek K, Koomlova M, Bajgar J (2012) A comparison of the efficacy of newly developed reversible inhibitors of acetylcholinesterase with commonly used pyridostigmine as pharmacological pretreatment of soman-poisoned mice. Bas Clin Pharmacol Toxicol 110:322–326. https://doi.org/10.1111/j.1742-7843.2011.00808.x
Korabecny J, Spilovska K, Mezeiova E, Benek O, Juza R, Kaping D, Soukup O (2019) A systematic review on donepezil-based derivatives as potential cholinesterase inhibitors for Alzheimer’s disease. Curr Med Chem 26:5625–5648. https://doi.org/10.2174/0929867325666180517094023
Kosasa T, Kuriya Y, Matsui K, Yamanishi Y (1999) Effect of donepezil hydrochloride (E2020) on basal concentration of extracellular acetylcholine in the hippocampus of rats. Eur J Pharmacol 380:101–107. https://doi.org/10.1016/S0014-2999(99)00545-2
Layish I, Krivoy A, Rotman E, Finkelstein A, Tashma Z, Yehezkelli Y (2005) Pharmacologic prophylaxis against nerve agent poisoning. Isr Med Assoc J 7:182–187
Leadbeater L, Inns RH, Ralands JM (1985) Treatment of poisoning by soman. Fund Appl Toxicol 5:S225–S231. https://doi.org/10.1016/0272-0590(85)90132-0
Lorke DE, Hasan MY, Nurulain SM, Shafiullah M, Kuca K, Petroianu GA (2011) Pretreatment for acute exposure to diisopropylfluorophosphate: in vivo efficacy of various acetylcholinesterase inhibitors. J Appl Toxicol 31:515–523. https://doi.org/10.1002/jat.1589
Lorke DE, Kalasz H, Petroianu GA, Tekes K (2008) Entry of oximes into the brain: a review. Curr Med Chem 15:743–753. https://doi.org/10.2174/092986708783955563
Lorke DE, Petroianu GA (2019) Reversible cholinesterase inhibitors as pretreatment for exposure to organophosphates. A review. J Appl Toxicol 39(1):101–116. https://doi.org/10.1002/jat.3662
Lupp A, Lücking CH, Koch R, Jackisch R, Feuerstein TJ (1992) Inhibitory effects of the antiparkinsonian drugs memantine and adamantine on N-methyl-D-aspartate-evoked acetylcholine release in the rabbit caudate nucleus in vitro. J Pharmacol Exp Ther 263(2):717–724
Marotta G, Basagni F, Rosini M, Minarini A (2020) Memantine derivatives as multitarget agents in Alzheimer’s disease. Molecules 25: No. 4005. https://doi.org/10.3390/molecules 25174005
Marrs TC, Rice P, Vale JA (2006) The role of oximes in the treatment of nerve agent poisoning in civilian casualties. Toxicol Rev 25:297–323. https://doi.org/10.2165/00139709-200625040-00009
McDonough JH, Shih TM (1997) Neuropharmacological mechanisms of nerve agent-induced seizure and neuropathology. Neurosci Biobehav Rev 21(5):559–579. https://doi.org/10.1016/S0149-7634(96)00050-4
McLean MJ, Gupta RC, Dettbarn WD, Wamil AW (1992) Prophylactic and therapeutic efficacy of memantine against seizures produced by soman in the rat. Toxicol Appl Pharmacol 112:95–103. https://doi.org/10.1016/0041-008X(92)90284-Y
Mercey G, Verdelet T, Renou J, Kliachyna M, Baati R, Nachon F, Jean L, Renard PY (2012) Reactivators of acetylcholinesterase inhibited by organophosphorus nerve agents. Acc Chem Res 45:756–766. https://doi.org/10.1021/ar2002864
Miller SA, Blick DW, Kerenyi SZ, Murphy MR (1993) Efficacy of physostigmine as a pretreatment for organophosphate poisoning. Pharmacol Biochem Behav 44:343–347. https://doi.org/10.1016/0091-3057(93)90472-6
Misik J, Kassa J (2014) A comparison of cholinesterase inhibitors in the treatment of quinuclidinyl benzilate-induced behavioural deficit in rats performing the multiple T-maze. J Appl Biomed 12(4):211–217. https://doi.org/10.1016/j.jab.2014.01.006
Myhrer T, Aas P (2016) Pretreatment and prophylaxis against nerve agent poisoning: are undesirable behavioral side effects unavoidable? Neurosci Biobehav Rev 71:657–670. https://doi.org/10.1016/jneubiorev.2016.10.017
Nguyen K, Hoffman H, Chakkamparambil B, Grossberg GT (2021) Evaluation of rivastigmine in Alzheimer’s disease. Neurodegen Dis Manag 11(1):35–48. https://doi.org/10.2217/nmt-2020-0052
Nordberg A (2006) Mechanisms behind the neuroprotective actions of cholinesterase inhibitors in Alzheimer disease. Alzheimer Dis Assoc Disord 20(2):S12–S18. https://doi.org/10.1097/01.wad.0000213804.59187.2d
Parsons CG, Danysa W, Dekundy A, Pulte I (2013) Memantine and cholinesterase inhibitors: complementary mechanisms in the treatment of Alzheimer’s disease. Neurotox Res 24:358–369. https://doi.org/10.1007/s12640-013-9398-z
Patocka J, Jun D, Bajgar J, Kuca K (2006) Prophylaxis against nerve agent intoxication. Def Sci J 56:775–784. https://doi.org/10.14429/dsj.56.1941
Philippens IHCHM, Wolthuis OL, Busker RW, Langenberg JP, Melchers BPC (1996) Side effects of physostigmine as a pretreatment in guinea pigs. Pharmacol Biochem Behav 55:99–105. https://doi.org/10.1016/0091-3057(96)83115-7
Rong X, Jiang LW, Qu MJ, ul Hassan SS, Liu ZC, (2021) Enhancing therapeutic efficacy of donepezil by combined therapy: a comprehensive review. Curr Pharm Des 27(3):332–344. https://doi.org/10.2174/1381612826666201023144836
Shih TM, McDonough JH (1997) Neurochemical mechanisms in soman-induced seizures. J Appl Toxicol 17(4):255–264. https://doi.org/10.1002/(SICI)1099-1263(199707)17:4%3c255:AID-JAT441%3e3.0.CO;2-D
Shih TM, McDonough JH, Koplovitz I (1999) Anticonvulsants for soman-induced seizure activity. J Biomed Sci 6:86–96. https://doi.org/10.1007/BF02256439
Stojiljkovic MP, Skrbic R, Jokanovic M, Kilibarda V, Vukovic M (2019) Prophylactic potential of memantine against soman poisoning in rats. Toxicology 416:62–74. https://doi.org/10.1016/j.tox.2019.01.012
Takada-Takatori Y, Kume T, Sugimoto M, Katsuki H, Sugimoto H, Akaik A (2006) Acetylcholinesterase inhibitors used in treatment of Alzheimer’s disease prevent glutamate neurotoxicity via nicotinic acetylcholine receptors and phosphatidylinositol 3-kinase cascade. Neuropharmacology 51:474–486. https://doi.org/10.1016/j.neuropharm.2006.04.007
Tallarida R, Murray R (1987) Manual of pharmacological calculation with computer programs. Springer-Verlag, New York
Tariot PN, Federoff HJ (2003) Current treatment for Alzheimer Disease and future prospects. Alzheimer Dis Assoc Dis 17(Suppl 4):S105–S113. https://doi.org/10.1097/00002093-200307004-00005
Tsai MC, Chen ML, Lo SC, Tsai GC (1989) Effects of memantine on the twitch tension of mouse diaphragm. Eur J Pharmacol 160:133–140. https://doi.org/10.1016/0014-2999(89)90662-6
Tsukada H, Sato K, Kakiuchi T, Nishiyama S (2000) Age-related impairment of coupling mechanism between neuronal activation and functional cerebral blood flow response was restored by cholinesterase inhibition: PET study with microdialysis in the awake monkey brain. Brain Res 857:158–164. https://doi.org/10.1016/S0006-8993(99)02394-X
Valis M, Masopust J, Vysata O, Hort J, Dolezal R, Tomek J, Misik J, Kuca K, Karasova JZ (2017) Concentration of donepezil in the cerebrospinal fluid of AD patients: evaluation of dosage sufficiency in standard treatment strategy. Neurotox Res 31(1):162–168. https://doi.org/10.1007/s12640-016-9672-y
Valis M, Herman D, Vanova N, Masopust J, Vysata O, Hort J, Pavelek Z, Klimova B, Kuca K, Misik J, Karasova JZ (2019) The concentration of memantine in the cerebrospinal fluid of Alzheimer’s disease patients and its consequence to oxidative stress biomarkers. Front Pharmacol 10:943. https://doi.org/10.3389/fphar.2019.00943
Zeb MW, Rias A, Szigeti K (2017) Donepezil: a review of pharmacological characteristics and role in the management of Alzheimer disease. Clin Med Insights Ger 10: No 1179553017695258. https://doi.org/10.1177/1179553017695258
Zemek F, Drtinova L, Nepovimova E, Sepsova V, Korabecny J, Klimes J, Kuca K (2014) Outcomes of Alzheimer’s disease therapy with acetylcholinesterase inhibitors and memantine. Expert Opin Drug Saf 13:759–774. https://doi.org/10.1517/14740338.2014.914168
Zhou ZJ, Dai XF, Gu X, Sun YG, Zheng G, Zheng J (2005) Memantine alleviates toxicity induced by dichlorvos in rats. J Occup Health 47(2), 96–101. https://doi.org/10.1539/joh.47.96
Acknowledgements
The authors would like to thank Ing. J. Hatlapatkova and Bc. I. Kaderabkova for their skillful technical assistance.
Funding
The study was supported by the Ministry of Defence of the Czech Republic through a long-term organization development plan — Medical Aspects of Weapons of Mass Destruction.
Author information
Authors and Affiliations
Contributions
Both authors contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by both authors. The first draft of the manuscript was written by J. Kassa, and both authors commented on precious versions of the manuscript. Both authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All procedures and protocols used in the study were in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. Handling of experimental animals was approved by the Ethics Committee of the Faculty of Military Health Sciences in Hradec Králové (Czech Republic) by approval number 163135/2020–684800.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kassa, J., Karasova, J.Z. Memantine and Its Combination with Acetylcholinesterase Inhibitors in Pharmacological Pretreatment of Soman Poisoning in Mice. Neurotox Res 39, 1487–1494 (2021). https://doi.org/10.1007/s12640-021-00394-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-021-00394-2