Abstract
Background
End-stage renal disease is considered to influence coronary microcirculation and left ventricular (LV) diastolic function. We investigated whether differences exist in LV diastolic function indices and coronary flow reserve (CFR) between patients on hemodialysis (HD) and peritoneal dialysis (PD).
Methods
A complete transthoracic echocardiographic study was performed on 21 HD and 22 PD patients and LV diastolic function was evaluated. CFR was estimated using transthoracic Doppler echocardiography on the left anterior descending artery, during high-dose dypiridamole infusion.
Results
HD and PD groups did not differ regarding Doppler-derived diastolic indices, but they significantly differed in the frequency of severe LV hypertrophy (38.1% in HD vs 4.5% in PD group, p = 0.009) and grade II diastolic dysfunction (42.9% in HD vs 4.5% in PD group, p = 0.004). No patient had restrictive filling pattern. There was no difference in the prevalence of arterial hypertension and diabetes mellitus in patients with grade II vs less than grade II dysfunction. Mean CFR was similar in the HD and PD groups (2.25 ± 0.65 vs 2.36 ± 0.76, p = 0.635) and lower in patients with grade II diastolic dysfunction (1.87 ± 0.43 vs 2.44 ± 0.72, p = 0.023) and diabetes (1.70 ± 0.59 vs 2.39 ± 0.68, p = 0.04). LV mass index was negatively associated with CFR (r = − 0.308, p = 0.045).
Conclusion
Patients on HD had more advanced diastolic dysfunction compared to PD, independently of the presence of hypertension and diabetes. CFR did not differ between HD and PD patients, but it was significantly lower in diabetics and in patients with more advanced diastolic dysfunction.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Left ventricular (LV) diastolic dysfunction displays a high prevalence and a strong prognostic importance for cardiovascular events in patients with end-stage renal disease (ESRD) [1,2,3,4]. Although its associations with LV hypertrophy (LVH), arterial hypertension and diabetes mellitus (DM) have been consistently investigated in the ESRD population [5,6,7,8], studies comparing the influence of different dialysis methods on LV diastolic function are rather sparse and contradictive [9, 10].
ESRD precipitates coronary microvascular disease through several pathophysiologic mechanisms [11,12,13,14]. Coronary microvascular function, in the absence of epicardial artery stenoses, is reflected by the coronary flow reserve (CFR), which can be estimated non-invasively in the left anterior descending artery (LAD) with transthoracic Doppler echocardiography during vasodilator stress testing [15,16,17]. Low CFR is common among dialysis patients without coronary artery disease (CAD) [18,19,20] and predicts increased cardiovascular risk in chronic kidney disease (CKD) patients and renal transplant recipients [21,22,23]. Few studies in the ESRD population have demonstrated the relation of CFR to LV mass index (LVMI) and LV diastolic function [24, 25], while the effect of the dialysis method has not been studied so far.
The purpose of our study was to detect potential differences in LV diastolic function indices and transthoracic echocardiography-derived CFR between patients on hemodialysis (HD) and peritoneal dialysis (PD).
Materials and methods
Study population
In this cross-sectional study, after screening 157 adult patients on a dialysis program (97 on HD and 60 on PD) at University Hospital of Ioannina and General Hospital of Ioannina “G. Hatzikosta”, Greece, we enrolled 21 patients on hemodialysis (HD group) and 22 on peritoneal dialysis (PD group). All patients were at least 3 months on dialysis therapy (HD: three times per week; PD: on continuous ambulatory). Exclusion criteria were: age > 75 years (n = 25), congestive heart failure or left ventricular ejection fraction (LVEF) < 50% (n = 8), known or suspected CAD (including any regional wall motion abnormality on echocardiogram, n = 30), severe valvular heart disease (n = 7), cardiomyopathy (dilated, hypertrophic or restrictive, n = 4), constrictive pericarditis, atrial fibrillation or atrial flutter (n = 8), frequent ventricular or atrial extrasystoles (n = 1), atrioventricular conduction abnormalities or left bundle branch block (n = 2), bronchial asthma or severe chronic obstructive pulmonary disease (n = 7), critical clinical status (n = 15), known dipyridamole hypersensitivity, insufficient echocardiographic imaging (n = 6) and refusal to provide informed consent (n = 1). A comprehensive medical history with standard demographic and clinical parameters was recorded, including age, weight, height, cause of CKD, time since initiation of dialysis therapy (“time on dialysis”), prescribed medications, arterial hypertension, DM, dyslipidemia, smoking habit, family history for CAD and other comorbidities. Blood samples were obtained after an overnight fast and before mid-week dialysis session in HD patients and on the outpatient visit day in PD patients.
Basic echocardiography and diastolic function assessment
A complete transthoracic echocardiogram was performed in HD patients the morning after a dialysis day and in PD patients with an empty abdomen. A commercially available ultrasound machine was used (Vivid 7 Pro; GE Vingmed Ultrasound A/S, Horten, Norway) with a 1.5–3.6 MHz transducer, capable of second harmonic imaging. LV dimensions were measured according to the current cardiac chamber quantification guidelines [26]. Left ventricular mass (LVM) was evaluated using the linear method, according to the Devereux formula [27]. LVMI was calculated by dividing the LVM by the body surface area (BSA). Mild LVH was defined by an LVMI 96–108 g/m2 in women and 116–131 g/m2 in men, moderate LVH by an LVMI 109–121 g/m2 in women and 132–148 g/m2 in men, while severe LVH by an LVMI > 121 g/m2 in women and > 148 g/m2 in men. Left atrial volume (LAV) was measured using the biplane method of disks and then indexed to BSA to render the LAV index (LAVI). LAVI was considered abnormal for values > 34 ml/m2. LV systolic function was assessed by the LV ejection fraction (LVEF), measured with the modified biplane Simpson’s method. Tricuspid annular plane systolic excursion (TAPSE) was measured by M-mode echocardiography between end-diastole and peak systole. Pulmonary artery systolic pressure measurement was based on the tricuspid regurgitant jet velocity with the addition of the right atrial pressure, estimated by the inferior vena cava size and dynamics, according to current guidelines [26].
LV diastolic function was evaluated taking into account all diastolic indices recommended by the current guidelines [28]. The transmitral inflow pattern was depicted with pulse-wave (PW) Doppler echocardiography and used to measure the peak mitral early (E) and late (A) diastolic velocities, the E-wave deceleration time (DT) and the isovolumic relaxation time (IVRT). Tissue Doppler was applied on the septal and lateral portions of the mitral annulus to obtain its peak early (e′) and late (a′) diastolic velocities, as averaged values of their septal and lateral components. An E/e′ ratio > 14 was considered to reflect elevated LV filling pressures. Pulmonary vein systolic (PVS), diastolic (PVD), and atrial flow reversal (PVAr) velocities were also obtained. A comprehensive interpretation of the various diastolic indices facilitated the classification of the diastolic dysfunction into grade 0 (normal diastolic function), grade I (impaired relaxation), grade II (pseudonormalization) and grades III–IV (restricted filling).
Coronary flow reserve measurement
Following the baseline echocardiogram, CFR was evaluated using high-dose dipyridamole intravenous infusion (0.84 mg/Kg over 6 min). Before dipyridamole administration, the distal LAD was visualized in a modified apical four-chamber view as a predominantly diastolic color signal. By applying the PW Doppler, the baseline coronary blood flow was recorded and its peak diastolic velocity was calculated as the average of its three highest measurements. Likewise, after the dipyridamole infusion, the hyperemic coronary peak diastolic velocity was measured and CFR was expressed by the ratio of hyperemic to baseline velocities. CFR < 2.0 was considered abnormal [29, 30]. Immediately after hyperemic coronary flow recording, the LV regional wall motion was assessed, based on established dipyridamole stress echocardiography protocol [15, 16]. During dipyridamole infusion and recovery, all patients had continuous electrocardiographic monitoring and their blood pressure was measured every 2 min. All patients abstained from methylxanthines-containing beverages (coffee, tea, chocolate etc.) for at least 24 h before the study. Depending on the presence of symptoms, an optional dose of intravenous aminophylline (125–250 mg) was administered at the end of the study to reverse the effects of dipyridamole.
Statistical analysis
Statistical analysis was performed using the SPSS software (IBM SPSS Statistics, version 23). Continuous variables are expressed as mean ± SD and categorical variables as percentages. To investigate if continuous variables were described by the normal distribution, the Kolmogorov–Smirnov Z test was used. The Student’s t test and the Mann–Whitney test were used to compare normally and not normally distributed data respectively. For categorical data, the χ2 and the Fischer’s exact test were used as appropriate. Association between continuous variables was determined with the Pearson’s correlation coefficient. p values were always two-sided and a value of p < 0.05 was considered statistically significant.
Results
Demographic, clinical, and laboratory data
With regard to the entire study sample (n = 43), mean age was 56 ± 13.6 years (range 26–75 years), 20 patients (46.5%) were female and mean body mass index (BMI) was 24.4 ± 4.3 kg/m2. The median time on dialysis was 24 months (interquartile range 13–72 months). Primary kidney disease was glomerulonephritis in 7 patients (16.3%), hypertensive nephropathy in 6 (14%), diabetic nephropathy in 4 (9.3%), polycystic kidney disease in 3 (7%), drug-induced in 3 (7%), other in 5 (16.1%), and of unknown etiology in 15 (34.9%). The baseline patient characteristics of the HD and PD groups are summarized in Table 1. No significant difference was found between the two groups with respect to age, gender, BMI, time on dialysis and prevalence of hypertension, dyslipidemia, and smoking habit. The prevalence of DM was higher in the PD group, as was the use of renin–angiotensin–aldosterone system inhibitors, calcium channel blockers, and diuretics. Mean serum albumin was lower in the PD group; no other significant difference was detected regarding laboratory values.
Basic echocardiography and diastolic function
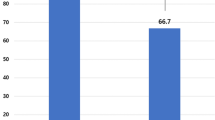
The basic echocardiographic measurements are shown in Table 2. Mean interventricular septal and LV posterior wall thickness was increased in the whole study sample (normal limits 6–9 mm, according to current guidelines [26]). LV cavity and wall dimensions, LVMI, LVEF, and LAVI did not significantly differ between the HD and PD groups. In both female and male subjects in HD, mean LVMI was above established upper normal limits [26] and higher than their PD counterparts, even though on statistically non-significant levels. Nevertheless, the prevalence of severe LVH was significantly higher in the HD group (38.1% vs 4.5%, p = 0.009) (Fig. 1). More patients on HD had an increased LAVI compared to PD (47.6% vs 22.7%), although this finding did not reach statistical significance (p = 0.087).
Between the two study groups, there was no significant difference regarding almost all individual Doppler-derived diastolic function indices (Table 3). However, after classification according to the grade of diastolic dysfunction, the two groups were found to significantly differ in the frequency of grade II (pseudonormalization) diastolic dysfunction (42.9% in HD vs 4.5% in PD group, p = 0.004) (Fig. 2). No patient had restrictive filling pattern (grades III and IV). Moreover, there was no difference in the prevalence of arterial hypertension and diabetes mellitus in patients with grade II vs less than grade II dysfunction (p = 0.458 and p = 1.00, respectively) (Fig. 3). Pseudonormalized pattern was more frequent among subjects with severe LVH compared to those without severe LVH (55.6% vs 14.7%, p = 0.02).
Only two HD and one PD patients (p = 0.607) had elevated LV filling pressures, estimated by an E/e′ ratio > 14 [28]. Time on dialysis had a positive impact on E/e′ ratio in the HD group (r = 0.575, p = 0.006), but not in the PD group (r = − 0,165, p = 0,463). E/e′ ratio was also associated with LVMI (r = 0.442, p = 0.03) and LAVI (r = 0.449, p = 0.003) in the entire study sample. LAVI, in turn, had additionally positive correlation to age (r = 0.34, p = 0.026), mitral E-wave velocity (r = 0.48, p = 0.001) and LVMI (r = 0.703, p < 0.001). Furthermore, LAVI was abnormal more frequently in patients with grade II diastolic dysfunction, compared to those with a better (less than grade II) diastolic pattern (70% vs 24%, p = 0.019), and was also abnormal in all patients with severe LVH (100% vs 17.6% in those without severe LVH, p < 0.001). Prescribed medications and laboratory values were not found to have significant associations with LVMI, LAVI, E/e′ or diastolic dysfunction grade.
Coronary flow reserve
Mean CFR value in the entire study sample was 2.31 (± 0.70), without significant difference between the HD (2.25 ± 0.65) and PD (2.36 ± 0.76) groups (p = 0.635). Peak diastolic coronary flow velocities at baseline and at maximal hyperemia did not differ either between the two groups (Table 4). Eight HD (38.1%) and 6 PD (27.3%) patients had CFR < 2 (p = 0.449). Diabetic patients had lower CFR values compared to non-diabetics (1.70 ± 0.59 vs 2.39 ± 0.68, p = 0.04) (Fig. 4a). Patients receiving calcium channel blockers had lower CFR values as well (2.04 ± 0.58 vs 2.48 ± 0.73, p = 0.038). No other demographic, clinical or laboratory parameter was found to influence CFR, including age and time on dialysis therapy. After excluding patients with DM, the disparities between HD and PD groups regarding the mean CFR value (2.25 ± 0.65 vs 2.55 ± 0.71) and the frequency of CFR < 2 (38.1% vs 11.8%) were larger, although without reaching statistical significance (p = 0.127 and p = 0.136, respectively, Fisher’s exact test).
Significant CFR associations. a Diabetic patients had lower CFR values. b CFR was negatively associated to LVMI. c Mean CFR was lower in patients with grade II diastolic dysfunction compared to those with less than grade II (grade I and normal diastolic function). CFR coronary flow reserve, LVMI left ventricular mass index
Among the several measured echocardiographic parameters, LVMI was found to be negatively associated with CFR (r = − 0.308, p = 0.045) in the total study sample (Fig. 4b). Patients with grade II diastolic dysfunction had significantly lower CFR (1.87 ± 0.43 vs 2.44 ± 0.72, p = 0.023) (Fig. 4c). There was also a trend regarding the association of CFR to E/e′ (r = − 0.297, p = 0.053). No patient had wall motion abnormalities on dypiridamole stress echocardiogram.
Discussion
LVH is very common in the ESRD population and constitutes a strong and independent predictor for adverse cardiovascular outcomes [31,32,33]. A lot of studies have evaluated the LVM in patients on HD in comparison to PD, yielding contradictive results; some researchers found a higher frequency or severity of LVH, either in the HD [34, 35] or in the PD [10, 36, 37] population, while others did not detect any difference between these two renal replacement methods [9, 38]. In our study, HD patients had more frequently severe LVH, even if their mean LVMI value was higher in non-significant levels, compared to their PD counterparts.
Diastolic dysfunction is the dominant LV physiology in ESRD and is closely related etiologically to LVH [4, 39]. In the present study, consistent with these data is the more frequent pseudonormalized pattern among patients with severe LVH. Patients on HD had more advanced diastolic dysfunction compared to patients on PD, whereas hypertension and DM were not found to influence the diastolic pattern, implying that hemodialysis itself may hypothetically play some role for the more advanced diastolic dysfunction. Τhis observation goes along with the strong positive correlation between time on HD therapy and E/e′ ratio. Nevertheless, it should not be misinterpreted, as the additive and independent impact of hypertension and DM on diastolic function has been well demonstrated in the ESRD context [5, 6, 40].
LAVI reflects the long-term effect of elevated LV filling pressures. In our study, significant differences between HD and PD patients were detected neither in the mean value of LAVI nor in the frequency of abnormally increased LAVI, even though an abnormal LAVI was more common in the HD group. As expected, LAVI was positively associated with E/e’ and LVMI and it was more frequently abnormal in subjects with advanced diastolic dysfunction.
Studies comparing the diastolic function between HD and PD patients are rather sparse and contradictive. Günal et al. [9] did not detect significant differences in diastolic function indices between patients on HD and PD, after applying strict salt intake and volume control. On the contrary, de Bie et al. [6] found a higher percentage of HD patients among those dialysis patients with elevated LV filling pressures, while, in a more recent study, Ellouali et al. [10] reported a higher prevalence of diastolic dysfunction (defined as E/e′ > 13) in the PD group. In this latter study, the echocardiogram in the HD group was performed soon after a dialysis session. This could explain, in part, the discrepancy with our study, where HD participants were characterized by a more advanced diastolic pattern compared to those on PD. The absence of restrictive filling pattern from our study and the small number of participants with E/e′ > 14 are, most likely, attributable to the exclusion of patients with congestive heart failure.
Impaired CFR is common among ESRD patients without CAD, reflecting coronary microvascular dysfunction [18,19,20]. To the best of our knowledge, the present study is the first to compare CFR between patients on HD and PD and did not result in any significant difference, despite the slightly higher mean CFR value and the lower frequency of CFR < 2 in the PD group. These differences became more pronounced, but still did not reach statistical significance, after excluding diabetic patients, a subgroup characterized by significantly lower CFR, as expected [41]. Nevertheless, these findings should be interpreted in the light of our small sample size, possibly attenuating the existing difference between the two dialysis methods.
Only a few studies have documented the relation of transthoracic echocardiography-derived CFR to LVM and LV diastolic function in ESRD patients without CAD. Niizuma et al. [18] found a positive correlation between LVMI and peak coronary flow velocity at baseline in HD patients. Bozbas et al. [25] detected a significant association of CFR to LVMI, time on dialysis and diastolic dysfunction in samples of HD patients and renal transplant recipients. More recently, Papamichail et al. [24] reported impaired diastolic function indices in HD patients with a very low CFR value. Accordingly, we have found a negative correlation between CFR and LVMI and lower CFR values in patients with advanced (grade II) diastolic dysfunction.
Limitations
Our study sample was highly selected, after applying strict exclusion criteria, to avoid the impact of several common ESRD comorbidities, like heart failure, CAD, and severe valvular disease, on diastolic function and CFR. Thus, it cannot represent the entire ESRD population and this is partly reflected in the absence of diabetic patients in the HD group. Furthermore, the small number of participants may have mitigated statistically significant differences in individual indices between the two study groups, like LVMI, LAVI, and CFR. This could also explain the absence of statistically overt effect of hypertension and DM on LV diastolic pattern. Finally, we did not perform coronary angiography to exclude underdiagnosed CAD as the reason for low CFR values, owing to the lack of clinical indications and the invasive nature of the procedure. Nevertheless, no wall motion abnormality was detected on dipyridamole stress echocardiogram.
Conclusion
In the present study, patients on HD had more advanced diastolic dysfunction compared to patients on PD, independently of the presence of hypertension and diabetes. CFR was not found to differ between HD and PD patients, but it was significantly lower in diabetics and in patients with pseudonormalized diastolic pattern. Larger studies are required to validate our results and to detect further differences between patients on these two methods of renal replacement therapy.
References
Antlanger M, Aschauer S, Kopecky C, et al. Heart Failure with preserved and reduced ejection fraction in hemodialysis patients: prevalence, disease prediction and prognosis. Kidney Blood Press Res. 2017;42(1):165–76.
Wu CK, Lee JK, Wu YF, et al. Left ventricular diastolic dysfunction in peritoneal dialysis: a forgotten risk factor. Medicine (Baltimore). 2015;94(20):e819.
Han JH, Han JS, Kim EJ, et al. Diastolic dysfunction is an independent predictor of cardiovascular events in incident dialysis patients with preserved systolic function. PLoS ONE. 2015;10(3):e0118694.
Pecoits-Filho R, Bucharles S, Barberato SH. Diastolic heart failure in dialysis patients: mechanisms, diagnostic approach, and treatment. Semin Dial. 2012;25(1):35–41.
Barberato SH, Bucharles SG, Sousa AM, et al. Prevalence and prognostic impact of diastolic dysfunction in patients with chronic kidney disease on hemodialysis. Arq Bras Cardiol. 2010;94(4):457–62.
de Bie MK, Ajmone Marsan N, Gaasbeek A, et al. Left ventricular diastolic dysfunction in dialysis patients assessed by novel speckle tracking strain rate analysis: prevalence and determinants. Int J Nephrol. 2012;2012:963504.
Rosello A, Torregrosa I, Solis MA, et al. Study of diastolic function in peritoneal dialysis patients. Comparison between pulsed doppler and tissue doppler. Nefrologia. 2007;27(4):482–8.
Kimura H, Takeda K, Tsuruya K, et al. Left ventricular mass index is an independent determinant of diastolic dysfunction in patients on chronic hemodialysis: a tissue doppler imaging study. Nephron Clin Pract. 2011;117(1):c67–c73.
Gunal AI, Ilkay E, Kirciman E, et al. Blood pressure control and left ventricular hypertrophy in long-term CAPD and hemodialysis patients: a cross-sectional study. Perit Dial Int. 2003;23(6):563–7.
Ellouali F, Berkchi F, Bayahia R, et al. Comparison of the effects of dialysis methods (haemodialysis vs peritoneal dialysis) on diastolic left ventricular function dialysis methods and diastolic function. Open Cardiovasc Med J. 2016;10:171–8.
Ahmadmehrabi S, Tang WHW. Hemodialysis-induced cardiovascular disease. Semin Dial. 2018;31(3):258–67.
Malyszko J. Mechanism of endothelial dysfunction in chronic kidney disease. Clin Chim Acta. 2010;411(19–20):1412–20.
Meyer C, Heiss C, Drexhage C, et al. Hemodialysis-induced release of hemoglobin limits nitric oxide bioavailability and impairs vascular function. J Am Coll Cardiol. 2010;55(5):454–9.
Gross ML, Ritz E. Hypertrophy and fibrosis in the cardiomyopathy of uremia–beyond coronary heart disease. Semin Dial. 2008;21(4):308–18.
Steeds RP, Wheeler R, Bhattacharyya S, et al. Stress echocardiography in coronary artery disease: a practical guideline from the British Society of Echocardiography. Echo Res Pract. 2019;6(2):G17–G33.
Sicari R, Nihoyannopoulos P, Evangelista A, et al. Stress echocardiography expert consensus statement: European Association of Echocardiography (EAE) (a registered branch of the ESC). Eur J Echocardiogr. 2008;9(4):415–37.
Dimitrow PP, Galderisi M, Rigo F. The non-invasive documentation of coronary microcirculation impairment: role of transthoracic echocardiography. Cardiovasc Ultrasound. 2005;3(18):1476–7120120.
Niizuma S, Takiuchi S, Okada S, et al. Decreased coronary flow reserve in haemodialysis patients. Nephrol Dial Transplant. 2008;23(7):2324–8.
Caliskan Y, Oflaz H, Demirturk M, et al. Coronary flow reserve dysfunction in hemodialysis and kidney transplant patients. Clin Transplant. 2008;22(6):785–93.
Tok D, Gullu H, Erdogan D, et al. Impaired coronary flow reserve in hemodialysis patients: a transthoracic Doppler echocardiographic study. Nephron Clin Pract. 2005;101(4):19.
Nakanishi K, Fukuda S, Shimada K, et al. Prognostic value of coronary flow reserve on long-term cardiovascular outcomes in patients with chronic kidney disease. Am J Cardiol. 2013;112(7):928–32.
Lakkas L, Naka KK, Bechlioulis A, et al. The prognostic role of myocardial strain indices and dipyridamole stress test in renal transplantation patients. Echocardiography. 2020;37(1):62–70.
Tona F, Silvestre C, Rigato M, et al. Coronary microvascular dysfunction predicts long-term outcome in simultaneous pancreas-kidney transplantation. Transplant Proc. 2016;48(2):344–8.
Papamichail N, Bechlioulis A, Lakkas L, et al. Impaired coronary microcirculation is associated with left ventricular diastolic dysfunction in end-stage chronic kidney disease patients. Echocardiography. 2020;00:1–10.
Bozbas H, Pirat B, Demirtas S, et al. Evaluation of coronary microvascular function in patients with end-stage renal disease, and renal allograft recipients. Atherosclerosis. 2009;202(2):498–504.
Lang RM, Badano LP, Mor-Avi V, et al. Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2015;28(1):1–39 e14.
Devereux RB, Alonso DR, Lutas EM, et al. Echocardiographic assessment of left ventricular hypertrophy: comparison to necropsy findings. Am J Cardiol. 1986;57(6):450–8.
Nagueh SF, Smiseth OA, Appleton CP, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2016;29(4):277–314.
Meimoun P, Tribouilloy C. Non-invasive assessment of coronary flow and coronary flow reserve by transthoracic Doppler echocardiography: a magic tool for the real world. Eur J Echocardiogr. 2008;9(4):449–57.
Simova I. Coronary flow velocity reserve assessment with transthoracic doppler echocardiography. Eur Cardiol. 2015;10(1):12–8.
Zoccali C, Benedetto FA, Mallamaci F, et al. Prognostic impact of the indexation of left ventricular mass in patients undergoing dialysis. J Am Soc Nephrol. 2001;12(12):2768–74.
Foley RN, Parfrey PS, Harnett JD, et al. Clinical and echocardiographic disease in patients starting end-stage renal disease therapy. Kidney Int. 1995;47(1):186–92.
Silberberg JS, Barre PE, Prichard SS, et al. Impact of left ventricular hypertrophy on survival in end-stage renal disease. Kidney Int. 1989;36(2):286–90.
Mitsnefes MM, Daniels SR, Schwartz SM, et al. Severe left ventricular hypertrophy in pediatric dialysis: prevalence and predictors. Pediatr Nephrol. 2000;14(10–11):898–902.
Tian JP, Wang T, Wang H, et al. The prevalence of left ventricular hypertrophy in Chinese hemodialysis patients is higher than that in peritoneal dialysis patients. Ren Fail. 2008;30(4):391–400.
Enia G, Mallamaci F, Benedetto FA, et al. Long-term CAPD patients are volume expanded and display more severe left ventricular hypertrophy than haemodialysis patients. Nephrol Dial Transplant. 2001;16(7):1459–64.
Lai S, Molfino A, Russo GE, et al. Cardiac, inflammatory and metabolic parameters: hemodialysis versus peritoneal dialysis. Cardiorenal Med. 2015;5(1):20–30.
Foley RN, Parfrey PS, Harnett JD, et al. Mode of dialysis therapy and mortality in end-stage renal disease. J Am Soc Nephrol. 1998;9(2):267–76.
Parfrey PS, Foley RN. The clinical epidemiology of cardiac disease in chronic renal failure. J Am Soc Nephrol. 1999;10(7):1606–15.
Wang AY, Wang M, Lam CW, et al. Left ventricular filling pressure by Doppler echocardiography in patients with end-stage renal disease. Hypertension. 2008;52(1):107–14.
Kibel A, Selthofer-Relatic K, Drenjancevic I, et al. Coronary microvascular dysfunction in diabetes mellitus. J Int Med Res. 2017;45(6):1901–29.
Funding
The authors received no financial support for the research, authorship, and publication of this article.
Author information
Authors and Affiliations
Contributions
IG: study conception, data acquisition, data analysis, and manuscript preparation. KKN and ED: study conception and design, critical appraisal, and study supervision. LL: study design, data acquisition, and critical appraisal. PM, AD, KK, and RK: enrollment process, data acquisition, and critical appraisal. LKM: critical appraisal and study supervision. CSK: study conception, data analysis, manuscript preparation, and study supervision. All authors provided important intellectual content to this work.
Corresponding author
Ethics declarations
Conflict of interest
Ioannis Gkirdis, Katerina K. Naka, Lampros Lakkas, Panagiota Manolakaki, Anila Duni, Konstantinos Koulousios, Rigas Kalaitzidis, Evangelia Dounousi, Lampros K. Michalis, and Christos S. Katsouras declare that they have no conflict of interest.
Human rights statement
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions.
Informed consent
Informed consent was obtained from all patients for being included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gkirdis, I., Naka, K.K., Lakkas, L. et al. Coronary microcirculation and left ventricular diastolic function: comparison between patients on hemodialysis and peritoneal dialysis. J Echocardiogr 19, 103–112 (2021). https://doi.org/10.1007/s12574-020-00493-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12574-020-00493-7