Abstract
Purpose of Review
During the 20 years since the introduction of intravascular ultrasound (IVUS) to catheterization laboratory, there has been growing evidence supporting the role of IVUS. In this article, we review clinical application of routine use of IVUS with recent evidences, a dominant strategy even in the era of drug-eluting stents.
Recent Findings
IVUS provides pre-procedural information to evaluate stenosis severity and plaque characteristics. In addition, IVUS helps optimal stent deployment, minimizing underexpansion and geographic miss, which are major mechanisms of stent failure. Large-scale clinical trials and meta-analyses have shown that the clinical benefits of IVUS guidance are maximized in complex lesions (left main coronary artery, long lesions and chronic total occlusion). Some recent studies have also supported the cost effectiveness of IVUS-guided PCI especially when there is a high risk of stent failure.
Summary
IVUS provides valuable information about lesion severity, lumen and vessel size, lesion length, and plaque characteristics. By determining appropriate stent sizes and optimizing stenting procedures, IVUS-guided PCI improves clinical outcomes especially in patients with high-risk coronary lesions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Since intravascular ultrasound (IVUS) was introduced in the early 1990s, it has brought profound scientific insights to the understanding of the pathophysiology of coronary artery disease and to clinical decision-making. With a spatial resolution of >150–200 μm, IVUS provides valuable information about stenosis severity, lumen and vessel morphology, lesion length, and plaque characteristics, complementing angiographic images. Current guidelines recommend the use of IVUS for assessing indeterminate left main coronary artery disease and cardiac allograft vasculopathy after heart transplantation (Class IIa). In addition, IVUS is useful for determining the mechanism of stent restenosis (Class IIa) [1, 2]. There are abundant prospective and retrospective data validating the clinical impact of IVUS-guided percutaneous coronary intervention (PCI) and imaging criteria for PCI optimization. Previously, clinical application of IVUS was shown in “Standards for the Acquisition, Measurement, and Reporting of Intravascular Ultrasound Studies: A Report of the ACC Task Force on Clinical Expert Consensus Documents” [3]. In this review, we discuss the role of IVUS in daily practice and its clinical implications, supported by more recent evidences.
Pre-procedural Lesion Evaluation
Severity of Stenosis
Lesion-specific fractional flow reserve (FFR) is considered to be the gold standard for assessing the physiologic significance of coronary artery disease [1, 2, 4,5,6,7]. The Fractional Flow Reserve versus Angiography for Multivessel Evaluation (FAME) trial, which included 1005 patients with multi-vessel disease, demonstrated that FFR-guided PCI resulted in a significantly lower rate of the composite endpoint of death, myocardial infarction (MI) and repeat revascularization compared with angiography-guided PCI [7]. With the current paradigm shift to functional angioplasty, the use of IVUS-derived minimal lumen area (IVUS-MLA) as a surrogate marker for ischemia-producing lesions has been under debate.
Non-left Main Coronary Artery
Table 1 summarizes much of the published data validating IVUS-MLA criteria for predicting an FFR of <0.75–0.80 in non-left main coronary artery (non-LMCA) lesions. Although IVUS-MLA thresholds varied from 2.0 to 4.0 mm2, their diagnostic accuracies were approximately 70% [8,9,10,11,12,13,14,15,16,17,18,19,20]. The overall mean MLA cutoff value from a recent meta-analysis was approximately 2.6 mm2 [21, 22]. Recent studies have attempted to identify subgroup-specific MLA cutoffs according to vessel size and lesion location because the FFR value is influenced by the amount of myocardium subtended to the post-stenotic segment [12,13,14,15, 16••, 19, 20]. For lesions of the mid left anterior descending (LAD) artery or those with a reference vessel diameter of <3.0 mm, the IVUS-MLA cutoff ranges from 2.0 to 2.6 mm2. For lesions of the proximal LAD artery or those with a reference vessel diameter of >3.0 mm, the IVUS-MLA cutoff ranges from 2.8 to 3.2 mm2 [10, 12, 15, 16••, 19, 20]. There is also a trend for the MLA cutoff for LAD artery lesions to have better accuracy for predicting FFR compared with those of other coronary vessels [12, 15, 16••]. However, the correlation between IVUS-MLA and FFR is only modest. In a recent meta-analysis including 11 clinical trials, the sensitivity and specificity of the IVUS-MLA cutoff of 2.6 mm2 for predicting an FFR of <0.80 were 79 and 65%, respectively [21]. Functional significance is influenced by many compounding factors besides MLA including plaque characteristics, lesion location and length, plaque burden, reference vessel size, and the amount of viable myocardium subtended by the lesion; so, IVUS-MLA criteria do not accurately predict ischemia-inducing lesions [23, 24]. It is therefore not recommended for determining stenosis severity for revascularization [1, 2, 25].
Nam et al. compared FFR-guided PCI (FFR < 0.8) with IVUS-guided PCI (MLA < 4.0 mm2) and showed similarly favorable clinical outcomes at 1-year follow-up [26]. In addition, de la Torre Herandez et al. reported no significant differences in 2-year clinical event rates between IVUS-guided PCI (MLA < 4 mm2 in vessels of >3 mm and MLA < 3.5 mm2 in vessels of 2.5–3 mm) and FFR-guided PCI (FFR < 0.75) [27]. Although IVUS-MLA has poor diagnostic accuracy and so cannot replace FFR measurement for assessing the functional significance of non-LMCA with its high negative predictive value, an IVUS-MLA larger than the cutoff may be useful for identifying lesions that can be safely deferred [13, 16••].
Left Main Coronary Artery
Identification of significant stenosis in LMCA is critically important because mortality benefit from revascularization is well established [28, 29]. However, coronary angiography alone has limitations when determining the significance of LMCA lesions and has considerable inter-observer variability [30]. Given the limitations of angiography, noninvasive functional tests such as myocardial perfusion imaging have complementary roles for identifying hemodynamic significance, while it is often non-contributive with balanced ischemia. Several prospective studies and meta-analyses have shown favorable clinical outcomes of FFR-guided deferral in intermediate LMCA [31,32,33,34]. A study evaluating 354 patients with LMCA disease showed that it is safe to defer revascularization with an IVUS-MLA of ≥6 mm2 [35••].
Some studies have correlated IVUS-derived MLA with FFR in isolated LMCA disease. Jasti et al. showed a strong correlation between FFR and IVUS-MLA in angiographic ambiguous LMCA stenosis [36]. The IVUS-MLA cutoff value used to predict an FFR of <0.75 was 5.9 mm2 (sensitivity, 93%; specificity, 95%). Kang et al. reported an IVUS-MLA of 4.8 mm2 as the best predictor of an FFR of <0.8 (sensitivity, 89%; specificity, 83%) in isolated intermediate LM lesions [37]. Recently, Park et al. investigated 112 patients with isolated ostial and shaft LCMA stenosis and showed that an IVUS-MLA of ≤4.5 mm2 was the optimal cutoff for an FFR of ≤0.8 (sensitivity, 77%; specificity, 82%) [38]. Although the IVUS-MLA thresholds varied among different ethnic groups, the overall diagnostic accuracies for predicting functional significance seemed better in LMCA versus non-LMCA, which might be explained by the simplicity of the morphological characteristics of LMCA lesions [37]. However, the presence of concomitant stenosis at the proximal LAD artery or the left circumflex artery limits the practical use of IVUS-MLA in making treatment decisions [24, 39, 40].
Plaque Characteristics and Lesion Morphology
The role of grayscale IVUS in plaque characterization is limited by its poor spatial resolution using 40–45 MHz IVUS transducers. Ultrasound reflection depends on acoustic impedance of the tissue, so the grayscale IVUS approach is to compare the “brightness” of the tissue to the surrounding adventitia (Fig. 1).
Attenuated Plaque
Attenuated plaque is an IVUS finding of hypoechoic or mixed atheromas with ultrasound attenuation but little evidence of calcium [41]. Virtual histology IVUS studies have shown that attenuated plaque corresponds to large necrotic cores and thin-cap fibroatheromas, and histopathologic studies have suggested that echo attenuation is related to microcalcification, hyalinized fibrous tissue, cholesterol crystals, and lipid pools [41,42,43,44]. Histopathologically, 91.4% of echo-attenuated plaques in one study corresponded to either fibroatheroma with a necrotic core or pathological intimal thickening with a lipid pool. Fibroatheromas were found in 97% of plaques with superficial IVUS attenuation [44].
Attenuated plaque on grayscale IVUS indicates a high risk for no-reflow or post-procedural creatine kinase (CK)-MB elevation. The HORIZONS-AMI trial showed that attenuated plaque was present in over 70% of patients with acute myocardial infarction (AMI) and the amount of attenuated plaque correlated with the likelihood of no-reflow after revascularization [45]. In addition, some studies have shown that attenuated plaque is associated with stent thrombosis (ST)-segment elevation MI (STEMI), peri-procedural myocardial necrosis, and no-reflow in patients with coronary artery disease undergoing PCI [45,46,47]. A recent meta-analysis of five clinical trials including 3833 patients showed that incidence of a thrombosis in MI (TIMI) score of 0–2 after PCI was significantly higher in patients with attenuated plaque (28.6%) than in those without attenuated plaque (5.8%) [48]. Endo et al. reported that peri-procedural no-reflow incidence was 18% in STEMI patients but was increased (up to 88%) in patients with long attenuated plaque (>5 mm) and plaque rupture [49]. A recent study demonstrated that large attenuated plaque with a maximal attenuation angle of ≥30° was related to unstable atherosclerotic plaque and predicted OCT-defined thin-cap fibroatheroma (sensitivity, 89%; specificity, 64%) and post-stenting peak CK-MB elevation [50].
Plaque Rupture
Plaque rupture is the most common type of plaque complication, accounting for 70% of AMIs [51, 52]. Studies have reported that IVUS-detected infarct-related plaque rupture in 16–56% of patients [53,54,55,56]. Hong et al. reported that IVUS detected plaque rupture in 66% of culprit lesions and 17% of non-infarct-related arteries in AMI patients [57]. Ruptured plaques cause symptoms in patients with small MLAs and thrombus formation, whereas silent plaque rupture is a form of wound healing that leads to lesion progression [58, 59]. Plaque ruptures without significant stenosis detected secondarily or incidentally at follow-up have not been shown to cause events. Plaque rupture is the most common cause of no-reflow and is associated with the worst clinical outcome [60,61,62]. Kusama et al. [63] showed that plaque rupture was associated with larger infarcts and a higher incidence of no-reflow after PCI. However, plaque rupture findings in IVUS do not always reflect the culprit lesion.
Thrombus
The IVUS hallmarks of thrombus (lobulated mass within the lumen, distinct interface between the presumed thrombus and underlying plaque, scintillating echoes, and blood flow within the thrombus) have limited sensitivity and specificity, and ruling out this diagnosis is only possible if all features are not present.
Positive and Negative Remodeling
Pathology and IVUS studies have demonstrated that coronary arteries are remodeled in response to plaque growth by expansion (positive remodeling) or constriction (negative remodeling) of the vessel wall [52, 64, 65]. Positive remodeling is regarded as compensatory enlargement to maintain coronary blood flow and is associated with a large lipid core, calcification, and macrophage infiltration, which might have a role in arterial cell matrix breakdown [66, 67]. Nakamura et al. [68] reported that among 125 symptomatic patients, positive remodeling at culprit lesions was found in 82% of those with AMI, 78% of those with unstable angina, and 33% of those with stable angina. Negative remodeling is suggested to be a result of an advanced process of atherosclerosis leading to constricting adventitial fibrosis behind plaque [52, 65,66,67]. Compared with positive remodeling, negatively remodeled lesions are longer, more calcified, and more stenosed [69, 70]. Recently, the PROSTECT sub-study [71] showed that positive and negative remodeling was associated with major adverse cardiac events related to non-culprit lesions. In addition, positive remodeling was suggested to be a predictor of peri-procedural MI and no-reflow [72, 73].
Calcification
IVUS is a more accurate tool for detecting calcium than angiography (Fig. 2). In a study including 1155 native-vessel target lesions, IVUS detected calcium in 73% of lesions (vs. 38% by angiography) [74]. However, IVUS did not detect calcium in 14.8% of the segments with micro-calcium deposits or deep calcium hidden behind large necrotic cores [44]. Although calcium is more frequently detected in stable than in unstable lesions, spotty calcium and calcified nodules are likely to be associated with vulnerable lesions [61, 75]. Calcium has been shown to be related to acute procedural complications including bleeding, stent thrombosis, target vessel revascularization, and MI after revascularization [76,77,78,79]. Worse outcomes after PCI in calcified lesions can be explained by the following. First, stent underexpansion (an important predictor of restenosis and stent thrombosis) occurs frequently in calcified lesions. Second, using high-pressure balloon inflation and plaque modification devices in severely calcified lesions is associated with peri-procedural adverse events including no-reflow, dissection, and perforation [78]. Thus, IVUS is indispensable for plaque evaluation and stent optimization in calcified lesions.
Stent Sizing
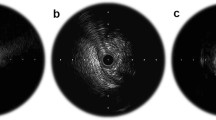
Sizing with the IVUS reference lumen dimension is safe and effective, whereas use of mid-wall or media-to-media dimensions is more aggressive and requires more experience and caution. Although angiography does not always eliminate foreshortening projection, IVUS accurately measures lesion length during motorized pullback, regardless of bend points or a tortuous or foreshortened lesion. Reference segment identification and stent length selection with IVUS ensures that residual plaques at the proximal and distal ends of a stenosis will be completely covered by the stent (Fig. 3).
Stent sizing by intravascular ultrasound. a Coronary stenosis (arrows) on angiography. b Intravascular ultrasound-measured lesion length using a longitudinal view. c A large plaque at the minimal lumen area site. d Measurement of lumen and vessel diameters at the proximal reference segment. e Measurement of lumen and vessel diameters at the distal reference segment
Stent Optimization
An optimally implanted stent has full and symmetrical expansion, complete stent–vessel wall apposition, no plaque prolapse, no dissections or other complications, and no residual edge plaque. IVUS-guided stent optimization is useful for minimizing mechanical problems that can lead to stent failure.
Expansion
The common causes of restenosis in both bare-metal stents (BMSs) and drug-eluting stents (DESs) are intimal hyperplasia and stent underexpansion. Stent underexpansion is correctable and preventable, so there has been a consistent effort to clarify the post-procedural minimal stent area (MSA) necessary to avoid restenosis. For preventing BMS restenosis, studies have reported a post-stenting MSA threshold of 6.4–6.5 mm2 [80,81,82]. For first-generation stents, the post-procedural MSA cutoff was reported as 5.0–5.5 mm2 with sirolimus-eluting stents (SESs) and 5.7 mm2 in paclitaxel-eluting stents [80, 82,83,84,85]. For newer generation stents, Song et al. showed that the optimal cutoff post-stenting MSA values for preventing restenosis were similar between zotarolimus-eluting stents, everolimus-eluting stents, and SESs (5.3, 5.4, and 5.5 mm2, respectively) (Table 2) [85].
Although stent optimization using an absolute MSA cutoff is practical and simple, various vessel sizes should be considered in real practice. In addition, an MSA of >5.0 mm2 may not be achievable in small vessels where stent expansion represented as the MSA/reference lumen area may be a predictor of an adequate lumen at follow-up [84]. The SIRIUS trial reported that the MSA threshold for predicting an adequate follow-up MLA in small coronary arteries (reference vessel diameter <2.8 mm) was 4.5 mm2 in SESs and 6 mm2 in BMSs [82]. For newer-generation DESs, Song et al. suggested an MSA cutoff of 4.9 mm2 in small vessel lesions with reference diameters of 2.5–3.0 mm [85].
To identify the MSA criteria for LMCA, Kang et al. [86•] evaluated 403 patients undergoing SES implantation in LMCA. Based on segmental analysis, the MSA cutoffs for predicting 9-month in-stent restenosis were 5.0 mm2 for the left circumflex artery ostium, 6.3 mm2 for the LAD ostium, 7.2 mm2 for the polygon of confluence, and 8.2 mm2 for the proximal LM, within the corresponding segments. Even in a two-stent group, lesions with complete expansion at all sites showed only a 6% restenosis rate; this was similar to that of a single stent group (6.3%) and non-bifurcation LMCA (4.5%) lesions. Furthermore, post-stenting underexpansion was an independent predictor of 2-year major adverse cardiac events (MACEs).
Residual Plaque Burden
Residual plaque burden is a predictor of late stent thrombosis and edge restenosis. Fujii et al. reported that the presence of a significant residual reference segment stenosis (defined as an edge lumen cross-sectional area of <4 mm2 and a plaque burden of >70%) was more common in the stent thrombosis group compared with the matched control group (67 vs. 9%, p < 0.001) [87]. Okabe et al. suggested that DES patients who developed stent thrombosis had smaller MSAs, more residual disease at the stent edges and larger plaque burdens [88]. Kang et al. evaluated newer generation DESs and suggested that 9-month edge restenosis was predicted by a post-stenting reference segment plaque burden of approximately >55%, and that this could be used to determine the optimal landing site. In addition, the cutoff values of residual plaque burden were similar for the proximal and distal reference segments (56.4 and 51.9%, respectively) (Table 2) [89•]. Angiographic stenosis is poorly correlated with IVUS-measured plaque burden especially in reference segments, so IVUS is an indispensable tool for stent optimization in daily practice [90].
Acute Malapposition
Incomplete stent apposition is defined as the separation of stent struts from the arterial wall with evidence of blood flow behind the strut, where the strut does not cross a side-branch. Acute malapposition is a common finding; across several studies, the prevalence detected by IVUS was 11.5–25% in stable angina patients [91,92,93,94] and 34–40% in STEMI patients [95, 96]. There is little evidence suggesting an association between isolated acute malapposition and adverse outcomes. Most acute stent malapposition resolves over time and does not affect the incidence of stent thrombosis or in-stent restenosis. Guo et al. [97] reported that 40% of acute malapposition in STEMI patients was resolved at 1-year follow-up mainly due to negative remodeling. Hong et al. showed that post-procedure incomplete stent apposition occurred in 7.2% of DES-treated lesions and was not associated with MACEs or even an increased amount of intimal hyperplasia [98, 99]. Similarly, the ADAPT-DES IVUS sub-study showed acute malapposition in 12.6% lesions, but a very low rate of stent thrombosis (0.65%) [96]. Most studies have reported no significant relationship between acute malapposition and the occurrence of MACEs including stent thrombosis [92, 96, 97, 100, 101], and therefore, aggressive additional inflation to eliminate malapposition is unwarranted. Incomplete apposition is not a major concern as long as the stent is well expanded, and underexpansion should be corrected even if there is complete apposition. Large-scale prospective studies are needed to clarify the natural history and clinical impact of malapposition in the long term.
Edge Dissection
With an effort to achieve maximal acute gain during PCI, unexpected vessel dissection can occur at the transition between the rigid stent struts and the adjacent arterial wall at a site of compliance mismatch (Fig. 4) [116]. In one study, IVUS detected edge dissections after 9.2% of DES implantations. Residual plaque eccentricity, lumen-to-stent-edge-area ratio, and stent edge symmetry predicted coronary stent edge dissections, and dissections in less diseased reference segments more often evolved into intramural hematomas [102]. In a HORIZONS-AMI sub-study, significant stent edge dissection (more than medial dissection with a lumen area of <4 mm2 or a dissection angle of ≥60°) was related to early stent thrombosis after primary PCI [101]. The ADAPT-DES IVUS sub-study reported that residual edge dissection was associated with target lesion revascularization at 1-year follow-up and suggested additional treatment with stents to produce a smaller effective lumen area (<5.1 mm2) [126]. Other studies have suggested the predictors of edge dissection as follows: calcified and lipid-rich plaques at the edges of stents, calcification angle, large plaque burden at stent edges, vessel over stretching, stent edge asymmetry, and residual plaque eccentricity [102,103,104,105]. In a current guideline, it is recommended that persistent and high-grade dissection with flow limitation in angiography is treated with prolonged balloon inflation or deployment of a second stent [3]. Conversely, non-flow-limiting minor dissections do not appear to impact on long-term clinical outcomes unless they result in lumen compromise. Low-grade and angiographically silent edge dissections may not be associated with adverse events [116,117,118, 121, 125]. However, some studies have reported a link between edge dissection and adverse clinical outcomes [85, 126, 127].
Intramural Hematoma
As a variant of dissection, intramural hematoma begins as a dissection of the media and propagates along the medial plane into more normal arterial segments without re-entering the lumen [106]. Blood accumulates in the medial space, the EEM expands outward, and the internal elastic membrane is pushed inward to cause lumen compromise. When contrast accumulates within the split media, echolucent contrast can be seen within the echogenic blood. Maehara et al. reported that the incidence of intramural hematomas detected by IVUS was 6.7% after stenting with BMSs [107] and Liu et al. reported the incidence as 3.2% after stenting with DESs [102]. Among IVUS-identified hematomas, 29% were not detected by angiography and 11% appeared as new angiographic stenosis. Moreover, intramural hematoma was associated with a high rate of MI, need for repeat revascularization, and sudden death. Therefore, IVUS has a pivotal role in detecting edge complications and helps with making clinical decisions.
Tissue Protrusion
In the HORIZONS-AMI sub-study, significant tissue protrusion with a lumen area of <4 mm2 was more prevalent in patients with early stent thrombosis after primary PCI [108]. In another study, tissue protrusion was associated with more stent thrombosis and no-reflow in patients with AMI, but was not associated with worse long-term outcomes after stent implantation for infarct-related arteries [109].
Clinical Impact of IVUS-Guided DES Implantation
While early randomized trials with DESs failed to prove superior clinical outcomes in IVUS-guided PCI [110,111,112], recent large-scale registries, randomized trials, and meta-analyses showed that IVUS guidance was associated with a lower rate of MACEs and of the hard endpoints of cardiovascular mortality, MI, and stent thrombosis (ST), compared with angiography-guided PCI [113,114,115,116,117,118]. In the sub-study of ADAPT-DES [119•], the largest prospective registry of 8583 patients (39% of patients treated with IVUS-guided PCI), the IVUS-guided group had lower rates of ST (0.6 vs. 1.0%), MI (2.5 vs. 3.7%), and MACEs (3.1 vs. 4.7%) at 1-year follow-up compared with the angiography-guided group, particularly in patients with acute coronary syndrome and complex lesions.
Table 3 summarizes randomized clinical trials comparing the clinical impact of IVUS- and angiography-guided PCI in the DES era [110,111,112, 116, 120,121,122]. A recent randomized multicenter trial, the Impact of Intravascular Ultrasound Guidance on Outcomes of Xience Prime Stents in Long Lesions (IVUS-XPL), evaluated 1400 patients with long coronary lesions (≥28 mm in stent length). This study proved that IVUS guidance was superior to angiography guidance in terms of improving long-term clinical outcomes [116]. IVUS-guided everolimus-eluting stent implantation significantly reduced the rate of 1-year MACEs (2.9 vs. 5.8%), which was mainly driven by a lower risk of target lesion revascularization (2.5 vs. 5.0%). In a meta-analysis conducted by Elgendy et al., IVUS-guided PCI was beneficial in reducing the rates of MACEs (6.5 vs.10.3%), ischemia-driven target lesion revascularization (4.1 vs. 6.6%), cardiac death (0.5 vs.1.2%), and stent thrombosis (0.6 vs.1.3%) compared with angiography-guided PCI [113]. Zhang et al. conducted a meta-analysis of 29,068 patients undergoing DES implantation and emphasized the benefit of IVUS guidance in reducing ST and MACEs [118].
In lesions with chronic total occlusion (CTO), IVUS can help resolve proximal cap ambiguity by identifying the position of the main branch and clarifying the guidewire position (in true or false lumens) during both antegrade and retrograde CTO crossing attempts. The AIR-CTO randomized controlled trials showed comparable rates of clinical events between IVUS- and angiography-guided PCI [112], whereas the prospective, randomized, multicenter CTO-IVUS trial showed a reduction in 1-year MACE rate after new-generation DES implantation with IVUS guidance [121].
A subgroup analysis from the MAIN-COMPARE registry showed that the 3-year mortality rate was reduced with IVUS compared with angiography guidance (6.3 vs. 13.6%) during LMCA stenting. In particular, the 3-year mortality rates for the 145 matched pairs of patients undergoing DES implantation were significantly lower with IVUS than with angiography guidance (4.7 vs. 16.0%) [123].
Even in the era of DESs, most studies have validated the superiority of IVUS-guided PCI (vs. angiography-guided PCI) to improve clinical outcomes [80,81,82,83, 85, 86•, 87, 89•, 101, 108, 124,125,126,127,128,129,130] by minimizing underexpansion and geographic miss and by treating PCI complications. In the ADAPT-DES study, longer stent lengths, higher inflation pressure, frequent post-dilatation, and larger stent sizes/balloons were used in the IVUS-guided group [119•]. Previous meta-analyses also reported a larger stent size and minimal lumen diameter in the IVUS-guided group (vs. the angiography-guided group) and supported the recommendation to expand the routine use of IVUS guidance [113, 117].
A recent economic analysis showed that IVUS-guided PCI is cost-effective especially in high-risk patients with diabetes, renal insufficiency, and acute coronary syndrome [131]. The incremental cost-effectiveness ratio remained lower than the implicit willingness-to-pay threshold at 1 year, and a negative incremental cost-effectiveness ratio was produced when the IVUS benefit covered full life expectancy. Further randomized trials with cost-effectiveness analyses are necessary to evaluate clinical and cost efficacy of IVUS-guided PCI in routine practice.
Assessment of Stent Failure
IVUS provides insight into the precise mechanisms of in-stent restenosis by giving detailed information about stent underexpansion, the extent and distribution of intimal tissue, plaque progression at the edges of stents, and vascular remodeling (Fig. 5). In a study evaluating lesions with DESs and in-stent restenosis, underexpansion (minimal stent area <5 mm2) and significant intimal hyperplasia (intimal area >50% of stent) were seen in 42 and 93%, respectively [132]. Although intimal hyperplasia is the predominant mechanism of in-stent restenosis, interventionists should focus on correction of stent underexpansion as a preventative mechanism during the procedure. A larger stent can provide more room for future intimal growth, so IVUS guidance may be helpful to avoid underexpansion especially in long lesions, small vessels, and other complex lesions. There is emerging evidence suggesting that chronic inflammation and/or incompetent endothelial function induces in-stent neoatherosclerosis, which is an important mechanism of in-stent restenosis and stent thrombosis in the late phase [133,134,135,136]. IVUS-documented causes of very late stent thrombosis have included in-stent plaque rupture, presumably the consequence of in-stent vulnerable neointima, another manifestation of in-stent neoatherosclerosis [137]. Although the low resolution of IVUS limits identifying macrophage and lipid infiltration within neointima, in-stent intimal rupture and calcified neointima may suggest the presence of advanced neoatherosclerosis.
Conclusion
For planning treatment strategies, IVUS provides valuable information about lesion severity, lumen and vessel size, lesion length, and plaque characteristics. By determining appropriate stent sizes and optimizing stenting procedures, IVUS-guided PCI improves clinical outcomes especially in patients with high-risk coronary lesions.
Abbreviations
- AMI:
-
Acute myocardial infarction
- BMS:
-
Bare-metal stent
- CTO:
-
Chronic total occlusion
- DES:
-
Drug-eluting stent
- FFR:
-
Fractional flow reserve
- ISR:
-
In-stent restenosis
- IVUS:
-
Intravascular ultrasound
- LAD:
-
Left anterior descending
- LMCA:
-
Left main coronary artery
- MLA:
-
Minimum lumen area
- MACE:
-
Major adverse cardiac events
- MI:
-
Myocardial infarction
- MSA:
-
Minimum stent area
- PCI:
-
Percutaneous coronary intervention
- SES:
-
Sirolimus-eluting stent
- ST:
-
Stent thrombosis
- STEMI:
-
ST-segment elevation MI
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Levine GN, Bates ER, Blankenship JC, Bailey SR, Bittl JA, Cercek B, et al. 2011 ACCF/AHA/SCAI guideline for percutaneous coronary intervention. A report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines and the Society for Cardiovascular Angiography and Interventions. J Am Coll Cardiol. 2011;58(24):e44–122. doi:10.1016/j.jacc.2011.08.007.
Kolh P, Windecker S, Alfonso F, Collet JP, Cremer J, Falk V, et al. 2014 ESC/EACTS guidelines on myocardial revascularization: the Task Force on myocardial revascularization of the European Society of Cardiology (ESC) and the European Association for Cardio-Thoracic Surgery (EACTS). Developed with the special contribution of the European Association of Percutaneous Cardiovascular Interventions (EAPCI). Eur J Cardiothorac Surg. 2014;46(4):517–92. doi:10.1093/ejcts/ezu366.
Mintz GS, Nissen SE, Anderson WD, Bailey SR, Erbel R, Fitzgerald PJ, et al. American College of Cardiology clinical expert consensus document on standards for acquisition, measurement and reporting of intravascular ultrasound studies (IVUS). A report of the American College of Cardiology task force on clinical expert consensus documents. J Am Coll Cardiol. 2001;37(5):1478–92.
Bech GJ, De Bruyne B, Pijls NH, de Muinck ED, Hoorntje JC, Escaned J, et al. Fractional flow reserve to determine the appropriateness of angioplasty in moderate coronary stenosis: a randomized trial. Circulation. 2001;103(24):2928–34.
De Bruyne B, Pijls NH, Kalesan B, Barbato E, Tonino PA, Piroth Z, et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N Engl J Med. 2012;367(11):991–1001. doi:10.1056/NEJMoa1205361.
Pijls NH, van Schaardenburgh P, Manoharan G, Boersma E, Bech JW, van't Veer M, et al. Percutaneous coronary intervention of functionally nonsignificant stenosis: 5-year follow-up of the DEFER study. J Am Coll Cardiol. 2007;49(21):2105–11. doi:10.1016/j.jacc.2007.01.087.
Tonino PA, De Bruyne B, Pijls NH, Siebert U, Ikeno F, van’t Veer M, et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009;360(3):213–24. doi:10.1056/NEJMoa0807611.
Takagi A, Tsurumi Y, Ishii Y, Suzuki K, Kawana M, Kasanuki H. Clinical potential of intravascular ultrasound for physiological assessment of coronary stenosis: relationship between quantitative ultrasound tomography and pressure-derived fractional flow reserve. Circulation. 1999;100(3):250–5.
Briguori C, Anzuini A, Airoldi F, Gimelli G, Nishida T, Adamian M, et al. Intravascular ultrasound criteria for the assessment of the functional significance of intermediate coronary artery stenoses and comparison with fractional flow reserve. Am J Cardiol. 2001;87(2):136–41.
Lee CH, Tai BC, Soon CY, Low AF, Poh KK, Yeo TC, et al. New set of intravascular ultrasound-derived anatomic criteria for defining functionally significant stenoses in small coronary arteries (results from Intravascular Ultrasound Diagnostic Evaluation of Atherosclerosis in Singapore [IDEAS] study). Am J Cardiol. 2010;105(10):1378–84. doi:10.1016/j.amjcard.2010.01.002.
Ben-Dor I, Torguson R, Gaglia MA Jr, Gonzalez MA, Maluenda G, Bui AB, et al. Correlation between fractional flow reserve and intravascular ultrasound lumen area in intermediate coronary artery stenosis. EuroIntervention: journal of EuroPCR in collaboration with the Working Group on Interventional Cardiology of the European Society of Cardiology. 2011;7(2):225–33. doi:10.4244/EIJV7I2A37.
Koo BK, Yang HM, Doh JH, Choe H, Lee SY, Yoon CH, et al. Optimal intravascular ultrasound criteria and their accuracy for defining the functional significance of intermediate coronary stenoses of different locations. JACC Cardiovascular interventions. 2011;4(7):803–11. doi:10.1016/j.jcin.2011.03.013.
Ben-Dor I, Torguson R, Deksissa T, Bui AB, Xue Z, Satler LF, et al. Intravascular ultrasound lumen area parameters for assessment of physiological ischemia by fractional flow reserve in intermediate coronary artery stenosis. Cardiovasc Revasc Med. 2012;13(3):177–82. doi:10.1016/j.carrev.2011.12.003.
Kang SJ, Ahn JM, Song H, Kim WJ, Lee JY, Park DW, et al. Usefulness of minimal luminal coronary area determined by intravascular ultrasound to predict functional significance in stable and unstable angina pectoris. Am J Cardiol. 2012;109(7):947–53. doi:10.1016/j.amjcard.2011.11.024.
Chen SL, Xu B, Chen JB, Xu T, Ye F, Zhang JJ, et al. Diagnostic accuracy of quantitative angiographic and intravascular ultrasound parameters predicting the functional significance of single de novo lesions. Int J Cardiol. 2013;168(2):1364–9. doi:10.1016/j.ijcard.2012.12.010.
•• Waksman R, Legutko J, Singh J, Orlando Q, Marso S, Schloss T, et al. FIRST: Fractional Flow Reserve and Intravascular Ultrasound Relationship Study. J Am Coll Cardiol. 2013;61(9):917–23. doi:10.1016/j.jacc.2012.12.012. This study is the first prospective, multicenter registry to examine the correlation of IVUS criteria with FFR values to determine the significance of intermediate coronary lesions. This study showed that the utility of IVUS-MLA as an alternative to FFR in intermediate lesions may be limited in accuracy and should be confirmed clinically.
Doh JH, Koo BK, Nam CW, Kim JH, Min JK, Nakazato R, et al. Diagnostic value of coronary CT angiography in comparison with invasive coronary angiography and intravascular ultrasound in patients with intermediate coronary artery stenosis: results from the prospective multicentre FIGURE-OUT (Functional Imaging criteria for GUiding REview of invasive coronary angiOgraphy, intravascular Ultrasound, and coronary computed Tomographic angiography) study. Eur Heart J Cardiovasc Imaging. 2014;15(8):870–7. doi:10.1093/ehjci/jeu009.
Han JK, Koo BK, Park KW, Ben-Dor I, Waksman R, Pichard A, et al. Optimal intravascular ultrasound criteria for defining the functional significance of intermediate coronary stenosis: an international multicenter study. Cardiology. 2014;127(4):256–62. doi:10.1159/000356480.
Naganuma T, Latib A, Costopoulos C, Takagi K, Naim C, Sato K, et al. The role of intravascular ultrasound and quantitative angiography in the functional assessment of intermediate coronary lesions: correlation with fractional flow reserve. Cardiovasc Revasc Med. 2014;15(1):3–7. doi:10.1016/j.carrev.2013.11.002.
Yang HM, Tahk SJ, Lim HS, Yoon MH, Choi SY, Choi BJ, et al. Relationship between intravascular ultrasound parameters and fractional flow reserve in intermediate coronary artery stenosis of left anterior descending artery: intravascular ultrasound volumetric analysis. Catheterization and cardiovascular interventions: official journal of the Society for Cardiac Angiography & Interventions. 2014;83(3):386–94. doi:10.1002/ccd.25088.
Nascimento BR, de Sousa MR, Koo BK, Samady H, Bezerra HG, Ribeiro AL, et al. Diagnostic accuracy of intravascular ultrasound-derived minimal lumen area compared with fractional flow reserve—meta-analysis: pooled accuracy of IVUS luminal area versus FFR. Catheter Cardiovasc Interv: Off J Soc Cardiac Angiography Interv. 2014;84(3):377–85. doi:10.1002/ccd.25047.
Jang JS, Shin HC, Bae JS, Jin HY, Seo JS, Yang TH, et al. Diagnostic performance of intravascular ultrasound-derived minimal lumen area to predict functionally significant non-left main coronary artery disease: a meta-analysis. Korean Circ J. 2016;46(5):622–31. doi:10.4070/kcj.2016.46.5.622.
Park SJ, Kang SJ, Ahn JM, Shim EB, Kim YT, Yun SC, et al. Visual-functional mismatch between coronary angiography and fractional flow reserve. JACC Cardiovasc Interv. 2012;5(10):1029–36. doi:10.1016/j.jcin.2012.07.007.
Pijls NH, Sels JW. Functional measurement of coronary stenosis. J Am Coll Cardiol. 2012;59(12):1045–57. doi:10.1016/j.jacc.2011.09.077.
Lotfi A, Jeremias A, Fearon WF, Feldman MD, Mehran R, Messenger JC, et al. Expert consensus statement on the use of fractional flow reserve, intravascular ultrasound, and optical coherence tomography: a consensus statement of the Society of Cardiovascular Angiography and Interventions. Catheter Cardiovasc Interv: Off J Soc Cardiac Angiography Interv. 2014;83(4):509–18. doi:10.1002/ccd.25222.
Nam CW, Yoon HJ, Cho YK, Park HS, Kim H, Hur SH, et al. Outcomes of percutaneous coronary intervention in intermediate coronary artery disease: fractional flow reserve-guided versus intravascular ultrasound-guided. JACC Cardiovasc Interv. 2010;3(8):812–7. doi:10.1016/j.jcin.2010.04.016.
de la Torre Hernandez JM, Lopez-Palop R, Garcia Camarero T, Carrillo Saez P, Martin Gorria G, Frutos Garcia A, et al. Clinical outcomes after intravascular ultrasound and fractional flow reserve assessment of intermediate coronary lesions. Propensity score matching of large cohorts from two institutions with a differential approach. EuroIntervention: Journal of EuroPCR in collaboration with the Working Group on Interventional Cardiology of the European Society of Cardiology. 2013;9(7):824–30. doi:10.4244/EIJV9I7A136.
Chaitman BR, Fisher LD, Bourassa MG, Davis K, Rogers WJ, Maynard C, et al. Effect of coronary bypass surgery on survival patterns in subsets of patients with left main coronary artery disease. Report of the collaborative study in coronary artery surgery (CASS). Am J Cardiol. 1981;48(4):765–77.
Yusuf S, Zucker D, Peduzzi P, Fisher LD, Takaro T, Kennedy JW, et al. Effect of coronary artery bypass graft surgery on survival: overview of 10-year results from randomised trials by the coronary artery bypass graft surgery Trialists collaboration. Lancet. 1994;344(8922):563–70.
Lindstaedt M, Spiecker M, Perings C, Lawo T, Yazar A, Holland-Letz T, et al. How good are experienced interventional cardiologists at predicting the functional significance of intermediate or equivocal left main coronary artery stenoses? Int J Cardiol. 2007;120(2):254–61. doi:10.1016/j.ijcard.2006.11.220.
Bech GJ, Droste H, Pijls NH, De Bruyne B, Bonnier JJ, Michels HR, et al. Value of fractional flow reserve in making decisions about bypass surgery for equivocal left main coronary artery disease. Heart. 2001;86(5):547–52.
Hamilos M, Muller O, Cuisset T, Ntalianis A, Chlouverakis G, Sarno G, et al. Long-term clinical outcome after fractional flow reserve-guided treatment in patients with angiographically equivocal left main coronary artery stenosis. Circulation. 2009;120(15):1505–12. doi:10.1161/CIRCULATIONAHA.109.850073.
Lindstaedt M, Yazar A, Germing A, Fritz MK, Holland-Letz T, Mugge A, et al. Clinical outcome in patients with intermediate or equivocal left main coronary artery disease after deferral of surgical revascularization on the basis of fractional flow reserve measurements. Am Heart J. 2006;152(1):156 e1–9. doi:10.1016/j.ahj.2006.03.026.
Mallidi J, Atreya AR, Cook J, Garb J, Jeremias A, Klein LW, et al. Long-term outcomes following fractional flow reserve-guided treatment of angiographically ambiguous left main coronary artery disease: a meta-analysis of prospective cohort studies. Catheter Cardiovasc Interv: Off J Soc Cardiac Angiography Interv. 2015;86(1):12–8. doi:10.1002/ccd.25894.
•• de la Torre Hernandez JM, Hernandez Hernandez F, Alfonso F, Rumoroso JR, Lopez-Palop R, Sadaba M, et al. Prospective application of pre-defined intravascular ultrasound criteria for assessment of intermediate left main coronary artery lesions results from the multicenter LITRO study. J Am Coll Cardiol. 2011;58(4):351–8. doi:10.1016/j.jacc.2011.02.064. This study is the multicenter, prospective registry that assessed the safety of deferred intermediate LMCA lesions with IVUS criteria. This study showed that MLA >6 mm 2 seems to be a safe value for deferring revascularization of the LMCA.
Jasti V, Ivan E, Yalamanchili V, Wongpraparut N, Leesar MA. Correlations between fractional flow reserve and intravascular ultrasound in patients with an ambiguous left main coronary artery stenosis. Circulation. 2004;110(18):2831–6. doi:10.1161/01.CIR.0000146338.62813.E7.
Kang SJ, Lee JY, Ahn JM, Song HG, Kim WJ, Park DW, et al. Intravascular ultrasound-derived predictors for fractional flow reserve in intermediate left main disease. JACC Cardiovasc Interv. 2011;4(11):1168–74. doi:10.1016/j.jcin.2011.08.009.
Park SJ, Ahn JM, Kang SJ, Yoon SH, Koo BK, Lee JY, et al. Intravascular ultrasound-derived minimal lumen area criteria for functionally significant left main coronary artery stenosis. JACC Cardiovasc Interv. 2014;7(8):868–74. doi:10.1016/j.jcin.2014.02.015.
Daniels DV, van't Veer M, Pijls NH, van der Horst A, Yong AS, De Bruyne B, et al. The impact of downstream coronary stenoses on fractional flow reserve assessment of intermediate left main disease. JACC Cardiovasc Interv. 2012;5(10):1021–5. doi:10.1016/j.jcin.2012.07.005.
Yong AS, Daniels D, De Bruyne B, Kim HS, Ikeno F, Lyons J, et al. Fractional flow reserve assessment of left main stenosis in the presence of downstream coronary stenoses. Circ Cardiovasc Interv. 2013;6(2):161–5. doi:10.1161/CIRCINTERVENTIONS.112.000104.
Wu X, Maehara A, Mintz GS, Kubo T, Xu K, Choi SY, et al. Virtual histology intravascular ultrasound analysis of non-culprit attenuated plaques detected by grayscale intravascular ultrasound in patients with acute coronary syndromes. Am J Cardiol. 2010;105(1):48–53. doi:10.1016/j.amjcard.2009.08.649.
Amano H, Wagatsuma K, Yamazaki J, Ikeda T. Virtual histology intravascular ultrasound analysis of attenuated plaque and ulcerated plaque detected by gray scale intravascular ultrasound and the relation between the plaque composition and slow flow/no reflow phenomenon during percutaneous coronary intervention. J Interv Cardiol. 2013;26(3):295–301. doi:10.1111/joic.12035.
Hara H, Tsunoda T, Moroi M, Kubota T, Kunimasa T, Shiba M, et al. Ultrasound attenuation behind coronary atheroma without calcification: mechanism revealed by autopsy. Acute Card Care. 2006;8(2):110–2. doi:10.1080/14628840600637781.
Pu J, Mintz GS, Biro S, Lee JB, Sum ST, Madden SP, et al. Insights into echo-attenuated plaques, echolucent plaques, and plaques with spotty calcification: novel findings from comparisons among intravascular ultrasound, near-infrared spectroscopy, and pathological histology in 2,294 human coronary artery segments. J Am Coll Cardiol. 2014;63(21):2220–33. doi:10.1016/j.jacc.2014.02.576.
Wu X, Mintz GS, Xu K, Lansky AJ, Witzenbichler B, Guagliumi G, et al. The relationship between attenuated plaque identified by intravascular ultrasound and no-reflow after stenting in acute myocardial infarction: the HORIZONS-AMI (Harmonizing outcomes with revascularization and stents in acute myocardial infarction) trial. JACC Cardiovasc Interv. 2011;4(5):495–502. doi:10.1016/j.jcin.2010.12.012.
Lee T, Kakuta T, Yonetsu T, Takahashi K, Yamamoto G, Iesaka Y, et al. Assessment of echo-attenuated plaque by optical coherence tomography and its impact on post-procedural creatine kinase-myocardial band elevation in elective stent implantation. JACC Cardiovasc Interv. 2011;4(5):483–91. doi:10.1016/j.jcin.2010.12.013.
Shiono Y, Kubo T, Tanaka A, Tanimoto T, Ota S, Ino Y, et al. Impact of attenuated plaque as detected by intravascular ultrasound on the occurrence of microvascular obstruction after percutaneous coronary intervention in patients with ST-segment elevation myocardial infarction. JACC Cardiovasc Interv. 2013;6(8):847–53. doi:10.1016/j.jcin.2013.01.142.
Jia R, Nie X, Li H, Zhu H, Pu L, Li X, et al. Impact of attenuated plaques on TIMI grade flow and clinical outcomes of coronary artery disease patients: a systematic review and meta analysis. J Thorac Dis. 2016;8(3):527–36. doi:10.21037/jtd.2016.02.51.
Endo M, Hibi K, Shimizu T, Komura N, Kusama I, Otsuka F, et al. Impact of ultrasound attenuation and plaque rupture as detected by intravascular ultrasound on the incidence of no-reflow phenomenon after percutaneous coronary intervention in ST-segment elevation myocardial infarction. JACC Cardiovasc Interv. 2010;3(5):540–9. doi:10.1016/j.jcin.2010.01.015.
Kang SJ, Ahn JM, Han S, Park DW, Lee SW, Kim YH, et al. Multimodality imaging of attenuated plaque using grayscale and virtual histology intravascular ultrasound and optical coherent tomography. Catheter Cardiovasc Interv: Off J Soc Cardiac Angiography Interv. 2016;88(1):E1–E11. doi:10.1002/ccd.25786.
Naghavi M, Libby P, Falk E, Casscells SW, Litovsky S, Rumberger J, et al. From vulnerable plaque to vulnerable patient: a call for new definitions and risk assessment strategies: part I. Circulation. 2003;108(14):1664–72. doi:10.1161/01.CIR.0000087480.94275.97.
Falk E, Nakano M, Bentzon JF, Finn AV, Virmani R. Update on acute coronary syndromes: the pathologists’ view. Eur Heart J. 2013;34(10):719–28. doi:10.1093/eurheartj/ehs411.
Fukuda D, Kawarabayashi T, Tanaka A, Nishibori Y, Taguchi H, Nishida Y, et al. Lesion characteristics of acute myocardial infarction: an investigation with intravascular ultrasound. Heart. 2001;85(4):402–6.
Rioufol G, Finet G, Ginon I, Andre-Fouet X, Rossi R, Vialle E, et al. Multiple atherosclerotic plaque rupture in acute coronary syndrome: a three-vessel intravascular ultrasound study. Circulation. 2002;106(7):804–8.
Kotani J, Mintz GS, Castagna MT, Pinnow E, Berzingi CO, Bui AB, et al. Intravascular ultrasound analysis of infarct-related and non-infarct-related arteries in patients who presented with an acute myocardial infarction. Circulation. 2003;107(23):2889–93. doi:10.1161/01.CIR.0000072768.80031.74.
Sano T, Tanaka A, Namba M, Nishibori Y, Nishida Y, Kawarabayashi T, et al. C-reactive protein and lesion morphology in patients with acute myocardial infarction. Circulation. 2003;108(3):282–5. doi:10.1161/01.CIR.0000079173.84669.4F.
Hong MK, Mintz GS, Lee CW, Kim YH, Lee SW, Song JM, et al. Comparison of coronary plaque rupture between stable angina and acute myocardial infarction: a three-vessel intravascular ultrasound study in 235 patients. Circulation. 2004;110(8):928–33. doi:10.1161/01.CIR.0000139858.69915.2E.
Fujii K, Kobayashi Y, Mintz GS, Takebayashi H, Dangas G, Moussa I, et al. Intravascular ultrasound assessment of ulcerated ruptured plaques: a comparison of culprit and nonculprit lesions of patients with acute coronary syndromes and lesions in patients without acute coronary syndromes. Circulation. 2003;108(20):2473–8. doi:10.1161/01.CIR.0000097121.95451.39.
Burke AP, Kolodgie FD, Farb A, Weber DK, Malcom GT, Smialek J, et al. Healed plaque ruptures and sudden coronary death: evidence that subclinical rupture has a role in plaque progression. Circulation. 2001;103(7):934–40.
Hayashi T, Kiyoshima T, Matsuura M, Ueno M, Kobayashi N, Yabushita H, et al. Plaque erosion in the culprit lesion is prone to develop a smaller myocardial infarction size compared with plaque rupture. Am Heart J. 2005;149(2):284–90. doi:10.1016/j.ahj.2004.06.020.
Jia H, Abtahian F, Aguirre AD, Lee S, Chia S, Lowe H, et al. In vivo diagnosis of plaque erosion and calcified nodule in patients with acute coronary syndrome by intravascular optical coherence tomography. J Am Coll Cardiol. 2013;62(19):1748–58. doi:10.1016/j.jacc.2013.05.071.
Higuma T, Soeda T, Abe N, Yamada M, Yokoyama H, Shibutani S, et al. A combined optical coherence tomography and intravascular ultrasound study on plaque rupture, plaque erosion, and calcified nodule in patients with ST-segment elevation myocardial infarction: incidence, morphologic characteristics, and outcomes after percutaneous coronary intervention. JACC Cardiovasc Interv. 2015;8(9):1166–76. doi:10.1016/j.jcin.2015.02.026.
Kusama I, Hibi K, Kosuge M, Nozawa N, Ozaki H, Yano H, et al. Impact of plaque rupture on infarct size in ST-segment elevation anterior acute myocardial infarction. J Am Coll Cardiol. 2007;50(13):1230–7. doi:10.1016/j.jacc.2007.07.004.
Glagov S, Weisenberg E, Zarins CK, Stankunavicius R, Kolettis GJ. Compensatory enlargement of human atherosclerotic coronary arteries. N Engl J Med. 1987;316(22):1371–5. doi:10.1056/NEJM198705283162204.
Bentzon JF, Otsuka F, Virmani R, Falk E. Mechanisms of plaque formation and rupture. Circ Res. 2014;114(12):1852–66. doi:10.1161/CIRCRESAHA.114.302721.
Varnava AM, Mills PG, Davies MJ. Relationship between coronary artery remodeling and plaque vulnerability. Circulation. 2002;105(8):939–43.
Burke AP, Kolodgie FD, Farb A, Weber D, Virmani R. Morphological predictors of arterial remodeling in coronary atherosclerosis. Circulation. 2002;105(3):297–303.
Nakamura M, Nishikawa H, Mukai S, Setsuda M, Nakajima K, Tamada H, et al. Impact of coronary artery remodeling on clinical presentation of coronary artery disease: an intravascular ultrasound study. J Am Coll Cardiol. 2001;37(1):63–9.
Mintz GS, Kent KM, Pichard AD, Satler LF, Popma JJ, Leon MB. Contribution of inadequate arterial remodeling to the development of focal coronary artery stenoses. An intravascular ultrasound study. Circulation. 1997;95(7):1791–8.
Maehara A, Mintz GS, Bui AB, Walter OR, Castagna MT, Canos D, et al. Morphologic and angiographic features of coronary plaque rupture detected by intravascular ultrasound. J Am Coll Cardiol. 2002;40(5):904–10.
Inaba S, Mintz GS, Farhat NZ, Fajadet J, Dudek D, Marzocchi A, et al. Impact of positive and negative lesion site remodeling on clinical outcomes: insights from PROSPECT. JACC Cardiovasc Imaging. 2014;7(1):70–8. doi:10.1016/j.jcmg.2013.10.007.
Mehran R, Dangas G, Mintz GS, Lansky AJ, Pichard AD, Satler LF, et al. Atherosclerotic plaque burden and CK-MB enzyme elevation after coronary interventions : intravascular ultrasound study of 2256 patients. Circulation. 2000;101(6):604–10.
Watanabe T, Nanto S, Uematsu M, Ohara T, Morozumi T, Kotani J, et al. Prediction of no-reflow phenomenon after successful percutaneous coronary intervention in patients with acute myocardial infarction: intravascular ultrasound findings. Circ J: Off J Jpn Circ Soc. 2003;67(8):667–71.
Mintz GS, Popma JJ, Pichard AD, Kent KM, Satler LF, Chuang YC, et al. Patterns of calcification in coronary artery disease. A statistical analysis of intravascular ultrasound and coronary angiography in 1155 lesions. Circulation. 1995;91(7):1959–65.
Mintz GS. Intravascular imaging of coronary calcification and its clinical implications. JACC Cardiovasc Imaging. 2015;8(4):461–71. doi:10.1016/j.jcmg.2015.02.003.
Ertelt K, Genereux P, Mintz GS, Reiss GR, Kirtane AJ, Madhavan MV, et al. Impact of the severity of coronary artery calcification on clinical events in patients undergoing coronary artery bypass grafting (from the acute catheterization and urgent intervention Triage strategy trial). Am J Cardiol. 2013;112(11):1730–7. doi:10.1016/j.amjcard.2013.07.038.
Genereux P, Madhavan MV, Mintz GS, Maehara A, Kirtane AJ, Palmerini T, et al. Relation between coronary calcium and major bleeding after percutaneous coronary intervention in acute coronary syndromes (from the acute catheterization and urgent intervention triage strategy and harmonizing outcomes with revascularization and stents in acute myocardial infarction trials). Am J Cardiol. 2014;113(6):930–5. doi:10.1016/j.amjcard.2013.11.053.
Genereux P, Madhavan MV, Mintz GS, Maehara A, Palmerini T, Lasalle L, et al. Ischemic outcomes after coronary intervention of calcified vessels in acute coronary syndromes. Pooled analysis from the HORIZONS-AMI (harmonizing outcomes with revascularization and stents in acute myocardial infarction) and ACUITY (acute catheterization and urgent intervention triage strategy) TRIALS. J Am Coll Cardiol. 2014;63(18):1845–54. doi:10.1016/j.jacc.2014.01.034.
Genereux P, Redfors B, Witzenbichler B, Arsenault MP, Weisz G, Stuckey TD, et al. Two-year outcomes after percutaneous coronary intervention of calcified lesions with drug-eluting stents. Int J Cardiol. 2016; doi:10.1016/j.ijcard.2016.12.150.
Doi H, Maehara A, Mintz GS, Yu A, Wang H, Mandinov L, et al. Impact of post-intervention minimal stent area on 9-month follow-up patency of paclitaxel-eluting stents: an integrated intravascular ultrasound analysis from the TAXUS IV, V, and VI and TAXUS ATLAS workhorse, long lesion, and direct stent trials. JACC Cardiovasc Interv. 2009;2(12):1269–75. doi:10.1016/j.jcin.2009.10.005.
Morino Y, Honda Y, Okura H, Oshima A, Hayase M, Bonneau HN, et al. An optimal diagnostic threshold for minimal stent area to predict target lesion revascularization following stent implantation in native coronary lesions. Am J Cardiol. 2001;88(3):301–3.
Sonoda S, Morino Y, Ako J, Terashima M, Hassan AH, Bonneau HN, et al. Impact of final stent dimensions on long-term results following sirolimus-eluting stent implantation: serial intravascular ultrasound analysis from the sirius trial. J Am Coll Cardiol. 2004;43(11):1959–63. doi:10.1016/j.jacc.2004.01.044. S0735109704005807 [pii]
Hong MK, Mintz GS, Lee CW, Park DW, Choi BR, Park KH, et al. Intravascular ultrasound predictors of angiographic restenosis after sirolimus-eluting stent implantation. Eur Heart J. 2006;27(11):1305–10. doi:10.1093/eurheartj/ehi882.
Okumura M, Ozaki Y, Ishii J, Kan S, Naruse H, Matsui S, et al. Restenosis and stent fracture following sirolimus-eluting stent (SES) implantation. Circ J: Off J Jpn Circ Soc. 2007;71(11):1669–77.
• Song HG, Kang SJ, Ahn JM, Kim WJ, Lee JY, Park DW, et al. Intravascular ultrasound assessment of optimal stent area to prevent in-stent restenosis after zotarolimus-, everolimus-, and sirolimus-eluting stent implantation. Catheter Cardiovasc Interv: Off J Soc Cardiac Angiography Interv. 2014;83(6):873–8. doi:10.1002/ccd.24560. This data showed that smaller stent area is predictive of angiographic restenosis of the second generation DES and optimal cutoff values of poststenting MSA for preventing restenosis were similar between ZES(5.3mm 2 ) vs. EES(5.4mm 2 ) vs. SES(5.5mm 2 ) .
• Kang SJ, Ahn JM, Song H, Kim WJ, Lee JY, Park DW, et al. Comprehensive intravascular ultrasound assessment of stent area and its impact on restenosis and adverse cardiac events in 403 patients with unprotected left main disease. Circ Cardiovasc Interv. 2011;4(6):562–9. doi:10.1161/CIRCINTERVENTIONS.111.964643. This paper showed that smaller IVUS-MSA predict angiographic ISR after DES implantation in LM disease and the best IVUS-MSA criteria to predict angiographic ISR on a segmental basis were 5.0 mm 2 for the LCX ostium, 6.3 mm 2 for the LAD ostium, 7.2 mm 2 for the POC, and 8.2 mm 2 for the proximal LM above the POC.
Fujii K, Carlier SG, Mintz GS, Yang YM, Moussa I, Weisz G, et al. Stent underexpansion and residual reference segment stenosis are related to stent thrombosis after sirolimus-eluting stent implantation: an intravascular ultrasound study. J Am Coll Cardiol. 2005;45(7):995–8. doi:10.1016/j.jacc.2004.12.066.
Okabe T, Mintz GS, Buch AN, Roy P, Hong YJ, Smith KA, et al. Intravascular ultrasound parameters associated with stent thrombosis after drug-eluting stent deployment. Am J Cardiol. 2007;100(4):615–20. doi:10.1016/j.amjcard.2007.03.072.
• Kang SJ, Cho YR, Park GM, Ahn JM, Kim WJ, Lee JY, et al. Intravascular ultrasound predictors for edge restenosis after newer generation drug-eluting stent implantation. Am J Cardiol. 2013;111(10):1408–14. doi:10.1016/j.amjcard.2013.01.288. This data showed that angiographic diameter stenosis in the reference segment after stenting was poorly correlated with plaque burden assessed by IVUS and edge restenosis was predicted by post-stenting reference segment plaque burden >55% after newer drug-eluting stent implantation.
Mintz GS, Painter JA, Pichard AD, Kent KM, Satler LF, Popma JJ, et al. Atherosclerosis in angiographically “normal” coronary artery reference segments: an intravascular ultrasound study with clinical correlations. J Am Coll Cardiol. 1995;25(7):1479–85.
Hoffmann R, Guagliumi G, Musumeci G, Reimers B, Petronio AS, Disco C, et al. Vascular response to sirolimus-eluting stents delivered with a nonaggressive implantation technique: comparison of intravascular ultrasound results from the multicenter, randomized E-SIRIUS, and SIRIUS trials. Catheter Cardiovasc Interv: Off J Soc Cardiac Angiography Interv. 2005;66(4):499–506. doi:10.1002/ccd.20542.
Kimura M, Mintz GS, Carlier S, Takebayashi H, Fujii K, Sano K, et al. Outcome after acute incomplete sirolimus-eluting stent apposition as assessed by serial intravascular ultrasound. Am J Cardiol. 2006;98(4):436–42. doi:10.1016/j.amjcard.2006.02.050.
Tanabe K, Serruys PW, Degertekin M, Grube E, Guagliumi G, Urbaszek W, et al. Incomplete stent apposition after implantation of paclitaxel-eluting stents or bare metal stents: insights from the randomized TAXUS II trial. Circulation. 2005;111(7):900–5. doi:10.1161/01.CIR.0000155607.54922.16.
Kandzari DE, Leon MB, Popma JJ, Fitzgerald PJ, O'Shaughnessy C, Ball MW, et al. Comparison of zotarolimus-eluting and sirolimus-eluting stents in patients with native coronary artery disease: a randomized controlled trial. J Am Coll Cardiol. 2006;48(12):2440–7. doi:10.1016/j.jacc.2006.08.035.
van der Hoeven BL, Liem SS, Dijkstra J, Bergheanu SC, Putter H, Antoni ML, et al. Stent malapposition after sirolimus-eluting and bare-metal stent implantation in patients with ST-segment elevation myocardial infarction: acute and 9-month intravascular ultrasound results of the MISSION! Intervention study. JACC Cardiovasc Interv. 2008;1(2):192–201. doi:10.1016/j.jcin.2008.02.003.
Wang B, Mintz GS, Witzenbichler B, Souza CF, Metzger DC, Rinaldi MJ, et al. Predictors and long-term clinical impact of acute stent malapposition: an assessment of dual antiplatelet therapy with drug-eluting stents (ADAPT-DES) intravascular ultrasound substudy. J Am Heart Assoc. 2016;5(12) doi:10.1161/JAHA.116.004438.
Guo N, Maehara A, Mintz GS, He Y, Xu K, Wu X, et al. Incidence, mechanisms, predictors, and clinical impact of acute and late stent malapposition after primary intervention in patients with acute myocardial infarction: an intravascular ultrasound substudy of the harmonizing outcomes with revascularization and stents in acute myocardial infarction (HORIZONS-AMI) trial. Circulation. 2010;122(11):1077–84. doi:10.1161/CIRCULATIONAHA.109.906040.
Hong MK, Mintz GS, Lee CW, Park DW, Lee SW, Kim YH, et al. Impact of late drug-eluting stent malapposition on 3-year clinical events. J Am Coll Cardiol. 2007;50(15):1515–6. doi:10.1016/j.jacc.2007.07.038.
Hong MK, Mintz GS, Lee CW, Park DW, Park KM, Lee BK, et al. Late stent malapposition after drug-eluting stent implantation: an intravascular ultrasound analysis with long-term follow-up. Circulation. 2006;113(3):414–9. doi:10.1161/CIRCULATIONAHA.105.563403.
Steinberg DH, Mintz GS, Mandinov L, Yu A, Ellis SG, Grube E, et al. Long-term impact of routinely detected early and late incomplete stent apposition: an integrated intravascular ultrasound analysis of the TAXUS IV, V, and VI and TAXUS ATLAS workhorse, long lesion, and direct stent studies. JACC Cardiovasc Interv. 2010;3(5):486–94. doi:10.1016/j.jcin.2010.03.007.
Choi SY, Witzenbichler B, Maehara A, Lansky AJ, Guagliumi G, Brodie B, et al. Intravascular ultrasound findings of early stent thrombosis after primary percutaneous intervention in acute myocardial infarction: a harmonizing outcomes with revascularization and stents in acute myocardial infarction (HORIZONS-AMI) substudy. Circ Cardiovasc Interv. 2011;4(3):239–47. doi:10.1161/CIRCINTERVENTIONS.110.959791.
Liu X, Tsujita K, Maehara A, Mintz GS, Weisz G, Dangas GD, et al. Intravascular ultrasound assessment of the incidence and predictors of edge dissections after drug-eluting stent implantation. JACC Cardiovascular interventions. 2009;2(10):997–1004. doi:10.1016/j.jcin.2009.07.012.
Chamie D, Bezerra HG, Attizzani GF, Yamamoto H, Kanaya T, Stefano GT, et al. Incidence, predictors, morphological characteristics, and clinical outcomes of stent edge dissections detected by optical coherence tomography. JACC Cardiovasc Interv. 2013;6(8):800–13. doi:10.1016/j.jcin.2013.03.019.
Gonzalo N, Serruys PW, Okamura T, Shen ZJ, Garcia-Garcia HM, Onuma Y, et al. Relation between plaque type and dissections at the edges after stent implantation: an optical coherence tomography study. Int J Cardiol. 2011;150(2):151–5. doi:10.1016/j.ijcard.2010.03.006.
Kobayashi N, Mintz GS, Witzenbichler B, Metzger DC, Rinaldi MJ, Duffy PL, et al. Prevalence, features, and prognostic importance of edge dissection after drug-eluting stent implantation: an ADAPT-DES intravascular ultrasound Substudy. Circ Cardiovasc Interv. 2016;9(7):e003553. doi:10.1161/CIRCINTERVENTIONS.115.003553.
Block PC, Myler RK, Stertzer S, Fallon JT. Morphology after transluminal angioplasty in human beings. N Engl J Med. 1981;305(7):382–5. doi:10.1056/NEJM198108133050706.
Maehara A, Mintz GS, Bui AB, Castagna MT, Walter OR, Pappas C, et al. Incidence, morphology, angiographic findings, and outcomes of intramural hematomas after percutaneous coronary interventions: an intravascular ultrasound study. Circulation. 2002;105(17):2037–42.
Choi SY, Maehara A, Cristea E, Witzenbichler B, Guagliumi G, Brodie B, et al. Usefulness of minimum stent cross sectional area as a predictor of angiographic restenosis after primary percutaneous coronary intervention in acute myocardial infarction (from the HORIZONS-AMI trial IVUS substudy). Am J Cardiol. 2012;109(4):455–60. doi:10.1016/j.amjcard.2011.10.005.
Hong YJ, Jeong MH, Choi YH, Song JA, Kim DH, Lee KH, et al. Impact of tissue prolapse after stent implantation on short- and long-term clinical outcomes in patients with acute myocardial infarction: an intravascular ultrasound analysis. Int J Cardiol. 2013;166(3):646–51. doi:10.1016/j.ijcard.2011.11.092.
Chieffo A, Latib A, Caussin C, Presbitero P, Galli S, Menozzi A, et al. A prospective, randomized trial of intravascular-ultrasound guided compared to angiography guided stent implantation in complex coronary lesions: the AVIO trial. Am Heart J. 2013;165(1):65–72. doi:10.1016/j.ahj.2012.09.017.
Kim JS, Kang TS, Mintz GS, Park BE, Shin DH, Kim BK, et al. Randomized comparison of clinical outcomes between intravascular ultrasound and angiography-guided drug-eluting stent implantation for long coronary artery stenoses. JACC Cardiovasc Interv. 2013;6(4):369–76. doi:10.1016/j.jcin.2012.11.009.
Tian NL, Gami SK, Ye F, Zhang JJ, Liu ZZ, Lin S, et al. Angiographic and clinical comparisons of intravascular ultrasound- versus angiography-guided drug-eluting stent implantation for patients with chronic total occlusion lesions: two-year results from a randomised AIR-CTO study. EuroIntervention : Journal of EuroPCR in Collaboration with the Working Group on Interventional Cardiology of the European Society of Cardiology. 2015;10(12):1409–17. doi:10.4244/EIJV10I12A245.
Elgendy IY, Mahmoud AN, Elgendy AY, Bavry AA. Outcomes with intravascular ultrasound-guided stent implantation: a meta-analysis of randomized trials in the era of drug-eluting stents. Circ Cardiovasc Interv. 2016;9(4):e003700. doi:10.1161/CIRCINTERVENTIONS.116.003700.
• Shin DH, Hong SJ, Mintz GS, Kim JS, Kim BK, Ko YG, et al. Effects of intravascular ultrasound-guided versus angiography-guided new-generation drug-eluting stent implantation: meta-analysis with individual patient-level data from 2,345 randomized patients. JACC Cardiovasc Interv. 2016;9(21):2232–9. doi:10.1016/j.jcin.2016.07.021. This paper is meta-analysis of three randomized trials included individual patient–level data treated with new-generation DES which showed that IVUS-guided DES implantation was associated with a reduction in MACEs for the treatment of complex coronary lesions.
Steinvil A, Zhang YJ, Lee SY, Pang S, Waksman R, Chen SL, et al. Intravascular ultrasound-guided drug-eluting stent implantation: an updated meta-analysis of randomized control trials and observational studies. Int J Cardiol. 2016;216:133–9. doi:10.1016/j.ijcard.2016.04.154.
Hong SJ, Kim BK, Shin DH, Nam CM, Kim JS, Ko YG, et al. Effect of intravascular ultrasound-guided vs angiography-guided everolimus-eluting stent implantation: the IVUS-XPL randomized clinical trial. JAMA. 2015;314(20):2155–63. doi:10.1001/jama.2015.15454.
Ahn JM, Kang SJ, Yoon SH, Park HW, Kang SM, Lee JY, et al. Meta-analysis of outcomes after intravascular ultrasound-guided versus angiography-guided drug-eluting stent implantation in 26,503 patients enrolled in three randomized trials and 14 observational studies. Am J Cardiol. 2014;113(8):1338–47. doi:10.1016/j.amjcard.2013.12.043.
Zhang YJ, Pang S, Chen XY, Bourantas CV, Pan DR, Dong SJ, et al. Comparison of intravascular ultrasound guided versus angiography guided drug eluting stent implantation: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2015;15:153. doi:10.1186/s12872-015-0144-8.
•• Witzenbichler B, Maehara A, Weisz G, Neumann FJ, Rinaldi MJ, Metzger DC, et al. Relationship between intravascular ultrasound guidance and clinical outcomes after drug-eluting stents: the assessment of dual antiplatelet therapy with drug-eluting stents (ADAPT-DES) study. Circulation. 2014;129(4):463–70. doi:10.1161/CIRCULATIONAHA.113.003942. This is substudy from ADAPT-DES, the largest study comparing outcomes of IVUS guidance versus angiographic guidance of DES implantation. This study showed that IVUS guidance was strongly associated with lower 1-year rates of definite/probable thrombosis, MI, and MACE, as well as ischemic TLR and TVR especially in ACS and complex coronary anatomy group.
Jakabcin J, Spacek R, Bystron M, Kvasnak M, Jager J, Veselka J, et al. Long-term health outcome and mortality evaluation after invasive coronary treatment using drug eluting stents with or without the IVUS guidance. Randomized control trial. HOME DES IVUS. Catheter Cardiovasc Interv: Off J Soc Cardiac Angiography Interv. 2010;75(4):578–83. doi:10.1002/ccd.22244.
Kim BK, Shin DH, Hong MK, Park HS, Rha SW, Mintz GS, et al. Clinical impact of intravascular ultrasound-guided chronic Total occlusion intervention with zotarolimus-eluting versus biolimus-eluting stent implantation: randomized study. Circ Cardiovasc Interv. 2015;8(7):e002592. doi:10.1161/CIRCINTERVENTIONS.115.002592.
Tan Q, Wang Q, Liu D, Zhang S, Zhang Y, Li Y. Intravascular ultrasound-guided unprotected left main coronary artery stenting in the elderly. Saudi Med J. 2015;36(5):549–53. doi:10.15537/smj.2015.5.11251.
Park SJ, Kim YH, Park DW, Lee SW, Kim WJ, Suh J, et al. Impact of intravascular ultrasound guidance on long-term mortality in stenting for unprotected left main coronary artery stenosis. Circ Cardiovasc Interv. 2009;2(3):167–77. doi:10.1161/CIRCINTERVENTIONS.108.799494.
Ziada KM, Kapadia SR, Belli G, Houghtaling PL, De Franco AC, Ellis SG, et al. Prognostic value of absolute versus relative measures of the procedural result after successful coronary stenting: importance of vessel size in predicting long-term freedom from target vessel revascularization. Am Heart J. 2001;141(5):823–31. doi:10.1067/mhj.2001.114199.
Liu X, Doi H, Maehara A, Mintz GS, Costa Jde R Jr, Sano K, et al. A volumetric intravascular ultrasound comparison of early drug-eluting stent thrombosis versus restenosis. JACC Cardiovasc Interv. 2009;2(5):428–34. doi:10.1016/j.jcin.2009.01.011.
Fujii K, Mintz GS, Kobayashi Y, Carlier SG, Takebayashi H, Yasuda T, et al. Contribution of stent underexpansion to recurrence after sirolimus-eluting stent implantation for in-stent restenosis. Circulation. 2004;109(9):1085–8. doi:10.1161/01.CIR.0000121327.67756.19.
Liu J, Maehara A, Mintz GS, Weissman NJ, Yu A, Wang H, et al. An integrated TAXUS IV, V, and VI intravascular ultrasound analysis of the predictors of edge restenosis after bare metal or paclitaxel-eluting stents. Am J Cardiol. 2009;103(4):501–6. doi:10.1016/j.amjcard.2008.10.010.
Cheneau E, Leborgne L, Mintz GS, Kotani J, Pichard AD, Satler LF, et al. Predictors of subacute stent thrombosis: results of a systematic intravascular ultrasound study. Circulation. 2003;108(1):43–7. doi:10.1161/01.CIR.0000078636.71728.40.
Sakurai R, Ako J, Morino Y, Sonoda S, Kaneda H, Terashima M, et al. Predictors of edge stenosis following sirolimus-eluting stent deployment (a quantitative intravascular ultrasound analysis from the SIRIUS trial). Am J Cardiol. 2005;96(9):1251–3. doi:10.1016/j.amjcard.2005.06.066.
Costa MA, Angiolillo DJ, Tannenbaum M, Driesman M, Chu A, Patterson J, et al. Impact of stent deployment procedural factors on long-term effectiveness and safety of sirolimus-eluting stents (final results of the multicenter prospective STLLR trial). Am J Cardiol. 2008;101(12):1704–11. doi:10.1016/j.amjcard.2008.02.053.
Alberti A, Giudice P, Gelera A, Stefanini L, Priest V, Simmonds M, et al. Understanding the economic impact of intravascular ultrasound (IVUS). Eur J Health Econ. 2016;17(2):185–93. doi:10.1007/s10198-015-0670-4.
Kang SJ, Mintz GS, Park DW, Lee SW, Kim YH, Whan Lee C, et al. Mechanisms of in-stent restenosis after drug-eluting stent implantation: intravascular ultrasound analysis. Circ Cardiovasc Interv. 2011;4(1):9–14. doi:10.1161/CIRCINTERVENTIONS.110.940320.
Kang SJ, Mintz GS, Akasaka T, Park DW, Lee JY, Kim WJ, et al. Optical coherence tomographic analysis of in-stent neoatherosclerosis after drug-eluting stent implantation. Circulation. 2011;123(25):2954–63. doi:10.1161/CIRCULATIONAHA.110.988436.
Nakazawa G, Otsuka F, Nakano M, Vorpahl M, Yazdani SK, Ladich E, et al. The pathology of neoatherosclerosis in human coronary implants bare-metal and drug-eluting stents. J Am Coll Cardiol. 2011;57(11):1314–22. doi:10.1016/j.jacc.2011.01.011.
Nakazawa G, Vorpahl M, Finn AV, Narula J, Virmani R. One step forward and two steps back with drug-eluting-stents: from preventing restenosis to causing late thrombosis and nouveau atherosclerosis. JACC Cardiovasc Imaging. 2009;2(5):625–8. doi:10.1016/j.jcmg.2009.01.011.
Yonetsu T, Kim JS, Kato K, Kim SJ, Xing L, Yeh RW, et al. Comparison of incidence and time course of neoatherosclerosis between bare metal stents and drug-eluting stents using optical coherence tomography. Am J Cardiol. 2012;110(7):933–9. doi:10.1016/j.amjcard.2012.05.027.
Lee CW, Kang SJ, Park DW, Lee SH, Kim YH, Kim JJ, et al. Intravascular ultrasound findings in patients with very late stent thrombosis after either drug-eluting or bare-metal stent implantation. J Am Coll Cardiol. 2010;55(18):1936–42. doi:10.1016/j.jacc.2009.10.077.
Morino Y, Tamiya S, Masuda N, Kawamura Y, Nagaoka M, Matsukage T, et al. Intravascular ultrasound criteria for determination of optimal longitudinal positioning of sirolimus-eluting stents. Circ J: Off J Jpn Circ Soc. 2010;74(8):1609–16.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Both authors declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Intravascular Imaging
Rights and permissions
About this article
Cite this article
Song, HG., Kang, SJ. Current Clinical Applications of Intravascular Ultrasound in Coronary Artery Disease. Curr Cardiovasc Imaging Rep 10, 28 (2017). https://doi.org/10.1007/s12410-017-9424-2
Published:
DOI: https://doi.org/10.1007/s12410-017-9424-2