Abstract
Cardiac risk assessment prior to kidney and liver transplantation is controversial. Given the paucity of available organs, selecting appropriate recipients with favorable short- and long-term cardiovascular risk profile is crucial. Using noninvasive cardiac imaging tools to guide cardiovascular risk assessment and management can also be challenging and controversial. In this article, we address the burden of coronary artery disease among kidney and liver transplant candidates and review the literature pertaining to the diagnostic accuracy and the prognostic value of noninvasive cardiac imaging techniques in this population.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cardiac risk assessment prior to liver and kidney transplantation is controversial. Due to the paucity of available organs, the selection of appropriate recipients with an acceptable post-operative and long-term cardiovascular risk profile is critical. The general perioperative cardiovascular evaluation guidelines and the appropriate use criteria are not applicable in, or intended for, this special population.1,2 The American Heart Association (AHA) and American College of Cardiology (ACC) have recently addressed the evaluation and management of cardiac disease in this population in a comprehensive consensus statement, endorsed by the American Society of Transplant Surgeons, the American Society of Transplantation, and the National Kidney Foundation.3 In this review, we will discuss coronary artery disease (CAD) burden in patients undergoing kidney and liver transplantation evaluation and expound on the use of cardiac imaging in the assessment of CAD in this population within the context of the recent AHA/ACC consensus statement.
Kidney Transplantation
In 2011, more than 17,000 kidney transplantations were performed in the United States. There are currently more than 100,000 patients listed for kidney transplantation with a median waiting time of 2.6 years.4 It is established that a successful kidney transplant improves the quality of life and reduces the mortality risk when compared with maintenance dialysis.5 Although transplantation confers the highest survival benefit among all the different renal replacement therapies, renal allograft recipients still have a high mortality rate as compared to the general population.6
Coronary Artery Disease Burden
Cardiovascular disease is nearly endemic in the end-stage renal disease (ESRD) population. Multiple investigators have shown that approximately 80% of patients being evaluated for kidney transplant had angiographic findings of at least mild coronary atherosclerosis, with up to 45% having significant disease.7,8 Congestive heart failure is also widely prevalent, as more than 50% of patients awaiting transplant carry this diagnosis.4 At all times after kidney transplantation, cardiovascular disease is the leading cause of death in patients with functioning allografts, accounting for nearly 40% of mortality, with the highest rates being in the peri-operative period.9 Therefore, careful cardiovascular evaluation is paramount prior to listing for kidney transplantation.
Coronary Disease Surveillance
The purpose of preoperative cardiac risk evaluation in ESRD patients is the reduction of cardiovascular short- and long-term morbidity and mortality. As in other aspects of cardiovascular disease, screening asymptomatic patients should be used only if the benefits of screening outweigh the harm, and if the findings would lead to management changes aimed at improving patient outcome. The ACC/AHA guidelines on perioperative cardiovascular evaluation and care for non-cardiac surgery were not designed for and not necessarily applicable to this special population.1
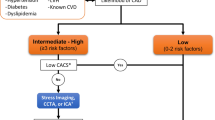
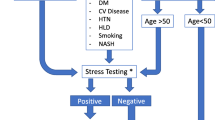
Several observational studies have identified independent risk predictors of cardiovascular disease and cardiac mortality in kidney transplant candidates. These include, but are not limited to: diabetes mellitus, left ventricular hypertrophy, age >50 years, more than 1 year on dialysis, tobacco abuse, hypertension, left ventricular ejection fraction <40%, and dyslipidemia.10-13 Despite a general agreement on these risk predictors, there has been some disagreement between the recommendations from various professional societies on how to select candidates for additional noninvasive evaluation.3,14-16 The 2012 AHA/ACC consensus statement on the evaluation and management of cardiac disease among kidney and liver transplant candidates states that “noninvasive stress testing may be considered in kidney transplantation candidates with no active cardiac conditions on the basis of the presence of multiple CAD risk factors regardless of functional status (Class IIb – Level of Evidence C)”.3 Relevant risk factors among transplantation candidates, as per the 2012 AHA/ACC statement, include: diabetes mellitus, prior cardiovascular disease, >1 year on dialysis, left ventricular hypertrophy, age >60 years, smoking, hypertension, and dyslipidemia.3 The statement indicated that “the specific number of risk factors that should be used to prompt testing remains to be determined, but the committee considers ≥3 to be reasonable”.3 The weak “class IIB - level of evidence C” recommendation highlights the lack of scientific evidence, not only as it pertains to risk stratification prior to transplantation, but more importantly, as to whether such testing and the resultant downstream interventions can improve the outcome of transplant recipients.
Despite the high prevalence of CAD among kidney transplant candidates, routine coronary angiography is not justified since the data is conflicting as to whether significant angiographic coronary stenoses alone can predict subsequent clinical events. De Lima et al reported that in patients undergoing transplant evaluation, there was a statistically significant increase in cardiac events in those with coronary stenosis ≥70%.17 In contrast, Hage et al showed that the prevalence and severity of CAD was not predictive of survival, and coronary revascularization did not impact survival except in ESRD patients with three-vessel disease.18 Furthermore, Jeloka et al showed that patients who underwent pre-transplant coronary revascularization still had a 43% cardiac event rate in the post-transplant period.19 Thus, “routine” coronary catheterization with subsequent revascularization may not reduce adverse event rates post-transplant. Therefore, noninvasive testing seems to be a reasonable approach. Exercise ECG stress, without imaging, is often not feasible in ESRD patients due to poor functional status as well as high prevalence of left ventricular hypertrophy which limits the specificity of the test.3 Moreover, ESRD patients often have uncontrolled hypertension which may impede performing a maximal symptom-limited exercise stress test. Currently, dobutamine stress echocardiography (DSE) and vasodilator stress myocardial perfusion imaging (MPI) with single photon emission computed tomography (SPECT) remain the mainstays of CAD surveillance in this population.
Vasodilator and catecholamine pharmacologic stress agents are believed to be safe in patients with ESRD. Although, regadenoson, a novel A2A receptor-agonist vasodilator stress agent, is predominantly cleared by the kidneys (57%), its use has been shown to be safe and well tolerated in ESRD patients, with the exception of excess gastrointestinal side effects as compared to controls.20,21 Intravenous aminophylline use can prevent these side effects.22,23
Diagnostic Accuracy of Noninvasive Testing
Myocardial Perfusion Imaging and Stress Echocardiography
Several studies have examined the diagnostic accuracy of DSE and SPECT-MPI in this population (Table 1). The pooled analysis of seven SPECT-MPI studies yielded a 66% sensitivity and 75% specificity for coronary stenosis ≥70% (Table 1). Figure 1 depicts examples of normal (A) and abnormal (B) SPECT-MPI from kidney transplant candidates. The diagnostic accuracy of DSE for coronary stenosis of ≥70% is comparable; a pooled analysis of nine studies yielded a sensitivity of 73% and a specificity of 88% (Table 1). A recent Cochrane database systematic review suggested that DSE had improved accuracy over SPECT-MPI in detecting coronary artery stenosis ≥50%. However, when only studies assessing the predictive accuracy for a coronary stenosis threshold of ≥70% were included, the results were not significantly different.24 The reason behind the relatively low MPI sensitivity for CAD in ESRD patients is unclear, but may be relevant in light of recent reports suggesting poor sensitivity of SPECT imaging in other study populations.25,26 Poor SPECT-MPI sensitivity may be related to the fact that all previously published reports utilized older SPECT technology, which lacks the use of attenuation correction and other hardware and software advancements. Furthermore, the low sensitivity of stress testing probably reflects the occasional discordance between coronary anatomy and physiology which may be particularly relevant in an asymptomatic patient population.27 It is well established in other settings that functionally insignificant lesions, though anatomically severe, may not translate into worse outcomes. Moreover, it has been extensively documented in nuclear cardiology literature that SPECT-MPI provides incremental prognostic value beyond coronary angiography.28 The predictive accuracy for DSE and SPECT-MPI seems comparable; therefore, the choice of testing modality should be based upon the expertise of the particular transplant center.3
SSS, Summed stress score; SDS, summed difference score; TID, transient ischemic dilation; LVEF, post-stress gated-SPECT left ventricular ejection fraction; LVMi, left ventricular mass indexed to the body surface area; LVEDVi, left ventricular end-diastolic volume indexed to the body surface area. (A) Rest-regadenoson stress 99mTc-tetrofosmine SPECT myocardial perfusion imaging of a 39-year-old African American man who was evaluated for kidney transplantation. He had been on hemodialysis for 3 years due to end-stage kidney disease caused by long standing uncontrolled hypertension. He has electrocardiographic signs of left ventricular hypertrophy. As illustrated, his myocardial perfusion was normal; the inferior wall defect was likely caused by diaphragmatic attenuation, as the gated-SPECT imaging was normal. Note left ventricular dilatation (LVEDVi = 84 mL·m−2; normal is <75 mL·m−2 for a male) due to chronic left ventricular volume overload caused by longstanding systemic shunting (arterial-venous fistula used for dialysis). (B) Rest-regadenoson stress 99mTc-tetrofosmine SPECT myocardial perfusion images of a 59-year-old man who was evaluated for kidney transplantation. He has a history of hypertension, dyslipidemia, long standing diabetes mellitus, and end-stage renal disease presumed to be due to diabetic nephropathy. He was recently initiated on hemodialysis. Note a severe, predominantly reversible perfusion abnormality in the apical anterior, apical lateral, and apical segments of the left ventricle. The gated SPECT images demonstrated stress-induced hypokinesis in the aforementioned segments. Coronary angiography confirmed severe stenosis in a large diagonal branch of the left anterior descending coronary artery.
While the superiority of positron emission tomography (PET) as compared to SPECT in the general population has been well described,29 the diagnostic accuracy of positron emission tomography specifically in ESRD patients has not been reported in the literature. This also holds true for hybrid imaging including PET/computed tomography (CT) and SPECT/CT.
Cardiac Computed Tomography
This technique is a less well studied noninvasive imaging modality in this population. Coronary CT angiography is frequently avoided in ESRD patients due to the high levels of coronary calcification causing “blooming artifacts”.30 However, Mao et al demonstrated that nearly one-third of pre-kidney transplant patients on dialysis may not have significant coronary calcifications and have interpretable coronary CT angiography with minimal to no CAD. Thus, in younger patients on short duration of dialysis, coronary CT angiography seems to be a good option.31 A few studies assessed the diagnostic accuracy of cardiac CT in comparison to invasive coronary angiography in patients with ESRD.32-37 Budoff and co-workers showed that coronary calcium score has a reasonable discriminative capacity for coronary atherosclerosis in ESRD patients, with areas under the curve of 0.77 and 0.75 for detecting coronary stenoses ≥50% and ≥70%, respectively.32 Rosario et al showed that Agatston’s score ≥400 was associated with a higher number of significant stenoses by invasive coronary angiography.34 Park et al reported that as compared to invasive coronary angiography, the sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) of coronary CT angiography for severe (≥70%) coronary stenosis was 100%, 59%, 44%, and 100%, respectively.35
The Prognostic Value of Noninvasive Cardiac Imaging
Identifying patients at risk for perioperative and long-term major adverse cardiac events is paramount in pre-transplant evaluation. Several studies evaluated the prognostic value of SPECT-MPI. Wong et al demonstrated that patients with a reversible defect on MPI were three times more likely to have a fatal cardiac event or death in the peri-operative period.38 Similarly, Iskandiran and co-workers showed that patients with a normal MPI pre-transplant were five times less likely to have a cardiac event by 42 months post-transplant.39 Most recently, in a retrospective analysis of 387 patients who underwent pre-transplant SPECT-MPI, Chew et al reported that patients with reversible perfusion defects are 4.4 times more likely to have a “soft end point” of coronary revascularization or admission with angina within 5 years post-transplant.40 They also showed that patients with a reversible perfusion defect trended toward having a statistically insignificant higher rate of “hard events” of cardiac death or MI (Figure 2).
Survival after kidney transplant based on perfusion imaging results. Kaplan-Meier curves for soft end-point and hard end-point events in myocardial perfusion imaging positive and negative groups. Hard end points included admission for an acute myocardial infarction or cardiac death. Soft end-points included admission for unstable angina, percutaneous coronary intervention, or coronary artery bypass grafting. From Chew et al Nephrology 2013;18:376-81, with permission.40
While there is no current data on the prognostication of perfusion PET imaging in ESRD patients, there is some information on the use of coronary flow reserve. Murthy et al showed that using PET coronary flow reserve improved risk stratification beyond comprehensive clinical assessment, LV systolic function and semi-quantitative measures of myocardial ischemia.41
A small number of studies have evaluated the prognostic value of DSE in this population. Bergeron et al demonstrated that having a normal pre-transplant DSE was associated with 80% survival after 3 years of follow-up. However, having any evidence of ischemia on pre-transplant DSE is associated with double the risk of death over the first 3 years post-transplant.42 Moreover, having a large area of ischemia (>25% of myocardial segments) was associated with an even higher death rate. Fixed wall motion abnormalities, indicating a scar, were associated with a higher death rate than a normal DSE, but not as high as patients with any evidence of ischemia.42 Furthermore, Tita et al showed that stress echocardiography had a NPV of 96% for major cardiac events occurring in the first year post transplantation, and that patients with an abnormal stress echo were seven times more likely to suffer from an event than those who had a normal test.43 In a meta-analysis of 12 studies (8 DSE and 4 SPECT-MPI studies) addressing prognostic value of noninvasive testing in kidney transplant recipients, Rabbat et al showed similar findings of increased risk for major adverse cardiac events in kidney transplant patients with abnormal stress tests.44 They showed that any abnormal stress test is associated with almost three times the risk of cardiac death (Table 2). They also showed that patients with a reversible abnormality on DSE or MPI (ischemia) were almost four times more likely to suffer from cardiac death or MI with even higher risks in the diabetic subpopulations.44 In summary, both DSE and SPECT-MPI provide comparably valuable short- and long-term prognostic data in kidney transplant recipients.3
A small number of outcome studies on coronary CT angiography in ESRD patients have shown that this imaging modality may be a reasonable alternative to other noninvasive techniques for prognostication. de Bie et al showed that not only was CT angiography interpretable in most patients, but that those with no significant CAD were less likely to have an incidence of cardiovascular events at 2-year follow-up.45 Furthermore, Shantouf et al showed that ESRD patients with a coronary artery calcium score of >400 had a significantly higher adjusted risk of death than those with a score of 0.46
Repeat Noninvasive Testing Awating Kidney Tranplantation
Given the prolonged wait time for a donor kidney (median 2.6 years), it is plausible that CAD severity may progress while awaiting a kidney transplant after the initial cardiovascular evaluation. This concern is particularly relevant among patients with multiple CAD risk factors. In a prospective study of 152 subjects, Rakhit et al showed that approximately 22% of ESRD patients developed new wall motion abnormalities on DSE during a 1.8-year follow-up period.47 However, an observational study of patients on the kidney transplant wait list in British Columbia (Canada) showed that periodic cardiac stress testing may be unnecessary, as there was no difference in the observed cardiovascular event rates between patients tested routinely vs. those tested only on the basis of symptoms.48 Clinical trials and outcome studies are needed to address the value of repeat testing awaiting kidney transplantation, as the wait time for organs is expected to continue to increase. In view of the paucity of data, the AHA/ACC consensus statement indicated that the usefulness of periodically screening for myocardial ischemia in asymptomatic kidney transplantation candidates while on the transplant waiting list to reduce the risk of major adverse cardiac events is uncertain (Class IIb—level of evidence C).
Conclusions and Recommendations
ESRD patients undergoing kidney transplant evaluation have a high burden of cardiovascular disease. Identifying patients at risk is paramount as cardiovascular disease remains the leading cause of mortality in patients pre- and post-transplant. In light of the recent AHA/ACC consensus, further cardiac evaluation should be performed in symptomatic patients being considered for kidney transplant. Noninvasive stress testing may be considered in asymptomatic patients based on the presence of three or more risk factors [diabetes mellitus, prior cardiovascular disease, >1 year on dialysis, left ventricular hypertrophy, age >60 years, smoking, hypertension, and dyslipidemia], regardless of functional status. Although they have only modest diagnostic accuracy, an abnormal SPECT-MPI or DSE has modest excellent prognostic NPV. On the other hand, an abnormal SPECT-MPI or DSE predicts increased peri-operative and long-term event rates. Therefore, the selection of either DSE or MPI should be based on the local expertise of the transplant center. Cardiac CT angiography is often interpretable in well-selected patients with ESRD, as coronary calcifications do not seem to be a major impediment to image interpretation. Cardiac CT angiography has an excellent diagnostic sensitivity and prognostic NPV; thus it is a reasonable diagnostic option, particularly for young patients on dialysis for a short period of time. The value of repeat testing in patients awaiting kidney transplantation is uncertain.
Areas for Future Research
Additional research is needed to better identify candidates for CAD surveillance prior to listing and to examine the value of repeat CAD surveillance while awaiting kidney transplantation. More importantly, outcome research and interventional trials are needed to define the impact of preoperative coronary revascularization in asymptomatic kidney transplant candidates on the perioperative and long-term outcomes after transplant.
Liver Transplant
There are currently 17,000 patients on the liver transplant waiting list. Although the number of donors has remained constant, the number of liver transplant candidates in the United States continues to rise annually, leading to longer pre-transplant wait time and older recipient age.49 Given the scarcity of donors and the significant cost involved, selecting patients with acceptable short- and long-term cardiovascular risk profiles is paramount. Cardiovascular evaluation in orthotopic liver transplant candidates is crucial, as cardiovascular death remains one of the leading causes of mortality in the first year post-transplant, second only to sepsis.50 Because of their baseline poor functional status, a significant proportion of patients with end-stage liver disease (ESLD) remain asymptomatic despite significant CAD, making the assessment and management particularly challenging.51
Coronary Artery Disease Burden
It was previously believed that ESLD patients were at lower CAD risk than the general population, as these patients tend to have low cholesterol levels, low blood pressure due to low systemic vascular resistance, and higher levels of circulating estrogens; all thought to be protective against cardiovascular disease.50 However, over the past two decades, studies have shown that the prevalence of CAD in this population can be as high as 27%, well above the general population.52 Proposed hypotheses for high CAD burden include the chronic inflammatory state associated with long-standing liver disease and high prevalence of smoking among patients with certain etiologies of ESLD.53 With improving survival of patients with ESLD, advanced age has also become a significant risk factor.54 Moreover, as non-alcoholic fatty liver disease has been an increasingly common cause of liver cirrhosis, diabetes mellitus, and metabolic syndrome have emerged as important CAD risk factors in ESLD.52,55
The impact of CAD on the outcome of liver transplant patients is significant. Plotkin et al reported 81% morbidity and 50% mortality among patients with CAD who underwent liver transplantation.56 This series, however, was limited by the lack of a control group. This limitation was addressed by Diedrich et al who evaluated 42 patients with known CAD who were age and gender matched to 42 controls without CAD. They reported nearly four times higher cumulative mortality and morbidity in the coronary disease group at both 1 and 3 years (Figure 3).57
Cumulative morbidity and mortality in liver transplant recipients based on coronary artery disease status. Morbidity is defined as new arrhythmias, myocardial infarction, or cardiac intervention. Adapted from Diedrich et al Transplant Proc 2008;40:3554-7, with permission.57
Cardiovascular morbidity and mortality has been attributed to the profound hemodynamic burden of liver transplantation, such as intra-operative hemorrhage, aggressive fluid and blood replacement, decreased pre-load secondary to inferior vena cava clamping, and electrolyte or acid-base disturbances.58 In addition, restoration of peripheral vascular resistance after liver transplantation causes a sudden postoperative increase in afterload, leading to acute ventricular failure in vulnerable patients. This is further exacerbated by reduced myocardial contractility from graft-produced cytokines, such as TNF-alpha.59 Additionally, immunosuppressants, such as calcineurin inhibitors, often cause a hypertensive response, further increasing afterload.59,60 The need therefore arises for careful evaluation of these patients prior to liver transplantation, addressing the perioperative and long-term cardiovascular risks.
Coronary Disease Surveillance
Given the considerable prevalence of CAD and its impact on the outcome of liver transplant recipients, there is a general agreement that CAD surveillance in “high risk” ESLD patients prior to transplantation is reasonable, irrespective of symptoms, and functional status.3,61 Nonetheless, there is less certainty regarding the definition of “high risk”. The AHA/ACC consensus statement indicates that noninvasive stress testing may be considered in liver transplant candidates with no active cardiac conditions on the basis of the presence of multiple CAD risk factors regardless of functional status. The statement defined the relevant risk factors in liver transplant candidates as diabetes mellitus, prior cardiovascular disease, left ventricular hypertrophy, age >60 years, smoking, hypertension, and dyslipidemia.3 The statement suggested that it would be “reasonable” to consider patients who have ≥3 risk factors as “high risk”. They left the choice of noninvasive testing to the local expertise. The panel gave this recommendation class IIb—level of evidence C (expert opinion), which underscores the lack of high quality data.3
CAD surveillance can be challenging in patients with ESLD. Due to excessive bleeding risk and frequent renal dysfunction, routine coronary angiography is not a feasible strategy.3 Since many patients tend to have poor functional capacity, exercise stress testing is often not an option.51 Thus, the majority of the patients undergo pharmacologic stress testing, which may be challenging as well. With dobutamine stress, ESLD patients are frequently unable to achieve their target heart rate due to autonomic dysfunction.62 In a study by Williams et al, 34 out of 60 (57%) patients who received dobutamine stress echo prior to liver transplant were unable to achieve their target heart rate despite maximum doses of dobutamine and atropine.62 On the other hand, hypotension among ESLD patients is a frequent limitation to using vasodilator stress with radionuclide MPI. Furthermore, patients with ESLD are maximally vasodilated at baseline, which may theoretically diminish the sensitivity of vasodilator stress MPI.63 Thus, some experts suggested that DSE may be preferred in this population.61 However, this recommendation is not supported by clinical data.
Vasodilator and catecholamine pharmacologic stress agents are believed to be safe in patients with ESLD. Although regadenoson, an A2A selective vasodilator stress agent, is partially excreted by the liver through the biliary tract, it has been shown to be safe and well tolerated among patients with ESLD.64
Diagnostic Accuracy of Noninvasive Testing
Myocardial Perfusion Imaging
Three studies have reported on the accuracy of SPECT-MPI as compared to invasive coronary angiography (Table 3). Figure 4 illustrates examples of normal (A) and abnormal (B) SPECT-MPI from liver transplant candidates. Davidson et al retrospectively analyzed 83 consecutive ESLD patients without known CAD who underwent dipyridamole- or adenosine-stress, dual-isotope SPECT-MPI followed by coronary angiography, irrespective of MPI findings. Abnormal MPI was defined as any perfusion abnormality regardless of reversibility. Severe CAD was defined as ≥70% diameter stenosis in one or more coronary segments.63 The sensitivity and specificity of SPECT-MPI were 37% and 63%, respectively; while the PPV and NPV were 22% and 77%, respectively. The prevalence of CAD in this population was 23% (Table 3).
SSS, Summed stress score; SDS, summed difference score; TID, transient ischemic dilation; LVEF, post-stress gated-SPECT left ventricular ejection fraction. (A) Rest-regadenoson stress 99mTc-tetrofosmine SPECT myocardial perfusion imaging of a 67-year-old man with end-stage liver disease who was evaluated for liver transplantation. He has a history of dyslipidemia and 50 pack-year tobacco use. As illustrated, his myocardial perfusion was normal; the inferior defect was likely caused by diaphragmatic attenuation as the gated-SPECT imaging was normal. (B) Rest-regadenoson stress 99mTc-tetrofosmine SPECT myocardial perfusion images of a 67-year-old woman who was evaluated for liver transplantation. She has a history of liver cirrhosis caused by autoimmune hepatitis. Note mild small reversible anteroseptal defect. Also note an apparent improved perfusion of the inferior wall on the post-stress images as compared to the rest images. The latter finding reflects stress-induced ischemia in the remainder of the myocardium, such that the inferior wall is being normalized relative to ischemic myocardium causing an apparent improvement in normalized count statistics in the inferior wall. Transient ischemic dilation further supports this interpretation. Coronary angiography showed left main coronary artery disease and severe stenosis in the proximal left anterior descending coronary.
More recently, Bhutani et al reported on 473 non-consecutive patients with ESLD who underwent adenosine or regadenoson 99mTc-tetrofosmin SPECT-MPI and were selected to undergo coronary angiography on the basis of MPI findings or the clinical risk profile.65 Patients with known CAD were excluded, and severe CAD was defined as ≥70% diameter stenosis. The prevalence of disease in this population was 10%. The sensitivity of SPECT-MPI was poor, while the specificity was acceptable, at 82% and 88% for adenosine and regadenoson stress, respectively (Table 3). The NPV for SPECT-MPI was 93% and 95% for adenosine and regadenoson stress, respectively, clearly inflated by low disease prevalence.65
In 2009, Aydinalp et al reported on 93 consecutive liver transplantation candidates who underwent dipyridamole-stress SPECT-MPI with thallium-201, followed by coronary angiography irrespective of risk factors, symptoms, or MPI findings. Abnormal MPI was defined as reversible perfusion defects and CAD was angiographically defined as ≥70% stenosis in one or more coronary segments or ≥50% stenosis corresponding with an abnormal MPI.66 The prevalence of CAD was only 6.5%. The sensitivity and specificity of SPECT-MPI were 100% and 61%, respectively, with PPV and NPV of 15% and 100%, respectively (Table 3).66 This remarkably higher sensitivity may simply be due to chance (small sample size and low disease prevalence), but it may also be due to the preferential use of exercise stress modality and radiotracer choice of 201Tl vs. 99mTc-based agents, as shown in Table 3. It has been postulated that vasodilator stress with regadenoson, adenosine, or dipyridamole may be a suboptimal stress modality in patients with ESLD since they are already in a vasodilated state at baseline.66 Thus, choosing stress modalities that do not primarily rely on coronary vasodilation, such as exercise or dobutamine stress, may be advantageous in this patient population.61 By the same token, radiotracers with higher extraction fraction and better linearity, such as 201Tl, may also be theoretically advantageous in demonstrating disparities in radioisotope uptake at high coronary flow states.67 It has been established that 201Tl is slightly more sensitive for reversible perfusion abnormalities than 99mTc-tetrofosmin.67 Although this advantage is clinically negligible in the general population, it may be significant in the vasodilated state of ESLD, resulting in superior sensitivity. Nonetheless, the potential impact of stress modality and radioisotope choice on the diagnostic performance of SPECT-MPI in this population remains speculative.
In addition, poor sensitivity of SPECT-MPI in this population may be attributed to dated technology used in the published studies, discordance between functional and anatomic significance of coronary lesions, and inferiority of this technique in asymptomatic individuals. However, it has been shown in other populations that SPECT-MPI is more predictive of patient outcome than coronary angiography, irrespective of diagnostic accuracy.28
PET perfusion imaging is generally better suited to overcome many of the limitations of SPECT-MPI, providing higher overall diagnostic accuracy.29 Nonetheless, PET imaging has not been specifically studied in liver transplant candidates.
Stress Echocardiography
As with SPECT-MPI, the diagnostic accuracy data with DSE as compared to coronary angiography has been conflicting. An earlier small study by Plotkin et al reported 100% sensitivity and specificity for DSE in detecting CAD, defined as coronary stenosis ≥70%.68 Another study by Donovan et al reported a 75% sensitivity for coronary stenosis ≥50%.69 Subsequent larger studies by Harenstein et al70 and Patel et al71 reported a substantially lower sensitivity and reasonable specificity (Table 3). The findings of the latter two studies were corroborated by Nguyen et al who analyzed pooled data of 110 patients from four studies. These authors reported pooled sensitivity, specificity, PPV, and NPV of 32%, 78%, 37%, and 75%, respectively.72
In reviewing the diagnostic accuracy data of SPECT-MPI and DSE, poor sensitivity and PPV with acceptable specificity and NPV seem to be the common theme, thus supporting the AHA/ACC consensus statement in basing the choice of noninvasive testing on the local expertise.3
Cardiac Computed Tomography
Coronary calcium score may help in identifying patients at risk for CAD. In a cohort of 85 liver transplant candidates, Kemmer et al demonstrated that only patients with coronary calcium scores ≥400 were found to have obstructive CAD, defined as ≥50% stenosis in ≥1 coronary artery.73 Furthermore, a few studies have recently demonstrated the feasibility and potential role of coronary CT angiography in the evaluation of liver transplant candidates.73-78 Limitations of coronary CT angiography include nephrotoxicity in this population with a high prevalence of renal dysfunction and some concern for hypotension with nitroglycerin and β-blocker use.79
Cardiac Magnetic Resonance Imaging
In an observational study of 51 liver transplant candidates, Reddy et al explored the feasibility of a comprehensive “one-stop shop” evaluation of cardiac function, structure, coronary disease, and viability along with thoracoabdominal vasculature and liver anatomy using magnetic resonance imaging.80 Although promising, the wide scale applicability of this technique is questionable. The diagnostic accuracy and outcome data of cardiac magnetic resonance in this population are lacking.80 Gadolinium related nephrotoxicity is also a concern.
Although the current data and AHA/ACC consensus statement do not identify a clear advantage for any noninvasive modality, certain techniques may be better suited for particular patient subsets. For example, patients with low baseline heart rate due to autonomic dysfunction or β-blockade (for the prevention of variceal bleeds) may benefit more from a cardiac CT or MPI rather than DSE, while among individuals with concomitant renal dysfunction MPI or DSE may be preferred over cardiac CT.
The Prognostic Value of Noninvasive Cardiac Imaging
The Prognostic Value of SPECT Myocardial Perfusion Imaging
Very few studies have evaluated the prognostic value of SPECT-MPI in liver transplant recipients. Zoghbi et al followed 87 patients who underwent adenosine (n = 24), dipyridamole (n = 51), or exercise (n = 12) 99mTc-sestamibi SPECT-MPI studies for an average of 21 months and found that a normal MPI had 99% and 96% NPV for peri-operative and late cardiac events, respectively.81 This suggests that the prognostic value of normal MPI remains excellent irrespective of angiographic coronary disease.
Additionally, Bradley et al retrospectively reviewed 291 patients who underwent liver transplantation after a SPECT-MPI and were followed for a median of 25 months.82 Among the 273 patients with low risk MPI (normal perfusion or a small perfusion defect), they reported a 2% rate of cardiac events (non-fatal myocardial infarction) within 30 days from transplantation, i.e., a prognostic NPV of 98%. Given the small sample size, little could be learned from this study regarding the outcome of patients with intermediate or high risk scans (n = 18).82 Both studies discussed above were limited by small sample size, retrospective design, and being derived from a single center experience.
The Prognostic Value of Stress Echocardiography
A few outcomes studies evaluated the use of DSE in predicting adverse events in liver transplant recipients. Safadi et al evaluated 356 patients who underwent DSE prior to liver transplantation and reported a NPV of 89% for death or nonfatal MI within 30 days, whereas the PPV was only 27%.83 Similarly, Williams et al evaluated a subset of 61 patients who had DSE prior to liver transplantation and found that, after excluding nondiagnostic studies, the NPV for adverse cardiac events was 80%. This investigation was limited by the fact that there were only two abnormal studies and 34 patients were unable to achieve their target heart rate.62 On the other hand, a study of 73 liver transplant recipients by Findlay et al found no correlation between pre-operative DSE results and post-transplant myocardial injury, defined by troponin elevation.84
Tsutsui et al sought to enhance the sensitivity of DSE by evaluating myocardial perfusion using micro-bubble ultrasound contrast, such as Optison® or Definity®. Among 85 patients who underwent liver transplantation, four had abnormal myocardial perfusion by echo contrast, 50% of whom (2/4) had perioperative adverse events vs. only 2% (2/81) among patients with normal echo contrast perfusion, thus suggesting a possible value of this imaging technique.85
Umphrey et al evaluated the outcome of 157 patients who underwent DSE prior to liver transplantation and found an increased rate of adverse cardiovascular events in patients who were unable to achieve their target heart rate (22% vs. 6%, P = .01) and those with low (<16,333) peak rate-pressure product (17% vs. 5%, P = .02).86 Although there have been no other confirmatory studies, this investigation suggests that additional testing may be warranted in patients unable to achieve their target heart rate during DSE.
The Prognostic Value of Cardiac Computed Tomography
A study by Cassagneau et al evaluated 52 patients who underwent coronary CT angiography prior to liver transplantation. A negative scan was defined as normal coronary CT angiography or non-obstructive disease. They reported a NPV of 95% for major cardiac events and 100% for major coronary events. However, the PPV was only 17% for both outcomes.78
Conclusions and Recommendations
CAD is prevalent among liver transplant candidates and has an important impact on perioperative and long-term outcome; thus, cardiac evaluation should be performed in symptomatic patients being considered for liver transplantation. In light of the recent AHA/ACC consensus statement, noninvasive stress testing may be considered in asymptomatic patients based on the presence of three or more risk factors [diabetes mellitus, prior cardiovascular disease, left ventricular hypertrophy, age >60 years, smoking, hypertension, and dyslipidemia] regardless of functional status. All published reports addressing the diagnostic and prognostic value of noninvasive cardiovascular imaging in this population are limited by small sample size and by their uncontrolled, retrospective, single-center design. Both DSE and SPECT have poor sensitivity, reasonable specificity, but good NPV. Irrespective of diagnostic accuracy, a normal DSE or SPECT-MPI is associated with low perioperative and long-term risk. However, the risk associated with abnormal MPI or DSE is less certain. Therefore, the selection of stress test modality should be based on the local expertise of the transplant center. Coronary CT angiography and coronary calcium score may have a role in the assessment of liver transplant candidates, but requires additional research.
Areas for Future Research
Prospective multicenter studies are needed to address deficiencies in the currently available data. Additional studies addressing the role of cardiac PET, cardiac CT, and myocardial perfusion echocardiography are needed. Well-designed prospective studies evaluating the impact of coronary revascularization on patient outcomes after liver transplantation are also needed.
References
Fleisher LA, Beckman JA, Brown KA, Calkins H, Chaikof EL, Fleischmann KE, et al. ACC/AHA 2007 guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery: Executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery) Developed in Collaboration With the American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Rhythm Society, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, and Society for Vascular Surgery. J Am Coll Cardiol 2007;50:1707-32.
Hendel RC, Berman DS, Di Carli MF, Heidenreich PA, Henkin RE, Pellikka PA, et al. ACCF/ASNC/ACR/AHA/ASE/SCCT/SCMR/SNM 2009 appropriate use criteria for cardiac radionuclide imaging: A report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, the American Society of Nuclear Cardiology, the American College of Radiology, the American Heart Association, the American Society of Echocardiography, the Society of Cardiovascular Computed Tomography, the Society for Cardiovascular Magnetic Resonance, and the Society of Nuclear Medicine. Endorsed by the American College of Emergency Physicians. J Am Coll Cardiol 2009;53:2201-29.
Lentine KL, Costa SP, Weir MR, Robb JF, Fleisher LA, Kasiske BL, et al. Cardiac disease evaluation and management among kidney and liver transplantation candidates: A scientific statement from the American Heart Association and the American College of Cardiology Foundation. J Am Coll Cardiol 2012;60:434-80.
U.S. Renal Data System. USRDS 2013 annual data report: Atlas of chronic kidney disease and end-stage renal disease in the United States. Bethesda, MD: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Disease; 2013. p. 2.
Port FK, Wolfe RA, Mauger EA, Berling DP, Jiang K. COmparison of survival probabilities for dialysis patients vs cadaveric renal transplant recipients. J Am Med Assoc 1993;270:1339-43.
Arend SM, Mallat MJ, Westendorp RJ, van der Woude FJ, van Es LA. Patient survival after renal transplantation; more than 25 years follow-up. Nephrol Dial Transplant 1997;12:1672-9.
Kumar N, Baker CS, Chan K, Duncan N, Malik I, Frankel A, et al. Cardiac survival after pre-emptive coronary angiography in transplant patients and those awaiting transplantation. Clin J Am Soc Nephrol 2011;6:1912-9.
Gowdak LH, de Paula FJ, Cesar LA, Martinez Filho EE, Ianhez LE, Krieger EM, et al. Screening for significant coronary artery disease in high-risk renal transplant candidates. Coron Artery Dis 2007;18:553-8.
Ojo AO, Hanson JA, Wolfe RA, Leichtman AB, Agodoa LY, Port FK. Long-term survival in renal transplant recipients with graft function. Kidney Int 2000;57:307-13.
Aakhus S, Dahl K, Wideroe TE. Cardiovascular disease in stable renal transplant patients in Norway: Morbidity and mortality during a 5-yr follow-up. Clin Transplant 2004;18:596-604.
Ducloux D, Kazory A, Chalopin JM. Predicting coronary heart disease in renal transplant recipients: A prospective study. Kidney Int 2004;66:441-7.
Jardine AG, Fellstrom B, Logan JO, Cole E, Nyberg G, Gronhagen-Riska C, et al. Cardiovascular risk and renal transplantation: Post hoc analyses of the Assessment of Lescol in Renal Transplantation (ALERT) Study. Am J Kidney Dis 2005;46:529-36.
Rigatto C, Foley R, Jeffery J, Negrijn C, Tribula C, Parfrey P. Electrocardiographic left ventricular hypertrophy in renal transplant recipients: Prognostic value and impact of blood pressure and anemia. J Am Soc Nephrol 2003;14:462-8.
Kasiske BL, Cangro CB, Hariharan S, Hricik DE, Kerman RH, Roth D, et al. The evaluation of renal transplantation candidates: Clinical practice guidelines. Am J Transplant 2001;1:3-95.
Abbud-Filho M, Adams PL, Alberu J, Cardella C, Chapman J, Cochat P, et al. A report of the Lisbon Conference on the care of the kidney transplant recipient. Transplantation 2007;83:S1-22.
K/DOQI Workgroup. K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis 2005;45:S1-153.
De Lima JJ, Sabbaga E, Vieira ML, de Paula FJ, Ianhez LE, Krieger EM, et al. Coronary angiography is the best predictor of events in renal transplant candidates compared with noninvasive testing. Hypertension 2003;42:263-8.
Hage FG, Smalheiser S, Zoghbi GJ, Perry GJ, Deierhoi M, Warnock D, et al. Predictors of survival in patients with end-stage renal disease evaluated for kidney transplantation. Am J Cardiol 2007;100:1020-5.
Jeloka TK, Ross H, Smith R, Huang M, Fenton S, Cattran D, et al. Renal transplant outcome in high-cardiovascular risk recipients. Clin Transplant 2007;21:609-14.
Aljaroudi W, Hermann D, Hage F, Heo J, Iskandrian AE. Safety of regadenoson in patients with end-stage renal disease. Am J Cardiol 2010;105:133-5.
Doukky R, Rangel MO, Wassouf M, Dick R, Alqaid A, Morales Demori R. The safety and tolerability of regadenoson in patients with end-stage renal disease: The first prospective evaluation. J Nucl Cardiol 2013;20:205-13.
Doukky R, Morales Demori R, Jain S, Kiriakos R, Mwansa V, Calvin JE. Attenuation of the side effect profile of regadenoson: A randomized double-blinded placebo-controlled study with aminophylline in patients undergoing myocardial perfusion imaging. “The ASSUAGE trial”. J Nucl Cardiol 2012;19:448-57.
Doukky R, Rangel MO, Dick R, Wassouf M, Alqaid A, Margeta B. Attenuation of the side effect profile of regadenoson: A randomized double-blind placebo-controlled study with aminophylline in patients undergoing myocardial perfusion imaging and have severe chronic kidney disease-the ASSUAGE-CKD trial. Int J Cardiovasc Imaging 2013;29:1029-37.
Wang LW, Fahim MA, Hayen A, Mitchell RL, Baines L, Lord S et al. Cardiac testing for coronary artery disease in potential kidney transplant recipients. Cochrane Database Syst Rev 2011;12:Cd008691.
Greenwood JP, Maredia N, Younger JF, Brown JM, Nixon J, Everett CC, et al. Cardiovascular magnetic resonance and single-photon emission computed tomography for diagnosis of coronary heart disease (CE-MARC): A prospective trial. Lancet 2012;379:453-60.
Rochitte CE, George RT, Chen MY, Arbab-Zadeh A, Dewey M, Miller JM, et al. Computed tomography angiography and perfusion to assess coronary artery stenosis causing perfusion defects by single photon emission computed tomography: The CORE320 study. Eur Heart J 2014;35:1120-30.
Tonino PA, De Bruyne B, Pijls NH, Siebert U, Ikeno F, van’ t Veer M, et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med 2009;360:213-24.
Iskandrian AS, Chae SC, Heo J, Stanberry CD, Wasserleben V, Cave V. Independent and incremental prognostic value of exercise single-photon emission computed tomographic (SPECT) thallium imaging in coronary artery disease. J Am Coll Cardiol 1993;22:665-70.
McArdle BA, Dowsley TF, deKemp RA, Wells GA, Beanlands RS. Does rubidium-82 PET have superior accuracy to SPECT perfusion imaging for the diagnosis of obstructive coronary disease? A systematic review and meta-analysis. J Am Coll Cardiol 2012;60:1828-37.
McCullough PA. Coronary artery disease. Clin J Am Soc Nephrol 2007;2:611-6.
Mao J, Karthikeyan V, Poopat C, Song T, Pantelic M, Chattahi J, et al. Coronary computed tomography angiography in dialysis patients undergoing pre-renal transplantation cardiac risk stratification. Cardiol J 2010;17:349-61.
Jug B, Kadakia J, Gupta M, Papazian J, Derakhshani A, Koplik S, et al. Coronary calcifications and plaque characteristics in patients with end-stage renal disease: A computed tomographic study. Coron Artery Dis 2013;24:501-8.
Sharples EJ, Pereira D, Summers S, Cunningham J, Rubens M, Goldsmith D, et al. Coronary artery calcification measured with electron-beam computerized tomography correlates poorly with coronary artery angiography in dialysis patients. Am J Kidney Dis 2004;43:313-9.
Rosario MA, Lima JJ, Parga JR, Avila LF, Gowdak LH, Lemos PA et al. Coronary calcium score as predictor of stenosis and events in pretransplant renal chronic failure. Arquivos brasileiros de cardiologia 2010;94:236-43, 52-60, 9-47.
Park MY, Choi SJ, Kim JK, Hwang SD, Suh JON, Seo HS, et al. Use of multidetector computed tomography for evaluating coronary artery disease in patients undergoing dialysis. Nephrology 2011;16:285-9.
Robinson J, Tan AU, Wilensky RL, Matthai W, Munoz M, Rosas SE. Electron-beam computerized tomography correlates with coronary angiogram in chronic kidney disease patients. Am J Nephrol 2007;27:247-52.
Haydar AA, Covic A, Colhoun H, Rubens M, Goldsmith DJ. Coronary artery calcification and aortic pulse wave velocity in chronic kidney disease patients. Kidney Int 2004;65:1790-4.
Wong CF, Little MA, Vinjamuri S, Hammad A, Harper JM. Technetium myocardial perfusion scanning in prerenal transplant evaluation in the United Kingdom. Transplant Proc 2008;40:1324-8.
Patel AD, Abo-Auda WS, Davis JM, Zoghbi GJ, Deierhoi MH, Heo J, et al. Prognostic value of myocardial perfusion imaging in predicting outcome after renal transplantation. Am J Cardiol 2003;92:146-51.
Chew CG, Unger S, Shakib S. Value of myocardial perfusion imaging in renal transplant evaluation. Nephrology (Carlton, VIC). 2013;18:376-81.
Murthy VL, Naya M, Foster CR, Hainer J, Gaber M, Dorbala S, et al. Coronary vascular dysfunction and prognosis in patients with chronic kidney disease. JACC Cardiovasc Imaging 2012;5:1025-34.
Bergeron S, Hillis GS, Haugen EN, Oh JK, Bailey KR, Pellikka PA. Prognostic value of dobutamine stress echocardiography in patients with chronic kidney disease. Am Heart J 2007;153:385-91.
Tita C, Karthikeyan V, Stroe A, Jacobsen G, Ananthasubramaniam K. Stress echocardiography for risk stratification in patients with end-stage renal disease undergoing renal transplantation. J Am Soc Echocardiogr 2008;21:321-6.
Rabbat CG, Treleaven DJ, Russell JD, Ludwin D, Cook DJ. Prognostic value of myocardial perfusion studies in patients with end-stage renal disease assessed for kidney or kidney-pancreas transplantation: A meta-analysis. J Am Soc Nephrol 2003;14:431-9.
de Bie MK, Buiten MS, Gaasbeek A, Boogers MJ, Roos CJ, Schuijf JD, et al. CT coronary angiography is feasible for the assessment of coronary artery disease in chronic dialysis patients, despite high average calcium scores. PLoS ONE 2013;8:e67936.
Shantouf RS, Budoff MJ, Ahmadi N, Ghaffari A, Flores F, Gopal A, et al. Total and individual coronary artery calcium scores as independent predictors of mortality in hemodialysis patients. Am J Nephrol 2010;31:419-25.
Rakhit DJ, Marwick TH, Armstrong KA, Johnson DW, Leano R, Isbel NM. Effect of aggressive risk factor modification on cardiac events and myocardial ischaemia in patients with chronic kidney disease. Heart 2006;92:1402-8.
Gill JS, Ma I, Landsberg D, Johnson N, Levin A. Cardiovascular events and investigation in patients who are awaiting cadaveric kidney transplantation. J Am Soc Nephrol 2005;16:808-16.
Xia VW, Taniguchi M, Steadman RH. The changing face of patients presenting for liver transplantation. Curr Opin Organ Transplant 2008;13:280-4.
Ehtisham J, Altieri M, Salame E, Saloux E, Ollivier I, Hamon M. Coronary artery disease in orthotopic liver transplantation: Pretransplant assessment and management. Liver Transplant 2010;16:550-7.
Jacob M, Copley LP, Lewsey JD, Gimson A, Rela M, van der Meulen JH. Functional status of patients before liver transplantation as a predictor of posttransplant mortality. Transplantation 2005;80:52-7.
Carey WD, Dumot JA, Pimentel RR, Barnes DS, Hobbs RE, Henderson JM, et al. The prevalence of coronary artery disease in liver transplant candidates over age 50. Transplantation 1995;59:859-64.
Gk H. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med 2005;352:1685-95.
Appleton CPHT. Reducing coronary artery disease events in liver transplant patients: Moving toward identifying the vulnerable patient. Liver Transplant 2008;14:1691-3.
Kadayifci A, Tan V, Ursell PC, Merriman RB, Bass NM. Clinical and pathologic risk factors for atherosclerosis in cirrhosis: A comparison between NASH-related cirrhosis and cirrhosis due to other aetiologies. J Hepatol 2008;49:595-9.
Plotkin JS, Scott VL, Pinna A, Dobsch BP, De Wolf AM, Kang Y. Morbidity and mortality in patients with coronary artery disease undergoing orthotopic liver transplantation. Liver Transplant Surg 1996;2:426-30.
Diedrich DA, Findlay JY, Harrison BA, Rosen CB. Influence of coronary artery disease on outcomes after liver transplantation. Transplant Proc 2008;40:3554-7.
Raval Z, Harinstein ME, Skaro AI, Erdogan A, DeWolf AM, Shah SJ, et al. Cardiovascular risk assessment of the liver transplant candidate. J Am Coll Cardiol 2011;58:223-31.
Moller S, Henriksen JH. Cirrhotic cardiomyopathy: A pathophysiological review of circulatory dysfunction in liver disease. Heart 2002;87:9-15.
Moller S, Dumcke CW, Krag A. The heart and the liver. Exp Rev Gastroenterol Hepatol 2009;3:51-64.
Murray KF, Carithers RL Jr. AASLD practice guidelines: Evaluation of the patient for liver transplantation. Hepatology 2005;41:1407-32.
Williams K, Lewis JF, Davis G, Geiser EA. Dobutamine stress echocardiography in patients undergoing liver transplantation evaluation. Transplantation 2000;69:2354-6.
Davidson CJ, Gheorghiade M, Flaherty JD, Elliot MD, Reddy SP, Wang NC, et al. Predictive value of stress myocardial perfusion imaging in liver transplant candidates. Am J Cardiol 2002;89:359-60.
Aljaroudi W, Iqbal F, Koneru J, Bhambhvani P, Heo J, Iskandrian AE. Safety of regadenoson in patients with end-stage liver disease. J Nucl Cardiol 2011;18:90-5.
Bhutani S, Tobis J, Gevorgyan R, Sinha A, Suh W, Honda HM, et al. Accuracy of stress myocardial perfusion imaging to diagnose coronary artery disease in end stage liver disease patients. Am J Cardiol 2013;111:1057-61.
Aydinalp A, Bal U, Atar I, Ertan C, Aktas A, Yildirir A, et al. Value of stress myocardial perfusion scanning in diagnosis of severe coronary artery disease in liver transplantation candidates. Transplant Proc 2009;41:3757-60.
Shanoudy H, Raggi P, Beller GA, Soliman A, Ammermann EG, Kastner RJ, et al. Comparison of technetium-99m tetrofosmin and thallium-201 single-photon emission computed tomographic imaging for detection of myocardial perfusion defects in patients with coronary artery disease. J Am Coll Cardiol 1998;31:331-7.
Plotkin JS, Benitez RM, Kuo PC, Njoku MJ, Ridge LA, Lim JW, et al. Dobutamine stress echocardiography for preoperative cardiac risk stratification in patients undergoing orthotopic liver transplantation. Liver Transplant Surg 1998;4:253-7.
Donovan CL, Marcovitz PA, Punch JD, Bach DS, Brown KA, Lucey MR, et al. Two-dimensional and dobutamine stress echocardiography in the preoperative assessment of patients with end-stage liver disease prior to orthotopic liver transplantation. Transplantation 1996;61:1180-8.
Harinstein ME, Flaherty JD, Ansari AH, Robin J, Davidson CJ, Rossi JS, et al. Predictive value of dobutamine stress echocardiography for coronary artery disease detection in liver transplant candidates. Am J Transplant 2008;8:1523-8.
Patel S, Kiefer TL, Ahmed A, Ali ZA, Tremmel JA, Lee DP, et al. Comparison of the frequency of coronary artery disease in alcohol-related versus non-alcohol-related endstage liver disease. Am J Cardiol 2011;108:1552-5.
Nguyen P, Plotkin J, Fishbein TM, Laurin JM, Satoskar R, Shetty K, et al. Dobutamine stress echocardiography in patients undergoing orthotopic liver transplantation: A pooled analysis of accuracy, perioperative and long term cardiovascular prognosis. Int J Cardiovasc Imaging 2013;29:1741-8.
Kemmer N, Case J, Chandna S, Neff GW. The role of coronary calcium score in the risk assessment of liver transplant candidates. Transplant Proc 2014;46:230-3.
McAvoy NC, Kochar N, McKillop G, Newby DE, Hayes PC. Prevalence of coronary artery calcification in patients undergoing assessment for orthotopic liver transplantation. Liver Transplant 2008;14:1725-31.
Keeling AN, Flaherty JD, Davarpanah AH, Ambrosy A, Farrelly CT, Harinstein ME, et al. Coronary multidetector computed tomographic angiography to evaluate coronary artery disease in liver transplant candidates: Methods, feasibility and initial experience. J Cardiovasc Med (Hagerstown, MD) 2011;12:460-8.
Jodocy D, Abbrederis S, Graziadei IW, Vogel W, Pachinger O, Feuchtner GM, et al. Coronary computer tomographic angiography for preoperative risk stratification in patients undergoing liver transplantation. Eur J Radiol 2012;81:2260-4.
Chae WY, Hwang S, Yoon YI, Kang MC, Moon DB, Song GW, et al. Clinical value of preoperative coronary risk assessment by computed tomographic arteriography prior to adult living donor liver transplantation. Transplant Proc 2012;44:415-7.
Cassagneau P, Jacquier A, Giorgi R, Amabile N, Gaubert JY, Cohen F, et al. Prognostic value of preoperative coronary computed tomography angiography in patients treated by orthotopic liver transplantation. Eur J Gastroenterol Hepatol 2012;24:558-62.
Poulin MF, Chan EY, Ibrahim SA, Doukky R. Integrating coronary computed tomographic angiography in the evaluation of liver transplant candidates: A novel approach [Abstract]. J Nuc Cardiol 2014;21:802.
Reddy ST, Thai NL, Fakhri AA, Oliva J, Tom KB, Dishart MK, et al. Exploratory use of cardiovascular magnetic resonance imaging in liver transplantation: A one-stop shop for preoperative cardiohepatic evaluation. Transplantation 2013;96:827-33.
Zoghbi GJ, Patel AD, Ershadi RE, Heo J, Bynon JS, Iskandrian AE. Usefulness of preoperative stress perfusion imaging in predicting prognosis after liver transplantation. Am J Cardiol 2003;92:1066-71.
Bradley SM, Soine LA, Caldwell JH, Goldberg SL. Screening stress myocardial perfusion imaging and eligibility for liver transplantation. Am J Cardiol 2010;105:1010-3.
Safadi A, Homsi M, Maskoun W, Lane KA, Singh I, Sawada SG, et al. Perioperative risk predictors of cardiac outcomes in patients undergoing liver transplantation surgery. Circulation 2009;120:1189-94.
Findlay JY, Keegan MT, Pellikka PP, Rosen CB, Plevak DJ. Preoperative dobutamine stress echocardiography, intraoperative events, and intraoperative myocardial injury in liver transplantation. Transplant Proc 2005;37:2209-13.
Tsutsui JM, Mukherjee S, Elhendy A, Xie F, Lyden ER, O’Leary E, et al. Value of dobutamine stress myocardial contrast perfusion echocardiography in patients with advanced liver disease. Liver Transplant 2006;12:592-9.
Umphrey LG, Hurst RT, Eleid MF, Lee KS, Reuss CS, Hentz JG, et al. Preoperative dobutamine stress echocardiographic findings and subsequent short-term adverse cardiac events after orthotopic liver transplantation. Liver Transplant 2008;14:886-92.
Gowdak LH, de Paula FJ, De Oliveira AL, Arantes RL, Cesar LA, Ramires JA, et al. Non-invasive screening for coronary artery disease in renal transplant candidates with diabetes is influenced by gender. Sophia Antipolis: European Society of Cardiology; 2010.
Boudreau RJ, Strony JT, duCret RP, Kuni CC, Wang Y, Wilson RF, et al. Perfusion thallium imaging of type I diabetes patients with end stage renal disease: Comparison of oral and intravenous dipyridamole administration. Radiology 1990;175:103-5.
Worthley MI, Unger SA, Mathew TH, Russ GR, Horowitz JD. Usefulness of tachycardic-stress perfusion imaging to predict coronary artery disease in high-risk patients with chronic renal failure. Am J Cardiol 2003;92:1318-20.
Schmidt A, Stefenelli T, Schuster E, Mayer G. Informational contribution of noninvasive screening tests for coronary artery disease in patients on chronic renal replacement therapy. Am J Kidney Dis 2001;37:56-63.
Vandenberg BF, Rossen JD, Grover-McKay M, Shammas NW, Burns TL, Rezai K. Evaluation of diabetic patients for renal and pancreas transplantation: Noninvasive screening for coronary artery disease using radionuclide methods. Transplantation 1996;62:1230-5.
Dahan M, Viron BM, Faraggi M, Himbert DL, Lagallicier BJ, Kolta AM, et al. Diagnostic accuracy and prognostic value of combined dipyridamole-exercise thallium imaging in hemodialysis patients. Kidney Int 1998;54:255-62.
Sharma R, Mehta RL, Brecker SJ, Gaze DC, Gregson H, Streather CP, et al. The diagnostic and prognostic value of tissue Doppler imaging during dobutamine stress echocardiography in end-stage renal disease. Coron Artery Dis 2009;20:230-7.
Sharma R, Pellerin D, Gaze DC, Gregson H, Streather CP, Collinson PO, et al. Dobutamine stress echocardiography and the resting but not exercise electrocardiograph predict severe coronary artery disease in renal transplant candidates. Nephrol Dial Transplant 2005;20:2207-14.
Ferreira PA, de Lima VC, Campos Filho O, Gil MA, Cordovil A, Machado CV, et al. Feasibility, safety and accuracy of dobutamine/atropine stress echocardiography for the detection of coronary artery disease in renal transplant candidates. Arq Bras Cardiol 2007;88:45-51.
Cai Q, Serrano R, Kalyanasundaram A, Shirani J. A preoperative echocardiographic predictive model for assessment of cardiovascular outcome after renal transplantation. J Am Soc Echocardiogr 2010;23:560-6.
West JC, Napoliello DA, Costello JM, Nassef LA, Butcher RJ, Hartle JE, et al. Preoperative dobutamine stress echocardiography versus cardiac arteriography for risk assessment prior to renal transplantation. Transplant Int 2000;13:S27-30.
Herzog CA, Marwick TH, Pheley AM, White CW, Rao VK, Dick CD. Dobutamine stress echocardiography for the detection of significant coronary artery disease in renal transplant candidates. Am J Kidney Dis 1999;33:1080-90.
Garcia-Canton C, Culegras C, Hernandez-Briz MJ, Palomar R, Moreno A, et al. Dobutamine stress echocardiography and stress 99M-technetium methoxyisobutylisonitrile SPECT in the assessment of coronary artery disease in kidney transplant candidates [Abstract]. J Am Soc Nephrol 1998;9:708A.
Gang S, Dabhi M, Rajapurkar MM. Ischaemia imaging in type 2 diabetic kidney transplant candidates—is coronary angiography essential? Nephrol Dial Transplant 2007;22:2334-8.
Disclosures
Rami Doukky received research support from Astellas Pharma U.S. and served on the Advisory Board of Astellas Pharma U.S. Other authors have no conflicts.
Author information
Authors and Affiliations
Corresponding author
Additional information
See related editorial, doi:10.1007/s12350-014-0006-2.
Rights and permissions
About this article
Cite this article
Parikh, K., Appis, A. & Doukky, R. Cardiac imaging for the assessment of patients being evaluated for kidney or liver transplantation. J. Nucl. Cardiol. 22, 282–296 (2015). https://doi.org/10.1007/s12350-014-9997-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-014-9997-y