Abstract
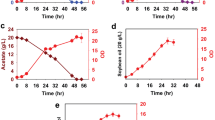
Dissecting the cellular metabolism of Cordyceps militaris is important for the efficient production of bioactive compounds of a target with medicinal and industrial applications. However, the metabolic functions during developmental stages in this fungus at a system level are still unexplored. In this study, we aimed to reveal the metabolic functions and regulation of C. militaris TBRC6039 relevant to its developmental stages, including mycelial growth (MY) and fruiting body (FB) stages through integrative transcriptome analysis. The transcriptome analysis showed that 9,256 genes of C. militaris were expressed in both stages. Of them, 1,877 genes, residing primarily in the cell division cycle and amino acid, carbohydrate, and lipid metabolisms, exhibited significant differences in transcript levels between the MY and FB stages. Through integration with genome-scale networks analysis, the unique reporter metabolites (e.g., α-D-glucose, β-D-glucose, D-galactose, triacylglycerol, and diacylglycerol) and key regulators (e.g., AtfA, Atf2, and Yap1 transcription factors) were identified in C. militaris when grown at FB stage, linking to up-regulation of the metabolic genes involved in galactose and polysaccharide metabolisms, as well as glycerolipid and glycerophospholipid biosynthesis. Moreover, the high cordycepin content is related to the upregulated genes in lipid metabolism during the FB stage. Our findings suggest that the transcriptional regulation of these metabolic pathways played a crucial role in specific developmental stages of C. militaris. This study serves for cultivation process improvement for overproduction of valuable metabolites in C. militaris through an emerging systems and synthetic biology approach.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Cui, J. D. (2015) Biotechnological production and applications of Cordyceps militaris, a valued traditional Chinese medicine. Crit. Rev. Biotechnol. 35: 475–484.
Elkhateeb, W. A., G. M. Daba, P. W. Thomas, and T.-C. Wen (2019) Medicinal mushrooms as a new source of natural therapeutic bioactive compounds. Egypt. Pharm. J. 18: 88–101.
Hur, H. (2008) Chemical ingredients of Cordyceps militaris. Mycobiology 36: 233–235.
Ashraf, S. A., A. E. O. Elkhalifa, A. J. Siddiqui, M. Patel, A. M. Awadelkareem, M. Snoussi, M. S. Ashraf, M. Adnan, and S. Hadi (2020) Cordycepin for health and wellbeing: a potent bioactive metabolite of an entomopathogenic Cordyceps medicinal fungus and its nutraceutical and therapeutic potential. Molecules 25: 2735.
Qin, P., Z. Wang, D. Lu, H. Kang, G. Li, R. Guo, Y. Zhao, R. Han, B. Ji, and Y. Zeng (2019) Neutral lipid content in lipid droplets: potential biomarker of cordycepin accumulation in cordycepin-producing fungi. Molecules 24: 3363.
Suparmin, A., T. Kato, H. Dohra, and E. Y. Park (2017) Insight into cordycepin biosynthesis of Cordyceps militaris: comparison between a liquid surface culture and a submerged culture through transcriptomic analysis. PLoS One 12: e0187052.
Chitcharoen, S. (2017) Metabolic footprinting of Cordyceps militaris culture using different carbon sources. M.S. thesis, Kasetsart University, Thailand.
Mao, X.-B., T. Eksriwong, S. Chauvatcharin, and J.-J. Zhong (2005) Optimization of carbon source and carbon/nitrogen ratio for cordycepin production by submerged cultivation of medicinal mushroom Cordyceps militaris. Process Biochem. 40: 1667–1672.
Raethong, N., H. Wang, J. Nielsen, and W. Vongsangnak (2020) Optimizing cultivation of Cordyceps militaris for fast growth and cordycepin overproduction using rational design of synthetic media. Comput. Struct. Biotechnol. J. 18: 1–8.
Wongsa, B., N. Raethong, P. Chumnanpuen, J. Wong-ekkabut, K. Laoteng, and W. Vongsangnak (2020) Alternative metabolic routes in channeling xylose to cordycepin production of Cordyceps militaris identified by comparative transcriptome analysis. Genomics 112: 629–636.
Yang, T., M. Guo, H. Yang, S. Guo, and C. Dong (2016) The blue-light receptor CmWC-1 mediates fruit body development and secondary metabolism in Cordyceps militaris. Appl. Microbiol. Biotechnol. 100: 743–755.
Oh, J., D.-H. Yoon, B. Shrestha, H.-K. Choi, and G.-H. Sung (2019) Metabolomic profiling reveals enrichment of cordycepin in senescence process of Cordyceps militaris fruit bodies. J. Microbiol. 57: 54–63.
Chen, Y., Y. Wu, L. Liu, J. Feng, T. Zhang, S. Qin, X. Zhao, C. Wang, D. Li, W. Han, M. Shao, P. Zhao, J. Xue, X. Liu, H. Li, E. Zhao, W. Zhao, X. Guo, Y. Jin, Y. Cao, L. Cui, Z. Zhou, Q. Xia, Z. Rao, and Y. Zhang (2019) Study of the whole genome, methylome and transcriptome of Cordyceps militaris. Sci. Rep. 9: 898.
Wang, F., Q. Liu, J. Zhang, K. Liu, K. Li, G. Liu, and C. Dong (2018) Comparative transcriptome analysis between a spontaneous albino mutant and its sibling strain of Cordyceps militaris in response to light stress. Front. Microbiol. 9: 1237.
Zheng, P., Y. Xia, G. Xiao, C. Xiong, X. Hu, S. Zhang, H. Zheng, Y. Huang, Y. Zhou, S. Wang, G.-P. Zhao, X. Liu, R. J. St Leger, and C. Wang (2011) Genome sequence of the insect pathogenic fungus Cordyceps militaris, a valued traditional Chinese medicine. Genome Biol. 12: R116.
Yin, Y., G. Yu, Y. Chen, S. Jiang, M. Wang, Y. Jin, X. Lan, Y. Liang, and H. Sun (2012) Genome-wide transcriptome and proteome analysis on different developmental stages of Cordyceps militaris. PLoS One 7: e51853.
Li, X., F. Wang, Q. Liu, Q. Li, Z. Qian, X. Zhang, K. Li, W. Li, and C. Dong (2019) Developmental transcriptomics of Chinese cordyceps reveals gene regulatory network and expression profiles of sexual development-related genes. BMC Genomics 20: 337.
Tong, X., H. Zhang, F. Wang, Z. Xue, J. Cao, C. Peng, and J. Guo (2020) Comparative transcriptome analysis revealed genes involved in the fruiting body development of Ophiocordyceps sinensis. PeerJ 8: e8379.
In-on, A., R. Thananusak, M. Ruengjitchatchawalya, W. Vongsangnak, and T. Laomettachit (2022) Construction of light-responsive gene regulatory network for growth, development and secondary metabolite production in Cordyceps militaris. Biology (Basel) 11: 71.
Thananusak, R., K. Laoteng, N. Raethong, Y. Zhang, and W. Vongsangnak (2020) Metabolic responses of carotenoid and cordycepin biosynthetic pathways in Cordyceps militaris under light-programming exposure through genome-wide transcriptional analysis. Biology (Basel) 9: 242.
Augustine, A., J. Imelda, and R. Paul Raj (2006) Biomass estimation of Aspergillus niger S14 a mangrove fungal isolate and A. oryzae NCIM 1212 in solid-state fermentation. J. Mar. Biol. Assoc. India 48: 139–146.
Jangbua, P., K. Laoteng, P. Kitsubun, M. Nopharatana, and A. Tongta (2009) Gamma-linolenic acid production of Mucor rouxii by solid-state fermentation using agricultural by-products. Lett. Appl. Microbiol. 49: 91–97.
Sakurai, Y., T. H. Lee, and H. Shiota (1977) On the convenient method for glucosamine estimation in koji. Agric. Biol. Chem. 41: 619–624.
Yang, T., J. Sun, T. Lian, W. Wang, and C. H. Dong (2014) Process optimization for extraction of carotenoids from medicinal caterpillar fungus, Cordyceps militaris (Ascomycetes). Int. J. Med. Mushrooms 16: 125–135.
Andrews, S., FastQC: a quality control tool for high throughput sequence data. http://www.bioinformatics.babraham.ac.uk/projects/fastqc/.
Bushnell, B. (2014) BBMap: a fast, accurate, splice-aware aligner. Proceedings of 9th Annual Genomics of Energy & Environment Meeting. March 17–20. Walnut Creek, CA, USA.
Li, H. and R. Durbin (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25: 1754–1760.
Li, H., B. Handsaker, A. Wysoker, T. Fennell, J. Ruan, N. Homer, G. Marth, G. Abecasis, R. Durbin, and 1000 Genome Project Data Processing Subgroup (2009) The sequence alignment/map format and SAMtools. Bioinformatics 25: 2078–2079.
Trapnell, C., A. Roberts, L. Goff, G. Pertea, D. Kim, D. R. Kelley, H. Pimentel, S. L. Salzberg, J. L. Rinn, and L. Pachter (2012) Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat. Protoc. 7: 562–578. (Erratum published 2014, Nat. Protoc. 9: 2513)
Grigoriev, I. V., R. Nikitin, S. Haridas, A. Kuo, R. Ohm, R. Otillar, R. Riley, A. Salamov, X. Zhao, F. Korzeniewski, T. Smirnova, H. Nordberg, I. Dubchak, and I. Shabalov (2014) MycoCosm portal: gearing up for 1000 fungal genomes. Nucleic Acids Res. 42(Database issue): D699–D704.
Love, M. I., W. Huber, and S. Anders (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15: 550.
Kolde, R. (2019) pheatmap: pretty heatmaps. R package version 1.0.12.
Patil, K. R. and J. Nielsen (2005) Uncovering transcriptional regulation of metabolism by using metabolic network topology. Proc. Natl. Acad. Sci. U. S. A. 102: 2685–2689.
Väremo, L., J. Nielsen, and I. Nookaew (2013) Enriching the gene set analysis of genome-wide data by incorporating directionality of gene expression and combining statistical hypotheses and methods. Nucleic Acids Res. 41: 4378–4391.
Xia, Y., F. Luo, Y. Shang, P. Chen, Y. Lu, and C. Wang (2017) Fungal cordycepin biosynthesis is coupled with the production of the safeguard molecule pentostatin. Cell Chem. Biol. 24: 1479–1489.e4.
Zhang, J., T. Jian, Y. Zhang, G. Zhang, and J. Ling (2021) Dynamic content changes of cordycepin and adenosine and transcriptome in Cordyceps kyushuensis Kob at different fermentation stages. Bioprocess Biosyst. Eng. 44: 1793–1803.
Phillips, T. (2008) Regulation of transcription and gene expression in eukaryotes. Nat. Educ. 1: 199.
Köhler, G. A., A. Brenot, E. Haas-Stapleton, N. Agabian, R. Deva, and S. Nigam (2006) Phospholipase A2 and phospholipase B activities in fungi. Biochim. Biophys. Acta 1761: 1391–1399.
Subramoni, S., Z. R. Suárez-Moreno, and V. Venturi (2010) Lipases as pathogenicity factors of plant pathogens. pp. 3269–3277. In: K. N. Timmis (ed.). Handbook of Hydrocarbon and Lipid Microbiology. Springer, Berlin, Germany.
Mondal, S., S. Baksi, A. Koris, and G. Vatai (2016) Journey of enzymes in entomopathogenic fungi. Pac. Sci. Rev. A 18: 85–99.
Campbell, K., J. Westholm, S. Kasvandik, F. Di Bartolomeo, M. Mormino, and J. Nielsen (2020) Building blocks are synthesized on demand during the yeast cell cycle. Proc. Natl. Acad. Sci. U. S. A. 117: 7575–7583.
Blank, H. M., O. Papoulas, N. Maitra, R. Garge, B. K. Kennedy, B. Schilling, E. M. Marcotte, and M. Polymenis (2020) Abundances of transcripts, proteins, and metabolites in the cell cycle of budding yeast reveal coordinate control of lipid metabolism. Mol. Biol. Cell 31: 1069–1084.
Konopka, J. B. and J. W. Thorner (2013) Pheromone receptors (yeast). pp. 441–446. In: W. J. Lennarz and M. D. Lane (eds.). Encyclopedia of Biological Chemistry. 2nd ed. Academic Press, London, UK.
Mouyna, I., L. Hartl, and J.-P. Latgé (2013) β-1,3-glucan modifying enzymes in Aspergillus fumigatus. Front. Microbiol. 4: 81.
Etxebeste, O., M. Ni, A. Garzia, N.-J. Kwon, R. Fischer, J.-H. Yu, E. A. Espeso, and U. Ugalde (2008) Basic-zipper-type transcription factor FlbB controls asexual development in Aspergillus nidulans. Eukaryot. Cell 7: 38–48.
Bayram, Ö., S. Krappmann, M. Ni, J. W. Bok, K. Helmstaedt, O. Valerius, S. Braus-Stromeyer, N.-J. Kwon, N. P. Keller, J.-H. Yu, and G. H. Braus (2008) VelB/VeA/LaeA complex coordinates light signal with fungal development and secondary metabolism. Science 320: 1504–1506.
Wu, M.-Y., M. E. Mead, S.-C. Kim, A. Rokas, and J.-H. Yu (2017) WetA bridges cellular and chemical development in Aspergillus flavus. PLoS One 12: e0179571.
Gibellini, F. and T. K. Smith (2010) The Kennedy pathway—de novo synthesis of phosphatidylethanolamine and phosphatidylcholine. IUBMB Life 62: 414–428.
John, E., K. B. Singh, R. P. Oliver, and K.-C. Tan (2021) Transcription factor control of virulence in phytopathogenic fungi. Mol. Plant Pathol. 22: 858–881.
Lara-Rojas, F., O. Sánchez, L. Kawasaki, and J. Aguirre (2011) Aspergillus nidulans transcription factor AtfA interacts with the MAPK SakA to regulate general stress responses, development and spore functions. Mol. Microbiol. 80: 436–454.
Leiter, É., T. Emri, K. Pákozdi, L. Hornok, and I. Pócsi (2021) The impact of bZIP Atf1 ortholog global regulators in fungi. Appl. Microbiol. Biotechnol. 105: 5769–5783.
Watson, G., Z. A. Ronai, and E. Lau (2017) ATF2, a paradigm of the multifaceted regulation of transcription factors in biology and disease. Pharmacol. Res. 119: 347–357.
Mendoza-Martínez, A. E., N. Cano-Domínguez, and J. Aguirre (2020) Yap1 homologs mediate more than the redox regulation of the antioxidant response in filamentous fungi. Fungal Biol. 124: 253–262.
Lee, B.-Y., S.-Y. Han, H. G. Choi, J. H. Kim, K.-H. Han, and D.-M. Han (2005) Screening of growth- or development-related genes by using genomic library with inducible promoter in Aspergillus nidulans. J. Microbiol. 43: 523–528.
Haas, H., K. Angermayr, I. Zadra, and G. Stöffler (1997) Overexpression of nreB, a new GATA factor-encoding gene of Penicillium chrysogenum, leads to repression of the nitrate assimilatory gene cluster. J. Biol. Chem. 272: 22576–22582.
Li, C., Q. Zhang, Y. Xia, and K. Jin (2021) MaAreB, a GATA transcription factor, is involved in nitrogen source utilization, stress tolerances and virulence in Metarhizium acridum. J. Fungi (Basel) 7: 512.
Meng, L., X. Lyu, L. Shi, Q. Wang, L. Wang, M. Zhu, I. Mukhtar, B. Xie, and W. Wang (2021) The transcription factor FvHmg1 negatively regulates fruiting body development in winter mushroom Flammulina velutipes. Gene 785: 145618.
Acknowledgements
The authors would like to thank Department of Zoology, SciKU Biodata Server, Faculty of Science, Kasetsart University for supports, resources and computing facilities. This research and innovation activity is funded by National Research Council of Thailand (NRCT). R.T. gratefully acknowledges supports from the Science Achievement Scholarship of Thailand (SAST) and Interdisciplinary Graduate Program in Bioscience, Faculty of Science, Kasetsart University. W.V. would like to thank Kasetsart University Research and Development Institute (KURDI), Omics Center for Agriculture, Bioresources, Food, and Health, Kasetsart University (OmiKU) and International SciKU Branding (ISB), Faculty of Science, Kasetsart University for supports.
This research was funded by The Thailand Research Fund, grant number RSA6180001 and National Research Council of Thailand (NRCT).
Author information
Authors and Affiliations
Contributions
R.T. performed experiments, analyzed the data, prepared figures and tables, and wrote the manuscript. K.L. assisted in experimental design, data interpretation and revised the manuscript. N.R. analyzed and interpreted the data, and revised the manuscript. M.K. assisted in data interpretation and revised the manuscript. W.V. designed all experiments, interpreted all results, supervised throughout the study and revised the manuscript. All authors reviewed and approved the final manuscript.
Corresponding author
Ethics declarations
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Neither ethical approval nor informed consent was required for this study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
12257_2022_207_MOESM1_ESM.pdf
Dissecting Metabolic Regulation in Mycelial Growth and Fruiting Body Developmental Stages of Cordyceps militaris through Integrative Transcriptome Analysis
Rights and permissions
About this article
Cite this article
Thananusak, R., Laoteng, K., Raethong, N. et al. Dissecting Metabolic Regulation in Mycelial Growth and Fruiting Body Developmental Stages of Cordyceps militaris through Integrative Transcriptome Analysis. Biotechnol Bioproc E 28, 406–418 (2023). https://doi.org/10.1007/s12257-022-0207-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-022-0207-5