Abstract
We present experimental data obtained with newly hatched veliger larvae of the gastropod Concholepas concholepas and juveniles of the mussel Perumytilus purpuratus exposed to three pCO2 levels. Egg capsules of C. concholepas were collected from three geographic locations in northern (Antofagasta), central (Las Cruces), and southern Chile (Calfuco), and then incubated throughout their entire intra-capsular life cycle at three nominal pCO2 levels, ~400, 700, and 1,000 ppm. Similarly, P. purpuratus were collected from both Las Cruces and Calfuco and exposed to the same pCO2 levels during 6 weeks. Hatched gastropod larvae and mussel juvenile were fed with the haptophyte Isochrysis galbana. Clearance and ingestion rates were estimated for newly hatched larvae, and for juvenile mussel these rates were measured at two observation times (3 and 6 weeks). Our results clearly showed a significant negative effect of elevated pCO2 on the clearance and ingestion for both C. concholepas larvae and P. purpuratus juveniles, which dropped between 15 up to 70 % under high pCO2 conditions. The present study has also shown large variations in the sensitivities of C. concholepas larvae from different local populations (i.e. Antofagasta, Las Cruces, and Calfuco). The influence of both corrosive upwelling waters and the influence of freshwater discharges from Maipo River may explain the minor negative effect of high pCO2 conditions in hatched larvae from Las Cruces’ egg capsules, which would suggest that they are inherently more tolerant to ocean acidification (OA) than organisms that live on regions with a lower pCO2 variability. The present study suggests the need for site-specific studies and reveals the important effect of low pH conditions on feeding activity. Furthermore, this study supports the notion that feeding is a key physiological process susceptible to the effects of OA in marine invertebrates.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Over the last two centuries, human activity has become an additional force in the global climate system, as exemplified by greenhouse gas emissions such as CO2 derived from the burning of fossil fuels (e.g. Caldeira and Wicket 2003; Orr et al. 2005). Although the ocean has partially absorbed the anthropogenic atmospheric CO2, this has come at the expense of a significant reduction in pH and the concentration of carbonate ion ([CO3 −2]), a process known as ocean acidification (OA). OA may promote changes in the carbonate chemistry of seawater (e.g. Sabine et al. 2004), resulting in a wide range of effects in marine organisms such as alteration in physiological processes (Kurihara et al. 2004; Fabry et al. 2008), changes in algal photosynthesis (Feng et al. 2009; Egge et al. 2009), plankton community structure (Engel et al. 2008; Huang et al. 2011), among others. Within the various marine organisms particularly vulnerable to OA, calcifying organisms, such as marine mollusks, are exceptionally vulnerable (Byrne 2011; Gazeau et al. 2013). They have been recognized for their great ecological and economic value, as a food source for humans. In consequence, any impacts of OA on sensitive life history traits of these organisms will potentially lead to adverse ecological and economic impacts.
New scientific evidences suggest that pH drop might affect significantly the physiology of marine mollusks (Chan et al. 2011; Navarro et al. 2013; Vargas et al. 2013) and, therefore, their ecological functions and interactions with lower and/or higher trophic levels (Widdicombe and Spicer 2008). The general reported effects of OA have been associated to changing growth and calcification patterns likely to be related to an altered energy budget allocation (Thomsen and Melzner 2010). Calcification process seems to be strongly controlled by a biological control. Previous studies conducted with mussel species (e.g. Melzner et al. 2011; Thomsen et al. 2013) suggest that shell growth upon ocean acidification scenarios can be less negatively affected when food availability is abundant and feeding is enhanced, considering the significant energetic effort need to keep inner shell integrity. Therefore, feeding could be one of the key physiological processes affected by OA. However, there have been scarce reports about the effect of OA on feeding behavior in both adult and/or juvenile/larval stages of marine invertebrates (Stumpp et al. 2011; Barton et al. 2012; Vargas et al. 2013; Navarro et al. 2013).
The eastern boundary Humboldt Current System (HCS) off Chile is characterized by spatial-environmental heterogeneity in oceanographic conditions and ecological patterns (Thiel et al. 2007). Coastal upwelling areas, such as found in northern-central HCS, are predicted to be strongly affected by OA and deoxygenation. Coastal upwelling waters have low dissolved O2 and are CO2 supersaturated resulting in lower coastal pH (Feely et al. 2008). In addition, non-seasonal freshwater discharges flow into some areas in Central Chile. Riverine waters are also acidic in comparison with oceanic waters, due to its low alkalinity and high dissolved inorganic carbon and pCO2 (Salisbury et al. 2008; Duarte et al. 2013). In consequence, marine mollusks inhabiting intertidal coastal environments are exposed to wide range of natural fluctuations of pH in their environment, which may determine a level of tolerance based on their natural range of exposure. Previous studies with mussels from different geographic locations have evidenced a wide range of negative effects, depending of the past environmental history (Range et al. 2013). Nonetheless, the extreme range of pH variability does not necessarily translate to extreme resistance to future OA (Hofmann et al. 2011).
Here, we present a laboratory study focused on two ecological important species of the rocky intertidal and subtidal communities distributed along the Chilean coast, the small-sized intertidal bivalve Perumytilus purpuratus and the economically important carnivorous gastropod Concholepas concholepas, known as “loco” (Chilean abalone). Both species have been selected for this study based in their wide latitudinal range along the Pacific coast (from 0° to 52°S for P. purpuratus and from 12°S to 55°S for C. concholepas) (Osorio and Bahamonde 1968; Stuardo 1979; Prado and Castilla 2006). P. purpuratus also play a highly relevant ecological role, forming conglomerated beds over intertidal rocks and therefore affecting habitat conditions and the local diversity of invertebrate species (Prado and Castilla 2006). Furthermore, both species constitute important components of the rocky intertidal food web (Prado and Castilla 2006). In nature, females of C. concholepas laid clumps of egg capsules to rocky substratum where they remain cemented for about 3 months (Manríquez and Castilla 2001). Here, an intra-capsular development period of 1 to 2 months is required for hatching (Gallardo 1973).
Most studies on the impact of OA on invertebrates have been relatively short term and mostly focused on one life history stage. However, a majority of marine invertebrates develop by means of benthic egg capsules and then on different larval stages and/or juvenile stages. In the present study, we have exposed juvenile stages of P. purpuratus and egg capsules of C. concholepas collected from different geographic locations along a latitudinal range and environmental histories to three different and constant pCO2 environments for many weeks (i.e. entire intra-capsular life cycle for the gastropod larvae and for 3 to 6 weeks for the mussel juveniles) (Fig. 1a). Then, we evaluated the ingestion and clearance rates of P. purpuratus juveniles and newly hatched C. concholepas veliger larvae originating from the treated egg capsules in order to evaluate effects of OA on feeding behavior of these species. We hypothesize in this study that physiological responses of both species under high pCO2 conditions are different depending on the geographic origin and therefore environmental histories of exposition to low pH/high pCO2 natural conditions.
Methods
Collection of Individuals
Newly hatched larvae of C. concholepas were obtained from egg capsules collected in rocky intertidal habitats (Antofagasta, 23°45′S, 70°26′W, exp I and II), and from egg capsules recently laid by females maintained in captivity in aquariums in Central Chile (Las Cruces, 33°30′S, 71°38′W, exp III and IV), and from southern Chile (Coastal Laboratory at Calfuco, 39°78′S, 73°39′W, exp V and VI). At Calfuco, as soon as they were laid, the egg capsules were identified, and a few days later, when the female had moved away from the oviposition site, they were transferred to rearing conditions. A similar procedure was carried out at Las Cruces, but once egg capsules were accessible, they were transported under wet conditions to Calfuco.
Juvenile individuals of P. purpuratus were randomly sampled in the rocky shore of two of the three locations previously mentioned: Las Cruces and Calfuco, accordingly to Osorio (2002). After collection, all animals were maintained in filtered seawater and translated to the lab in a thermobox within 2 h after sampling. Ninety individuals were cleaned, sized (12 ± 1.4 mm lengths), and then separated in three groups of 30 individuals each one and transferred to rearing conditions under different pCO2.
Seawater CO2 Manipulation
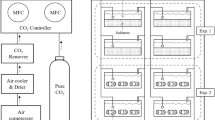
In our study, three plastic 280-L tanks that were used as acidification units to generate seawater to three nominal levels of pCO2 were set: 400, 700, and 1,000 ppm, hereafter referred to as “low,” “medium,” and “high” levels of pCO2. The pCO2 conditions chosen for the medium and high levels were selected taking into account the rate of change projected by the years 2070–2110 (i.e. based on rate of change in pH predicted by the most extreme scenario [RCP8.5 scenario] of atmospheric CO2 (Meinshausen et al. 2011). For the medium and high CO2 treatments, CO2 concentrations were modified by equilibrating the seawater with air containing different CO2 concentrations, as Findlay et al. (2008). Air/CO2 mixtures were produced using a bulk flow technique, where known flows of dry air (i.e. by compressing atmospheric air, 117 psi, and passing through a 1 μm particle) and ultra-pure (i.e. research grade) CO2 gas were supplied, via mass flow controller (MFC), and mixed before equilibration with sea water. Airflow in MFC was set manually to 5 L min−1 for both treatments, and CO2 flow was set manually to 1.33 and 4.25 mL min−1, to produce CO2 treatments of approximately 700 and 1,000 ppm, respectively. The CO2 of blended gas was monitored to allow fine regulation of CO2 through MFCs to reach each target pCO2 in seawater. During the experiments, seawater pH (total scale, pHT) were monitored in each tank every 3 days in a 25 mL cell thermostatted at 25.0 ± 0.1 °C for standardization, with a pH meter Metrohm® using a glass-combined double junction Ag/AgCl electrode following DOE potenciometric method (DOE, 1994). Temperature and salinity were monitored during incubations by using a small CTDO (Ocean Seven 305 Plus). Temperature averaged 13 °C in spring and from 15.4 to 16 °C during summer experiments (Table 1). Total alkalinity (TA) was also measured using the automated potentiometric titration method (Haraldsson et al. 1997). The pHT, AT, phosphate (Strickland and Parsons 1968), dissolved silicate (Strickland and Parsons 1968), temperature, and salinity data were used to calculate the rest of carbonate system parameters (e.g. pCO2, CO3 2−) and the saturation stage of Omega aragonite (Ωarag) using CO2SYS software (Lewis and Wallace 1998) set with Mehrbach solubility constants (Mehrbach et al.1973) refitted by Dickson and Millero (1987).
Experimental Rearing
Egg capsule rearing was achieved through two methodologies developed to maintain them during the entire intra-capsular developing period under contrasting pCO2 levels (i.e. 400, 700, and 1,000). The first methodology included a recirculation system in which the seawater equilibration took place in three large (250 L) plastic tanks, and electric pumps were used to move the equilibrated seawater into the rearing containers. A small opening (1 cm in diameter) was made within the upper part of each rearing beaker and sealed with 100 μm mesh size to allow seawater overflow and to prevent larval loss if hatching took place during the rearing. Seawater overflow was accumulated within a plastic container in which the rearing beakers were placed and then conducted by gravity through a silicone pipe into the large equilibration tanks. This rearing methodology was used with egg capsules collected in Antofagasta. The second methodology included 5-L Plexiglas containers filled once a day with equilibrated seawater generated in the large tanks (above). However, to maintain the required pCO2 levels between seawater changes, a continuous injection of a mix of air and CO2 was provided. This rearing methodology was used with egg capsules collected in Las Cruces and Calfuco. In both methodologies, the egg capsules were cleaned regularly, and developing took place within the described timing (ca. 2 months). Once maturity was achieved and hatching egg capsules were recorded, they were removed from the rearing containers to obtain the hatching larvae. Since differences in egg capsule developing were not detected and the contrasting pCO2 and pHT levels were achieved similarly through both methodologies (Table 1), the consequence or the type of egg capsule rearing was not analyzed in the present study.
Individuals of P. purpuratus from both Las Cruces (n = 90) and Calfuco (n = 90) were reared during a mean period of 3 to 6 weeks at the different pCO2 levels (low, medium, and high; 30 mussels per CO2 treatment) in three plastic 22-L containers filled with 15 L of corresponding equilibrated seawater. The continuous injection of air and the required CO2 was used to maintain the experimental levels between water changes. After water changes, the individuals were fed ad libitum (>10 μg Chl a L−1) with I. galbana and seawater in each bakers was renewed every day (from Monday to Friday) during mean incubation time of 10 weeks (Table 2).
Feeding Experiments with Veliger Larvae
Once a week, the rearing containers were washed and the egg capsules were carefully cleaned with the aid of a soft paintbrush. Mature egg capsules of C. concholepas bearing near-hatch larvae were easily recognizable by their brownish coloration (Manríquez and Castilla 2001), which under our rearing conditions were obtained after about 2 months. The detection of newly hatched larvae in the rearing containers marked the end of the intra-capsular period (from ~6.5 to ~8 weeks). Mature egg capsules in process of hatching were then removed from the rearing containers and placed in a small petri dish with 0.5 μm filtered seawater. Under a stereomicroscope, hatched larvae were carefully removed and transferred to experimental acclimation conditions. Experiments with newly hatched larvae of C. concholepas hatched from egg capsules from different locations were conducted on six separate occasions (Table 1): 20–21 June 2011 (exp I, Antofagasta), 02–03 August 2011 (exp II, Antofagasta), 18–19 October 2011 (exp III, Las Cruces), 25–26 January 2012 (exp IV, Las Cruces), 13–14 April 2012 (exp V, Calfuco), and 15–16 April 2012 (exp VI; Calfuco). Naturally induced hatched larvae of C. concholepas selected for the experiments were transferred to 1,000-mL acid-washed polycarbonate bottles filled with the filtered and equilibrated seawater from the three different pCO2 treatments and fed with ~4,000 to 5,000 cell mL−1 (2.2 ± 0.2 and 3.0 ± 0.3 μg Chl a L−1) of the haptophyte I. galbana sp. Care was taken to avoid air bubbles in the bottles. Three control bottles without larvae and three bottles with 30 newly hatched larvae (0.06 ind mL−1) for each treatment (low, medium, and high pCO2 levels) were incubated for approximately 24 h and periodically rotated by hand to avoid particle sedimentation. Bottles were immersed in a container with flow-through seawater system used for maintaining temperature fluctuation during a given experiment within 1°. In all experiments, 300-mL subsamples for determination of chlorophyll a (Chl a) were 0.7 μm filtered and dark extracted in acetone 95 % before measurement on a TD 700 Turner fluorometer (Strickland and Parsons 1968). The remaining volume was gently poured through a 20-μm sieve in order to check that incubated larvae were healthy and actively filtering after the incubation period. Clearance (CR) and ingestion rates (IR), measured as Chl a removal, were calculated according to Frost (1975) modified by Marin et al. (1986). Clearance and ingestion rates were calculated only when the differences in prey concentration between control and experimental bottles proved to be significant (t test: p < 0.05). Food availability (i.e. biomass) of I. galbana in each CO2 treatment was estimated from the T0 subsample. For all experiments, there were no significant differences in food availability (Chl a) among pCO2 treatments; ANOVA tests, exp I–II (F 2,12 = <1, p > 0.05), exp III–IV (F 2,12 = <1, p > 0.05), exp V–VI (F 2,12 = <1, p > 0.05). However, variations on food availability (Chl a) were evident between experiments (exp I > exp II, exp III < exp IV, exp V > exp VI) (not shown).
Feeding Experiments with P. purpuratus
Feeding experiments were conducted with individuals from both locations after ca. three (“time I”) and 6 weeks (“time II”) of CO2 exposition (Table 2). Experiments with juveniles of P. purpuratus were conducted on four separate occasions (Table 2): 29–30 January 2012 (exp I), 02–03 February 2012 (exp II), 12–13 April 2012 (exp III), and 17–18 April 2012 (exp IV). Individuals belonging to each experimental group were individually sorted into 140-mL acid-washed DBO amber bottles filled with filtered seawater (1 μm) and equilibrated with the respective CO2 treatment. Three control bottles without P. purpuratus and ten bottles with one individual for each treatment (low, medium, and high pCO2 levels) were incubated for approximately 4 h and periodically rotated by hand to avoid particle sedimentation. Previous to the experiment juvenile body mass (mb) was determined with an analytical balance (±0.01 mg). Food availability varied between 11.7 and 11.8 μg Chl a L−1, which correspond to a mean cell concentration of ~20,000 cell mL−1 of I. galbana. Bottles were immersed in a container with flow-through seawater system used for maintaining temperature fluctuation during a given experiment within 1° (Table 2). After to sieving through 80 mm (fecal material), 8 mL of all bottles were measured directly on TD 700 Turner fluorometer (Strickland and Parsons 1968). The length and maximal width of each valve from each individual were estimated, and with these measurements, wet and buoyant weight was then estimated accordingly with Palmer (1982). Clearance (CR) and ingestion rates (IR) were measured as Chl a removal and were calculated according to Coughlan (1969). For all experiments, there were no significant differences in food availability among CO2 treatments (Table 4). However, variations on food availability were significant between consecutive observation times performed in Calfuco: time I > time II (Table 4).
Statistical Analyses
Since experimental conditions (salinity, temperature, and food availability) were statistically similar on each couple of experiments performed with C. concholepas larvae at the three locations, results will be pooled in one single observation per location: Antofagasta, Las Cruces, and Calfuco. Such that salinity, temperature, food availability, and clearance and ingestion rates of C. concholepas larvae were compared among geographic locations and pCO2 treatments through a two-way ANOVA tests. When significant differences were found, either among locations or pCO2 concentrations, a least significant difference (LSD) post hoc test identified the source of such variability. A similar approach but to evaluate the role of incubation time (i.e. time I and time II) and pCO2 treatments was adopted to evaluate variability on clearance and ingestion rates of P. purpuratus.
Results
Experimental Conditions
The experimental setup and average environmental and carbonate chemistry parameters recorded during experiments with C. concholepas larvae are shown in Table 1. Saturation states for Aragonite varied significantly among all the different treatments and experiments (ANOVA, F 2,12 = 107.3, p < 0.0001) and during the winter and spring incubations (exp I to III) ranged from supersaturation (Ωarag = 2.0) to slightly undersaturated (Ωarag = 0.8) (Table 1). Meantime, experimental setup, environmental and carbonate chemistry parameters recorded during experiments with P. purpuratus are shown in Table 2. For incubations with organisms from Las Cruces, pHT 25 °C was maintained at 8.08, 7.86, and 7.73 and 8.07, 7.84, and 7.71 for exp I and II, respectively, which correspond to pCO2 levels of approximate 355, 661, and 916 and 372, 695, and 949 μatm, respectively. Saturation states for Aragonite varied less in relation to larvae experiments as well as undersaturation was never reached (Ωarag > 1) (Table 2).
Clearance and Ingestion Rate of Veliger Larvae upon High pCO2
Statistic comparison of experimental conditions (“temperature,” “salinity,” and “food availability”) and clearance and ingestion rates of newly hatched larvae of C. concholepas in relation to “location” and “pCO2” levels are showed in Table 3. Highest temperature (16.5 ± 1 °C), salinity (34.6 PSU), and food availability (2.84 ± .2 μg Chl a L−1) were recorded during experiments in summer with egg capsules from Las Cruces (Fig. 2a, Table 1). These environmental conditions varied significantly among experiments with organisms from different locations (one-way ANOVA, p < 0.05); however, all these variables remain similar upon the different pCO2 treatments (two-way ANOVA, with non-significant location × pCO2 interaction, F 1,45 = <1, p > 0.05). These results are highlighted in Table 3 since they support further evaluations of CO2 effects on larval feeding traits. Maximum clearance and ingestion rates were observed in newly hatched larvae from egg capsules from Las Cruces, then, followed by those from Antofagasta, and finally those from Calfuco (Fig. 2b, c). Although, larval size of newly hatched larvae were slightly larger in Las Cruces and Valdivia than those from capsules from Antofagasta, differences were not significant. Moreover, mean size of newly hatched larvae from egg capsules from these different geographic locations reared under contrasting pCO2 levels did not show differences in larval size (i.e. 250 ± 6, 245 ± 9, and 252 ± 7 μm at low, medium, and high pCO2 levels, respectively. Clearance rates varied between 2.6 ± 0.3 (Las Cruces) and 1.9 ± 0.4 (Calfuco) mL ind−1 day−1 (Fig. 2b). A similar pattern was observed on ingestion rates, which varied between 6.8 ± 0.8 (Las Cruces) and 4.1 ± 0.9 (Calfuco) μg Chl a ind−1 day−1 (Fig. 2c). Estimates from the different feeding experiments with Isochrysis diet clearly showed a significant reduction in clearance rate, between ~15 up to 60 % from the control condition at low pCO2 upon increasing pCO2 levels in seawater. This negative effect of high pCO2 on larval clearance and ingestion was more intense in veliger larvae from Calfuco (mean 14 % of ingestion reduction at nominal 700 μatm) and Las Cruces (mean 18 % of ingestion reduction at nominal 1,000 μatm) (Fig. 2b, c). Indeed, both clearance and ingestion rates varied significantly in relation to both variability factors, since the interaction between geographic location and different pCO2 levels resulted significant on both clearance and ingestion rates (two-way ANOVA, with significant location × pCO2 interaction, F 2,45 = 3, p = 0.04 and F 1,45 = 4, p = 0.01, respectively) (Table 3).
a Comparison of the initial food availability (μg Chl a L−1) before CO2 equilibration for feeding of newly hatched larvae of C. concholepas; vertical bars belong to total plankton biomass, b individual clearance (mL ind−1 day−1), and c ingestion rates (μgC Chl a ind−1 day−1) in six experiments including three CO2 treatments. Error bars denote standard error
Clearance and Ingestion Rate of Juvenile Mussels upon High pCO2
Statistic comparison of experimental conditions (temperature, salinity, and food availability) and clearance and ingestion rates of P. purpuratus in relation to “time of exposition” (i.e. 3 or 6 weeks) and “pCO2 levels” is showed in Tables 4 and 5. Temperature in observations conducted in early January to April 2012 decreased from ca. 16.9 to 15.7 °C (Table 1). Temperature and salinity did not vary significantly neither between observation times and pCO2 levels (one-way ANOVA, p > 0.05). However, food availability decreased between both observations (times I and II) only for experiments with P. purpuratus collected in Calfuco (one-way ANOVA, p = 0.0001) (Fig. 3a, Table 5).
a Comparison of the initial food availability (μg Chl a L−1) before CO2 equilibration for feeding of juvenile P. purpuratus; vertical bars belong to total plankton biomass, b weight-specific clearance (mL g−1 h−1), and c ingestion rates (μgC Chl a g−1 h−1) in two obseervations (I and II) including three CO2 treatments. Error bars denote standard error
Our results clearly evidenced that mussels significantly decreased their clearance and ingestion rates, when they were exposed to a high pCO2 level, with a reduction from 15 up to more than 70 %. Significant changes were observed in clearance rates in relation to pCO2 levels for individuals from both geographic locations (“Las Cruces” and “Calfuco”), decreasing from ~7,997 to 4,916 mL g−1 day−1 from low to high pCO2 levels, respectively (Fig. 3). The effect of high pCO2 was even more intense with the time of exposition for individuals collected in Las Cruces (two-way ANOVA, with significant time × pCO2 interaction, F 1,54 = 6.2, p = 0.004) (Table 4). The effect of high pCO2 on ingestion rates was only evident for individuals collected in Calfuco, since for those collected in Las Cruces, we only found significant differences at different exposition time, with a decrease between observation times of ca. 19 % (one-way ANOVA time, F 1,54 = 4, p = 0.05).
A frequency analysis performed on clearance rates of P. purpuratus collected from both Las Cruces and Calfuco evidenced that as time of exposition and pCO2 level increase, most individuals reduce their feeding activity, since the highest frequency is observed for lower ranges of clearance rates, but even more, new ranges of low clearance rates appears between observation time (Fig. 4).
Discussion
The periods of exposure used in our study for both gastropod egg capsules and juvenile mussels (42 to 56 days) allowed a better appraisal of the long-term effects on feeding performance of these species, which may contribute to reconcile the context of a permanent exposure under pCO2-driven OA scenarios. During our study, our clearance rates estimates in “control” treatment were in the mean range of published values for other veliger larvae (~1.5 to 3.5 mL ind−1 day−1). For instance, Baldwin and Newell (1991) found that veligers of the eastern oyster Crassostrea virginica cleared autotrophic 14C-labeled cells at 2 mL ind−1 day−1. Vargas et al. (2013) had previously reported clearance estimates for newly hatched larvae of C. concholepas larvae of 0.4 up to 7 mL ind−1 day−1, based on cell counts of natural food assemblages. Nevertheless, differences in clearance may be associated with several factors, including field food concentration, incubation temperature and, more importantly, differences in larval body size. Indeed, all feeding structures, such as length of the prototrochal ciliary band, prototrochal cilia, and the angular velocity of the cilia, scale with larval body size (Strathmann and Leise 1979). Indeed, although larval size in those individuals hatched from egg capsules from all locations were not significantly different, individuals from Antofagasta were slightly smaller than those from other locations. Furthermore, similar than our finding, a recent study have detected that levels of pCO2 do not affect morphological traits of newly hatched larvae of C. concholepas such as larval size during the egg capsule rearing. However, the same study detected that larval size is significantly affected by the interaction between pCO2 levels with female nested within source locality (Manríquez, personal communication).
Our clearance estimates for juveniles of P. purpuratus also are in the relatively same magnitude with clearance rates reported for juveniles and/or adults of other mussel species (~0.15 to 0.46 L g−1 h−1). For instance, for Mytilus edulis, Petersen et al. (2004) reported a clearance from 5.3 to 10 L g−1 h−1, and Denis et al. (1999) have reported for the Mediterranean mussel Mytilus galloprovincialis a similar clearance rate from ~0.2 to 0.4 L g−1 h−1. For another mussel species in Chilean waters, Choromytilus chorus, Toro et al. (2003) reported a clearance rate of ~0.7 to 2.2 L g−1 h−1.
During the last few years, several studies have reported the impacts of OA on the growth and development of larval and adult stages of shellfish (Miller et al. 2009). However, to date, there are few reports regarding the effect of OA on feeding behavior of marine invertebrates, especially on early life stages (Vargas et al. 2013). It is evident from our experiments that an important effect of CO2-driven OA is the radical decrease in larval and juvenile feeding of both gastropod and mussel species selected. Newly hatched larvae of the gastropod C. concholepas and juvenile stages of the mussel P. purpuratus decreased their clearance rate between 15 up to 70 % under high pCO2 in comparison to control conditions at low pCO2 levels. However, the underlying mechanism by which OA impacts the feeding process remains unclear for us.
Despite that feeding processes during early life stages of marine invertebrates are likely to be more sensitive to OA than in adults (Findlay et al. 2008), there are few reports regarding the effect of OA on feeding behavior of larval invertebrates (Stumpp et al. 2011; Barton et al. 2012). Recently, Vargas et al. (2013) reported a significant effect of elevated pCO2 (~1,000 ppm pCO2) on the intensity of C. concholepas larval feeding on natural food assemblages, which dropped by 60 %. Furthermore, these authors also reported that high pCO2 induced changes in the food selectivity of C. concholepas larvae, switching from large diatom cells to small nanoflagellate and cyanobacteria. In the present study, we have used as a single food source, the nanoflagellate I. galbana, a small algal cell, which should be selected under high pCO2 conditions, but clearance on this small algal cell also decreased significantly under this condition.
In recent years, numerous studies have demonstrated the negative effects of OA on marine bivalves. During a long-term exposure at high pCO2 (35 days), Navarro et al. (2013) showed that Mytilus chilensis showed a significant reduction in its clearance rate. Fernández-Reiriz et al. (2011) also found a reduction in the feeding rate by the clam Ruditapes decussatus. Recently, Range et al. (2013) reported a significant decrease in the clearance rate of R. decussatus during a long-term experiment under high pCO2 (i.e. 1,000 to 4,000 μatm). These authors used the same food item, I. galbana, at similar food concentration (0.8 to14.8 μg Chl a −1 L−1). Food concentration is another important issue as food availability has explicitly showed a potential counteractive effect on marine organisms under elevated pCO2 conditions (e.g. Holcomb et al. 2010, 2012; Melzner et al. 2011; Thomsen et al. 2013). Nevertheless, Chl a concentration in experiments with C. concholepas ranged from 2 to 3 μg Chl a −1 L−1, which is within the main range of Chl a concentration observed for Chilean coastal waters (Yuras et al. 2005), and in those conducted with P. purpuratus, mean Chl a concentration was similar than used by Range et al. (2013), resembling a bloom condition (i.e. 11 to 12 μg Chl a −1 L−1). In consequence, the decrease in larval filtration rate observed in our study could be associated to other physiological constraints, such as hypercapnia, which can also lead to additional metabolic costs for many marine organisms (Cummings et al. 2011; Stumpp et al. 2011), as well as the effect of uncompensated extracellular pH (Stumpp et al. 2012), affecting the filtration rate in these organisms. In any case, our results also support the notion that feeding is one of the key physiological processes affected by OA in marine invertebrates.
Substantial reductions of larvae ingestion occurred at mid-high CO2 levels among larvae from different locations. This was especially clear in the case of larvae from Las Cruces where ingestion reached a significant reduction of 18 ± 3 % (Kruskall-Wallis test H = 4, p < 0.05) at nominal CO2 level of 1,000 μatm in relation to the control situation. Thus, the present study has also shown large variations in the sensitivities of newly hatched larvae from different local populations (i.e. Antofagasta, Las Cruces, and Calfuco). Consequently, information about geographic variation is critical because many morphological, life history, and metabolic traits show variation across space (Beniash et al. 2010; Ramajo et al. 2013), which is often attributed to organism adaptation over environmental gradients (Levins 1968). For example, (Pascal et al. 2010) recorded that copepods associated with sediments with higher CO2 concentrations were better adapted to hypercapnic environments than copepods inhabiting sediments with lower CO2 levels. A similar hypothesis was proposed by (Widdicombe et al. 2009) to explain the different tolerances of two macrofauna communities to sea acidification. Cummings et al. (2011) showed that the expression of the chitin synthase (CHS) enzyme, which is a key in the synthesis of bivalve shells, was upregulated in individuals of the Antarctic bivalve Laternula elliptica exposed to hypercapnic conditions, indicating some degree of adaptation to sea acidification in this species. The coastal ecosystems of the Chilean Southeastern Pacific coast is characterized by an almost linear coastline (north of 40°S) and by strong pCO2 gradients between the atmosphere and the surface water, with high spatial and temporal variability in the northern-southern direction (Torres et al. 2011). Large upwelling areas are located northward of 37°S and determine the monthly fluctuation in sea surface temperature (SST) and pCO2 levels (Torres et al. 2011). Typically, quasi-permanent upwelling events, occurring in northern Chile, such as in our study site, Antofagasta, injecting low dissolved O2 and CO2 supersaturated waters in surface waters along the coast, resulting in lower ocean pH, resulting in a strong across-shore pH and pCO2 gradients, similar than occurring in other coastal upwelling areas (e.g. California Current System, Feely et al. 2008). However, in the present study, C. concholepas from the Antofagasta region were collected from Antofagasta Bay, an area already reported as an “upwelling shadow”; therefore, marine invertebrates in this region commonly are not affected by low oxygen/high pCO2 waters (Lagos et al. 2008). Nevertheless, other coastal areas such as our site in Las Cruces in Central Chile are affected by both upwelling and the influence of freshwater discharges from Maipo River, where typically pCO2 levels may increase as a response to organic matter remineralization (i.e. terrestrial and anthropogenic) and its low buffer capacity (Pérez personal communication). This local condition could greatly influence the adaption potential for organisms under a high pCO2 conditions. Indeed, the medium and high levels of pCO2 (i.e. 712 to 1,067 μatm, respectively) represent a natural condition which can be observed in Las Cruces (Lagos et al. 2013; Ramajo et al. 2013), especially in the site where egg capsules were collected and where typically the Maipo River plume arrive daily at the rocky shore (Vargas et al. 2006). In consequence, this natural variability may explain the minor negative effect of high pCO2 conditions in newly hatched larvae from this local population, which indeed may suggest they are inherently more tolerant to OA than organisms that live on regions with a lower pCO2 variability (e.g. Antofagasta). Finally, Calfuco is an area that is not directly affected by both upwelling and/or river discharge (Aguilera et al. 2013). The environmental history of pH exposure at the different selected sites in our study (i.e. Antofagasta, Las Cruces, and Calfuco) could induce differential physiological resilience of both C. concholepas and P. purpuratus and may explain the differential responses upon high pCO2 observed in this study. Similarly, Range et al. (2013) showed large variations in the sensitivities of the mussel M. galloprovincialis, the clams Chamelea gallina and R. decussatus upon high pCO2, among different local populations of the same species. Consequently, more attention should be given to understanding geographic differences in variability rather than differences in mean values in response to OA. These results clearly suggest the need for site-specific studies and local adaptive measures for supporting the ecological and economic roles of these species under future scenarios of climate change in South America.
Summarizing, our results highlight the negative effects of OA on the feeding process of economically and ecologically important marine species under projected rising atmospheric pCO2 scenarios. In spite of the fact that this incubation period encompasses a biologically relevant timescale, our approach also must confront the inherent decoupling between experimental periods and the timescale at which the OA process is projected to occur in nature. In addition, OA in nature will act simultaneously with other climate-related variables, including ocean warming, and the extent of low oxygen minimum zone in coastal areas (Doney et al. 2012). In consequence, these coastal areas should be further studied to assess the mechanisms determining the sensitivity of marine invertebrates under those multiple stressors. This condition may imply even worse scenarios, and it might have a larger effect on the metabolic process of marine invertebrates in Chilean waters. Finally, under high pCO2 scenarios, food quality can be deteriorated as increased CO2 can stimulate carbon fixation through photosynthesis and therefore reducing the nutrient content relative to carbon (Engel et al. 2008). This condition may have significant consequences for the consumer, as nutrient demand for somatic growth can be impaired (Urabe et al. 2003)
Considering that the gastropod C. concholepas supports small-scale fisheries that rely on natural stocks (Leiva and Castilla 2001) and P. purpuratus form dense beds that play an important role as bioengineers on intertidal rocky shores along Chilean coast (Prado and Castilla 2006), any negative impacts associated with OA that affect this species may produce significant socio-economic and ecological disruptions to the ecosystem and to the essential ecosystem services that it provides for humans.
References
Aguilera, V.M., C.A. Vargas, P.H. Manríquez, J.M. Navarro, and C. Duarte. 2013. Low-pH freshwater discharges drive spatial and temporal variations in life history traits of neritic copepod Acartia tonsa. Estuaries and Coasts 1–9.
Baldwin, B.S., and R.I.E. Newell. 1991. Omnivorous feeding by planktotrophic larvae of the eastern oyster Crassostrea virginica. Marine Ecology Progress Series 78: 285–301.
Barton, A., B. Hales, G.G. Waldbusser, C. Langdon, and R.A. Feely. 2012. The Pacific oyster, Crassostrea gigas, shows negative correlation to naturally elevated carbon dioxide levels: implications for near-term ocean acidification effects. Limnology and Oceanography 57: 698–710.
Beniash, E., A. Ivanina, N.S. Lieb, I. Kurochkin, and I.A. Sokolova. 2010. Elevated level of carbon dioxide affects metabolism and shell formation in oysters Crassostrea virginica. Marine Ecology Progress Series 419: 95–108.
Byrne, M. 2011. Impact of ocean warming and ocean acidification on marine invertebrate life history stages: vulnerabilities and potential for persistence in a changing ocean. Oceanography and Marine Biology: An Annual Review 49: 1–42.
Caldeira, K., and M.E. Wickett. 2003. Anthropogenic carbon and ocean pH. Nature 425: 365.
Chan, K.Y.K., D. Grünbaum, and M.J. O’Donnell. 2011. Effects of ocean-acidification-induced morphological changes on larval swimming and feeding. The Journal of Experimental Biology 214: 3857–3867.
Coughlan, J. 1969. The estimation of filtering rate from the clearance of suspensions. Marine Biology 2: 356–358.
Cummings, V., J. Hewitt, A. Van Rooyen, K. Currie, S. Beard, S. Thrush, and V. Metcalf. 2011. Ocean acidification at high latitudes: potential effects on functioning of the antarctic bivalve Laternula elliptica. PLoS One 6: e16069.
Denis, L., E. Alliot, and D. Grzebyk. 1999. Clearance rate responses of Mediterranean mussels, Mytilus galloprovincialis, to variations in the flow, water temperature, food quality and quantity. Aquatic Living Resources 12: 279–288.
Dickson, A.G., and F.J. Millero. 1987. A comparison of the equilibrium constants for the dissociation of carbonic acid in seawater media. Deep Sea Research Part A: Oceanographic Research Papers 34: 1733–1743.
DOE. 1994. Handbook of methods for the analysis of the various parameters of the carbon dioxide system in sea water, version 2. In Report ORNL/CDIAC-74, ed. Dickson, A.G. and C. Goyet. Oak Ridge: Oak Ridge National Laboratory.
Doney, S.C., M. Ruckelshaus, J.E. Duffy, J.P. Barry, F. Chan, C.A. English, and L.D. Talley. 2012. Climate change impacts on marine ecosystems. Annual Review of Marine Science 4: 11–37.
Duarte, C.A., I.E. Hendriks, T.S. Moore, Y.S. Olsen, A. Steckbauer, L. Ramajo, J. Carstensen, J.A. Trotter, and M. McCulloch. 2013. Is ocean acidification an open-ocean syndrome? Understanding anthropogenic impacts on seawater pH. Estuaries and Coasts 36(2): 221–236.
Egge, J.K., J.F. Thingstad, A. Larsen, A. Engel, J. Wohlers, R. Bellerby, and U. Riebesell. 2009. Primary production during nutrient-induced blooms at elevated CO2 concentrations. Biogeosciences 6: 877–885.
Engel, A., K.G. Schulz, U. Riebesell, R. Bellerby, B. Delille, and M. Schartau. 2008. Effects of CO2 on particle size distribution and phytoplankton abundance during a mesocosm bloom experiment (PeECE II). Biogeosciences 5: 509–521.
Fabry, V.J., B.A. Seibel, R.A. Feely, and J.C. Orr. 2008. Impacts of ocean acidification on marine fauna and ecosystem processes. ICES Journal of Marine Science: Journal du Conseil 65: 414–432.
Feely, R.A., C.L. Sabine, J.M. Hernandez-Ayon, D. Ianson, and B. Hales. 2008. Evidence for upwelling of corrosive “acidified” water onto the continental shelf. Science 320: 1490–1492.
Feng, Y., C.E. Hare, K. Leblanc, J.M. Rose, Y. Zhang, G.R. DiTullio, and D.A. Hutchins. 2009. The effects of increased pCO2 and temperature on the North Atlantic spring bloom: I. The phytoplankton community and biogeochemical response. Marine Ecology Progress Series 388: 13–25.
Fernández-Reiriz, M.J., P. Range, X.A. Álvarez-Salgado, and U. Labarta. 2011. Physiological energetics of juvenile clams Ruditapes decussatus in a high CO2 coastal ocean. Marine Ecology Progress Series 433: 97–105.
Findlay, H.S., M.A. Kendall, J.I. Spicer, C. Turley, and S. Widdicombe. 2008. Novel microcosm system for investigating the effects of elevated carbon dioxide and temperature on intertidal organisms. Aquatic Biology 3: 51–62.
Frost, B.W. 1975. A threshold feeding behavior in Calanus pacificus. Limnology and Oceanography 1: 263–266.
Gallardo, C.S. 1973. Desarrollo intracapsular de Concholepas concholepas (Brugière) (Gastropoda, Muricidae). Publicación ocasional. Museo nacional de Historia Natural (Chile) 16.
Gazeau, F., L.M. Parker, S. Comeau, J.P. Gattuso, W.A. O'Connor, S. Martin, H.O. Pörtner, and P.M. Ross. 2013. Impacts of ocean acidification on marine shelled molluscs. Marine Biology 160: 2207–2245.
Haraldsson, C., L.G. Anderson, M. Hassellöv, S. Hulth, K. Olsson. 1997. Rapid, high precision potentiometric titration of alkalinity in ocean and sediment pore waters. Deep-Sea Research Part I: Oceanographic Research Papers 44: 2031–2044.
Hofmann, G.E., J.E. Smith, K.S. Johnson, U. Send, L.A. Levin, F. Micheli, and T.R. Martz. 2011. High-frequency dynamics of ocean pH: a multi-ecosystem comparison. PLoS One 6: e28983.
Holcomb, M., D.C. McCorkle, and A.L. Cohen. 2010. Long-term effects of nutrient and CO2 enrichment on the temperate coral Astrangia poculata (Ellis and Solander, 1786). Journal of Experimental Marine Biology and Ecology 386: 27–33.
Holcomb, M., A.L. Cohen, and D.C. McCorkle. 2012. An investigation of the calcification response of the scleractinian coral Astrangia poculata to elevated pCO2 and the effects of nutrients, zooxanthellae and gender. Biogeosciences 9: 29–39.
Huang, B., W. Xiang, X. Zeng, K.P. Chiang, H. Tian, J. Hu, and H. Hong. 2011. Phytoplankton growth and microzooplankton grazing in a subtropical coastal upwelling system in the Taiwan Strait. Continental Shelf Research 31: 48–56.
Kurihara, H., S. Shimode, and Y. Shirayama. 2004. Sub-lethal effects of elevated concentration of CO2 on planktonic copepods and sea urchins. Journal of Oceanography 60: 743–750.
Lagos, N.A., J.C. Castilla, and B.R. Broitman. 2008. Spatial environmental correlates of intertidal recruitment: a test using barnacles in Northern Chile. Ecological Monographs 78: 245–261.
Lagos, N.A., L. Prado, L. Ramajo, C.A. Vargas, R. Torres, P.H. Manríquez, J.M. Navarro, M.A. Lardies, C. Duarte, and G. Saldías. 2013. Divergent population and community patterns on faunal assemblages associated to intertidal mussel beds: influence of corrosive freshwater input on benthic ecosystems. In this issue.
Leiva, G.E., and J.C. Castilla. 2001. A review of the world marine gastropod fishery: evolution of catches, management and the Chilean experience. Reviews in Fish Biology and Fisheries 11: 283–300.
Levins, R. 1968. Evolution in changing environments. Princeton: Princeton University Press.
Lewis, E. and D. Wallace. 1998. Program developed for CO2 system calculations. Carbon Dioxide Information Analysis Center, Oak Ridge, Tennessee, USA.
Manríquez, P.H., and J.C. Castilla. 2001. Significance of marine protected areas in central Chile as seeding grounds for the gastropod Concholepas concholepas. Marine Ecology Progress Series 215: 201–211.
Marin, V., M.E. Huntley, and B. Frost. 1986. Measuring feeding rates of pelagic herbivores: analysis of experimental design and methods. Marine Biology 93: 49–58.
Mehrbach, C., C.H. Culberson, J.E. Hawley, and R.N. Pytkowicz. 1973. Measurement of the apparent dissociation constants of carbonic acid in seawater at atmospheric pressure. Limnology and Oceanography 18: 897–907.
Meinshausen, M., S.J. Smith, K. Calvin, J.S. Daniel, M.L.T. Kainuma, J.-F. Lamarque, K. Matsumoto, S.A. Montzka, S. Raper, K. Riahi, A. Thomson, G.J.M. Velders, and D.P.P. van Vuuren. 2011. The RCP greenhouse gas concentrations and their extensions from 1765 to 2300. Climatic Change 109: 213–241.
Melzner, F., P. Stange, K. Trübenbach, J. Thomsen, I. Casties, U. Panknin, and M.A. Gutowska. 2011. Food supply and seawater pCO2 impact calcification and internal shell dissolution in the blue mussel Mytilus edulis. PLoS One 6: e24223.
Miller, A.W., A.C. Reynolds, C. Sobrino, and G.F. Riedel. 2009. Shellfish face uncertain future in high CO2 world: influence of acidification on oyster larvae calcification and growth in estuaries. PLoS One 4: e5661.
Navarro, J.M., R. Torres, K. Acuña, C. Duarte, P.H. Manriquez, M. Lardies, and V. Aguilera. 2013. Impact of medium-term exposure to elevated pCO2 levels on the physiological energetics of the mussel Mytilus chilensis. Chemosphere 90: 1242–1248.
Orr, J.C., V.J. Fabry, O. Aumont, L. Bopp, S.C. Doney, R.A. Feely, and A. Yool. 2005. Anthropogenic ocean acidification over the twenty-first century and its impact on calcifying organisms. Nature 437: 681–686.
Osorio, C. 2002. Moluscos marinos en Chile, especies de importancia económica. Facultad de Ciencias Universidad de Chile. 211 pp.
Osorio, C., and N. Bahamonde. 1968. Moluscos bivalvos en pesquerías chilenas. Biología Pesquera 3: 69–128.
Palmer, A.R. 1982. Growth in marine gastropods: a non-destructive technique for independently measuring shell and body weight. Malacologia 23: 63–73.
Pascal, P.Y., J.W. Fleeger, F. Galvez, and K.R. Carman. 2010. The toxicological interaction between ocean acidity and metals in coastal meiobenthic copepods. Marine Pollution Bulletin 60: 2201–2208.
Petersen, K.J., S. Bougrier, A.C. Smaal, P. Garen, S. Robert, J.E.N. Larsen, and E. Brummelhuis. 2004. Intercalibration of mussel Mytilus edulis clearance rate measurements. Marine Ecology Progress Series 267: 187–194.
Prado, L., and J.C. Castilla. 2006. The bioengineer Perumytilus purpuratus (Mollusca: Bivalvia) in central Chile: biodiversity, habitat structural complexity and environmental heterogeneity. Journal of the Marine Biological Association of the United Kingdom 86: 417–421.
Ramajo, L., A. Baltanás, R. Torres, P.H. Manríquez, A. Rodriguez-Navarro, and N. Lagos. 2013. Geographic variation in shell morphology of juvenile snails (Concholepas concholepas) along the physical-chemical gradient of the Chilean coast. Journal of Marine Biological Association of the United Kingdom. doi:10.1017/S0025315413000891.
Range, P., M.A. Chícharo, R. Ben-Hamadou, D. Piló, M.J. Fernandez-Reiriz, U. Labarta, and L. Chícharo. 2013. Impacts of CO2-induced seawater acidification on coastal Mediterranean bivalves and interactions with other climatic stressors. Regional Environmental Change 1–12.
Sabine, C.L., R.A. Feely, N. Gruber, R.M. Key, K. Lee, J.L. Bullister, and A.F. Rios. 2004. The oceanic sink for anthropogenic CO2. Science 305: 367–371.
Salisbury, J.E., D. Vandemark, C.W. Hunt, J.W. Campbell, W.R. McGillis, and W.H. McDowell. 2008. Seasonal observations of surface waters in two Gulf of Maine estuary-plume systems: relationships between watershed attributes, optical measurements and surface pCO2. Estuarine, Coastal and Shelf Science 77: 245–252.
Strathmann, R.R., and E. Leise. 1979. On feeding mechanisms and clearance rates of molluscan veligers. The Biological Bulletin 157: 524–535.
Strickland, J.D.H., and T.R. Parsons. 1968. Determination of reactive phosphorus. A practical handbook of seawater analysis. Fisheries Research Board of Canada, Bulletin 167: 49–56.
Stuardo, J. 1979. Sobre la clasificación, distribución y variación de Concholepas concholepas (Bruguiere, 1789): Un estudio de taxonomía Beta. Biología Pesquera Chile 12: 5–38.
Stumpp, M., S. Dupont, M.C. Thorndyke, and F. Melzner. 2011. CO2 induced seawater acidification impacts sea urchin larval development II: gene expression patterns in pluteus larvae. Comparative Biochemistry and Physiology-Part A: Molecular & Integrative Physiology 160: 320–330.
Stumpp, M., K. Trübenbach, D. Brennecke, M.Y. Hu, and F. Melzner. 2012. Resource allocation and extracellular acid–base status in the sea urchin Strongylocentrotus droebachiensis in response to CO2 induced seawater acidification. Aquatic Toxicology 110: 194–207.
Thiel, M., E.C. Macaya, E. Acuña, W.E. Arntz, H. Bastías, K. Brokordt, P. Camus, J.C. Castilla, L.R. Castro, M. Cortés, C.P. Dumont, R. Escribano, M. Fernández, J.A. Gajardo, C.F. Gaymer, I. Gomez, A.E. González, H.E. González, P.A. Haye, J.E. Illanes, J.L. Iriarte, D.L. Lancellotti, G. Luna-Jorquera, C. Luxoro, P.H. Manriquez, V. Marín, P. Muñoz, S.A. Navarrete, E. Pérez, E. Poulin, J. Sellanes, H.H. Sepúlveda, W. Stotz, F. Tala, A. Thomas, C.A. Vargas, J.A. Vásquez, and J.M.A. Vega. 2007. The Humboldt Current system of northern and central Chile oceanographic processes, ecological interactions and socioeconomic feedback. Oceanography and Marine Biology: An Annual Review 45: 195–344.
Thomsen, J., and F. Melzner. 2010. Seawater acidification does not elicit metabolic depression in the blue mussel Mytilus edulis. Marine Biology 157: 2667–2676. doi:10.1007/s00227-010-1527-0.
Thomsen, J., I. Castles, C. Pansch, A. Körtzinger, and F. Melzner. 2013. Food availability outweighs ocean acidification effects in juvenile Mytilus edulis: laboratory and field experiments. Global Change Biology 19: 1017–1027.
Toro, B., J.M. Navarro, and H. Palma-Fleming. 2003. Relationship between bioenergetics responses and organic pollutants in the giant mussel, Choromytilus chorus (Mollusca: Mytilidae). Aquatic Toxicology 63: 257–269.
Torres, R., S. Pantoja, N. Harada, H.E. González, G. Daneri, M. Frangopulos, and M. Fukasawa. 2011. Air-sea CO2 fluxes along the coast of Chile: from CO2 outgassing in central northern upwelling waters to CO2 uptake in southern Patagonian fjords. Journal of Geophysical Research 116, C09006.
Urabe, J., J. Togari, and J.J. Elser. 2003. Stoichiometric impacts of increased carbon dioxide on a planktonic herbivore. Global Change Biology 9: 818–825.
Vargas, C.A., D.A. Narváez, A. Piñones, S.A. Navarrete, N.A. Lagos. 2006. River plume dynamic infuences transport of barnacle larvae in the inner shelf o¡ central Chile. Journal of Marine Biological Association of United Kingdom 86: 1057–1065.
Vargas, C.A., M. de la Hoz, V. Aguilera, V. San Martín, P.H. Manríquez, J.M. Navarro, and N.A. Lagos. 2013. CO2-driven ocean acidification reduces larval feeding efficiency and change food selectivity in the mollusk Concholepas concholepas. Journal of Plankton Research 35(5): 1059–1068
Widdicombe, S. and J.L. Spicer. 2008. Predicting the impact of ocean acidification on benthic biodiversity: what can animal physiology tell us? Journal of Experimental Marine Biology and Ecology 366: 187–197.
Widdicombe, S., S.L. Dashfield, C.L. McNeill, H.R. Needham, A. Beesley, A. McEvoy, and J.A. Berge. 2009. Effects of CO2 induced seawater acidification on infaunal diversity and sediment nutrient fluxes. Marine Ecology Progress Series 379: 59–75.
Yuras, G., O. Ulloa, and S. Hormazabal. 2005. On the annual cycle of coastal and open ocean satellite chlorophyll off Chile (18–40°S). Geophysical Research Letters 32, L23604. doi:10.1029/2005GL023946.
Acknowledgments
We acknowledge the skillful help of all assistants involved in this research, especially to Haydee Müller, Karin Acuña, Loreto Mardones, and María Elisa Jara for their logistic support. This work was supported by the Proyecto Anillos ACT-132 by CONICYT (Comisión Nacional de Investigación Científica y Tecnológica) (http://www.eula.cl/anillos_acidificacion); Fondecyt Grant 1090624 (TOA-SPACE Project), and Millennium Nucleus Center for the Study of Multiple-Drivers on Marine Socio-Ecological Systems (MUSELS) funded by MINECON NC120086. Finally, the Millennium Institute of Oceanography (IMO) funded by MINECON IC120019 also supported this work during the final stage. CAV is supported by Red Doctoral REDOC.CTA, MINEDUC project UCO1202 at U. de Concepción.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Wayne S. Gardner
Rights and permissions
About this article
Cite this article
Vargas, C.A., Aguilera, V.M., Martín, V.S. et al. CO2-Driven Ocean Acidification Disrupts the Filter Feeding Behavior in Chilean Gastropod and Bivalve Species from Different Geographic Localities. Estuaries and Coasts 38, 1163–1177 (2015). https://doi.org/10.1007/s12237-014-9873-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-014-9873-7