Abstract
This study evaluated the antioxidant activity and total phenolic content (TPC) of Butia odorata fruit extracted with different solvents, as well as the phenolic composition using UHPLC-MS/MS analysis. In addition, the antitumor potential of B. odorata fruit against various cancer cell lines (Caco-2, HeLa, SiHa, C33a) was evaluated for the first time. The TPC and antioxidant activity (DPPH• and ORAC assays) varied according to the extraction solvents, with the highest values obtained for the methanol:water (80:20, v/v) extracts. Thirteen phenolic compounds were quantified in the ethanol:water (80:20, v/v) extracts of B. odorata fruit by UHPLC-MS/MS, with the main compounds identified as (+)-catechin (259 ± 18 mg kg−1), (−)-epicatechin (211 ± 12 mg kg−1) and rutin (161 ± 2 mg kg−1). Sinapic and ellagic acids, trans-resveratrol, naringenin, and apigenin were reported in B. odarata fruit for the first time. Furthermore, the B. odorata fruit showed antitumor activity against cervical cancer cell lines (SiHa and C33a), providing preliminary evidence for further assessment of its anticancer potential.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The study of phenolic compounds has grown in the last decades due to the recognition of their antioxidant properties (Shahidi and Ambigaipalan 2015). These compounds can inhibit or reduce oxidative damage caused by pro-oxidant species, such as reactive oxygen and nitrogen species, preventing several diseases associated with oxidative stress, such as cancer, diabetes, cardiovascular, and neurodegenerative diseases (Pisoschi and Pop 2015). Fruits are one of the main dietary sources of phenolic compounds, presenting potential health beneficial effects (Roleira et al. 2015; Shahidi and Ambigaipalan 2015).
Butia odorata (Barb. Rodr.) Noblick is a native fruit tree species from South America belonging to the Arecaceae family. In Brazil, it is predominantly found in Rio Grande do Sul State (Hoffmann et al. 2014). The fruit, known as jelly palm or butiá, is ovoid to depressed globose, ranging from yellow to orange in appearance, with an acid–sweet taste. The consumption of B. odorata fruit is greatly appreciated either fresh or processed into juice, ice cream, jam, and pulp (Ferrão et al. 2013; Hoffmann et al. 2014; Cruxen et al. 2017). Although the biological potential of the fruit is still underexploited, Hoffmann et al. (2017a, 2017b) and Vinholes et al. (2017) recently evaluated the in vitro antioxidant activity by different radical scavenging assays and Vinholes et al. (2017) also evaluated the in vitro anti-hyperglycemic potential of B. odorata fruit. Moreover, a few recent studies have shown that B. odorata fruit is a potential source of phenolic compounds, carotenoids, and vitamin C (Beskow et al. 2015; Hoffmann et al. 2017a). The main phenolic compounds reported in B. odorata fruit were rutin, (+)-catechin, (−)-epicatechin, hesperetin, isorhamnetin-O-rutinoside, and chlorogenic, hydroxybenzoic, and gallic acids (Beskow et al. 2015; Hoffmann et al. 2017a; Hoffmann et al. 2018).
Currently, reliable analysis of the biological potential of fruits and vegetables, which are complex matrices, are increasingly evidenced. This fact can be associated with the accessibility to advanced analytical techniques, such as ultra-high-performance liquid chromatography coupled to tandem mass spectrometry (UHPLC-MS/MS), as well as the use of cell culture assays. UHPLC-MS/MS is a powerful tool for both quantification and structural characterization of individual compounds in complex matrices and has been reported for determination of phenolic compounds in fruit extracts (Bataglion et al. 2015; Medina et al. 2017; Nicácio et al. 2017). Cell culture assays are valuable tools in screening extracts and compounds with potential biological effects, such as antitumor activity.
In this context, this work aimed to evaluate the antioxidant activity and total phenolic content (TPC) of B. odorata fruit extracted with different solvents, as well as to determine the phenolic compounds using UHPLC-MS/MS analysis. Additionally, the antitumor activity of B. odorata fruit was evaluated against colon (Caco-2) and cervical (HeLa, SiHa and C33a) cancer cell lines, for the first time.
Materials and Methods
Chemicals
Phenolic standards, quercetin, (+)-catechin, (−)-epicatechin, epicatechin gallate, kaempferol, rutin, naringenin, apigenin, luteolin, trans-resveratrol, and gallic, chlorogenic, p-coumaric, sinapic, p-hydroxybenzoic, protocatechuic, trans-cinnamic, syringic acids were obtained from Sigma-Aldrich (St. Louis, MO, USA); vanillic, ferulic, and ellagic acids were obtained from Fluka (Buchs, SG, Switzerland). All phenolic standards presented purity higher than 95%. Stock standard solutions of individual phenolic compounds (1 mg mL−1) were prepared in methanol and stored at − 18 °C, light protected. Working standard mixture solutions were prepared by appropriate dilution of the stock solutions in methanol. HPLC-grade methanol, acetonitrile, ethanol, and ethyl acetate were purchased from Sigma-Aldrich (St. Louis, MO, USA). Ultrapure water was obtained from a Milli-Q ultrapure water purification system (Millipore, Burlington, MA, USA). Formic acid used in the mobile phase was purchased from Sigma-Aldrich (St. Louis, MO, USA).
Folin–Ciocalteu’s phenol reagent, 2,2-diphenyl-1-picrylhydrazyl (DPPH•), 2,2-azobis(2-methylpropanimidamide) dihydrochloride (AAPH), (±)-6-hydroxy-2,5,7,8-tetramethylchromane-2-carboxylic acid (Trolox) were supplied from Sigma-Aldrich (St. Louis, MO, USA). Disodium fluorescein was obtained from Fluka (Buchs, SG, Switzerland), sodium carbonate from J.T.Baker (Phillipsburg, NJ, USA), and monobasic and dibasic potassium phosphate from Synth (Diadema, SP, Brazil).
Dulbecco’s modified Eagle’s medium (DMEM) and fetal bovine serum (FBS) were obtained from Invitrogen (Carlsbad, CA, USA); 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) and dimethyl sulphoxide (DMSO) were obtained from Sigma-Aldrich (St. Louis, MO, USA). Cell lines were supplied from American Type Culture Collection (Manassas, VA, USA).
Butia odorata fruit samples
B. odorata fruit samples (≈ 2 kg) were collected in Selbach, Rio Grande do Sul, Brazil (28° 37' 34" S and 52° 57' 12" W). The plant was identified by MSc. Kelen Pureza Soares and a voucher specimen (HUEM 30052) was deposited in the Herbarium of the State University of Maringá (HUEM). The ripe fruits were washed and manually depulped, rejecting only the seeds. Then, the fruits were frozen and lyophilized. Freeze-dried samples were homogenized, vacuum-packed, and stored at − 18 °C until the preparation of the extracts.
Extraction conditions
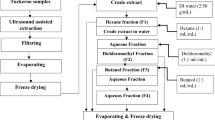
Freeze-dried sample (1 g) was extracted with 10 mL of the following solvents: (1) methanol; (2) methanol:water (80:20, v/v); (3) ethyl acetate; (4) acetonitrile; (5) acetonitrile:water (80:20, v/v); (6) ethanol; and (7) ethanol:water (80:20, v/v). Extraction was performed in 50-mL centrifuge tubes (Greiner Bio-one, Monroe, NC, USA) at 30 °C for 30 min, in an ultrasonic bath (Elmasonic P30H, Singen, BW, Germany). After extraction, the mixture was centrifuged at 6535g for 10 min, and the supernatant was filtered through 0.22-μm PTFE syringe filters. The extractions were performed in triplicate.
TPC determination
TPC was performed according to Singleton and Rossi (1965). Initially, 250 μL of the sample extracts obtained with the different extraction solvents and in an appropriate dilution (ethanol, methanol, methanol:water, ethanol:water, and acetonitrile:water (diluted 25 times); ethyl acetate and acetonitrile (without dilution)) were mixed with 250 μL Folin–Ciocalteu’s phenol reagent (diluted in distilled water (1:1, v/v)), 500 μL of saturated sodium carbonate solution, and 4 mL of distilled water. This mixture was kept in the dark for 25 min and was then centrifuged at 4529g for 10 min. The absorbance was measured at 725 nm using a spectrophotometer (Genesys 10, Thermo Scientific, Waltham, MA, USA). Solutions of gallic acid (0–180 mg L−1) were used to prepare the analytical curve (y = 0.0058x − 0.0179, r2 = 0.993). The results were expressed as milligrams of gallic acid equivalents (GAE) per gram of freeze-dried sample (mg GAE g−1).
Antioxidant activity
DPPH• radical scavenging activity
DPPH• assay was performed according to Ma et al. (2011). First, 25 μL of the sample extracts obtained with the different extraction solvents and in an appropriate dilution (ethanol, methanol, methanol:water, ethanol:water, and acetonitrile:water (diluted 5 times); ethyl acetate and acetonitrile (without dilution)) were added to 2 mL of a DPPH• methanolic solution (6.25 × 10−5 mol L−1). After 30 min of incubation in the dark, the absorbance of the mixture was measured at 517 nm using a spectrophotometer (Genesys 10, Thermo Scientific, Waltham, MA, USA). Trolox solutions (0–2000 μmol L−1) were used to prepare the analytical curve (y = − 2.98 × 10−4x + 0.689, r2 = 0.995). The results were expressed as micromoles of Trolox equivalents (TE) per gram of freeze-dried sample (μmol TE g−1).
Oxygen radical absorbance capacity assay
Oxygen radical absorbance capacity (ORAC) assay was performed according to Ou et al. (2001). Dilutions of extracts and the preparation of the assay reagents were done using phosphate buffer solution (monobasic potassium phosphate and dibasic potassium phosphate, 75 mmol L−1, pH = 7.4). First, 25 μL of the sample extracts obtained with the different extraction solvents and in an appropriate dilution (acetonitrile and ethyl acetate (diluted 100 times); ethanol (diluted 1000 times); methanol, ethanol:water, methanol:water, and acetonitrile:water (diluted 2500 times)) and 150 μL of fluorescein solution (4 nmol L−1) were transferred to a 96-well microplate. After incubation (37 °C for 5 min, light protected), 25 μL of a freshly prepared AAPH solution (161 mmol L−1) was added. Immediately, the fluorescence was measured, and further measurements were performed every 1 min during 30 min by a microplate reader at an excitation wavelength of 485 nm and an emission wavelength of 535 nm (Wallac Victor™, Perkin-Elmer, Waltham, MA, USA). Trolox solutions (0–50 μmol L−1) were used to prepare the analytical curve (y = 0.261x + 2.07, r2 = 0.993). The results were expressed as micromoles of Trolox equivalents (TE) per gram of freeze-dried sample (μmol TE g−1).
Determination of phenolic compounds by UHPLC-MS/MS
Phenolic compounds in B. odorata fruit were determined using an Acquity UPLC H-Class system (Waters, Milford, MA, USA) coupled to a Xevo TQD triple-quadrupole mass spectrometer, equipped with a Z spray™ source (Waters, Milford, MA, USA). The chromatographic separation was performed on an Acquity UPLC® BEH C18 column (50 mm × 2.1 mm i.d., 1.7 μm) operating at a flow rate of 0.15 mL min−1 and a temperature of 30 °C. The mobile phase used was (A) ultrapure water (acidified with 0.1% formic acid) and (B) methanol. Gradient elution was performed as follows: 0–0.01 min (10% B), 0.01–4 min (10–70% B), 4–8 min (70–100% B), 8–9 min (100% B), 9–11 min (100–50% B), 11–12.5 min (50–10% B), 12.5–15 min (10% B). The injection volume was 1.5 μL.
The mass spectrometer conditions were as follows: source temperature, 130 °C; capillary voltage, 3.0 kV; extractor voltage, 3.0 V; and desolvation gas temperature, 550 °C. Both the cone gas and desolvation gas were nitrogen and flows were 50 and 700 L h−1, respectively. Argon was used as the collision gas at a constant pressure of 3 × 10−3 mbar. Data acquisition and processing were done using MassLynx and QuanLynx software version 4.1 (Waters, Milford, MA, USA). The ionization source (ESI) was set in negative ion mode, and the mass spectrometer was operated in MS/MS mode, using selected reaction monitoring (SRM). Table S1 summarizes the precursor and product ions, associated with the instrumental parameters for fragmentation, and the retention times for the phenolic compound standards.
For the quantification of B. odorata fruit extract [ethanol:water (80:20, v/v)], analytical curves were constructed at five concentration levels by the standard addition method, due to the complexity of the sample. The results were expressed as milligrams per kilogram of freeze-dried sample (mg kg−1). The linear dynamic range and the calculation of the limit of detection (LOD) and limit of quantitation (LOQ) were carried out after knowing the endogenous amount of phenolic compounds present in B. odorata fruit. LOD and LOQ values were calculated by the signal-to-noise ratio defined as 3 and 10 times the baseline noise, respectively.
Antitumor activity
For antitumor activity, colon (Caco-2) and cervical (HeLa, SiHa and C33a) human cancer cell lines were evaluated. Non-tumor murine fibroblast (L929) and human keratinocyte (HaCaT) cells were used as controls in the assay. All cell lines were cultured in DMEM supplemented with 10% FBS and maintained at 37 °C in a humidified atmosphere with 5% CO2. The antitumor activity was performed using the MTT assay (Mosmann 1983), which measured the cytotoxic effects of the B. odorata extract in cancer cell lines. For this, cells (2.5 × 105 cells mL−1) were plated in a 96-well plate and incubated under the same conditions described above for 24 h. After incubation, the medium was replaced with treatment media containing different concentrations of B. odorata extract (0–1000 μg mL−1). Treatment media were prepared by evaporating the ethanol:water (80:20, v/v) from B. odorata extract under vacuum (35 °C) in a rotary evaporator (Fisatom, Brazil), then re-dissolved in DMSO and culture medium to obtain a final concentration of 0.1% (v/v) of DMSO. After treatment for 48 h, the medium was removed, and MTT solution (2 mg mL−1) was added, followed by incubation for 4 h. The formazan crystals formed from mitochondrial enzymatic metabolism of viable cells were dissolved in DMSO (150 μL per well) and the absorbance was measured using a microplate reader (Power Wave XS, BioTek, Winooski, VT, USA) at 570 nm. Antitumor activity was expressed as half maximal inhibitory concentration (IC50), defined as the concentration of the sample that inhibited 50% of cell growth compared to the control (untreated cells).
Statistical analysis
All experiments were performed in triplicate (n = 3) and expressed as mean ± standard deviation (SD). Data were submitted to one-way analysis of variance (ANOVA) with a comparison of means by Tukey’s test (p < 0.05), using Statistica 7.0 software (StatSoft, Tulsa, OK, USA).
Results and discussion
TPC and antioxidant activity
Considering the complexity involved in the in vivo action of antioxidants, different in vitro assays have been developed to estimate, in a simple experimental way, the antioxidant activity of plant materials. Several assays are frequently used for this purpose, such as DPPH•, ORAC, ABTS•+, and FRAP (López-Alarcón and Denicola 2013). In this study, the extracts of B. odorata fruit obtained with the different extraction solvents were submitted to the analysis of antioxidant activity by DPPH• and ORAC assays and TPC using Folin–Ciocalteu method (Fig. 1). The solvent employed in the extraction is one of the most important factors that affects the extraction efficiency since it is responsible for solubilizing the target compounds, allowing their extraction from the matrix (Naczk and Shahidi 2006; Meneses et al. 2013).
As shown in the Fig. 1, the TPC values ranged from 0.38 ± 0.04 to 19.3 ± 0.6 mg GAE g−1, and antioxidant activity values ranged from 3.47 ± 0.04 to 64 ± 7 μmol TE g−1 and from 2.4 ± 0.4 to 278 ± 15 μmol TE g−1, for DPPH• and ORAC assays, respectively. Methanol, methanol:water (80:20, v/v), acetonitrile:water (80:20, v/v), ethanol, and ethanol:water (80:20, v/v) B. odorata fruit extracts showed greater TPC (Fig. 1a) and antioxidant activity values (Fig. 1b) than acetonitrile and ethyl acetate B. odorata fruit extracts, as reported in other works (Nguyen et al. 2015; Onivogui et al. 2016; Rotta et al. 2017).
Among the B. odorata fruit extracts obtained with the different extraction solvents, methanol:water (80:20, v/v) extract showed the highest TPC (19.3 ± 0.6 mg GAE g−1) and antioxidant activity values by DPPH• assay (64 ± 7 μmol TE g−1). These results reinforce the antioxidant properties of B. odorata fruits, since other studies have shown the TPC in B. odorata fruit and its antioxidant activity by DPPH• assay (Hoffmann et al. 2017a, 2017b; Vinholes et al. 2017).
For the ORAC assay, methanol:water (80:20, v/v) and ethanol:water (80:20, v/v) extracts exhibited the highest antioxidant activity with values of 278 ± 15 and 265 ± 15 μmol TE g−1, respectively, and no significant difference was observed between these values. The ORAC assay is based on the scavenging of peroxyl radicals (ROO•), a relevant reactive oxygen species, which is produced as an intermediate of many in vivo oxidation reactions as a result of reaction of oxygen molecules with carbon-centered radicals. ROO• can cause oxidation of essential biomolecules, mainly proteins, playing an important role in the development of various human diseases (Leinisch et al. 2017). According to the study of Vinholes et al. (2017), B. odorata fruit also showed scavenging capacity against other physiologically reactive species, such as superoxide anion (O2•−), hydroxyl (•OH), and nitric oxide (•NO) radicals, evidencing its antioxidant potential.
UHPLC-MS/MS analysis
The chromatographic method developed was used for the analysis of 21 phenolic compounds (Fig. S1). First, the extraction of the selected phenolic compounds using different solvents was evaluated, as shown in Fig. 2. It can be observed that the amounts of the phenolic compounds extracted varied according to the solvents employed in the extraction step, and in general, methanol, methanol:water (80:20, v/v), acetonitrile:water (80:20, v/v), ethanol, and ethanol:water (80:20, v/v) solvents extracted greater amounts of the target phenolic compounds than acetonitrile and ethyl acetate solvents. Moreover, Fig. 2 reveals that ethanol:water (80:20, v/v) extracted the highest amounts of rutin and naringenin while sinapic, chlorogenic, and ellagic acids, trans-resveratrol, quercetin, luteolin, apigenin, and (−)-epicatechin were extracted with similar higher amounts than other solvents. Thus, ethanol:water (80:20, v/v) was the extraction solvent selected for the quantification of target phenolic compounds in B. odorata fruit. Noteworthy, the solvent that extracted the highest amounts of target phenolic compounds was not the same as that which gave the highest TPC value, i.e., methanol:water (80:20, v/v). This difference can happen because the Folin–Ciocalteu method is used for the determination of all phenolic compounds present in the sample. Moreover, the reagent used in this method does not react only with phenols but will react with any reducing substance (Tan and Lim 2015).
Extraction efficiency of phenolic acids (a) and flavonoids/stilbene (b) phenolic compounds found in B. odorata fruit. HYD: p-hydroxybenzoic acid; COU: p-coumaric acid; SIN: sinapic; FER: ferulic acid; ELA: ellagic acid; RES: trans-resveratrol; QUE: quercetin; LUT: luteolin; NAR: naringenin. API: apigenin; CAT: (+)-catechin; EPI: (−)-epicatechin; CLO: chlorogenic acid; RUT: rutin. Results are presented as mean ± SD (n = 3). Bars with different letters indicate means with significant differences (p < 0.05)
Table 1 shows the analytical curves used for the quantification of the phenolic compounds, as well as the linear range, correlation coefficients, LOD, and LOQ. The method developed presented good linearity, with correlation coefficients greater than 0.99 for all phenolic compounds analyzed. The LOQ values ranged between 0.25–400 μg kg−1.
Among the 21 phenolic compounds evaluated in this study, 13 were found at concentrations greater than the method LOQ and thus quantified, as shown in Table 2. Several of the phenolic compounds determined in the B. odorata fruit have already been reported in the literature (Beskow et al. 2015; Hoffmann et al. 2017a, 2017b; Hoffmann et al. 2018), except sinapic and ellagic acids, trans-resveratrol, naringenin, and apigenin, which were reported for the first time.
The major compounds found were (+)-catechin (259 ± 18 mg kg−1), (−)-epicatechin (211 ± 12 mg kg−1), and rutin (161 ± 2 mg kg−1). These compounds have several benefits to human health because they present several biological activities, including antioxidant, cytoprotective, anticarcinogenic, neuroprotective, and cardioprotective activities (Aron and Kennedy 2008; Costa and Silva 2014; Ganeshpurkar and Saluja 2016). Similar phenolic composition data are reported by Hoffmann et al. (2017a) that determined 13 phenolic compounds for methanolic extracts of B. odorata fruit, and identified (−)-epicatechin (463.0 ± 23.4 μg 100 g−1 fresh sample), rutin (426.7 ± 5.0 μg 100 g−1 fresh sample) and (+)-catechin (189.2 ± 23.9 μg 100 g−1 fresh sample) as the main phenolic compounds. Instead, Beskow et al. (2015) quantified 11 phenolic compounds in different genotypes of B. odorata fruit extracted and hydrolyzed using acidified methanol and found that gallic (117.10–234.29 mg 100 g−1 fresh sample) and hydroxybenzoic (106.52–150.14 mg 100 g−1 fresh sample) acids were the major phenolic compounds. Recently, Hoffmann et al. (2018) determined 22 phenolic compounds in four species of Butia (B. odorata, B. yatay, B. paraguayensis, and B. catarinensis) extracted with aqueous methanol (75%) and B. odorata, in general, showed the highest content of phenolic compounds, with the highest abundance as (+)-catechin and (−)-epicatechin.
Antitumor activity
Several studies have demonstrated that phenolic compounds are associated with significant antitumor activity (Al-Hazzani and Alshatwi 2011; Di Domenico et al. 2012; Roleira et al. 2015). Considering the results obtained in this study, especially the extensive phenolic composition (Table 2), the ethanol:water (80:20, v/v) extract of B. odorata was tested against colon (Caco-2) and cervical (HeLa, SiHa and C33a) cancer cell lines for its antitumor effects, as described in Table 3.
Antitumor activity varied across the different cancer cell lines (Table 3). SiHa and C33a showed the highest activity, with IC50 of 528 ± 7 and 411 ± 92 μg mL−1, respectively. Furthermore, the incubation of B. odorata extract with murine fibroblast (L929) and human keratinocyte (HaCaT) cells did not affect cellular viability (50% cytotoxity concentration, CC50 > 1000 μg mL−1), confirming that the responses obtained in cancer cells were not due to its toxic action.
Cervical cancer is the second leading cause of cancer death in women worldwide. The main agent promoting cervical cancer is the human papilloma viruses (HPVs), mainly HPV 16 and HPV 18 (Di Domenico et al. 2012; Moga et al. 2016). Among cervical cell lines used in this study, SiHa and HeLa cell lines contain HPV 18 and 16, respectively, whereas the C33a cell line did not include HPVs. The variation in antitumor activity among these cell lines may suggest different action mechanisms of B. odorata extract that should be further studied.
Other investigators have evidenced the growth–inhibition properties of several phenolic compounds identified in B. odorata fruit extract, such as (+)-catechin against cervical (SiHa) (Al-Hazzani and Alshatwi 2011) and prostate (LNCaP and PC3) cancer cells (Kampa et al. 2000); rutin against cervical (HeLa), colorretal (SW-480), and breast (MDA-MB-231) cancer cells (Alonso-Castro et al. 2013); and (−)-epicatechin against prostate (LNCaP and PC3) cancer cells (Kampa et al. 2000). Thus, phenolic compounds present in the B. odorata extract may contribute to the antitumor activity found for SiHa and C33a.
Conclusion
This study evidences the antioxidant potential of B. odorata fruit, which the total phenolic content and antioxidant activity values varied according to the solvents employed in the extraction step of fruit antioxidants. Moreover, 13 phenolic compounds were quantified in B. odorata fruit, which (+)-catechin, (−)-epicatechin, and rutin were found in the highest concentration levels, and sinapic and ellagic acids, trans-resveratrol, naringenin, and apigenin were reported in B. odorata fruit for the first time. In addition, B. odorata fruit demonstrated antitumor activity against two cervical cancer cell lines, SiHa and C33a, evaluated by the MTT assay, providing preliminary evidence for further assessment of its anticancer potential.
References
Al-Hazzani AA, Alshatwi AA (2011) Catechin hydrate inhibits proliferation and mediates apoptosis of SiHa human cervical cancer cells. Food Chem Toxicol 49:3281–3286
Alonso-Castro AJ, Domínguez F, García-Carrancá A (2013) Rutin exerts antitumor effects on nude mice bearing SW480 tumor. Arch Med Res 44:346–351
Aron PM, Kennedy JA (2008) Flavan-3-ols: nature, occurrence and biological activity. Mol Nutr Food Res 52:79–104
Bataglion GA, Silva FMA, Eberlin MN, Koolen HHF (2015) Determination of the phenolic composition from Brazilian tropical fruits by UHPLC–MS/MS. Food Chem 180:280–287
Beskow GT, Hoffmann JF, Teixeira AM, Fachinello JC, Chaves FC, Rombaldi CV (2015) Bioactive and yield potential of jelly palms (Butia odorata Barb. Rodr.). Food Chem 172:699–704
Costa R, Silva L (2014) Health benefits of nongallated and gallated flavan-3-ols: a prospectus. In: Kinsey AL (ed) Recent advances in gallate research. Nova Science Publishers, New York, pp 1–191
Cruxen CES, Hoffmann JF, Zandoná GP, Fiorentini AM, Rombaldi CV, Chaves FC (2017) Probiotic butiá (Butia odorata) ice cream: development, characterization, stability of bioactive compounds, and viability of Bifidobacterium lactis during storage. LWT – Food Sci Technol 75:379–385
Di Domenico F, Foppoli C, Coccia R, Perluigi M (2012) Antioxidants in cervical cancer: chemopreventive and chemotherapeutic effects of polyphenols. Biochim Biophys Acta (BBA) - Mol Basis Dis 1822:737–747
Ferrão TS, Ferreira DF, Flores DW, Bernardi G, Link D, Barin JS, Wagner R (2013) Evaluation of composition and quality parameters of jelly palm (Butia odorata) fruits from different regions of Southern Brazil. Food Res Int 54:57–62
Ganeshpurkar A, Saluja AK (2016) The pharmacological potential of rutin. Saudi Pharm J 25:149–164
Hoffmann JF, Barbieri RL, Rombaldi CV, Chaves FC (2014) Butia spp. (Arecaceae): An overview. Sci Hortic 179:122–131
Hoffmann JF, Zandoná GP, Santos PS, Dallmann CM, Madruga FB, Rombaldi CV, Chaves FC (2017a) Stability of bioactive compounds in butiá (Butia odorata) fruit pulp and nectar. Food Chem 237:638–644
Hoffmann JF, Carvalho IR, Barbieri RL, Rombaldi CV, Chaves FC (2017b) Butia spp. (Arecaceae) LC-MS-based metabolomics for species and geographical origin discrimination. J Agric Food Chem 65:523–532
Hoffmann JF, Crizel RL, Madruga NA, Barbieri RL, Rombaldi CV, Chaves FC (2018) Flavan-3-ol, flavanone, flavone, flavonol, phenolic acid, and stilbene contents of four Butia species (Arecaceae). Fruits 73:125–137
Kampa M, Hatzoglou A, Notas G, Damianaki A, Bakogeorgou E, Gemetzi C, Castanas E (2000) Wine antioxidant polyphenols inhibit the proliferation of human prostate cancer cell lines. Nutr Cancer 37:223–233
Leinisch F, Mariotti M, Rykaer M, Lopez-Alarcon C, Hägglund P, Davies MJ (2017) Peroxyl radical- and photo-oxidation of glucose 6-phosphate dehydrogenase generates cross-links and functional changes via oxidation of tyrosine and tryptophan residues. Free Radic Biol Med 112:240–252
López-Alarcón C, Denicola A (2013) Evaluating the antioxidant capacity of natural products: A review on chemical and cellular-based assays. Anal Chim Acta 763:1–10
Ma X, Wu H, Liu L, Yao Q, Wang S, Zhan R, Xing S, Zhou Y (2011) Polyphenolic compounds and antioxidant properties in mango fruits. Sci Hortic 129:102–107
Medina S, Collado-González J, Ferreres F, Londoño-Londoño J, Jiménez-Cartagena C, Guy A, Durand T, Galano J, Gil-Izquierdo A (2017) Quantification of phytoprostanes – bioactive oxylipins – and phenolic compounds of Passiflora edulis Sims shell using UHPLC-QqQ-MS/MS and LC-IT-DAD-MS/MS. Food Chem 229:1–8
Meneses NGT, Martins S, Teixeira JA, Mussatto SI (2013) Influence of extraction solvents on the recovery of antioxidant phenolic compounds from brewer’s spent grains. Sep Purif Technol 108:152–158
Moga MA, Dimienescu OG, Arvatescu CA, Mironescu A, Dracea L, Ples L (2016) The role of natural polyphenols in the prevention and treatment of cervical cancer–an overview. Molecules 21:1055–1087
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Naczk M, Shahidi F (2006) Phenolics in cereals, fruits and vegetable: occurrence, extraction and analysis. J Pharm Biomed Anal 41:1523–1542
Nguyen VT, Bowyer MC, Vuong QV, Altena IAV, Scarlett CJ (2015) Phytochemicals and antioxidant capacity of Xao tam phan (Paramignya trimera) root as affected by various solvents and extraction methods. Ind Crop Prod 67:192–200
Nicácio AE, Rotta EM, Boeing JS, Barizão EO, Kimura E, Visentainer JV, Maldaner L (2017) Antioxidant activity and determination of phenolic compounds from Eugenia involucrata DC. fruits by UHPLC-MS/MS. Food Anal Methods 10:2718–2728
Onivogui G, Letsididi R, Diaby M, Wang L, Song Y (2016) Influence of extraction solvents on antioxidant and antimicrobial activities of the pulp and seed of Anisophyllea laurina R. Br. ex Sabine fruits. Asian Pac J Trop Biomed 6:20–25
Ou B, Hampsch-Woodill M, Prior RL (2001) Development and validation of an improved oxygen radical absorbance capacity assay using fluorescein as the fluorescent probe. J Agric Food Chem 49:4619–4626
Pisoschi AM, Pop A (2015) The role of antioxidants in the chemistry of oxidative stress: a review. Eur J Med Chem 97:55–74
Roleira FMF, Tavares-da-Silva EJ, Varela CL, Costa SC, Silva T, Garrido J, Borges F (2015) Plant derived and dietary phenolic antioxidants: anticancer properties. Food Chem 183:235–258
Rotta EM, Haminiuk CWI, Maldaner L, Visentainer JV (2017) Determination of antioxidant activity and phenolic compounds of Muntingia calabura Linn. peel by HPLC-DAD and UPLC-ESI-MS/MS. Int J Food Sci Technol 52:954–963
Shahidi F, Ambigaipalan P (2015) Phenolics and polyphenolics in foods, beverages and spices: Antioxidant activity and health effects – a review. J Funct Foods 18:820–897
Singleton VL, Rossi JAJr (1965) Colorimetry of total phenolics with phosphomolybdic–phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Tan JBL, Lim YY (2015) Critical analysis of current methods for assessing the in vitro antioxidant and antibacterial activity of plant extracts. Food Chem 172:814–822
Vinholes J, Lemos G, Barbieri RL, Franzon RC, Vizzotto M (2017) In vitro assessment of the antihyperglycemic and antioxidant properties of araçá, butiá and pitanga. Food Biosci 19:92–100
Acknowledgments
The authors thank MSc. Kelen Pureza Soares and the Herbarium of the State University of Maringá for the identification of the B. odorata plant.
Funding
This work received financial support from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (Capes) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 67 kb)
Rights and permissions
About this article
Cite this article
Boeing, J.S., Barizão, É.O., Rotta, E.M. et al. Phenolic Compounds from Butia odorata (Barb. Rodr.) Noblick Fruit and Its Antioxidant and Antitumor Activities. Food Anal. Methods 13, 61–68 (2020). https://doi.org/10.1007/s12161-019-01515-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-019-01515-6