Abstract
Purpose
Ovarian cancer (OC) is a common malignancy, and IFN-γ, a multifunctional cytokine, is unveiled to impede the multiplication and enhance apoptosis in diverse tumor cells in previous research. Nonetheless, its function and mechanism in OC are blurred.
Methods
OC cell lines SKOV3 and OVCAR3 were dealt with different concentrations (0–40 ng/ml) of IFN-γ. CCK-8 experiment was utilized to examine cell multiplication; Flow cytometry was executed to detect apoptosis and cell cycle; Wound healing assay was utilized to detect cell migration; and Transwell experiment was implemented to examine cell invasion. qRT-PCR analysis was applied to detect STAT5, STAT3, JAK2 and JAK3 mRNA expression in OC cell lines. Western blot experiment was applied to detect the protein and phosphorylation levels of SOCS1, STAT5 and STAT3.
Results
IFN-γ suppressed OC cell multiplication in a concentration-dependent manner. Relative to the control group, IFN-γ restrained OC cell migration, invasion, enhanced apoptosis and prevented cell transformation from G0/G1 to S phase. Further analysis revealed that IFN-γ up-modulated SOCS1 expression and impeded STAT5 and STAT3 protein phosphorylation levels, and knockdown of SOCS1 partially counteracted the inhibitory effect of IFN-γ on STAT5 and STAT3 protein phosphorylation levels.
Conclusion
IFN-γ represses OC progression by facilitating SOCS1 to suppress STAT3 and STAT5 protein phosphorylation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ovarian cancer (OC) is a prevalent malignancy. OC patients are usually asymptomatic in the early stages and there is a lack of effective screening and diagnostic methods. As a result, approximately 70% of OC patients have progressed to advanced disease stage by the time of diagnosis [1]. Recently, despite significant advances in surgery and chemotherapy, the 5-year survival rate of patients is still less than 50% [2]. Given the limited options for targeted therapies for OC, there is a need to find novel targets for OC treatment [3].
The immune system helps to destroy cancer cells and exogenous infections. Interferon gamma (IFN-γ) is a multifunctional cytokine produced mainly by T-helper cells, cytotoxic T cells, natural killer cells, and macrophages that enhances apoptosis and lysis of tumor cells [4]. INF-γ has immunomodulatory, antiviral and antitumor effects [5], and can restrain cell multiplication and induce apoptosis in different tumors, such as non-small cell lung cancer (NSCLC) and melanoma [6]. In OC, IFN-γ is unearthed to impede OC cell multiplication and enhances apoptosis through in vivo and in vitro experiments [7]. In addition, IFN-γ receptor overexpression is linked to a better prognosis in OC patients [8]. Nonetheless, the role and modulatory mechanisms of INF-γ in OC are undefined.
IFN-γ functions by binding to the cell surface receptor IFN-γ receptor (IFNGR), thereby activating The Janus kinase (JAK)–signal transducer of activators of transcription (STAT) signaling pathway. The JAK–STAT signaling pathway consists of three main components, namely tyrosine kinase-associated receptor, tyrosine kinase JAK, and transcription factor STAT, which is implicated in diverse biological processes such as cell multiplication, differentiation, apoptosis, and immune regulation [9]. Reportedly, the JAK–STAT signaling pathway participates in the development of multiple tumors, such as gastric, breast, and colorectal cancers [10]. In OC, the activation of the JAK–STAT signaling pathway is demonstrated to be linked to OC progression [11]. Therefore, inhibition of the JAK–STAT signaling pathway may be one of the strategies for OC treatment. Nonetheless, it is blurred whether IFN-γ can be implicated in OC progression by modulating the JAK–STAT signaling pathway.
Suppressor of cytokine signaling (SOCS) family proteins are inhibitors of the JAK–STAT signaling pathway [12]. SOCS molecules are unearthed to negatively modulate this signaling pathway by modulating STAT protein tyrosine phosphorylation levels. Many cytokines, such as the T-helper (Th) 1cytokines IFN-γ and IL-2, and the Th2 cytokine TNF-α can rapidly induce SOCS [13]. SOCS1 overexpression in OC patients is studied to be associated with better patient prognosis [14]. Nevertheless, it is blurred whether INF-γ can modulate SOCS1 in OC.
This work probed the role of IFN-γ in the multiplication, migration, invasion, apoptosis and cell cycle regulation of OC cell lines and identified its mechanism. IFN-γ was unveiled to restrain JAK–STAT signaling pathway through up-modulation of SOCS1 protein to suppress OC cell multiplication, migration and invasion, and to enhance apoptosis and prevent cell transformation from G0/G1 phase to S phase.
Methods
Cell culture
Two OC cell lines (SKOV3 and OVCAR3) were procured from Shanghai Cell Bank, Chinese Academy of Sciences (Shanghai, China). All cells were cultured in DMEM medium containing 10% FBS, 100 U/mL penicillin, and 100 U/mL streptomycin and incubated at 37 °C in 5% CO2.
Cell transfection
SOCS1 short hairpin RNA and negative control shRNA (sh-NC) were designed and synthesized by JRDUN Biotechnology co. (Shanghai). The above vectors were transfected into cells using a lentiviral vector (Addgene).
qRT-PCR analysis
Total RNA was isolated from the cell lines using TRIzol reagent (Invitrogen, Shanghai, China). RNA was reverse transcribed to cDNA using AMV reverse transcriptase (Fermentas, USA), and finally SYBRmix Ex Taq II (Takara, Dalian, China) and StepOnePlus real-time PCR system (Thermo Fisher Scientific, Inc., Waltham, MA. USA) for qRT-PCR analysis. The relative expression of RNA genes was calculated using the 2−ΔΔCt method, with GAPDH as an internal reference. The primer sequences were as follows:
SOCS1, 5ʹ-CGACACGCACTTCCGCACATT-3' (forward) and 5ʹ-TGGGTCCCGAGGCCATCTTCAC-3ʹ (reverse); STAT5, 5ʹ-TCCCTATAACATGTACCCACA-3ʹ (forward), and 5ʹ-ATGGTCTCATCCAGGTCGAA-3ʹ (reverse); JAK2, 5ʹ-ATCCACCCAACCATGTCTTCC-3ʹ (forward), and 5ʹ-ATTCCATGCCGATAGGCTCTG-3ʹ (reverse); JAK3, 5ʹ-CCTGATCGTGGTCCAGAGAG-3ʹ (forward), and 5ʹ-GCAGGGATCTTGTGAAATGTCAT-3ʹ (reverse); STAT3, 5ʹ-ATCACGCCTTCTACAGACTGC-3ʹ (forward), and 5'-CATCCTGGAGATTCTCTACCACT-3ʹ (reverse); GAPDH, 5′-CTTTGGTATCGTGGAAGGACTC-3′ (forward) and 5′-GTAGAGGCAGGGATGATGTTCT-3ʹ (reverse).
CCK-8 experiment
The transfected cells were treated with trypsin, inoculated with 1 × 103 cells/well in a 96-well plate and incubated in the water-saturated carbon dioxide incubator (5%). 10 μL of CCK-8 reagent (Dojindo Laboratories, Kumamoto, Japan) was supplemented to each well at 12, 24 and 48 h of inoculation and incubated for 2 h at 37 °C. The absorbance at 450 nm/well was subsequently measured using a microplate reader (Bio-Rad, Hercules, CA, USA). The inhibiting rate of cell proliferation (%) = (OD of control group − OD of experimental group)/(OD of control group − OD of blank group) × 100%.
Apoptosis and cell cycle analysis
Cells were trypsinized with trypsin and inoculated on 24-well plates and collected by centrifugation after 48 h. Cells were resuspended using PBS solution and counted using the Annexin V-Propidium Iodine (PI) staining kit (Becton Dickinson, for apoptosis) or PI staining kit (for cycle, Beyotime) in a FACSCalibur cytometer (FACSCalibur, BD Biosciences) to detect apoptosis rate and cell cycle.
Wound healing experiment
For the wound healing experiment, the OC cells were transfected and planted in 12-well plates till confluence, and then scratched with a micropipette tip. Cell migration images were captured at 0 h and 48 h after scratching.
Transwell experiment
Cell invasion experiments were executed using 24-well Transwell chambers (Corning Incorporated, Corning, MA, USA) pre-coated with Matrigel (BD Bioscience, Franklin Lakes, NJ, USA) on the bottom of the upper compartment. 1 × 105 cells were planted in 150 µL of serum-free medium in the upper chamber of Transwell chamber. The lower compartment was supplemented with 600 μL complete medium and incubated at 37 °C for 24 h. The cells in the upper compartment were wiped off, and the cells on the lower surface of the upper compartment were fixed using 95% ethanol and stained with 0.1% crystal violet for 25 min. Counting was implemented using a microscope (Olympus, Japan).
Western blot
The total protein was isolated from the cells using RIPA lysis buffer (Beyotime, Shanghai, China) and the protein was quantified using the BCA detecting kit (Beyotime, Shanghai, China). 30 μg of protein specimens was spiked in 10% SDS-PAGE (Sigma Aldrich, St. Louis, MO, USA) after electrophoresis and transferred to PVDF membranes (Millipore, Billerica, MA, USA). Subsequently, the PVDF membrane was blocked with 5% skimmed milk and anti-SOCS1 (1:1000, ab9870, Abcam Inc., Cambridge, UK), anti-STAT5 (1:1000, ab230670, Abcam Inc., Cambridge, UK), anti-p-STAT5(1:1000, ab32364, Abcam Inc., Cambridge, UK), anti-STAT3(1:1000, ab68153, Abcam Inc., Cambridge, UK), anti-p-STAT3(1:1000, ab76315, Abcam Inc., Cambridge, UK), anti-GAPDH (1:1000, ab9485, Abcam Inc., Cambridge, UK) were supplemented to incubate overnight at 4 °C. Thereafter, the membrane was incubated with the horseradish peroxidase-conjugated secondary antibody (1:2000, ab150077, Abcam Inc., Cambridge, UK) for 1 h, and the ECL luminescence reagent (Thermo Fisher Scientific, Waltham, MA, USA) was utilized to detect protein bands.
Statistical analysis
Data were collected after all assays were carried out 3 times, and the results were expressed as mean ± standard deviation. Statistical analysis and plotting were executed using GraphPad Prism V5.0 (GraphPad Software, Inc., La Jolla, CA, USA). Student's t-test was utilized for analysis of differences between two groups, and one-way ANOVA was applied for analysis of differences between three or more groups. * P < 0.05, ** P < 0.01 and *** P < 0.001 signified statistical significance.
Results
IFN-γ impedes OC cell multiplication, migration and invasion
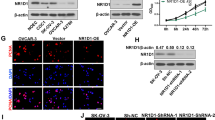
Analysis of the GEPIA database revealed that IFN-γ overexpression was linked to a better prognosis in OC patients (P = 0.013, Fig. 1A). To probe the function of IFN-γ in OC, SKOV3 and OVCAR3 cell lines were dealt with different concentrations of IFN-γ (0, 5, 10, 20, 40 ng/ml). CCK-8 results revealed that 20, 40 ng/ml IFN-γ remarkably impeded the multiplication of SKOV3 and OVCAR3 cells relative to 0 ng/ml IFN-γ, and IFN-γ restrained cell multiplication in a dose-dependent manner (Fig. 1B, C). A concentration of 40 ng/ml of IFN-γ was selected for follow-up experiments. Wound healing experiment demonstrated that IFN-γ remarkably impeded OC cell migration relative to the control group (Fig. 1D, E); Transwell experiment validated that the cell invasion was markedly lower in the IFN-γ-treated group relative to the control group (Fig. 1F). The above data unearthed that IFN-γ impeded OC cell multiplication, migration and invasion.
IFN-γ restrains the multiplication, migration and invasion of ovarian cancer cells. A GEPIA database analyzed the relationship between IFN-γ expression and prognosis in OC; B, C CCK-8 experiment verified the multiplication of OC cells SKOV3 and OVCAR3 after treatment with different concentrations of IFN-γ (0–40 ng/ml); D, E Wound healing assay was employed to analyze the effect of IFN-γ on the migration of SKOV3 and OVCAR3 cells; F Transwell assay was utilized to analyze the effect of IFN-γ on SKOV3 and OVCAR3 cell invasion. * P < 0.05; ** P < 0.01; *** P < 0.001
IFN-γ restrains apoptosis and modulates cell cycle in OC cells
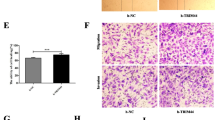
The role of IFN-γ on apoptosis and cell cycle in SKOV3 and OVCAR3 cells was studied using flow cytometry. The data unveiled that IFN-γ remarkably repressed apoptosis of SKOV3 and OVCAR3 cells (Fig. 2A, B). Similarly, the G0/G1 phase of the cell cycle was remarkably long and the S phase was remarkably shorter in the IFN-γ-treated group relative to the untreated group (Fig. 3C, D). Thus, IFN-γ impeded OC cell apoptosis and prevented cell transformation from G0/G1 phase to the S phase.
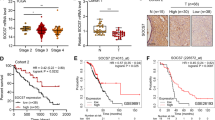
IFN-γ suppresses the malignant phenotype of OC by augmenting SOCS1 expression. A Correlation of IFN-γ and SOCS1 expression in OC was analyzed by GEPIA database; B, C qRT-PCR and Western blot experiments were applied to detect the effects of IFN-γ on SOCS1 mRNA and protein in SKOV3 and OVCAR3 cells; D, E qRT-PCR and Western blot assays were utilized to detect SOCS1 mRNA and protein expression after transfection of SKOV3 and OVCAR3 cells with SOCS1 shRNA; F, G CCK-8 was adopted to detect cell multiplication after SKOV3 and OVCAR3 cells were transfected with SOCS1 shRNA and treated with IFN-γ in SKOV3 and OVCAR3 cells; K Wound healing assay was implemented to detect cell migration after transfection of SKOV3 and OVCAR3 cells with SOCS1 shRNA and treatment of SKOV3 and OVCAR3 cells with IFN-γ; L Transwell experiment was executed to detect cell invasion after transfection of SKOV3 and OVCAR3 cells with SOCS1 shRNA and treatment of SKOV3 and OVCAR3 cells with IFN-γ; M Flow cytometry was utilized to detect the apoptosis rate of SKOV3 and OVCAR3 cells after transfection of SOCS1 shRNA and IFN-γ treatment of SKOV3 and OVCAR3 cells; * P < 0.05; ** P < 0.01; *** P < 0.001.
IFN-γ suppresses the malignant phenotype of OC by enhancing SOCS1 expression
Analysis of the correlation between IFN-γ and SOCS1 using the GEPIA database revealed that SOCS1 mRNA and IFN-γ were positively correlated in OC tissues (Fig. 3A). Treatment of ovarian cancer cell lines SKOV3 and OVCAR3 with IFN-γ revealed that IFN-γ remarkably augmented SOCS1 mRNA expression and protein (Fig. 3B, C). Subsequently, it was verified whether IFN-γ could exert a tumor-suppressive effect in OC through SOCS1. SOCS1 shRNA or control shRNA was transfected into SKOV3 and OVCAR3 cells, respectively, and a SOCS1 under-expression model was successfully constructed (Fig. 3D, E). Functional experiment demonstrated that knockdown of SOCS1 partially counteracted the inhibitory effects of IFN-γ on the multiplication migration and invasion of SKOV3 and OVCAR3 cells, and the promotion of apoptosis (Fig. 3F, M). Thus, the above data confirmed that IFN-γ impeded the malignant phenotype of OC by augmenting SOCS1 expression.
IFN-γ impedes JAK/STAT signaling pathway via SOCS1
SOCS1 is an inhibitor of the JAK/STAT signaling pathway. To verify whether IFN-γ could affect the JAK/STAT signaling pathway through the modulation of SOCS1, OC cell lines were treated with IFN-γ. qRT-PCR experiments confirmed that STAT5 and STAT3 mRNA were substantially reduced, while other protein expression of the JAK/STAT pathway such as JAK2, JAK3 was barely affected by IFN-γ. Western blot experiments unearthed that p-STAT5 and p-STAT3 expression was remarkably reduced, while STAT5 and STAT3 expression was not remarkably altered. Compensation experiments validated that knockdown of SOCS1 partially counteracted the inhibitory effect of IFN-γ on p-STAT5 and p-STAT3 expression. Thus, the above data validated that in OC, IFN-γ impeded p-STAT3 and p-STAT5 expression by enhancing SOCS1 (Fig. 4).
IFN-γ restrains JAK/STAT signaling pathway via SOCS1. A Effect of IFN-γ treatment on STAT5, JAK2, JAK3 and STAT3 mRNA expression in OVCAR3 cells was examined by qRT-PCR; B Western blot assay was utilized to detect STAT5, p-STAT5, STAT3 and p-STAT3 protein expression after IFN-γ treatment of OVCAR3 cells; C Western blot was conducted to detect p-STAT5 and p-STAT3 protein expression in OVCAR3 cells transfected with SOCS1 shRNA and treated with IFN-γ. ** P < 0.01; *** P < 0.001
Discussion
IFN-γ is unveiled to have antitumor activity, and many studies and clinical trials have demonstrated that IFN-γ can be used as a target for tumor therapy [15]. In cervical cancer, IFN-γ is unveiled to facilitate autophagy in cervical cancer cells through induction of IDO1 expression [16]. In gastric cancer, IFN-γ is unearthed to remarkably up-modulate in tumor tissue relative to paracancerous tissue [17]. In hepatocellular carcinoma, IFN-γ restrains tumor growth and enhances tumor cell apoptosis [18]. In OC, IFN-γ treatment is unveiled to cause depolarization of the mitochondrial membrane, release of cytochrome C into the cytoplasm and activation of caspase 9, which has antiproliferative effects [19]. GEPIA analysis unearths that IFN-γ overexpression is linked to better survival of OC patients. Further analysis reveals that IFN-γ represses OC cell multiplication, migration and invasion, and induces apoptosis and facilitates cell transformation from G0/G1 phase to S phase. Thus, IFN-γ exhibits antitumor activity in OC in vitro experiments.
Dysregulation of the JAK–STAT signaling pathway may result in diverse diseases such as skin diseases, cancer, and immune system disorders [20]. JAK/STAT signaling pathway is aberrantly activated in OC, and it’s associated with development and progression of OC. Previous study confirmed that inhibition of JAK/STAT3 signaling pathways could significantly suppress OC tumor growth and decreased peritoneal dissemination in vivo [21]. Furthermore, activation of JAK/STAT signaling accelerates tumorigenesis in OC [22]. In addition, research has shown that IFN-γ could inhibit JAK/STAT signaling, thus leading to decreases of the glioma cells invasiveness and tumorigenesis [23]. STATs are a member of cytoplasmic transcription factors whose role is to transmit signals from the cell membrane to the nucleus [24]. STAT3 and STAT5 are crucial in tumor formation by enhancing cell multiplication, impeding apoptosis, inducing angiogenesis, facilitating invasion, and restraining antitumor immunity [25]. In OC, inhibition of STAT3 and STAT5 impedes the malignant behavior of the cells, indicating that STAT3 and STAT5 may be latent therapeutic targets in OC [26]. Additionally, the activation of the STAT3 pathway and pY-STAT3 overexpression is unveiled to be linked to higher clinical stage, lower differentiation, lymph node metastasis, and shorter patient survival in OC patients [27]. Besides, STAT5 is related to cisplatin resistance in OC [28]. In this work, IFN-γ was unearthed to suppress STAT3 and STAT5 protein phosphorylation in OC.
SOCS interacts with cytokines such as IFN-γ and is the most potent inhibitor of the JAK/STAT pathway [29, 30]. Numerous studies have demonstrated SOCS1 play vital roles in various malignant processes and SOCS1 gene silencing was observed in human hepatic carcinoma, multiple melanoma, gastric cancer and pancreatic cancer [31]. In OC, previous study has demonstrated that low SOCS1 mRNA expression level was significantly associated with unfavorable prognosis in OC patients and SOCS1 may serve as prognostic biomarkers for OC patients [14]. A significant increase in SOCS1 protein expression was observed in human villous trophoblasts cells treated with IFN-γ [32]. In this work, SOCS1 was unveiled to be remarkably elevated in IFN-γ-treated OC cell lines. Further identification of the function of SOCS1 in the modulation of the malignant phenotype of OC by IFN-γ revealed that knockdown of SOCS1 partially counteracted the inhibitory effects of IFN-γ on the multiplication, migration, and invasion of OC cells as well as the modulatory effects on apoptosis and cell cycle. Hence, IFN-γ exerts oncogenic effects in OC by augmenting SOCS1 expression, and knockdown of SOCS1 partially counteracted the inhibitory effect of IFN-γ on STAT3 and STAT5 protein phosphorylation. Hence, the above data imply that IFN-γ restrains the JAK/STAT signaling pathway through SOCS1.
In conclusion, IFN-γ is revealed to be high in OC patient tissues and cell lines, and IFN-γ impeded OC progression by enhancing SOCS1 to repress STAT3 and STAT5 protein phosphorylation.
References:
Matulonis UA, Sood AK, Fallowfield L, Howitt BE, Sehouli J, Karlan BY. Ovarian cancer. Nat Rev Dis Primers. 2016;2:16061.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J Clin. 2021;71:7–33.
DiSilvestro P, Alvarez SA. Maintenance treatment of recurrent ovarian cancer: Is it ready for prime time? Cancer Treat Rev. 2018;69:53–65.
Gomez-Lomeli P, Bravo-Cuellar A, Hernandez-Flores G, et al. Increase of IFN-gamma and TNF-alpha production in CD107a + NK-92 cells co-cultured with cervical cancer cell lines pre-treated with the HO-1 inhibitor. Cancer Cell Int. 2014;14:100.
Kursunel MA, Esendagli G. The untold story of IFN-gamma in cancer biology. Cytokine Growth Factor Rev. 2016;31:73–81.
Jorgovanovic D, Song M, Wang L, Zhang Y. Roles of IFN-gamma in tumor progression and regression: a review. Biomark Res. 2020;8:49.
Wall L, Burke F, Barton C, Smyth J, Balkwill F. IFN-gamma induces apoptosis in ovarian cancer cells in vivo and in vitro. Clin Cancer Res. 2003;9:2487–96.
Duncan TJ, Rolland P, Deen S, Scott IV, Liu DT, Spendlove I, Durrant LG. Loss of IFN gamma receptor is an independent prognostic factor in ovarian cancer. Clin Cancer Res. 2007;13:4139–45.
O’Shea JJ, Schwartz DM, Villarino AV, Gadina M, McInnes IB, Laurence A. The JAK-STAT pathway: impact on human disease and therapeutic intervention. Annu Rev Med. 2015;66:311–28.
Owen KL, Brockwell NK, Parker BS. JAK-STAT signaling: a double-edged sword of immune regulation and cancer progression. Cancers (Basel). 2019;11:2002.
Ruan Z, Yang X, Cheng W. OCT4 accelerates tumorigenesis through activating JAK/STAT signaling in ovarian cancer side population cells. Cancer Manag Res. 2019;11:389–99.
Croker BA, Kiu H, Nicholson SE. SOCS regulation of the JAK/STAT signaling pathway. Semin Cell Dev Biol. 2008;19:414–22.
Li Y, Chu N, Rostami A, Zhang GX. Dendritic cells transduced with SOCS-3 exhibit a tolerogenic/DC2 phenotype that directs type 2 Th cell differentiation in vitro and in vivo. J Immunol. 2006;177:1679–88.
Yang M, Chen H, Zhou L, Huang X, Su F, Wang P. Identification of SOCS family members with prognostic values in human ovarian cancer. Am J Transl Res. 2020;12:1824–38.
Mojic M, Takeda K, Hayakawa Y. The dark side of IFN-gamma: its role in promoting cancer immunoevasion. Int J Mol Sci. 2017;19:89.
Yang SL, Tan HX, Niu TT, et al. The IFN-gamma-IDO1-kynureine pathway-induced autophagy in cervical cancer cell promotes phagocytosis of macrophage. Int J Biol Sci. 2021;17:339–52.
Su Z, Sun Y, Zhu H, et al. Th17 cell expansion in gastric cancer may contribute to cancer development and metastasis. Immunol Res. 2014;58:118–24.
Li J, Zeng M, Yan K, Yang Y, Li H, Xu X. IL-17 promotes hepatocellular carcinoma through inhibiting apoptosis induced by IFN-gamma. Biochem Biophys Res Commun. 2020;522:525–31.
Barton C, Davies D, Balkwill F, Burke F. Involvement of both intrinsic and extrinsic pathways in IFN-gamma-induced apoptosis that are enhanced with cisplatin. Eur J Cancer. 2005;41:1474–86.
Xin P, Xu X, Deng C, et al. The role of JAK/STAT signaling pathway and its inhibitors in diseases. Int Immunopharmacol. 2020;80:106210.
Wen W, Liang W, Wu J, et al. Targeting JAK1/STAT3 signaling suppresses tumor progression and metastasis in a peritoneal model of human ovarian cancer. Mol Cancer Ther. 2014;13:3037–48.
Ruan Z, Yang X, Cheng W. OCT4 accelerates tumorigenesis through activating JAK/STAT signaling in ovarian cancer side population cells. Cancer Manag Res. 2018;11:389–99.
Delen E, Doğanlar O. The dose dependent effects of ruxolitinib on the invasion and tumorigenesis in gliomas cells via inhibition of interferon gamma-depended JAK/STAT signaling pathway. J Korean Neurosurg Soc. 2020;63:444–54.
Darnell JJ. STATs and gene regulation. Science. 1997;277:1630–5.
Yu H, Jove R. The STATs of cancer—new molecular targets come of age. Nat Rev Cancer. 2004;4:97–105.
Silver DL, Naora H, Liu J, Cheng W, Montell DJ. Activated signal transducer and activator of transcription (STAT) 3: localization in focal adhesions and function in ovarian cancer cell motility. Cancer Res. 2004;64:3550–8.
Rosen DG, Mercado-Uribe I, Yang G, et al. The role of constitutively active signal transducer and activator of transcription 3 in ovarian tumorigenesis and prognosis. Cancer. 2006;107:2730–40.
Wu CJ, Sundararajan V, Sheu BC, Huang RY, Wei LH. Activation of STAT3 and STAT5 signaling in epithelial ovarian cancer progression: mechanism and therapeutic opportunity. Cancers (Basel). 2019;12:24.
Bachmann J, Raue A, Schilling M, et al. Division of labor by dual feedback regulators controls JAK2/STAT5 signaling over broad ligand range. Mol Syst Biol. 2011;7:516.
Lindemann C, Hackmann O, Delic S, Schmidt N, Reifenberger G, Riemenschneider MJ. SOCS3 promoter methylation is mutually exclusive to EGFR amplification in gliomas and promotes glioma cell invasion through STAT3 and FAK activation. Acta Neuropathol. 2011;122:241–51.
Liang Y, Xu WD, Peng H, Pan HF, Ye DQ. SOCS signaling in autoimmune diseases: molecular mechanisms and therapeutic implications. Eur J Immunol. 2014;44:1265–75.
Liu H, Wang W, Liu C. Increased expression of IFN-γ in preeclampsia impairs human trophoblast invasion via a SOCS1/JAK/STAT1 feedback loop. Exp Ther Med. 2021;21:112.
Funding
This work was supported by Suzhou science and technology development plan (application basis) (SYS201729) and Jiangsu province maternal and child health research project (F201709).
Author information
Authors and Affiliations
Contributions
AHG designed the study and drafted the manuscript. AHG and YRH were responsible for the collection and analysis of the experimental data. YRH and WPZ revised the manuscript critically for important intellectual content. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by Ethical Committee of the Second Affiliated Hospital of Soochow University and conducted in accordance with the ethical standards.
Informed consent
Subjects signed the informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, A.H., Hu, Y.R. & Zhu, W.P. IFN-γ inhibits ovarian cancer progression via SOCS1/JAK/STAT signaling pathway. Clin Transl Oncol 24, 57–65 (2022). https://doi.org/10.1007/s12094-021-02668-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-021-02668-9