Abstract
Purpose
Over the last decade a dramatic improvement in the treatment and prognosis of human epidermal growth factor receptor-2 (HER2) positive metastatic breast cancer (MBC) has been achieved. This study aimed to describe pattern, timing of metastases, and time to progression (TTP) of MBC patients (pts) treated with multiple lines of therapy with trastuzumab and/or lapatinib.
Methods
Clinical-pathologic features, treatment-lines and metastatic sites were collected from the institutional database; TTP was evaluated for each treatment-line. A meta-analysis of treatment-line estimates was performed; Q test and I 2-index were used to detect and estimate heterogeneity. Cox’s proportional hazards model and Fine and Gray’s proportional subhazards model in a competing risks setting were used to detect differences in hazard rate and to estimate relative risks.
Results
112 pts were analyzed. The median number of treatment-lines administered was 6 (range 1–17) and 524 (86 %) disease progression events were observed (median follow up 4.2 years). Distribution of metastases at baseline remained consistent across all lines. Having a given site affected by metastasis was a major risk factor of progression in that site. Hormone-receptor-positive pts resulted more likely to progress on bone (HR = 1.88). Elderly pts were less likely to progress on CNS (HR = 0.73). Median TTP resulted superior to 5 months up to the 6th line of treatment, reaching a plateau at the 9th treatment-line.
Conclusions
These data suggest that risk factors for progression in HER2 positive MBC do not significantly differ between various distributions of metastases, and that MBC pts benefit from anti-HER2 therapy even in late treatment-lines.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
About 20 % of invasive breast cancers (BC) exhibit protein overexpression and/or gene amplification of human epidermal growth factor receptor-2 (HER2) [1]. This subpopulation (hereinafter HER2+ BC) exhibits poor prognosis and a more aggressive phenotype with early relapses and increased rate of BC specific death [1]. Trastuzumab, a humanized monoclonal antibody directed against the extracellular domain of HER2, received Food and Drug Administration (FDA) approval in 1998, after phase II–III studies revealing that combination with chemotherapy increased overall response rate (ORR), time to progression (TTP), and overall survival (OS), compared with chemotherapy alone in patients with HER2+ metastatic BC (MBC) [2, 3]. More recently, trastuzumab, in combination with or after adjuvant chemotherapy, has improved disease-free and OS in patients with early stage BC [4, 5], and pathologic complete remission, disease-free and OS in the neoadjuvant setting [6].
15 % Of patients treated with adjuvant trastuzumab relapse. The majority of MBC patients present disease progression at most within 12–18 months from initial treatment [7]. Although phase II/III studies [8] and international guidelines [9] support the use of trastuzumab at relapse, its optimal duration in patients with long-term disease is unclear. A phase III trial of capecitabine with/without trastuzumab in trastuzumab-pretreated patients demonstrated the benefit of continuing it after progression in terms of ORR, TTP, and OS [8]. More recently, the FDA approved the HER2 tyrosine kinase inhibitor lapatinib in combination with capecitabine and the trastuzumab-emtansine conjugate drug, also known as T-DM1 [10], for the treatment of HER2+ MBC patients on progression following trastuzumab- and taxane-based chemotherapy.
Despite the increasing number of anti-HER2 agents and patients coping with MBC for long periods, few studies have investigated the natural history of HER2+ MBC, including factors that determine metastases characteristics [11–13].
Herein, we planned a mono-institutional cross-sectional study to investigate the clinical outcome of MBC patients receiving approved HER2-targeted therapies as a part of routine clinical care and we specifically conducted a detailed analysis of the metastatic timing, patterns, and factors influencing metastases.
Materials and methods
All patients with HER2+ MBC treated at the National Cancer Institute of Milan with at least one line, not necessarily the first one, of HER2-targeted therapy were considered eligible. Following the institutional review board approval, baseline demographics, treatment history, clinical and survival data were retrospectively collected from medical records. The observation period was considered from the first-line therapy up to the date of last contact. The database was locked to June 2013.
Patients were required to have HER2+ primary tumor defined by immunohistochemical (IHC) staining of 3+ (strong complete membrane staining in >10 % of tumor cells) or 2+ (weak to moderate complete membrane staining in >10 % of tumor cells) with fluorescence in situ hybridization (FISH) or chromogenic in situ hybridization (CISH) positivity defined as a HER2 gene signals to chromosome 17 signals (HER2:CEP17) ratio ≥2. This definition was applied to 82 patients included in the dataset. The remaining 30 patients were defined HER2+ with either a 3+ uniform and intense membrane staining of >30 % of invasive tumor cells and/or a FISH result of more than six HER2:CEP17 ratio of more than 2.2, as per ASCO CAP guidelines 2007 [14]. Two endpoints were collected for each treatment-line: specific site of disease progression and TTP; site of disease progression was classified as visceral, soft tissue (ST, including cutaneous and subcutaneous localizations, nodes, pleura and peritoneum), bone, and central nervous system (CNS); TTP was defined as the time from the first drug administration to tumor progression; patients who were alive and progression-free were right-censored at the time of the last contact; patients who were dead and progression-free were right-censored at the date of death; patients follow-up consisted of physical examination, laboratory analysis, and imaging exams every 8–12 weeks. In symptomatic patients and/or in case of laboratory abnormalities suspect for progression, imaging exams were anticipated, according to good clinical practice.
Statistical analysis
Treatment-lines were considered as a finite sequence of heterogeneous and dependent trials in which study endpoints were repeatedly collected (see detailed information on "Appendix", paragraph “Meta-analysis model”) [15]; for each treatment-line, TTP distribution was estimated by the product-limit method of Kaplan and Meier [16]; to detect the plateau reached by the TTP distribution along treatment-lines, a Cox regression model was used (see detailed information on "Appendix", paragraph “TTP plateau”). To test the influence of disease progression in a specific site as a risk factor for the same site progression in successive treatment lines (“carryover” effect) a logistic regression model was used (see detailed information on "Appendix", paragraph “Carryover effect”). To estimate the statistical association between covariates and the sites of disease progression, a survival analysis in presence of competing risks (e.g. other sites that could be affected by disease progression) was performed; the semiparametric regression model proposed by Fine and Gray [17] was applied [e.g. proportionality of the hazards of the cumulative incidence function (CIF)]; the cause-specific Hazard Ratio (HRcpRisk) was estimated for the following complementary causes: the specific site (e.g. visceral, ST, bone, CNS) and the competing site (for instance visceral or ST or bone is the competing site of the specific site CNS); each HRcpRisk was estimated across treatment-lines using a random-effects meta-analysis model; heterogeneity between treatment-lines estimates was evaluated using the Q statistic and the I 2 index (see detailed information on "Appendix", paragraph "Meta-analysis model"); every Fine and Gray regression model used to estimate the HRcpRisk was a multivariable regression model with five predictor variables: the evaluated characteristics (e.g. age, DFI, ER/PgR, anti-HER2 therapy as reported in Table 3) and the number of previous disease progressions affecting the specific site and the other three sites until the treatment-line involved; the last four predictor variables were introduced in each Fine and Gray regression model to adjust it for the ‘carryover effect’. Baseline covariate distributions were summarized using descriptive statistics (median and range for continuous variables, and absolute and percentage frequencies for categorical variables). Because of the descriptive nature of this study, hypothesis testing was applied qualitatively and not formally (e.g. no threshold for statistical significance level was defined). Statistical analysis was performed using SAS version 9.2 (SAS Institute, Cary, NC, USA); the statistical software R version 2.15.1 [18] was used to perform the survival analysis in presence of competing risks; the R function “crr” available in the “cmprsk” package [19] was used to estimate the HRcpRisk parameter; survival curves and bar charts were plotted using Stata version 12.1 (StataCorp, College Station, Texas 77845 USA).
Results
Patient characteristics
A total of 112 MBC patients with HER2+ disease treated with at least one line of therapy with anti-HER2 agents were identified. These patients were diagnosed with MBC between January 1995 and December 2012 and treated with anti-HER2 therapies between November 2000 and December 2012. The median age was 50 years (range 26–74 years). Approximately 15 % of patients had metastases at initial diagnosis. Among the 84 patients with early or locally advanced BC at diagnosis, 52 (62 %) received adjuvant therapy, 14 (17 %) neoadjuvant therapy and 10 (12 %) received both; 23 (27 %) patients were pretreated with trastuzumab. Hormone receptors (estrogen receptor-ER and/or progesterone receptor-PgR) were positive in about 70 % of cases. Sites of disease at diagnosis were ST (58 %), bone (42 %), liver (32 %), lung (27 %), and CNS (4 %). Of note, about half of the patients presented oligometastatic disease. Patient demographics and pathological characteristics are summarized in Table 1.
Treatment characteristics
Each treatment-line stopped for tumor progression and/or death for any cause. No patients included in the dataset discontinued treatment due to toxicity and/or consent withdrawal. Patients received a median of 6 (range 1–17) lines of therapy for MBC. In 79 patients (70.5 %) the first-line contained a HER2-targeted drug: trastuzumab in 68 cases (86 %), lapatinib in 11 cases (14 %). The remaining 33 patients received anti-HER2 therapy as a part of second (13 patients), third (7 patients) and >3rd (13 patients) line of treatment. Pertuzumab was prescribed to two patients in 2nd and 3rd line. The proportion of patients receiving HER2-targeted therapy was on average 70 % (range 67–75 %) across all treatments up to line 9 and 69 % (range 0–100 %) up to line 17, suggesting that anti-HER2 agents were prescribed upfront and maintained during treatments even in heavily pre-treated patients.
Patients received anti-HER2 treatments despite progression up to line 16. First-line contained chemotherapy in 95 cases (85 %) and endocrine therapy in 17 cases (15 %). The most common first-line regimen was taxane-based (51 patients, 45 %), followed by vinorelbine with/without capecitabine (26 patients, 23 %); capecitabine (plus lapatinib) was preferentially used in later lines (range 8.1–24.4 % from line 1 to line 9). During the course of metastatic disease, chemotherapy was progressively replaced by endocrine therapy (Fig. 1); the proportion of patients treated with endocrine therapy increased by 10 % for each subsequent line [Odds Ratio (OR): 1.10; 95 % CI 1.04–1.17; p = 0.001]. Among patients treated with endocrine treatment, first to third-line most common therapies included aromatase inhibitors (letrozole, anastrozole and exemestane in 82, 79, 71 % of cases, respectively). The use of aromatase inhibitors declined to 50 % in late lines, with a parallel increase in the use of fulvestrant from line 6 up to line 10.
Pattern of progression
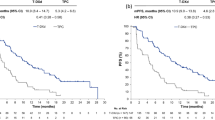
After a median follow-up of 4.2 years (range 0.2–18.1 years), 25 out of 112 (22.3 %) patients were dead; the progression rate at the first treatment was 77.7 %. The hazard rate of progression disease increased at each subsequent treatment-line of 28.9 % (95 % CI 17.3–41.6 %, p < 0.0001); this increment progressively reduced along the treatment-lines (HR quadratic term: 0.986, 95 % CI 0.978–0.993, p = 0.0002) until reaching a plateau at the 9th treatment line (95 % CI 6th–20th treatment line); the median TTP was 9.9 months (95 % CI 8.0–12.6 months) in the first-line treatment and 3.9 months (95 % CI 2.0–6.0 months) in the 9th line. Figure 2 reported the median TTP (which represents also the duration of treatment) until the 12th treatment-line (Fig. 2a) and the parameters’ estimates of the Cox model with treatment-line as linear and quadratic term (Fig. 2b). As reported in Table 2, visceral disease was the most frequent site of disease progression; on average along the treatment-lines the proportion of patients interested by disease progression in visceral site was 48.6 % (95 % CI 44.2–52.9 %) followed by ST (40.7 %; 95 % CI 36.6–44.9 %), bone (29.0 %; 95 % CI 24.4–33.5 %) and CNS (15.6 %; 95 % CI 12.6–18.7 %); for each site, heterogeneity along the treatment-lines was almost all explained by random error (range I 2 index: 0.0–20.3 %).
Determinants of pattern of progression
Progression sites showed a dragging effect: the risk of progression for each specific organ was higher if that organ was previously affected by metastases with an overall OR of 1.9 (95 % CI 1.7–2.1), 1.5 (95 % CI 1.4–1.6), 1.8 (95 % CI 1.6–2.0), and 4.2 (95 % CI 3.1–5.6) for visceral, ST, bone, and CNS disease, respectively (p < 0.0001) (Table 3). Considering all lines of treatment, age was not a risk factor for disease progression in visceral, ST, or bone involvement; an inverse correlation between the risk of CNS metastasis and patients’ age was observed (HRcpRisk, [10 years]: 0.73; 95 % CI 0.53–1.0, p = 0.07) although the risk estimate was poorly consistent between treatment-lines (I 2 = 51 %). The hazard ratio (HR) of disease progression showed a 24 % reduction (HR [10 years]: 0.76; 95 % CI 0.54–1.06, p = 0.10) for every 10 years of disease-free interval (DFI) with all heterogeneity along the treatment-lines explained by random error (I 2 = 0 %); the HRcpRisk of CNS progression showed a 64 % reduction for every 10 years of DFI (HRcpRisk, [10 years]: 0.36; 95 % CI 0.11–1.20, p = 0.10; I 2 = 0 %). An increased progression risk in bone lesions was found in ER and/or PR positive disease (HRcpRisk: 1.88; 95 % CI 0.97–3.64, p = 0.06; I 2 = 0 %). The HR of bone progression showed a 34 % of reduction with anti-HER2 therapy (HRcpRisk: 0.66; 95 % CI 0.45–0.98, p = 0.04; I 2 = 0 %).
Discussion
This study aimed to analyze the clinical outcome of MBC patients receiving approved anti-HER2 therapies and the determinants of metastatic timing and patterns of spreading.
The natural history of HER2+ tumors radically changed with the introduction of trastuzumab as part of 1st line regimens. For about 10 years, it was the only HER2-targeted therapy available in clinical practice, and then lapatinib was introduced. More recently, new anti-HER2 agents have been developed.
Although our data should be interpreted with caution, as the retrospective design may have selected patients with better prognosis, this results support the administration of HER2-targeted therapies during subsequent treatment-lines, regardless the combination with chemo- or endocrine-therapy. The study by Blackwell et al. [20, 21] has already highlighted a benefit in continuing trastuzumab beyond progression, comparing lapatinib with/without trastuzumab. Herein we support the concept that maintenance should be carried over the third line of treatment, and that development of new drugs should not be limited to first-lines, but also to later treatments.
About half of patients presented an oligometastatic disease. 58 and 42 % of patients had ST and bone metastases respectively, presenting an indolent disease. This reflects the heterogeneity of HER2+ MBC as a group of tumors with different patterns of presentation and prognosis. The unfavorable prognosis associated with sites of disease such as CNS and viscera is well recognized and may be due to more aggressive disease and less activity of anticancer treatments at these sites. In this scenario, the impact of different sites of metastasis on the outcome of HER2+ BC patients treated with targeted therapies is still unclear. We investigated whether the disease site could be considered as a predictor of response to treatment. With the limits of the cross-sectional study and the small number of patients, our data suggest that there is no interaction between anti-HER2 treatment efficacy and the sites of metastases, and that treatment does not affect the progression of metastatic sites. The risk of progression for each specific organ was higher if that organ was previously affected by disease, indicating that progression sites showed a “carry-over effect”. An increased progression risk in bone lesions was found in hormone-receptor-positive disease. Young patients had an increased risk of developing CNS metastasis, but it did not reach statistical significance. These data are consistent with previous adjuvant studies that analyzed the pattern of disease recurrence [4, 5] and, together with the “carry-over effect”, may suggest different and personalized follow-up schedules [11–13]. Approximately one-third of women with HER2+ MBC will develop brain metastases [22–25]; in our clinical records, only 4.5 % of patients present CNS disease. This difference is not easy to explain and may simply reflect the nature of our study, i.e. database derived, in contrast to published literature, in which there may be reporting bias; in fact we may hypothesize that brain metastases may not have been diagnosed in all affected patients.
Another possible explanation could be that most of the MBC patients (77 %) included in our study had not received adjuvant trastuzumab and it is well recognized that the incidence of CNS disease as the first site of recurrence was relatively uncommon before the extensive use of adjuvant trastuzumab [26]. Several studies suggest a potential organotropism of lapatinib for the CNS [27]. A retrospective exploratory analysis of EMILIA trial showed a similar efficacy of TDM1 versus lapatinib plus capecitabine in terms of CNS disease progression [28]; a benefit of TDM1 in terms of OS was reported for patients with brain metastases at baseline. On the other hand, retrospective experimental data support the possibility that the protection exerted by trastuzumab is lower in bone metastases [29]. Conversely, it has been shown that women who never received trastuzumab were more likely to develop bone metastasis than brain metastasis [30]. Although these studies suggesting that specific sites can differentially benefit from anti-HER2 therapies, the potential predictive role of metastatic sites for these treatments is still debated. Therefore, choosing therapy on the basis of the disease presentation and pattern of progression still remains a challenge.
It should be noted that, in the present study, the patient population came from daily clinical practice and the tumor response assessment was performed in a less rigorous fashion than in clinical trials. This may provide a justification for the longer median duration of treatment reported. This study is proof of the concept that treatment exerts its effect in a cytostatic rather that cytotoxic manner and our data support the use of HER2 inhibitors at relapse, as well as (mono-) chemotherapy or endocrinotherapy beyond 2nd line in highly motivated, fit patients, responders to first-lines, with an oligometastatic, indolent disease, without comorbidity/complications to achieve a better quality of life.
References
Slamon DJ, Clark GM, Wong SG, Levin WJ, Ullrich A, McGuire WL. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science. 1987;235(4785):177–82.
Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344:783–92.
Marty M, Cognetti F, Maraninchi D, Snyder R, Mauriac L, Tubiana-Hulin M, et al. Randomized phase II trial of the efficacy and safety of trastuzumab combined with docetaxel in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer administered as first-line treatment: the M77001 study group. J Clin Oncol. 2005;23:4265–74.
Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE Jr, Davidson NE, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005;353(16):1673–84.
Piccart-Gebhart MJ, Procter M, Leyland-Jones B, Goldhirsch A, Untch M, Smith I, et al. Herceptin Adjuvant (HERA) Trial Study Team. Trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer. N Engl J Med. 2005;353(16):1659–72.
Buzdar AU, Ibrahim NK, Francis D, Booser DJ, Thomas ES, Theriault RL, et al. Significantly higher pathologic complete remission rate after neoadjuvant therapy with trastuzumab, paclitaxel, and epirubicin chemotherapy: results of a randomized trial in human epidermal growth factor receptor 2-positive operable breast cancer. J Clin Oncol. 2005;23(16):3676–85 Epub 2005 Feb 28.
Baselga J, Cortés J, Kim SB, Im SA, Hegg R, Im YH, et al. Pertuzumab plus trastuzumab plus docetaxel for metastatic breast cancer. N Engl J Med. 2012;366(2):109–19. doi:10.1056/NEJMoa1113216 (Epub 2011 Dec 7).
von Minckwitz G, du Bois A, Schmidt M, Maass N, Cufer T, de Jongh FE. et al Trastuzumab beyond progression in human epidermal growth factor receptor 2-positive advanced breast cancer: a German breast group 26/breast international group 03-05 study. J Clin Oncol. 2009;27(12):1999–2006. doi:10.1200/JCO.2008.19.6618.
Giordano SH, Temin S, Kirshner JJ, Chandarlapaty S, Crews JR, Davidson NE, et al. Systemic therapy for patients with advanced human epidermal growth factor receptor 2- positive breast cancer: American society of clinical oncology clinical practice guideline. J Clin Oncol. 2014;32(19):2078–99. doi:10.1200/JCO.2013.54.0948 (Epub 2014 May 5).
Verma S, Miles D, Gianni L, Krop IE, Welslau M, Baselga J, et al. Trastuzumab emtansine for HER2-positive advanced breast cancer. N Engl J Med. 2012;367(19):1783–91. doi:10.1056/NEJMoa1209124.
Yardley DA, Kaufman PA, Brufsky A, Yood MU, Rugo H, Mayer M, et al. Treatment patterns and clinical outcomes for patients with de novo versus recurrent HER2-positive metastatic breast cancer. Breat Cancer Res Treat. 2014;145(3):725–34.
Vaz-Luis I, Ottesen RA, Hughes ME, Marcom PK, Moy B, Rugo HS, et al. Impact of hormone receptor status on patterns of recurrence and clinical outcomes among patients with human epidermal growth factor-2-positive breast cancer in the National Comprehensive Cancer Network: a prospective cohort study. Breast Cancer Res. 2012;14(5):R129.
Maki DD, Grossman RI. Patterns of disease spread in metastatic breast carcinoma: influence of estrogen and progesterone receptor status. AJNR Am J Neuroradiol. 2000;21(6):1064–6.
Wolff AC, Hammond ME, Schwartz JN, Hagerty KL, CraigAllred D, Cote RJ, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for human epidermal growth factor receptor 2 testing in breast cancer. J Clin Oncol. 2007;25:118–45.
DerSimonian R, Laird N. Meta-analysis in clinical trials. Control Clin Trials. 1986;7:177–88.
Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J Am Stat Assoc. 1958;53(282):457–81.
Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509.
R Core Team. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2012.
Gray B. cmprsk: Subdistribution analysis of competing risks. 2013. R package version 2.2–6. http://www.CRAN.R-project.org/package=cmprsk.
Blackwell KL, Burstein HJ, Storniolo AM, Rugo H, Sledge G, Koehler M, et al. Randomized study of Lapatinib alone or in combination with trastuzumab in women with ErbB2-positive, trastuzumab-refractory metastatic breast cancer. J Clin Oncol. 2010;28(7):1124–30. doi:10.1200/JCO.2008.21.4437.
Blackwell KL, Burstein HJ, Storniolo AM, Rugo HS, Sledge G, Aktan G, et al. Overall survival benefit with lapatinib in combination with trastuzumab for patients with human epidermal growth factor receptor 2-positive metastatic breast cancer: final results from the EGF104900 Study. J Clin Oncol. 2012;30(21):2585–92. doi:10.1200/JCO.2011.35.6725.
Bendell JC, Domchek SM, Burstein HJ, Harris L, Younger J, Kuter I, et al. Central nervous system metastases in women who receive trastuzumab-based therapy for metastatic breast carcinoma. Cancer. 2003;97:2972–7.
Clayton AJ, Danson S, Jolly S, Ryder WD, Burt PA, Stewart AL, et al. Incidence of cerebral metastases in patients treated with trastuzumab for metastatic breast cancer. Br J Cancer. 2004;91:639–43.
Lin NU, Winer EP. Brain metastases: the HER2 paradigm. Clin Cancer Res. 2007;13:1648–55.
Stemmler HJ, Kahlert S, Siekiera W, Untch M, Heinrich B, Heinemann V. Characteristics of patients with brain metastases receiving trastuzumab for HER2 overexpressing metastatic breast cancer. Breast. 2006;15:219–25.
Olson EM, Abdel-Rasoul M, Maly J, Wu CS, Lin NU, Shapiro CL. Incidence and risk of central nervous system metastases as site of first recurrence in patients with HER2-positive breast cancer treated with adjuvant trastuzumab. Ann Oncol. 2013;24(6):1526–33.
Bachelot T, Romieu G, Campone M, Diéras V, Cropet C, Dalenc F, et al. Lapatinib plus capecitabine in patients with previously untreated brain metastases from HER2-positive metastatic breast cancer (LANDSCAPE): a single-group phase 2 study. Lancet Oncol. 2013;14(1):64–71.
Krop IE, Lin NU, Blackwell K, Guardino E, Huober J, Lu M, et al. Trastuzumab emtansine (T-DM1) versus lapatinib plus capecitabine in patients with HER2-positive metastatic breast cancer and central nervous system metastases: a retrospective, exploratory analysis in EMILIA. Ann Oncol. 2015;26(1):113–9. doi:10.1093/annonc/mdu486.
Campiglio M, Bufalino R, Sasso M, Ferri E, Casalini P, Adamo V, et al. Effect of adjuvant trastuzumab treatment in conventional clinical setting: an observational retrospective multicenter Italian study. Breast Cancer Res Treat. 2013;141(1):101–10.
Lower EE, Drosick DR, Blau R, Brennan L, Danneman W, Hawley DK. Increased rate of brain metastasis with trastuzumab therapy not associated with impaired survival. Clin Breast Cancer. 2003;4(2):114–9.
Conflict of interest
The authors disclose no potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
D. Serpico and L. Porcu contributed equally to this paper.
Appendix
Appendix
Meta-analysis model
Treatment lines were considered as a finite sequence of heterogeneous, dependent, and random trials, i.e. experiments, in which study endpoints were repeatedly collected; to summarize these repeated experiments and to evaluate the heterogeneity of the repeated estimates, a random-effects meta-analysis model was applied and the following statistics were considered: the point and 95 % confidence interval of the combined estimate, the Q statistic and the I 2 index; the between-studies variance was estimated using the DerSimonian and Laird method [11].
TTP plateau
To demonstrate that a plateau was reached by the TTP distribution after a certain number of treatment lines and to estimate the treatment line interested by the plateau, a Cox regression model was used; the number of treatment lines was introduced both as a linear and a quadratic predictor.
Carryover effect
To test the influence of disease progression in a specific site as a risk factor for the same site-progression in successive treatment lines (“carryover” effect), the following statistics was computed: for each site of disease progression and for each k = 0, 1, 2, … , n we defined the k-sample group as follows: this group was arranged by patients experiencing the first disease progression event in the evaluated site for the k-time (note: the 0-sample group was arranged by patients starting the first advanced treatment line without the evaluated site as metastatic site); the ratio between the subsequent disease progression events concerning the specific site and the total number of subsequent disease progression events whichever site occurred was calculated; this ratio could be mathematically expressed as a weighted average of the proportion of disease progression events affecting the evaluated site for each patient weighted by the number of subsequent treatment lines associated with a disease progression event:
where S i is the number of interested events of first disease progression for patient n°i; Tot i : number of subsequent treatment lines involved by first disease progression for patient n°i.
The logistic regression model was used to estimate the Odds Ratio (OR) between the k + 1-sample and the k-sample odds statistics and to statistically test the presence of a “carryover” effect.
Rights and permissions
About this article
Cite this article
Serpico, D., Porcu, L., Tessari, A. et al. Disease progression pattern in metastatic breast cancer patients treated with anti-HER2 therapies. Clin Transl Oncol 17, 530–538 (2015). https://doi.org/10.1007/s12094-015-1274-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-015-1274-2