Abstract
Methane is a potent greenhouse gas and produced mainly by methanogens. Few studies have specifically dealt so far with methanogens in estuarine environments. In this study, diversity and distribution of methanogens were investigated by clone library and T-RFLP analysis in a Jiulong River estuarine sediment core which contained clear sulfate–methane-transition zone. The majority of obtained sequences in clone libraries and T-RF peaks from T-RFLP analysis were assigned mainly to Methanosaeta, Methanomicrobiales and Methanosarcinales/ANME. The fragments of Methanosarcinales/ANME were most dominant group (mean 51 %) and composed largely of ANME-2a. In addition, Methanosaeta and Methanomicrobiales accounted for 21 and 28 % of all fragments. Therefore, the presence of Methanomicrobiales, Methanosaeta and ANME-2a was indicative of acetoclastic methanogenesis, hydrogenotrophic methanogenesis, and anaerobic methane oxidation in Jiulong River estuarine sediments. This study provided the important knowledge towards understanding methane cycling association of representative of methanogens involved in estuarine environments.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
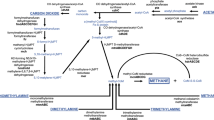
Methanogen is widespread in marine sediments where contain the largest global reservoir of methane and dominate the terminal steps of organic mater degradation to produce methane when depletion of sulfate occurs in anaerobic environments [1]. Methane is an important greenhouse gas and has so far contributed an estimated 20 % of postindustrial global warming [2]. Marine sediments produce methane between 75 and 320 Tg year−1 which methanogens are mainly responsible for [3]. However, nearly all methane is oxidized microbially in the marine sediment [4]. Till date, numerous studies on methanogen communities mainly focused on seep, gas hydrate, continental-margin, lake and tidal sediments with universal archaeal primers which were not specific to methanogen [4–7]. These results failed to detect the whole methanogen communities because they only represented a minor part of archaea in these environments. Recently methanogen-specific 16S rRNA gene primers were reported, which provided a useful tool to detect methanogen communities in marine sediments [8, 9].
Estuaries are an important link between land and the open sea, and well known as natural deposits of organic matter which come from marine, terrestrial and anthropogenic sources. These organic matters are finally used to produce methane when sulfate is depleted in anaerobic environments. So estuaries contribute importantly to marine CH4 emissions [10]. Methane production and consumption are spatially separated and conducted by different archaeal groups in the subsurface sediment environments [2]. Jiulong River Estuary located at south tropical region in Fujian Province, southern China. It is one of the largest river/estuary systems in southern China with an area of 14741 km2. This area have a high load of anthropogenic nutrients from fish dike farming, industrial wastewater, domestic sewage due to economic and population developments [11]. However the communities of methanogens were not addressed in Jiulong River Estuary. In this study, methanogens were investigated by applying clone library construction and T-RFLP analysis with methanogen-specific 16S rRNA gene primers. Finally, methane cycling association of representative of methanogens was documented in Jiulong River estuarine environments.
Materials and Methods
Sediment Collection and Description
A sediment core of 1.5 m named as JL08 were collected by gravity coring from Jiulong River Estuary (24°24′48.6″ N, 117°56′30.5″ E), in Fujian province, China. The sediment core was sectioned into 3 cm slices with sterile falcon tubes on a clean bench and stored with dry ice quickly. Finally the samples were stored aseptically at −80 °C until analysis.
Construction of Clone Libraries and DNA Sequencing
Total genomic DNA was extracted from subsamples of sediment core using SDS-based DNA extraction method [12]. The methanogen-related 16S rRNA fragments were amplified by PCR using primers 355F (3′-CAGGCGCGAAAACTTTAC-5′) and 1068R (3′-ATGCTTCACAGTACGAAC-5′) from layers above sulfate-methane-transition zone (SMTZ) (31–34 cm), within (71–74 cm) and below (151–154 cm) in core JL08. These primers have previously been developed to specifically target members of Methanosarcinales and Methanomicrobiales [8]. All reactions were carried out in 50 μl mixtures containing approximately 10–100 ng sediment DNA, 10 × reaction buffer, 200 μM dNTP, 10 pmol of each primer, 1.5 mM MgC12 and 5U Taq polymerase. Negative controls were performed with water instead of the template. The reactions were performed in a T3 thermocycler (Biometra, Germany) using 30 cycles consisting of 94 °C for 1 min, 52 °C for 1 min and 72 °C for 1 min, followed by a final extension step at 72 °C for 10 min. Reaction products were checked by agarose gel electrophoresis. The clone libraries were constructed by using the TA cloning kit (TaKaRa) following the manufacturer’s instructions. The positive clones contained 16S rRNA gene fragments were picked randomly for sequencing (Sangon Inc., Shanghai, China).
Phylogenetic and Diversity Analysis
The obtained 16S rRNA gene sequences were submitted to the Chimera–Check program at the Ribosomal Database to detect the presence of chimeric artifacts. The sequences bearing more than 97 % similarity were considered as the identical phylotype. Phylogenetic trees were inferred, and bootstrap analysis (1000 replicates) was performed with the MEGA 4.0 by the neighbor-joining method [13]. Then coverage was calculated by using the following formula: C = 1−(n1/N), where n1 is the number of phylotype that occurred only once in the clone library and N is the total number of clones analyzed. The Shannon-Wiener index and evenness (equitability) were calculated using the equations from Krebs [14].
T-RFLP Analysis
Amplification of methanogen 16S rRNA genes from all depths were performed using the same methanogen-specific primers with a 5′ label of 6-carboxyfluorescein (FAM) attached to the forward primer as described by the 16S rRNA gene library construction. Dual restriction digestion of the PCR amplicon was performed by incubating 100–200 ng of the PCR amplicons with 5 U of TaqI (Takara) at 65 °C for 2 h followed by incubation at 37 °C for 2 h with 5 U of added RsaI (Takara). Restriction product was diluted to 500 μl with sterile water and passed through Microcon YM-10 centrifugal filters (Millipore) to desalt. Desalted restriction products were analyzed on an ABI 3100 Prism Genetic Analyzer automated capillary sequencer and peak heights quantified using GeneScan Analysis v3.7 software (Applied Biosystems) as described previously [8, 9]. Signals with a peak height below 100 relative fluorescent units (rfu) were excluded from the analysis.
Nucleotide Sequence Accession Numbers
The nucleotide obtained in this study were deposited in the NCBI GenBank database under accession numbers (JQ866664–JQ866685).
Results and Discussion
Profiles of Sulfate and Methane
The sulfate concentration was highest (23 mM) at the sediment layer 16 cm and only 5.8 mM in top sediment layer which reflected influences of water-column mixing, and declined with the depth to less than 2.0 mM below 86 cm along Jiulong River estuarine sediment core. The methane concentration was less than 2.3 μM in the top above 60 cm, but increased gradually below 61 cm to a maximum value of 5.8 μM at 91 cm. Therefore, there is a clear SMTZ in Jiulong River estuarine sediments at an interval from 60 to 86.0 cm (Fig. 1(a)), similar to large areas of estuary and marine sediments where methane controlled by diffusion. In these sediments, anaerobic methane oxidation (AOM) and methanogenesis are finely balanced.
Distribution and Phylogenetic Analysis
Three clone libraries were constructed from the PCR-amplified methanogen-specific 16S rRNA genes. A total of 146 clones from the libraries were randomly selected for sequencing. A similarity analysis of all sequences presented 20 different representative clones. The coverage values of the libraries were shown to be from 80 to 95 % (Table 1). The limited diversity of 16S rRNA gene sequences was observed by the analysis of the Shannon-Wiener index (Table 1). BLASTN search of phylotype sequences indicated that majority of the retrieved methanogen sequences clustered closely with uncultured methanogen sequences came from environments like lake and marine sediments or methanogenic digester sludge rather than cultured isolates. Phylogenetic analysis was used to outline the phylogenetic positons of the detected phylotypes (Fig. 2). The majority of obtained sequences in clone libraries and T-RF peaks from T-RFLP analysis were assigned mainly to Methanosaeta, Methanomicrobiales and Methanosarcinales/ANME.
Neighbor-joining tree showing phylogenetic positions of the methanogen retrieved during this study. Phylotype name refer to sample M31 (31–33 cm at depth interval), M71 (71–74 cm at depth interval), and M151 (151-154 cm at depth interval). Bootstrap values equal to or larger than 0.5 are indicated. The values were determined using 1,000 replicates
The group of Methanosarcinales/ANME was most dominant (41 % of total sequences and 51 % of total fragments) in which ANME-2a could be identified by phylogenetic analysis of 16S rRNA sequences (Fig. 2). The peaks of Methanosarcinales/ANME from T-RFLP analysis were prevailing (mean 63 %), relatively constant blow 44 cm, and accounted for 19.3 % averagely above 34 cm in the sediment core. The most predominant phylotype affiliated with ANME-2a (38 % of total sequences) in libraries was represented by M151-3 (32 % of total sequences) and was closely related to AOM-Clone-E10 (97 %) from anaerobic methane-oxidizing sulfate-reducing enrichment [15]. Till date, it was well known that ANME-2a was responsible for anaerobic methane oxidation in a variety of environments including clod seep, mud volcano, deep sea sediment, hydrothermal vent and coastal sediment [2]. Thus Methanosarcinales/ANME could be safely related to methane oxidizing archaea in our study.
The group of Methanomicrobiales represented 28 % of total sequences and 27 % of total fragments .The peaks of Methanomicrobiales were to a maximum at layer of 3–6 cm (67 %), deceased to 31.8 % at layer of 31–34 cm, and relatively constant blow 44 cm (mean 21 %) in the sediment core. The phylotype M151-5 was related (95 %) to Methanoregula formicica isolated from methanogenic sludge and represented 12 % of total sequences [16]. The phylotype M151-17 was indentical (97 %) to Methanolinea tarda NOBI-1 isolated from a methanogenic digester sludge [17] and presented 8 % of total sequences. The phylotype M151-60 was identical (95 %) with Methanoculleus receptaculi isolated from Shengli oil field, China [18], and ocuppied 7 % of total sequences.
The group of Methanosaeta accounted for 27 % of total sequences and 21 % of fragments mean for all depth. Methanosaeta-related fragments increased suddenly from 11 to 34 cm (mean 42 %) and deceased to 12 % at the bottom along the core. The phylotypes (M151-24 and M151-4) were related (95–97 %) to Methanosaeta concilii [19]. The phylotypes (M31-30, M31-41, M151-2 and M151-16) were related (95–98 %) to Methanosaeta harundinacea strain 6Ac isolated from a UASB reactor which used acetate exclusively for growth and methane production [20]. In addition, the phylotype M33-30 were the most detected phylotype (12 % of total sequences).
Considering their range of utilizing substrates, the majority of Methanosaeta, Methanomicrobiales and Methanosarcinales/ANME could be related to acetoclastic methanogenen, hydrogenotrophic methanogen and methane oxidizing methanogen. The results from Skagerrak and brackish Lake sediments indicated that the presence and relative abundance of methanogen sequences fit with the comparative rate measurements of H2/CO2 and acetate methanogenesis at the same depth [8, 9]. Thus the relative abundance of methanogen fragments could be explained activity of methanogen to some extend in Jiulong River estuarine sediments.
Anaerobic methane oxidation represents an effective barrier to methane released from sediments. Methanosarcinales/ANME was most dominant group in Jiulong River estuarine sediments. The sequences of Methanosarcinales/ANME in clone libraries were dominated by ANME-2a which was characterized as anaerobic methane oxidizing archaea. Then, why do they contribute major part even if blow SMTZ in Jiulong River estuarine sediments? The primer bias could be excluded because of previous analysis [8, 9]. The possible reason could be specific sulfate and methane flux in estuarine environments. Sulfate concentration is only 5.8 mM in top sediment layer and 23 mM at the sediment layer 16 cm, which reflected influences of water-column mixing due to intertidal activities. The frequent alterations of depth of SMTZ due to sulfate and methane flux resulted in accumulations of methane oxidizing methanogens in a wide range of sediments.
Hydrogenotrophic methangenesis was predominant in marine environments because of a result of sulfate reduction [8, 9]. On the contrary, acetoclastic methanogenensis contributed approximately 70 % of total methane production in freshwater environments because of low sulfate concentration [21, 22]. In this study, the fragments affiliated with Methanosaeta and Methanomicrobiales accounted for 21 and 27 % of all fragments averagely. The dominance of Methanomicrobiales and Methanosaeta indicated that both acetoclastic and hydrogenotrophic methanogenesis were the main methanogenic pathway in Jiulong River estuarine sediments. This result exhibited the methanogenic character of transition zone from fresh water environments to marine environments in Jiulong River estuarine sediments.
In this study, diversity and distribution of methanogen were investigated by clone library and T-RFLP in a Jiulong River estuarine sediment core. The results showed that the group of Methanosarcinales/ANME was most dominant and ANME-2a was mainly responsible for AOM. Acetoclastic and hydrogenotrophic methanogenesis could be the main methanogenic pathway in Jiulong River estuarine sediments. This study rovided the important knowledge towards understanding methane cycling and methanogen involved in estuarine environments.
References
Kvenvolden KA (1988) Methane hydrate—a major reservoir of carbon in the shallow geosphere? Chem Geol 71:41–51
Knittel K, Boetius A (2009) Anaerobic oxidation of methane: progress with an unknown process. Annu Rev Microbiol 63:311–334
Valentine DL (2002) Biogeochemistry and microbial ecology of methane oxidation in anoxic environments: a review. Antonie Van Leeuwenhoek 81:271–282
Orphan VJ, House CH, Hinrichs KU, McKeegan KD et al (2001) Methane-consuming archaea revealed by directly coupled isotopic and phylogenetic analysis. Science 293:484–487
Harrison BK, Zhang H, Berelson W, Orphan VJ (2009) Variations in archaeal and bacterial diversity associated with the sulfate-methane transition zone in continental margin sediments (Santa Barbara Basin California). Appl Environ Microbiol 75:1487–1499
Jiang L, Zheng Y, Chen J, Xiao X et al (2011) Stratification of archaeal communities in shallow sediments of the Pearl River Estuary Southern China. Antonie Van Leeuwenhoek 99:739–751
Ahmed N, Johri S, Sultan P, Abdin MZ et al (2011) Phylogenetic characterization of archaea in saltpan sediments. Indian J Microbiol 51:132–137
Banning N, Brock F, Fry JC, Parkes RJ et al (2005) Investigation of the methanogen population structure and activity in a brackish lake sediment. Environ Microbiol 7:947–960
Parkes RJ, Cragg BA, Banning N, Brock F et al (2007) Biogeochemistry and biodiversity of methane cycling in subsurface marine sediments (Skagerrak Denmark). Environ Microbiol 9:1146–1161
Shalini A, Ramesh R, Purvaja R, Barnes J (2006) Spatial and temporal distribution of methane in an extensive shallow estuary south. India J Earth Syst Sci 115:451–460
Song C, Wenzhuo L, Aiyu X (1998) Transfer of organic mather in Jiulong Jiang Esturine waters 1995 cruises. J Oceanogr Taiwan Strait 17:180–184
Zhou J, Bruns MA, Tiedje JM (1996) DNA recovery from soils of diverse composition. Appl Environ Microbiol 62:316–322
Kreb CJ, Elzinga C, Salzer D, Willoughby J et al (1989) Ecological methodology. Harper & Row, New York
Jagersma GC, Meulepas RJW, Gieteling J et al (2009) Microbial diversity and community structure of a highly active anaerobic methanemethane-oxidizing sulfate-reducing enrichment. Environ Microbiol 11:3223–3232
Yashiro Y, Sakai S, Ehara M, Miyazaki M et al (2011) Methanoregula formicica sp nov a methane-producing archaeon isolated from methanogenic sludge. Int J Syst Evol Microbiol 61:53–59
Imachi H, Sakai S, Sekiguchi Y, Hanada S et al (2008) Methanolinea tarda gen nov sp a methane-producing archaeon isolated from a methanogenic digester sludge. Int J Syst Evol Microbiol 58:294–301
Cheng L, Qiu TL, Li X, Wang WD et al (2008) Isolation and characterization of Methanoculleus receptaculi sp nov from Shengli oil field China. FEMS Microbiol Lett 285:65–71
Mori K, Iino T, Suzuki K, Yamaguchi K et al (2012) Aceticlastic and NaCl-requiring methanogen “Methanosaeta pelagica” sp nov isolated from marine tidal flat sediment. Appl Environ Microbiol 78:3416–3423
Ma K, Liu X, Dong X (2006) Methanosaeta harundinacea sp nov a novel acetate-scavenging methanogen isolated from a UASB reactor. Int J Syst Evol Microbiol 56:127–131
Conrad R (1999) Contribution of hydrogen to methane production and control of hydrogen concentrations in methanogenic soils and sediments. FEMS Microbiol Ecol 28:193–202
Kotsyurbenko OR, Chin KJ, Glagolev MV, Stubner S et al (2004) Acetoclastic and hydrogenotrophic methane production and methanogenic populations in an acidic West-Siberian peat bog. Environ Microbiol 6:1159–1173
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 40. Mol Biol Evol 24:1596–1599
Acknowledgments
This work has been partly supported by Natural Science Foundation of Yunnan University(2012CG014, 2012CG009, 2012CG007).
Conflict of interest
We declare that no financial/commercial conflicts of interest exist in this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, J., Yin, X. Stratified Communities of Methanogens in the Jiulong River Estuarine Sediments, Southern China. Indian J Microbiol 53, 432–437 (2013). https://doi.org/10.1007/s12088-013-0397-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12088-013-0397-9