Abstract
Thyroid hormones (THs) play a critical function in fundamental signaling of the body regulating process such as metabolism of glucose and lipids, cell maturation and proliferation, and neurogenesis, to name just a few. THs trigger biological effects both by directly affecting gene expression through the interaction with nuclear receptors (genomic effects) and by activating protein kinases and/or ion channels (short-term effects). For years, a close relationship between the THs hormones and the central nervous system (CNS) has been described, not only for neuronal cells but also for glial development and differentiation. A deficit in thyroid hormones triiodothyronine (T3) and thyroxine (T4) is observed in the hypothyroid condition, generated by a iodine deficiency or an autoimmune response of the body. In the hypothyroid condition, several cellular deregulation and alterations have been described in dendrite spine morphology, cell migration and proliferation, and impaired synaptic transmission in the hippocampus, among others. The aim of this review is to describe the role of the thyroid hormones with focus in brain function and neurodegenerative disorders.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Thyroid hormones (TH) are crucial for multiple body functions, from development in prenatal stages, such as neural growth and differentiation, up to adulthood functions, like memory and concentration, maintenance of metabolic rate, equilibrium of reactive oxygen species (ROS), cardiovascular function, thermogenesis, and feeding [1, 2]. Also, in the last years, THs has been involved in more complex processes including the regulation of some clock genes expression and memory/learning task [3].
A deficit in thyroid hormones triiodothyronine (T3) and thyroxine (T4) is observed in the hypothyroid condition, produced mainly by an iodine deficiency or an autoimmune response of the body [4,5,6,7]. This condition affects 1–2% of the population, where the incidence increases 10 times more in women than in man, while in iodine-deficient areas, equivalent to one third of the world’s population, the prevalence increases up to 80% [5]. The main locations affected by this problem are South Asia and sub-Saharan Africa. During pregnancy, the fetus relies on the mother’s thyroid hormones until the 12–14 weeks of gestation [8]. Nowadays, hypothyroidism during pregnancy affects 2.5% of women, and congenital hypothyroidism has an incidence of one newborn in 3500–4000 births [5]. The impact of a deficiency during the development period has shown impaired growth and neurodevelopment; alterations in dendrite spine morphology, cell migration and proliferation; and impaired synaptic transmission in hippocampus has been detected [5, 9, 10]. Furthermore, a relationship between suboptimal thyroid function during pregnancy with low child intelligence quotient (IQ) and attention-deficit/hyperactivity disorder (ADHD) has been established [6].
Postnatal studies showing alterations in thyroid hormones have revealed perturbations in glucose metabolism, hypothalamus energy imbalance, and hippocampal structure and synaptic plasticity, affecting the general brain function [11, 12]. Adult neurodegenerative diseases has also been related with hypothyroidism, specifically both in Alzheimer’s disease (AD) and Parkinson’s disease, were a thyroid hormone imbalance has been reported in numerous cases [13]. Furthermore, several pathways or potential biomarkers involved in these diseases are related to thyroid hormone targets, but the specific relation is still unknown [14, 15]. The aim of this review is to understand the complexity of thyroid hormone actions with focus in neurodegenerative diseases from developmental stages up to senile stages.
Mechanism of Action of Thyroid Hormones
Thyroid hormone action is influenced by several cellular mechanisms, from the amount of local ligand conversion, specialized membrane transporters, thyroid hormone receptor isoform, crosstalk with other key cellular pathways and nuclear corepressors and coactivators. This denotes the complex mechanisms that underlies the action of thyroid hormones [16,17,18].
At the cellular level, TH exerts an effect in the metabolism of proteins, carbohydrates, and lipids, affecting numerous processes, including metabolic pathways and growth and inflammatory processes [19]. The uptake of thyroid hormones by the cells is mediated through specific transporters that are expressed in different proportions and cell type depending on the developing stage. The main transporters in the brain are the monocarboxylate transporter (MCT), L-type amino acid transporters (SLC7), and the organic anion transporter polypeptides (OATPs) that may express differentially in oligodendrocyte, astrocyte, neurons, microglia, and endothelial cells [16, 20]. Once inside the cells, thyroid hormones generate their physiological actions through the interaction of T3 with thyroid hormone nuclear receptors (TR), which exists in four isoforms, these are α1, β1, β2, and β3. These nuclear receptors act as transcription factors that activates with the binding of T3, and as transcriptional repressors in the absence of this hormone [21, 22]. These thyroid hormone receptors are also expressed in different proportions in organs. TRα1 is predominantly expressed in the brain, heart, and skeletal muscle, while TRβ1 is expressed extensively; TRβ2 is expressed in the brain, retina, and inner ear and TRβ3 is expressed in kidney, liver, and lung [2]. Thyroid hormones have numerous target genes (Table 1), among them in the brain are Reln, which product, Reelin, acts as an extracellular matrix protein produced by neurons and which activity has effects in neuronal migration [23, 26]; Nrgn (RC3/neurogranin), a brain-specific protein kinase C (PKC) substrate, that has been implicated in the hippocampal plasticity process such as long-term potentiation (LTP) [32]; and Shh, a key developmental morphogen, are some of the genes upregulated with T3 treatment. There are also genes downregulated in rats were hypothyroidism was induced (Table 2), for instance, brain-derived neurotrophic factor (BDNF), which has been implicated in structural changes at synapses and Laminin, a guidance molecule in neuronal migration [33, 34].
Thyroid hormones have complex actions because of their impact in other signal transduction pathways and direct interaction with other nuclear receptors [36]. Within the metabolic pathways that are interconnected with the action of thyroid hormones are the retinoic acid and glucocorticoids during neural development. This was demonstrated through gene expression studies of key enzymes involved in the retinoic acid synthesis and degradation such as Aldh1a3 and Aldh1a1 that corresponds to the aldehyde dehydrogenase enzyme. Retinoic acid has a role in relevant processes such as sleep, learning, and memory, and also stimulates MCT8 expression and thyroid transport [37]. Both Aldh1a3 and Aldh1a1 were upregulated and downregulated by T3, respectively. This was related as well with a synergist effect of T3 and dexamethasone (synthetic glucocorticoid), were the latter increases the expression of Aldh1a1. This was all in fine coordination with the subtype of the thyroid hormone receptor at a nuclear level [26]. Moreover, the Klf9 gene, which mediates effects of glucocorticoids on neuronal structure and function, is also upregulated by thyroid hormone. Findings in the hippocampus and cerebellum demonstrate that this interaction occurs through a T3 response element in the 5′ flanking region of the Klf9 gene [26, 29].
Further pathways involving thyroid hormones have been elucidated in other organs such as the liver, were thyroid hormone control cholesterol metabolism through the stimulation of low-density lipoprotein (LDL) receptor expression, the inhibition of the enzyme that degradates cholesterol to bile acids (CYP7A1), and through the crosstalk with proliferator peroxisome receptor (PPAR) elements [38]. These studies emphasize the complexity of thyroid hormone actions and the numerous crosstalk’s that impact in different organs.
Hypothyroidism
Hypothyroidism is a clinical condition in which the thyroid gland produces insufficient levels of the thyroid hormones T3 and T4, leading to high thyroid stimulating hormone (TSH) values, and has a major consequence which is reflected in a slowdown of the cell metabolism. The causes of this condition are iodine deficiency (water, plants, and foods) and autoimmune disease caused by Hashimoto’s thyroiditis or thyrotoxicosis caused by Graves’s disease. The treatment of hypothyroidism is through the administration of levothyroxine, that together with the action of deiodinases of the body, T4 will transform it to T3, were the latter is the hormone that produces the main effect at a transcriptional level. This therapy is capable of regulating the TSH levels to the stipulated “normal values.” Nevertheless, a subgroup of patients still presents symptoms of hypothyroidism [38,39,40]. Hypothyroidism may be triggered in all the stages of development, from pregnancy as subclinical hypothyroidism and congenital hypothyroidism, in which the latter have a greater impact in neurodevelopment retardation and metabolic impairments such as poor feeding and failure in weight gain and growth, up to adulthood where even the subclinical hypothyroidism has been related to depression and dementia [41,42,43]. Thyroid hormones are implicated in metabolic alterations in several organs, where the brain is fundamental.
Hypothyroidism during Development
The effect of a deficiency in thyroid hormones depends on the stage in which is presented; for instance, congenital hypothyroidism can produce impairment of growth and development of the skeletal system, while if it is presented in childhood it could cause poor visuomotor and visuospatial abilities, delayed speech and language development, neuromotor deficiencies, and poor attention and memory skills. In telencephalon, studies have revealed that maternal hypothyroidism during pregnancy leads to an inhibition in neurogenesis and impairments in learning and memory. Furthermore, specifically in the hippocampus, when analyzing structural and growth properties in this region from an early postnatal hypothyroid condition, a significant reduction was found in the surface of the hippocampus, being CA1 the more affected region [44]. Also, studies in early postnatal rat development found that the number of granule cells of the dentate gyrus of the hippocampus is diminished in hypothyroid animals, while in the CA1 region occurs an irreversible neuronal cell death after postnatal hypothyroidism induction. In the CA3 region, there is a reduction of the volume of the pyramidal cell layer, but not in the number of these (Fig. 1) [45, 46].
Hypothyroidism during hippocampus development. Hypothyroidism in the early postnatal stage triggers in the hippocampus structural and growth defects, such as irreversible neuronal death in the CA1 region, decrease of the volume of pyramidal cell layer in CA3, and a decrease in the number of granule cells in the dentate gyrus
The most common method for analyzing the effect in developing animals is by the administration of propylthiouracil (PTU) to pregnant rats and lactating dams through the water. Studies in target genes of thyroid hormones have been made in rat development exposed to PTU, specifically in the regions of the cerebral cortex, corpus callosum, dentate gyrus, and cerebellar vermis. All these areas showed changes in genes involved in neuronal differentiation, development, cell migration, synaptic function, and axonogenesis. Alterations in genes of the ephrin signaling and the ionotropic glutamate receptor AMPA were observed, which are involved in neuronal migration. Furthermore, genes encoding cyclooxygenase 2 (COX2) were downregulated in the hippocampal dentate gyrus, which has been related with neurogenesis [47, 48]. Moreover, studies in vitro revealed that T3 promotes the neuronal differentiation of mouse embryonic neural stem cells. The action was through the inhibition by TRα1 over the JAK/STAT3 signaling activity. The final effect contributes to early neurogenesis in the embryonic telencephalon [49].
Functional properties of the hippocampus observed through electrophysiology have also been addressed. Studies have shown reductions in synaptic transmission and impairment of short-term synaptic plasticity and LTP in the dentate gyrus electrophysiological recordings. The Morris Water Maze task revealed impairment in spatial learning that was dependent of the PTU dose. Also, thyroid hormone insufficiency during late gestational periods has shown permanent alterations in synaptic transmission and plasticity in adult animals in this area [50, 51]. In CA1 recordings, paired pulse inhibition of the population spike is also reduced, but in contrast, LTP magnitude was increased; this is supported by the fact that the phosphorylation of the extracellular signal-regulated kinases was increased. These alterations are hypothesized to be due to differential expression or localization of N-methyl-d-aspartate (NMDA) receptors and voltage-dependent calcium channels (VDCC) on pyramidal cell soma versus dendrites triggered by an early thyroid hormone insufficiency [52].
Also, the administration of low doses of PTU, in the drinking water, from gestational (day 14) to early postnatal ages (21 days) is enough to induce a middle impair in cognitive performance in males. These results were correlated with a decrease in the expression of some neurotransmitters such as glutamate and glycine in the hippocampus. Additionally, a decrease in the expression of N-methyl-D-aspartate receptor subunits, which play a role in the neurotransmission related with the cognitive performance in the hippocampus was observed [53]. Also, the induction of hypothyroidism during pregnancy induces changes in the DNA hypermethylation of some important genes for brain function such us brain-derived neurotrophic factor gene (Bdnf), which is critical for several brain functions including the neuroplasticity related with the process of memory and learning in the hippocampus [54].
The levels of thyroid hormones are critical in all stages; for instance, in hypothyroidism induced during early postnatal stages, several changes in the brain function has been described, including decreases in the cognitive performance and LTP, along with an increase in several markers for oxidative stress such as 4-hydroxy-2-nonenal [55]. Together, all these results confirm the importance of thyroid hormones for brain function in several ages; however, the target for these effects has not been completely studied.
Relationship between Hypothyroidism and Metabolism
Thyroid hormones are crucial for the functioning of almost all organs, and a deregulation in the levels of TH has been described to impact the cardiovascular system, adipose tissue, liver control of glucose levels, growth process, fertility, and the CNS [21, 56,57,58]. For instance, a study in young rats demonstrates that in a dose-dependent manner, PTU has a negative effect on body and organ weights, where kidney and heart decreases their weight, while the testes, brain, and thyroid gland increases [59].
Taking into consideration that glucose is one of the main supply of energy of the body, the regulation and maintenance of glycemic levels is essential for the correct functioning of the organism. THs have a role in the regulation of carbohydrates, and it has been reported that hypothyroidism can reduced the glucose availability, alter the correct absorption of glucose, and delay gluconeogenesis, reducing insulin synthesis. In fact, hypothyroidism has been associated with metabolic diseases, such as diabetes, were thyroid dysfunction has been found in up to an 11% of diabetic patients [56]. Considering that glucose is the main supply of the brain, and cognitive performance depends on it, an energy imbalance may affect major functions in the brain. Studies with PET in patients with hypothyroidism has shown a reduction in brain regional activity, specifically in the bilateral amygdala, hippocampus, perigenual anterior cingulate cortex (ACC), left sub-genual ACC, and right posterior cingulate cortex [60].These regions are essential for emotions, incorporation of attentional information, and memory [61, 62]. Regardless of the imbalance in the levels of thyroid hormones, whether it is a decrease or excess of hormones, the glucose metabolism in the brain is diminished. Patients with hyperthyroidism also present a deregulation in cerebral glucose metabolism, where lower levels have been detected in the limbic system, frontal lobes, and temporal lobes. Improvements after thyroid hormone treatments were observed in memory and constructive abilities; nevertheless, not all of the brain regions return to normal activity levels; this could be due to mood disorders for instance; therefore, further studies to reach an optimal conditions are needed [63]. Moreover, a study in which the glucose transporter 1 (GLUT1) was analyzed in hypo-hyperthyroid rats in different age conditions showed decreased levels in the expression of this transporter in both conditions in young rats, while it was increased in hyperthyroid aged rats; therefore, age condition in glucose metabolism must be considered as well. Mice with primary hypothyroidism in early postnatal stages also showed a decline in GLUT1, while GLUT3 has no changes, and hexokinase enzyme activity was enhanced by a compensatory mechanism [64, 65].
Adult cognitive functions are also imbalanced in hypothyroid conditions. The hippocampus is a region of the brain where neurogenesis occurs, a process that has an important role in learning and memory [66]. Studies with TRα1 and TRα2 knockouts reveal a key role of TRα1 in the regulation of the survival of adult hippocampal progenitors, indicating that a deficiency in this nuclear receptor enhances hippocampal progenitor survival; this has been explained by the possible role of TRα1 in regulating commitment and maturation of neuronal progenitors [67, 68].
Another relevant area has been investigated, this is the hypothalamus, were a deregulation of thyroid hormones leads to energy imbalance in thermogenesis. A model with a mutation in TRα1 presents a hypermetabolic state with increased thermogenesis and energy expenditure. This has direct effects in the weight of the animals, demonstrated by the fact that a high-fat diet (HFD) did not normalize the body weight; therefore, the central effect of thyroid hormones overrides the peripheral effects [12, 69]. Furthermore, there is evidence of the pathways involved in the regulation of this energy balance, where energy expenditure is controlled via AMP kinase and feeding with the mTOR pathway [70].
In addition, diabetes mellitus (DM), a highly prevalent and complex metabolic disorder in modern societies worldwide, has been associated with higher risk of cognitive impairments, dementia, and other behavioral/neurological changes [71]. These alterations have been associated with an increase in the levels of insulin but correlated with an increase in the insulin resistance. In fact, diabetic rats induced by alloxan present hypothyroidism and increased expression of proinflammatory cytokines, which are known to reduce insulin sensitivity [72]. In diabetic rate treated with T3 for 4 weeks was observed a recover of the insulin signaling and the glucose levels [73].
Relationship of Hypothyroidism with Brain Disorders
As previously described, hypothyroidism affects several brain functions during development and adulthood, therefore impairs a great number of pathways in diseases such as congenital hypothyroidism and autism in childhood, but also in elderly diseases such as Parkinson’s and Alzheimer’s diseases (Fig. 2) [74,75,76].
Hypothyroidism and neurodegenerative diseases. Hypothyroidism is related to several neurodegenerative diseases, among them are autism, AD, and Parkinson’s disease. In autism, specifically in the putamen region, a reduction in thyroid hormone target genes are downregulated, such as TTRa, TTRb, TTRc, BDNF, Relin, and MCT8, while in the orbitofrontal cortex occurs an increase in the oxidative stress marker 3-nitrotyrosine. In AD patients, the levels of rT3 and rT3/T4 are increased in the cerebrospinal fluid. Moreover, a negative regulation of T3 over the transcriptional activity of APP has been observed, along with an increase of the processing in APP, therefore Aβ amount increases. Also, tau hyperphosphorylation, the neuropathological marker of AD, increases in rats with hypothyroidism. Finally, in early Parkinson’s disease, the increase of 3-nytrotyrosine correlates with a decrease in thyroperoxidase
Autism and Thyroid Dysfunction
Nowadays, the idea of a relationship between thyroid hormone deregulation and autism spectrum disorders (ASD) is gaining force. ASD includes autism and Asperger’s syndrome, and these are neurobiological disorders with a developmental disturbance of communication that impairs social interaction and communication. It affects 1 in 132 persons, with a higher prevalence in males. The degree of severity of the symptoms may vary, from intellectual and language disability up to epilepsy. The specific cause of this disorder is yet unknown, but it has a genetic component, and also it is thought that a combination of genes with the environment may contribute to the severity of this disorder [77]. Considering that human thyroid hormones are essential for brain development, an impairment of TH during pregnancy has been associated with autism. Evidence shows association between family history of hypothyroidism with an increase in the risk of childhood autism, with a higher risk in children with maternal hypothyroidism. More specifically, a postmortem study of patients with ASD was conducted to analyze thyroid hormones target genes. Results showed a brain region-specific disruption of TH and gene expression in autism, specifically in the putamen, a region that influences reinforcement and implicit learning, the levels of genes such as TTR (a, b, and c), BDNF, RELN, and MCT8 where downregulated [78,79,80]. Interestingly, the levels of T3 were decreased and the oxidative stress marker, 3-nitrotyrosine (3-NT), was increased in the orbitofrontal cortex area in male patients with ASD (Fig. 2).
A Danish nationwide study revealed increased risk of ASD when the mother presented hypothyroidism during pregnancy, and a Rotterdam study showed a fourfold increase in the risk of autism in children with maternal hypothyroxinemia [76, 81]. Furthermore, autoimmune disease has been associated with ASD, through the measure of maternal TPO-Ab antibody. Specifically, autoimmune thyroid disease has been associated with ASD that presented regression, a phenomenon were established skills in communication or social interaction are lost [82, 83].
Parkinson’s Disease and Hypothyroidism
Parkinson’s disease (PD) is the second most prevalent neurodegenerative disease presented mainly in later years of life, characterized by impaired motor function, slow movement, tremor, and postural instability, among others. Different causes have been associated to this disease, such as psychiatric disorders, socioeconomic status, autoimmune disease, and hormone dysfunctions [84, 85].
Parkinson’s disease and hypothyroidism are two disorders that are common in the elderly population, and they share many clinical characteristics. In the past, several studies has found a relationship between hypothyroidism and PD, for instance Berger and Kelley [86] reported that hypothyroidism has a 6.6% of incidence among Parkinson’s patients, but in the recent years no significant difference has been found between control and PD patients regarding to hypothyroidism, except for a nationwide epidemiological study from Sweden with 310,000 patients, were among the autoimmune disease as a risk factor for PD, Hashimoto’s disease/hypothyroidism is one of them [75]. In PD, several clinical biomarkers have been proposed to detect this disease in early stages, where one of them is the presence of the oxidative marker 3-NT in serum in early Parkinsonian patients. In relation with thyroid hormones, one study described an increase of the enzyme thyroperoxidase that was negatively correlated with serum 3-nitrotyrosine protein in patients with early Parkinson’s disease (Fig. 2); therefore, they propose a possible role of the thyroid gland and thyroperoxidase in the nitrosylation of serum proteins, thus influencing Parkinsonian nitrosative stress [87].
Hypothyroidism and Alzheimer’s Disease
Nowadays the population aging is advancing, and dementia affects more than 25 million people worldwide were Alzheimer’s disease (AD), a neurodegenerative disease, is the most common form of dementia, having an incidence in people over 65 of about 20 per 1000 person-year and 15 per 1000 in Europe and the USA, respectively. The amount of people affected increases almost exponentially when the age is above 85. AD is still a disease without any medical treatment, which clinically is diagnosed once the pathology has already began [88]. It is characterized by a decline in cognitive functions and physical disability, leading to institutionalization and a deterioration in quality of life. Different factors may impact in the development of the disease, such as genetic, vascular, psychosocial, and dietary, among others. Neuropathologically, it is characterized by the presence of aggregated amyloid protein, tau hyperphosphorylation, increased reactive oxygen species, and decreased glucose consumption [88,89,90,91].
In the last years, thyroid hormone imbalance in AD has been addressed as another factor to consider in this pathology, considering that both diseases increase their prevalence with age. Thyroid hormone levels in the cerebrospinal fluid from diagnosed AD patients revealed increased rT3 levels and increased rT3 to T4 ratio, regardless the euthyroid systemic condition when these hormones were measured in serum. This is in accordance with findings of an association between AD and abnormal function of the hypothalamic-pituitary thyroid axis. This is fundamental when considering that T3 negatively regulates the transcriptional activity of the β-amyloid precursor protein gene (APP), an important risk factor of AD. This was observed in human neuroblastoma cells and in primary cultures of rat neurons. Further studies revealed that hypothyroidism induced the amyloidogenic pathway, increasing the amount of Aβ peptides in the hippocampus. In addition, high and low levels of TSH have been associated with increased risk of developing AD in women (Fig. 2) [74, 92,93,94,95].
Further studies in adult rats with hypothyroidism revealed that several neuropathological signs of Alzheimer were present, such as tau hyperphosphorylation in the hippocampus, increased levels of proinflammatory cytokines, and altered expression of signaling molecules implicated with synaptic plasticity and memory. In addition, spatial memory was impaired [14].
In AD, several studies have revealed a deregulation in molecular pathways, where the Wnt pathway has gained force in the last years [96, 97]. The Wnt pathway is activated by Wnt ligands and exerts an effect in many cellular processes during embryonic development, such as cell proliferation, cell polarity, cell migration, and neural patterning and also influences adult homeostasis. This pathway may be classified as a canonical (Wnt/β-catenin) or non-canonical pathway, where the first one regulates the amount of β-catenin, a protein that functions as a transcriptional co-activator of gene expression of the T cell factor/lymphoid enhancer factor (TCF/LEF) [98, 99]. Normally, the β-catenin protein is being degradated by a destruction complex, in which one of the proteins, the glycogen synthase kinase 3 (GSK3), is constantly phosphorylating β-catenin, leading to its proteasomal degradation in the cytoplasm and therefore preventing to reach the nucleus. When a Wnt ligand binds to a Frizzled receptor and its co-receptor, it activates the Wnt pathway and inhibits the phosphorylation of β-catenin, with the consequent activation of gene expression. The non-canonical Wnt pathway is divided into the Planar Cell Polarity pathway (PCP) and the Wnt/Ca+2 pathway. The PCP pathway activates the small GTPases Rho and Rac that activates other pathways related to actin polymerization. The Wnt/Ca+2 pathway depends on G proteins that activates the enzyme phospholipase C, with the consequent production of diacylglycerol and inositol triphosphate (IP3). IP3 is capable of stimulate the intracellular Ca+2 release from the ER, that in turn activates proteins sensitive to Ca+2, such as protein kinase C (PKC) and calcium/calmodulin-dependent kinase II (CamKII) and calcineurin [100,101,102]. Considering that both the Wnt pathway and thyroid hormones has as targets the expression of genes, a possible interaction of these two pathways has been proposed.
Wnt and Thyroid Hormones Relation
Currently, the evidence of an interaction between thyroid hormones and the Wnt pathway is validated by several studies in tissues and cell lines. In thyroid cell line, Pax8, a gene involved in morphogenesis of the thyroid gland was found to modulate the expression of Wnt4 at a transcriptional level, through the direct binding of the promotor of Wnt4, and the consequent activation of the expression of this pathway [103, 104].
The regulation that thyroid hormones has over the Wnt pathway has been studied to analyze the temporal expression of these genes. A study that uses the cell line GC, that expresses functional thyroid hormone receptors (TRs), revealed through microarray that T3 proliferation induction repressed the expression of key regulators of the Wnt pathway and inhibits transcriptional elements of the β-catenin/TCF complex. Specifically, the following components were downregulated: β-catenin, TCF4, Dishevelled-1, Frizzled, and c-jun. In accordance, the expression of axin and APC (repressors of the Wnt pathway) were increased (Fig. 3) [105].
Relation between thyroid hormones and the Wnt pathway. Several components of the Wnt pathway are modulated by T3 availability. In the GC cell line, the stimulation with T3 triggers a decreased in Fzzd, Dvl, β-catenin, and TCF4, while an increase of APC and Axin is produced. In bone formation, the Wnt pathway is normally activated because of the stabilization of β-catenin by TRβ. Contrary, T3 treatment inhibits this pathway, most likely through the competition for TRβ. In oligodendrocytes, the stimulation with T3, produced an increase of the endogenous inhibitor of the Wnt pathway, sFRP1
Thyroid hormones have a role in bone formation during growth and Wnt canonical signaling has also been implicated in this process. During this process, the Wnt pathway is activated, increasing bone mass through the stimulation of osteoblastic bone formation, most likely due to physical interaction between TRβ and β-catenin, that stabilizes β-catenin. In contrast, T3 treatment inhibited the Wnt signaling due to the competition of the binding to the receptor. Studies in transgenic mice ThrbPV/PV, characterized by an impaired pituitary-thyroid axis and high levels of thyroid hormones, in which the mice developed thyroid carcinogenesis, revealed that T3 failed in the inhibition of the Wnt pathway [106, 107]. Furthermore, the induction of hyper- or hypothyroidism correlated with this results, while the hyperthyroid condition produces an increase in bone formation that correlated with low levels of DKK1; the contrary occurred with the hypothyroid condition (Fig. 3) [108]. Studies in hepatocytes has also demonstrate the role of T3 over the Wnt pathway through the modulation of other genes; for instance, T3 union to the thyroid receptor activates the transcription of Klf9, and both transcription factors modulate pathways such as Notch, FGF (AD amyloid secretase pathway), TGFβ, and the Wnt pathways [109]. Since Klf9 is a relevant gene in the modulation of neurogenesis and glucocorticoids in brain, the interaction of T3-Klf9-Wnt could be an interesting pathway to study in the CNS.
There are few studies in CNS that relates Wnt with thyroid hormones. Among these, experiments in oligodendrocyte progenitor cells treated with T3 hormone showed an increase in secreted Frizzled-related protein-1 (SFRP1), an inhibitor of the Wnt pathway [110].
Conclusions
Thyroid hormones are critical for the correct brain function, modulating critical roles including neurogenesis, cognitive performance, and energy brain metabolism (Fig. 4). The blood levels of T3 and T4 are regulated by several mechanisms that involve the participation of several tissues. However, for several conditions including genetic patterns or environment interaction, a decrease in blood levels of thyroid hormones may be the trigger, causing a wide deregulation downstream. When this hypothyroidism is established in the early stages of development, it produces brain alterations that could affect patients even in adult stages. Until now, several reports has correlated the function of thyroid hormones with several neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease and autism. Nevertheless, little is known about the specific cellular mechanism that could be altered by the dysfunction induced by the decrease in thyroid hormone levels. Therefore, it is important to develop efficient tools against the consequences of hypothyroidism in early stages of life and in this manner prevent some brain pathologies in the adulthood.
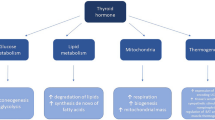
Effects of thyroid hormones in normal and pathologic conditions. The thyroid gland is in charge of thyroid hormone production, which impacts the general metabolism in aspects such as bone formation, growth, carbohydrates, and lipid metabolism. Also, in normal conditions, controls brain process, such as neurogenesis, synaptic transmission, synaptic structure, cellular proliferation and migration, and glucose metabolism (in this case, the thyroid hormones could affect several parameters including expression of glucose transporters, enzyme activities, and mitochondrial biogenesis). Nevertheless, these hormones are also involved in neurological disorders, such as AD, depression, and autism spectrum disorders, among others
References
Bernal J (2000) Thyroid hormones in brain development and function. Endotext 3:1–48. https://doi.org/10.1038/ncpendmet0424
Cheng SY, Leonard JL, Davis PJ (2010) Molecular aspects of thyroid hormone actions. Endocr Rev 31:139–170
Angelousi A, Kassi E, Nasiri-Ansari N, Weickert MO, Randeva H, Kaltsas G (2018) Clock genes alterations and endocrine disorders. Eur J Clin Invest 48:e12927. https://doi.org/10.1111/eci.12927
Lazarus JH (2002) Epidemiology and prevention of thyroid disease in pregnancy. Thyroid 12:861–865. https://doi.org/10.1089/105072502761016485
Vanderpump MPJ (2011) The epidemiology of thyroid disease. Br Med Bull 99:39–51. https://doi.org/10.1093/bmb/ldr030
Korevaar TIM, Muetzel R, Medici M, Chaker L, Jaddoe VWV, de Rijke YB, Steegers EAP, Visser TJ et al (2016) Association of maternal thyroid function during early pregnancy with offspring IQ and brain morphology in childhood: a population-based prospective cohort study. Lancet Diabetes Endocrinol 4:35–43. https://doi.org/10.1016/S2213-8587(15)00327-7
Modesto T, Tiemeier H, Peeters RP, Jaddoe VWV, Hofman A, Verhulst FC, Ghassabian A (2015) Maternal mild thyroid hormone insufficiency in early pregnancy and attention-deficit/hyperactivity disorder symptoms in children. JAMA Pediatr 169:838–845. https://doi.org/10.1001/jamapediatrics.2015.0498
Zimmermann MB (2009) Iodine deficiency. Endocr Rev 30:376–408
Koromilas C, Liapi C, Schulpis KH, Kalafatakis K, Zarros A, Tsakiris S (2010) Structural and functional alterations in the hippocampus due to hypothyroidism. Metab Brain Dis 25:339–354
Yu J, Tang Y-Y, Feng H-B, Cheng X-X (2014) A behavioral and micro positron emission tomography imaging study in a rat model of hypothyroidism. Behav Brain Res 271:228–233. https://doi.org/10.1016/j.bbr.2014.06.019
Jang C, Oh SF, Wada S, Rowe GC, Liu L, Chan MC, Rhee J, Hoshino A et al (2016) A branched-chain amino acid metabolite drives vascular fatty acid transport and causes insulin resistance. Nat Med 22:421–426. https://doi.org/10.1038/nm.4057
Martínez-Sánchez N, Alvarez CV, Fernø J, Nogueiras R, Diéguez C, López M (2014) Hypothalamic effects of thyroid hormones on metabolism. Best Pract Res Clin Endocrinol Metab 28:703–712
Munhoz RP, Teive HAG, Troiano AR et al (2004) Parkinson’s disease and thyroid dysfunction. Parkinsonism Relat Disord 10:381–383. https://doi.org/10.1016/j.parkreldis.2004.03.008
Chaalal A, Poirier R, Blum D, Gillet B, le Blanc P, Basquin M, Buée L, Laroche S et al (2014) PTU-induced hypothyroidism in rats leads to several early neuropathological signs of Alzheimer’s disease in the hippocampus and spatial memory impairments. Hippocampus 24:1381–1393. https://doi.org/10.1002/hipo.22319
Garcia-Moreno JM, Chacon J (2002) Hypothyroidism concealed by Parkinson’s disease. Rev Neurol 35:741–742
Bernal J, Guadaño-Ferraz A, Morte B (2015) Thyroid hormone transporters-functions and clinical implications. Nat Rev Endocrinol 11:406–417
Hammes SR, Davis PJ (2015) Overlapping nongenomic and genomic actions of thyroid hormone and steroids. Best Pract Res Clin Endocrinol Metab 29:581–593. https://doi.org/10.1016/j.beem.2015.04.001
Mullur R, Liu Y-Y, Brent GA (2014) Thyroid hormone regulation of metabolism. Physiol Rev 94:355–382. https://doi.org/10.1152/physrev.00030.2013
Bowers J, Terrien J, Clerget-Froidevaux MS, Gothié JD, Rozing MP, Westendorp RGJ, van Heemst D, Demeneix BA (2013) Thyroid hormone signaling and homeostasis during aging. Endocr Rev 34:556–589
Jansen J, Friesema ECH, Milici C, Visser TJ (2005) Thyroid hormone transporters in health and disease. Thyroid 15:757–768. https://doi.org/10.1089/thy.2005.15.757
Bernal J (2002) Action of thyroid hormone in brain. J Endocrinol Investig 25:268–288. https://doi.org/10.1007/BF03344003
Ortiga-Carvalho TM, Sidhaye AR, Wondisford FE (2014) Thyroid hormone receptors and resistance to thyroid hormone disorders. Nat Rev Endocrinol 10:582–591
Alvarez-Dolado M, Ruiz M, Del Río JA et al (1999) Thyroid hormone regulates reelin and dab1 expression during brain development. J Neurosci 19:6979–6993
Sui L, Ren W-W, Li B-M (2010) Administration of thyroid hormone increases reelin and brain-derived neurotrophic factor expression in rat hippocampus in vivo. Brain Res 1313:9–24. https://doi.org/10.1016/j.brainres.2009.12.010
Pathak A, Sinha RA, Mohan V, Mitra K, Godbole MM (2011) Maternal thyroid hormone before the onset of fetal thyroid function regulates reelin and downstream signaling cascade affecting neocortical neuronal migration. Cereb Cortex 21:11–21. https://doi.org/10.1093/cercor/bhq052
Gil-Ibáñez P, Bernal J, Morte B (2014) Thyroid hormone regulation of gene expression in primary cerebrocortical cells: role of thyroid hormone receptor subtypes and interactions with retinoic acid and glucocorticoids. PLoS One 9. https://doi.org/10.1371/journal.pone.0091692
Diez D, Grijota-Martinez C, Agretti P, de Marco G, Tonacchera M, Pinchera A, Morreale de Escobar G, Bernal J et al (2008) Thyroid hormone action in the adult brain: gene expression profiling of the effects of single and multiple doses of triiodo-L-thyronine in the rat striatum. Endocrinology 149:3989–4000. https://doi.org/10.1210/en.2008-0350
Potter GB, Zarach JM, Sisk JM, Thompson CC (2002) The thyroid hormone-regulated corepressor hairless associates with histone deacetylases in neonatal rat brain. Mol Endocrinol 16:2547–2560. https://doi.org/10.1210/me.2002-0115
Bonett RM, Hu F, Bagamasbad P, Denver RJ (2009) Stressor and glucocorticoid-dependent induction of the immediate early gene kruppel-like factor 9: implications for neural development and plasticity. Endocrinology 150:1757–1765. https://doi.org/10.1210/en.2008-1441
Gilbert ME, Sanchez-Huerta K, Wood C (2016) Mild thyroid hormone insufficiency during development compromises activity-dependent neuroplasticity in the hippocampus of adult male rats. Endocrinology 157:774–787. https://doi.org/10.1210/en.2015-1643
Desouza LA, Sathanoori M, Kapoor R, Rajadhyaksha N, Gonzalez LE, Kottmann AH, Tole S, Vaidya VA (2011) Thyroid hormone regulates the expression of the sonic hedgehog signaling pathway in the embryonic and adult mammalian brain. Endocrinology 152:1989–2000. https://doi.org/10.1210/en.2010-1396
Kaleka KS, Gerges NZ (2016) Neurogranin restores amyloid β-mediated synaptic transmission and long-term potentiation deficits. Exp Neurol 277:115–123. https://doi.org/10.1016/j.expneurol.2015.12.013
Farwell AP, Tranter MP, Leonard JL (1995) Thyroxine-dependent regulation of integrin-laminin interactions in astrocytes. Endocrinology 136:3909–3915. https://doi.org/10.1210/endo.136.9.7649099
Farwell AP, Dubord-Tomasetti SA (1999) Thyroid hormone regulates the expression of laminin in the developing rat cerebellum. Endocrinology 140:4221–4227. https://doi.org/10.1210/endo.140.9.7007
Royland JE, Parker JS, Gilbert ME (2008) A genomic analysis of subclinical hypothyroidism in hippocampus and neocortex of the developing rat brain. J Neuroendocrinol 20:1319–1338. https://doi.org/10.1111/j.1365-2826.2008.01793.x
Brent GA (2012) Mechanisms of thyroid hormone action. J Clin Invest 122:3035–3043. https://doi.org/10.1172/JCI60047
Dräger UC (2006) Retinoic acid signaling in the functioning brain. Sci STKE 2006(324):pe10
Fuhrer D, Brix K, Biebermann H (2015) Understanding the healthy thyroid state in 2015. Eur Thyroid J 4:1–8. https://doi.org/10.1159/000431318
Gereben B, McAninch EA, Ribeiro MO, Bianco AC (2015) Scope and limitations of iodothyronine deiodinases in hypothyroidism. Nat Rev Endocrinol 11:642–652
Saravanan P, Chau WF, Roberts N, Vedhara K, Greenwood R, Dayan CM (2002) Psychological well-being in patients on “adequate” doses of L-thyroxine: results of a large, controlled community-based questionnaire study. Clin Endocrinol 57:577–585. https://doi.org/10.1046/j.1365-2265.2002.01654.x
Blum MR, Wijsman LW, Virgini VS, Bauer DC, den Elzen WPJ, Jukema JW, Buckley BM, de Craen AJM et al (2016) Subclinical thyroid dysfunction and depressive symptoms among the elderly: a prospective cohort study. Neuroendocrinology 103:291–299. https://doi.org/10.1159/000437387
Jia Y, Zhong S, Wang Y, Liu T, Liao X, Huang L (2015) The correlation between biochemical abnormalities in frontal white matter, hippocampus and serum thyroid hormone levels in first-episode patients with major depressive disorder. J Affect Disord 180:162–169. https://doi.org/10.1016/j.jad.2015.04.005
Kratzsch J, Pulzer F (2008) Thyroid gland development and defects. Best Pract Res Clin Endocrinol Metab 22:57–75. https://doi.org/10.1016/j.beem.2007.08.006
Farahvar A, Meisami E (2007) Novel two-dimensional morphometric maps and quantitative analysis reveal marked growth and structural recovery of the rat hippocampal regions from early hypothyroid retardation. Exp Neurol 204:541–555. https://doi.org/10.1016/j.expneurol.2006.10.012
Madeira MD, Cadete-Leite A, Andrade JP, Paula-Barbosa MM (1991) Effects of hypothyroidism upon the granular layer of the dentate gyrus in male and female adult rats: a morphometric study. J Comp Neurol 314:171–186. https://doi.org/10.1002/cne.903140116
Madeira MD, Sousa N, Lima-Andrade MT, Calheiros F, Cadete-Leite A, Paula-Barbosa MM (1992) Selective vulnerability of the hippocampal pyramidal neurons to hypothyroidism in male and female rats. J Comp Neurol 322:501–518. https://doi.org/10.1002/cne.903220405
Rodger J, Salvatore L, Migani P (2012) Should i stay or should i go? Ephs and ephrins in neuronal migration. NeuroSignals 20:190–201. https://doi.org/10.1159/000333784
Shiraki A, Saito F, Akane H, Takeyoshi M, Imatanaka N, Itahashi M, Yoshida T, Shibutani M (2014) Expression alterations of genes on both neuronal and glial development in rats after developmental exposure to 6-propyl-2-thiouracil. Toxicol Lett 228:225–234. https://doi.org/10.1016/j.toxlet.2014.04.018
Chen C, Zhou Z, Zhong M, Zhang Y, Li M, Zhang L, Qu M, Yang J et al (2012) Thyroid hormone promotes neuronal differentiation of embryonic neural stem cells by inhibiting STAT3 signaling through TRα1. Stem Cells Dev 21:2667–2681. https://doi.org/10.1089/scd.2012.0023
Gilbert ME, Sui L (2006) Dose-dependent reductions in spatial learning and synaptic function in the dentate gyrus of adult rats following developmental thyroid hormone insufficiency. Brain Res 1069:10–22. https://doi.org/10.1016/j.brainres.2005.10.049
Gilbert ME (2004) Alterations in synaptic transmission and plasticity in area CA1 of adult hippocampus following developmental hypothyroidism. Dev Brain Res 148:11–18. https://doi.org/10.1016/j.devbrainres.2003.09.018
Sui L, Anderson WL, Gilbert ME (2005) Impairment in short-term but enhanced long-term synaptic potentiation and ERK activation in adult hippocampal area CA1 following developmental thyroid hormone insufficiency. Toxicol Sci 85:647–656. https://doi.org/10.1093/toxsci/kfi095
Amano I, Takatsuru Y, Khairinisa MA, et al (2018) Effects of mild perinatal hypothyroidism on cognitive function of adult male offspring. Endocrinology 159(4):1910–1921. https://doi.org/10.1210/en.2017-03125
Kawahori K, Hashimoto K, Yuan X et al (2018) Mild maternal hypothyroxinemia during pregnancy induces persistent DNA hypermethylation in the hippocampal brain-derived neurotrophic factor gene in mouse offspring. Thyroid 28:395–406. https://doi.org/10.1089/thy.2017.0331
Salazar P, Cisternas P, Codocedo JF, Inestrosa NC (2017) Induction of hypothyroidism during early postnatal stages triggers a decrease in cognitive performance by decreasing hippocampal synaptic plasticity. Biochim Biophys Acta Mol basis Dis 1863:870–883. https://doi.org/10.1016/j.bbadis.2017.01.002
Wang C (2013) The relationship between type 2 diabetes mellitus and related thyroid diseases. J Diabetes Res 2013:390534
Iwen KA, Schröder E, Brabant G (2013) Thyroid hormones and the metabolic syndrome. Eur Thyroid J 2:83–92. https://doi.org/10.1159/000351249
Wakim AN, Polizotto SL, Buffo MJ, Marrero MA, Burholt DR (1993) Thyroid hormones in human follicular fluid and thyroid hormone receptors in human granulosa cells. Fertil Steril 59:1187–1190. https://doi.org/10.1016/S0015-0282(16)55974-3
Mellert W, Deckardt K, Walter J, Gfatter S, van Ravenzwaay B (2003) Detection of endocrine-modulating effects of the antithyroid acting drug 6-propyl-2-thiouracil in rats, based on the “Enhanced OECD Test Guideline 407”. Regul Toxicol Pharmacol 38:368–377
Bauer M, Silverman DHS, Schlagenhauf F, London ED, Geist CL, van Herle K, Rasgon N, Martinez D et al (2009) Brain glucose metabolism in hypothyroidism: a positron emission tomography study before and after thyroid hormone replacement therapy. J Clin Endocrinol Metab 94:2922–2929. https://doi.org/10.1210/jc.2008-2235
Burgess N, Maguire EA, O’Keefe J (2002) The human hippocampus and spatial and episodic memory. Neuron 35:625–641
Davidson RJ, Lewis DA, Alloy LB, Amaral DG, Bush G, Cohen JD, Drevets WC, Farah MJ et al (2002) Neural and behavioral substrates of mood and mood regulation. Biol Psychiatry 52:478–502
Miao Q, Zhang S, Guan YH, Ye HY, Zhang ZY, Zhang QY, Xue RD, Zeng MF et al (2011) Reversible changes in brain glucose metabolism following thyroid function normalization in hyperthyroidism. AJNR Am J Neuroradiol 32:1034–1042. https://doi.org/10.3174/ajnr.A2449
Khan JY, Rajakumar RA, Devaskar UP, Weissfeld LA, Devaskar SU (1999) Effect of primary congenital hypothyroidism upon expression of genes mediating murine brain glucose uptake. Pediatr Res 45:718–725
Mooradian AD, Girgis W, Shah GN (1997) Thyroid hormone-induced GLUT-1 expression in rat cerebral tissue: effect of age. Brain Res 747:144–146. https://doi.org/10.1016/S0006-8993(96)01110-9
Yau S, Li A, So K-F (2015) Involvement of adult hippocampal neurogenesis in learning and forgetting. Neural Plast 2015:717958–717913. https://doi.org/10.1155/2015/717958
Kapoor R, van Hogerlinden M, Wallis K, Ghosh H, Nordstrom K, Vennstrom B, Vaidya VA (2010) Unliganded thyroid hormone receptor alpha1 impairs adult hippocampal neurogenesis. FASEB J 24:4793–4805. https://doi.org/10.1096/fj.10-161802
Munoz A, Wrighton C, Seliger B et al (1993) Thyroid hormone receptor/c-erbA: Control of commitment and differentiation in the neuronal/chromaffin progenitor line PC12. J Cell Biol 121:423–438
Sjögren M, Alkemade A, Mittag J, Nordström K, Katz A, Rozell B, Westerblad H, Arner A et al (2007) Hypermetabolism in mice caused by the central action of an unliganded thyroid hormone receptor α1. EMBO J 26:4535–4545. https://doi.org/10.1038/sj.emboj.7601882
Varela L, Martínez-Sánchez N, Gallego R, Vázquez MJ, Roa J, Gándara M, Schoenmakers E, Nogueiras R et al (2012) Hypothalamic mTOR pathway mediates thyroid hormone-induced hyperphagia in hyperthyroidism. J Pathol 227:209–222. https://doi.org/10.1002/path.3984
Biessels GJ, Kappelle LJ (2005) Increased risk of Alzheimer’s disease in type II diabetes: insulin resistance of the brain or insulin-induced amyloid pathology? Biochem Soc Trans 33:1041–1044
Panveloski-Costa AC, Silva Teixeira S, Ribeiro IMR, Serrano-Nascimento C, das Neves RX, Favaro RR, Seelaender M, Antunes VR et al (2016) Thyroid hormone reduces inflammatory cytokines improving glycaemia control in alloxan-induced diabetic wistar rats. Acta Physiol 217:130–140. https://doi.org/10.1111/apha.12647
Prieto-Almeida F, Panveloski-Costa AC, Crunfli F, da Silva Teixeira S, Nunes MT, Torrão AS (2018) Thyroid hormone improves insulin signaling and reduces the activation of neurodegenerative pathway in the hippocampus of diabetic adult male rats. Life Sci 192:253–258. https://doi.org/10.1016/j.lfs.2017.11.013
Tan ZS, Beiser A, Vasan RS et al (2008) Thyroid function and the risk of Alzheimer disease: The Framingham study. Arch Intern Med 168:1514–1520. https://doi.org/10.1001/archinte.168.14.1514
Li X, Sundquist J, Sundquist K (2012) Subsequent risks of Parkinson disease in patients with autoimmune and related disorders: a nationwide epidemiological study from Sweden. Neurodegener Dis 10:277–284. https://doi.org/10.1159/000333222
Andersen SL, Laurberg P, Wu CS, Olsen J (2014) Attention deficit hyperactivity disorder and autism spectrum disorder in children born to mothers with thyroid dysfunction: a Danish nationwide cohort study. BJOG 121:1365–1374. https://doi.org/10.1111/1471-0528.12681
Baxter AJ, Brugha TS, Erskine HE, Scheurer RW, Vos T, Scott JG (2014) The epidemiology and global burden of autism spectrum disorders. Psychol Med 45:1–13. https://doi.org/10.1017/s003329171400172x
Khan A, Harney JW, Zavacki AM, Sajdel-Sulkowska EM (2014) Disrupted brain thyroid hormone homeostasis and altered thyroid hormone-dependent brain gene expression in autism spectrum disorders. J Physiol Pharmacol 65:257–272
Sadamatsu M, Kanai H, Xu X, Liu Y, Kato N (2006) Review of animal models for autism: implication of thyroid hormone. Congenit Anom (Kyoto) 46:1–9
Wu S, Ding Y, Wu F, Li R, Xie G, Hou J, Mao P (2015) Family history of autoimmune diseases is associated with an increased risk of autism in children: a systematic review and meta-analysis. Neurosci Biobehav Rev 55:322–332
Roman GC, Ghassabian A, Bongers-Schokking JJ et al (2013) Association of gestational maternal hypothyroxinemia and increased autism risk. Ann Neurol 74:733–742. https://doi.org/10.1002/ana.23976
Brown AS, Surcel H-M, Hinkka-Yli-Salomäki S, Cheslack-Postava K, Bao Y, Sourander A (2015) Maternal thyroid autoantibody and elevated risk of autism in a national birth cohort. Prog Neuro-Psychopharmacol Biol Psychiatry 57:86–92. https://doi.org/10.1016/j.pnpbp.2014.10.010
Molloy CA, Morrow AL, Meinzen-Derr J, Dawson G, Bernier R, Dunn M, Hyman SL, McMahon WM et al (2006) Familial autoimmune thyroid disease as a risk factor for regression in children with autism spectrum disorder: a CPEA study. J Autism Dev Disord 36:317–324. https://doi.org/10.1007/s10803-005-0071-0
Heinzel S, Roeben B, Ben-Shlomo Y, Lerche S, Alves G, Barone P, Behnke S, Berendse HW et al (2016) Prodromal markers in Parkinson’s disease: limitations in longitudinal studies and lessons learned. Front Aging Neurosci 8:147
Sveinbjornsdottir S (2016) The clinical symptoms of Parkinson’s disease. J Neurochem 139(Suppl 1):318–324
Berger JR, Kelley RE (1981) Thyroid function in Parkinson disease. Neurology 31:93–95. https://doi.org/10.1212/WNL.31.1.93
Fernández E, García-Moreno J-M, Martín de Pablos A, Chacón J (2014) May the thyroid gland and thyroperoxidase participate in nitrosylation of serum proteins and sporadic Parkinson’s disease? Antioxid Redox Signal 21:2143–2148. https://doi.org/10.1089/ars.2014.6072
Querfurth HW, LaFerla FM (2010) Alzheimer’s disease. N Engl J Med 362:329–344. https://doi.org/10.1056/NEJMra0909142
Cerpa W, Dinamarca MC, Inestrosa NC et al (2008) Structure-function implications in Alzheimer’s disease: effect of Abeta oligomers at central synapses. Curr Alzheimer Res 5:233–243. https://doi.org/10.2174/156720508784533321
Chen Z, Zhong C (2013) Decoding Alzheimer’s disease from perturbed cerebral glucose metabolism: implications for diagnostic and therapeutic strategies. Prog Neurobiol 108:21–43. https://doi.org/10.1016/j.pneurobio.2013.06.004
Qiu C, Kivipelto M, Von Strauss E (2009) Epidemiology of Alzheimer’s disease: occurrence, determinants, and strategies toward intervention. Dialogues Clin Neurosci 11:111–128
Contreras-Jurado C, Pascual A (2012) Thyroid hormone regulation of APP (β-amyloid precursor protein) gene expression in brain and brain cultured cells. Neurochem Int 60:484–487. https://doi.org/10.1016/j.neuint.2012.01.027
Ghenimi N, Alfos S, Redonnet A et al (2010) Adult-onset hypothyroidism induces the amyloidogenic pathway of amyloid precursor protein processing in the rat hippocampus. J Neuroendocrinol 22:951–959. https://doi.org/10.1111/j.1365-2826.2010.02002.x
Sampaolo S, Campos-Barros A, Mazziotti G, Carlomagno S, Sannino V, Amato G, Carella C, di Iorio G (2005) Increased cerebrospinal fluid levels of 3,3′,5′-triiodothyronine in patients with Alzheimer’s disease. J Clin Endocrinol Metab 90:198–202. https://doi.org/10.1210/jc.2004-1083
Yong-Hong L, Xiao-Dong P, Chang-Quan H et al (2013) Hypothalamic-pituitary-thyroid axis in patients with Alzheimer disease (AD). J Investig Med 61:578–581. https://doi.org/10.2310/JIM.0b013e318280aafb
Vargas JY, Ahumada J, Arrázola MS, Fuenzalida M, Inestrosa NC (2015) WASP-1, a canonical Wnt signaling potentiator, rescues hippocampal synaptic impairments induced by Aβ oligomers. Exp Neurol 264:14–25. https://doi.org/10.1016/j.expneurol.2014.11.005
Vargas JY, Fuenzalida M, Inestrosa NC (2014) In vivo activation of Wnt signaling pathway enhances cognitive function of adult mice and reverses cognitive deficits in an Alzheimer’s disease model. J Neurosci 34:2191–2202. https://doi.org/10.1523/JNEUROSCI.0862-13.2014
Inestrosa NC, Arenas E (2010) Emerging roles of Wnts in the adult nervous system. Nat Rev Neurosci 11:77–86. https://doi.org/10.1038/nrn2755
Willert K, Nusse R (2012) Wnt proteins. Cold Spring Harb Perspect Biol 4:a007864. https://doi.org/10.1101/cshperspect.a007864
Komiya Y, Habas R (2008) Wnt signal transduction pathways. Organogenesis 4:68–75
MacDonald BT, Tamai K, He X (2009) Wnt/beta-catenin signaling: components, mechanisms, and diseases. Dev Cell 17:9–26. https://doi.org/10.1016/j.devcel.2009.06.016
Varela-nallar L, Alfaro IE, Serrano FG et al (2010) Wingless-type family member 5A (Wnta-5a) stimulates synaptic differentiation and function of glutamatergic synapses. Proc Natl Acad Sci U S A 107:10–15. https://doi.org/10.1073/pnas.1010011107 DCSupplemental.www.pnas.org/cgi/doi/10.1073/pnas.1010011107
Filippone MG, Di Palma T, Lucci V, Zannini M (2014) Pax8 modulates the expression of Wnt4 that is necessary for the maintenance of the epithelial phenotype of thyroid cells. BMC Mol Biol 15:21. https://doi.org/10.1186/1471-2199-15-21
Pasca di Magliano M, Di Lauro R, Zannini M (2000) Pax8 has a key role in thyroid cell differentiation. Proc Natl Acad Sci U S A 97:13144–13149. https://doi.org/10.1073/pnas.240336397
Miller LD, Park KS, Guo QM, Alkharouf NW, Malek RL, Lee NH, Liu ET, Cheng SY (2001) Silencing of Wnt signaling and activation of multiple metabolic pathways in response to thyroid hormone-stimulated cell proliferation. Mol Cell Biol 21:6626–6639. https://doi.org/10.1128/MCB.21.19.6626-6639.2001
Guigon CJ, Zhao L, Lu C, Willingham MC, Cheng SY (2008) Regulation of beta-catenin by a novel nongenomic action of thyroid hormone beta receptor. Mol Cell Biol 28:4598–4608. https://doi.org/10.1128/MCB.02192-07
O’Shea PJ, Kim DW, Logan JG et al (2012) Advanced bone formation in mice with a dominant-negative mutation in the thyroid hormone receptor β gene due to activation of Wnt/β-catenin protein signaling. J Biol Chem 287:17812–17822. https://doi.org/10.1074/jbc.M111.311464
Rosenfeld CS (2016) Sexspecific placental responses in fetal. Development 156:3422–3434. https://doi.org/10.1210/en.2015
Cvoro A, Devito L, Milton FA, Noli L, Zhang A, Filippi C, Sakai K, Suh JH et al (2015) A thyroid hormone receptor/KLF9 axis in human hepatocytes and pluripotent stem cells. Stem Cells 33:416–428. https://doi.org/10.1002/stem.1875
Chew L-J, Shen W, Ming X, Senatorov VV, Chen HL, Cheng Y, Hong E, Knoblach S et al (2011) SRY-box containing gene 17 regulates the Wnt/-catenin signaling pathway in oligodendrocyte progenitor cells. J Neurosci 31:13921–13935. https://doi.org/10.1523/JNEUROSCI.3343-11.2011
Acknowledgments
This work was supported by grants from the Basal Center of Excellence in Aging and Regeneration (CONICYT-AFB 170005) to N.C.I., FONDECYT (no. 1160724) to N.C.I., and FONDECYT (no. 11160651) to P.C. We also thank our special grant “Lithium in Health and Disease” from the Sociedad Química y Minera de Chile (SQM).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Salazar, P., Cisternas, P., Martinez, M. et al. Hypothyroidism and Cognitive Disorders during Development and Adulthood: Implications in the Central Nervous System. Mol Neurobiol 56, 2952–2963 (2019). https://doi.org/10.1007/s12035-018-1270-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-018-1270-y