Abstract
Adrenal incidentalomas (AI) are one of the most frequent reasons for consultation in Endocrinology, as they are present in 3–10% of the general population. Up to 20% of them may have autonomous cortisol secretion (ACS), a term that refers to AI carriers with biochemical evidence of excess cortisol, but without the “specific” clinical signs of Cushing’s syndrome. As ACS is associated with an increased risk of diabetes, obesity, high blood pressure (HBP), osteoporosis, cardiovascular events, and global mortality; its correct identification is of great importance. There are different laboratory assays to detect ACS, but all of them have some limitations. The dexamethasone suppression test is the most accepted for screening. However, there is no consensus on the cutoff point that should be used. Low levels of ACTH and DHEA-S and high urinary free cortisol are also associated with ACS, but in isolation they are of little value to establish the diagnosis. Considering its clinical implications and the lack of consensus in the diagnosis and in which is the most appropriate management of these patients, this review offers a quick reference guide of ACS, presenting an exhaustive review of the topic: its definition, epidemiology, diagnosis, clinical implications, treatment, and follow-up.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
By definition, and adrenal incidentaloma (AI) is an asymptomatic adrenal lesion detected in imaging tests not performed due to suspected adrenal disease [1]. Moreover, most reviews and recommendations require a size ≥1 cm and exclude from the definition adrenal lesions detected in the extension study of extra-adrenal tumor disease [2,3,4,5] and on the screening of patients with hereditary syndromes [6].
Generally, AI are nonfunctioning cortico-adrenal adenomas that do not require a specific treatment approach. However, the remaining 10–15% may be conditions that require some type of therapeutic intervention (e.g., adrenal carcinoma, pheochromocytoma, or Cushing's syndrome (CS)) [6].
A term widely used in the context of AI was “subclinical CS”. However, this terminology has become somewhat obsolete and it is currently more advisable to talk about autonomous cortisol secretion (ACS). This term aims to define AI patients with biochemical evidence of excess of cortisol, but without the “specific” clinical signs of CS (mainly the lack of catabolic characteristics such as myopathy and skin fragility).
It is important to identify these patients since ACS is associated with increased morbidity (diabetes, obesity, hypertension, osteoporosis, and cardiovascular events) and mortality [7, 8]. However, ACS is not easy to diagnose, mainly due to the lack of consensus on its definition and the fact that the detection of “specific” findings of CS is doctor-dependent.
In this article, we will review the definition, epidemiology, diagnosis, clinical implications, treatment, and prognosis of ACS. We will focus on the main tools for its diagnosis and describe the strengths and pitfalls of different diagnostic techniques that are currently being used, and the complications associated to ACS.
Epidemiology
Adrenal incidentalomas
Due to the increase in the use of imaging tests and their better resolution, incidentalomas are an increasing clinical problem. They are most frequently located at thyroid level (50%), followed by the pituitary (10–20%), and in the adrenals (5%) [9].
AI are one of the most frequently attended problems in general Endocrinology consultation; as radiological studies report their presence in 3% of 50-year-old patients increasing up to 10% in the elderly [3, 4, 10, 11]. They are more frequent in women (female-to-male ratio: 1.7) and in Caucasians. Their prevalence increases in obese, diabetic, and hypertensive patients [12]. They are very rare in children and adolescents (0.3–0.4% of childhood tumors). In this scenario, there is a high risk of malignancy, hence special attention should be paid to them [13].
The concept of AI includes a wide spectrum of diseases There is a marked variability of the underlying etiology depending on whether it is a surgical or clinical series, however, the most common cause in all of them is the non-functioning adrenal cortical adenoma (80%). The prevalence of malignancy and functionality is higher in the surgical series, since the lesions of larger size or with clinical–biochemical suspicion of functionality are generally operated (Table 1) [2, 3, 6, 10, 12, 14].
Autonomous cortisol secretion
The biochemical hypercortisolism or ACS is present in up to 20% of AI and is the most frequent hormonal alteration in AI. Its prevalence is very variable in the different series, mostly justified by the lack of consensus on its diagnosis (different diagnostic tests and cutoff points used in the different studies) [15]. Bulow et al. [16] reported a prevalence of 2%; Libe et al. [17] 18%; and Terzolo et al. [18] 5–20% (review of different series).
It is slightly more frequent in women and in patients older than 50 years [18]. Like AI, its prevalence increases in patients with diabetes, obesity, high blood pressure (HBP) and osteoporosis. It accounts for 15–20% of cases of endogenous CS, and is present in 0.2–2% of the general adult population [19].
Adrenal lesions with ACS usually correspond to cortico-adrenal adenomas (>85% of cases). However, there are reported cases of cortisol-producing myelolipomas [20], primary macro- or micronodular hyperplasia and in also in some adrenal carcinomas, where ACS is found [21].
Diagnosis of ACS
ACS is associated with increased morbidity and mortality, hence it is important to identify its presence. Therefore, hypercortisolism screening should be performed in all AI carriers.
Three criteria must be fulfilled to strictly speak of ACS [18]:
-
1.
Presence of AI.
-
2.
Absence of typical CS data. The main problem at this point is that CS is a continuous clinical spectrum, ranging from mild to clearly manifest forms, and the detection will be markedly conditioned by the clinician’s experience.
-
3.
Hormone study demonstrating ACS (independent ACTH). It is at this point where there are serious limitations, since there is no clear differentiation between excess cortisol and normality (depends in part on the personal preference of the clinician), considerable overlap in hormonal outcomes and lack of consensus on which is the best diagnostic strategy. Therefore, some authors propose a combination of several parameters to establish the diagnostic criteria. Moreover, coexistence of false positives of the tests is frequent in patients with AI (severe obesity, poorly controlled diabetes, psychiatric illness…). The different diagnostic tools currently available are shown in Table 2.
Table 2 Tests for ACS diagnosis (cutoff points, limitations, and advantages)
Dexamethasone suppression test
Currently, DST (1 mg) is the most used test in the diagnosis of ACS [6]. However, there is still no clear consensus about the cutoff point that should be used for the screening of ACS in AI [6, 7, 14, 22].
This parameter is altered in patients with AI, between 3 and 100% of cases, depending on the cutoff point and the dose of dexamethasone used; therefore, the sensitivity (44–100%) and specificity (24–100%) described are very variable [3, 4, 10, 12, 18, 23,24,25,26,27].
It is a simple and economical assay to perform that may be done on an outpatient setting. The standard test consists in the administration of 1 mg of dexamethasone at 23PM and the determination of plasma cortisol between 8.00–9.00 a.m. the following day.
Cutoff points used
The guidelines of the National Institute of Health (NIH) and the American Association of Clinical Endocrinologists and Endocrine Surgeons (AACE/AAES) agree that hypersecretion can be ruled out when DST is <5 μg/dl [4, 6, 25]. The sensitivity of this cutoff point is relatively low [44–58%), but it is the one that offers greater specificity (up to 100% according to some authors). The guidelines of the French Endocrine Society (FES) [28], the Italian Association of Clinical Endocrinologists (AME position statment) [14] and the European Endocrinology Society (ESE) [6] advocate a considerably lower cutoff point of 1.8 μg/dl to rule out ACS. This cutoff point has been established based on different studies demonstrating an increase in morbidity and mortality above these serum values [29, 30]. These results offer a higher sensitivity (75–100%), but low specificity (67–72%). Some European studies chose an intermediate point of 3 μg/dl, based on the fact that it offers the best sensitivity-specificity balance for morbidity and mortality above this cutoff point [7] (Table 3).
In general, we could say that ACS can be ruled out with DST <1.8 μg/dl and confirm its presence with levels >5 μg/dl. However, in patients with intermediate values in the DST there is no consensus on what attitude should be taken, so, there are several aspects still unclear in this subgroup of patients (see below, Section special situations: possible ACS).
Some authors propose higher doses of dexamethasone (3 or 8 mg), but no advantages have been shown compared with the conventional dose of 1 mg [31]. Others use the 2-day low-dose test (0.5 mg/6 h for 2 days); but since it is a more uncomfortable test to perform, it should be reserved for confirmatory diagnosis or in the context of psychiatric illness, alcoholism or diabetes [15, 32].
Limitations
For the correct interpretation of the results, the circumstances responsible for false positives in the test must be taken into account [33] (Table 4).
The simultaneous determination of dexamethasone in plasma can help confirm that sufficient concentrations are reached (>5.6 nmol/L (0.22 μg/dl)) to interpret the test result reliably, but its cost prevents its incorporation into a regular screening test [34, 35].
Urinary free cortisol
Its determination offers an integrated assessment of the plasma concentration of cortisol in 24 h, which is not affected by variations in CBG, as occurs under estrogen therapy or with mitotane.
Cutoff points used
When interpreting the results of this test is very important to take into account the laboratory method used; with radioimmunoassay a figure of <150 μg/day is normal, while with high-performance liquid chromatography the normal values are 40–50 μg/day. The latter is a more specific test for the detection of hypercortisolism [15, 36, 37].
Limitations
One of the more frequent problems is the incorrect sample collection. In these circumstances the simultaneous measurement of creatinine in urine increases its performance [38]. On the other hand, the guidelines of the ESE recommend collecting a second sample if the first is normal and there is suspicion of CS (to rule out cyclic CS or mild hypercortisolism) [6, 39]. Renal function should be taken into account (risk of false negatives with estimated glomerular filtrations <30–45 ml/min/1.73 m2), and also that the patient does not make excessive water intake or take preparations containing glucocorticoids [40].
UFC concentrations are elevated in 5–20% of patients with ACS. This is because the sensitivity to detect minimal elevations of cortisol is low, and therefore has a limited applicability as a screening test in this context.
Urinary cortisol metabolites
An alternative method for the diagnosis of ACS is the measurement of 24-h urinary cortisol metabolites by liquid tandem mass spectrometry (LC–MS/MS). Recent studies suggest that they present a better correlation with metabolic complications and are an earlier marker of hypercortisolim compared to other parameters [41].
Cutoff points used and limitations
Given that it is an experimental marker, the cutoff points that should be used for each metabolite are unknown. Its main limitations are the lack of the availability of the test, the need for 24-h urine collection and the lack of validation until the current date.
ACTH
The determination of ACTH in plasma is essential to know if CS is ACTH-dependent or independent.
Cutoff points used
Once hypercortisolism is confirmed, an ACTH level below 10 ng/mL confirms that it is an adrenal CS, and a value >20 ng/mL determines that it is an ACTH dependent CS. In case of intermediate values, it could be considered to expand the study with other tests such as stimulation test with CRH or DST for ACTH) [42]. However, their results should be interpreted with caution due to the lack of studies that validate these results.
Limitations
In patients with ACS, the degree of hypercortisolism may not be enough to completely suppress the HPA axis, and ACTH levels may not be suppressed. The specificity of a low ACTH as an isolated parameter in ACS is approximately 50% (38–60%), so it should be used in combination with other tests to guide the diagnosis of ACS.
The processing of the sample should be carried out with special caution, given its lability, which may result in a falsely low result. In a multicenter Italian study, they found a low inter-laboratory reproducibility in the ACTH assay, only 60% of the samples with low ACTH were correctly classified [43].
Night serum cortisol
It assesses the normality of the circadian rhythm of cortisol. In general, its performance in sensitivity and specificity is high, around 95%. Serum cortisol at 23 h should be used as a second line test since it requires patient hospitalization. However, it has a better correlation with several clinical conditions associated with hypercortisolism (mainly with cardiovascular risk) compared to other tests, and it is an early marker of autonomy and excess of cortisol [44].
Cutoff points used
Cutoff normal value depends on the extraction conditions and the range of normality of each center; but generally all cortisol values at 23 h >7.5 μg/dl are considered pathological [45].
Limitations
The main limitation lies in the need of hospitalization for its proper collection and the possibility of false negatives in patients with cyclic hypercortisolism (complete HPA axis integrity may exist in the quiescence phase).
Salivary cortisol and cortisone
These tests have the advantage that they are not affected by the concentrations of CBG, are economic and can be done at an outpatient setting.
Several recent studies have found that normal nocturnal salivary cortisol does not rule out ACS in patients with AI, therefore it should not be used as a screening test [46, 47]. In the recent study of Ceccato daily salivary cortisol rhythm was maintained in AI patients, without significant differences between patients with ACS and nonfunctioning adenomas (NFA), concluding that it was not an adequate parameter to detect ACS in these patients [48]. Several studies have found salivary cortisone a better biomarker for free cortisol than salivary cortisol [49].
Cutoff point used
Depending on the laboratory method used, there are different ranges of normality and differences in the sensitivity and specificity of the tests [45, 50, 51]. The methods that offer the best results are ELISA and LC–MS/MS [52]. With these techniques, normal nocturnal serum cortisol levels (23–24PM) are <145 ng/dL(4 nmol/L).
Limitations
The usefulness of salivary cortisol in AI is low (sensitivity of 22–76%) given that it does not discriminate ACS from normality. In cigarette smokers, salivary cortisol levels are higher than in nonsmokers [53], and the results may be altered in patients working at night or in shifts, as is the case with 23PM-serum cortisol.
The diagnostic ability of salivary cortisone in ACS can be compared, according to some studies, to that of nocturnal serum cortisol.
Dehydroepiandrosterone sulfate
Low serum levels of dehydroepiandrosterone sulfate (DHEAS) are one of the most frequent alterations in patients with ACS. According to the AACE/AAES guidelines [25], a low level of DHEAS supports the diagnosis of ACS. However, other authors consider that it is not a reliable parameter [54, 55] and the FES [28], korean [56] and ESE [6] guidelines do not recommend the systematic measurement of DHEAS in patients with AI.
Cutoff points used
The cutoff point used for the screening of ACS is variable according to the different studies. Yener found that a level <40 μg/dl shows a good diagnostic capacity (73.7%), being the one that offers a better balance between sensitivity (68%) and specificity (75%) [57]. Furthermore, a recent study investigating the serum steroid profiling by LC–MS/MS in AI, found that low DHEA had good accuracy in predicting ACS (sensitivity and specificity of 71 and 76% for a cutoff of 0.93 ng/mL) [58].
Limitations
It should be taken into account that DHEAS levels decrease physiologically with age [55]. Therefore, its specificity in the diagnosis of ACS decreases in the elderly population. The mass spectrometry-based assays are the best available techniques for the measurement of DHEAS, thanks to multianalytical capability at high level of specificity and sensitivity [58].
Clinical implications of ACS
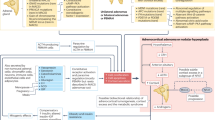
In ACS, by definition there are no specific signs or symptoms of hypercortisolism, but it is documented a higher frequency of several metabolic disorders, including HBP, type 2 diabetes (T2DM), obesity, osteoporosis, and increased mortality [29, 59] (Fig. 1).
Metabolic syndrome
There are basically two theories to explain the association between metabolic syndrome and ACS. One of them propose that it is mediated by the excess of cortisol (Terzolo) finds that triglyceride levels (considered an indirect marker of insulin resistance) were higher in patients with ACS regardless of BMI, attributing an effect mediated by cortisol [60]. On the contrary, others studies (Masserini) have found that it is mainly related to age and hyperinsulinism and in the absence of alterations in glucidic metabolism, ACS has no effect on lipid metabolism, that is, there is no direct effect of cortisol per se [61].
Lifestyle modifications are recommended, especially dietary control, physical exercise and weight control in patients with ACS and metabolic syndrome.
Type 2 diabetes
Excess glucocorticoids alter the glucose metabolism through several complex patho-physiological mechanisms and are associated with various degrees of glucose intolerance, such as altered basal glycemia, carbohydrate intolerance, and overt diabetes [62].
There is a higher prevalence of T2DM in patients with ACS, estimated in the range 20–75% (variable according to the diagnostic criteria used). In this regard, because basal glycemia levels may be normal it is recommended to use oral glucose tolerance test or glycated hemoglobin for diagnosis [62]. The risk of T2DM increases proportionally with the degree of hypercortisolism, the patients' age and the size of the adenoma [62].
The prevalence of ACS in T2DM varies between 0 and 9.4% [63,64,65], and the risk increases in those with poor metabolic control, microvascular complications, obesity and HBP [66].
Regarding the antidiabetic therapy for glycemic control in ACS patients, the same recommendations for T2DM may be used.
High blood pressure
Between 40 and 90% of patients have HBP. There seems to be a relationship with the duration of CS [67, 68] and a tendency toward higher BP than in patients with CS [67]. It is the alteration that improves more after surgery in patients with ACS [69].
The ESE guidelines recommend screening for HBP and T2DM in all patients with possible ACS and ACS [6].
Regarding the treatment of HBP, the ABC study group formulated a treatment algorithm that is specifically tailored for the management of hypercortisolism-related hypertension. They propose as a first-line treatment the use of any of the two blockers of the renin–angiotensin system, in light of the many evidences supporting a major alteration of this pathway in endogenous hypercortisolism, and for their cardioprotective effects [70].
Osteoporosis
ACS can lead to an increased risk of osteoporosis and vertebral fractures [71]. ACS can be detected in nearly 5% of patients with osteoporosis and in 10% with osteoporotic vertebral fractures [71]. The prevalence of osteoporosis in ACS is higher than in NFA (48 vs. 13%) [72]. It is partially explained by a decrease in bone mineral density (BMD) and a deterioration in bone quality [71]. The role of dual X-ray absorptiometry in predicting the risk of fractures is unknown (given that there is greater deterioration of the microarchitecture than of the BMD) [73]. Most studies reported a reduction in BMD at the spine assessed by either DXA [73, 74] or quantitative computed tomography [71]. TBS has also been reported to be lower and to predict the development of fracture in this group of patients [73]. ACS is reported to be associated with a decrease in blood osteocalcin levels, but no consistent changes in other markers or bone formation or markers of bone resorption [75].
Besides, it seems that there is an individual susceptibility to develop bone complications due to hypercortisolism. This is conditioned by different polymorphisms in the glucocorticoid receptor [76] and is a function of the activity of 11beta-hydroxyisteroid dehydrogenase [77].
Guidelines and recommendations for the pharmacological treatment of glucocorticoid-induced osteoporosis [78, 79]. recommend to start pharmacological treatment if the risk of fracture is moderate or high (regarding the age, BMD, menopausal status, and history of previous fracture) [75].
Cardiovascular disease and mortality
Several studies show an increase in mortality in ACS patients, mostly from cardiovascular causes. Nevertheless, it is still unclear if the mortality rate is higher than that of the general population [80, 81]. Patients with ACS had a higher rate of cardiovascular disease and death than NFA patients [30]. Cardiovascular disease and infections are the most frequent causes of death in ACS [29]. There are several evidences that support the increase in cardiovascular risk in ACS; among them, the higher waist-to-hip ratio, higher percentage of visceral fat, larger number of atheromatous plaques, and increased intima-media thickness, which have been observed in these patients in several studies [29, 81,82,83].
We recommend treat intensively HBP, diabetes and all the cardiovascular risk factors in these patients (taking into account the risk/benefit balance) with the aim of reduce the global cardiovascular risk, so it should be considered the indication of statins, antihyperglycemics and antihypertensive drugs in ACS patients.
Other associations
Some authors have found an association with thromboembolic disease [84], autoimmune thyroid disease [85], and psychiatric diseases, among others.
Follow-up evolution
The follow-up recommendations in patients with AI are designed to detect malignant and functioning tumors (mainly ACS [3]). Some studies report that the risk of tumor hyperfunctionality reaches a peak at 3–4 years of follow-up, although there is still no clear consensus about the time of follow-up necessary prior to discharge [86].
Risk of ACS in NFA
The risk that a patient with a negative functionality study progresses toward ACS is variable according to the different series ranging from 6.6 to 31% according to the different criteria used to diagnose ACS and the follow-up period. A higher risk of progression to ACS has been observed in adrenal lesions >2.5–3 cm and bilateral lesions [86, 87].
Until data will be available from large prospective studies, a reasonable approach may be to repeat the DST annually at least for 5 years in NFA (and consider also ACTH and UFC levels in patients with possible ACS). Similarly, we recommend that a careful monitoring of comorbidities potentially related to hypercortisolism should be performed annually.
Risk of overt CS in ACS
The risk of evolution from ACS to CS is very low (<1% according to multiple studies [3, 27, 80, 86]). Some series [86, 88] reported that a size >3 cm and unilateral uptake of iodocholesterol were considered predictive factors of overt CS.
In patients with ACS an annual clinical reassessment for cortisol excess and comorbidities potentially related to cortisol excess should be performed [6]
Treatment
The indication for surgical resection of an AI needs to be discussed by a multidisciplinary group once the recommended morphological and biochemical workup is completed. It is important to consider the patient's age, life expectancy, comorbidities, etc. The laparoscopic approach is the first line approach for the great majority of AI, once carcinoma has been ruled out.
Patients with ACS and possible ACS are at risk for transient insufficiency of the HPA axis after surgery. This will be prevented with intra and postoperative glucocorticoid supplementation treatment, preferably by hydrocortisone [6].
Several smaller studies have shown improvement of glucose metabolism and body weight after surgical resection [54, 89]. Furthermore in a prospective randomized study of 45 ACS patients found that 70% of operated patients showed improved of diabetes and HBP and patients who received conservative treatment showed deterioration in the glucemic and BP control [90]. These data suggest that surgical treatment is more beneficial than conservative treatment. Previous prospective, nonrandomized studies found similar results [83, 91,92,93].
Special situations
Bilateral AI
Bilateral AI (BAI) represent about 15% of AI [26, 94]. The distribution of the etiologies of BAI differs from that of unilateral AI. The most common causes are metastasis, primary bilateral macronodular adrenal hyperplasia (BMAH) and bilateral cortical adenomas [95]. BMAH and bilateral cortical adenomas are usually linked with a cortisol excess. In BMAH the cortisol secretion is partly regulated by the expression of multiple aberrant G protein-coupled receptors [96]. They are present in up to 87% of patients [97].
The hormonal evaluation in BAI is similar to unilateral AI, but in contrast to unilateral AI, adrenal insufficiency and congenital adrenal hyperplasia (specially 21-hydroxylase deficiency) should be excluded [6]. It is known that ACS is more prevalent in BAI than in unilateral adenomas patients For example, in the Vassiliadi retrospective study (41.5 vs. 12.2%) [98]; Vassilatou prospective study (35.1 vs. 17.9%) [99] and Pasternak in a surgical serie (21.7 vs. 6.2%) [100].
The management of these patients is the most complicated issue. Bilateral adrenalectomy may be considered if UFC levels are >3–4 times the upper limit of normal (ULN) with both adrenals having relatively symmetrical size [95]. However, many groups reported the benefits of unilateral adrenalectomy in small series of patients with BMAH and modest cortisol secretion (<2–3 times ULN): with a remission rate of hypercortisolism reaching 97% with a recurrence rate of 23% [101, 102].
Possible ACS
Nowadays, one of the most concerning issues are the patients with intermediate values in the DST (1.8–5 mcg/dl) (possible ACS) [6]. We must bear in mind that the result of the DST should be interpreted more as a continuous variable instead of categorical (yes/no) [6].
Exist evidence that patients with DST between 1.8 and 5 mcg/dl have higher risk of T2DM, hypertension, cardiovascular disease and mortality, and this risk increased during the follow-up [6, 8, 30]. So, we should look for comorbidities potentially related to cortisol excess in these patients [6].
None of the different additional tests evaluated in different studies was convincing enough to establish the diagnosis of ACS in these patients [6]. However, in a large number of studies a combination of DST and other parameters (specially high UFC and low ACTH) are used to improve the sensitivity and specificity [17, 47, 103]. Furthermore, the FES [28] and Korean guidelines [56] and AME position statement [14] recommend that patient with DST > 1.8 mcg/dl should receive second line investigation, with at the very minimum a determination of UFC, serum, or salivary cortisol at midnight and ACTH.
Most of the panel members of the ESE guidelines recommend to repeat the DST after 3–12 months and re-evaluate comorbidities [6]. This attitude seems reasonable, especially in patients with DST closer to 5 mcg/dl if we take into account that a significant percentage evolves to ACS or developed potentially cortisol related complications during the follow-up [8, 30].
Our recommendation in patients with DST between 1.8 and 5 mcg/dl is to extend the study with a second-line test (mainly with UFC and ACTH) if there are comorbidities potentially related to excess cortisol (T2DM, hypertension, disease cardiovascular, etc). In all other cases the DST should be repeated after 6–12 months (before or after depending on the results of the previous DST).
Conclusion
ACS is the most common hormonal alteration in AI, present in approximately 20% of these patients. It is associated with an increased risk of T2DM, HBP, obesity, dyslipidemia, and global cardiovascular risk. Therefore, it is of great importance to correctly identify these patients.
There are different tests for the screening of hypercortisolism. However, all of them have some limitations. In the absence of a single test considered as the gold standard, it is necessary the combination of different tests to reach an adequate diagnostic capacity.
The indication of surgery in ACS patients should be based mainly in the hormonal evaluation and the comorbidities potentially mediated by cortisol.
References
J. Wagner, D.C. Aron, Incidentalomas—A ‘disease’ of modern imaging technology. Best Pract. Res. Clin. Endocrinol. Metab. 26(1), 3–8 (2012). https://doi.org/10.1016/j.beem.2011.08.006
G. Favia, F. Lumachi, S. Basso, D.F. D’Amico, Management of incidentally discovered adrenal masses and risk of malignancy. Surgery 128(6), 918–924 (2000). https://doi.org/10.1067/msy.2000.109965
L. Barzon, N. Sonino, F. Fallo, G. Palu, M. Boscaro, Prevalence and natural history of adrenal incidentalomas. Eur. J. Endocrinol. 149(4), 273–285 (2003)
M.M. Grumbach, B.M. Biller, G.D. Braunstein, K.K. Campbell, J.A. Carney, P.A. Godley et al. Management of the clinically inapparent adrenal mass (‘incidentaloma’). Ann. Intern. Med. 138(5), 424–429 (2003)
P. Anagnostis, A. Karagiannis, K. Tziomalos, A.I. Kakafika, V.G. Athyros, D.P. Mikhailidis, Adrenal incidentaloma: a diagnostic challenge. HormONES 8(3), 163–184 (2009)
M. Fassnacht, W. Arlt, I. Bancos, H. Dralle, J. Newell-Price, A.M. Sahdev et al. Management of adrenal incidentalomas: European Society of Endocrinology Clinical Practice Guideline in collaboration with the European Network for the Study of Adrenal Tumors. Eur. J. Endocrinol. 175(2), G1–G34 (2016). https://doi.org/10.1530/EJE-16-0467
I. Chiodini, Diagnosis and treatment of subclinical hypercortisolism. J. Clin. Endocrinol. Metab. 96(5), 1223–1236 (2011). https://doi.org/10.1210/jc.2010-2722. Epub 2011 Mar 2
G. Di Dalmazi, V. Vicennati, E. Rinaldi, A.M. Morselli-Labate, E. Giampalma, C.G. Mosconi et al. Progressively increased patterns of subclinical cortisol hypersecretion in adrenal incidentalomas differently predict major metabolic and cardiovascular outcomes: a large cross-sectional study. Eur. J. Endocrinol. 166(4), 669–677 (2012). https://doi.org/10.1530/EJE-11-1039. Epub 2012 Jan 20
A.A. Gheorghisan-Galateanu, M. Carsote, A. Valea, Incidentaloma: from general practice to specific endocrine frame. J. Pak. Med Assoc. 67(6), 917–922 (2017)
F. Mantero, M. Terzolo, G. Arnaldi, G. Osella, A.M. Masini, A.F. Alì et al. A survey on adrenal incidentaloma in Italy. J. Clin. Endocrinol. Metab. 85(2), 637–644 (2000)
S. Bovio, A. Cataldi, G. Reimondo, P. Sperone, S. Novello, A. Berruti et al. Prevalence of adrenal incidentaloma in a contemporary computerized tomography series. J. Endocrinol. Invest. 29(4), 298–302 (2006)
R.T. Kloos, M. Korobkin, N.W. Thompson, I.R. Francis, B. Shapiro, M.D. Gross, Incidentally discovered adrenal masses. Cancer Treat. Res. 89, 263–292 (1997)
S.K. Mayer, L.L. Oligny, C. Deal, S. Yazbeck, N. Gagné, H. Blanchard, Childhood adrenocortical tumors: case series and reevaluation of prognosis--a 24-year experience. J. Pediatr. Surg. 32(6), 911–915 (1997)
M. Terzolo, A. Stigliano, I. Chiodini, P. Loli, L. Furlani, G.,M. Arnaldi et al. AME position statement on adrenal incidentaloma. Eur. J. Endocrinol. 164(6), 851–870 (2011). https://doi.org/10.1530/EJE-10-1147. Epub 2011 Apr 6
L.K. Nieman, B.M. Biller, J.W. Findling, J. Newell-Price, M.O. Savage, P.M. Stewart et al. The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 93(5), 1526–1540 (2008). https://doi.org/10.1210/jc.2008-0125. Epub 2008 Mar 11
B. Bülow, S. Jansson, C. Juhlin, L. Steen, M. Thorén, H. Wahrenberg et al. Adrenal incidentaloma—follow-up results from a Swedish prospective study. Eur. J. Endocrinol. 154(3), 419–423 (2006)
R. Libè, C. Dall’Asta, L. Barbetta, A. Baccarelli, P.R. Beck-Peccoz et al. Long-term follow-up study of patients with adrenal incidentalomas. Eur. J. Endocrinol. 147(4), 489–494 (2002)
M. Terzolo, A. Pia, G.M. Reimondo, Subclinical Cushing’s syndrome: definition and management. Clin. Endocrinol. 76(1), 12–18 (2012). https://doi.org/10.1111/j.1365-2265.2011.04253.x
I. Chiodini, C.E. Vainicher, V. Morelli, S. Palmieri, E. Cairoli, A.S. Salcuni et al. Endogenous subclinical hypercortisolism and bone: a clinical review. Eur. J. Endocrinol. 175(6), R265–R282 (2016)
M. Boronat, A. Moreno, S. Ramón y Cajal, E. Pineda, T. Lucas, J. Estrada, Subclinical Cushing’s syndrome due to adrenal myelolipoma. Arch. Pathol. Lab Med. 121(7), 735–737 (1997)
K. Duan, K.G. Hernandez, O. Mete, Clinicopathological correlates of adrenal cushing’s syndrome. Postgrad. Med J. 91(1076), 331–342 (2015). https://doi.org/10.1136/postgradmedj-2014-202612rep
G.M. Goddard, A. Ravikumar, A.C. Levine, Adrenal mild hypercortisolism. Endocrinol. Metab. Clin. North Am. 44(2), 371–379 (2015). https://doi.org/10.1016/j.ecl.2015.02.009
N.S. Ross, Epidemiology of Cushing’s syndrome and subclinical disease. Endocrinol. Metab. Clin. North Am. 23(3), 539–546 (1994)
M. Reincke, J. Nieke, G.P. Krestin, W. Saeger, B. Allolio, W. Winkelmann, Preclinical Cushing's syndrome in adrenal incidentalomas—comparison with adrenal Cushing's syndrome. J. Clin. Endocrinol. Metab. 75(3), 826–832 (1992)
M.A. Zeiger, G.B. Thompson, Q.Y. Duh, A.H. Hamrahian, P. Angelos, D. Elaraj et al. American Association of Clinical Endocrinologists and American Association of Endocrine Surgeons Medical Guidelines for the Management of Adrenal Incidentalomas: executive summary of recommendations. Endocr. Pract. 15(5), 450–453 (2009)
W.F. Young Jr., Clinical practice. The incidentally discovered adrenal mass. N. Engl. J. Med. 356(6), 601–610 (2007)
T.J. Cawood, P.J. Hunt, D. O’Shea, D. Cole, S.T. Soule, Recommended evaluation of adrenal incidentalomas is costly, has high false-positive rates and confers a risk of fatal cancer that is similar to the risk of the adrenal lesion becoming malignant; time for a rethink? Eur. J. Endocrinol. 161(4), 513–527 (2009). https://doi.org/10.1530/EJE-09-0234. Epub 2009 May 13
A. Tabarin, S. Bardet, J. Bertherat, B. Dupas, O. Chabre, E. Hamoir et al. Exploration and management of adrenal incidentalomas. French Society of Endocrinology Consensus. Ann. Endocrinol. 69(6), 487–500 (2008). https://doi.org/10.1016/j.ando.2008.09.003. Epub 2008 Nov 20
M. Debono, M. Bradburn, M. Bull, B. Harrison, R.J. Ross, J. Newell-Price, Cortisol as a marker for increased mortality in patients with incidental adrenocortical adenomas. J. Clin. Endocrinol. Metab. 99(12), 4462–4470 (2014). https://doi.org/10.1210/jc.2014-3007
G. Di Dalmazi, V. Vicennati, S. Garelli, E. Casadio, E. Rinaldi, E. Giampalma et al. Cardiovascular events and mortality in patients with adrenal incidentalomas that are either non-secreting or associated with intermediate phenotype or subclinical Cushing’s syndrome: a 15-year retrospective study. Lancet Diabetes Endocrinol. 2(5), 396–405 (2014). https://doi.org/10.1016/S2213-8587(13)70211-0. Epub 2014 Jan 29
T. Katabami, R. Obi, N. Shirai, S. Naito, N. Saito, Discrepancies in results of low-and high-dose dexamethasone suppression tests for diagnosing preclinical Cushing’s syndrome. Endocr. J. 52(4), 463–469 (2005)
S. Tsagarakis, P. Kokkoris, C. Roboti, C. Malagari, J. Kaskarelis, V. Vlassopoulou et al. The low-dose dexamethasone suppression test in patients with adrenal incidentalomas: comparisons with clinically euadrenal subjects and patients with Cushing’s syndrome. Clin. Endocrinol. 48(5), 627–633 (1998)
L.K. Nieman, B.M. Biller, J.W. Findling, J. Newell-Price, M.O. Savage, P.M. Stewart et al. The Diagnosis of Cushing’s Syndrome: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 93(5), 1526–1540 (2008). https://doi.org/10.1210/jc.2008-0125. Epub 2008 Mar 11
A.W. Meikle, Dexamethasone suppression tests: usefulness of simultaneous measurement of plasma cortisol and dexamethasone. Clin. Endocrinol. 16(4), 401–408 (1982)
C. Hempen, S. Elfering, A.H. Mulder, F.A. van den Bergh, R.G. Maatman, Dexamethasone suppression test: Development of a method for simultaneous determination of cortisol and dexamethasone in human plasma by liquid chromatography/tandem mass spectrometry. Ann. Clin. Biochem. 49(Pt 2), 170–176 (2012). https://doi.org/10.1258/acb.2011.011004. Epub 2012 Jan 12
G. Arnaldi, A. Angeli, A.B. Atkinson, X. Bertagna, F. Cavagnini, G.P. Chrousos et al. Diagnosis and complications of Cushing’s syndrome: a consensus statement. J. Clin. Endocrinol. Metab. 88(12), 5593–5602 (2003)
J. Newell-Price, Diagnosis/differential diagnosis of Cushing’s syndrome: a review of best practice. Best. Pract. Res Clin. Endocrinol. Metab. 23, S5–S14 (2009)
S. Santos, E. Santos, S. Gaztambide, J. Salvador, Diagnóstico y diagnóstico diferencial del síndrome de Cushing. Endocrinol. Nutr. 56(2), 71–84 (2009). https://doi.org/10.1016/S1575-0922(09)70555-8. Epub 2009 May 1
F. Ceccato, G. Antonelli, M. Barbot, M. Zilio, L. Mazzai, R. Gatti et al. The diagnostic performance of urinary free cortisol is better than the cortisol: cortisone ratio in detecting de novo Cushing’s syndrome: the use of a LC–MS/MS method in routine clinical practice. Eur. J. Endocrinol. 171(1), 1–7 (2014). https://doi.org/10.1530/EJE-14-0061. Epub 2014 Apr 17
F. Ceccato, M. Boscaro, Cushing’s syndrome: screening and diagnosis. High Blood Press Cardiovasc Prev. 23(3), 209–215 (2016). https://doi.org/10.1007/s40292-016-0153-4
A. Kotłowska, T. Puzyn, K. Sworczak, P. Stepnowski, P. Szefer, Metabolomic biomarkers in urine of cushing’s syndrome patients. Int J. Mol. Sci. 18(2), pii: E294 (2017). https://doi.org/10.3390/ijms18020294
J. Newell-Price, X. Bertagna, A.B. Grossman, L.K. Nieman, Cushing’s syndrome. Lancet 367(9522), 1605–1617 (2006)
F. Pecori Giraldi, A. Saccani, F. Cavagnini, Assessment of ACTH assay variability: a multicenter study. Eur. J. Endocrinol. 164(4), 505–512 (2011). https://doi.org/10.1530/EJE-10-0962. Epub 2011 Jan 20
M. Terzolo, S. Bovio, A. Pia, P.A. Conton, G. Reimondo, C.T. Dall’Asta et al. Midnight serum cortisol as a marker of increased cardiovascular risk in patients with a clinically inapparent adrenal adenoma. Eur. J. Endocrinol. 153(2), 307–315 (2005)
D.A. Papanicolaou, N. Mullen, I. Kyrou, L.K. Nieman, Nighttime salivary cortisol: A useful test for the diagnosis of Cushing’s syndrome. J. Clin. Endocrinol. Metab. 87(10), 4515–4521 (2002)
M. Doi, N. Sekizawa, Y. Tani, K. Tsuchiya, R. Kouyama, T. Tateno et al. Late-night salivary cortisol as a screening test for the diagnosis of Cushing’s syndrome in Japan. Endocr. J. 55(1), 121–126 (2008). Epub 2008 Jan 17
B. Masserini, V. Morelli, S. Bergamaschi, F. Ermetici, C. Eller-Vainicher, A.M. Barbieri et al. The limited role of midnight salivary cortisol levels in the diagnosis of subclinical hypercortisolism in patients with adrenal incidentaloma. Eur. J. Endocrinol. 160(1), 87–92 (2009). https://doi.org/10.1530/EJE-08-0485. Epub 2008 Oct 3
F. Ceccato, M. Barbot, N. Albiger, G. Antonelli, M. Zilio, M. Todeschini et al. Daily salivary cortisol and cortisone rhythm in patients with adrenal incidentaloma. Endocrine 59(3), 510–519 (2018). https://doi.org/10.1007/s12020-017-1421-3. Epub 2017 Sep 27
I. Perogamvros, B.G. Keevil, D.W. Ray, P.J. Trainer, Salivary cortisone is a potential biomarker for serum free cortisol. J. Clin. Endocrinol. Metab. 95(11), 4951–4958 (2010). https://doi.org/10.1210/jc.2010-1215. Epub2010 Aug 4
M. Castro, P.C. Elias, A.R. Quidute, F.P. Halah, A.C. Moreira, Out-patient screening for Cushing’s syndrome: the sensitivity of the combination of circadian rhythm and overnight dexamethasone suppression salivary cortisol tests. J. Clin. Endocrinol. Metab. 84(3), 878–882 (1999)
K. Mészáros, G. Karvaly, Z. Márta, B. Magda, J. Tőke, N. Szücs et al. Diagnostic performance of a newly developed salivary cortisol and cortisone measurement using an LC–MS/MS method with simple and rapid sample preparation. J. Endocrinol. Invest 41(3), 315–323 (2018). https://doi.org/10.1007/s40618-017-0743-6. Epub 2017 Aug 16
S.K. Baid, N. Sinaii, M. Wade, D. Rubino, L.K. Nieman, Radioimmunoassay and tandem mass spectrometry measurement of bedtime salivary cortisol levels: a comparison of assays to establish hypercortisolism. J. Clin. Endocrinol. Metab. 92(8), 3102–3107 (2007). Epub 2007 Jun 5
E. Badrick, C. Kirschbaum, M. Kumari, The relationship between smoking status and cortisol secretion. J. Clin. Endocrinol. Metab. 92(3), 819–824 (2007). Epub 2006 Dec 19
A. Tanabe, M. Naruse, T. Nishikawa, T. Yoshimoto, T. Shimizu, T. Seki et al. Autonomy of cortisol secretion in clinically silent adrenal incidentaloma. Horm. Metab. Res. 33(7), 444–450 (2001)
Z. Bencsik, I. Szabolcs, Z. Kovács, A. Ferencz, A. Vörös, I. Kaszás et al. Low dehydroepiandrosterone sulfate (DHEA-S) level is not a good predictor of hormonal activity in nonselected patients with incidentally detected adrenal tumors. J. Clin. Endocrinol. Metab. 81(5), 1726–1729 (1996)
J.M. Lee, M.K. Kim, S.H. Ko, J.M. Koh, B.Y. Kim, S.W. Kim et al. Clinical Guidelines for the management of adrenal incidentaloma. Endocrinol. Metab. 32(2), 200–218 (2017). https://doi.org/10.3803/EnM.2017.32.2.200
S. Yener, H. Yilmaz, T. Demir, M. Secil, A. Comlekci, DHEAS for the prediction of subclinical Cushing’s syndrome: perplexing or advantageous? Endocrine 48(2), 669–676 (2015). https://doi.org/10.1007/s12020-014-0387-7. Epub 2014 Aug 22
G. Di Dalmazi, F. Fanelli, M. Mezzullo, E. Casadio, E. Rinaldi, S. Garelli et al. Steroid profiling by LC-MS/MS in nonsecreting and subclinical cortisol-secreting adrenocortical adenomas. J. Clin. Endocrinol. Metab. 100(9), 3529–3538 (2015). https://doi.org/10.1210/JC.2015-1992. Epub2015 Jul 10
A.S. Salcuni, V. Morelli, C. Eller Vainicher, S. Palmieri, E. Cairoli, A. Spada et al. Adrenalectomy reduces the risk of vertebral fractures in patients with monolateral adrenal incidentalomas and subclinical hypercortisolism. Eur. J. Endocrinol. 174(3), 261–269 (2016). https://doi.org/10.1530/EJE-15-0977. Epub 2015 Dec 2
M. Terzolo, A. Pia, A. Alì, G. Osella, G. Reimondo, S. Bovio et al. Adrenal incidentaloma: a new cause of the metabolic syndrome? J. Clin. Endocrinol. Metab. 87(3), 998–1003 (2002)
B. Masserini, V. Morelli, S. Palmieri, C. Eller-Vainicher, V. Zhukouskaya, E. Cairoli et al. Lipid abnormalities in patients with adrenal incidentalomas: Role of subclinical hypercortisolism and impaired glucose metabolism. J. Endocrinol. Invest. 38(6), 623–628 (2015). https://doi.org/10.1007/s40618-014-0232-0. Epub 2015 Jan 23
C. Scaroni, M. Zilio, M. Foti, M. Boscaro, Glucose metabolism abnormalities in cushing syndrome: from molecular basis to clinical management. Endocr. Rev. 38(3), 189–219 (2017). https://doi.org/10.1210/er.2016-1105
E. Ellis, P.K. Chin, P.J. Hunt, H. Lunt, J.G. Lewis, S.G. Soule, Is late-night salivary cortisol a better screening test for possible cortisol excess than standard screening tests in obese patients with Type 2. Diabetes? N. Z. Med J. 125(1353), 47–58 (2012)
K. Mullan, N. Black, A. Thiraviaraj, P.M. Bell, C. Burgess, S.J. Hunter et al. Is there value in routine screening for Cushing’s syndrome in patients with diabetes? J. Clin. Endocrinol. Metab. 95(5), 2262–2265 (2010). https://doi.org/10.1210/jc.2009-2453. Epub 2010 Mar 17
L. Gagliardi, I.M. Chapman, P. O’Loughlin, D.J. Torpy, Screening for subclinical Cushing’s syndrome in type 2 diabetes mellitus: low false-positive rates with nocturnal salivary cortisol. Horm. Metab. Res. 42(4), 280–284 (2010). https://doi.org/10.1055/s-0029-1246191. Epub 2010 Jan 29
G.B. Cansu, S. Atılgan, M.K. Balcı, R. Sarı, S. Özdem, H.A. Altunbaş, Which type 2 diabetes mellitus patients should be screened for subclinical Cushing’s syndrome? Hormone. 16(1), 22–32 (2017). https://doi.org/10.14310/horm.2002.1716
A.M. Isidori, C. Graziadio, R.M. Paragliola, A. Cozzolino, A.G. Ambrogio, A. Colao et al. The hypertension of Cushing’s syndrome: controversies in the pathophysiology and focus on cardiovascular complications. J. Hypertens. 33(1), 44–60 (2015). https://doi.org/10.1097/HJH.0000000000000415
T. Mancini, B. Kola, F. Mantero, M. Boscaro, G. Arnaldi, High cardiovascular risk in patients with Cushing’s syndrome according to 1999WHO/ISH guidelines. Clin. Endocrinol. 61(6), 768–777 (2004)
Bancos I., Alahdab F., Crowley R.K., Chortis V., Delivanis D.A., Erickson D6 et al. Therapy of endocrine disease: improvement of cardiovascular risk factors after adrenalectomy in patients with adrenal tumors and subclinical Cushing’s syndrome: a systematic review and meta-analysis. Eur. J. Endocrinol. 2016;175(6):R283–R295. Epub 2016 Jul 22.
M. Grasso, M. Boscaro, C. Scaroni, F. Ceccato, Secondary Arterial Hypertension: From Routine Clinical Practice to Evidence in Patients with Adrenal Tumor. High. Blood Press Cardiovasc Prev. 25(4), 345–354 (2018). https://doi.org/10.1007/s40292-018-0288-6. Epub 2018 Nov 10
I. Chiodini, M. Torlontano, V. Carnevale, G. Guglielmi, M. Cammisa, V. Trischitta et al. Bone loss rate in adrenal incidentalomas: a longitudinal study. J. Clin. Endocrinol. Metab. 86(11), 5337–5341 (2001)
V. Morelli, C. Eller-Vainicher, A.S. Salcuni, F. Coletti, L. Iorio, G. Muscogiuri et al. Risk of new vertebral fractures in patients with adrenal incidentaloma with and without subclinical hypercortisolism: a multicenter longitudinal study. J. Bone Miner. Res. 26(8), 1816–1821 (2011). https://doi.org/10.1002/jbmr.398
I. Chiodini, V. Morelli, B. Masserini, A.S. Salcuni, C. Eller-Vainicher, R. Viti et al. Bone mineral density, prevalence of vertebral fractures, and bone quality in patients with adrenal incidentalomas with and without subclinical hypercortisolism: an Italian multicenter study. J. Clin. Endocrinol. Metab. 94(9), 3207–3214 (2009). https://doi.org/10.1210/jc.2009-0468. Epub 2009 Jun 23
C. Eller-Vainicher, V. Morelli, F.M. Ulivieri, S. Palmieri, V.V. Zhukouskaya, E. Cairoli et al. Bone quality, as measured by trabecular bone score in patients with adrenal incidentalomas with and without subclinical hypercortisolism. J. Bone Miner. Res. 27(10), 2223–2230 (2012). https://doi.org/10.1002/jbmr.1648
R.S. Hardy, H. Zhou, M.J. Seibel, M.S. Cooper, Glucocorticoids and bone: consequences of endogenous and exogenous excess and replacement therapy. Endocr. Rev. 39(5), 519–548 (2018). https://doi.org/10.1210/er.2018-00097
V. Morelli, F. Donadio, C. Eller-Vainicher, V. Cirello, L. Olgiati, C. Savoca et al. Role of glucocorticoid receptor polymorphism in adrenal incidentalomas. Eur. J. Clin. Invest 40(9), 803–811 (2010). https://doi.org/10.1111/j.1365-2362.2010.02330.x. Epub 2010 Jun 23
V.V. Zhukouskaya, C. Eller-Vainicher, A. Gaudio, E. Cairoli, F.M. Ulivieri, S.,V. Palmieri et al. In postmenopausal female subjects with type 2 diabetes mellitus vertebral fractures are independently associated with cortisol secretion and sensitivity. J. Clin. Endocrinol. Metab. 100(4), 1417–1425 (2015). https://doi.org/10.1210/jc.2014-4177. Epub 2015 Jan 15
L. Buckley, G. Guyatt, H.A. Fink, M. Cannon, J. Grossman, K.E. Hansen et al. 2017 American College of Rheumatology Guideline for the Prevention and Treatment of Glucocorticoid-Induced Osteoporosis. Arthritis Care Res. 69(8), 1095–1110 (2017). https://doi.org/10.1002/acr.23279. Epub 2017 Jun 6
S. Lekamwasam, J.D. Adachi, D. Agnusdei, J. Bilezikian, S. Boonen, F. Borgström et al. A framework for the development of guidelines for the management of glucocorticoid-induced osteoporosis. Osteoporos. Int. 23(9), 2257–2276 (2012). https://doi.org/10.1007/s00198-012-1958-1. Epub 2012 Mar 21
G.P. Bernini, A. Moretti, C. Oriandini, M. Bardini, C. Taurino, A. Salvetti, Long-term morphological and hormonal follow-up in a single unit on 115 patients with adrenal incidentalomas. Br. J. Cancer 92(6), 1104–1109 (2005)
S. Yener, M. Baris, A. Peker, O. Demir, B. Ozgen, M. Secil, Autonomous cortisol secretion in adrenal incidentalomas and increased visceral fat accumulation during follow-up. Clin. Endocrinol. 87(5), 425–432 (2017). https://doi.org/10.1111/cen.13408. Epub2017 Aug 2
M. Debono, J. Newell-Price, Subclinical hypercortisolism in adrenal incidentaloma. Curr. Opin. Endocrinol. Diabetes Obes. 22(3), 185–192 (2015). https://doi.org/10.1097/MED.0000000000000151
R. Rossi, L. Tauchmanova, A. Luciano, M. Di Martino, C. Battista, L. Del Viscovo et al. Subclinical Cushing’s syndrome in patients with adrenal incidentaloma: clinical and biochemical features. J. Clin. Endocrinol. Metab. 85(4), 1440–1448 (2000)
R. Świątkowska-stodulska, A. Skibowska-bielińska, P. Wiśniewski, K. Sworczak, Activity of selected coagulation factors in overt and subclinical hypercortisolism. Endocr. J. 62(8), 687–694 (2015). https://doi.org/10.1507/endocrj.EJ14-0539. Epub 2015 May 13
M. Karakose, B. Karbek, M. Sahin, M.S. Arslan, O. Topaloglu, G. Erden et al. The association of autoimmune thyroiditis and non-functional adrenal incidentalomas with insulin resistance. Arch. Endocrinol. Metab. 59(1), 42–46 (2015). https://doi.org/10.1590/2359-3997000000008
L. Barzon, C. Scaroni, N. Sonino, F. Fallo, A. Paoletta, M. Boscaro, Risk factors and long-term follow-up of adrenal incidentalomas. J. Clin. Endocrinol. Metab. 84(2), 520–526 (1999)
V. Morelli, A. Scillitani, M. Arosio, I. Chiodini, Follow-up of patients with adrenal incidentaloma, in accordance with the European society of endocrinology guidelines: could we be safe? J. Endocrinol. Invest. 40(3), 331–333 (2017). https://doi.org/10.1007/s40618-016-0558-x. Epub 2016 Oct 15
L. Barzon, F. Fallo, N. Sonino, M. Boscaro, Development of overt Cushing’s syndrome in patients with adrenal incidentaloma. Eur. J. Endocrinol. 146(1), 61–66 (2002)
I.C. Mitchell, R.J. Auchus, K. Juneja, A.Y. Chang, S.A. Holt, W.H. Snyder et al. Subclinical Cushing’s syndrome is not subclinical: improvement after adrenalectomy in 9 patients. Surgery 142(6), 900–905 (2007)
A. Toniato, I. Merante-Boschin, G. Opocher, M.R. Pelizzo, F. Schiavi, E. Ballotta, Surgical versus conservative management for subclinical cushing syndrome in adrenal incidentalomas: a prospective randomized study. Ann. Surg. 249(3), 388–391 (2009). https://doi.org/10.1097/SLA.0b013e31819a47d2
S. Midorikawa, H. Sanada, S. Hashimoto, T. Suzuki, T. Watanabe, The improvement of insulin resistance in patients with adrenal incidentaloma by surgical resection. Clin. Endocrinol. 54(6), 797–804 (2001)
R. Emral, A.R. Uysal, M. Asik, S. Gullu, D. Corapcioglu, V. Tonyukuk et al. Prevalence of subclinical Cushing’s syndrome in 70 patients with adrenal incidentaloma: clinical, biochemical and surgical outcomes. Endocr. J. 50(4), 399–408 (2003)
M. Tsuiki, A. Tanabe, S. Takagi, M. Naruse, K. Takano, Cardiovascular risks and their long-term clinical outcome in patients with subclinical Cushing’s syndrome. Endocr. J. 55(4), 737–745 (2008). Epub 2008 May 28
J. Patrova, I. Jarocka, H. Wahrenberg, H. Falhammar, Clinical outcomes in adrenal incidentaloma: experience from one center. Endocr. Pract. 21(8), 870–877 (2015). https://doi.org/10.4158/EP15618.OR. Epub 2015 Jun 29
I. Bourdeau, N. El Ghorayeb, N. Gagnon, A. Lacroix, Management of endocrine disease: Differential diagnosis, investigation and therapy of bilateral adrenal incidentalomas. Eur. J. Endocrinol. 179(2), R57–R67 (2018). https://doi.org/10.1530/EJE-18-0296. Epub 2018 May 10
N. El Ghorayeb, I. Bourdeau, A. Lacroix, Multiple aberrant hormone receptors in Cushing’s syndrome. Eur. J. Endocrinol. 173(4), M45–M60 (2015). https://doi.org/10.1530/EJE-15-0200. Epub 2015 May 13
R. Libé, J. Coste, L. Guignat, F. Tissier, H. Lefebvre, G. Barrande et al. Aberrant cortisol regulations in bilateral macronodular adrenal hyperplasia: a frequent finding in a prospective study of 32 patients with overt or subclinical Cushing’s syndrome. Eur. J. Endocrinol. 163(1), 129–138 (2010). https://doi.org/10.1530/EJE-10-0195. Epub 2010 Apr 8
D.A. Vassiliadi, G. Ntali, T. Stratigou, M. Adali, S. Tsagarakis, Aberrant cortisol responses to physiological stimuli in patients presenting with bilateral adrenal incidentalomas. Endocrine 40(3), 437–444 (2011). https://doi.org/10.1007/s12020-011-9490-1. Epub 2011 May 20
E. Vassilatou, A. Vryonidou, D. Ioannidis, S.A. Paschou, M. Panagou, I. Tzavara, Bilateral adrenal incidentalomas differ from unilateral adrenal incidentalomas in subclinical cortisol hypersecretion but not in potential clinical implications. Eur. J. Endocrinol. 171(1), 37–45 (2014). https://doi.org/10.1530/EJE-13-0848. Epub 2014 Apr 17
J.D. Pasternak, C.D. Seib, N. Seiser, J.B. Tyrell, C. Liu, R.M. Cisco et al. Differences between bilateral adrenal incidentalomas and unilateral lesions. JAMA Surg. 150(10), 974–978 (2015). https://doi.org/10.1001/jamasurg.2015.1683
E. Debillon, F.L. Velayoudom-Cephise, S. Salenave, P. Caron, P. Chaffanjon, T. Wagner et al. Unilateral adrenalectomy as a first-line treatment of cushing’s syndrome in patients with primary bilateral macronodular adrenal hyperplasia. J. Clin. Endocrinol. Metab. 100(12), 4417–4424 (2015). https://doi.org/10.1210/jc.2015-2662. Epub 2015 Oct 9
Y. Xu, W. Rui, Y. Qi, C. Zhang, J. Zhao, X. Wang et al. The role of unilateral adrenalectomy in corticotropin-independent bilateral adrenocortical hyperplasias. World J. Surg. 37(7), 1626–1632 (2013). https://doi.org/10.1007/s00268-013-2059-9
V. Morelli, G. Reimondo, R. Giordano, S. Della Casa, C. Policola, S. Palmieri et al. Long-term follow-up in adrenal incidentalomas: an Italian multicenter study. J. Clin. Endocrinol. Metab. 99(3), 827–834 (2014). https://doi.org/10.1210/jc.2013-3527. Epub 2014 Jan 1
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Araujo-Castro, M., Sampedro Núñez, M.A. & Marazuela, M. Autonomous cortisol secretion in adrenal incidentalomas. Endocrine 64, 1–13 (2019). https://doi.org/10.1007/s12020-019-01888-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-019-01888-y