Abstract
Purpose of Review
The aim of the study is to assess historical anatomical and functional definitions of Wernicke’s area in light of modern lesion and neuroimaging data.
Recent Findings
“Wernicke’s area” has become an anatomical label usually applied to the left posterior superior temporal gyrus and adjacent supramarginal gyrus. Recent evidence shows that this region is not critical for speech perception or for word comprehension. Rather, it supports retrieval of phonological forms (mental representations of phoneme sequences), which are used for speech output and short-term memory tasks. Focal damage to this region produces phonemic paraphasia without impairing word comprehension, i.e., conduction aphasia. Neuroimaging studies in recent decades provide evidence for a widely distributed temporal, parietal, and frontal network supporting language comprehension, which does not include the anatomically defined Wernicke area.
Summary
The term Wernicke’s area, if used at all, should not be used to refer to a zone critical for speech comprehension.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Functional brain imaging and MRI lesion-deficit correlation studies over the past several decades have dramatically reawakened interest in the neurobiology of language and aphasia while raising questions about many aspects of classical aphasia models. Several recent reviews focused on the concept of “Wernicke’s area” and how it might be interpreted in light of this new information [1, 2, 3•, 4•]. Here, I attempt to reconcile and synthesize some of these views and to present a clinically useful and straightforward model of Wernicke aphasia and related posterior aphasia syndromes.
Unpacking “Speech Comprehension”
One point garnering universal consensus recently is that there appears to be nothing like a localized “speech comprehension center” in the brain. One reason for this conclusion is that speech comprehension is itself not a single process. Imagine a task in which a patient is asked to decide if two meaningless speech sounds, say “kah” and “gah,” are identical, a task called phoneme discrimination. To perform the task, a complex cortical auditory network must identify the phonemes (consonant and vowel sounds) in each stimulus and hold these identities briefly in memory, but word meanings play no role in the task. Now imagine a task in which a patient is asked to decide which of two pictures matches the spoken word “frog.” In this case, the phonemes of frog must be identified, but there is an additional, qualitatively different process of retrieving a meaning or concept associated with the particular string of phonemes. Both of these processes, the first dealing with the auditory information in the stimulus and the second with retrieving an associated word concept, are necessary for speech comprehension. As might be expected, these distinct processes also have very different localization in the brain; thus, it serves no useful purpose to think of speech comprehension as a single, localized process.
Wernicke understood this two-stage model of comprehension well (Fig. 1), as did many subsequent authors who distinguished between a “sensory speech area” that processes the sounds of speech and a more broadly distributed “concept field” that stores word meanings [5,6,7]. Unfortunately, this critical distinction seems not to have survived translation into the clinical arena and was lost altogether from late twentieth century introductory neurology textbooks. As discussed below, extensive evidence from functional imaging and lesion-deficit correlation studies showing distinct phoneme perception and semantic memory networks in the brain can only be understood in terms of this two-stage model, and any discussion of what Wernicke’s area might mean must begin with an acceptance of this distinction.
Wernicke’s 1874 drawings (slightly modified from the original for clarity) illustrating centers and pathways supporting language. Figure 3 shows the word sound center (a 1 ) in the STG, which analyzes incoming speech sounds and guides the motor speech center (b) during speech output. Figure 4 shows, in addition, pathways linking the word sound center to the centers for tactile (c) and optical (d) memory images, which Wernicke believed were necessary for retrieval of the word concept. (“The concept is nothing more than the path cd.” [5])
A second aspect of Wernicke’s original model also needs clarification. In Wernicke’s view, the “word sound images” activated during speech perception are also activated during speech production. Their role in speech production, according to this view, is to provide a kind of template for the sounds to be produced, thereby “guiding” the motor system during articulation [5]. Damage to the sound images was therefore believed to not only impair speech perception but also result in errors (paraphasia) during speech production. As discussed below, the weight of the evidence suggests instead that there are separate neural systems in the superior temporal region for phoneme perception and for guiding motor output during speech production. A debate has ensued regarding which of these systems ought properly to be labeled Wernicke’s area [1].
Anatomical Definitions of Wernicke’s Area
Accompanying the confusion about speech comprehension processes was a gradual tendency by neurologists to attribute these processes to the posterior superior temporal gyrus (pSTG) and to label this structure Wernicke’s area. Wernicke originally localized his “auditory images” to the entire STG, based on what was at the time a well-educated guess that subcortical auditory pathways probably projected to the STG [5]. Subsequent generations of neurologists refined the localization to pSTG, or to pSTG and adjacent supramarginal gyrus [8,9,10,11]. Many authors over the years have disputed this localization, variously including parts of the middle temporal gyrus, angular gyrus, and even inferior temporal gyrus in their definition of Wernicke’s area [12,13,14,15,16], though in most of these cases, it is unclear whether the claims referred to Wernicke’s phoneme perception process or to a nonspecific speech comprehension function.
Much of modern thinking on this topic is influenced by the work of Geschwind, who followed earlier authors in emphasizing the role of the pSTG in Wernicke aphasia [17]. Geschwind was heavily influenced by findings of left-right asymmetry in the size of the planum temporale, which is the cortex posterior to Heschl’s gyrus on the dorsal STG. This structure is larger on the left side in approximately two thirds of right-handed people [18,19,20]. Geschwind and others interpreted this asymmetry as confirming a central role for the pSTG in language [21,22,23] and as the site where damage causes Wernicke aphasia. Many late twentieth century textbooks and review articles thus equate the pSTG with Wernicke’s area [17, 24,25,26]. Today, the label is reinforced on numerous internet sites, including Wikipedia (http://en.wikipedia.org/wiki/Wernicke's_area).
What has remained unclear as this evolution of terminology has proceeded is what actual process or processes are carried out by the pSTG and surrounding areas. The following sections review some more recent evidence on this critical issue.
Posterior Superior Temporal Gyrus and Conduction Aphasia
Autopsy reports from the pre-CT era are replete with cases of conduction aphasia resulting from focal damage to the pSTG [11, 27, 28], often accompanied by ad hoc explanations of why speech comprehension remained intact. CT and MRI studies have since confirmed that conduction aphasia is a typical result of focal lesions of the pSTG and SMG, sometimes including the posterior insula [29,30,31,32, 33•]. The constellation of phonemic paraphasia and intact comprehension that characterizes conduction aphasia is also elicited by cortical electrical stimulation of the pSTG and adjacent cortex [34,35,36,37].
According to the classical Wernicke-Lichtheim aphasia model, intact comprehension is not possible if Wernicke’s sensory speech area is damaged; thus, the combination of paraphasic speech and intact comprehension was explained as a disconnection between the intact sensory speech area and the intact motor speech area. Damage to this connection, later identified as the arcuate fasciculus, prevented the word sound images in Wernicke’s area from properly guiding the motor area, resulting in paraphasia.
An alternative view, strongly favored by evidence accumulating over several decades, is that conduction aphasia results from damage to a cortical region separate from both sensory speech perception and speech articulation systems. The function of this region, which includes the pSTG and adjacent cortex in the superior temporal sulcus and supramarginal gyrus, is to store and mentally activate phonological (speech sound) forms [38]. To demonstrate this function, consider that in reading this sentence, it is possible without much effort to recognize that the word snow rhymes with the word blow but not with plow. This “hearing in the mind” occurs without the need for any external speech input or overt speech production, indicating that there are ensembles of neurons in the brain that, when activated, produce a “mental image” of speech. A modern term for this process is phonological representation, where “representation” refers to both the mental image activated and the process of activation, and “phonological” specifies that it is the spoken form (as opposed to the written form or the meaning) of the word that is activated. Alternative terms in the literature include phonological encoding, phonological access, phonological retrieval, and others.
In this alternative account, phonological paraphasia occurs when brain damage disrupts the phonological representation itself, causing deletion, duplication, reordering, and other errors in the phoneme sequence that makes up the representation [39,40,41]. Phonological representation is a necessary stage prior to all speech output tasks, including spontaneous speech, naming, reading aloud, and repetition; thus, impairment causes paraphasia in all of these tasks. Importantly, the same phonological representations are also used to maintain speech sounds in short-term memory, such as when holding a phone number or other meaningless list in mind for a brief period [33•, 42•].
Four types of evidence support this new view of conduction aphasia and the pSTG. First are the previously mentioned studies of phonological paraphasia elicited by electrical cortical stimulation [34,35,36,37], which demonstrate that cortical disruption in this region (without white matter damage) can produce the paraphasic deficit, and do so without disrupting speech comprehension. Second are a large number of functional imaging studies that link neural activation in this region specifically with activation of phonological representations or maintenance of phonological representations during short-term memory tasks [33•, 42•, 43,44,45,46,47,48,49,50,51,52] (Fig. 2). Third is a recent lesion-deficit correlation study that identified sites where damage from stroke impairs performance on a silent rhyme matching task illustrated by the snow-blow-plow example above [53•]. This task requires no motor speech output, so failure cannot be ascribed to a disconnection between auditory and motor representations. Impairment on this task was correlated with damage to the left pSTG and supramarginal gyrus, indicating that patients with lesions in this region are unable to retrieve an internal mental image of the phonemes represented by the written words. Notably, lesions in this region did not cause impairment on a control task in which patients had to match written words on similarity of meaning, indicating that the deficit is specifically one of retrieving a phonological representation. Finally, the deficits observed with cortical degeneration in this brain region are consistent with an impairment of phonological representation. This syndrome, known as logopenic variant primary progressive aphasia (lvPPA), is characterized by phonological paraphasia, impaired verbal short-term memory, and intact word comprehension [54,55,56]. Other components of the lvPPA syndrome include anomia and varying degrees of sentence comprehension impairment. These components can also be explained by phonological representation failure, if one assumes that a severe deficit could cause complete failure of phonological retrieval (i.e., anomia) as well as inability to maintain a string of words in short-term memory during sentence comprehension.
As this discussion suggests, the concept of phonological representation is central to understanding many language tasks, and impairments in this process account for a variety of related deficits. The broad evidence linking this function to pSTG and adjacent inferior parietal regions suggests that the anatomical zone traditionally designated Wernicke’s area plays a much larger role in speech production and verbal short-term memory tasks than in speech comprehension.
Phoneme Perception and Phoneme Deafness
Wernicke and other early aphasiologists believed that speech comprehension deficits from temporal lobe damage were the result of inability to recognize speech sounds. Damage to the sensory speech area was thought to prevent activation of the auditory images of spoken words. Modern evidence, however, suggests that sensory perception of phonemes (i.e., ability to distinguish between different speech sounds) is intact in most aphasia patients with comprehension impairment and that the deficit in these patients arises instead from inability to assign meaning to the speech sounds [57, 58]. Sparing of phoneme perception, even in patients with large temporal lobe lesions, is due to the fact that phoneme perception occurs bilaterally and can be supported independently by either hemisphere in isolation [59, 60]. Although patients with true phoneme perception impairment have been reported, this “pure word deafness” syndrome is rare because it generally requires bilateral temporal lobe lesions [7, 61, 62].
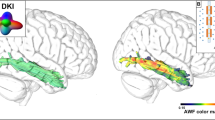
Functional neuroimaging studies have consistently localized phoneme perception to the lateral STG and adjacent superior temporal sulcus [63, 64, 65•]. The most consistent activation occurs in the mid-portion of the gyrus, anterior to the classical Wernicke area (Fig. 3). Consistent with the lesion evidence, activation in this region is bilateral, though typically stronger on the left side. Given its location, some authors have advocated “moving” the Wernicke area designation from pSTG to this more anterior location [65•, 66]. Two considerations weigh against this proposal. First is the evidence, mentioned above, that phoneme perception is intact in most patients with comprehension impairment. Thus, damage to this anterior phoneme perception region, even when it occurs, is not the cause of comprehension impairment in Wernicke aphasia. Second, Wernicke believed that one major function of his sensory speech center was to “guide” speech production. Although he was mistaken about the details of this auditory-motor transformation process, it turns out that damage to the pSTG reliably causes the phonological paraphasia he was attempting to explain. From a theoretical point of view, therefore, the pSTG has as much or more claim to the title of Wernicke’s area than the more anterior phoneme perception zone.
Meta-analysis of 22 fMRI studies comparing brain activation to speech vs. nonspeech control sounds, highlighting a mid-STG region (larger on the left) where responses are reliably stronger for speech sounds (adapted with permission from DeWitt I, Rauschecker JP. Phoneme and word recognition in the auditory ventral stream. Proc Natl Acad Sci U S A. 2012;109:E505–14). The more posterior region usually called Wernicke’s area is indicated with a dashed line
Semantic Memory and Comprehension Impairment
Given the considerable evidence, reviewed above, that lesions of the STG are not responsible for comprehension impairments in Wernicke aphasia, the question naturally arises: Where are the lesions responsible for this impairment? Examination of the extensive pre-CT era literature on this topic shows that lesions associated with Wernicke aphasia (including Wernicke’s two original cases; Fig. 4) are relatively large, typically including portions of the middle temporal gyrus (MTG) and angular gyrus in addition to STG and supramarginal gyrus [67], suggesting that these regions surrounding the STG may be critical for comprehension. More recently, several large, quantitative lesion-deficit mapping studies demonstrated an association between comprehension impairment and damage to MTG, angular gyrus, anterior STG, and several areas in left prefrontal cortex, but no association with damage to pSTG or SMG [68,69,70,71,72]. A smaller literature reports on lesions associated with transcortical sensory aphasia, a syndrome in which language comprehension is impaired but phonological processing is intact, as exemplified by the patients’ preserved ability to repeat aloud words and sentences they cannot understand. This syndrome is associated with large lesions affecting the left inferolateral temporal lobe (MTG, inferior temporal gyrus, fusiform gyrus) and angular gyrus, sparing the STG and supramarginal gyrus [73,74,75,76]. Notably, left frontal lobe lesions can also produce this syndrome [77,78,79].
A depiction of the lesions in Wernicke’s two original cases, inferred from the text description, with the principal areas cited as damaged listed below each drawing. In retrospect, Wernicke’s main conclusion (“We can thus assert with certainty that the softening of the first temporal convolution in the case of Rother was the only brain lesion which could have caused the localized symptom of aphasia.”[5]) appears somewhat premature
Comprehension impairment is a defining feature of the semantic variant of primary progressive aphasia (svPPA), also known as semantic dementia [80]. Similar to transcortical sensory aphasia, patients with svPPA are unable to comprehend words and sentences normally, but show normal phonological skills in speech repetition and other tasks. Cortical degeneration in this condition is focused in the anterior half of the temporal lobe, well outside the standard Wernicke area [3•, 81, 82]. The comprehension impairment in svPPA has been extensively studied and shown to be due to loss of basic concept knowledge, or semantic memory [80, 83,84,85]. Patients lose their knowledge of basic facts, such as the color, shape, size, and function of common objects.
Considered together, the data from stroke patients and patients with svPPA suggest that language comprehension depends on a large, distributed network including much of the inferolateral and anterior temporal lobe, angular gyrus, and prefrontal cortex. Extensive evidence from functional neuroimaging studies supports this conclusion. A meta-analysis of 120 such studies, focusing on retrieval of semantic information during word and sentence comprehension tasks, identified a widespread network of brain regions involved in this function, including angular gyrus, MTG, ventral temporal lobe, medial parietal cortex, medial prefrontal cortex, and inferior lateral prefrontal regions [86]. These results closely mirror the previously mentioned lesion data. Together, the data converge on the conclusion that many cortical regions support language comprehension, whereas the classical Wernicke area is one of the few brain regions that does not.
Middle Temporal Gyrus: a Language Comprehension Hub?
Much recent research on language comprehension networks in the brain has turned to the problem of understanding the roles of various nodes in this large system. At a broad level, frontal lobe components of the system, such as the pars orbitalis of the inferior frontal gyrus (Brodmann area 47) and the superior frontal gyrus, appear to be involved in initiating and controlling the activation of semantic information, rather than in long-term storage of the information per se [87,88,89,90]. Conversely, the temporal and inferior parietal components of the system are believed to function as the actual repositories of concept knowledge. Within this large posterior subnetwork, there appear to be many subregions defined by different types of knowledge content and varying degrees of intramodal and multimodal integration [91,92,93,94,95,96]. Defining these knowledge subnetworks is an ongoing area of active research.
The MTG, particularly its posterior half, occupies a unique position in the larger comprehension network, in that it is situated just below the pSTG/supramarginal phonological system, and may provide an interface or “hub” linking the inferior and anterior temporal lobe semantic memory system with the phonological system. In a study exploring the functional and structural connectivity of five regions in the comprehension network, Turken and Dronkers [97••] demonstrated that the posterior MTG has the most extensive connectivity of these regions, linking with angular gyrus, anterior and inferior temporal lobe, inferior and superior frontal gyri, and homologous nodes in the right hemisphere (Fig. 5a). This broad connectivity is due in large part to the fact that posterior MTG lies at a crossroad of several large white matter tracts linking these regions, including the inferior longitudinal, middle longitudinal, arcuate, and inferior fronto-occipital fasculi.
a Resting-state fMRI functional connectivity of five regions implicated in a large voxel-based lesion study of spoken language comprehension impairment [71]. The region covering the posterior half of the middle temporal gyrus (yellow) has the most widespread connectivity (adapted with permission from [97••]). b Results of another voxel-based lesion study, showing left hemisphere regions where damage produces greater impairment of spoken language comprehension than impairment of picture naming (adapted with permission from [98])
One likely role for such a hub is to facilitate comprehension of sentences, a complex task that requires analyzing the meanings of multiple words and the syntactic role of each word, while combining this information gradually into an overall meaning. Because these processes must unfold over several seconds as the sentence is presented, the phonologic, semantic, and syntactic information extracted must be maintained in short-term memory for a period of time. The function of the posterior MTG might therefore be to integrate word meanings activated throughout the semantic memory network, while maintaining the word forms and meanings in short-term memory through interactions with the phonological system and frontal lobe control systems. In a recent voxel-based lesion study, Pillay et al. tested this hypothesis by identifying lesions associated with phrase and sentence comprehension deficits [98]. Performance on a picture naming task was included as a control for nonspecific phonological and executive dysfunction. The posterior half of the left MTG was the only location where damage caused greater phrase and sentence comprehension impairment than naming impairment (Fig. 5b). Thus, the posterior MTG appears to be particularly critical for sentence comprehension, probably due to its widespread connectivity with semantic memory, phonological, and frontal control networks.
A Model of Posterior Language Networks and Related Aphasias
Figure 6 presents a visual summary of key temporal and parietal lobe language components and their participation in some common tasks used to assess aphasia. These tasks include
-
1.
Speech repetition, which requires phoneme perception (yellow region in Fig. 6), followed by transformation of the auditory input into a phonological representation (Wernicke’s area, blue), which is then transformed into a motor sequence. Unilateral lesions in the phoneme perception area typically cause no impairment because this function is represented in both hemispheres. Lesions to the phonological representation system result in phonological paraphasia.
-
2.
Single-word comprehension, which for spoken words require phoneme perception, followed by activation of an associated concept in the semantic memory network (red and green regions in Fig. 6). Damage to the semantic memory component is typically extensive in patients with single-word comprehension impairment, the most severe deficits occurring with bilateral damage to the anterior temporal component [3].
-
3.
Sentence comprehension, which in addition to the processes engaged by word comprehension, requires integration of multiple word meanings and maintenance of word sounds and meanings in short-term memory. This integration process is particularly vulnerable to lesions in the mid-to-posterior MTG.
-
4.
Sentence (spontaneous speech) production, which requires integration of multiple word concepts, followed by activation of corresponding phonological representations, followed by transformation into a motor sequence. Lesions confined to Wernicke’s area (phonological representation) result in phonological paraphasia during spontaneous speech, whereas lesions in the semantic system sparing Wernicke’s area result in verbal paraphasia (incorrect word selection), anomia, and “empty” speech lacking content.
A schematic model of posterior language zones and their involvement in several language tasks. The region often called Wernicke’s area (blue) supports activation of phonological representations, which play a central role in all speech output tasks (including reading, not shown here) and a supporting role in holding words in phonological short-term memory during sentence comprehension. The posterior half of the MTG (green) is proposed to serve as a comprehension “hub” that integrates the meaning of multiword utterances during sentence comprehension and production. Lesions in this latter region likely account for most of the language comprehension impairments observed in patients with Wernicke aphasia
From this model, it is easy to understand the Wernicke aphasia syndrome as a commonly occurring combination of damage to phonological representations in Wernicke’s area (resulting in the paraphasic speech characteristic of the syndrome) together with varying amounts of damage to the surrounding semantic memory network, resulting in varying degrees of comprehension impairment. When the surrounding damage is confined to the MTG, the comprehension impairment is likely to affect mainly sentence comprehension and spare single-word recognition (for example, ability to match words with pictures).
It should be emphasized that all of these processes are under varying degrees of “control” by frontal systems. Posterior inferior frontal regions (precentral gyrus and pars opercularis) interact closely with Wernicke’s area to maintain phonological representations in short-term memory, and more anterior inferior frontal regions (pars orbitalis and triangularis) likely control activation of particular concepts in semantic memory depending on context and task demands [87,88,89,90].
Conclusion: Is Wernicke’s Area a Useful Concept?
Current controversies regarding the meaning of Wernicke’s area are understandable given the incompatibilities between classical notions of this region and the modern empirical evidence just reviewed. To summarize,
-
The anatomical region now almost universally called Wernicke’s area (pSTG and adjacent supramarginal gyrus) likely plays no role in speech comprehension. The main function of this region is phonological representation prior to speech production.
-
The classical concept of a single phonological representation supporting both speech comprehension and speech production is not supported by the evidence. Phonological representation and phoneme perception show different localization in fMRI studies. Distinct lesions produce phonological paraphasia and phoneme deafness.
-
The classical notion that comprehension impairment in Wernicke aphasia results from damage to a sensory speech perception process is mistaken. The comprehension deficit in this syndrome is unrelated to phoneme perception ability and instead reflects an inability to activate word and sentence meaning.
Given these significant incompatibilities, does the term Wernicke’s area retain any clinical utility? As discussed above, proposals to move Wernicke’s area to the more anterior phoneme perception zone seem ill considered on both theoretical and historical grounds. As discussed elsewhere, the notion of redefining Wernicke’s area to include all the regions where damage causes comprehension impairment would render the term anatomically meaningless [2]. Tremblay and Dick [4•] recently proposed that the terms Wernicke’s area and “Broca’s area” both be abandoned, though the argument in the case of Wernicke’s area was based on small variations in how the term is applied anatomically, rather than on the more substantial theoretical problems outlined above.
In my view, the use of the term Wernicke’s area is problematic and confusing because of its continued strong association with speech comprehension. As discussed above, speech comprehension is not a unitary process, and the anatomical region typically labeled Wernicke’s area plays little or no role in this complex function. Given these considerations, the term should never be used to mean “speech comprehension area.” It may retain some usefulness as an anatomical label for the cortex surrounding the lower bank of the posterior Sylvian fissure (pSTG and posterior supramarginal gyrus) and as shorthand for “cortex involved in phonological representation” or “cortex where a focal lesion causes conduction aphasia.”
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
DeWitt I, Rauschecker JP. Wernicke’s area revisited: parallel streams and word processing. Brain Lang. 2013;127(2):181–91.
Binder JR. The Wernicke area: modern evidence and a reinterpretation. Neurology. 2015;85:2170–5.
• Mesulam MM, Thompson CK, Weintraub S, Rogalski EJ. The Wernicke conundrum and the anatomy of language comprehension in primary progressive aphasia. Brain. 2015;138(8):2423–37. This study uses data from a large cohort of patients with primary progressive aphasia to examine the neural correlates of language comprehension, concluding that word comprehension depends mainly on the left anterior temporal lobe.
• Tremblay P, Dick AS. Broca and Wernicke are dead, or moving past the classic model of language neurobiology. Brain Lang. 2016;162:60–71. This paper presents interesting survey data on current use of the terms Broca's area and Wernicke's area among language scientists and clinicians, and argues that a lack of agreement renders these terms largely meaningless.
Wernicke C. Der aphasische Symptomenkomplex. Cohn & Weigert: Breslau; 1874.
Lichtheim L. On aphasia. Brain. 1885;7:433–84.
Henschen SE. On the hearing sphere. Acta Otolaryngol (Stockh). 1918-1919;1:423–86.
Bastian HC. On different kinds of aphasia, with special reference to their classification and ultimate pathology. Br Med J. 1887;2:931–6. 85-90
Déjerine J. Anatomie des centres nerveux. Paris: Rueff; 1895.
Pick A. Aphasia. Berlin: Springer; 1931.
Kleist K. Sensory aphasia and amusia. London: Pergamon Press; 1962.
Starr MA. The pathology of sensory aphasia, with an analysis of fifty cases in which Broca’s centre was not diseased. Brain. 1889;12:82–101.
Marie P. On aphasia in general and agraphia in particular according to the teaching of Professor Charcot. Reprinted from Le Progres Medical, Series 2, 1888; 7: 81-84. Pierre Marie's Papers on Speech Disorders. New York: Hafner; 1971.
Goldstein K. Language and language disturbances. New York: Grune & Stratton; 1948.
Penfield W, Roberts L. Speech and brain-mechanisms. New York: Atheneum; 1959.
Bogen JE, Bogen GM. Wernicke’s region—where is it? Ann N Y Acad Sci. 1976;290:834–43.
Geschwind N. Aphasia. N Engl J Med. 1971;284(12):654–6.
Geschwind N, Levitsky W. Human brain: left-right asymmetries in temporal speech region. Science. 1968;161:186–7.
Wada JA, Clarke R, Hamm A. Cerebral hemispheric asymmetry in humans. Arch Neurol. 1975;32:239–46.
Steinmetz H, Volkmann J, Jäncke L, Freund H-J. Anatomical left-right asymmetry of language-related temporal cortex is different in left- and right-handers. Ann Neurol. 1991;29:315–9.
Galaburda AM, LeMay M, Kemper T, Geschwind N. Right-left asymmetries in the brain. Structural differences between the hemispheres may underlie cerebral dominance. Science. 1978;199:852–6.
Witelson SF, Kigar DL. Sylvian fissure morphology and asymmetry in men and women: bilateral differences in relation to handedness in men. J Comp Neurol. 1992;323:326–40.
Foundas AL, Leonard CM, Gilmore R, et al. Planum temporale asymmetry and language dominance. Neuropsychologia. 1994;32:1225–31.
Benson DF. Aphasia, alexia and agraphia. New York: Churchill Livingstone; 1979.
Mayeux R, Kandel ER. Natural language, disorders of language, and other localizable disorders of cognitive function. In: Kandel ER, Schwartz J, editors. Principles of Neural Science. 2nd ed. New York: Elsevier Science Publishing Co.; 1985. p. 688–703.
Mesulam MM. Large-scale neurocognitive networks and distributed processing for attention, language, and memory. Ann Neurol. 1990;28:597–613.
Benson DF, Sheremata WA, Bouchard R, et al. Conduction aphasia. A clinicopathological study. Arch Neurol. 1973;28:339–46.
Boller F. Destruction of Wernicke’s area without language disturbance. A fresh look at crossed aphasia. Neuropsychologia. 1973;11:243–6.
Damasio H, Damasio AR. The anatomical basis of conduction aphasia. Brain. 1980;103:337–50.
Palumbo CL, Alexander MP, Naeser MA. CT scan lesion sites associated with conduction aphasia. In: Kohn SE, editor. Conduction aphasia. Hillsdale, NJ: Lawrence Erlbaum; 1992. p. 51–75.
Axer H, Keyserlingk AG, Berks G, Keyserlingk DF. Supra- and infrasylvian conduction aphasia. Brain Lang. 2001;76:317–31.
Fridriksson J, Kjartansson O, Morgan PS, et al. Impaired speech repetition and left parietal lobe damage. J Neurosci. 2010;30:11057–61.
• Buchsbaum BR, Baldo J, D’Esposito M, et al. Conduction aphasia, sensory-motor integration, and phonological short-term memory: an aggregate analysis of lesion and fMRI data. Brain Lang. 2011;119:119–28. This paper presents a multi-faceted review and meta-analysis of neuroimaging data regarding phonological processing.
Anderson JM, Gilmore R, Roper S, et al. Conduction aphasia and the arcuate fasciculus: a reexamination of the Wernicke-Geschwind model. Brain Lang. 1999;70:1–12.
Quigg M, Fountain NB. Conduction aphasia elicited by stimulation of the left posterior superior temporal gyrus. J Neurol Neurosurg Psychiatry. 1999;66:393–6.
Corina DP, Loudermilk BC, Detwiler L, et al. Analysis of naming errors during cortical stimulation mapping: implications for models of language representation. Brain Lang. 2010;115:101–12.
Roux FE, Durand JB, Jucla M, et al. Segregation of lexical and sub-lexical reading processes in the left perisylvian cortex. PLoS One. 2012;7(11):e50665.
Levelt WJM. Speaking: from intention to articulation. Cambridge, MA: MIT Press; 1989.
Caplan D, Waters G. Issues arising regarding the nature and consequences of reproduction conduction aphasia. In: Kohn SE, editor. Conduction aphasia. Hillsdale, NJ: Lawrence Erlbaum; 1992. p. 117–49.
Wilshire CE, McCarthy RA. Experimental investigations of an impairment in phonological encoding. Cogn Neuropsychol. 1996;13(7):1059–98.
Dell GS, Schwartz MF, Martin N, et al. Lexical access in aphasic and nonaphasic speakers. Psychol Rev. 1997;104(4):801–38.
• Acheson DJ, Hamidi M, Binder JR, Postle BR. A common neural substrate for language production and verbal working memory. J Cogn Neurosci. 2011;23:1358–67. This study presents combined fMRI and transcranial magnetic stimulation evidence for a common posterior STG localization for phonological representation and phonological short-term memory .
Booth JR, Burman DD, Meyer JR, et al. Functional anatomy of intra- and cross-modal lexical tasks. NeuroImage. 2002;16:7–22.
Xu B, Grafman J, Gaillard WD, et al. Neuroimaging reveals automatic speech coding during perception of written word meaning. NeuroImage. 2002;17(2):859–70.
Hickok G, Buchsbaum B, Humphries C, Muftuler T. Auditory-motor interaction revealed by fMRI: speech, music, and working memory in area Spt. J Cogn Neurosci. 2003;15(5):673–82.
Indefrey P, Levelt WJM. The spatial and temporal signatures of word production components. Cognition. 2004;92(1–2):101–44.
Buchsbaum BR, Olsen RK, Koch P, Berman KF. Human dorsal and ventral auditory streams subserve rehearsal-based and echoic processes during verbal working memory. Neuron. 2005;48(4):687–97.
Burton MW, Locasto PC, Krebs-Noble D, Gullapalli RP. A systematic investigation of the functional neuroanatomy of auditory and visual phonological processing. NeuroImage. 2005;26(3):647–61.
Graves WW, Grabowski TJ, Mehta S, Gordon JK. A neural signature of phonological access: distinguishing the effects of word frequency from familiarity and length in overt picture naming. J Cogn Neurosci. 2007;19(4):617–31.
Bruno JL, Zumberge A, Manis FR, et al. Sensitivity to orthographic familiarity in the occipito-temporal region. NeuroImage. 2008;39:1988–2001.
Wilson SM, Isenberg AK, Hickok G. Neural correlates of word production stages delineated by parametric modulation of psycholinguistic variables. Hum Brain Mapp. 2009;30:3596–608.
Wilson LB, Tregellas JR, Slason E, et al. Implicit phonological priming during visual word recognition. NeuroImage. 2011;55(2):724–31.
• Pillay SB, Stengel BC, Humphries C, et al. Cerebral localization of impaired phonological retrieval during rhyme judgment. Ann Neurol. 2014;76:738–46. This study presents lesion evidence linking the classical Wernicke's area with pre-articulatory (i.e., silent) retrieval of phonological word forms.
Gorno-Tempini ML, Brambati SM, Ginex V, et al. The logopenic/phonological variant of primary progressive aphasia. Neurology. 2008;71(16):1227–34.
Rohrer JD, Ridgway GR, Crutch SJ, et al. Progressive logopenic/phonological aphasia: erosion of the language network. NeuroImage. 2010;49:984–93.
Leyton CE, Ballard KJ, Piguet O, Hodges JR. Phonologic errors as a clinical marker of the logopenic variant of PPA. Neurology. 2014;82(18):1620–7.
Blumstein SE, Baker E, Goodglass H. Phonological factors in auditory comprehension in aphasia. Neuropsychologia. 1977;15:19–30.
Blumstein SE, Tartter VC, Nigro G, Statlender S. Acoustic cues for the perception of place of articulation in aphasia. Brain Lang. 1984;22:128–49.
Boatman D, Hart J, Lesser RP, et al. Right hemisphere speech perception revealed by amobarbital injection and electrical interference. Neurology. 1998;51(2):458–64.
Hickok G, Okada K, Barr W, et al. Bilateral capacity for speech sound processing in auditory comprehension: evidence from Wada procedures. Brain Lang. 2008;107:179–84.
Buchman AS, Garron DC, Trost-Cardamone JE, et al. Word deafness: one hundred years later. J Neurol Neurosurg Psychiatry. 1986;49:489–99.
Poeppel D. Pure word deafness and the bilateral processing of the speech code. Cogn Sci. 2001;25:679–93.
Binder JR, Frost JA, Hammeke TA, et al. Human temporal lobe activation by speech and nonspeech sounds. Cereb Cortex. 2000;10:512–28.
Liebenthal E, Binder JR, Spitzer SM, et al. Neural substrates of phonemic perception. Cereb Cortex. 2005;15:1621–31.
• DeWitt I, Rauschecker JP. Phoneme and word recognition in the auditory ventral stream. Proc Natl Acad Sci U S A. 2012;109:E505–14. This careful meta-analysis of fMRI speech perception studies provides strong evidence that auditory cortex specialized for speech perception lies anterior to the classical Wernicke's area .
Scott SK, Blank C, Rosen S, Wise RJS. Identification of a pathway for intelligible speech in the left temporal lobe. Brain. 2000;123:2400–6.
Binder JR. Wernicke aphasia: a disorder of central language processing. In: D’Esposito ME, editor. Neurological foundations of cognitive neuroscience. Cambridge, MA: MIT Press; 2002. p. 175–238.
Kertesz A, Lau WK, Polk M. The structural determinants of recovery in Wernicke’s aphasia. Brain Lang. 1993;44:153–64.
Dronkers NF, Redfern BB, Ludy CA. Lesion localization in chronic Wernicke’s aphasia. Brain Lang. 1995;51:62–5.
Damasio H, Tranel D, Grabowski T, et al. Neural systems behind word and concept retrieval. Cognition. 2004;92:179–229.
Dronkers NF, Wilkins DP, Van Valin RD, et al. Lesion analysis of the brain areas involved in language comprehension. Cognition. 2004;92:145–77.
Thothathiri M, Kimberg DY, Schwartz MF. The neural basis of reversible sentence comprehension: evidence from voxel-based lesion symptom mapping in aphasia. J Cogn Neurosci. 2012;24(1):212–22.
Damasio H. Cerebral localization of the aphasias. In: Sarno MT, editor. Acquired aphasia. Orlando: Academic Press; 1981. p. 27–50.
Kertesz A, Sheppard A, MacKenzie R. Localization in transcortical sensory aphasia. Arch Neurol. 1982;39:475–8.
Alexander MP, Hiltbrunner B, Fischer RS. Distributed anatomy of transcortical sensory aphasia. Arch Neurol. 1989;46:885–92.
Rapcsak SZ, Rubens AB. Localization of lesions in transcortical aphasia. In: Kertesz A, editor. Localization and neuroimaging in neuropsychology. San Diego: Academic Press; 1994. p. 297–329.
Otsuki M, Soma Y, Koyama A, et al. Transcortical sensory aphasia following left frontal infarction. J Neurol. 1998;245:69–76.
Maeshima S, Osawa A, Nakayama Y, Miki J. Transcortical sensory aphasia following infarction in the left frontal lobe. Eur Neurol. 2004;52(2):125–8.
Sethi NK, Burke L, Torgovnick J, Arsura E. Transcortical sensory aphasia as a result of left frontal cortical-subcortical infarction. A case report. Eur Neurol. 2007;57(1):52–3.
Hodges JR, Patterson K, Oxbury S, Funnell E. Semantic dementia: progressive fluent aphasia with temporal lobe atrophy. Brain. 1992;115:1783–806.
Rohrer JD, Warren JD, Modat M, et al. Patterns of cortical thinning in the language variants of frontotemporal lobar degeneration. Neurology. 2009;72:1562–9.
Rogalski E, Cobia D, Harrison TM, et al. Progression of language decline and cortical atrophy in subtypes of primary progressive aphasia. Neurology. 2011;76:1804–10.
Bozeat S, Lambon Ralph MA, Patterson K, et al. Nonverbal semantic impairment in semantic dementia. Neuropsychologia. 2000;38:1207–15.
Rogers TT, Garrard P, McClelland JL, et al. Structure and deterioration of semantic memory: a neuropsychological and computational investigation. Psychol Rev. 2004;111(1):205–35.
Patterson K, Nestor PJ, Rogers TT. Where do you know what you know? The representation of semantic knowledge in the human brain. Nat Rev Neurosci. 2007;8:976–87.
Binder JR, Desai R, Conant LL, Graves WW. Where is the semantic system? A critical review and meta-analysis of 120 functional neuroimaging studies. Cereb Cortex. 2009;19:2767–96.
Thompson-Schill SL, D’Esposito M, Aguirre GK, Farah MJ. Role of left inferior prefrontal cortex in retrieval of semantic knowledge: a reevaluation. Proc Natl Acad Sci U S A. 1997;94:14792–7.
Wagner AD, Pare-Blagoev EJ, Clark J, Poldrack RA. Recovering meaning: left prefrontal cortex guides semantic retrieval. Neuron. 2001;31:329–38.
Novick JM, Trueswell JC, Thompson-Schill SL. Cognitive control and parsing: reexamining the role of Broca’s area in sentence comprehension. Cognitive, Affective, & Behavioral Neuroscience. 2005;5(3):263–81.
Jefferies E, Lambon Ralph MA. Semantic impairment in stroke aphasia versus semantic dementia: a case-series comparison. Brain. 2006;129:2132–47.
Damasio AR. Time-locked multiregional retroactivation: a systems-level proposal for the neural substrates of recall and recognition. Cognition. 1989;33:25–62.
Simmons WK, Barsalou LW. The similarity-in-topography principle: reconciling theories of conceptual deficits. Cogn Neuropsychol. 2003;20(3):451–86.
Meyer K, Damasio A. Convergence and divergence in a neural architecture for recognition and memory. Trends Neurosci. 2009;32(7):376–82.
Binder JR, Desai RH. The neurobiology of semantic memory. Trends Cogn Sci. 2011;15(11):527–36.
Meteyard L, Rodriguez Cuadrado S, Bahrami B, Vigliocco G. Coming of age: a review of embodiment and the neuroscience of semantics. Cortex. 2012;48:788–804.
Fernandino L, Binder JR, Desai RH, et al. Concept representation reflects multimodal abstraction: a framework for embodied semantics. Cereb Cortex. 2016;26:2018–34.
•• Turken AU, Dronkers NF. The neural architecture of the language comprehension network: converging evidence from lesion and connectivity analyses. Frontiers in Systems Neuroscience. 2011;5:Article 1. This paper presents a thorough, multimodal examination of connectivity within a broadly distributed network supporting sentence comprehension, suggesting a special role for the posterior left MTG in this function.
Pillay SB, Binder JR, Humphries C, et al. Lesion localization of speech comprehension deficits in chronic aphasia. Neurology. 2017;88(10):970–5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Jeffrey R. Binder declares no potential conflicts of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Behavior
Rights and permissions
About this article
Cite this article
Binder, J.R. Current Controversies on Wernicke’s Area and its Role in Language. Curr Neurol Neurosci Rep 17, 58 (2017). https://doi.org/10.1007/s11910-017-0764-8
Published:
DOI: https://doi.org/10.1007/s11910-017-0764-8