Abstract
Purpose of Review
This review seeks to present an overview of the recent literature on the importance of CPAP and antihypertensive treatment adherence in blood pressure control of hypertensive patients, especially those with obstructive sleep apnea.
Recent Findings
Although it is unquestionable that a good adherence to CPAP and antihypertensive drugs is crucial to improvements in sleep-related symptoms, blood pressure levels (even the modest reductions of 2–2.5 mmHg achieved by CPAP treatment) and future cardiovascular risk, this adherence decreases over time, despite efforts made toward behavioral intervention and monitoring. Curiously, although taking a drug would seem to be easier than the use of CPAP treatment, based on current information, it seems that the compliance with drug treatment in hypertensive subjects is not better than that achieved with CPAP treatment in OSA patients with hypertension. However, some studies have shown some phenotypes of hypertensive and OSA patients with good adherence and better hypertensive effect, such as those with uncontrolled blood pressure (resistant and refractory hypertension), severe forms of sleep apnea, and more sleep-related symptoms, especially a higher degree of diurnal hypersomnia.
Summary
The positive effect of antihypertensive drugs and CPAP treatment on blood pressure levels depends on the degree of treatment adherence, especially in forms of uncontrolled hypertension, but this adherence decreases over time. Educational programs and new devices are needed to improve adherence to treatment in these patients, along with fuller understanding of the different patterns and phenotypes of non-adherence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
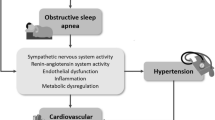
Arterial hypertension (AHT) is probably the cardiovascular disease that has been most consistently associated with sleep apnea syndrome (OSA) [1•]. Furthermore, it is also the cardiovascular disease in which treatment with continuous positive airway pressure (CPAP) has demonstrated efficacy in the most studies, with, according to different meta-analyses, a modest reduction of between 2 and 2.5 mmHg of systolic (SBP) or diastolic blood pressure (DBP) [2••]. There are some circumstances, however, in which the effect of CPAP appears to be greater, as in situations of lack of hypertensive control (i.e., resistant or refractory hypertension) [3••], patients with more severe OSA [4], and patients with good adherence to CPAP treatment [5•].
Some questions still remain unanswered about the importance of good adherence to antihypertensive drugs and CPAP treatment in hypertensive OSA patients. Is a different number of daily hours of CPAP use necessary to obtain an optimal response, according to the different clinical or cardiovascular outcomes? Is there a ground effect below which CPAP has no effect on BP levels, or a ceiling effect above which no greater efficiency is achieved? Is there a relationship between adherence to CPAP and adherence with other antihypertensive drugs? In other words, is there a non-compliant patient phenotype with respect to all the treatments? What is the most effective way of measuring a patient’s adherence? This review will attempt to address these and other issues related to the influence of adherence with CPAP and antihypertensive drugs on hypertensive patients with OSA.
Adherence to CPAP in OSA Patients
Continuous positive airway pressure (CPAP) therapy is the treatment of choice for most patients with severe or symptomatic OSA [6]. CPAP is a physical therapy based on the delivery of pressurized air into the nostrils, usually using a nasal interface, in order to avoid the repetitive episodes of upper airway obstruction during sleep characteristic of OSA. As in the case of other chronic treatments, adherence to CPAP is essential to attain any of its beneficial effects, but this adherence is also difficult to achieve [7•]. Fortunately, CPAP machines have an internal time counter that permits objective and detailed daily monitoring.
At present, the definition of adherence to CPAP is controversial and is based on consensus rather than on high-quality evidence. Given that respiratory events occur during sleep, CPAP should ideally be used every night and during the entire sleep period. In practice, however, this occurs only in a minority of subjects, so experts have agreed to define good adherence as an average use of CPAP for at least 4 h/night during 70% of nights [8].
This definition has some flaws, however. First, optimal adherence levels seem to differ, depending on the analyzed outcome [9, 10]. Adherence rates as low as 3 h/night may be sufficient to improve subjective daytime sleepiness and other OSA symptoms, whereas higher adherence rates of around 5 or 5.5 h/night are required to decrease BP measurements, particularly in non-sleepy patients [11]. Second, a dose-response relationship has been consistently shown between CPAP adherence and improvements in clinical outcomes and BP levels [12]. Third, this definition does not take into account different patterns of CPAP use that may override any cardiovascular benefit, despite a theoretically adequate average use [13]. For instance, some patients may use the device on working days but not at weekends, or they may sleep with CPAP for the first hours of the night and then remove it and sleep without it for the final hours, when the density of REM sleep is greater. Since there is evidence suggesting that OSA during REM sleep is associated with incident hypertension and cardiovascular outcomes, patients with predominantly REM-OSA who do not wear the CPAP device during all sleep time may be at greater risk of cardiovascular consequences [14].
Overall, adherence rates are poor, and between 30 and 85% of treated OSA patients use CPAP < 4 h/night [8, 12,13,14,15,16]. Unfortunately, suboptimal adherence is not easy to predict. There is much controversy and lack of agreement as to the best predictors of adherence to CPAP therapy in patients with OSA. Some variables such as sleepiness, age, gender, side effects, or OSA severity have been reported as influencing adherence in several studies but not in others [13, 15, 17,18,19,20,21,22]. It has been hypothesized that asymptomatic patients may have poorer adherence than sleepy OSA patients, as they may not perceive any substantial benefits from the treatment. The results of recent randomized controlled trials seem to support this hypothesis and suggest that patients from cardiovascular settings may have poorer adherence because they are usually less symptomatic and do not seek advice for sleep complaints [19, 23]. In contrast, however, two studies focusing on non-sleepy OSA patients have reported similar adherence rates to those enrolling symptomatic patients [18, 24], and one study conducted in non-sleepy patients with resistant hypertension followed up for 57 months observed good adherence rates in 74.5% of the sample [25••].
A recent review that analyzed CPAP adherence over a 20-year timeframe (1994–2015) reported an overall non-adherence rate, based on a 7 h/night sleep time, of 34.1%, and an average use of 4.5 h/night, without any improvements in adherence over this period—not even in recent years, despite efforts made toward behavioral intervention, telemedicine, and remote monitoring [26].
Adherence to Antihypertensive Treatment
Adherence to pharmacological treatment is one of the most important aspects that need to be considered when subjects do not achieve an adequate BP control [27••]. It is necessary and important to assess adherence to the pharmacological treatment because the magnitude of the antihypertensive effect will be influenced by this adherence. Moreover, a prescription of more antihypertensive drugs may lead to an increased risk of side effects and pharmaceutical interactions.
The adherence with antihypertensive therapy reported by different studies varies according to the methodology used. In a systematic review and meta-analysis involving 28 studies and 13,668 hypertensive subjects from 15 different countries, in which adherence was assessed using the 8-item Morisky medication adherence scale (MMAS-8), Abegaz et al. [28] found that 45% of hypertensive patients were non-adherent. Yang et al. [29] found, in a large cohort study of hypertensive patients in which adherence was assessed through the analysis of medical and pharmacy database records, that only 51.7% of patients presented high adherence to the treatment. In another study, the annual drop in pharmacological adherence was 4.3%, which was associated with a 1.68-fold increase in uncontrolled BP at follow-up [30]. In addition to poor BP control, non-adherence to antihypertensive treatment has been associated with an increased risk of cardiovascular events [28, 30].
Several factors have been associated with a drop in antihypertensive adherence. Some of these are modifiable: the number of pills or drugs; specific antihypertensive medication classes on tolerance, dosage, and side effects; associated symptoms of depressions; limited income [30, 31]; and social factors such as the organization and coverage of the health care system [31]. There are also non-modifiable factors, however: age, race, gender [31], associated treatment of co-morbidity, and marital status [30, 32]. Lower age, non-white race, and higher medication for co-morbidity have all been related with poorer adherence. All these factors could be helpful for identifying patients at risk of decreasing adherence to antihypertensive treatment and, therefore, poorer blood pressure control [30]. Moreover, these key data reported in the studies might be useful for planning strategies to improve adherence. For example, the side effects of antihypertensive drugs, such as excessive urination and decreased sexual drive, could have a predictive value for identifying patients with low adherence [33]. Therefore, any avoidance of or reduction in medication with these side effects in particular profiles of hypertensive subjects, or discussion of them, might be helpful in improving adherence. Another factor is the number of pills prescribed, as the higher this number, the worse the adherence. Thus, any reduction in this number by using fixed combinations of antihypertensive drugs results in an improvement in adherence [34, 35].
Furthermore, going beyond the identification of modifiable and non-modifiable factors, it might be useful to assess, in some way the adherence. Although direct methods (urine and blood samples, directly observed therapy) are considered the methods linked to the highest estimation of adherence, indirect methods (pill count, questionnaires, checking pharmacy database records, and using electronic monitoring systems) should not be undervalued, because beyond being cheaper, it reports complementary information. This means that direct methods confirm to us that the patient swallowed down the drugs, but those methods do not give any information regarding the behavior in daily life adherence of the patient out of the office. Therefore, it is considered that the best estimation of the medication adherence is achieved by combining methods of the two types, a direct objective one and an another indirect, more subjective [36,37,38].
The benefits of improving adherence with treatment are unquestionable. Not only good adherence to antihypertensive treatment implies clinically significant reductions in BP [39], but also it has been associated with a reduced risk of coronary and cerebrovascular events [40] and mortality [41]. Even modest reductions in BP achieved through the strategy of improving adherence with medication have been associated with improvements in mortality. A reduction in SBP of 3 mmHg has been associated with an 8% reduction in stroke mortality and a 5% reduction in mortality from coronary heart disease [40].
Effect of CPAP Adherence on BP Levels
The impact of CPAP treatment on BP control in OSA patients with hypertension has long been a topic of interest, and several studies and meta-analyses have investigated it. All these studies have reported different factors that might influence the BP response to CPAP in OSA patients, such as being treatment-naïve hypertensive or receiving associated drug treatment, as well as the time that has passed since the start of the CPAP treatment; the presence or not of sleepiness; and the degree or phenotype of hypertension. One key factor that may continue having a crucial impact, over and above all these circumstances, is adherence with CPAP in terms of hours per night of use of the device. This last aspect might be such important that probably it might explain, to a great extent, the lack of benefits in terms of reduction of cardiovascular events with the treatment that has been reported by the studies addressed to this issue [23].
Campos-Rodriguez et al. [41] performed a randomized trial with hypertensive and OSA (AHI ≥ 10 events/h) subjects that included patients who were randomized to therapeutic or subtherapeutic CPAP. By intention to treat, no statistically significant reductions in BP after 4 weeks of treatment were observed in the therapeutic CPAP group, compared with the subtherapeutic group. No significant correlation was observed with respect to CPAP adherence. Along similar lines, the same author [42] evaluated the long-term effect of CPAP treatment in a study with 55 patients. After 24 months of CPAP treatment, the results by intention-to-treat showed a reduction of 2.2 mmHg (95%CI − 4.2 to − 0.1 mmHg; p = 0.03) in diastolic BP, but no changes were observed in other ambulatory blood pressure monitoring (ABPM) parameters. However, a subgroup of patients with a CPAP adherence > 5.3 h/night achieved statistically significant BP reductions for 24-h mean BP, 24-h systolic BP, 24-h diastolic BP, and night-time BP [42].
Furthermore, the results of a trial performed by Duran-Cantolla et al. [43•] with 340 patients with untreated hypertension, diagnosed in the office and randomized to CPAP/sham and 2 weeks of follow-up, showed by intention to treat in the CPAP group a decrease of 1.5 mmHg (95% CI 0.4 to 2.7 mmHg; p = 0.01) in the mean 24-h ambulatory BP, while the mean nocturnal blood pressure decreased by 2.1 mmHg (95% CI 0.5 to 3.6 mmHg; p = 0.01). Taking into account CPAP adherence, greater effects of CPAP treatment on BP were observed and the mean 24-h ambulatory blood pressure of the CPAP group decreased 1.9 mmHg (95% CI 0.5 to 3.4 mmHg; p = 0.01). Moreover, when only those patients presenting hypertension confirmed by an ABPM were selected, a better response to CPAP treatment was also observed. In the meta-analysis of 32 studies performed by Montesi et al. [44], the results showed that CPAP treatment compared with placebo or standard care produces a modest reduction in mean diurnal SBP (− 2.58 mmHg) and DBP (− 2.01 mmHg), while a drop in SBP (− 4.09 mmHg) and DBP (− 1.85 mmHg) was observed in the nocturnal BP. Another meta-analysis conducted by Fava et al. [45] found that CPAP treatment produces a moderate decrease in systolic and diastolic BP, as well as reporting that the reduction in BP was higher in those studies whose subjects presented more severe OSA and remarking that CPAP adherence is an important aspect to consider when seeking to increase reductions in BP [45, 46].
As regards the effect of the associated symptoms of OSA on BP response to CPAP, Barbé et al. [11] evaluated the effect of CPAP treatment on BP in non-sleepy hypertensive patients. The results showed a decrease of 1.89 mmHg (95% CI − 3.90 to 0.11 mmHg; p = 0.0654) in systolic blood pressure and of 2.19 mmHg (95% CI − 3.46 to − 0.93 mmHg; p = 0.0008) in diastolic blood pressure after 12 months of CPAP treatment. The reductions in BP were greater in those patients who used CPAP treatment for more than 5.6 h/night.
All the various studies assessing the effect of CPAP treatment on BP decrease have reported a modest and variable reduction of around 2 mmHg although this decrease cannot be considered negligible at population level regarding its potential benefits. Beyond this, evaluations of different subgroups of patients have shown that reductions in BP after CPAP treatment may be even greater in those patients with uncontrolled BP, known as resistant hypertensive subjects.
The Specific Case of Resistant/Refractory Hypertension
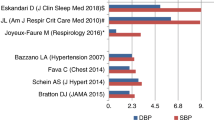
The effect of CPAP treatment on patients with refractory or resistant hypertension, especially when it remains uncontrolled, is usually greater than that observed in patients with controlled hypertension [3,4,5, 47, 48]. As mentioned above, in patients with hypertension, the decrease in BP levels correlates with the number of hours of CPAP used [11, 41,42,43,44,45,46]. Does the same thing happen in patients with RH? Since the first study on the effect of CPAP on blood pressure levels in patients with RH published by Logan et al. in 2003 [49], a significant number of studies have been published in this respect, including observational studies, clinical trials, and systematic meta-analyses/reviews [3, 43, 47, 48, 50,51,52,53,54,55,56,57,58,59,60,61,62,63]. In summary, in those studies in which the correlation between the hours of CPAP use and the decrease in BP (or other outcomes) in patients with RH was analyzed, a positive correlation was observed (Table 1). Thus, in the largest RCT conducted to date, the HIPARCO study [5•], there was a positive linear correlation between CPAP use (in hours/day) and the decrease in 24-h mean BP (r = 0.29; p = 0.006), 24-h SBP (r = 0.25, p = 0.02), and DBP (r = 0.30; p = 0.005). Moreover, a linear regression analysis showed an improvement in BP figures of 1.3 mmHg (95% CI 0.4 to 2.2) for mean BP, of 1.9 mmHg (95% CI 0.6 to 3.3) in SBP, and of 1.0 mmHg (95% CI 0.1 to 1.8) in DBP for each additional hour of CPAP use (Fig. 1). Lozano et al. [50] observed no significant differences in changes in ABPM values between the conventional and the CPAP groups at the 3-month follow-up in those patients with a CPAP use under the median (5.8 h/night), but there were significant reductions in mean daytime diastolic BP (6.12 mmHg, CI 1.45; 10.82, p = 0.004), 24-h systolic BP (9.71 mmHg, CI 0.20; 19.22, p = 0.046), and 24-h diastolic BP (6.98 mmHg, CI 1.86; 12.1, p = 0.009) in those patients who used CPAP for more than 5.8 h/night. Based in part on the results of these two studies, Iftikhar et al. [4] performed a meta-analysis in 2014 on the effect of CPAP on BP levels in RH patients and found a statistically significant correlation between hours of CPAP use and DBP response (pooled R = 0.26; p = 0.0039), based on the HIPARCO study and Lozano’s study. However, it is important to mention that other RCTs have not found a correlation between adherence to CPAP and a better response to hypertension [56, 63] (Table 1).
Correlation between changes in 24-h systolic and diastolic blood pressure and number of hours of CPAP use (with permission of JAMA, reference (5))
One important question to take into account is whether patients with RH have a sufficient degree of CPAP adherence, as their degree of adherence could be reduced because they are usually pauci-symptomatic (with an ESS < 10). In this respect, Campos-Rodriguez et al. [25••] conducted a prospective, multicenter, and observational study in 177 patients with RH and OSA with a median age of 58 years, median BMI of 30.8, and median ESS of 9. Good adherence was defined as a mean use of CPAP for at least 4 h/day. After a follow-up of 57.6 (IQR 42–72) months, the median CPAP use was 5.7 (IQR 3.9–6.6) hours per night, with 74.5% of patients showing a good adherence. The presence of a previous stroke was the only baseline variable associated with non-adequate adherence (HR 4; 95% CI 1.92–8.31). Remarkably, as is the case with almost all patients with OSA, an adequate adherence during the first month of treatment was clearly associated with good long-term adherence (HR 14.4; 4.94–56). These results showed that good long-term adherence to CPAP in patients with RH is possible, even in those with no symptoms or diurnal hypersomnia. So, taking into account the results of the meta-analysis and RCTs, the adherence of CPAP in patients with RH is similar to that seen in other OSA groups, even when the RH patients present less symptoms.
Future Challenges
The threshold of adherence to CPAP associated with a reduction in BP measures is still unknown and needs to be defined, both in symptomatic and asymptomatic patients (in case these are different). It is also essential:
To identify patterns of adherence that may be associated with poor outcomes, beyond the total number of hours of CPAP use (e.g., patients who use CPAP only on working days, only in the first hours of the sleep, on alternate days).
To identify risk factors for non-adherence and evaluate whether there are any specific subgroups of hypertensive patients who are more prone to poor adherence with CPAP. Personalized medicine could be of great help in this respect.
To investigate what strategies could be implemented to optimize adherence in clinical settings, including educational programs, telemedicine, and remote monitoring.
To develop an easy, reliable, and accurate method to measure adherence to antihypertensive medication.
To develop combinations of drugs that facilitate adherence to antihypertensive medication therapy.
Conclusions
Although taking a drug would seem to be easier than using CPAP treatment, it appears, based on the information currently available that the adherence with drug treatment in hypertensive subjects is not better than that achieved with CPAP treatment in OSA patients with hypertension.
A significant adherence in terms of the number of hours of CPAP use per night is crucial because, over and above any improvement in OSA symptoms, a decrease in BP, and subsequent decrease in cardiovascular events, therefore also reduces cardiovascular risk. Furthermore, the key role played by CPAP adherence in achieving these cardiovascular benefits is found in different profiles of patients with OSA (sleepy and non-sleepy, or varying severity of hypertension).
Unfortunately, the threshold of adherence to CPAP associated with a reduction in BP is still unknown, but it seems that at least 5–6 h/night, and preferably all the sleeping time, are required to obtain a maximal benefit on BP levels. Nevertheless, despite the improvements made in CPAP devices and interfaces, adherence rates to this therapy are still poor, and they do not seem to have improved over the last two decades.
In patients with resistant hypertension, there is a correlation between the number of hours of CPAP treatment and the decrease in blood pressure levels, as well as a higher percentage of patients who recover their dipper pattern during the night. Moreover, despite the lack of sleep-related symptoms, good long-term adherence to CPAP in resistant hypertension is possible, with numbers similar to those seen in the general population of OSA patients. Further studies are needed to corroborate some important aspects about adherence to CPAP and antihypertensive drugs in resistant hypertension [64•].
Clinical Remarks (Bullets)
-
1.
The non-adherence to the antihypertensive treatment reported in studies is high, and it is associated with an increased risk of cardiovascular events. To improve this adherence, it is important to identify those modifiable factors that could be related to poor adherence.
-
2.
CPAP treatment produces a modest and variable decrease in blood pressure in patients with hypertension and OSA. However, adherence to CPAP treatment (≥ 4 h/night) seems to be an important factor as regards a better response in BP after CPAP treatment.
-
3.
A CPAP adherence of at least 5–6 h per night is required to achieve a decrease in BP measures.
-
4.
Given that adherence to CPAP is variable, and in many cases suboptimal, patients should be closely followed in order to detect poor adherence and instigate suitable measures to fix it.
-
5.
In patients with resistant/refractory hypertension and obstructive sleep apnea, good adherence to CPAP seems to be crucial for achieving a good response in blood pressure levels.
-
6.
Although patients with OSA and resistant hypertension usually have limited sleep-related symptoms, their long-term adherence with CPAP treatment is acceptable.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
• Javaheri S, Barbe F, Campos-Rodriguez F, Dempsey JA, Khayat R, Javaheri S, et al. Sleep apnea: types, mechanisms, and clinical cardiovascular consequences. J Am Coll Cardiol. 2017;69:841–58. Meta de hypertension. A comprehensive review of sleep apnea and the mechanisms linking this disease with cardiovascular disease, as well as the most important clinical results of all the recent studies on this topic.
•• Hu X, Fan J, Chen S, Yin Y, Zrenner B. The role of continuous positive airway pressure in blood pressure control for patients with obstructive sleep apnea and hypertension: a meta-analysis of randomized controlled trials. J Clin Hypertension. 2015;17:215–22. This is the last meta-analysis of randomized controlled trials on the effect of CPAP treatment on blood pressure levels.
•• Lei Q, Lv Y, Li K, Ma L, Du G, Xiang Y, et al. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: a systematic review and meta-analysis of six randomized controlled trials. J Bras Pneumol. 2017;43:373–9. This is the latest meta-analysis on the effect of CPAP treatment on blood pressure levels in patients with resistant hypertension.
Iftikhar IH, Valentine CW, Bittencourt LRA, Cohen DL, Fedson AC, Gíslason T, et al. Effects of continuous positive airway pressure on blood pressure in patients with resistant hypertension and obstructive sleep apnea: a meta-analysis. J Hypertens. 2014;32:2341–50.
• Martínez-García M-A, Capote F, Campos-Rodríguez F, Lloberes P, Díaz de Atauri MJ, Somoza M, et al. Effect of CPAP on blood pressure in patients with obstructive sleep apnea and resistant hypertension: the HIPARCO randomized clinical trial. JAMA. 2013;310:2407–15. The largest randomized clinical trial to date on the effect of CPAP on blood pressure levels and circadian pattern in patients with resistant hypertension.
Patil SP, Ayappa IA, Caples SM, Kimoff RJ, Patel SR, Harrod CG. Treatment of adult obstructive sleep apnea with positive airway pressure: an American Academy of Sleep Medicine systematic review, meta-analysis, and GRADE assessment. J Clin Sleep Med. 2019;15:301–34.
• Bakker JP, Weaver TE, Parthasarathy S, Aloia MS. Adherence to CPAP: what should we be aiming for, and how can we get there? Chest. 2019;155:1272–87. This is an excellent and updated review of the definitions and strategies for optimizing adherence to CPAP.
Weaver TE, Grunstein RR. Adherence to continuous positive airway pressure therapy: the challenge to effective treatment. Proc Am Thorac Soc. 2008;5:173–8.
Antic NA, Catcheside P, Buchan C, Hensley M, Naughton MT, Rowland S, et al. The effect of CPAP in normalizing daytime sleepiness, quality of life, and neurocognitive function in patients with moderate to severe OSA. Sleep. 2011;34:111–9.
Weaver TE, Maislin G, Dinges DF, Bloxham T, George CFP, Greenberg H, et al. Relationship between hours of CPAP use and achieving normal levels of sleepiness and daily functioning. Sleep. 2007;30:711–9.
Barbé F, Durán-Cantolla J, Capote F, de la Peña M, Chiner E, Masa JF, et al. Long-term effect of continuous positive airway pressure in hypertensive patients with sleep apnea. Am J Respir Crit Care Med. 2010;181:718–26.
Campos-Rodriguez F, Martinez-Garcia MA, Reyes-Nuñez N, Caballero-Martinez I, Almeida-Gonzalez CV, Catalan-Serra P, et al. Long-term continuous positive airway pressure compliance in females with obstructive sleep apnoea. Eur Respir J. 2013;42:1255–62.
Baratta F, Pastori D, Bucci T, Fabiani M, Fabiani V, Brunori M, et al. Long-term prediction of adherence to continuous positive air pressure therapy for the treatment of moderate/severe obstructive sleep apnea syndrome. Sleep Med. 2018;43:66–70.
Alzoubaidi M, Mokhlesi B. Obstructive sleep apnea during rapid eye movement sleep: clinical relevance and therapeutic implications. Curr Opin Pulm Med. 2016;22:545–54.
Jacobsen AR, Eriksen F, Hansen RW, Erlandsen M, Thorup L, Damgård MB, et al. Determinants for adherence to continuous positive airway pressure therapy in obstructive sleep apnea. In: Heiser C, editor. PLoS One. 2017;12:e0189614.
Kohler M, Smith D, Tippett V, Stradling JR. Predictors of long-term compliance with continuous positive airway pressure. Thorax. 2010;65:829–32.
Pelletier-Fleury N, Rakotonanahary D, Fleury B. The age and other factors in the evaluation of compliance with nasal continuous positive airway pressure for obstructive sleep apnea syndrome. A Cox’s proportional hazard analysis. Sleep Med. 2001;2:225–32.
Campos-Rodriguez F, Martinez-Alonso M, Sanchez-de-la-Torre M, Barbe F. Long-term adherence to continuous positive airway pressure therapy in non-sleepy sleep apnea patients. Sleep Med. 2016;17:1–6.
Chai-Coetzer CL, Luo Y-M, Antic NA, Zhang X-L, Chen B-Y, He Q-Y, et al. Predictors of long-term adherence to continuous positive airway pressure therapy in patients with obstructive sleep apnea and cardiovascular disease in the SAVE study. Sleep. 2013;36:1929–37.
Furukawa T, Suzuki M, Ochiai M, Kawashima H, Yokoyama N, Isshiki T. Long-term adherence to nasal continuous positive airway pressure therapy by hypertensive patients with preexisting sleep apnea. J Cardiol. 2014;63:281–5.
Meurice JC. Predictive factors of long-term compliance with nasal continuous positive airway pressure treatment in sleep apnea syndrome. Chest. 1994;105:429.
Sin DD, Mayers I, Man GCW, Pawluk L. Long-term compliance rates to continuous positive airway pressure in obstructive sleep apnea: a population-based study. Chest. 2002;121:430–5.
McEvoy RD, Antic NA, Heeley E, Luo Y, Ou Q, Zhang X, et al. CPAP for prevention of cardiovascular events in obstructive sleep apnea. N Engl J Med. 2016;375:919–31.
Gagnadoux F, Le Vaillant M, Paris A, Pigeanne T, Chollet S, Masson P, et al. Adherence to positive airway pressure in non-sleepy patients with obstructive sleep apnoea. Eur Respir J. 2013;42:863–6.
•• Campos-Rodriguez F, Navarro-Soriano C, Reyes-Nuñez N, Torres G, Caballero-Eraso C, Lloberes P, et al. Good long-term adherence to continuous positive airway pressure therapy in patients with resistant hypertension and sleep apnea. J Sleep Res. 2019;28:e12805. This is the first long-term analysis of CPAP adherence conducted in patients with OSA and resistant hypertension. The study concluded that good adherence can be achieved in this population, even though they were mostly non-sleepy.
Rotenberg BW, Murariu D, Pang KP. Trends in CPAP adherence over twenty years of data collection: a flattened curve. J Otolaryngol Head Neck Surg. 2016;45:43.
•• Williams B, Mancia G, Spiering W, Agabiti Rosei E, Azizi M, Burnier M, et al. ESC/ESH guidelines for the management of arterial hypertension: the Task Force for the management of arterial hypertension of the European Society of Cardiology (ESC) and the European Society of Hypertension. J Hypertens. 2018;36(10):1953–2041. This is the latest European guideline to include all the important aspects of and changes in the management of the hypertension.
Abegaz TM, Shehab A, Gebreyohannes EA, Bhagavathula AS, Elnour AA. Nonadherence to antihypertensive drugs: a systematic review and meta-analysis. Medicine (Baltimore). 2017;96(4):e5641.
Yang Q, Chang A, Ritchey MD, Loustalot F. Antihypertensive medication adherence and risk of cardiovascular disease among older adults: a population-based cohort study. J Am Heart Assoc. 2017;6(6).
Krousel-Wood M, Joyce C, Holt E, Muntner P, Webber LS, Morisky DE, et al. Predictors of decline in medication adherence: results from the cohort study of medication adherence among older adults. Hypertension. 2011;58(5):804–10.
Burnier M, Brent M, Egan. Adherence in hypertension: a review of prevalence, risk factors, impact, and management. Circ Res. 2019;124:1124–40.
Chapman RH, Petrilla AA, Benner JS, Schwartz JS, Tang SS. Predictors of adherence to concomitant antihypertensive and lipid-lowering medications in older adults: a retrospective, cohort study. Drugs Aging. 2008;25(10):885–92.
Tedla YG, Bautista LE. Drug side effect symptoms and adherence to antihypertensive medication. Am J Hypertens. 2016;29(6):772–9.
Gupta AK, Arshad S, Poulter NR. Compliance, safety, and effectiveness of fixed-dose combinations of antihypertensive agents: a meta-analysis. Hypertension. 2010;55:399–407.
Bangalore S, Kamalakkannan G, Parkar S, Messerli FH. Fixed-dose combinations improve medication compliance: a meta-analysis. Am J Med. 2007;120(8):713–9.
Hameed MA, Dasgupta I. Medication adherence and treatment-resistant hypertension: a review. Drugs Context. 2019;8:1–11.
Gupta P, Patel P, Horne, Buchanan H, Williams B, Tomaszewski M. How to screen for non-adherence to antihypertensive therapy. Curr Hypertens Rep. 2016;18(12):89.
Durand H, Hayes P, Morrissey EC, Newell J, Casey M, Murphy AW, et al. Medication adherence among patients with apparent treatment-resistant hypertension: systematic review and meta-analysis. J Hypertens. 2017;35(12):2346–57.
Peacock E, Krousel-Wood M. Adherence to antihypertensive therapy. Med Clin North Am 2017. 2017;101(1):229–45.
Corrao G, Parodi A, Nicotra F, Zambon A, Merlino L, Cesana G, et al. Better compliance to antihypertensive medications reduces cardiovascular risk. J Hypertens. 2011;29(3):610–8.
Campos-Rodriguez F. Effect of continuous positive airway pressure on ambulatory BP in patients with sleep apnea and hypertension. A Placebo-Controlled Trial Chest. 2006;129(6):1459–67.
Campos-Rodriguez F, Perez-Ronchel J, Grilo-Reina A, Lima-Alvarez J, Benitez MA, Almeida-Gonzalez C. Long-term effect of continuous positive airway pressure on BP in patients with hypertension and sleep apnea. Chest. 2007;132(6):1847–52.
• Duran-Cantolla J, Aizpuru F, Montserrat JM, Ballester E, Teran-Santos J, Aguirregomoscorta JI, et al. Continuous positive airway pressure as treatment for systemic hypertension in people with obstructive sleep apnoea: randomised controlled trial. BMJ. 2010;341:c5991. An important randomized control trial for evaluating the effect on BP of CPAP treatment; it includes a large sample size of non-treated hypertensive patients with obstructive sleep apnea.
Montesi SB, Edwards BA, Ph D, Malhotra A, Bakker JP, Ph D. The effect of continuous positive airway pressure treatment on blood pressure: a systematic review and meta-analysis of randomized controlled trials. J Clin Sleep Med. 2012;8(5):587–96.
Fava C, Dorigoni S, Dalle Vedove F, Danese PharmD E, Montagnana M, Cesare Guidi G, et al. Effect of CPAP on blood pressure in patients with OSA/hypopnea: a systematic review and meta-analysis. Chest. 2014;145:762–71.
Bazzano LA, Khan Z, Reynolds K, He J. Effect of nocturnal nasal continuous positive airway pressure on blood pressure in obstructive sleep apnea. Hypertension. 2007;50(2):417–23.
Feldstein CA. Blood pressure effects of CPAP in nonresistant and resistant hypertension associated with OSA: a systematic review of randomized clinical trials. Clin Exp Hypertens. 2016;38:337–46.
Varounis C, Katsi V, Kallikazaros IE, Tousoulis D, Stefanadis C, Parissis J, et al. Effect of CPAP on blood pressure in patients with obstructive sleep apnea and resistant hypertension: a systematic review and meta-analysis. Int J Cardiol. 2014;15(175):195–8.
Logan AG, Tkacova R, Perlikowski SM, Leung RS, Tisler A, Floras JS, et al. Refractory hypertension and sleep apnoea: effect of CPAP on blood pressure and baroreflex. Eur Respir J. 2003;21:241–7.
Lozano L, Tovar JL, Sampol G, Romero O, Jurado MJ, Segarra A, et al. Continuous positive airway pressure treatment in sleep apnea patients with resistant hypertension: a randomized, controlled trial. J Hypertens. 2010;28:2161–8.
de Oliveira AC, Martinez D, Massierer D, Gus M, Gonçalves SC, Ghizzoni F, et al. The antihypertensive effect of positive airway pressure on resistant hypertension of patients with obstructive sleep apnea: a randomized, double-blind, clinical trial. Am J Respir Crit Care Med. 2014;190:345–7.
Lloberes P, Sampol G, Espinel E, Segarra A, Ramon MA, Romero O, et al. A randomized controlled study of CPAP effect on plasma aldosterone concentration in patients with resistant hypertension and obstructive sleep apnea. J Hypertens. 2014;32:1650–7.
• Navarro-Soriano C, Martínez-García MA, Torres G, Barbé F, Caballero-Eraso C, Lloberes P, et al. Effect of continuous positive airway pressure in patients with true refractory hypertension and sleep apnea: a post-hoc intention-to-treat analysis of the HIPARCO randomized clinical trial. J Hypertens. 2019;37:1269–75. The only study to date on the effect of CPAP on blood pressure levels in patients with true refractory hypertension, demonstrating that this effect is even better than that seen in resistant hypertension.
Joyeux-Faure M, Baguet JP, Barone-Rochette G, Faure P, Sosner P, Mounier-Vehier C, et al. Continuous positive airway pressure reduces night-time blood pressure and heart rate in patients with obstructive sleep apnea and resistant hypertension: the RHOOSAS randomized controlled trial. Front Neurol. 2018;9:318.
de Souza F, Muxfeldt ES, Margallo V, Cortez AF, Cavalcanti AH, Salles GF. Effects of continuous positive airway pressure treatment on aldosterone excretion in patients with obstructive sleep apnoea and resistant hypertension: a randomized controlled trial. J Hypertens. 2017;35:837–44.
Pedrosa RP, Drager LF, de Paula LKG, Amaro ACS, Bortolotto LA, Lorenzi-Filho G. Effects of OSA treatment on BP in patients with resistant hypertension. A randomized trial. Chest. 2013;144:1487–94.
Dernaika TA, Kinasewitz GT, Tawk MM. Effects of nocturnal continuous positive airway pressure therapy in patients with resistant hypertension and obstructive sleep apnea. J Clin Sleep Med. 2009;5:103–7.
Martínez-García MA, Gómez-Aldaraví R, Soler-Cataluña JJ, Martínez TG, Bernácer-Alpera B, Román-Sánchez P. Positive effect of CPAP treatment on the control of difficult-to-treat hypertension. Eur Respir J. 2007;29:951–7.
Walia HK, Griffith SD, Foldvary-Schaefer N, Thomas G, Bravo EL, Moul DE, et al. Longitudinal effect of CPAP on BP in resistant and nonresistant hypertension in a large clinic-based cohort. Chest. 2016;149:747–55.
Frenţ ŞM, Tudorache VM, Ardelean C, Mihăicuţă S. Long-term effects of nocturnal continuous positive airway pressure therapy in patients with resistant hypertension and obstructive sleep apnea. Pneumologia. 2014;204:207–11.
Liu L, Cao Q, Guo Z, Dai Q. Continuous positive airway pressure in patients with obstructive sleep apnea and resistant hypertension: a meta-analysis of randomized controlled trials. J Clin Hypertens (Greenwich). 2016;18:153–8.
Navarro-Soriano C, Martínez-García MA, Torres G, Barbé F, Caballero-Eraso C, Lloberes P, et al. Factors associated with the changes from a resistant to a refractory phenotype in hypertensive patients: a Pragmatic Longitudinal Study. Hypertens Res. 2019;42:1708–15. https://doi.org/10.1038/s41440-019-0285-8.
Muxfeldt ES, Margallo V, Costa LMS, Guimaraes G, Cavalcante AH, Azevedo JCM, et al. Effects of continuous positive airway pressure treatment on clinic and ambulatory blood pressures in patients with obstructive sleep apnea and resistant hypertension. A randomized controlled trial. Hypertension. 2015;65:736–42.
• Sapiña-Beltrán E, Torres G, Martínez-Alonso M, Sánchez-de-la-Torre M, Franch M, Bravo C, et al. Rationale and methodology of the SARAH trial: long-term cardiovascular outcomes in patients with resistant hypertension and obstructive sleep apnea. Arch Bronconeumol. 2018;54:518–23. Recent study on the methodology of the SARAH study, which will be the largest study in the world on the long-term impact of OSA and CPAP treatment on patients with resistant hypertension in terms of cardiovascular risk and blood pressure control.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest relevant to this manuscript.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Sleep and Hypertension
All the authors had access to the data and played a role in writing the manuscript.
Rights and permissions
About this article
Cite this article
Posadas, T., Campos-Rodriguez, F., Sapiña-Beltrán, E. et al. Obstructive Sleep Apnea and Arterial Hypertension: Implications of Treatment Adherence. Curr Hypertens Rep 22, 12 (2020). https://doi.org/10.1007/s11906-020-1015-y
Published:
DOI: https://doi.org/10.1007/s11906-020-1015-y