Abstract
Eastern Europe and Central Asia represent one of the few regions globally where there is a continued increase in the incidence of HIV infection. For example, in Eastern Europe the rate of diagnosed cases of HIV infection per 100 000 population has increased from 11.7 in 2004 to 22.5 in 2011. Initially propelled by injection drug use, heterosexual transmission has now become a major driver of new infections in the region. Nonetheless substance use remains an important factor, with its control limited by challenges in scaling up harm reduction efforts. While most countries have implemented opioid substitution therapy programs, their scale remains very limited. Similarly, coverage of needles syringe programs across the region is variable. Complicating the control of HIV has been the emergence of non-injection drugs and inadequate access to antiretroviral therapy. In addition, structural barriers and stigma toward HIV infected people may contribute to the high proportion of late presentations for HIV care. Finally in the wake of the HIV epidemic, high rates of hepatitis C infection and tuberculosis have been noted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There is increasing evidence that HIV incidence is in decline among general populations worldwide [1]. Eastern Europe and Central Asia, however, are notable exceptions as will be detailed in this article. This is highlighted by a 13 % increase in new HIV infections in Eastern Europe and Central Asia between 2006 to 2012 [2]. In addition, while other regions have seen declines in death rates, Eastern Europe and Central Asia have experienced a 2.5 times increase between 2001 and 2012 [2].
A number of factors place the emerging democracies of Eastern Europe and Central Asia at high risk for HIV infection [3]. First, the political transition in the early 1990s led to dramatic declines in income, a significant increase in unemployment, and widened income inequities. These economic dislocations also led to an expansion of informal and criminal economies [4]. Second, the highly structured public health system rooted in the Soviet tradition has been unable to effectively transition to meet post-Soviet challenges. The Soviet model was based on a highly centralized and hierarchical sanitary-epidemiologic system, which was characterized by a large labor force and minimal emphasis on technology [3]. Thus, while surveillance was formally enforced, health promotion efforts remained rudimentary [5]. However, there was minimal attention to feedback and quality improvement. These changes have been confounded by dramatic increases in injection drug use (IDU), the enormous production of opiates from the Central Asian state of Afghanistan [6] and consequent drug trafficking.
Initially the HIV epidemic in the region was driven by people who inject drugs (PWIDs). However, while the driving patterns vary substantially between countries in Eastern Europe [7], surveillance data [8] and recent analysis of the modes of transmission [2] suggest that PWID currently account for up to one third of new HIV infections in Eastern Europe. In many countries of the region heterosexual transmission has now become the leading mode of HIV transmission.
Nonetheless, substance use remains an important factor. While opiod substitution therapy (OST) has been shown to reduce HIV incidence in drug users [9], it is variably and inadequately accessible. Its use is proscribed in Russia and access to these services is inadequate in other countries of the region. This is in spite of the fact that in 2005, WHO added methadone and buprenorphine to the WHO model list of essential medicines for opioid addiction treatment. Implementation of other interventions to reduce transmission such as needle exchange has also been limited. Antiretroviral therapy (ART) in the region became widely (but not universally) available after 2002. However, as noted below, access to therapy may be reduced in the setting of substance use.
Complicating control of HIV has been the emergence and acceleration of different non-injection drugs in some regions of the world. Certain non-injection drugs, particularly stimulants, lead to an elevated rate of HIV transmission due to their association with high-risk sexual behaviors [10, 11•]. The use of new compounds is increasing in many countries including Eastern Europe and Central Asia, which has seen the arrival of synthetic heroin substitutes and home-made ‘street’ drugs such as krokodil and ‘China white’ [12] locally produced opiate analogues (so called ‘designer drugs’) [13–15]. Some forms of non-injection substance abuse, particularly amphetamine-types [16–18] and krokodil has increased in Russia, Kazakhstan, Estonia, and Ukraine due to changes in heroin availability, purity, and price, all of which are associated with heroin ‘droughts’, police interdiction, legislative changes targeting poppy imports, and rising poverty levels in Russia since the 2008 global economic downturn [19]. These new compounds increase the risk of HIV, overdose and mortality rates among PWID. The epidemic in the region is characterized by a number of varying patterns. The subsequent sections address two important regions, that of Eastern Europe (assessing the countries of Belarus, Estonia, Latvia, Lithuania, Moldova, Russia, and Ukraine) and Central Asia (assessing the countries of Kazakhstan, Kyrgyzstan, Uzbekistan, Tajikistan, Turkmenistan).
Eastern Europe
Epidemiology
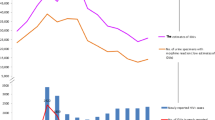
Across Eastern Europe the rate of diagnosed cases of HIV infection per 100 000 population has increased from 11.7 in 2004 to 22.5 in 2011 [8]. The highest rates of new HIV infections in 2011 occurred in Russia (47/100 000), Ukraine (38/100 000), and Estonia (27/100 000) [8]. Notably, the epidemic continues to rise in the most populous countries of the region. For example, in the first six months of 2012, Russia reported a 12 % increase in the number of new HIV cases compared to the same period in 2011 [20]. With the rise in incidence, prevalence is also rising from 2.6/100 000 in 2004 to 5.7/100 000 in 2011 [8]. The highest AIDS prevalence in 2011 was reported in Moldova (11.4/100 000), Ukraine (9.0/100 000), and Belarus (5.4/100 000). According to WHO surveillance data, the most common HIV associated conditions diagnosed in Eastern Europe in 2011 were pulmonary tuberculosis (36 %), wasting syndrome (25 %), and esophageal candidiasis (23 %) [8].
Sex Work
A recent systematic review of the burden of HIV among female sex workers (FSW) documented scarcity of the data characterizing HIV risk among female sex workers in parallel to the disproportionately high burden of disease in this population group [21]. According to results of a study conducted among FSWs in Russia (Kazan, Krasnoyarsk, and Tomsk) in 2011, 3.9 % of women were HIV infected. Those with a history of IDU, and/or with client-perpetrated physical and sexual abuse contributing significantly to the HIV risk [22]. Significantly higher HIV prevalence is reported among FSWs in Moldova (6.9 % among women from Chisinau, and 24.7 % in Balti) with injection drugs again being associated with HIV seropositivity [23].
Prisons
HIV infection rates above 10 % have been documented in correctional institutions in the region [24]. Recent nationwide studies in Estonia and Ukraine have documented a high HIV prevalence among inmates – 15.6 % in Estonia (Kivimets K, Ministry of Justice, Estonia, personal communication) and 19.4 % in Ukraine (among individuals being released from prisons within six months) [25]. Data assessing the implementation and effectiveness of HIV prevention and harm reduction programs introduced in prisons in these countries are limited or missing.
Migration
Migration adds another significant layer of complexity for both HIV transmission and provision of prevention and care. The most common push factor for migration is seeking economic improvement. Apart from economic migration, political and social factors may force people to migrate. Particularly relevant to HIV epidemiology are people who are persecuted because of their sexual identity (e.g., men who have sex with men [MSM]). Eastern Europe experiences migration both within and from the region.
Low socioeconomic status, lack of access to services, separation from family, and limited risk awareness all contribute to migrants’ HIV vulnerability. In St. Petersburg, 30 % of male migrants reported multiple female partners in the past 3 months. Condom use was low, ranging from 35 % with permanent to 52 % with casual partners. Central Asian migrants had very low AIDS knowledge, low levels of substance use, moderate sexual risk behaviors, high levels of depression, and poor social supports [26, 27].
Co-Infections
Tuberculosis (TB)
TB is a common opportunistic infection in Eastern Europe. In 2011, TB notification rates per 100 000 varied from 25.4 in Estonia to 150.7 in Moldova, and the estimated proportion of new cases of TB (including relapses) who were HIV positive varied from 0.9 % in Lithuania to 20 % in Ukraine [28].
In the WHO European area, the highest rates of resistance to at least one anti-TB drug were reported in Estonia, Latvia, and Lithuania (34-41 %). Multi-drug resistant (MDR) TB was also most frequently observed in the same three Baltic countries, occurring in 11–23 % of all new cases and 29-58 % among previously treated cases [28].
A recent study comparing data on 55 patients with HIV plus MDR TB in Eastern Europe (between 2004 and 2006) to 89 patients whose Mycobacterium tuberculosis isolates were susceptible to isoniazid and rifampicin documented that patients with HIV and MDR TB were young and predominantly male with high rates of IDU, imprisonment, and HCV co-infection. The majority (84 %) of patients with MDR TB had no history of previous TB drug exposure suggesting that most MDR TB infections resulted from transmission of drug resistant Mycobacterium tuberculosis. The use of non-standardized TB treatment was common, and the use of antiretroviral therapy was infrequent. Mortality was high among HIV infected patients with MDR and they were more likely to die than the other group (65.5 % vs. 27.0 %, p < 0.0001) [29]. Extensively drug resistant TB (XDR TB) has also been reported from the region. A recent study of patients with locally confirmed pulmonary MDR found XDR rates varying from 6.5 % in Estonia to 11.3 % in Russia [30].
HCV
Eastern Europe has high rates of HIV/HCV co-infection, which keep rising – currently up to 80 % among people living with HIV and seeking treatment in Estonia and Ukraine, and over 90 % in samples tested in Russia [31].
Additionally, HCV/HIV infected persons in Eastern Europe do not have access to HCV treatment with the new, oral, direct-acting antivirals (which offer a potential HCV cure). Even access to pegylated interferon is a challenge as the average price for a 48-week treatment course in the region is US$14,500 [32]. According to the Eurasian Harm Reduction Network (EHRN) mapping experiment, the quality of treatment offered is substandard due to the lack of treatment guidelines prioritizing up-to-date, internationally recognized, HCV diagnostics and treatment protocols; HCV testing access and uptake are extremely low; and awareness of HCV, including risk factors and treatment, is limited among both healthcare providers and patients [32].
HIV Prevention and Care
A systematic review of data for provision and coverage of HIV prevention, treatment, and care services for PWIDs was published in 2010 [33] documenting that needle and syringe programs (NSPs) were present in nearly all countries in Eastern Europe and CA, and only five countries did not have OST programs (Russia, Armenia, Tajikistan, Turkmenistan, Uzbekistan).
Programs to provide PWID with access to NSPs are generally considered to be among the most effective means of reducing HIV and hepatitis C (HCV) transmission among PWID [1, 2]. However, coverage of NSPs is variable across Eastern Europe, and generally low (below 35 needle–syringes distributed per PWID per year); with the lowest coverage reported from Russia (four needle–syringes distributed per PWID per year) [32], although in Russia and in other countries, PWIDs can purchase needles in pharmacies. High NSP coverage (over 100 needle–syringes distributed per PWID per year) has been described in Estonia, Moldova [32, 34], and Lithuania [34, 35].
HIV Care
Limited data are available on health and health/risk behavior characteristics of people accessing HIV care in Eastern European countries. Given the large number of PWID’s with HIV, studies conducted in Russia [36] and Estonia [37] document that people accessing HIV treatment services frequently have an IDU background (Russia, St. Petersburg: PWID history 80.3 % of men, 48.7 % of women (15); Estonia: 64 % of HIV-positive individuals receiving care [37]. A cohort study in Europe (Collaboration of Observational HIV Epidemiological Research Europe, COHERE) analyzing the occurrence and factors associated with late presentation for HIV care (defined as HIV diagnosis with a CD4+ cell count <350/mm3 or an AIDS diagnosis within 6 months of HIV diagnosis) documented that 47.2 % and 28.7 % of patients in HIV care in Eastern European sites were later presenters or late presenters with advanced disease, respectively. Importantly, there was an increase in late presentation among male and female PWIDs from Eastern Europe between 2000 and 2011 [38]. Officially, HIV care and ART are universally available for those in need in the region. However, a qualitative study by Sharang et al. delineating systemic barriers to accessing ART among PWID in Yekaterinburg, Russia identified three core themes: ‘labyrinthine bureaucracy’ governing access to ART; a ‘system Catch 22’ created by an expectation that access to ART was conditional on treated drug use in a setting of limited drug treatment opportunity; and ‘system verticalization’, where a lack of integration across HIV, tuberculosis, and drug treatment compromised access to ART. The authors concluded that systemic factors play a key role in shaping access to ART [39].
Rapid scaling-up of antiretrovirals (ARV) has been reported from Estonia, where data collected from repeated cross-sectional respondent-driven samples of PWID have documented increased ARV coverage among PWID (from 0 % in 2005 to 62 % in 2013) [40].
In addition to the structural level barriers, stigma toward HIV infected people operating at multiple levels has been documented [41]. In Russia (St. Petersburg), 25 % of people living with HIV (PLH) had been refused general health care, 11 % refused employment, 7 % fired, and 6 % forced from family homes [42]. Negative attitudes toward treating HIV-positive and marginalized patients were found to be common among medical students in Kazan, Russia [43]. Other related barriers that have been described include criminalization of drug use, limited knowledge of HIV/AIDS, services and entitlements, shortages of commodities and personnel, and organizational, economic, and geographical barriers [41]. The described system-level barriers and stigmatization may partly explain the high proportion of late presentations for HIV care and the low acceptance and adherence to ARV. For example, in Ukraine, in 2008-2010, only 54 % of 108,116 people who tested HIV positive enrolled in care [44]; in Russia (St. Petersburg) of those PLH who were offered ARV (n = 266), 16 % refused ARV regimens [43].
Opioid Substitution Therapy (OST)
Most countries have implemented OST programs (with Russia and four other nations as notable exceptions); however, overall, the scale of programs was very limited in the region with only one person receiving OST for every 100 PWIDs [33]. One of the success stories has been the launching of opioid substitution therapy in Ukraine in 2004 (initially limited to buprenorphine use followed by methadone since 2007) [45]. The number of people in Ukraine receiving OST has increased threefold from 2504 in 2009 to 7090 receiving treatment in 143 facilities by mid-2012. However, even given its success, there is less than 3 % OST coverage among PWID in Ukraine. Similar coverage (2 %) is seen in Estonia [34].
Central Asia
Epidemiology of HIV
While HIV incidence has declined globally, UNAIDS recently identified nine countries where HIV incidence increased 25 % or more between 2001 and 2011 [1, 46]; five of these nine countries are in Central Asia (i.e., Kazakhstan, Kyrgyzstan, Uzbekistan, Tajikistan, Turkmenistan) [46]. As with Eastern Europe, new HIV infections in Central Asia have risen primarily among PWID, sex partners of men who inject drugs, men who have sex with men (MSM), female sex workers, and migrant workers [47–50]. More than half of the recently-diagnosed HIV infections in Central Asia have occurred in the PWID population [47, 51, 52]. While it is estimated that 1 % of adults inject drugs in Central Asia, the number exceeds 10 % in areas along major drug trafficking routes, representing one of the highest rates of IDU in the world, which illuminates the rise of the HIV epidemic in this region [47, 51, 52].
Recent data from national integrated bio-behavioral surveys (IBBS) among PWID show that HIV prevalence ranges from 0 to 13.1 % in different parts in Kazakhstan [53], 3-32.4 % in Kyrgyzstan [54], 0.27-.4 % in Tajikistan [55], and 0-25 % in Uzbekistan [56]. At the end of 2012, data showed that 33 % of Uzbekistan’s cumulative registered cases and 72 % of those in Kazakhstan were transmitted through the use of shared injection equipment [57, 58]. These statistics, however, may not be fully accurate given that only registered PWID in these countries are included in the surveys.
In the past three years, there has also been a steady rise in the incidence of sexual transmission of HIV as PWID in Central Asia have spread the virus to their sexual partners [59]. Official data from Kazakhstan in 2011 showed heterosexual activity as the primary mode of transmission, which represented 50.7 % of new cases and surpassed IDU, which represented 47 % of new cases [53]. The rise in sexual HIV transmission, particularly to the sexual partners of PWID, has been reported in each of the Central Asian countries except Turkmenistan [59, 60••]. Although data on HIV and drug use from Turkmenistan remain very scarce; it is estimated that the HIV epidemic is similar to other Central Asia countries because of shared historical and geographical contexts [59]. These statistics, however, may be underreported by the surveillance data.
Another key population affected by HIV in Central Asia is MSM. National reports estimate HIV prevalence among MSM to range from 1 % to 2 % in Kazakhstan, Kyrgyzstan, Tajikistan, and Uzbekistan in 2011 [61] and as high as 6.8 % in Tashkent, Uzbekistan [47, 51]. In a study among MSM in Kazakhstan, HIV prevalence was 20.2 % [Berry et al., Association between human rights violations and HIV risk behaviors among men who have sex with men (MSM) in Almaty, unpublished]. In a meta-analysis paper, lifetime prevalence of same sex sexual activity among men in Central Asia and Eastern Europe countries was found to range between 6-27 % [62]. The paper does not provide these data by country.
Sex work has also emerged as a major driver of HIV among females who inject drugs (FWID) in Central Asia [10, 11•, 63, 64••] where the dual risks of injection and sex trading influence rates of HIV transmission [65]. Although it is estimated that a third of FWID engage in sex trading globally, data about this population is limited in Central Asia. More research and surveillance on this key population group is needed. MSM and FWID, in general and especially those who trade sex, face social and structural barriers that impede their access to HIV prevention such as a lack of access to health care and HIV services, poverty, criminalization of MSM, sex work, imprisonment, and the confiscation of condoms as evidence of commercial sex work [10, 59, 63]. This situation underscores the urgent need to address sexual and drug risks and structural barriers to HIV prevention services and policies for people who use drugs, specifically for FWID who trade sex.
While some evidence is growing on IDU among youth in Central Asia, this population has not yet been included as a key risk population in the region. A recent review of studies conducted in Ukraine indicated that youth who were homeless and orphaned were more likely to be HIV-positive and engaging in IDU, compared to youth with stable housing who resided with their parents [46, 49, 66]. A similar situation may be occurring in Central Asia. Age-appropriate HIV prevention services are needed for young people who use drugs, as are additional studies to inform prevention development.
Co-infections
IDU, increased use of synthetic heroin substitutes, and mixing different drugs and alcohol has led to elevated risks for morbidity and mortality from HIV, HCV, TB, and overdose. The prevalence of HCV infection is exceptionally high and the rates of MDR-TB in PWID in Central Asia are among the highest in the world [67].
TB
Overall tuberculosis incidence rates in Kazakhstan, Uzbekistan, Tajikistan, and Kyrgyzstan are extremely high [68] and the percentage of both new cases and previously treated cases of TB that demonstrate multidrug-resistance in Kazakhstan, Uzbekistan, Tajikistan, and Kyrgyzstan are among the highest in the world [50, 68]. All of these countries of Central Asia are among the 27 high- burden countries for MDR-TB in the world. The percentages both of new cases and previously treated cases of tuberculosis that demonstrate multidrug-resistance in Kazakhstan, Uzbekistan, Tajikistan, and Kyrgyzstan are among the highest in the world [69].
According to WHO, the percentage of TB cases with HIV co-infection is 2 % in Kazakhstan and Tajikistan and 3 % in Uzbekistan [67]. In these three countries, between 82-100 % of TB patients have been tested for HIV [67]. Data from Kyrgyzstan on testing TB patients for HIV are sparse and unreliable.
This situation requires substantial effort and improvement in diagnosis and treatment to control Central Asia’s staggering TB epidemic, especially that of MDR-TB. Furthermore, there is limited attention to adherence to TB treatment protocols for people who use drugs. Effective programs have been created in a number of regions around the world and these models should be adopted in Central Asia.
HCV
HCV imposes a major disease burden in regions that have high numbers of PWID, such as Central Asia, Eastern Europe, and Russia. While robust data on the magnitude of HIV/HCV co-infections are lacking, the higher prevalence and increased risk of HCV relative to HIV transmission through sharing contaminated injection equipment suggests that the majority of PWID infected with HIV will also be co-infected with HCV [70••]. In a recent study with 364 couples (728 individuals) from Almaty, Kazakhstan, where at least one member of the dyad reported recent IDU, 90.2 % of PWID were HCV positive and 15.5 % of those who did not report injecting drugs were HCV positive. The prevalence of HIV and HCV co-infection among the total sample was 20.7 % [18].
Prisoners are a unique population of PWID affected by HCV. In Kyrgyzstan, where 26 % of incarcerated men and women are estimated to be HIV positive, 38 % test positive for HCV [71]. In Kazakhstan, 91 % of HIV infected prisoners are HCV positive and 40 % of the general prison population have antibodies to HCV [47, 72]. A sample of 491 PWID in Dushanbe, Tajikistan reported HCV prevalence of 61 % with 50 to 51 (98 %) also tested HCV antibody positive, suggesting near universal HIV/HCV co-infection [16].
The high prevalence of HCV observed among PWID highlights the need for an urgent focus on HCV testing and treatment among this most at risk group in Central Asia. Needle exchange programs (NEP), OST, and ART programs can serve as appropriate settings to help reduce HIV and HCV co-infections. Improving HCV treatment outcomes among PWID should include education and training for service providers and primary care physicians on understanding HCV risks and recognizing the importance of receiving treatment. HCV prevention and treatment needs to be integrated into services for patients who are at risk for HIV as well as for those who are HIV positive. A long-term strategy for the control of HIV/HCV co-infection should encompass the prevention of both HIV and HCV and co-infection treatment. Finally, reduction of stigma and discrimination related to both HCV and PWID are required in order to promote the uptake of HCV testing and treatment programs.
HIV Testing and Counseling
The WHO European Action Plan for 2012-2015 released in 2011 calls for reducing the number of undiagnosed people by increasing early uptake of HIV testing and counseling services, especially in the critical populations mentioned above [73]. Specifically, the Action Plan sets as a target having more than 90 % of PWID aware of their HIV status by 2015. Despite notable progress in scaling up HIV testing in Central Asia, the number of key populations (such as PWID, MSM, and FSWs who inject drugs) who have been tested remains well below WHO’s recommended coverage of more than 90 % [73]. Coverage of PWID with recent testing (during the last 12 months) increased in Kazakhstan (from 52 to 65 %) and in Tajikistan (from 40 to 54 %) from 2009 through 2011. However, testing dropped from 40 % to 36 % in Kyrgyzstan and from 34 % to 29 % in Uzbekistan [74]. A recent study conducted in Almaty, Kazakhstan with 580 PWID found that 25 % of the study sample had never been tested for HIV [18]. Coverage of PWID with a recent HIV test is higher than coverage among MSM with an HIV test in all Central Asian countries, except for Uzbekistan (29 % for PWID versus 31 % for MSM in 2011). For example 77.1 % of FSWs were tested in Kazakhstan versus 64.7 % of PWID in 2011 and this can be explained by mandatory testing of detained FSWs in all Central Asian countries. HIV home-based self-testing is not available in Central Asia. There is an urgent need to increase HIV testing among these key populations in Central Asia.
HIV Care
Currently, the Ministries of Health in Central Asian countries recommend ART for HIV-infected individuals with CD4+ cell count <350/mm3 or WHO clinical stage III/IV which is analogous with the 2010 WHO guidelines [75]. As of January 2013, the proportion of estimated ART-eligible PWID on treatment among those who were enrolled in HIV care in Kazakhstan, Kyrgyzstan, and Tajikistan ranged between 51 % and 88 % [53, 55, 56]. However these statistics must be considered with caution because a large number of PWID are not enrolled in care and ART coverage may actually be much lower [76, 77]. Solid data on ART among other key populations are limited and access to ART treatment is limited by several barriers.
Barriers to HIV Testing, Treatment and Care
A number of structural barriers to HIV testing and treatment among key at-risk populations in Central Asia have been identified in multiple level domains [78]. Barriers to HIV testing and treatment exist within the health sector (i.e., discriminatory practices of health care providers, violation of confidentiality, logistic, and geographical barriers). Negative attitudes deprive PWID, MSM, and FSWs of needed services. Informal charges are often instituted for what should be anonymous, free health care. Low levels of commitment by staff and unwillingness to work with these key at-risk populations among government health care providers often result in clients having negative experiences in accessing HIV services and prevent them from using such services again [79].
Discrimination against PWID such as punitive drug laws, stigmatization of HIV/AIDS and drug use, police harassment and violence affect access to HIV testing and treatment [49]. Criminalization of drug use and discriminatory practices among government service providers restrict access of PWID to NEPs and NGOs where HIV services are located. Police often arrest PWID who access harm reduction services, confiscate drugs and syringes, and/or extract bribes for the possession of syringes/needles [78, 80].
There are a number of organizational barriers to accessing government-run clinic-based HIV testing and counseling services for PWID in Central Asia such as inconvenient hours, distant locations, and transportation costs [77, 81]. Despite increasing the number of HIV service sites, most are located in urban communities and are consolidated in certain areas, which limits access to HIV testing and treatment for suburban and rural PWID [81].
Women who inject drugs suffer additional structural barriers to accessing HIV testing and treatment including gender-based violence and inequalities in accessing health services [82, 83]. They also face more difficulties in obtaining child care, and paying for transportation to get to distant HIV services sites [81].
Conclusion
To meet the needs of key populations especially PWID, their sexual partners, MSM, sex workers and their social networks, it is critical to establish comprehensive programs that include harm reduction services, that integrate HIV prevention, care, and treatment services, and that target common co-morbidities, particularly HCV, TB, and the consequences of drug overdose. Most importantly, redressing structural barriers is critical to enhancing access to such services. This process must involve overcoming all the aforementioned barriers, especially discriminatory practices and laws and transforming the health system to enable ready access and quality of care.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Beyrer C, Abdool Karim Q. The changing epidemiology of HIV in 2013. Curr Opin HIV AIDS. 2013;8(4):306–10.
World AIDS Day Report: 2012. Switzerland: UNAIDS, 2012. 2012.
Coker RJ, Atun RA, McKee M. Health-care system frailties and public health control of communicable disease on the European Union’s new eastern border. Lancet. 2004;383:1389–92.
UNICEF. A decade of transition, Regional Monitoring Report, No.8. Florence: UNICEF Innocenti Research Center; 2001.
Vlassov V. Is there epidemiology in Russia? J Epidemiol Commun Health. 2000;54:740–44.
Beyrer C, DW, Celentano D. Public Health Aspects of HIV/AIDS in Low and Middle Income Countries., in “The HIV/AIDS Epidemics in the former Soviet Union, Central Asia and Iran.” I.D.C.a.C.B. (eds). Editor. 2008, New York:Springer Press.
Gouws E, Cuchi P, H.I.V.I.b.M.o.T. International Collaboration on Estimating. Focusing the HIV response through estimating the major modes of HIV transmission: a multi-country analysis. Sex Transm Infect. 2012;88(Supp 2):i76–85.
Europe., E.C.f.D.P.a.C.W.R.O.f. HIV/AIDS surveillance in Europe 2011, in Stockholm: European Centre for Disease Prevention and Control. 2012.
Sullivan LE, Fudala PJ, Fiellin DA. Decreasing inernational HIV transmission: the role of expanding access to opioid agonist therapies for injection drug users. Addiction. 2005;100:150–8.
Platt L, et al. Factors mediating HIV risk among female sex workers in Europe: a systematic review and ecological analysis. BMJ Open 2013; 3(7).
Wagner KD et al. Drug-using male clients of female sex workers who report being paid for sex: HIV/sexually transmitted infection, demographic, and drug use correlates. Sex Transm Dis. 2013. doi:10.1097/OLQ.0b013e31829569ec. The paper examines HIV/STI among 170 drug-using male clients of FSWs in Tijuana, Mexico to determine the extent to which these men report being paid for sex and the association with positive HIV/STI results. Findings suggest that drug-using male clients of FSWs may be characterized by unique risk profiles that require tailored HIV prevention interventions. The findings have important implications for prevention for FSWs.
Talu A et al. HIV infection and risk behaviour of primary fentanyl and amphetamine injectors in Tallinn, Estonia: implications for intervention. Int J Drug Policy. 2010;21(1):56–63.
Grund JP, Latypov A and Harris M. Breaking worse: the emergence of krokodil and excessive injuries among people who inject drugs in Eurasia. Int J Drug Policy. 2013.
El-Bassel N, Strathdee S and El Sadr W. People who inject drugs in Central Asia and HIV: Confronting the perfect storm. Drug Alcohol Depend. 2013.
Thekkemuriyi DV, JS, Pillai U. ‘Krokodil’-A Designer Drug from Across the Atlantic, with Serious Consequences. Am J Med. 2013;(13): 00879-6.
Beyrer C et al. Characterization of the emerging HIV type 1 and HCV epidemics among injecting drug users in Dushanbe, Tajikistan. AIDS Res Hum Retrovir. 2009;25(9):853–60.
Walsh N, Maher L. HIV and viral hepatitis C coinfection in people who inject drugs: implications of new direct acting antivirals for hepatitis C virus treatment. Curr Opin HIV AIDS. 2012;7(4):339–44.
El-Bassel N, et al. hiv among injection drug users and their intimate partners in Almaty, Kazakhstan. AIDS Behav. 2013: 1–11.
Grund J-PC, Latypov A and Harris M. Breaking worse: the emergence of krokodil and excessive injuries among people who inject drugs in Eurasia. Int J Drug Policy. 2013.
Europe BN. Russia reports 12% rise in HIV - 200 new cases a day. 28 November 2012.
Baral S et al. Burden of HIV among female sex workers in low-income and middle-income countries: a systematic review and meta-analysis. Lancet Infect Dis. 2012;12(7):538–49.
Decker MR, WA, Moguilnyi V, Peryshkina A, Ostrovskaya M, Nikita M, Kuznetzova J, et al. Female sex workers in three cities in Russia: HIV prevalence, risk factors and experience with targeted HIV prevention. AIDS Behav. 2013. doi: 10.1007/s10461-013-0577-y.
Zohrabyan L, JL, Scutelniciuc O, Iovita A, Todirascu L, Costin T, Plesca V, et al. Determinants of HIV infection among female sex workers in two cities in the Republic of Moldova: the role of injection drug use and sexual risk. AIDS Behav. 2013;17(8): 2588–96.
Dolan K et al. HIV in prison in low-income and middle-income countries. Lancet Infect Dis. 2007;7(1):32–41.
van der Werf MJ, HV, Noori T. Is tuberculosis crossing borders at the Eastern boundary of the European Union?. Eur J Public Health. 2013;23(6): 1058–63.
Amirkhanian YA et al. Male labor migrants in Russia: HIV risk behavior levels, contextual factors, and prevention needs. J Immigr Minor Health. 2011;13(5):919–28.
Weine S et al. HIV sexual risk behaviors and multilevel determinants among male labor migrants from Tajikistan. J Immigr Minor Health. 2013;15(4):700–10.
Europe, E.C.f.D.P.a.C.W.R.O.f. Tuberculosis surveillance and monitoring in Europe 2013. 2013.
Post FA et al. Multi-drug-resistant tuberculosis in HIV positive patients in Eastern Europe. J Infect. 2013. doi:10.1016/j.jinf.2013.09.034.
Dalton T, Cegielski P, Akksilp S, Asencios L, Campos Caoili J, Cho S-N, et al. Prevalence of and risk factors for resistance to second-line drugs in people with multidrug-resistant tuberculosis in eight countries: a prospective cohort study. Lancet. 2012;380(9851): 1406–17.
Network, C.a.E.E.H.R. Hepatitis C Among Injecting Drug Usersin the New EU Member States and Neighboring Countries: SITUATION, GUIDELINES AND RECOMMENDATIONS. 2007.
Ocheret D, BD, Sultangaziev A, Matuizaite E. Eurasian Harm reduction Network. Current situation regarding access to hepatitis C treatment in Eastern Europe and Central Asia. 2012.
Mathers BM et al. HIV prevention, treatment, and care services for people who inject drugs: a systematic review of global, regional, and national coverage. Lancet. 2010;375(9719):1014–28.
Hurley SF, Jolley DJ, Kaldor JM. Effectiveness of needle-exchange programmes for prevention of HIV infection. Lancet. 1997;349(9068):1797–800.
Uusküla A, DJD, Kals M, Rüütel K, Abel-Ollo K, Talu A, Sobolev I. Expanded syringe exchange programs and reduced HIV infection among new injection drug users in Tallinn, Estonia. BMC Public Health. 2011;30(11): 517.
Vasquez C, LD, Nikolaenko S, Yatsishin S, Lesnikova D, Cox D, Pankovich J, et al. St Petersburg-CIHR Canadian HIV Trials Network Collaboration. Gender disparities in HIV risk behavior and access to health care in St. Petersburg, Russia. AIDS Patient Care STDS. 2013;27(5): 304–10.
Uusküla A, LK, Raag M, Šmidt J, Semjonova S, Kogan J, Amico KR, et al. Antiretroviral therapy (ART) adherence and correlates to nonadherence among people on ART in Estonia. AIDS Care. 2012;24(12): 1470–9.
Mocroft A et al. Risk factors and outcomes for late presentation for HIV-positive persons in europe: results from the collaboration of observational HIV epidemiological research europe study (COHERE). PLoS Med. 2013;10(9):e1001510.
Sarang A, RT, Sheon N. Systemic barriers accessing HIV treatment among people who inject drugs in Russia: a qualitative study. Health Policy Plan. 2013;28(7): 681–9.
Des Jarlais DC, U.A. Multiple HIV Prevention Packages for IDUs in Estonia. Presentation at the NIH MP3 Investigators meeting, NIH, Bethesda, USA 2013.
Spicer N, et al. ‘It’s risky to walk in the city with syringes’: understanding access to HIV/AIDS services for injecting drug users in the former Soviet Union countries of Ukraine and Kyrgyzstan. Global Health. 2011;7(7): 22.
Amirkhanian YA et al. People with HIV in HAART-era Russia: transmission risk behavior prevalence, antiretroviral medication-taking, and psychosocial distress. AIDS Behav. 2011;15(4):767–77.
Bikmukhametov DA et al. Bias in medicine: a survey of medical student attitudes towards HIV-positive and marginalized patients in Russia, 2010. J Int AIDS Soc. 2012;15(2):17372.
Kiriazova TK, PO, Perehinets IB, Neduzhko OO. Association of injecting drug use and late enrolment in HIV medical care in Odessa Region, Ukraine. HIV Med. 2013;14(Supp 3): 38–41.
Opioid treatment in Ukraine risks losing momentum, in Bulletin of the World Health Organization. 2013. p. 81–156.
World AIDS. Day Report: 2012. Switzerland: UNAIDS; 2012.
UNAIDS. Country report on progress in implementation of the global response to HIV infection, Kazakhstan 2012. 2012, Joint United Nations Programme on HIV/AIDS Geneva, Switzerland.
Donoghoe M. Why is the HIV epidemic in Eastern Europe and Central Asia the fastest growing in the world and what can we do to stop it? , in XIX International AIDS Conference. 2012: Washington, D.C.
Jolley E, et al. HIV among people who inject drugs in Central and Eastern Europe and Central Asia: a systematic review with implications for policy. BMJ Open. 2012;2(5).
Thorne C et al. Central Asia: hotspot in the worldwide HIV epidemic. Lancet Infect Dis. 2010;10(7):479–88.
UNAIDS. Country report on progress in implementation of the global response to HIV infection, Kyrgyz Republic 2012. 2012, Joint United Nations Programme on HIV/AIDS Geneva, Switzerland.
UNAIDS. Country report on progress in implementation of the global response to HIV infection, Uzbekistan 2012. 2012, Joint United Nations Programme on HIV/AIDS Geneva, Switzerland.
Kazakhstan R.A.C.-. Report on AIDS service activities for 2012. 2012, Republican AIDS Center of the Republic of Kazakhstan: Almaty.
Kyrgyzstan R.N.C.-. Program monitoring data. 2013, Republican Center of Narcology of the Kyrgyz Republic.
Tajikistan R.A.C.-. 2011. Republican AIDS Center of the Republic of Tajikistan: Dushanbe.
Uzbekistan R.A.C.-. Results of the sentinel surveillance study. 2011, Republican AIDS Center of the Republic of Uzbekistan: Tashkent.
Yusopov O. National report on the drug situation in the Republic of Kazakhstan for 2011. 2012, National scientific-practical center on medical and social problems of drug addiction/CADAP: Astana-Prague.
Mravcik V. National report on the drug situation in the Republic of Uzbekistan for 2011. 2012: Prague.
Boltaev A, et al. The scaling up of HIV prevention for people who inject drugs in Central Asia: a review of structural challenges and ways forward. Drug Alcohol Depend. 2013.
Boltaev A et al. Evaluation of a PilotMedication-assisted therapy program in kazakhstan: successes, challenges, and opportunities for scaleup advances. Prev Med. 2012. doi:10.1155/2012/308793. The paper reviews the current status and trends of national efforts in CA countries to control HIV among people who inject drugs and key structural and health systems-related challenges. The paper underscores that scaling up prevention and treatment demand attention to structural, health-care and social factors that facilitate HIV risk andimpede utilization of harm reductionservices.
UNAIDS. UNAIDS Report on the Global AIDS Epidemic 2012 (Annexes). Geneva: UNAIDS; 2012.
Cáceres C et al. Estimating the number of men who have sex with men in low and middle income countries. Sex Transm Infect. 2006;82 suppl 3:iii3–9.
Morris MD et al. Factors associated with pathways toward concurrent sex work and injection drug use among female sex workers who inject drugs in northern Mexico. Addiction. 2013;108:161–70.
Syvertsen JL et al. Where sex ends and emotions begin’: love and HIV risk among female sex workers and their intimate, non-commercial partners along the Mexico-US border. Cult Health Sex. 2013;15(5):540–54. This study among female sexworkers utilizes qualitative data to understand emotional relationships between females and their non-commercial partners, pointing to the role of intimacyin decisions about drug and sexual risk behaviors.
Uusküla A et al. Condom use and partnership intimacy among drug injectors and their sexual partners in Estonia. Sex Transm Infect. 2012;88(1):58–62.
Hillis SD et al. HIV seroprevalence among orphaned and homeless youth: no place like home. AIDS. 2012;26:105–10.
WHO. Global tuberculosis report. Geneva: World Health Organization; 2012.
Schluger NW et al. Tuberculosis, drug use and HIV infection in Central Asia: an urgent need for attention. Drug Alcohol Depend. 2013;132:S32–6.
Schluger NW, E-BN, Hermosilla S, Terlikbayeva A, Darisheva M, Aifah A, et al. Tuberculosis, drug use and HIV infection in Central Asia: An urgent need for attention. Drug Alcohol Depend. 2013;132: S32–6.
Walsh N, Maher L. HIV and HCV among people who inject drugs in Central Asia. Drug Alcohol Depend. 2013. doi:10.1016/j.drugalcdep.2013.06.030. The paper examines the relationship between HCV and HV among people who use drugs in CA .It shows that while the HIV treatment response has, HCV diagnosis and treatment in the region for people who use drugs remain limited. Improving access to HCV diagnosis and treatment is urgently needed in CA especially for people who use drugs.
Larney S et al. The incidence and prevalence of hepatitis C in prisons and other closed settings: results of a systematic review and meta‐analysis. Hepatology. 2013. doi:10.1002/hep.26387.
PEPFAR. Central Asia Region Operational Plan Report FY 2010. Washington, D.C: United States Government; 2010.
WHO. European action plan for HIV/AIDS 2012-2015. Geneva: World Health Organization; 2011.
UNAIDS. UNAIDS Global AIDS Response Progress Reporting: Kazakhstan, Kyrgyzstan, Tajikistan, Uzbekistan. 2012, UNAIDS: Available at: http://www.unaids.org/en/dataanalysis/knowyourresponse/countryprogressreports/2012countries/.
WHO. Antiretroviral Therapy for HIV infection in adults and adolescents: recommendations for a Public Health Approach. 2010 Revision. Austria: WHO; 2010.
Deryabina A et al. Report on the Evaluation of HIV integrated biobehavioral surveillance in the Republic of Kazakhstan. Almaty: ICAP; 2012.
Deryabina A. Mapping of key HIV services, assessment of their quality, and analysis of gaps and needs of most-at-risk populations in Chui Oblast and Bishkek City, Kyrgyzstan. Arlington: USAID’s AIDS Support and Technical Assistance Resources, AIDSTAR-One, Task Order 1; 2011.
Spicer N et al. Circus monkeys or change agents? Civil society advocacy for HIV/AIDS in adverse policy environments. Soc Sci Med. 2011;73(12):1748–55.
Wolfe D, Carrieri MP, Shepard D. Treatment and care for injecting drug users with HIV infection: a review of barriers and ways forward. Lancet. 2010;376(9738):355–66.
Mimiaga MJ et al. “We fear the police, and the police fear us”: structural and individual barriers and facilitators to HIV medication adherence among injection drug users in Kiev, Ukraine. AIDS Care. 2010;22(11):1305–13.
Grabbe KL et al. Increasing access to HIV counseling and testing through mobile services in Kenya: Strategies, utilization and cost-effectiveness. J Acquir Immune Defic Syndr. 2010;54(3):317.
El-Bassel N, Terlikbaeva A, Pinkham S. HIV and women who use drugs: double neglect, double risk. Lancet (Br Ed). 2010;376(9738):312.
Wood RA. Issues of gender in injection drug use: examining contextual circumstances of women’s first injecting experience and factors associated with treatment engagement. 2010.
Acknowledgements
Dr. DeHovitz acknowledges partial support from grant from Fogarty International Center and National Institute of Drug Abuse, (D43 TW000233). Dr. El-Bassel acknowledges the partial support received from a grant from the National Institute of Drug Abuse (R01DA033168).
Compliance with Ethics Guidelines
ᅟ
Conflict of Interest
Jack DeHovitz, Anneli Uuskula, and Nabila El-Bassel declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
DeHovitz, J., Uuskula, A. & El-Bassel, N. The HIV Epidemic in Eastern Europe and Central Asia. Curr HIV/AIDS Rep 11, 168–176 (2014). https://doi.org/10.1007/s11904-014-0202-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11904-014-0202-3