Abstract
Purpose of Review
The goal of the present review is to explore the relationship between dietary changes and alterations in gut microbiota that contribute to disorders of gut motility and obesity.
Recent Findings
We review the microbiota changes that are seen in obesity, diarrhea, and constipation and look at potential mechanisms of how dysbiosis can predispose to these. We find that microbial metabolites, particularly short chain fatty acids, can lead to signaling changes in the host enterocytes. Microbial alteration leading to both motility disorders and obesity may be mediated by the release of hormones including glucagon-like peptides 1 and 2 (GLP-1, GLP-2) and polypeptide YY (PYY). These pathways provide avenues for microbiota-targeted interventions that can treat both disorders of motility and obesity.
Summary
In summary, multiple mechanisms contribute to the interplay between the microbial dysbiosis, obesity, and dysmotility.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The human body is comprised of trillions of microorganisms that reside in the respiratory tract, gut, skin, and urogenital tracts and is collectively termed the human microbiome. New high throughput technologies make it possible to look at the ecosystem of microorganisms in our body as a whole. This has led to exponential growth in the field linking alterations in the microbiome to various disease states including obesity, and disorders of gut motility [1,2,3,4].
The Interplay Between Diet and the Microbiome
In the intestine, the microbiome has numerous important functions including maintaining the integrity of the gut wall, preventing overgrowth of harmful organisms, and breaking down and absorption of certain nutrients. Animal studies have demonstrated that alterations in the microbiome may lead to phenotypic changes in the host such as weight gain or loss. In a seminal study, Backhed et al. showed that transplantation of stool from conventionally raised mice to germ-free rodents (absent of microbiota) led to 60% increase in body fat and increased insulin resistance and that these changes were independent of food intake [5]. This provided early evidence that the microbiome may play an important independent role in energy harvesting and storage. The intestinal tract is largely comprised of the dominant phyla Bacteroides and Firmicutes, accounting for about 75% of the total population [6]. Turnbaugh et al. demonstrated mice with a mutation in the leptin gene leading to morbid obesity (ob/ob) had higher relative abundance of Firmicutes and proportional reduction in Bacteroides. Furthermore, stool transplants of these genetically obese mice to their lean littermates resulted in weight gain in the latter with microbiome distribution resembling that of the obese mice [7]. Several other studies in animal models and humans support a similar pattern of increased ratio of Firmicutes/Bacteroides associated with obesity and the reverse seen with weight loss [3, 8,9,10]. Other studies, however, have shown either the opposite or no relationship [11,12,13], suggesting that the microbial changes related to obesity are likely more complex, possibly with greater impact at lower phylogenetic levels and functional characteristics of the microbiome as a whole.
Dietary changes can lead to rapid changes in the gut microbial environment at the genera and species levels. Wu et al. assessed the relationship between nutrient content and microbial patterns and identified that diets high in animal protein and saturated fat had increased abundance of the Bacteroides genera whereas those high in fiber had relatively higher abundance in Prevotella [14]. A separate study looked at differences in gut microbiota among individuals placed on a Mediterranean diet (relatively high fat diet with large proportion of monounsaturated fat) compared to those on a low-fat, high complex carbohydrate diet [15]. After 1 year of follow-up, those on the low-fat, high complex carbohydrate diets had higher levels of Prevotella and reduced Roseburia whereas those on the Mediterranean diet had increased abundance of Roseburia and Oscillospira [15]. In agreement with these findings, increase in Prevotella has been seen with higher intake of plant-based fibers [16] likely reflecting increased ability of the microbe to extract calories from these resistant starches [17]. These findings provide support that changes in macronutrient content can directly impact microbiota at these lower phylogenetic levels.
The Role of Microbiota in Metabolism and Obesity
Obesity is a chronic condition that affects 35% of the US population [18] and is growing in prevalence both in the USA and globally [19]. Even with aggressive lifestyle interventions with diet and exercise, many individuals still struggle with losing 3–5% of their body weight [20, 21]. A study by Pedrogo and colleagues suggested that certain gut microbes may help predict likelihood of successful weight loss with lifestyle interventions. In comparing individuals who were and were not able to achieve 5% weight loss, they found that increased abundance of Phascolarctobacterium was associated with greater likelihood of achieving weight loss while Dialister was associated with failure [22]. Furthermore, functional analysis of the microbial genes also showed that those who did not achieve 5% weight loss had higher abundance of enzymes involved in carbohydrate metabolism [22]. This suggests that there may be a direct relationship between host and microbial metabolism. In another study in which overweight and obese adults were treated with 6 weeks of calorie restricted diet, increased abundance of a mucin-degrading bacterium, Akkermansia muciniphila, at baseline was associated with greater improvement in markers of glucose metabolism and adiposity [23].
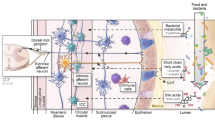
Several studies have shown that individual organisms and groups of related organisms or phylotypes with specific metabolic functions play an important role in the development of obesity. In a study of lean and obese young Chinese individuals, Liu et al. found that people who were obese had reduced abundance of certain Bacteroides species, including B. thetaiotaomicron, B. intestinalis, and B. ovatus as compared to matched controls [24]. Furthermore, using mouse models, they found that gavage with B. thetaiotaomicron alleviated diet-induced obesity from high fat diets. This effect may be driven by increased metabolism of glutamate, leading to lower serum glutamate levels in B. thetaiotaomicron-supplemented animals [24]. These studies not only show that that the microbiota may be predictive of likelihood for weight loss but that introduction of certain organisms can improve the metabolism resulting in weight loss (Fig. 1).
Microbiome and Disorders of Gut Motility
Recent studies have examined the connections between the gut microbiota and disorders of gastrointestinal motility, specifically constipation and diarrhea.
Constipation
Gut dysbiosis and dysmotility and constipation have been associated with high fat diets [25•, 26]. Short chain fatty acids (SCFAs) including butyrate, propionate, and acetate are the end products of dietary fiber fermentation and may affect various metabolic pathways [7]. After being broken down from otherwise indigestible polysaccharides, they are absorbed by host cells and utilized as a direct energy source and in cell signaling pathways [27, 28]. Studies of mouse models of constipation have demonstrated that production of the SCFA, butyrate, is decreased in the setting of gut dysbiosis and suggest that repletion of butyrate may decrease symptoms of dysmotility [29, 30]. Hwang et al. investigated the changes in microbial composition triggered by different diets and the consequent changes in butyrate levels. They identified that different diets cause a reduction in butyrate production through separate metabolic pathways. For example, high fat diets decrease butyrate levels through reductions in Lactobacillus, whereas low-fat diets and loperamide decrease butyrate levels due to reductions in Roseburia and Oscillibacter [29]. Lower levels of bile acids also cause symptoms of constipation, longer transit time, and decreased colonic muscle contraction [30]. Furthermore, when butyrate and bile acids are supplemented, many of the constipation symptoms reverse [30]. Studies suggest the effect of metabolites of gut microbiota on dysmotility may be mediated by changing serotonin levels [31,32,33].
Diarrhea
Just as the microbiota can affect stool transit time, the transit time can also affect the composition of the microbiota. In vitro models of the colon have demonstrated that as transit time increases, there is a decrease in biomass and species diversity [34]. There is also an increase in SCFAs that corresponds to increased carbohydrate fermentation. The increase of SCFAs reflects the increased growth of microbiota that produce them, such as Firmicutes, Bacteroidetes, and Actinobacteria [34,35,36]. In studies of adults with motility disorders, Vandeputte and colleagues demonstrated similar findings. They found that increased stool consistency (looser stool with high transit time) is inversely related to microbiota diversity and positively associated with the Bacteroidetes/Firmicutes ratio [37]. They hypothesized that transit time may determine which microbiota proliferate by changing the amount and duration of nutrients the microbiota are exposed to [37].
Most of the evidence examining the link between gut microbiota and diarrhea comes from studies of post-infectious diarrhea. Studies have demonstrated that diarrhea-causing pathogens decrease the diversity of the gut microbiota, although the pathogen-specific changes are not consistent across studies [38,39,40]. After an infection resolves, the diversity of the microbiota recovers and diarrheal symptoms improve. Fecal microbiota transplants may speed the process of relieving diarrheal symptoms in patients with dysbiosis and diarrhea [41, 42], and have been found to restore microbiotal diversity some of the time [41].
Pathogenesis: Common Pathways of Obesity and Dysmotility
There are multiple theories as to how the gut microbiota is associated with obesity and gut dysmotility. Each of the theories considers different facets of metabolism and dysmotility from neuromuscular dysfunction and alterations to the gut wall, to microbial metabolites to common hormonal triggers of motility and metabolic functions.
Several mechanisms contribute to the effect of the microbiome on metabolism and obesity. The microbiota are involved in breakdown of otherwise non-digestible compounds thus affecting energy harvesting capacity and leading to weight gain depending on the microbial milieu [5, 7, 43]. Dietary fibers play an important role in shaping the microbiota and can protect against deleterious changes induced by high fat diets [44]. Dietary supplementation with fibers that increase levels of SCFAs as well as direct consumption of SCFAs has been shown to have beneficial effects on lipid and glucose metabolism as well as weight reduction in animals and humans [44,45,46,47]. Increasing evidence suggests that the mechanism of how SCFAs alter metabolism and obesity may be related to cell signaling pathways leading to release of incretin and other gut hormones.
Incretin and other gut hormones are released by the entero-endocrine cells of the gut and include glucose-dependent insulinotropic polypeptide (GIP), peptide YY (PYY), and glucagon-like peptide-1 and 2 (GLP-1 and GLP-2). They are involved in regulating gut motility, satiety, and glucose metabolism [48]. In mouse models, administration of SCFAs, butyrate, and propionate were found to protect against diet-induced obesity and reduce food intake, and these effects were, in part, mediated by increased secretion of incretin hormones [49]. Sodium butyrate administration lead to increased plasma levels of GLP-1, GIP, and PYY while sodium propionate lead to increased GIP only [49]. In another study in humans, oral administration of the fiber, inulin combined with propionate acutely stimulated secretion of PYY and GLP-1 and was associated with reduced energy intake [50]. Despite the stimulating effects of SCFAs supplementation on incretin hormones, they were not shown to directly influence gastric emptying [50]. Using mouse models of type 2 diabetes, Grasset et al. found evidence of GLP-1 resistance in mice fed high fat diet and that these changes correlated with microbial dysbiosis [51]. Furthermore, GLP-1-mediated insulin secretion was absent in germ-free mice, suggesting that microbiota are needed for the action of GLP-1 [51]. In another study of humans with type 2 diabetes, increased dietary fiber promoted SCFA-producing bacteria, which lead to increased production of GLP-1 contributing to improved glycemic control [52•].
In addition to the effects of microbial metabolites on incretin hormone secretion and action, gut microbiota may affect metabolism by directly altering the integrity of the gut wall. Using human colonic tissue, Chassaing et al. found that microbiota encroach within the gut wall and the degree of encroachment directly correlated with dysglycemia in adults [53]. Finally, endotoxemia with the microbial product, lipopolysaccharide (LPS), may lead to inflammatory changes that increase the risk of obesity. In a recent study by Fabbiano et al., the authors found that the microbiota of calorically restricting mice have increased abundance in the families Lactobacillaceae, Erysipelotrichaceae, and Verrucomicrobiaeceae, the latter largely represented by A. muciniphila [54]. Metabolic improvements were associated with lower levels of LPS and may be mediated by suppression of LPS-TLR4 signaling [54]. Reintroduction of LPS prevented the metabolic improvements seen with caloric restriction [54].
Studies conducted in mice and rats have identified an association between dysmotility and changes to the enteric nervous system mediated by the gut microbiota. Mice fed high fat diets have increased amounts of Bacteroidetes, Firmicutes, Escherichia coli, and Bifidobacteria [25•], as well as damage to and loss of enteric neurons [25•, 26, 55]. The mechanism linking gut dysbiosis to neuronal injury is still unclear but several hypotheses have been proposed. Toll-like receptors (TLRs) are an important part of the innate immune system, acting as a first line of defense in recognizing microbial molecules and triggering an inflammatory response [56]. They are present throughout the intestine and are often the first interaction between gut microbiota and the host immune system [56]. In in vitro studies, Anitha et al. demonstrated that LPS combined with palmitate from the high fat diet activated TLR4 to cause neuronal apoptosis [25•]. Additionally, they found that administering an oligofructose prebiotic to mice fed high fat diets caused levels of LPS to decrease and prevented neuronal damage [25•]. In other mouse studies, Reichardt et al. corroborated the findings that gut dysbiosis, free fatty acids, and TLR4 activation are associated with enteric neuronal death and delayed transit time [26]. Other research groups have identified a correlation between changes in TLR2 signaling, gut dysbiosis, and damage to both the structure and function of the enteric nervous system [55, 57]. Gut dysbiosis has been linked not only to neuronal loss but also to decreased neuromuscular contractility and impaired neurotransmission [55, 58]. Neuronal loss, slowed signal transmissions, and decreased contractility can all lead to slower gastrointestinal transit. Although studies suggest that gut microbiota have a role in this, that role still needs further clarification.
Other studies have investigated the role of microbial metabolites, including SCFAs and secondary bile acids in dysmotility. Both SCFAs and secondary bile acids can increase levels of serotonin, a hormone that regulates intestinal motility [32, 59]. Serotonin has been shown to upregulate receptors for hormones that cause neuromuscular excitation and trigger contraction of the smooth muscle in the gastrointestinal tract [60]. Serotonin secretion can be affected by the gut microbiome [33]. In rat studies using a probiotic mixture to alter the makeup of the gut microbiota, Deng and colleagues found that microbiota that produce SCFAs also stimulate serotonin synthesis to increase intestinal motility [31].
These studies identify common mechanisms driving enteric nervous system injury and obesity. In summary, we identify common inflammatory pathways involving TLRs and LPS as well as changes in microbial metabolites that may drive both disease processes.
Interventions in Disorders of Obesity and Dysmotility
Microbiota contribute to motility disorders through a variety of mechanisms, but most of them share a common starting point in gut dysbiosis. Consequently, prior interventions have looked at methods to restore the gut microbiome or prevent dysbiosis from occurring.
Dietary Interventions in Obesity
The impact of different types of diets and dietary supplements on the microbiome and their effect on obesity and metabolism have been studied at length. Animal studies have looked at the impact of dietary supplements and foods on the microbiota. Foods and supplements including antioxidants, camu camu extract (Brazilian fruit with rich phytochemical properties), and fungus, Hirsutella sinensis, have all been shown to blunt weight gain and inflammatory changes associated with high fat diets as well as to promote growth of certain gut microorganisms [61,62,63]. Supplementation of camu camu in mice fed high fat diets altered the microbiota while blunting fat gain, metabolic inflammation, and endotoxemia [61,62,63]. In another animal study using pigs, addition of alfalfa to the diet led to increased production of butyrate along with PYY and precursors of GLP-1 and GLP-2 [64]. The effect of direct supplementation of an inulin-propionate ester was studied in a randomized, double-blind, placebo-controlled study of 50 healthy volunteers and provides insight into the potential mechanism by which the microbiota may affect obesity and metabolism [50]. During a 24-week study period, subjects experienced reduction in weight and intra-abdominal adiposity, had improved lipid profiles, and had protection against deteriorating insulin resistance seen in the control group. Subjects receiving proprionate had increased postprandial PYY and GLP-1 levels and reduced overall energy intake [50]. In another study, Karl et al. compared the effects of whole versus refined grains on metabolism among healthy adults [65]. They found that diets rich in whole grains lead to overall favorable energetics with net daily energy loss as compared to refined grains [65]. These changes were also accompanied by trends towards reduction in pro-inflammatory taxa Enterobacteriaceae and increased butyrate-producing Roseburia and Lachnospira associated with the whole grain diet [65].
Numerous foods and supplements have been shown to alter the microbiome in a manner that favors increased energy expenditure and provide promising avenues for future obesity research. More mechanistic studies are needed to determine whether the microbiota changes actually lead to metabolic improvements.
Dietary Interventions in Dysmotility
The foods that people eat affect the composition of their microbiota, which can in turn impact motility [35, 66]. Thus, studies have been conducted looking at how alterations in diet reverse symptoms of dysmotility. In patients with diarrhea secondary to irritable bowel syndrome (IBS), studies have examined the efficacy of limiting the amount of fermentable carbohydrates consumed to decrease diarrheal symptoms in a diet termed “low FODMAP (fermentable oligosaccharides, monosaccharides, disaccharides, and polyols) diet.” Fermentable carbohydrates increase the amount of water and gas in the intestines, which leads to softer, more frequent stools [67•, 68]. A 2016 meta-analysis of IBS studies found that symptom severity scores improve in patients consuming a FODMAP diet [69]. In a placebo-controlled trial of adults with dysmotility, Staudacher et al. demonstrated that patients who receive low FODMAP dietary advice experience fewer symptoms of IBS compared to those who receive placebo dietary advice. They also found that low FODMAP dietary advice is correlated with decreases in the population of Bifidobacteria, a genus of microbiota that is inversely related to symptoms [67•]. Importantly, Staudacher’s group found that the Bifidobacteria could be restored with consumption of a multi-strain probiotic in addition to the low FODMAP diet [67•]. Although dietary interventions are promising options for treatment of dysmotility, restricting foods linked to symptoms can also have undesired effects on the microbiota composition with other deleterious health outcomes. More research is needed to identify which aspects of diet would have the most targeted effects on dysmotility.
Medications
GLP-1 receptor agonists (GLP-1RA) used in the treatment of type 2 diabetes are medications that mimic the function of native GLP-1 and are of particular interest due to their additional effect on weight by increasing satiety and slowing gastric emptying [70]. Studies looking at the effect of GLP-1 RAs on the microbiome have shown that these medications may act in part by altering the microbiota to favor weight reduction [71, 72]. Treatment with GLP-1RA, liraglutide, was associated with enrichment of A. muciniphila which has previously been associated with weight loss and improved metabolism following weight loss interventions [23, 71]. Thus, the mechanism of GLP-1 action in weight loss and metabolism may be in part mediated by microbiota changes.
Several medications have been shown to improve symptoms of dysmotility, but newer studies have examined the effects of those medications on the gut microbiota. In a study of rats with colitis, Zheng and colleagues demonstrated that sulfasalazine reverses gut dysbiosis in part through increasing populations of bacteria that produce short chain fatty acids or lactic acid [73]. However, not all medications that relieve symptoms change the microbiome. In a study of humans with constipation, Kang and colleagues demonstrated that administering lubiprostone does not alter the makeup of the gut microbiome but still increases butyrate production [74].
Rifaximin has been studied in the improvement of diarrheal symptoms. In a randomized, double-blinded, human study of patients with diarrhea, Fodor and colleagues demonstrated that treatment with rifaximin transiently decreases gut bacteria associated with poor health, such as Peptostreptococcaceae and Enterobacteriaceae, but the study did not investigate the relationship between the microbes and symptomatology [75]. Thus, although some of the medications that improve symptoms of dysmotility also change the makeup of the gut microbiome, the pathway connecting symptoms and the gut microbiome is still unclear.
Pre- and Probiotics
Prebiotics are non-living non-digestible materials that can alter the microbiota when consumed, and they have been studied both in obesity and dysmotility. They can be fermented by bacteria into metabolites that play an important role in metabolic pathways of both of the bacteria and host organism. In a recently published study by Zou et al., dietary supplementation with prebiotic, inulin, was shown to protect mice from microbiota and inflammatory changes associated with high fat diets [44]. Both animal and human studies have shown that supplementation with inulin may lead to alterations in the microbiota and microbial metabolites as well as improved glucose metabolism and reduced adiposity [76,77,78,79,80,81]. Intake of soluble fibers may lead to changes in the microbiota that directly alter energy expenditure. Wang et al. demonstrated that mice fed soluble fibers have increased oxygen consumption and energy expenditure, and reduced white fat mass and overall weight that is independent of caloric intake [10]. Mice with diet-induced obesity who were supplemented with soluble fiber had shifts in their microbiota with lower Firmicutes/Bacteroides ratio and increased microbial diversity [10]. In a study by Miyamoto et al., supplementation of soluble fiber, barley β-glucan, to mice fed high fat diets led to less weight gain than conventionally raised mice. These changes were associated with increased levels of SCFAs, particularly butyrate, and higher levels of PYY and GLP-1 [82]. These changes were not seen in germ-free mice given the same β-glucan supplement, suggesting a critical role of the microbiota in the metabolic effects of soluble fibers [82]. In humans, addition of dietary fibers such as inulin has been shown to increase levels of SCFAs leading to increased levels of hormones important in glucose metabolism and hunger such as ghrelin [80] and adiponectin [83]. In a study of obese women, administration of inulin-type fructans led to increases in Bifidobacterium with reduction in LPS and endotoxin as well as slight reduction in fat mass [77]. In another study of patients with type 2 diabetes, treatment with inulin alone or in combination with butyrate during a 45-day study period leads to reductions in waist circumference with a trend towards increased GLP-1 concentration [79]. In contrast, other studies in humans have failed to show effect of inulin on satiety and incretin hormones, GLP-1 or PYY [84, 85]. Taken together, the findings of these animal and human studies provide evidence that increased dietary fibers may lead to alterations in the gut microbiota favoring improved metabolic profile potentially leading to weight loss.
Probiotics are living microorganisms that are ingested orally in order to directly alter the microbiota. Use of probiotics has been studied in treatment of obesity-related disorders. In a double-blind randomized trial, Simon et al. studied the effect of supplementation of Lactobacillus reuteri over 3 weeks on glucose-tolerant lean and obese individuals [86]. They found that intake of the probiotic leads to increase in glucose-stimulated GLP-1 and GLP-2 release along with increased insulin secretion but with no change body mass seen during the study period. Another probiotic, Bifidobacterium animalis, combined with fiber was administered to healthy adults over a 6-month period and lead to 4.5% or 1.4 kg weight loss, with pronounced reduction in central adiposity and waist circumference [87].
Pre- and probiotics have also been studied in treatment of motility disorders. In a study in adults with constipation, Blatchford and colleagues demonstrated that kiwifruit administration leads to an increase in Faecalibacterium prausnitzii, a butyrate-producing microorganism that produces with anti-inflammatory properties [88]. Studies in rats with diarrheal symptoms have examined the effects of the Chinese prebiotic, Wuji Wan, and demonstrated that its relief of diarrheal symptoms is mediated by an increase in gut microbiota diversity. Administration of Wuji Wan increased Akkermansia, Bacteroides, and Parasutterella, and decreases Lactobacillus and Prevotella. Wuji Wan also increased the amount of mucin and goblet cells, stabilizing the gut mucosa to block bacterial translocation [89]. This suggests that changing the microbiotal makeup can relieve symptoms of dysmotility. In a mouse study, Jeong and colleagues demonstrated that administration of Lactobacillus kefiranofaciens, a microorganism in a probiotic used to relieve constipation also known as kefir, increases Firmicutes, Bacteroidetes, Lactobacillus, and Prevotella, and decreases Proteobacteria, Enterobacteriaceae, and Clostridium [90]. In a study of constipated adults, Kim and colleagues demonstrated that a mix of probiotics reduces symptoms of constipation and alters the makeup of the gut microbiome [91]. Patients who received the probiotic had similar increases in Lactobacillus, Bifidobacterium, and Bacteroides species. Hatanaka and colleagues found similar improvement of diarrheal symptoms and microbiotal changes with administration of Bacillus subtilis spores [92]. Although multiple studies have shown probiotics relieve symptoms of dysmotility [93], the mechanisms involved need to be further examined.
Gastric Bypass Surgery
Bariatric bypass surgery has been used for the management of obesity and comorbid conditions, primarily diabetes, and has been shown to have dramatic results both in reduction of obesity and obesity-related complications. Not surprisingly, bypass surgery also results in dramatic changes in gut microbiota. There is significant interest in the potential role of gut microbiota changes in the dramatic effect of bariatric surgery on obesity and metabolism. Bariatric surgery leads to long-term alterations in the gut microbiota composition and increased abundance of microorganisms associated with improved insulin resistance and weight loss, such as A. muciniphila and Bacteroides thetaiotaomicron [24, 94,95,96,97]. These organisms are also important in maintenance of gut wall integrity. Some patients undergoing bariatric surgery may have remission of diabetes. Murphy et al. demonstrated that elevations in Roseburia species correlated with likelihood of diabetes remission [98]. Furthermore, transplantation of gut microbiota from individuals who underwent bariatric surgery to germ-free mice lead to reductions in body fat and increased lean mass in those animals as compared to mice receiving stool transplant from obese subjects who did not have surgery [94]. This study supports a direct role of microbiota shifts contributing to the weight loss effects of bypass surgery. In addition to the effects bypass on the microbiota, pre-treatment with microbiota-altering agents may affect degree of weight loss following surgery. In a small study of subjects undergoing Roux-en-Y gastric bypass, supplementation with the prebiotic, fructo-oligosaccharide, was associated with greater weight loss following surgery [99].
Fecal Microbiota Transplantation
Fecal microbiota transplants are currently only FDA approved for treatment of Clostridium difficile infection. However, numerous mouse studies and one human study have demonstrated that directly altering the microbiome can affect host metabolism. In humans, transplantation of stool from lean to obese individuals leads to improved insulin sensitivity in the latter along with increased levels of butyrate-producing bacteria, Roseburia intestinalis [100]. Several trials looking at fecal transplantation for morbid obesity are ongoing (NCT03391817; NCT02530385). The findings of these trials may show whether directly changing the microbiota can provide a new avenue for management of obesity.
Summary
Evidence is mounting on the critical role that the microbiome plays in obesity and dysmotility. However, some conflicting findings in the effects of certain microbes and their metabolites reveal the importance of identifying pathogenic mechanisms rather than correlations between microbiota and disease. Here we discussed the recent literature on the relationship between diet, microbiome, obesity, and motility. Multiple mechanisms contribute to the pathogenesis of microbiota in these disease states and provide avenues for future interventions.
Abbreviations
- SCFAs:
-
Short chain fatty acids
- TLR:
-
Toll-like receptor
- FODMAP:
-
Fermentable oligosaccharides, monosaccharides, disaccharides, and polyols
- GLP:
-
Glucagon-like peptide
- GIP:
-
Glucose-dependent insulinotropic polypeptide
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Turnbaugh PJ, Backhed F, Fulton L, Gordon JI. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe. 2008;3(4):213–23. https://doi.org/10.1016/j.chom.2008.02.015.
Zhang X, Shen D, Fang Z, Jie Z, Qiu X, Zhang C, et al. Human gut microbiota changes reveal the progression of glucose intolerance. PLoS One. 2013;8(8):e71108. https://doi.org/10.1371/journal.pone.0071108.
Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444(7122):1022–3. https://doi.org/10.1038/4441022a.
Larsen N, Vogensen FK, van den Berg FW, Nielsen DS, Andreasen AS, Pedersen BK, et al. Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS One. 2010;5(2):e9085. https://doi.org/10.1371/journal.pone.0009085.
Backhed F, Ding H, Wang T, Hooper LV, Koh GY, Nagy A, et al. The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci U S A. 2004;101(44):15718–23. https://doi.org/10.1073/pnas.0407076101.
Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, et al. Diversity of the human intestinal microbial flora. Science. 2005;308(5728):1635–8. https://doi.org/10.1126/science.1110591.
Turnbaugh PJ, Ley RE, Mahowald MA, Magrini V, Mardis ER, Gordon JI. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444(7122):1027–31. https://doi.org/10.1038/nature05414.
Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE, et al. A core gut microbiome in obese and lean twins. Nature. 2009;457(7228):480–4. https://doi.org/10.1038/nature07540.
Koliada A, Syzenko G, Moseiko V, Budovska L, Puchkov K, Perederiy V, et al. Association between body mass index and Firmicutes/Bacteroidetes ratio in an adult Ukrainian population. BMC Microbiol. 2017;17(1):120. https://doi.org/10.1186/s12866-017-1027-1.
Wang H, Hong T, Li N, Zang B, Wu X. Soluble dietary fiber improves energy homeostasis in obese mice by remodeling the gut microbiota. Biochem Biophys Res Commun. 2018;498(1):146–51. https://doi.org/10.1016/j.bbrc.2018.02.017.
Schwiertz A, Taras D, Schafer K, Beijer S, Bos NA, Donus C, et al. Microbiota and SCFA in lean and overweight healthy subjects. Obesity (Silver Spring). 2010;18(1):190–5. https://doi.org/10.1038/oby.2009.167.
Duncan SH, Lobley GE, Holtrop G, Ince J, Johnstone AM, Louis P, et al. Human colonic microbiota associated with diet, obesity and weight loss. Int J Obes. 2008;32(11):1720–4. https://doi.org/10.1038/ijo.2008.155.
Finucane MM, Sharpton TJ, Laurent TJ, Pollard KS. A taxonomic signature of obesity in the microbiome? Getting to the guts of the matter. PLoS One. 2014;9(1):e84689. https://doi.org/10.1371/journal.pone.0084689.
Wu GD, Chen J, Hoffmann C, Bittinger K, Chen YY, Keilbaugh SA, et al. Linking long-term dietary patterns with gut microbial enterotypes. Science. 2011;334(6052):105–8. https://doi.org/10.1126/science.1208344.
Haro C, Montes-Borrego M, Rangel-Zuniga OA, Alcala-Diaz JF, Gomez-Delgado F, Perez-Martinez P, et al. Two healthy diets modulate gut microbial community improving insulin sensitivity in a human obese population. J Clin Endocrinol Metab. 2016;101(1):233–42. https://doi.org/10.1210/jc.2015-3351.
David LA, Maurice CF, Carmody RN, Gootenberg DB, Button JE, Wolfe BE, et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2014;505(7484):559–63. https://doi.org/10.1038/nature12820.
Flint HJ, Bayer EA, Rincon MT, Lamed R, White BA. Polysaccharide utilization by gut bacteria: potential for new insights from genomic analysis. Nat Rev Microbiol. 2008;6(2):121–31. https://doi.org/10.1038/nrmicro1817.
Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of childhood and adult obesity in the United States, 2011-2012. JAMA. 2014;311(8):806–14. https://doi.org/10.1001/jama.2014.732.
Ng M, Fleming T, Robinson M, Thomson B, Graetz N, Margono C, et al. Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet. 2014;384(9945):766–81. https://doi.org/10.1016/S0140-6736(14)60460-8.
O'Brien MJ, Perez A, Scanlan AB, Alos VA, Whitaker RC, Foster GD, et al. PREVENT-DM comparative effectiveness trial of lifestyle intervention and metformin. Am J Prev Med. 2017;52(6):788–97. https://doi.org/10.1016/j.amepre.2017.01.008.
Glechner A, Keuchel L, Affengruber L, Titscher V, Sommer I, Matyas N, et al. Effects of lifestyle changes on adults with prediabetes: a systematic review and meta-analysis. Prim Care Diabetes. 2018;12:393–408. https://doi.org/10.1016/j.pcd.2018.07.003.
Muniz Pedrogo DA, Jensen MD, Van Dyke CT, Murray JA, Woods JA, Chen J, et al. Gut microbial carbohydrate metabolism hinders weight loss in overweight adults undergoing lifestyle intervention with a volumetric diet. Mayo Clin Proc. 2018;93(8):1104–10. https://doi.org/10.1016/j.mayocp.2018.02.019.
Dao MC, Everard A, Aron-Wisnewsky J, Sokolovska N, Prifti E, Verger EO, et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut. 2016;65(3):426–36. https://doi.org/10.1136/gutjnl-2014-308778.
Liu R, Hong J, Xu X, Feng Q, Zhang D, Gu Y, et al. Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nat Med. 2017;23(7):859–68. https://doi.org/10.1038/nm.4358.
• Anitha M, Reichardt F, Tabatabavakili S, Nezami BG, Chassaing B, Mwangi S, et al. Intestinal dysbiosis contributes to the delayed gastrointestinal transit in high-fat diet fed mice. Cell Mol Gastroenterol Hepatol. 2016;2(3):328–39. https://doi.org/10.1016/j.jcmgh.2015.12.008 The study suggests that intestinal dysbiosis in mice fed high-fat diets contributes to delayed intestinal motility and these changes may be mediated by TLR4-dependant neuronal loss.
Reichardt F, Chassaing B, Nezami BG, Li G, Tabatabavakili S, Mwangi S, et al. Western diet induces colonic nitrergic myenteric neuropathy and dysmotility in mice via saturated fatty acid- and lipopolysaccharide-induced TLR4 signalling. J Physiol. 2017;595(5):1831–46. https://doi.org/10.1113/JP273269.
Brown AJ, Goldsworthy SM, Barnes AA, Eilert MM, Tcheang L, Daniels D, et al. The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J Biol Chem. 2003;278(13):11312–9. https://doi.org/10.1074/jbc.M211609200.
Bergman EN. Energy contributions of volatile fatty acids from the gastrointestinal tract in various species. Physiol Rev. 1990;70(2):567–90. https://doi.org/10.1152/physrev.1990.70.2.567.
Hwang N, Eom T, Gupta SK, Jeong SY, Jeong DY, Kim YS et al. Genes and gut bacteria involved in luminal butyrate reduction caused by diet and loperamide. Genes (Basel). 2017;8(12). doi:https://doi.org/10.3390/genes8120350.
Ge X, Zhao W, Ding C, Tian H, Xu L, Wang H, et al. Potential role of fecal microbiota from patients with slow transit constipation in the regulation of gastrointestinal motility. Sci Rep. 2017;7(1):441. https://doi.org/10.1038/s41598-017-00612-y.
Deng Y, Li M, Mei L, Cong LM, Liu Y, Zhang BB, et al. Manipulation of intestinal dysbiosis by a bacterial mixture ameliorates loperamide-induced constipation in rats. Benefic Microbes. 2018;9(3):453–64. https://doi.org/10.3920/BM2017.0062.
Fukumoto S, Tatewaki M, Yamada T, Fujimiya M, Mantyh C, Voss M, et al. Short-chain fatty acids stimulate colonic transit via intraluminal 5-HT release in rats. Am J Phys Regul Integr Comp Phys. 2003;284(5):R1269–76. https://doi.org/10.1152/ajpregu.00442.2002.
Yano JM, Yu K, Donaldson GP, Shastri GG, Ann P, Ma L, et al. Indigenous bacteria from the gut microbiota regulate host serotonin biosynthesis. Cell. 2015;161(2):264–76. https://doi.org/10.1016/j.cell.2015.02.047.
Tottey W, Feria-Gervasio D, Gaci N, Laillet B, Pujos E, Martin JF, et al. Colonic transit time is a driven force of the gut microbiota composition and metabolism: in vitro evidence. J Neurogastroenterol Motil. 2017;23(1):124–34. https://doi.org/10.5056/jnm16042.
Wang L, Hu L, Yan S, Jiang T, Fang S, Wang G, et al. Effects of different oligosaccharides at various dosages on the composition of gut microbiota and short-chain fatty acids in mice with constipation. Food Funct. 2017;8(5):1966–78. https://doi.org/10.1039/c7fo00031f.
Hoek M, Merks RMH. Emergence of microbial diversity due to cross-feeding interactions in a spatial model of gut microbial metabolism. BMC Syst Biol. 2017;11(1):56. https://doi.org/10.1186/s12918-017-0430-4.
Vandeputte D, Falony G, Vieira-Silva S, Tito RY, Joossens M, Raes J. Stool consistency is strongly associated with gut microbiota richness and composition, enterotypes and bacterial growth rates. Gut. 2016;65(1):57–62. https://doi.org/10.1136/gutjnl-2015-309618.
Gallardo P, Izquierdo M, Vidal RM, Chamorro-Veloso N, Rossello-Mora R, O’Ryan M, et al. Distinctive gut microbiota is associated with diarrheagenic Escherichia coli infections in Chilean children. Front Cell Infect Microbiol. 2017;7:424. https://doi.org/10.3389/fcimb.2017.00424.
Singh P, Teal TK, Marsh TL, Tiedje JM, Mosci R, Jernigan K, et al. Intestinal microbial communities associated with acute enteric infections and disease recovery. Microbiome. 2015;3:45. https://doi.org/10.1186/s40168-015-0109-2.
The HC, Florez de Sessions P, Jie S, Pham Thanh D, Thompson CN, Nguyen Ngoc Minh C, et al. Assessing gut microbiota perturbations during the early phase of infectious diarrhea in Vietnamese children. Gut Microbes. 2018;9(1):38–54. https://doi.org/10.1080/19490976.2017.1361093.
Liu SX, Li YH, Dai WK, Li XS, Qiu CZ, Ruan ML, et al. Fecal microbiota transplantation induces remission of infantile allergic colitis through gut microbiota re-establishment. World J Gastroenterol. 2017;23(48):8570–81. https://doi.org/10.3748/wjg.v23.i48.8570.
Johnsen PH, Hilpusch F, Cavanagh JP, Leikanger IS, Kolstad C, Valle PC, et al. Faecal microbiota transplantation versus placebo for moderate-to-severe irritable bowel syndrome: a double-blind, randomised, placebo-controlled, parallel-group, single-centre trial. Lancet Gastroenterol Hepatol. 2018;3(1):17–24. https://doi.org/10.1016/S2468-1253(17)30338-2.
Ley RE, Backhed F, Turnbaugh P, Lozupone CA, Knight RD, Gordon JI. Obesity alters gut microbial ecology. Proc Natl Acad Sci U S A. 2005;102(31):11070–5. https://doi.org/10.1073/pnas.0504978102.
Zou J, Chassaing B, Singh V, Pellizzon M, Ricci M, Fythe MD, et al. Fiber-mediated nourishment of gut microbiota protects against diet-induced obesity by restoring IL-22-mediated colonic health. Cell Host Microbe. 2018;23(1):41–53 e4. https://doi.org/10.1016/j.chom.2017.11.003.
Gao Z, Yin J, Zhang J, Ward RE, Martin RJ, Lefevre M, et al. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes. 2009;58(7):1509–17. https://doi.org/10.2337/db08-1637.
Todesco T, Rao AV, Bosello O, Jenkins DJ. Propionate lowers blood glucose and alters lipid metabolism in healthy subjects. Am J Clin Nutr. 1991;54(5):860–5.
Lindstrom J, Peltonen M, Eriksson JG, Louheranta A, Fogelholm M, Uusitupa M, et al. High-fibre, low-fat diet predicts long-term weight loss and decreased type 2 diabetes risk: the Finnish Diabetes Prevention Study. Diabetologia. 2006;49(5):912–20. https://doi.org/10.1007/s00125-006-0198-3.
Holst JJ. The physiology of glucagon-like peptide 1. Physiol Rev. 2007;87(4):1409–39. https://doi.org/10.1152/physrev.00034.2006.
Lin HV, Frassetto A, Kowalik EJ Jr, Nawrocki AR, Lu MM, Kosinski JR, et al. Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS One. 2012;7(4):e35240. https://doi.org/10.1371/journal.pone.0035240.
Chambers ES, Viardot A, Psichas A, Morrison DJ, Murphy KG, Zac-Varghese SE, et al. Effects of targeted delivery of propionate to the human colon on appetite regulation, body weight maintenance and adiposity in overweight adults. Gut. 2015;64(11):1744–54. https://doi.org/10.1136/gutjnl-2014-307913.
Grasset E, Puel A, Charpentier J, Collet X, Christensen JE, Terce F, et al. A specific gut microbiota dysbiosis of type 2 diabetic mice induces GLP-1 resistance through an enteric NO-dependent and gut-brain axis mechanism. Cell Metab. 2017;26(1):278. https://doi.org/10.1016/j.cmet.2017.06.003.
• Zhao L, Zhang F, Ding X, Wu G, Lam YY, Wang X, et al. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science. 2018;359(6380):1151–6. https://doi.org/10.1126/science.aao5774 This was a randomized clinical study showing that high fiber intake promoted increased production of short-chain fatty acids and improved glycemic control in patients with type 2 diabetes mellitus.
Chassaing B, Raja SM, Lewis JD, Srinivasan S, Gewirtz AT. Colonic microbiota encroachment correlates with dysglycemia in humans. Cell Mol Gastroenterol Hepatol. 2017;4(2):205–21. https://doi.org/10.1016/j.jcmgh.2017.04.001.
Fabbiano S, Suarez-Zamorano N, Chevalier C, Lazarevic V, Kieser S, Rigo D, et al. Functional gut microbiota remodeling contributes to the caloric restriction-induced metabolic improvements. Cell Metab. 2018;28:907–921.e7. https://doi.org/10.1016/j.cmet.2018.08.005.
Caputi V, Marsilio I, Filpa V, Cerantola S, Orso G, Bistoletti M, et al. Antibiotic-induced dysbiosis of the microbiota impairs gut neuromuscular function in juvenile mice. Br J Pharmacol. 2017;174(20):3623–39. https://doi.org/10.1111/bph.13965.
Iwasaki A, Medzhitov R. Toll-like receptor control of the adaptive immune responses. Nat Immunol. 2004;5(10):987–95. https://doi.org/10.1038/ni1112.
Brun P, Giron MC, Qesari M, Porzionato A, Caputi V, Zoppellaro C, et al. Toll-like receptor 2 regulates intestinal inflammation by controlling integrity of the enteric nervous system. Gastroenterology. 2013;145(6):1323–33. https://doi.org/10.1053/j.gastro.2013.08.047.
Johnson S, Gerding DN. Fecal fixation: fecal microbiota transplantation for Clostridium difficile infection. Clin Infect Dis. 2017;64(3):272–4. https://doi.org/10.1093/cid/ciw735.
Bunnett NW. Neuro-humoral signalling by bile acids and the TGR5 receptor in the gastrointestinal tract. J Physiol. 2014;592(14):2943–50. https://doi.org/10.1113/jphysiol.2014.271155.
Utsumi D, Matsumoto K, Amagase K, Horie S, Kato S. 5-HT3 receptors promote colonic inflammation via activation of substance P/neurokinin-1 receptors in dextran sulphate sodium-induced murine colitis. Br J Pharmacol. 2016;173(11):1835–49. https://doi.org/10.1111/bph.13482.
Zheng J, Yuan X, Zhang C, Jia P, Jiao S, Zhao X, et al. N-Acetylcysteine alleviates gut dysbiosis and glucose metabolic disorder in high-fat diet-fed mice. J Diabetes. 2018;11:32–45. https://doi.org/10.1111/1753-0407.12795.
Anhe FF, Nachbar RT, Varin TV, Trottier J, Dudonne S, Le Barz M, et al. Treatment with camu camu (Myrciaria dubia) prevents obesity by altering the gut microbiota and increasing energy expenditure in diet-induced obese mice. Gut. 2018;68:453–64. https://doi.org/10.1136/gutjnl-2017-315565.
Wu TR, Lin CS, Chang CJ, Lin TL, Martel J, Ko YF, et al. Gut commensal Parabacteroides goldsteinii plays a predominant role in the anti-obesity effects of polysaccharides isolated from Hirsutella sinensis. Gut. 2018;68:248–62. https://doi.org/10.1136/gutjnl-2017-315458.
Wang J, Qin C, He T, Qiu K, Sun W, Zhang X, et al. Alfalfa-containing diets alter luminal microbiota structure and short chain fatty acid sensing in the caecal mucosa of pigs. J Anim Sci Biotechnol. 2018;9:11. https://doi.org/10.1186/s40104-017-0216-y.
Karl JP, Meydani M, Barnett JB, Vanegas SM, Goldin B, Kane A, et al. Substituting whole grains for refined grains in a 6-wk randomized trial favorably affects energy-balance metrics in healthy men and postmenopausal women. Am J Clin Nutr. 2017;105(3):589–99. https://doi.org/10.3945/ajcn.116.139683.
Walker AW, Ince J, Duncan SH, Webster LM, Holtrop G, Ze X, et al. Dominant and diet-responsive groups of bacteria within the human colonic microbiota. ISME J. 2011;5(2):220–30. https://doi.org/10.1038/ismej.2010.118.
• Staudacher HM, MCE L, Farquharson FM, Louis P, Fava F, Franciosi E, et al. A diet low in FODMAPs reduces symptoms in patients with irritable bowel syndrome and a probiotic restores Bifidobacterium species: a randomized controlled trial. Gastroenterology. 2017;153(4):936–47. https://doi.org/10.1053/j.gastro.2017.06.010 This study demonstrated that a diet low in fermentible carbohydrates was associated with improvement in gastrointestinal symptoms among patients with irritable bowel syndrome and correlated with lower abundance of Bifidobacterium species.
Eswaran SL, Chey WD, Han-Markey T, Ball S, Jackson K. A randomized controlled trial comparing the low FODMAP diet vs. modified NICE guidelines in US adults with IBS-D. Am J Gastroenterol. 2016;111(12):1824–32. https://doi.org/10.1038/ajg.2016.434.
Marsh A, Eslick EM, Eslick GD. Does a diet low in FODMAPs reduce symptoms associated with functional gastrointestinal disorders? A comprehensive systematic review and meta-analysis. Eur J Nutr. 2016;55(3):897–906. https://doi.org/10.1007/s00394-015-0922-1.
van Can J, Sloth B, Jensen CB, Flint A, Blaak EE, Saris WH. Effects of the once-daily GLP-1 analog liraglutide on gastric emptying, glycemic parameters, appetite and energy metabolism in obese, non-diabetic adults. Int J Obes. 2014;38(6):784–93. https://doi.org/10.1038/ijo.2013.162.
Wang L, Li P, Tang Z, Yan X, Feng B. Structural modulation of the gut microbiota and the relationship with body weight: compared evaluation of liraglutide and saxagliptin treatment. Sci Rep. 2016;6:33251. https://doi.org/10.1038/srep33251.
Zhao L, Chen Y, Xia F, Abudukerimu B, Zhang W, Guo Y, et al. A Glucagon-like peptide-1 receptor agonist lowers weight by modulating the structure of gut microbiota. Front Endocrinol (Lausanne). 2018;9:233. https://doi.org/10.3389/fendo.2018.00233.
Zheng H, Chen M, Li Y, Wang Y, Wei L, Liao Z, et al. Modulation of gut microbiome composition and function in experimental colitis treated with sulfasalazine. Front Microbiol. 2017;8:1703. https://doi.org/10.3389/fmicb.2017.01703.
Kang DW, DiBaise JK, Ilhan ZE, Crowell MD, Rideout JR, Caporaso JG, et al. Gut microbial and short-chain fatty acid profiles in adults with chronic constipation before and after treatment with lubiprostone. Anaerobe. 2015;33:33–41. https://doi.org/10.1016/j.anaerobe.2015.01.005.
Fodor AA, Pimentel M, Chey WD, Lembo A, Golden PL, Israel RJ, et al. Rifaximin is associated with modest, transient decreases in multiple taxa in the gut microbiota of patients with diarrhoea-predominant irritable bowel syndrome. Gut Microbes. 2018;10:1–28. https://doi.org/10.1080/19490976.2018.1460013.
Chassaing B, Miles-Brown J, Pellizzon M, Ulman E, Ricci M, Zhang L, et al. Lack of soluble fiber drives diet-induced adiposity in mice. Am J Physiol Gastrointest Liver Physiol. 2015;309(7):G528–41. https://doi.org/10.1152/ajpgi.00172.2015.
Salazar N, Dewulf EM, Neyrinck AM, Bindels LB, Cani PD, Mahillon J, et al. Inulin-type fructans modulate intestinal Bifidobacterium species populations and decrease fecal short-chain fatty acids in obese women. Clin Nutr. 2015;34(3):501–7. https://doi.org/10.1016/j.clnu.2014.06.001.
Alexander C, Cross TL, Devendran S, Neumer F, Theis S, Ridlon JM, et al. Effects of prebiotic inulin-type fructans on blood metabolite and hormone concentrations and faecal microbiota and metabolites in overweight dogs. Br J Nutr. 2018;120(6):711–20. https://doi.org/10.1017/S0007114518001952.
Roshanravan N, Mahdavi R, Alizadeh E, Jafarabadi MA, Hedayati M, Ghavami A, et al. Effect of butyrate and inulin supplementation on glycemic status, lipid profile and glucagon-like peptide 1 level in patients with type 2 diabetes: a randomized double-blind, placebo-controlled trial. Horm Metab Res. 2017;49(11):886–91. https://doi.org/10.1055/s-0043-119089.
Rahat-Rozenbloom S, Fernandes J, Cheng J, Wolever TMS. Acute increases in serum colonic short-chain fatty acids elicited by inulin do not increase GLP-1 or PYY responses but may reduce ghrelin in lean and overweight humans. Eur J Clin Nutr. 2017;71(8):953–8. https://doi.org/10.1038/ejcn.2016.249.
Guess ND, Dornhorst A, Oliver N, Frost GS. A randomised crossover trial: the effect of inulin on glucose homeostasis in subtypes of prediabetes. Ann Nutr Metab. 2016;68(1):26–34. https://doi.org/10.1159/000441626.
Miyamoto J, Watanabe K, Taira S, Kasubuchi M, Li X, Irie J, et al. Barley beta-glucan improves metabolic condition via short-chain fatty acids produced by gut microbial fermentation in high fat diet fed mice. PLoS One. 2018;13(4):e0196579. https://doi.org/10.1371/journal.pone.0196579.
Prykhodko O, Sandberg J, Burleigh S, Bjorck I, Nilsson A, Fak Hallenius F. Impact of rye kernel-based evening meal on microbiota composition of young healthy lean volunteers with an emphasis on their hormonal and appetite regulations, and blood levels of brain-derived neurotrophic factor. Front Nutr. 2018;5:45. https://doi.org/10.3389/fnut.2018.00045.
van der Beek CM, Canfora EE, Kip AM, Gorissen SHM, Olde Damink SWM, van Eijk HM, et al. The prebiotic inulin improves substrate metabolism and promotes short-chain fatty acid production in overweight to obese men. Metabolism. 2018;87:25–35. https://doi.org/10.1016/j.metabol.2018.06.009.
Lee I, Shi L, Webb DL, Hellstrom PM, Riserus U, Landberg R. Effects of whole-grain rye porridge with added inulin and wheat gluten on appetite, gut fermentation and postprandial glucose metabolism: a randomised, cross-over, breakfast study. Br J Nutr. 2016;116(12):2139–49. https://doi.org/10.1017/S0007114516004153.
Simon MC, Strassburger K, Nowotny B, Kolb H, Nowotny P, Burkart V, et al. Intake of Lactobacillus reuteri improves incretin and insulin secretion in glucose-tolerant humans: a proof of concept. Diabetes Care. 2015;38(10):1827–34. https://doi.org/10.2337/dc14-2690.
Stenman LK, Lehtinen MJ, Meland N, Christensen JE, Yeung N, Saarinen MT, et al. Probiotic with or without fiber controls body fat mass, associated with serum zonulin, in overweight and obese adults-randomized controlled trial. EBioMedicine. 2016;13:190–200. https://doi.org/10.1016/j.ebiom.2016.10.036.
Blatchford P, Stoklosinski H, Eady S, Wallace A, Butts C, Gearry R, et al. Consumption of kiwifruit capsules increases Faecalibacterium prausnitzii abundance in functionally constipated individuals: a randomised controlled human trial. J Nutr Sci. 2017;6:e52. https://doi.org/10.1017/jns.2017.52.
Chen Y, Xiao S, Gong Z, Zhu X, Yang Q, Li Y, et al. Wuji Wan formula ameliorates diarrhea and disordered colonic motility in post-inflammation irritable bowel syndrome rats by modulating the gut microbiota. Front Microbiol. 2017;8:2307. https://doi.org/10.3389/fmicb.2017.02307.
Jeong D, Kim DH, Kang IB, Kim H, Song KY, Kim HS, et al. Modulation of gut microbiota and increase in fecal water content in mice induced by administration of Lactobacillus kefiranofaciens DN1. Food Funct. 2017;8(2):680–6. https://doi.org/10.1039/c6fo01559j.
Kim SE, Choi SC, Park KS, Park MI, Shin JE, Lee TH, et al. Change of fecal flora and effectiveness of the short-term VSL#3 probiotic treatment in patients with functional constipation. J Neurogastroenterol Motil. 2015;21(1):111–20. https://doi.org/10.5056/jnm14048.
Hatanaka M, Yamamoto K, Suzuki N, Iio S, Takara T, Morita H, et al. Effect of Bacillus subtilis C-3102 on loose stools in healthy volunteers. Benefic Microbes. 2018;9(3):357–65. https://doi.org/10.3920/BM2017.0103.
Ford AC, Quigley EM, Lacy BE, Lembo AJ, Saito YA, Schiller LR, et al. Efficacy of prebiotics, probiotics, and synbiotics in irritable bowel syndrome and chronic idiopathic constipation: systematic review and meta-analysis. Am J Gastroenterol. 2014;109(10):1547–61; quiz 6, 62. https://doi.org/10.1038/ajg.2014.202.
Tremaroli V, Karlsson F, Werling M, Stahlman M, Kovatcheva-Datchary P, Olbers T, et al. Roux-en-Y gastric bypass and vertical banded gastroplasty induce long-term changes on the human gut microbiome contributing to fat mass regulation. Cell Metab. 2015;22(2):228–38. https://doi.org/10.1016/j.cmet.2015.07.009.
Palleja A, Kashani A, Allin KH, Nielsen T, Zhang C, Li Y, et al. Roux-en-Y gastric bypass surgery of morbidly obese patients induces swift and persistent changes of the individual gut microbiota. Genome Med. 2016;8(1):67. https://doi.org/10.1186/s13073-016-0312-1.
Liou AP, Paziuk M, Luevano JM Jr, Machineni S, Turnbaugh PJ, Kaplan LM. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci Transl Med. 2013;5(178):178ra41. https://doi.org/10.1126/scitranslmed.3005687.
Cortez RV, Petry T, Caravatto P, Pessoa R, Sanabani SS, Martinez MB, et al. Shifts in intestinal microbiota after duodenal exclusion favor glycemic control and weight loss: a randomized controlled trial. Surg Obes Relat Dis. 2018;14:1748–54. https://doi.org/10.1016/j.soard.2018.07.021.
Murphy R, Tsai P, Jullig M, Liu A, Plank L, Booth M. Differential changes in gut microbiota after gastric bypass and sleeve gastrectomy bariatric surgery vary according to diabetes remission. Obes Surg. 2017;27(4):917–25. https://doi.org/10.1007/s11695-016-2399-2.
Fernandes R, Beserra BT, Mocellin MC, Kuntz MG, da Rosa JS, de Miranda RC, et al. Effects of prebiotic and synbiotic supplementation on inflammatory markers and anthropometric indices after Roux-en-Y gastric bypass: a randomized, triple-blind, placebo-controlled pilot study. J Clin Gastroenterol. 2016;50(3):208–17. https://doi.org/10.1097/MCG.0000000000000328.
Vrieze A, Van Nood E, Holleman F, Salojarvi J, Kootte RS, Bartelsman JF, et al. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology. 2012;143(4):913–6 e7. https://doi.org/10.1053/j.gastro.2012.06.031.
Funding
SS is funded by NIH grant number NIH RO1 DK080684 and a VA Merit Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Maya Fayfman, Kristen Flint, and Shanthi Srinivasan declare no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Neurogastroenterology and Motility Disorders of the Gastrointestinal Tract
Rights and permissions
About this article
Cite this article
Fayfman, M., Flint, K. & Srinivasan, S. Obesity, Motility, Diet, and Intestinal Microbiota—Connecting the Dots. Curr Gastroenterol Rep 21, 15 (2019). https://doi.org/10.1007/s11894-019-0680-y
Published:
DOI: https://doi.org/10.1007/s11894-019-0680-y