Abstract
A strong case for the deregulation of epigenetic chromatin modifications in the development and progression of various chronic complications of diabetes has emerged from recent experimental observations. Clinical trials of type 1 and type 2 diabetes patients highlight the importance of early and intensive treatment and the prolonged damage of hyperglycemia on organs such as the kidney. The functional relationship between the regulation of chromatin architecture and persistent gene expression changes conferred by prior hyperglycemia represents an important avenue of investigation for explaining diabetic nephropathy. While several studies implicate epigenetic changes at the chromatin template in the deregulated gene expression associated with diabetic nephropathy, the molecular determinants of metabolic memory in renal cells remain poorly understood. There is now strong evidence from experimental animals and cell culture of persistent glucose-driven changes in vascular endothelial gene expression that may also have relevance for the microvasculature of the kidney. Exploration of epigenetic mechanisms underlying the hyperglycemic cue mediating persistent transcriptional changes in renal cells holds novel therapeutic potential for diabetic nephropathy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It is well documented that type 1 and type 2 diabetic patients experience accelerated rates of clinically defined macrovascular complications such as atherosclerosis, cardiovascular disease, and stroke, as well as microvascular complications of the retina, nervous system, and kidney [1, 2]. Prolonged exposure to the insults of the diabetic milieu is associated with deregulated gene expression and key intracellular signaling pathways that drive deleterious changes across multiple organ systems. The association of chronic hyperglycemia with the development and progression of diabetic complications is striking. Despite significant clinical efforts to control blood glucose, chronic diseases, including diabetic nephropathy, are a major cause of mortality in individuals with diabetes [3]. The increasingly noxious conditions of the hyperglycemic kidney promote numerous pathophysiological changes in gene expression that drive structural alterations associated with diabetic nephropathy [4]. Understanding the molecular mechanisms that intersect glucose-mediated signaling cues with gene expression represents a major opportunity and challenge for the development of novel therapeutic approaches for diabetic kidney disease.
Epigenetic Regulation Unites the Diabetic Milieu and Vascular Complications
Significant progress in the field of chromatin biology has begun to distill the molecular mechanisms of transcriptional changes observed in human diseases [5–10], revealing potential for novel avenues of therapy [11]. Moreover, epigenetic processes link cellular environment to genomic regulation, and there is a compelling case for such markedly flexible interactions as a driving force behind the occurrence and progression of diabetic vascular disease [12•]. Chromatin modifications are integral elements of genomic structural organization and transcriptional regulation. Mounting evidence directly links the modified chromatin landscape underlying transcriptional changes with diabetes and its complications.
The concept of metabolic or glycemic memory was popularized by seminal epidemiological studies of type 1 diabetes, where despite stringent long-term glycemic control, initial periods of suboptimal blood glucose continued to be a risk factor for chronic vascular complications, including nephropathy [13–17]. Pioneering studies of experimental animal models had suggested memories of prior hyperglycemia [18, 19]. More than 2 decades following these initial observations, there is now considerable appreciation for the role of sustained gene-regulating events conferred by hyperglycemia on the development and persistence of the vascular complications of diabetes. Recent insights from cultured vascular cells identify the importance of posttranslational modification of histone tails in the persistent inflammatory response to glycemic variability [20, 21••, 22]. These observations of glucose-mediated chromatin and transcriptional changes in vascular cells of aortic and retinal endothelial origin indicate the possibility of similar mechanisms operating in the renal microvasculature.
It is anticipated that a better understanding of the molecular events mediating chromatin modification and transcriptional regulation will help to identify novel approaches to inhibit, attenuate, or reverse the persistent deleterious consequences of diabetes in the kidney. In this review, we explore associations between regulatory modification of the chromatin template and transcriptional changes that drive the phenotype of diabetic kidney disease. Specific examples highlight the epigenetic component of nephropathy in cultured renal cells and human samples. Drawing from observations of vascular models of hyperglycemic variability, we discuss potential molecular mechanisms underlying the persistent development of diabetic nephropathy beyond restoration of euglycemia.
The Diabetic Milieu of the Kidney
Diabetes is the leading cause of nephropathy and end stage renal disease [23]. Characterized clinically by hypertension, proteinuria, and deterioration in glomerular filtration [24], the development of diabetic nephropathy is largely driven by activation of key metabolic and hemodynamic pathways by hyperglycemia. In particular, advanced glycation end-product formation, overproduction of reactive oxygen species (ROS), increased polyol and hexosamine pathway flux link high circulating glucose concentrations to declining kidney function. Systemic and glomerular hypertension through amplification of the vasoactive renin-angiotensin system, signaling through TGF-β and other growth factors, as well as activation of protein kinase C, p38 MAPK, and inflammatory pathways, are also associated with diabetic nephropathy [3, 25–30]. These intracellular signaling events promote key structural alterations including mesangial matrix expansion, podocyte injury, and glomerular basement membrane thickening, as well as changes associated with later stages of the disease, including epithelial-to-mesenchymal transition, fibrosis, and extracellular matrix (ECM) accumulation leading to glomerulosclerosis [31–34]. The reorganization of glomerular architecture through pathological signaling is thought to be responsible for the decrease in kidney function [35, 36]. Diabetes-specific changes in gene expression associated with structural adaptation of the kidney have accordingly been observed for various renal components, including podocytes, mesangial, and tubular epithelial cells [37–39].
A growing body of evidence strongly implicates altered expression of transcription factors, adhesion molecules, chemokines, and growth factors in glomerular structural remodeling. Recent gene array analysis of glomeruli isolated from diabetic and control rats demonstrated widespread differential expression of genes associated with ECM interactions, TGF-β, and renin-angiotensin pathways [40•]. These observations parallel earlier findings from gene expression profiling of kidneys of type 1 [41] and glomerular endothelial cells isolated from type 2 [42] diabetic mouse models that revealed numerous differentially expressed genes with both protective and pathogenic roles in disease progression. The pathways identified include cell adhesion, PKC, TGF-β and angiotensin signaling, extracellular matrix deposition, growth, and cell survival [41, 42]. Multiple transcriptional changes identified by massive parallel mRNA sequencing following TGF-β stimulation of tubular epithelial cells were also evident in renal biopsies from patients with diabetic nephropathy [43•]. Independent array analyses of microdissected glomerular and tubulointerstitial tissue from patients with diabetic kidney disease revealed differential expression of immunological and inflammatory pathways in both renal components [44, 45]. Further characterization of disease-associated changes by deep sequencing and transcriptome profiling will provide a better definition of the disease-associated gene expression profiles of individual renal cell types.
Persistent Glycemic Memories
Effective glycemic control is recognized as an integral component in the clinical treatment of diabetes. Clinical studies addressing the potential for intensive blood-glucose-lowering strategies to attenuate diabetic vascular complications have confirmed the clinical relevance of strict metabolic regulation to nephropathy and other diabetic complications. Relative to conventional treatment, reduced progression of kidney disease following intensive regimens of insulin therapy was observed in several small-scale studies of type 1 diabetes. Clinical trials such as the Steno study [46, 47], the KROC study [48, 49], the Oslo study [50], and the Stockholm Diabetes Intervention Study [51] reported significant attenuation of markers of diabetic nephropathy, including urinary albumin excretion.
Important observations of the persistent benefit of early and aggressive optimization of blood glucose were revealed by large-scale multicenter clinical trials. The Diabetes Control and Complications Trial compared conventional and intensive insulin therapy of type 1 diabetes. Over a median period of 6.5 years, the risk for microalbuminuria and overt nephropathy was decreased by intensive glycemic control [13, 14]. The Epidemiology of Diabetes Intervention and Complications observational follow-up study of the same cohort examined the long-term benefits of intensive therapy following normalization of blood glucose and subsequently glycated hemoglobin across both groups for more than a decade [15–17]. Throughout this period, consistent divergence in indices of vascular complications, including nephropathy, began to emerge across groups formerly assigned to conventional and intensive treatment. Specifically, previous periods of inadequate blood glucose control significantly increased incidences of microalbuminuria and of the risk of renal complications, revealing the persistence of cellular memory despite intensive insulin therapy.
The significance of competent glycemic control to the development and progression of nephropathy in type 2 diabetes was illustrated by the Action in Diabetes and Vascular disease: Preterax and Diamicron-MR Controlled Evaluation (ADVANCE) study [52]. Of particular significance, there was a 21 % relative reduction in progression of albuminuria and nephropathy when insulin therapy was intensified late in the disease [53]. Similarly, the large-scale United Kingdom Prospective Diabetes Study correlated early intensive glycemic control with abrogated occurrence and progression of diabetic nephropathy in patients with type 2 diabetes. Observational follow-up over the subsequent decade demonstrated the sustained benefit of glucose control with regard to renal and retinal events well beyond a period of prior intensive therapy [54, 55]. The smaller Steno-2 clinical trial examined the effects of intensive multifactorial behavior modification and therapy for hypertension, dyslipidemia, and hyperglycemia in a cohort of patients with type 2 diabetes and microalbuminuria. Equivalent and significantly reduced risk for the development of nephropathy was observed after 4 and 8 years of intensified treatment, further emphasizing the benefit of early intervention [56, 57].
The consistency across these studies is striking: that early glycemic control significantly delays the onset of diabetic nephropathy in both type 1 and type 2 diabetes. Unraveling the mechanisms driving these key findings is paramount to a better understanding of the persistent deleterious effects of prior hyperglycemia in the diabetic kidney. In consideration of the changes to cellular signaling characteristic of diabetic nephropathy, these observations now indicate that the persistent gene expression changes confer the disease phenotype. The additional layers of dynamic transcriptional control conferred by epigenetic regulatory mechanisms [58] functionally associate the deleterious environmental signaling of the diabetic kidney with changes in gene expression that may be sustained despite glycemic correction.
Chromatin Modifications Underlying Transcriptional Changes
The centrality of epigenetic regulation to transcription-driven cellular processes is unequivocal. The control of gene expression is mediated by the accessibility of the dynamic chromatin polymer of DNA and histone proteins to transcription factors and core regulatory machinery [59]. The eukaryotic genome is distinguished by modifications at the chromatin template that instruct local structural reorganization and transcriptional expression. Recent experimental focus on the chromatinization of specific environmental cues has revealed a role for epigenetic events in sensitizing transcriptional responses to external stimuli. Specific chromatin signatures and transcriptional responses mediated by the effects of chronic exposure to the diabetic milieu indicate critical epigenetic events that mediate signaling pathways relevant to diabetic vascular disease [60]. While strongly implicated in other complications of diabetes, the association of histone modification patterns to gene expression changes in diabetic nephropathy has only recently emerged. The structural and transcriptional changes observed in the diabetic kidney owing to a microvasculature highly vulnerable to the increasingly hypertensive and inflammatory local environment provides strong candidature for histone modifications and DNA methylation changes [12•, 61]. The functional relationship between the regulation of chromatin architecture and altered signaling pathways represents an important axis for explaining diabetic nephropathy.
Covalent methylation of DNA primarily at CG dinucleotides leading to stable gene silencing and the recruitment of remodeling complexes is extensively characterized in various human diseases [62–64]. The importance of this robust modification to the development and persistence of diabetes and associated complications is evidenced in studies of cell models and in preclinical as well as human studies [65]. Although the role of DNA methylation in diabetic kidney gene expression remains poorly characterized, recent studies have shown that changes to the methylation status of key genes are associated with diabetic nephropathy [66, 67]. For instance, DNA methylation profiling of a small cohort of patients with type 1 diabetes revealed changes at CG loci associated with the disease. Moreover, the duration of diabetes prior to the onset of nephropathy correlated to CG methylation at those genes [67].
Histone amino tails are extensively modified posttranslation by the enzymatic activity of a growing list of chromatin regulators. Modifications promote electrostatic charge disruption and create high-affinity sites for specific recruitment of chromatin remodeling complexes, as well as the functional exchange between repressed and active states of gene expression [68, 69]. The interconnectivity of this highly regulated network of enzymes, modifications, and constituent protein complexes has driven the proposition of a regulatory histone code that significantly extends the information potential of the genome [70]. The vast majority of modifications localize to H3 and H4 histone tails, although the characterized list of chemical moieties assigned to other histones is expanding [71, 72]. Prominent modifications include transcriptionally permissive lysine acetylation, as well as methylation of lysine and arginine residues, where the association with transcriptional repression or activation is dependent on the position of the residue within the histone tail and the degree of modification. In addition, lysine residues can be sumoylated or ubiquitinated, and serine and tyrosine side-chains phosphorylated [73].
Numerous studies strongly implicate histone-modifying responses to environmental cues that drive gene expression changes associated with diabetes. Increasingly, histone modifications are investigated in the context of diabetic nephropathy. Analysis of experimental models of advanced glomerulosclerosis in diabetic mice revealed striking global enrichment of H3 histones acetylated at K9 and K27, dimethylated at K4, and phosphorylated at S10 [74]. By contrast, a separate study reported decreased acetylation and phosphorylation of H3 histones in the kidneys of diabetic rats. Indeed, the method of western blot analysis of acidic preparations of histones employed by these studies lacks the resolution to detect enrichment patterns at discrete loci necessary for association to specific gene activity [75]. To this end, immunoprecipitation of soluble chromatin from high-glucose and TGF-β-stimulated rat mesangial cells revealed enrichment of H3 methylated at K4 and depletion of the repressive K9 methylation mark at several profibrotic promoters. Interestingly, specific activation of Col1a1 and CTGF was found to be dependent on K4 mono-methylation of H3 histones (H3K4m1) by the Set7 lysine methyltransferase [76•].
These observations underscore the need to characterize chromatin modifications associated with diabetic nephropathy in order to better understand the disease process. Histone modifications represent a potential interface for diabetic transcriptional changes and chromatin architecture. Moreover, recent experimental evidence implicates histone modifications in chromatinization of persistent gene expression patterns that could explain the continued deleterious effects of previous hyperglycemia described by clinical studies [60]. While examination of metabolic memory in cells of renal origin is indispensable to such a connection, few studies have directly investigated this phenomenon in the kidney. A potential role for transcriptional memory can be inferred from animal and cell culture studies of similar models of glucose-mediated vascular injury.
Experimental Models of Vascular Memory
Primary insights into metabolic memory were first communicated following observations from several important animal studies. Of particular relevance in the context of this review, streptozotocin-induced diabetes in rats with glomerular basement material accumulation was shown to be irreversible despite correction of blood glucose by islet cell transplantation [77]. A comparable approach revealed a pattern of perpetually elevated fibronectin expression in renal cortices despite reinstitution of euglycemia [78]. Similar observations were described for experiments examining the progression of diabetic retinopathy in rodent [19] and canine [18] models. More recently, insulin treatment of rats at the onset of diabetes attenuated the glomerular hypertrophy, mesangial matrix expansion, and renal p38 MAPK pathway activation observed for nontreated diabetic animals and animals that received insulin therapy 8 weeks after confirmation of diabetes [79].
Observations of cultured vascular cells illustrate how critical gene-activating chromatin modifications may prime future transcriptional and metabolic profiles. To this end, stimulation with high glucose markedly increased fibronectin and collagen expression in primary vascular endothelial cells that remained elevated through multiple generations of culture in normal glucose. Significantly, this study represents the first postulation of sustained glucose-mediated changes at the chromatin template underlying transcription [78]. More recently, glucose-driven nuclear localization and promoter-specific modifications associate the Set7 methyltransferase with transcriptional responses to glycemic signaling in the vasculature [80]. Strikingly, specific Set7-mediated H3K4m1 enrichment paralleling transcriptional activation of the RELA promoter mediated by high glucose continued beyond reinstitution of euglycemia in vascular endothelial models of glycemic variability. This gene encodes the p65 subunit of NFκB, and transactivation of targets downstream of this transcription factor further associate hyperglycemia with inflammatory pathway activation [20, 21••]. A critical observation describing the inverse correlation of persistent glucose-mediated depletion of the repressive methylation of K9 on RELA H3 histones and enrichment of the lysine-specific demethylase 1 further substantiates chromatinization of sustained regulatory changes induced by transient hyperglycemia and identifies a cooperative program of methyl writing and erasure [21••]. In accordance with these observations, prolonged activation of NFκB pathways was observed in the renal cortices of db/db mice [81]. The complex transcriptional modulation conferring memory undoubtedly necessitates further uncharacterized functional roles for histone-modifying enzymes and other chromatin regulators. For instance, non-histone methylation by Set7 potentially represents additional mechanisms of transcriptional memory [82]. In addition, recent genetic polymorphisms of genes encoding histone methyltransferases revealed novel associations to diabetic vascular disease [83].
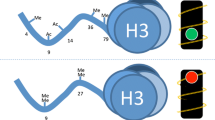
Hyperglycemia-driven ROS overproduction is strongly linked to the development and persistence of diabetic vascular complications and, importantly, to changes in the diabetic kidney [84]. Overexpression of genes (SOD2 and UCP1) that prevent ROS accumulation abolished transcriptional activation of hyperglycemic markers in vascular cells, including RELA [21••, 22]. Interestingly, the phenomenon of glycemic memory is now known to extend to SOD2 in cell culture, also by changes to histone methylation. Specifically, high glucose-mediated trimethylation at K20 of H3 histones and concomitant down-regulation of SOD2 persisted despite subsequent culture under normal glucose conditions [85•]. This model of vascular glycemic memory is schematized in Fig. 1.
The persistent influence of hyperglycemia on the vasculature. Overproduction of vascular endothelial reactive oxygen species (ROS) downstream of hyperglycemic exposure resulting from SOD2 transcriptional repression and ROS-promoted transcriptional activation of RELA by Set7 are sustained through culture under physiological glycemic conditions by persistent changes in histone methylation. Overexpression of SOD2 or specific knockdown of Set7 attenuated the hyperglycemia-induced activation of RELA and inflammatory gene expression
Finally, mesangial epithelial cells have been noted to share numerous structural and function characteristics with smooth muscle cells (SMCs), and studies of transcriptional memory may be applicable across these cell types [74, 86]. To this end, ex vivo culture of SMCs from diabetic db/db mice revealed a persistent inflammatory and atherogenic phenotype for up to 8 weeks, indicating that the phenomenon of memory may not be restricted to endothelial components of the vasculature [87].
Conclusions and Future Considerations for Past Memories
Despite current treatment modalities aimed at blood pressure and glycemic control, an alarming proportion of diabetic patients continue to develop nephropathy often leading to renal failure requiring dialysis and/or transplantation [88, 89]. Clinical studies have shown that intensive glycemic control at early stages of the disease is critical, that the development of diabetic microvascular complications is gradual, and that the lasting effects of hyperglycemic events largely drive the disease process. Until now, most studies have relied on microarray approaches or analysis of discrete loci, and additional investigations are required to definitively connect changes in epigenetic chromatin regulation with the pathological gene expression and signaling pathway activation associated with diabetic nephropathy. The characterization of mechanisms underlying these observations is paramount to a better understanding of diabetic nephropathy and the development of novel and effective treatments that could be applicable to both type 1 and type 2 diabetes.
Studies in cell culture demonstrate the importance of chromatin modifications to transcriptional changes initiated by glucose in the vasculature. Delineating these complex series of molecular events represents a formidable challenge. With significant advances in genome-wide sequencing and bioinformatics tools for analyzing, interpreting, and integrating large and complex data sets [90], the anticipated complete description of chromatin modifications may soon unveil novel methods for selectively and precisely interrupting the epigenetic pathways that promote nephropathy and other diabetic complications. To this end, genome-wide analysis of primary human vascular cells has revealed distinguishable H3 histone acetylation and DNA methylation patterns closely associated to gene expression changes in response to hyperglycemia [91]. Moreover, histone methylation at key promoters drives gene expression that is not attenuated by the reversal of high glucose.
Future studies should aim to substantiate this phenomenon of glycemic memory in the microvasculature of the kidney. The striking nuclear translocation and persistent transcriptional activation exhibited by Set7 in vascular endothelial cells under hyperglycemic conditions strongly indicate this lysine methyltransferase as a prime candidate for interrogation in renal models of glycemic damage and memory. This regulatory role for Set7 operates downstream of ROS, a major agonist of diabetic renal damage. In addition, expression of Set7 is increased upon TGF-β stimulation of rat mesangial cells. Combined with the role of this enzyme in glucose-driven mesangial fibrotic gene expression [76•], these observations support the case for altered histone methylation in the legacy of glucose and the pathology of diabetic kidney disease.
References
Recently published papers of particular interest are highlighted as: • Of importance •• Of major importance
Forbes JM, Cooper ME. Mechanisms of diabetic complications. Physiol Rev. 2013;93:137–88.
Campos C. Chronic hyperglycemia and glucose toxicity: pathology and clinical sequelae. Postgrad Med. 2012;124:90–7.
Ayodele OE, Alebiosu CO, Salako BL. Diabetic nephropathy–a review of the natural history, burden, risk factors and treatment. J Natl Med Assoc. 2004;96:1445–54.
Forbes JM, Fukami K, Cooper ME. Diabetic nephropathy: where hemodynamics meets metabolism. Exp Clin Endocrinol Diabetes. 2007;115:69–84.
Rhee I, Bachman KE, Park BH, et al. DNMT1 and DNMT3b cooperate to silence genes in human cancer cells. Nature. 2002;416:552–6.
Laird PW, Jaenisch R. The role of DNA methylation in cancer genetic and epigenetics. Annu Rev Genet. 1996;30:441–64.
Herman JG, Baylin SB. Promoter-region hypermethylation and gene silencing in human cancer. Curr Top Microbiol Immunol. 2000;249:35–54.
Balgkouranidou I, Liloglou T, Lianidou ES. Lung cancer epigenetics: emerging biomarkers. Biomark Med. 2013;7:49–58.
Waldmann T, Schneider R. Targeting histone modifications-epigenetics in cancer. Curr Opin Cell Biol. 2013.
Webster AL, Yan MS, Marsden PA. Epigenetics and cardiovascular disease. Can J Cardiol. 2013;29:46–57.
Chin L, Gray JW. Translating insights from the cancer genome into clinical practice. Nature. 2008;452:553–63.
• Cooper ME, El-Osta A. Epigenetics: mechanisms and implications for diabetic complications. Circ Res. 2010;107:1403–13. Excellent review of the potential role of epigenetic mechansims and pathways implicated in diabetic vascular complications with insight into hyperglycemic memory.
[no authors listed] The Diabetes Control and Complications Trial (DCCT). Design and methodologic considerations for the feasibility phase. The DCCT Research Group. Diabetes 1986: 35: 530–545.
[no authors listed] The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N Engl J Med 1993: 329: 977–986.
[no authors listed] Epidemiology of Diabetes Interventions and Complications (EDIC). Design, implementation, and preliminary results of a long-term follow-up of the Diabetes Control and Complications Trial cohort. Diabetes Care 1999: 22: 99–111.
[no authors listed] Retinopathy and nephropathy in patients with type 1 diabetes four years after a trial of intensive therapy. The Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications Research Group. N Engl J Med 2000: 342: 381–389.
[no authors listed] Sustained effect of intensive treatment of type 1 diabetes mellitus on development and progression of diabetic nephropathy: the Epidemiology of Diabetes Interventions and Complications (EDIC) study. JAMA 2003: 290: 2159–2167.
Engerman RL, Kern TS. Progression of incipient diabetic retinopathy during good glycemic control. Diabetes. 1987;36:808–12.
Hammes HP, Klinzing I, Wiegand S, et al. Islet transplantation inhibits diabetic retinopathy in the sucrose-fed diabetic Cohen rat. Invest Ophthalmol Vis Sci. 1993;34:2092–6.
El-Osta A, Brasacchio D, Yao D, et al. Transient high glucose causes persistent epigenetic changes and altered gene expression during subsequent normoglycemia. J Exp Med. 2008;205:2409–17.
•• Brasacchio D, Okabe J, Tikellis C, et al. Hyperglycemia induces a dynamic cooperativity of histone methylase and demethylase enzymes associated with gene-activating epigenetic marks that coexist on the lysine tail. Diabetes. 2009;58:1229–36. This important study demonstrates that transcriptional activation of RELA is linked with persistent chromatin modifications that are regulated by histone modifying enzymes.
Ihnat MA, Thorpe JE, Kamat CD, et al. Reactive oxygen species mediate a cellular ‘memory’ of high glucose stress signalling. Diabetologia. 2007;50:1523–31.
Gilbertson DT, Liu J, Xue JL, et al. Projecting the number of patients with end-stage renal disease in the United States to the year 2015. J Am Soc Nephrol. 2005;16:3736–41.
Steinke JM, Mauer M. Lessons learned from studies of the natural history of diabetic nephropathy in young type 1 diabetic patients. Pediatr Endocrinol Rev. 2008;5 Suppl 4:958–63.
Wada J, Makino H. Inflammation and the pathogenesis of diabetic nephropathy. Clin Sci (Lond). 2013;124:139–52.
Henique C, Tharaux PL. Targeting signaling pathways in glomerular diseases. Curr Opin Nephrol Hypertens. 2012;21:417–27.
Mariappan MM. Signaling mechanisms in the regulation of renal matrix metabolism in diabetes. Exp Diabetes Res. 2012;201(2):749812.
Lan HY. Transforming growth factor-beta/Smad signalling in diabetic nephropathy. Clin Exp Pharmacol Physiol. 2012;39:731–8.
Gupta A, Gupta P, Biyani M. Targeted therapies in diabetic nephropathy: an update. J Nephrol. 2011;24:686–95.
Liu W, Lan T, Xie X, et al. S1P2 receptor mediates sphingosine-1-phosphate-induced fibronectin expression via MAPK signaling pathway in mesangial cells under high glucose condition. Exp Cell Res. 2012;318:936–43.
Ziyadeh FN. Mediators of diabetic renal disease: the case for tgf-Beta as the major mediator. J Am Soc Nephrol. 2004;15 Suppl 1:S55–7.
Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes. 2005;54:1615–25.
Leehey DJ, Singh AK, Alavi N, et al. Role of angiotensin II in diabetic nephropathy. Kidney Int Suppl. 2000;77:S93–8.
Kang SW, Adler SG, Lapage J, et al. p38 MAPK and MAPK kinase 3/6 mRNA and activities are increased in early diabetic glomeruli. Kidney Int. 2001;60:543–52.
Brosius 3rd FC. New insights into the mechanisms of fibrosis and sclerosis in diabetic nephropathy. Rev Endocr Metab Disord. 2008;9:245–54.
Kanwar YS, Wada J, Sun L, et al. Diabetic nephropathy: mechanisms of renal disease progression. Exp Biol Med (Maywood). 2008;233:4–11.
Bondeva T, Ruster C, Franke S, et al. Advanced glycation end-products suppress neuropilin-1 expression in podocytes. Kidney Int. 2009;75:605–16.
Bondeva T, Roger T, Wolf G. Differential regulation of Toll-like receptor 4 gene expression in renal cells by angiotensin II: dependency on AP1 and PU.1 transcriptional sites. Am J Nephrol. 2007;27:308–14.
Yonemoto S, Machiguchi T, Nomura K, et al. Correlations of tissue macrophages and cytoskeletal protein expression with renal fibrosis in patients with diabetes mellitus. Clin Exp Nephrol. 2006;10:186–92.
• Zhang Q, Xiao X, Li M, et al. Gene expression profiling in glomeruli of diabetic nephropathy rat. Exp Biol Med (Maywood). 2012;237:903–11. This study examined transcriptome profiles of diabetic and control rat gloemruli. Furhermore, bioinformatic analysis revealed enrichment for pathways associated with diabetic nephropathy.
Wada J, Zhang H, Tsuchiyama Y, et al. Gene expression profile in streptozotocin-induced diabetic mice kidneys undergoing glomerulosclerosis. Kidney Int. 2001;59:1363–73.
Brunskill EW, Potter SS. Gene expression programs of mouse endothelial cells in kidney development and disease. PLoS One. 2010;5:e12034.
• Brennan EP, Morine MJ, Walsh DW, et al. Next-generation sequencing identifies TGF-beta1-associated gene expression profiles in renal epithelial cells reiterated in human diabetic nephropathy. Biochim Biophys Acta. 2012;1822:589–99. This study describes transcriptome profiling of renal tubular epithelial cells following stimulation with TGF-β. Numerous transcriptional changes were confirmed in renal biopsies of patients with diabetic nephropathy.
Woroniecka KI, Park AS, Mohtat D, et al. Transcriptome analysis of human diabetic kidney disease. Diabetes. 2011;60:2354–69.
Tang W, Gao Y, Li Y, et al. Gene networks implicated in diabetic kidney disease. Eur Rev Med Pharmacol Sci. 2012;16:1967–73.
[no authors listed] Effect of 6 months of strict metabolic control on eye and kidney function in insulin-dependent diabetics with background retinopathy. Steno study group. Lancet 1982: 1: 121–124.
Feldt-Rasmussen B, Mathiesen ER, Deckert T. Effect of two years of strict metabolic control on progression of incipient nephropathy in insulin-dependent diabetes. Lancet. 1986;2:1300–4.
[no authors listed] Blood glucose control and the evolution of diabetic retinopathy and albuminuria. A preliminary multicenter trial. The Kroc Collaborative Study Group. N Engl J Med 1984: 311: 365–372.
Bending JJ, Viberti GC, Bilous RW, et al. Eight-month correction of hyperglycemia in insulin-dependent diabetes mellitus is associated with a significant and sustained reduction of urinary albumin excretion rates in patients with microalbuminuria. Diabetes. 1985;34 Suppl 3:69–73.
Dahl-Jorgensen K, Brinchmann-Hansen O, Hanssen KF, et al. Effect of near normoglycaemia for two years on progression of early diabetic retinopathy, nephropathy, and neuropathy: the Oslo study. Br Med J (Clin Res Ed). 1986;293:1195–9.
Reichard P, Nilsson BY, Rosenqvist U. The effect of long-term intensified insulin treatment on the development of microvascular complications of diabetes mellitus. N Engl J Med. 1993;329:304–9.
Patel A, MacMahon S, Chalmers J, et al. Effects of a fixed combination of perindopril and indapamide on macrovascular and microvascular outcomes in patients with type 2 diabetes mellitus (the ADVANCE trial): a randomised controlled trial. Lancet. 2007;370:829–40.
Patel A, MacMahon S, Chalmers J, et al. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med. 2008;358:2560–72.
[no authors listed] Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998: 352: 837–853.
Holman RR, Paul SK, Bethel MA, et al. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med. 2008;359:1577–89.
Gaede P, Tarnow L, Vedel P, et al. Remission to normoalbuminuria during multifactorial treatment preserves kidney function in patients with type 2 diabetes and microalbuminuria. Nephrol Dial Transplant. 2004;19:2784–8.
Gaede P, Vedel P, Parving HH, et al. Intensified multifactorial intervention in patients with type 2 diabetes mellitus and microalbuminuria: the Steno type 2 randomised study. Lancet. 1999;353:617–22.
Murr R. Interplay between different epigenetic modifications and mechanisms. Adv Genet. 2010;70:101–41.
Svejstrup JQ. The RNA, polymerase II transcription cycle: cycling through chromatin. Biochim Biophys Acta. 2004;1677:64–73.
El-Osta A. Glycemic memory. Curr Opin Lipidol. 2012;23:24–9.
Reddy MA, Natarajan R. Role of epigenetic mechanisms in the vascular complications of diabetes. Subcell Biochem. 2012;61:435–54.
Fuks F, Hurd PJ, Wolf D, et al. The methyl-CpG-binding protein MeCP2 links DNA methylation to histone methylation. J Biol Chem. 2003;278:4035–40.
Meehan RR, Lewis JD, Bird AP. Characterization of MeCP2, a vertebrate DNA binding protein with affinity for methylated DNA. Nucleic Acids Res. 1992;20:5085–92.
Zhang Y, Ng HH, Erdjument-Bromage H, et al. Analysis of the NuRD subunits reveals a histone deacetylase core complex and a connection with DNA methylation. Genes Dev. 1999;13:1924–35.
Keating ST, El-Osta A. Epigenetic changes in diabetes. Clin Genet 2013. [epub ahead of print]
Sapienza C, Lee J, Powell J, et al. DNA methylation profiling identifies epigenetic differences between diabetes patients with ESRD and diabetes patients without nephropathy. Epigenetics. 2011;6:20–8.
Bell CG, Teschendorff AE, Rakyan VK, et al. Genome-wide DNA methylation analysis for diabetic nephropathy in type 1 diabetes mellitus. BMC Med Genomics. 2010;3:33.
Roth SY, Denu JM, Allis CD. Histone acetyltransferases. Annu Rev Biochem. 2001;70:81–120.
Havas K, Whitehouse I, Owen-Hughes T. ATP-dependent chromatin remodeling activities. Cell Mol Life Sci. 2001;58:673–82.
Strahl BD, Allis CD. The language of covalent histone modifications. Nature. 2000;403:41–5.
Kassner I, Barandun M, Fey M et al. Crosstalk between SET7/9-dependent methylation and ARTD1-mediated ADP-ribosylation of histone H1.4. Epigenetics Chromatin 2013: 6: 1.
Tweedie-Cullen RY, Brunner AM, Grossmann J, et al. Identification of combinatorial patterns of post-translational modifications on individual histones in the mouse brain. PLoS One. 2012;7:e36980.
Muers M. Chromatin: a haul of new histone modifications. Nat Rev Genet. 2011;12:744.
Sayyed SG, Gaikwad AB, Lichtnekert J, et al. Progressive glomerulosclerosis in type 2 diabetes is associated with renal histone H3K9 and H3K23 acetylation, H3K4 dimethylation and phosphorylation at serine 10. Nephrol Dial Transplant. 2010;25:1811–7.
Gaikwad AB, Gupta J, Tikoo K. Epigenetic changes and alteration of Fbn1 and Col3A1 gene expression under hyperglycaemic and hyperinsulinaemic conditions. Biochem J. 2010;432:333–41.
• Sun G, Reddy MA, Yuan H, et al. Epigenetic histone methylation modulates fibrotic gene expression. J Am Soc Nephrol. 2010;21:2069–80. This important study implicates the Set7 lysine methyltransferase in mesangial gene expression in response to TGF-β and hyperglycemia. This is the first study to show that enzymes and histone modifications involved in vascular glycemic memory are responsive to glucose in renal cells.
Gotzsche O, Gundersen HJ, Osterby R. Irreversibility of glomerular basement membrane accumulation despite reversibility of renal hypertrophy with islet transplantation in early experimental diabetes. Diabetes. 1981;30:481–5.
Roy S, Sala R, Cagliero E, et al. Overexpression of fibronectin induced by diabetes or high glucose: phenomenon with a memory. Proc Natl Acad Sci U S A. 1990;87:404–8.
Fang D, Guan H, Liu J, et al. Early intensive insulin therapy attenuates the p38 pathway in the renal cortex and indices of nephropathy in diabetic rats. Endocr J. 2012;59:81–90.
Okabe J, Orlowski C, Balcerczyk A, et al. Distinguishing hyperglycemic changes by Set7 in vascular endothelial cells. Circ Res. 2012;110:1067–76.
Starkey JM, Haidacher SJ, LeJeune WS, et al. Diabetes-induced activation of canonical and noncanonical nuclear factor-kappaB pathways in renal cortex. Diabetes. 2006;55:1252–9.
Keating ST, El-Osta A. Chromatin modifications associated with diabetes. J Cardiovasc Transl Res. 2012;5:399–412.
Syreeni A, El-Osta A, Forsblom C, et al. Genetic examination of SETD7 and SUV39H1/H2 methyltransferases and the risk of diabetes complications in patients with type 1 diabetes. Diabetes. 2011;60:3073–80.
Nishikawa T, Edelstein D, Brownlee M. The missing link: a single unifying mechanism for diabetic complications. Kidney Int Suppl. 2000;77:S26–30.
• Zhong Q, Kowluru RA. Epigenetic changes in mitochondrial superoxide dismutase in the retina and the development of diabetic retinopathy. Diabetes. 2011;60:1304–13. This study reveals a strong association between mitochondrial superoxide dismutase and the development of diabetic retinopathy.
Schmid H, Boucherot A, Yasuda Y, et al. Modular activation of nuclear factor-kappaB transcriptional programs in human diabetic nephropathy. Diabetes. 2006;55:2993–3003.
Villeneuve LM, Reddy MA, Lanting LL, et al. Epigenetic histone H3 lysine 9 methylation in metabolic memory and inflammatory phenotype of vascular smooth muscle cells in diabetes. Proc Natl Acad Sci U S A. 2008;105:9047–52.
Hovind P, Tarnow L, Rossing K, et al. Decreasing incidence of severe diabetic microangiopathy in type 1 diabetes. Diabetes Care. 2003;26:1258–64.
Yokoyama H, Okudaira M, Otani T, et al. Higher incidence of diabetic nephropathy in type 2 than in type 1 diabetes in early-onset diabetes in Japan. Kidney Int. 2000;58:302–11.
El-Osta A. Remodeling is at the heart of chromatin: the heartaches of chromatin. Epigenetics. 2011;6:884–7.
Pirola L, Balcerczyk A, Tothill RW, et al. Genome-wide analysis distinguishes hyperglycemia regulated epigenetic signatures of primary vascular cells. Genome Res. 2011;21:1601–15.
Acknowledgments
The authors acknowledge grant, scholarship, and fellowship support from the National Health and Medical Research Council (NHMRC), the Diabetes Australia Research Trust (DART), the Juvenile Diabetes Research Foundation International (JDRF), the National Heart Foundation of Australia (NHF), and the Australian Postgraduate Award (APA). A. E-O is a Senior Research Fellow supported by the NHMRC. This article was supported in part by the Victorian Government’s Operational Infrastructure Support Program.
Conflict of interest
Samuel T. Keating declares that he has no conflict of interest.
Assam El-Osta declares that he has no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keating, S.T., El-Osta, A. Glycemic Memories and the Epigenetic Component of Diabetic Nephropathy. Curr Diab Rep 13, 574–581 (2013). https://doi.org/10.1007/s11892-013-0383-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11892-013-0383-y