Abstract
In obesity, the vascular complication is a result of insulin resistance, such as decreased capillary recruitment in skeletal muscle from endothelial insulin resistance. Recent progress in the study of obesity-associated inflammation suggests that vasculature dysfunction occurs in adipose tissue before insulin resistance. In obesity, capillary density and function fail to meet the demand of adipose tissue growth. The failure leads to microcirculation dysfunction from an impaired blood perfusion, which results in a local hypoxia response in adipose tissue. The hypoxia response in adipocytes and macrophages is a new cellular basis for the chronic inflammation. The obesity-associated inflammation has both positive and negative effects in the body. At the early stage, it amplifies the hypoxia signal to stimulate vasculature remodeling locally, and promotes systemic energy expenditure against obesity. At the late stage, it causes adipose tissue dysfunction for insulin resistance. These points suggest that in obesity, adipose tissue vascularization controls chronic inflammation and influences systemic insulin sensitivity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Obesity-associated chronic inflammation occurs primarily in adipose tissue, and contributes to pathogenesis of insulin resistance. There are several hypotheses to explain the origin of inflammation in adipose tissue. Those involve in free fatty acids (FFAs)/toll-like receptor 4, fatty acid derivatives (diaglyceride or ceramide), endoplasmic reticulum (ER) stress, oxidative stress, adipocyte death and PKR (double-stranded RNA-dependent protein kinase) [1]. Although these factors directly induce inflammatory response in adipose tissue, it is not clear why these factors are increased in obesity. On the other side, anti-inflammatory therapies are not successful in the treatment of type 2 diabetes [2]. These facts suggest there are a lot of unknowns about obesity-associated inflammation. The origin and biological significance of the inflammation are two major questions to address the failure in anti-inflammation-based therapy. Recent studies have provided answers to both questions.
The character of obesity-associated inflammation has been widely documented in literature. The inflammation contributes to systemic insulin resistance through inhibition of adipose tissue function in fat storage and hormones/cytokines secretion. Inflammation enhances lipolysis and reduces re-esterification of FFAs in adipocytes, which leads to FFA elevation in circulation and ectopic fat deposition for lipotoxicity. Inhibition of adiponectin expression by inflammation leads to hypoadiponectinemia for insulin resistance. Lipotoxicity and hypoadiponectinemia impair insulin receptor substrate (IRS) function in the insulin signaling pathway in liver and muscle. Studies from many groups including ours suggest that activation of serine kinases (IKK, JNK, PKC, and S6K) by FFA derivatives or inflammatory cytokines contributes to the IRS inhibition through serine phosphorylation. The adipocyte inhibition by inflammation relates to functional suppression of peroxisome proliferator-activated receptor-γ (PPAR-γ), which is a nuclear receptor for thiazolidinedione/long-chain fatty acids and required for maintenance of the adipocyte functions. Our study suggests that nuclear factor-κB (NF-κB) activation by inflammatory cytokine (tumor necrosis factor-α [TNF-α]) results in the PPAR-γ inhibition through a corepressor exchange program [3].
Adipose Tissue Hypoxia
Recent studies from multiple groups including ours consistently suggest that adipose tissue hypoxia (ATH) is a root of chronic inflammation in obesity. The hypoxia is likely a result of reduction in adipose tissue blood flow (ATBF), which is supported by studies in humans and animals. ATBF (mL/min/g) reduction was first indicated in obese individuals by a decreased rate in the radioisotope clearance in subcutaneous fat in 1966 [4]. The reduction was confirmed in an animal experiment in dogs in 1967. The most convincing evidence is reported in rats in 1984 [5], in which ATBF reduction was tested using radiolabeled microsphere in Sprague-Dawley rats for age-related (450 days) obesity. In the study, blood flow was determined with 51Cr-labeled microsphere in epididymal, mesenteric, retroperitoneal, and subcutaneous fat. The result demonstrates that the fat blood flow is different among distinct fat pads. The highest flow is in mesenteric fat and lowest in epididymal fat. In obesity, the blood flow (mL/min/vol or mL/min/mm2) is decreased in epididymal, retroperitoneal, mesenteric, and subcutaneous fat pads. A similar observation was made later in Zucker obese rats in 1987 [6], in which 51Cr or 103Ru-labeled microsphere (dextran) was used in the analysis of blood flow in five difference adipose tissues including dorsal subcutaneous, retroperitoneal, inguinal, epididymal, and mesenteric fat pads. The blood flow (mL/min) was normalized in three ways: pad, gram, and cell surface (mm2). When normalized with cell surface, the blood flow (mL/min/mm2) was significantly (>50%) reduced in all five fat pads by obesity [6]. The blood flow was not changed in nonadipose tissues in the obese rats, suggesting that the reduction is fat-specific. Now, it is known that in obese individuals, ATBF is 30% to 40% lower (P < 0.02–0.05) than that of lean controls [7].
The ATBF reduction is a result of obesity, but not a result of insulin resistance. ATBF was compared in two rat models of type 2 diabetes with or without obesity [8]. Both obese Zucker rats and nonobese Goto-Kakizaki (GK) rats suffer insulin resistance. The ATBF reduction was observed only in the obese diabetic rats (obese Zucker rat), but not in the nonobese diabetic GK rats, suggesting that the ATBF reduction is a result of obesity, not insulin resistance. In obesity, the ATBF reduction is associated with insulin resistance [9, 10], suggesting a role of ATBF reduction in the pathogenesis of insulin resistance. Although the association has been known for years, the cause/effect relationship was unclear for several years. The relationship received strong support in 2007 when a hypoxia response was found in obese mice to induce adipose tissue inflammation [11] and adipose tissue dysfunction [12]. Study of adipose tissue oxygen tension is found in literature of 2004 to 2005 about wound healing in obese patients [13, 14]. The reports show that oxygen supply is reduced in subcutaneous fat of obese patients. The authors conclude that the reduced oxygenation is a potential mechanism for the delayed wound healing and increased infection risk in obese patients after surgery. However, these studies did not address the relationship of low oxygenation and insulin resistance in obesity. Hypoxia was proposed as a possible cause of adipose tissue inflammation in 2004 in a review article [15]. The article suggests that adipose tissue may suffer a hypoxia response that contributes to the chronic inflammation. This hypothesis received strong support when the relationship was systematically investigated in obese mice during 2004 to 2007 and reported in 2007 by our group [11]. The study provides a direct link of ATH to the pathogenesis of insulin resistance.
In addition to the ATBF reduction, the increase in adipocyte size may contribute to the hypoxia response in adipose tissue as well, which is discussed in our report [11]. In tissue, oxygen can only defuse about 120 μm. When adipocyte diameter increases to (or above) 120 μm, oxygen will not be able to reach the cells beyond 120 μm from the capillary. The diameter of a large adipocyte can be over 150 μm [16]. This distance effect remains to be tested in the adipose tissue in obesity. There are four different assays in the study of ATH as being reviewed [17].
Hypoxia Induction of Adipose Tissue Inflammation
Hypoxia promotes inflammatory response through direct and indirect effects in adipose tissue. The direct effect is activation of multiple signaling pathways in adipocytes and macrophages from the hypoxia-related stresses. Transcription factors such as hypoxia-inducible factor-1α (HIF-1α) and NF-κB are the important signaling molecules for hypoxia to induce the inflammatory responses. The nuclear factors stimulate expression of many proinflammatory molecules through transcriptional activation. In the indirect approach, hypoxia induces adipocyte death (eg, apoptosis and necrosis) and lipolysis.
HIF-1α is a master signal mediator of hypoxia signal. In 2005, HIF-1α was shown to be increased in adipose tissue of obese patients and its expression was reduced after surgery-induced weight loss [18]. In 2006, the increase in HIF-1α expression was confirmed in adipose tissue by microarray and immunohistostaining [19]. In the primary cell culture, HIF-1α protein was induced in human adipocytes by hypoxia [20]. HIF-1α mRNA and protein are induced by insulin as well. Therefore, in obese conditions, HIF-1α induction in fat tissue may be a result of hyperinsulinemia and ATH. The role of hypoxia in HIF-1α activation was reported in the adipose tissue of obese mice in 2007 by our group and Hosogai’s group independently [11, 12]. The human study was conducted in obese patients and reported in 2009 by Pasarica, et al. [21].
Regulation of HIF-1α activity is well documented in the literature. HIF-1α stays in the nucleus and is not detectable in the cytoplasm under normoxia. HIF-1α and HIF-1β are the two subunits of HIF-1, whose activity is regulated at multiple levels including expression (mRNA and protein) and protein degradation. Hypoxia does not induce HIF-1α mRNA in cultured cells, but does induce the mRNA in tissue under ischemia [22]. Under normal oxygen conditions, HIF-1α protein with a half-life of less than 5 min is subject to rapid degradation in the proteasome. The protein stability is induced by hypoxia and several growth factors including insulin, insulin-like growth factor, and epidermal growth factor through activation of PI3K-AKT and MAPK-ERK pathways. Under hypoxic conditions, HIF-1α protein increases dramatically through inhibition of protein degradation by hypoxia. In cell culture, HIF-1α protein peaks at 4 to 8 h and returns to the basal level 16 h later in response to ambient hypoxia. HIF-1α translocates to the nucleus and dimerizes with HIF-1β (hydrocarbon receptor nuclear translocator [ARNT]) to form the active HIF-1 protein, which binds to the promoter DNA of target genes and induces gene transcription. The core DNA sequence of HIF-1 element is 5′-RCGTG-3′. HIF-1α is also upregulated by certain transition metals (Co2+, Ni2+, and Mn2+) and by iron chelating. HIF-1α expression is required for embryonic survival in mice.
Activation of NF-κB by hypoxia is widely investigated in the cancer, immunology, and cardiovascular diseases. In response to hypoxia, NF-κB is disassociated from the inhibitor protein (IκBα) in the absence of IκBα degradation. The disassociation leads to nuclear translocation and transcriptional activation of inflammatory cytokines. In the literature, there was no clear documentation about the role of hypoxia-induced NF-κB activation in adipose tissue before our report in 2007 [11]. We detected hypoxia and NF-κB activation in the adipose tissue at the same time. In cell culture, hypoxia activated NF-κB in adipocytes and macrophages to induce expression of proinflammatory cytokines (TNF-α and monocyte chemoattractant protein-1 [MCP-1]). ER stress and oxidative stress may be involved in NF-κB activation by hypoxia according to the literature.
Hypoxia induces macrophage infiltration into adipose tissue by stimulating expression of chemokines, which attract macrophages into adipose tissue. Adipose tissue macrophages have been under active investigation since 2004 when macrophage infiltration was identified in obese mice [23–26]. MCP-1 (CCL2) is proposed to be a major chemokine for macrophage infiltration into adipose tissue. This activity of MCP-1 is supported by several studies using transgenic mice with either global MCP-1 knockout or fat-specific overexpression of MCP-1. However, later studies suggest that macrophage infiltration was not associated with MCP-1 in both lean and obese mice [27, 28]. We propose that macrophage migration inhibition factor (MIF) is one of the factors mediating the hypoxia signal in the macrophage infiltration in adipose tissue [11]. This possibility is supported by adipocyte expression of MIF in response to hypoxia [11]. MIF is a 114 amino acid protein that circulates in homotrimeric, dimeric, and monomeric forms. MIF expression is increased by hypoxia and glucocorticoids. Study of MIF knockout mice suggests that MIF is required for normal function of macrophage. It remains to be understood how MIF is linked to insulin resistance. Such a link is suggested by the possible role of MIF in macrophage infiltration into adipose tissue [11].
Our study suggests that hypoxia may promote macrophage infiltration through induction of cell apoptosis or necrosis [29••]. Adipocyte death induces macrophage infiltration into adipose tissue to clean the dead adipocytes [30]. This activity of dead adipocytes is supported by an observation that macrophages are located around the dead adipocytes in the adipose tissue [31]. Macrophages are also located in the hypoxic areas in adipose tissue of obese mice [32]. However, it is unknown what causes the cell death in adipose tissue. Our study suggests that the adipocyte death is a result of hypoxia response [29••]. In response to hypoxia, adipocytes undergo apoptosis and necrosis for cell death. The cell death may contribute to FFA elevation in circulation because dead cells may release FFA into blood stream. This possibility is verified in an experiment using a ischemia model, in which plasma FFA was induced by acute hypoxia from a femoral arterial clamp in the rat hind leg [29••]. This possibility is supported by a study of newborn mice, in which plasma FFA is induced by systemic hypoxia [33].
Our study demonstrates that hypoxia induces lipolysis in adipocytes [29••]. Plasma FFAs are positively associated with increased lipolysis in adipose tissue. The lipolysis is a result of reduced insulin action in adipocytes, which occurs under fasting or insulin resistance. To understand hypoxia impact on lipid metabolism in adipocytes, we examined lipolysis in adipocytes under hypoxia. The study shows that hypoxia induces adipocyte lipolysis through inhibition of insulin signaling pathway [29••]. Hypoxia inhibits insulin receptor protein in 3T3-L1 adipocytes and reduces signaling activity of insulin receptor [29••]. Inhibition of PPAR-γ expression is another mechanism of hypoxia-induced lipolysis. In a study of hypoxia in the regulation of adipogenesis, Yun et al. [34] observed that hypoxia inhibited PPAR-γ activity in a cellular model. Activation of AMP-activated protein kinase (AMPK) by hypoxia may contribute to lipolysis in adipocytes as well. AMPK is a serine kinase that serves as a sensor for energy supply in cells. It is known that hypoxia activates AMPK through inhibition of mitochondrial respiration or oxidative stress [35].
Hypoxia induces ER stress [36]. ER stress induces adipose tissue dysfunction in obese mice [37]. In search for the mechanism of obesity-associated JNK activation, ER stress was found in the adipose tissue in obese mice [37]. Inhibition of ER stress genetically or chemically provides protection of the mice from obesity-associated insulin resistance [38]. However, those studies do not explain why ER stress occurs in obesity. This point is addressed in a study by Hosogai et al. [12] in which hypoxia was shown to induce ER stress in 3T3-L1 adipocytes. In addition to the JNK activation, the ER stress may induce NF-κB activity as well under hypoxia condition.
Hypoxia inhibits mitochondrial function. White adipocytes have much less mitochondria relative to the brown adipocytes. However, mitochondrial dysfunction may contribute to malfunction in white adipose tissue [39]. Mitochondria are reduced in adipose tissue of obese people [39]. However, it is not clear why and how the mitochondria number is reduced. It is known that mitochondria respiration and biogenesis are inhibited by hypoxia. HIF-1α is a major mediator of the hypoxia signal in the inhibition of mitochondrial function. Therefore, hypoxia is a potential cause of the mitochondrial inhibition in adipocyte in obesity. This possibility remains to be tested.
Several groups consistently report that hypoxia inhibits adiponectin expression in adipocytes [11, 12, 20, 40]. ATH provides a new mechanism for adiponectin reduction in obesity. Inflammation was proposed to inhibit adiponectin expression in obesity, although the relative importance of inflammation is controversial in obesity. In classic chronic inflammatory/autoimmune diseases (eg, rheumatoid arthritis, systemic lupus erythematosus, inflammatory bowel disease, type 1 diabetes, and cystic fibrosis), adiponectin levels are elevated rather than decreased. In obesity, inhibition of adiponectin expression by hypoxia provides an excellent alternative mechanism to inflammation.
Leptin is reported as a HIF-1α target gene [41, 42]. Leptin is a primary hormone produced by adipose tissue and plasma leptin is positively associated with body weight (or adiposity). In adipose tissue, leptin mRNA is positively associated with adiposity. In cell culture, leptin expression is induced by hypoxia in adipocytes [43]. However, this activity of hypoxia is controversial because leptin was not induced when classical hypoxia response genes were induced by hypoxia in adipocytes [44].
Causes of ATBF Reduction
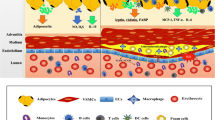
Our study suggests that a reduction in capillary density contributes to ATH [45]. We observed that capillary density was reduced in adipose tissue of obese mice [45], which was confirmed in other studies later [21, 46, 47]. Capillary density is determined by angiogenesis that requires proliferation and tube formation of endothelial cells. Endothelial proliferation was driven by growth factors including vascular endothelial growth factor (VEGF) and fibroblast growth factor-2. The tube formation and capillary maturation are controlled by a different set of cytokines including platelet-derived growth factor (PDGF), transforming growth factor-β (TGF-β), and angiopoietin. We observed that VEGF expression was not increased in response to hypoxia in the adipose tissue of ob/ob mice, although expression of other hypoxia response genes was upregulated [11]. This defect was associated with a reduced endothelial density in the tissue [45]. The evidence suggests that angiogenesis is deficient in the adipose tissue of obese mice, and is a mechanism for the reduction in capillary density, which accounts for ATBF reduction in obesity. The details of molecular events underlying the angiogenic defect remain to be investigated in the obese condition.
Blood perfusion is regulated by vasodilation or vasoconstriction. An increase in vasoconstriction in obesity is another possible mechanism for the ATBF reduction. This possibility is supported by the literature about angiotensin II (Ang II). Ang II is a serum peptide for vasoconstriction. Ang II is a component in the renin-angiotensin system, and produced after hydrolysis of Ang I by angiotensin-converting enzyme. Ang II acts on both the type 1 (AT1) and type 2 (AT2) receptors. In obesity, the Ang II activity is increased in adipose tissue and in circulation. This may contribute to the ATBF reduction through an increase in vasoconstriction. Additionally, the Ang II inhibitors are known to enhance blood perfusion in adipose tissue [48]. The inhibitors also decrease inflammation in adipose tissue, and increase systemic insulin sensitivity. The relative significance remains to be tested in vivo for the capillary reduction and vasoconstriction in the pathogenesis of ATH in obesity.
Benefits of Obesity-Associated Inflammation
Inflammation originates from the hypoxia response in adipose tissue at the early stage in obesity. When metabolic disorders occur, metabolites of lipid and glucose will join the force to promote inflammation at the late stage of obesity. Those metabolites include oxygen species, diaglyceride, and ceramide, which induce inflammation through activation of IKK and JNK. What is the biological significance of inflammation in the adipose hypoxia response? We believe that inflammation serves as a signal amplifier for hypoxia in the tissue remodeling process during adipose tissue expansion. Angiogenesis is a major event in the extracellular matrix remodeling program and is extensively documented in cancer biology and developmental biology. Pathogenic angiogenesis is related to tumor metastasis and diabetic retinopathy. The major driving force for angiogenesis is hypoxia. Angiogenesis is required for adipocyte differentiation and tissue growth [49, 50]. Inhibition of angiogenesis reduces adipose tissue growth and prevents obesity. Regulation of angiogenesis by inflammation in adipose tissue is supported by several recent studies about macrophage functions in adipose tissue [28, 45, 51].
In adipose tissue, many cytokines produced by adipocytes and macrophages are proangiogenic factors [50]. These cytokines include leptin, adiponectin, VEGF, hepatocyte growth factor (HGF), TNF-α, MIF-1, interleukin-8, PDGF, TGF-β, and angiopoietin.
Adipocytes and macrophages have different activities in the regulation of angiogenesis. Adipocytes produce cell type-specific proangiogenic factors (leptin and adiponectin), and also cell type-unspecific proangiogenic factors, such as VEGF and HGF. VEGF is a powerful angiogenic factor to stimulate proliferation of endothelial cells. Adipocytes express more VEGF than stromal-vascular cells, which contain macrophages, fibroblasts, and endothelial cells. We compared primary adipocytes and macrophages in VEGF expression, and observed that adipocytes express more VEGF than macrophages in mice (Ye, Unpublished data). Fat pad location determines VEGF expression levels. Visceral fat (omentum) expresses more VEGF than subcutaneous fat in mice. This location effect was verified in a transplantation study of 3T3-L1 adipocytes, which express more VEGF mRNA in the mesenteric area than in subcutaneous area [52]. In obese patients, serum VEGF is positively associated with visceral fat content [53], suggesting that fat tissue controls serum VEGF level. Visceral fat is most important among all of the fat pads in the control of serum VEGF. The molecular basis is that visceral fat expresses more VEGF than subcutaneous fat [52, 54]. It is not clear why VEGF expression by adipocytes varies with fat pad location.
Macrophages may serve as a signal amplifier for hypoxia in the stimulation of angiogenesis in adipose tissue. In response to hypoxia, macrophages are able to secrete almost all of the proangiogenic factors [55]. PDGF is a good example for this activity of macrophages. Adipocytes produce a high level of VEGF, but they cannot produce much PDGF [45]. Our data suggest that differentiated 3T3-L1 cells lost their ability in expression of PDGF [45], and gained capacity in VEGF expression. VEGF mainly induces proliferation of endothelial cells. It does not stimulate maturation of capillary. PDGF is able to induce tube formation and recruitment of pericytes for capillary maturation. Therefore, in adipose tissue, proangiogenic factors from adipocytes may not be sufficient for neovascularization. Macrophages may facilitate the neovascularation by producing PDGF. Without macrophages, angiogenesis was significantly reduced in adipose tissue in lean mice [28].
In addition to adipose angiogenesis, inflammation serves as a signal to stimulate energy expenditure in obesity. This concept is supported by abundant literature on cancer cachexia and caloric restriction [56••]. Inflammation stimulates energy expenditure by enhancing fuel mobilization and heat production. The former is supported by inflammation induction of fatty acid release from adipose tissue through lipolysis and adipocyte death. The latter is based on elevated basal metabolic rates in our NF-κB transgenic mice, which exhibit chronic inflammation and elevated body temperature [57]. To understand inflammation effects on insulin sensitivity, we made fat-specific NF-κB overexpressing mice by expressing the P65 subunit under the aP2 gene promoter. The mice exhibit a chronic inflammation in adipose tissue on chow diet. Macrophage infiltration and proinflammatory cytokine expression are elevated in the tissue. They have a normal body weight at birth and less fat content in adulthood. Their body temperature is 1° higher than the wild-type mice on chow diet. The transgenic mice have a significant increase in food intake and energy expenditure. On a high-fat diet, they are protected from obesity and insulin resistance. These observations suggest that chronic inflammation promotes energy expenditure in vivo.
Clinical Relevance of ATH
In clinic, obstructive sleep apnea (OSA) is associated with insulin resistance. The OSA syndrome is characterized by recurrent collapse of the upper airway during sleep leading to periods of intermittent hypoxia (IH) and fragmentation of sleep. The prevalence of OSA is 40% to 60% in obese subjects. OSA increases risk of hypertension, coronary artery disease, and stroke. Because systemic hypoxia is associated with insulin resistance and glucose intolerance, hypoxia is proposed as a risk factor for OSA-associated insulin resistance.
In a recent study, systemic hypoxia is shown to induce insulin resistance in lean mice [58]. The insulin resistance was examined with the hyperinsulinemic-euglycemic clamp. The result suggests that hypoxia induced systemic insulin resistance as glucose infusion rate was decreased. A reduction in glucose utilization in the oxidative muscle fibers was observed, suggesting that muscle developed insulin resistance. The role of liver was excluded because hepatic glucose output was not changed by the hypoxia treatment. A role of autonomic nervous system (ANS) was excluded because ANS blocker did not improve the insulin resistance. This study cannot exclude the role of adipose tissue response in the muscle insulin resistance because the adipose tissue function and blood FFA were not examined.
In addition to insulin resistance, the ATH concept suggests a new mechanism for insulin sensitization by several factors such as physical exercise, fasting, weight loss, and Ang II inhibitors. ATBF is increased in response to stress such as exercise, mental stress, fasting, and nutrient intake. ATBF is increased by the Ang II inhibitor, epinephrine, insulin, and nitric oxide. Insulin sensitivity is improved under these conditions. An improvement in oxygen supply may contribute to the mechanism of insulin sensitization in these conditions. This possibility remains to be tested in experiments.
Conclusions
Recent studies have provided compelling evidence for the role of vasculature in the control of adipose tissue growth and function. These studies are leading us to a new view for the adipose tissue vasculature in obesity and its complications. The ATH concept states the importance of vasculature and provides a cellular mechanism for adipose inflammation in obesity. The biological basis of ATH is related to reduction in ATBF, which reflects a failure in compensatory angiogenesis or vasodilation. The beneficial effect of inflammation in obesity is another new concept. It is now believed that inflammation may serve as a signal amplifier for hypoxia to stimulate vasculature remodeling during adipose tissue expansion. In the whole body, inflammation promotes energy expenditure to fight against obesity. Inflammation will exhibit the side effects for insulin resistance and complications at the late stage of obesity when the chronic inflammation is overactivated. These new insights may help us to understand why anti-inflammatory therapy is not successful in the treatment of insulin resistance.
References
Papers of particular interest, published recently, have been highlighted as: •• Of major importance
Nakamura T, Furuhashi M, Li P, Cao H, Tuncman G, Sonenberg N, et al. Double-stranded RNA-dependent protein kinase links pathogen sensing with stress and metabolic homeostasis. Cell. 2010;140:338–48.
Shoelson SE, Goldfine AB. Getting away from glucose: fanning the flames of obesity-induced inflammation. Nat Med. 2009;15:373–4.
Gao Z, He Q, Peng B, Chiao PJ, Ye J. Regulation of nuclear translocation of HDAC3 by I{kappa}B{alpha} is required for tumor necrosis factor inhibition of peroxisome proliferator-activated receptor gamma function. J Biol Chem. 2006;281:4540–7.
Larsen OA, Lassen NA, Quaade F. Blood flow through human adipose tissue determined with radioactive xenon. Acta Physiol Scand. 1966;66:337–45.
Crandall DL, Goldstein BM, Huggins F, Cervoni P. Adipocyte blood flow: influence of age, anatomic location, and dietary manipulation. Am J Physiol. 1984;247:R46–51.
West DB, Prinz WA, Francendese AA, Greenwood MR. Adipocyte blood flow is decreased in obese Zucker rats. Am J Physiol. 1987;253:R228–233.
Bolinder J, Kerckhoffs DA, Moberg E, Hagstrom-Toft E, Arner P. Rates of skeletal muscle and adipose tissue glycerol release in nonobese and obese subjects. Diabetes. 2000;49:797–802.
Kampf C, Bodin B, Kallskog O, Carlsson C, Jansson L. Marked increase in white adipose tissue blood perfusion in the type 2 diabetic GK rat. Diabetes. 2005;54:2620–7.
Jansson PA, Larsson A, Lonnroth PN. Relationship between blood pressure, metabolic variables and blood flow in obese subjects with or without non-insulin-dependent diabetes mellitus. Eur J Clin Invest. 1998;28:813–8.
Karpe F, Fielding BA, Ilic V, Macdonald IA, Summers LK, Frayn KN. Impaired postprandial adipose tissue blood flow response is related to aspects of insulin sensitivity. Diabetes. 2002;51:2467–73.
Ye J, Gao Z, Yin J, He H. Hypoxia is a potential risk factor for chronic inflammation and adiponectin reduction in adipose tissue of ob/ob and dietary obese mice. Am J Physiol Endocrinol Metab. 2007;293:E1118–28.
Hosogai N, Fukuhara A, Oshima K, Miyata Y, Tanaka S, Segawa K, et al. Adipose tissue hypoxia in obesity and its impact on adipocytokine dysregulation. Diabetes. 2007;56:901–11.
Kabon B, Nagele A, Reddy D, Eagon C, Fleshman JW, Sessler DI, et al. Obesity decreases perioperative tissue oxygenation. Anesthesiology. 2004;100:274–80.
Fleischmann E, Kurz A, Niedermayr M, Schebesta K, Kimberger O, Sessler DI, et al. Tissue oxygenation in obese and non-obese patients during laparoscopy. Obes Surg. 2005;15:813–9.
Trayhurn P, Wood IS. Adipokines: inflammation and the pleiotropic role of white adipose tissue. Br J Nutr. 2004;92:347–55.
Bluher M, Wilson-Fritch L, Leszyk J, Laustsen PG, Corvera S, Kahn CR. Role of insulin action and cell size on protein expression patterns in adipocytes. J Biol Chem. 2004;279:31902–9.
Ye J. Emerging role of adipose tissue hypoxia in obesity and insulin resistance. Int J Obes. 2009;33:54–66.
Cancello R, Henegar C, Viguerie N, Taleb S, Poitou C, Rouault C, et al. Reduction of macrophage infiltration and chemoattractant gene expression changes in white adipose tissue of morbidly obese subjects after surgery-induced weight loss. Diabetes. 2005;54:2277–86.
Higami Y, Barger JL, Page GP, Allison DB, Smith SR, Prolla TA, et al. Energy restriction lowers the expression of genes linked to inflammation, the cytoskeleton, the extracellular matrix, and angiogenesis in mouse adipose tissue. J Nutr. 2006;136:343–52.
Wang B, Wood IS, Trayhurn P: Dysregulation of the expression and secretion of inflammation-related adipokines by hypoxia in human adipocytes. Pflugers Arch. 2007;455:479–492.
Pasarica M, Sereda OR, Redman LM, Albarado DC, Hymel DT, Roan LE, et al. Reduced adipose tissue oxygenation in human obesity: evidence for rarefaction, macrophage chemotaxis, and inflammation without an angiogenic response. Diabetes. 2009;58:718–25.
Yu AY, Frid MG, Shimoda LA, Wiener CM, Stenmark K, Semenza GL. Temporal, spatial, and oxygen-regulated expression of hypoxia-inducible factor-1 in the lung. Am J Physiol. 1998;275:L818–826.
Xu H, Barnes GT, Yang Q, Tan G, Yang D, Chou CJ, et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest. 2003;112:1821–30.
Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante Jr AW. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112:1796–808.
Di Gregorio GB, Yao-Borengasser A, Rasouli N, Varma V, Lu T, Miles LM, et al. Expression of CD68 and macrophage chemoattractant protein-1 genes in human adipose and muscle tissues: association with cytokine expression, insulin resistance, and reduction by pioglitazone. Diabetes. 2005;54:2305–13.
Fain JN. Release of interleukins and other inflammatory cytokines by human adipose tissue is enhanced in obesity and primarily due to the nonfat cells. Vitam Horm. 2006;74:443–77.
Inouye KE, Shi H, Howard JK, Daly CH, Lord GM, Rollins BJ, et al. Absence of CC chemokine ligand 2 does not limit obesity-associated infiltration of macrophages into adipose tissue. Diabetes. 2007;56:2242–50.
Cho C-H, Jun Koh Y, Han J, Sung H-K, Jong Lee H, Morisada T, et al. Angiogenic role of LYVE-1-positive macrophages in adipose tissue. Circ Res. 2007;100:e47–57.
•• Yin J, Gao Z, He Q, Ye J. Role of hypoxia in obesity-induced disorders of glucose and lipid metabolism in adipose tissue. Am J Physiol Endocrinol Metab. 2009;296:E333–42. This is the first comprehensive study of hypoxia in the regulation of adipocyte function (insulin signaling, lipolysis, FFA elevation, adipocyte apoptosis, and necrosis). It also provides background information about adipose tissue hypoxia through literature review.
Cinti S, Mitchell G, Barbatelli G, Murano I, Ceresi E, Faloia E, et al. Adipocyte death defines macrophage localization and function in adipose tissue of obese mice and humans. J Lipid Res. 2005;46:2347–55.
Strissel KJ, Stancheva Z, Miyoshi H, Perfield 2nd JW, DeFuria J, Jick Z, et al. Adipocyte death, adipose tissue remodeling, and obesity complications. Diabetes. 2007;56:2910–8.
Rausch ME, Weisberg S, Vardhana P, Tortoriello DV. Obesity in C57BL/6J mice is characterized by adipose tissue hypoxia and cytotoxic T-cell infiltration. Int J Obes (Lond). 2008;32:451–63.
Weinberger B, Carbone T, England S, Kleinfeld AM, Hiatt M, Hegyi T. Effects of perinatal hypoxia on serum unbound free fatty acids and lung inflammatory mediators. Biol Neonate. 2001;79:61–6.
Yun Z, Maecker HL, Johnson RS, Giaccia AJ. Inhibition of PPAR gamma 2 gene expression by the HIF-1-regulated gene DEC1/Stra13: a mechanism for regulation of adipogenesis by hypoxia. Dev Cell. 2002;2:331–41.
Liu L, Cash TP, Jones RG, Keith B, Thompson CB, Simon MC. Hypoxia-induced energy stress regulates mRNA translation and cell growth. Mol Cell. 2006;21:521–31.
Koumenis C, Naczki C, Koritzinsky M, Rastani S, Diehl A, Sonenberg N, et al. Regulation of protein synthesis by hypoxia via activation of the endoplasmic reticulum kinase PERK and phosphorylation of the translation initiation factor eIF2alpha. Mol Cell Biol. 2002;22:7405–16.
Ozcan U, Cao Q, Yilmaz E, Lee A-H, Iwakoshi NN, Ozdelen E, et al. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science. 2004;306:457–61.
Ozcan U, Yilmaz E, Ozcan L, Furuhashi M, Vaillancourt E, Smith RO, et al. Chemical chaperones reduce ER stress and restore glucose homeostasis in a mouse model of type 2 diabetes. Science. 2006;313:1137–40.
Keijer J, van Schothorst EM. Adipose tissue failure and mitochondria as a possible target for improvement by bioactive food components. Curr Opin Lipidol. 2008;19:4–10.
Chen B, Lam KS, Wang Y, Wu D, Lam MC, Shen J, et al. Hypoxia dysregulates the production of adiponectin and plasminogen activator inhibitor-1 independent of reactive oxygen species in adipocytes. Biochem Biophys Res Commun. 2006;341:549–56.
Ambrosini G, Nath AK, Sierra-Honigmann MR, Flores-Riveros J. Transcriptional activation of the human leptin gene in response to hypoxia. Involvement of hypoxia-inducible factor 1. J Biol Chem. 2002;277:34601–9.
Grosfeld A, Andre J, Hauguel-De Mouzon S, Berra E, Pouyssegur J, Guerre-Millo M. Hypoxia-inducible factor 1 transactivates the human leptin gene promoter. J Biol Chem. 2002;277:42953–7.
Wang B, Wood IS, Trayhurn P. Hypoxia induces leptin gene expression and secretion in human preadipocytes: differential effects of hypoxia on adipokine expression by preadipocytes. J Endocrinol. 2008;198:127–34.
Yasumasu T, Takahara K, Nakashima Y. Hypoxia inhibits leptin production by cultured rat adipocytes. Obes Res. 2002;10:128.
Pang C, Gao Z, Yin J, Zhang J, Jia W, Ye J. Macrophage infiltration into adipose tissue may promote angiogenesis for adipose tissue remodeling in obesity. Am J Physiol Endocrinol Metab. 2008;295:E313–22.
Halberg N, Khan T, Trujillo ME, Wernstedt-Asterholm I, Attie AD, Sherwani S, et al. HIF 1 alpha induces fibrosis and insulin resistance in white adipose tissue. Mol Cell Biol. 2009;29:4467–83.
Gealekman O, Guseva N, Hartigan C, Apotheker S, Gorgoglione M, Gurav K, Tran KV, Straubhaar J, Nicoloro S, Czech MP, Thompson M, Perugini RA, Corvera S: Depot-specific differences and insufficient subcutaneous adipose tissue angiogenesis in human obesity. Circulation. 2011;123:186–194.
Goossens GH, McQuaid SE, Dennis AL, van Baak MA, Blaak EE, Frayn KN, et al. Angiotensin II: a major regulator of subcutaneous adipose tissue blood flow in humans. J Physiol. 2006;571:451–60.
Fukumura D, Ushiyama A, Duda DG, Xu L, Tam J, Krishna V, et al. Paracrine regulation of angiogenesis and adipocyte differentiation during in vivo adipogenesis. Circ Res. 2003;93:e88–97.
Cao Y. Angiogenesis modulates adipogenesis and obesity. J Clin Invest. 2007;117:2362–8.
Nishimura S, Manabe I, Nagasaki M, Hosoya Y, Yamashita H, Fujita H, et al. Adipogenesis in obesity requires close interplay between differentiating adipocytes, stromal cells, and blood vessels. Diabetes. 2007;56:1517–26.
Miyazawa-Hoshimoto S, Takahashi K, Bujo H, Hashimoto N, Yagui K, Saito Y. Roles of degree of fat deposition and its localization on VEGF expression in adipocytes. Am J Physiol Endocrinol Metab. 2005;288:E1128–1136.
Miyazawa-Hoshimoto S, Takahashi K, Bujo H, Hashimoto N, Saito Y. Elevated serum vascular endothelial growth factor is associated with visceral fat accumulation in human obese subjects. Diabetologia. 2003;46:1483–8.
Ledoux S, Queguiner I, Msika S, Calderari S, Rufat P, Gasc JM, et al. Angiogenesis associated with visceral and subcutaneous adipose tissue in severe human obesity. Diabetes. 2008;57:3247–57.
Murdoch C, Muthana M, Lewis CE. Hypoxia regulates macrophage functions in inflammation. J Immunol. 2005;175:6257–63.
•• Ye J, Keller J. Regulation of energy metabolism by inflammation: a feedback response in obesity and calorie restriction. Aging. 2010;2:361–8. This is a review article that provides comprehensive information about inflammation in the regulation of energy metabolism. The positive activity of inflammation is emphasized for the first time in obesity in this article.
Tang T, Zhang J, Yin J, Staszkiewicz J, Gawronska-Kozak B, Mynatt R, et al. Uncoupling of inflammation and insulin resistance by NF-kB in transgenic mice through induction of energy expenditure. J Biol Chem. 2010;285:4637–44.
Iiyori N, Alonso LC, Li J, Sanders MH, Garcia-Ocana A, O’Doherty RM, Polotsky VY, O’Donnell CP: Intermittent hypoxia causes insulin resistance in lean mice independent of autonomic activity. Am J Respir Crit Care Med. 2007;175:851–857.
Acknowledgements
This manuscript was prepared with support from National Institutes of Health (NIH) funds (DK68036 and DK85495) to J. Ye.
Disclosure
No potential conflict of interest relevant to this article was reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ye, J. Adipose Tissue Vascularization: Its Role in Chronic Inflammation. Curr Diab Rep 11, 203–210 (2011). https://doi.org/10.1007/s11892-011-0183-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11892-011-0183-1