Abstract
Purpose of Review
Laparoscopic hemicolectomy (LHC) is widely used to treat benign and malignant colorectal disease. However, the optimal method to restore intestinal continuity is still controversial; hence, this study aims to compare the safety and efficacy of intracorporeal anastomosis (IA) with extracorporeal anastomosis (EA).
Recent Findings
Current literature comprises largely of retrospective cohort studies and three significant randomized controlled trials examining the differences between IA and EA in mostly right-sided colectomies. The evidence points to superior intraoperative and postoperative outcomes such as reduced incision length, incidence of wound infection and length of stay for IA, but with incongruous findings on outcomes such as anastomotic leakage and lymph node yield. Likewise, existing meta-analyses have reached differing conclusions about some of these key outcomes.
Summary
This paper provides a timely update to existing reviews and shows that IA is associated with superior intraoperative, postoperative and long-term recovery outcomes. We provide more conclusive evidence that lymph node yield for cancer patients is improved in IA. Furthermore, lymph node yield and lowered readmission rate appeared to be more pronounced in patients with higher BMI through regression analysis. Our key recommendation is for surgeons to acquire a high level of proficiency in performing IA to improve patient outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Since its inception in the 1990s [1], laparoscopic hemicolectomy (LHC) is widely acknowledged as the standard of care in the treatment of benign and malignant colorectal diseases. Currently, extracorporeal anastomosis (EA) is the most common technique for anastomotic closure in LHC [2]. It entails exteriorisation of the bowel outside the abdominal cavity, which facilitates visual assessment of bowel viability for anastomosis and decreases the risk of colonic content leakage [3]. However, the increased mesenteric tractions can result in serosal injuries, mesenteric bleeding and devascularisation that contributes to postoperative ileus [4]. This increased traction is further associated with other morbidities such as pain-associated reduction in pulmonary function and wound infections leading to prolonged hospital stay [5,6,7]. In addition, the midline extraction site, an option chosen more frequently as compared to other sites for the accommodation of the transverse colon extraction in EA, is associated with higher rates of incisional hernias [8].
In contrast, by obviating the need for external bowel mobilisation, intracorporeal anastomosis (IA) confers notable advantages such as a decreased risk of ileus [4]. Moreover, a midline extraction site is not a requirement for IA. However, IA is known to be technically demanding as it requires a mastery of skills in laparoscopic surgery especially in hand-sewn suturing [9]. Other drawbacks include an inability to perform bowel palpation prior to transection as well as exposure of peritoneum to potential intraluminal contamination [10]. Nevertheless, a majority of previous meta-analyses that compared IA and EA showed better postoperative recovery and fewer complications [11, 12, 13, 14, 15•, 16] when IA is used.
However, existing meta-analysis have not extensively analysed clinically important outcomes such as lymph node yield and have reached differing conclusions about the impact of IA on anatomic leak rates [16]. This review provides an updated comparison between IA and EA by including papers published in the previous 2 years, utilising a more comprehensive search that comprises both right- and left-sided, robotic assisted, and laparoscopic hemicolectomies, and analysing hitherto unreported outcomes such as lymph node yield. Sensitivity analysis was conducted, when appropriate (at least two papers in a subgroup is required), to reduce biases and better estimate the true effect-size. Meta-regression which was lacking in previous reviews was used to examine the potential sources of heterogeneity in our findings and to identify patient characteristics most suited for IA.
Materials and Methods
Search Strategy
This systematic review and meta-analysis follows the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) guidelines [17]. Relevant articles were identified by searching Medline, Embase and Cochrane electronic databases from inception through 21 September 2020. Search terms consisted of MeSH terms and keywords relating to “Colectomy” or “Laparoscopy” or “Intracorporeal anastomosis” and “Extracorporeal anastomosis”. The detailed search strategies are presented in Online Resource 1. All relevant abstracts were imported into EndNote X9 and duplicates were removed.
Criteria for the Selection of Studies
Studies were eligible for inclusion if (1) patients underwent elective laparoscopic or robotic right- or left-sided hemicolectomies, (2) studies compared outcomes of interest between intracorporeal and extracorporeal anastomosis, (3) studies were randomized controlled trials (RCTs) or observational studies (cohort and case–control studies only), and (4) written in the English language. Studies were excluded if they (1) were review articles, conference abstracts, non-human studies, case reports, or cross-sectional studies; (2) included emergency surgeries; (3) lacked comparison between intracorporeal and extracorporeal anastomosis; (4) included only open surgery in comparison; (5) did not analyse postoperative complications; or (6) were written in languages apart from the English language.
Intraoperative, postoperative and long-term recovery outcomes were extracted and analysed. Intraoperative outcomes included operation time, incision size, conversion to open surgery, number of lymph nodes harvested and intraoperative blood loss. A broad range of postoperative complications were examined, encompassing anastomotic leak rate, surgical site infections, dehiscence, intra-abdominal abscess, pneumonia, deep vein thrombosis, pulmonary embolism, acute myocardial infarction, overall bleeding, anastomotic bleeding, intra-abdominal bleeding, gastrointestinal bleeding, evisceration, ileus, bowel obstruction, Clostridioides difficile colitis, postoperative blood transfusions, urinary tract infections, urinary retention, reoperations, time to oral feeding, time to bowel movement, time to flatus, time to mobilisation, time to intestinal recovery and length of hospital stay. Long-term recovery outcomes comprised incidence of hernia formation and readmissions.
Data Extraction and Assessment of Quality
Predefined data were extracted from the selected articles into a structured proforma by TL and SAU independently. Data extracted from each paper included the general information of the study (author’s name, article title, publication year, geographical region of the study, study design and indication for surgery), patient demographics (age, gender and body mass index (BMI)), clinical characteristics, operative details and outcomes of interest, as defined above. For continuous outcomes where mean and SD were not available, available data were transformed according to existing formulae; median and range were transformed using formulae of Hozo et al. [18], while median and interquartile range were transformed using calculations formulae of Wan et al. [19]. Quality assessment for the included articles was done by two independent authors using the Newcastle–Ottawa Quality Assessment Form for Cohort Studies [20] and the Jadad Scale [21] for clinical trials. The Newcastle–Ottawa Quality Assessment Form for Cohort Studies [20] is designed to assess the quality of nonrandomised studies in meta-analyses and evaluates studies on three domains: the selection of the study groups; the comparability of the groups; the outcome of interest for cohort studies. The Jadad scale [21] for randomised controlled trials is created to assess the methodological quality of a clinical trial, by assessing the effectiveness of blinding.
Statistical Analysis
Meta-analyses and sensitivity analyses were performed with Cochrane Review Manager 5.3 [22] while meta-regression analyses were conducted using RStudio [23]. OR and corresponding 95% CIs were calculated for dichotomous variables while weighted mean differences (WMD) and corresponding 95% CI were calculated for continuous variables. The DerSimonian–Laird (DL) random-effects method [24] was then used to estimate pooled ORs and pooled WMDs regardless of inter-study heterogeneity as assessed by Cochran Q statistics and I2 statistics. Significance was determined by p < 0.05. Studies were characterised based on study type (RCT or observational study) and location of colectomy (right or left sided) for sensitivity analyses in several outcomes including lymph node yield, anastomotic leak rate, reoperations and readmissions. Meta-regression analyses were subsequently conducted for selected outcomes with variance estimates obtained using the DL approach and Knapp–Hartung method. Publication bias was assessed by visual inspection of funnel plots (Online Resource 2).
Results
Study and Patient Characteristics
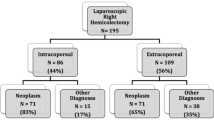
A summary of the study selection process can be found in Fig. 1. The search strategy yielded 579 records, of which the full texts of 367 manuscripts were evaluated. Forty-one retrospective cohort studies [25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65], three RCTs [66••, 67, 68] and three case–control studies [69,70,71] that fulfilled the predefined inclusion criteria were incorporated for final analysis, with 19 studies from Italy, 13 from the USA, three studies from Canada, two from Spain, two from Israel, and one each from France, China, Japan, Poland, Romania, Turkey, Argentina and Portugal. These 47 articles reported on 7001 patients of which 3152 underwent IA (45%) and 3849 underwent EA (55%). The key characteristics and quality assessments of included studies as well as demographic and operative variables of the patient population are detailed in Table 1. Most patients underwent hemicolectomy for benign growths, malignant neoplasia or inflammatory bowel disease.
Complications between Patients in IA Group versus EA Group
Full results for all intraoperative, postoperative and long-term recovery outcomes are reported in Table 2. Key results are further detailed below.
Intraoperative Complications
Pooled analysis of 6570 patients (42 studies; 2957 with IA and 3613 with EA) revealed a significantly longer operative time in the IA group compared to the EA group (p < 0.01; WMD = 13.32 min; 95% CI, 6.57 to 20.06, I2 = 92%). Among the 2467 patients (21 studies; 1187 with IA and 1280 with EA) evaluated, a significantly smaller incision size was found in the IA group compared to the EA group (p < 0.01; WMD = − 2.21 cm; 95% CI, − 2.85 to − 1.58, I2 = 97%). Evaluation of 4509 patients (24 studies; 2081 with IA and 2428 with EA) revealed that IA was associated with a significantly lower odds of conversion to open surgery compared to EA (p < 0.01; OR = 0.33; 95% CI, 0.24 to 0.46, I2 = 56%). Assessment of 3386 patients (21 studies; 1479 with IA and 1907 with EA) revealed a lower estimated blood loss for patients in the IA group compared with the EA group (p < 0.01; WMD = − 15.73 ml; 95% CI, − 21.53 to − 9.93, I2 = 49%).
The number of lymph nodes harvested in pooled analysis of 3394 patients (25 studies; 1706 with IA and 1688 with EA) undergoing colectomy for colon cancer was significantly higher in the IA group compared with the EA group (p < 0.01; WMD = 1.29; 95% CI, 0.46 to 2.12, I2 = 72%; Fig. 2). Sensitivity analysis of right-sided colectomies confirmed that lymph node yield was greater in the IA groups (p < 0.01; WMD = 1.33; 95% CI, 0.43 to 2.22, I2 = 74%; Online Resource 4 (Supplementary Fig. 2b)).
Postoperative Complications
Analysis of 5839 patients (33 studies; 2579 with IA and 3260 with EA) showed a significantly lower odds of anastomotic leakage in the IA group compared to the EA group (p < 0.01; OR = 0.60; 95% CI, 0.42 to 0.87, I2 = 0%; Fig. 3). However, sensitivity analysis of three RCTs revealed no significant difference in the odds of anastomotic leakage between the two approaches (p = 0.47; OR = 1.63; 95% CI, 0.43 to 6.22, I2 = 34%; Online Resource 4 (Supplementary Fig. 3c)). The odds of surgical site and wound infection among 6342 patients (38 studies; 2759 with IA and 3583 with EA) was lower in the IA group compared with the EA group (p < 0.01; OR = 0.62; 95% CI, 0.49 to 0.78, I2 = 21%; Fig. 4). The odds of other postoperative complications including intra-abdominal abscess, postoperative ileus, bowel obstruction, postoperative blood transfusion and grade 4 to 5 Clavien–Dindo complications were found to be similar in both groups. Out of 436 patients (4 studies; 193 with IA and 243 with EA) evaluated, the odds of gastrointestinal (GI) bleeding was found to be lower in the IA group compared to the EA group (p = 0.02; OR = 0.27; 95% CI, 0.09 to 0.83, I2 = 0%). Investigation of 3356 patients (18 studies; 1417 with IA and 1939 with EA) revealed that patients in the IA group had a significantly lower odds of undergoing reoperation compared with the EA group (p = 0.02; OR = 0.60; 95% CI 0.40 to 0.91, I2 = 0%; Fig. 5). Sensitivity analysis of right-sided colectomies likewise indicated reduced odds of reoperations in the IA group (p = 0.02; OR = 0.60; 95% CI 0.39 to 0.92, I2 = 0%; Online Resource 4 (Supplementary Fig. 5b)). Sensitivity analysis with solely RCTs was not possible because only Bollo et al. reported reoperation rates [72].
Analysis of 2079 patients (13 studies; 862 with IA and 1217 with EA) revealed that IA was associated with a shorter time to oral feeding compared with EA (p < 0.01; WMD = − 0.74 days; 95% CI, − 1.09 to − 0.39, I2 = 73%). Among the 4059 patients (22 studies; 1722 with IA and 2337 with EA), it was observed that a significantly shorter time to first bowel movement was found in the IA group compared to the EA group (p < 0.01; WMD = − 15.04 h; 95% CI, − 19.11 to − 10.96, I2 = 71%). Pooled analysis of 3584 patients (24 studies; 1620 with IA and 1964 with EA) revealed that IA patients experienced significantly shorter time to first flatus compared to the EA patients (p < 0.01; WMD = − 8.28 h; 95% CI, − 12.07 to − 4.50, I2 = 83%). Evaluating 1362 patients (4 studies; 564 with IA and 798 with EA), a significantly shorter time to mobilisation was found in the IA group compared with the EA group (p = 0.04; WMD = − 0.28 days; 95% CI, − 0.55 to − 0.01, I2 = 79%). Investigations of 5848 patients (40 studies; 2534 with IA and 3314 with EA) revealed that IA was associated with a significantly shorter length of stay compared to the EA group (p < 0.01; WMD = − 0.83 days; 95% CI, − 1.08 to − 0.57, I2 = 67%). Among the 199 patients (2 studies; 83 with IA and 116 with EA) evaluated, no significant difference was found in the time to intestinal function recovery between the IA group and the EA group (p = 0.06; WMD = − 1.12 days; 95% CI, − 2.29 to 0.06, I2 = 98%).
Long-Term Recovery
The odds of hernias in 1792 patients (16 studies; 880 with IA and 912 with EA) was significantly lower in the IA group compared to the EA group (p < 0.01; OR = 0.28; 95% CI, 0.18 to 0.44, I2 = 0%). A total of 2081 patients (17 studies; 895 with IA and 1186 with EA) were evaluated, and it was found that patients in the IA group had a significantly lower odds of readmissions compared with the EA group (p = 0.01; OR = 0.57; 95% CI 0.37 to 0.89, I2 = 0%; Fig. 6). However, sensitivity analysis of RCTs revealed no significant difference of the odds of readmissions between IA and EA (p = 0.26; OR = 0.32; 95% CI, 0.05 to 2.28, I2 = 30%; Online Resource 4 (Supplementary Fig. 6c)).
Meta-Regression Analyses
Meta-regression analyses of study and patient characteristics (sample size, age, gender, BMI, aetiology, tumour size, prior abdominal surgery, ASA class) with key outcomes (operative time, anastomotic leak, number of lymph nodes harvested, readmissions and reoperations) were conducted and reported in Online Resource 3.
A significant association was found between BMI and mean number of lymph nodes harvested (25 studies; β = 0.427, SE = 0.0992, p = 0.0003; Online Resource 4 (Supplementary Fig. 2d)), showing that the increased lymph node yield seen in IA is more pronounced for patients with higher BMI. Rate of readmissions was also found to be significantly associated with BMI (14 studies; β = − 0.3628, SE = 0.1123, p = 0.0072; Online Resource 4 (Supplementary Fig. 6d)) which indicates that the lowered readmission linked to IA is more evident in patients with increased BMI. Further analyses failed to highlight any significant associations between the remaining patient-specific factors and key outcomes.
Discussion and Conclusion
Minimally invasive surgery (MIS), which comprises laparoscopic and robotic approaches, has emerged as an increasingly popular alternative to open colectomies for the treatment of both benign and malignant colonic diseases. In MIS, resection is followed by an anastomotic procedure, either IA or EA, to restore intestinal continuity. The benefits of transitioning from the prevailing EA technique to its more modern IA counterpart have been reviewed by numerous studies but are still a matter of debate. This paper found a significant improvement in intraoperative outcomes for the IA group pertaining to reduced blood loss, lower rates of conversion to open surgery and smaller incision sizes, in addition to a reduction in the risks of postoperative and long-term surgical complications such as hernias, SSI and wound infections (Table 2) which are largely consistent with current literature [15•, 16]. In the present study, a significantly improved lymph node yield in favour of IA was additionally found (Fig. 2). Not surprisingly, AL rates remained similar after subgroup analysis of RCTs (Online Resource 4 (Supplementary Fig. 3c)), which is contrary to previous meta-analysis [16], while meta-regression analysis further suggested that the advantages of IA may be more pronounced in patients with higher BMI.
Sufficient lymph node yield is important for precise cancer staging, containment of metastatic tumour spread [73, 74] and serves as an accurate prognostic marker [75], yet it has been poorly investigated in previous meta-analyses. The present study found only two reviews, Wu et al. and Ricci et al., that analysed the lymph node yield of 1006 patients from 8 studies and 1532 patients from 12 studies, respectively. Both separately concluded that there were no significant differences between IA and EA for this outcome [12, 76]. In contrast, this meta-analysis incorporated 3394 patients from 25 studies and, with improved statistical power, found a significantly increased number of lymph nodes harvested in IA compared with EA (Fig. 2). While the anastomotic technique (IA or EA) itself is unlikely to have a direct effect on the lymph node harvest, it is possible that the increased ease of performing central vascular ligation (CVL) in IA due to clearer visualisation of the mesenteric root when the bowel is not externalized [77] plays a role. CVL involves central ligation of the main arteries and veins at their roots, thus allowing for more meticulous lymph node dissection [77]. Along with an increased uptake of intermediate and central nodes through higher ligation of the vascular pedicle [78], the improvement in the lymph node yield can be explained. These results contribute to the existing base of evidence supporting the use of IA for radical dissection of colonic neoplasms.
Another infrequently analysed outcome is the readmission rate which was lower in the IA group in the primary meta-analysis (Fig. 6). While this may be a consequence of improved intraoperative characteristics and reduced postoperative complications [79], a sensitivity analysis of three RCTs showed no significant difference between readmission rates (Online Resource 4 (Supplementary Fig. 6c)). Further large-scale RCTs are required to determine the true effect of IA on readmission rates.
Meta-regression analyses of cohort studies and RCTs additionally revealed that the advantages of increased lymph node yield and lowered readmission rate in IA are more prominent in patients with higher BMI (Online Resource 4 (Supplementary Fig. 2d) and Online Resource 4 (Supplementary Fig. 6d)), suggesting that IA may be especially beneficial for obese individuals. For patients with thicker abdominal walls as seen in those with higher BMI, the difference in incision lengths required for IA and EA is further pronounced. This is because intracorporeal dissection in IA enables extraction of specimens one limb at a time whereby a smaller incision can be made, while simultaneous evisceration of both limbs of the bowel to facilitate extracorporeal dissection in EA necessitates a longer incision. This translates to poorer postoperative outcomes such as readmission rates among patients with higher BMI. In addition, thickened abdominal pannus renders exteriorisation of the specimen difficult [80], making extracorporeal vessel root ligation and wider mesenteric dissection more challenging. As such, a larger number of lymph nodes can be harvested in IA for these patients.
IA was also associated with lower odds of GI bleeding and improved intestinal recovery which manifested as reduced time to oral feeding, time to first bowel movement, time to first flatus, time to mobilisation and length of stay. By obviating the need for bowel exteriorisation, a process that introduces additional tractional forces and physical trauma, intracorporeal anastomosis reduces the probability of traction injury to mesenteric blood supply and decreases the risk of serosal tears thereby improving postoperative function [16]. The present study also found that IA was also associated with decreased odds of hernia formation, SSI and wound infection (Fig. 4). The use of smaller incisions, and Pfannenstiel instead of midline incisions, may explain these results [81, 82]. Pfannenstiel incisions also provide superior cosmesis and reduced postoperative pain [83]; however, these outcomes could not be evaluated in this meta-analysis.
Emile et al. [16] and Aiolfi et al. [15•] conducted a meta-analysis of cohort studies and found that IA reduced AL rates but had no significant effect on reoperation rates. This study’s primary analysis of cohort studies supports their AL finding (Fig. 3) but indicated that IA significantly reduces reoperation rates (Fig. 5). Sensitivity analysis of AL excluding all cohort studies, however, showed that the odds were not significantly different in combined analysis of the three included RCTs (Online Resource 4 (Supplementary Fig. 3c)). AL and reoperation rates are most commonly affected by factors affecting wound healing such as the surgical technique and experience, preoperative steroid use [84] as well as patient comorbidities [85]; there is no a priori reason why these factors would be affected by bowel exteriorisation. The decreased AL rate observed in the primary meta-analysis was instead likely due to methodological and operative differences between study groups that occur in non-randomised cohort studies, for instance an oversewing of the anastomosis in IA as suggested by Grams et al. [86]. A biased AL rate could additionally explain for biased reoperation rate, although this could not be confirmed due to insufficient RCTs included in this review. This meta-analysis thus suggests that the choice to restore intestinal continuity intracorporeally or extracorporeally does not affect the odds of AL or reoperation rates between IA and EA.
It has been suggested that intra-abdominal anastomosis formation could lead to increased risk of intraperitoneal opening of contaminated bowel [87], thereby increasing intra-abdominal abscess rates. However, this meta-analysis found no evidence to support this. Antibiotic prophylaxis [88,89,90] and mechanical bowel preparation [90, 91] thus appear to be adequate in curtailing the risk of infection when appropriate care and precautions are taken during surgery. Another major concern is the technical difficulty of laparoscopic suturing in IA as opposed to manual sewing in EA, which could increase the operative time of IA. This study confirms this finding and quantifies that IA takes 13 min longer than EA. While familiarisation with IA would help overcome the steep learning curve and should narrow the mean difference in operation time between the two groups [92], no significant association was found when total sample size was used as a surrogate measure for experience in meta-regression. Further studies are warranted to investigate this and should account for the overall surgical experience of the surgeon with precise characterisation of intraoperative and postoperative outcomes for individual surgeons.
Strengths and Limitations
This paper updates the preceding meta-analyses by including recent studies published in the past 2 years. By investigating a comprehensive range of outcomes and utilising a broadened search criterion, our review provides clarification on the clinical benefits of IA through more accurate evaluation based on a larger pool of evidence. Sensitivity analysis of RCTs overcame selection and operative biases in non-randomised cohort studies to better estimate the true effect of IA for contentious outcomes. Meta-regression identified obese patients as a patient subtype for whom IA may be more efficacious, and this should be investigated in future studies. Separately, the use of meta-regression analyses also helped to identify potential confounding factors that may lead to biased findings.
This paper presents with a few principal limitations, mostly pertaining to significant heterogeneity (reflected by the high I2 values of selected outcomes) across studies, which have been considered through subgroup and regression analysis. Included studies failed to provide details regarding the definition or clinical measurement of outcomes such as hernia formation as well as incision size, and in many cases did not standardise or report surgeon experience. Imprecise definitions of conversion to open surgery protocols as well as a lack of postoperative management protocol could have affected the postoperative recovery, complication rates and long-term recovery outcomes. However, it was likely that all colorectal surgery patients were managed by the same protocols, limiting the risk that differences in postoperative outcomes between IA and EA patients are due to such protocol differences. The vast majority of included studies are cohort studies which are inherently at risk for both case selection bias and possible surgeon bias. Sensitivity analysis of RCTs helped to mitigate this problem for selected outcomes. Lastly, we are unable to comment if the improvement in postoperative outcomes is clinically significant due to lack of well-defined minimal clinically important differences (MCID).
Conclusion
In the context of LHC, this updated meta-analysis strengthens existing findings that IA superior to EA and reveals for the first time that IA is associated with an ameliorated lymph node yield without a difference in anastomotic leak rates. The benefits of IA pertaining to readmission rate and lymph node yield may also be more pronounced in patients with higher BMI while large-scale, randomised controlled trials are warranted to further verify these findings.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Blackmore AE, Wong MTC, Tang CL. “Evolution of laparoscopy in colorectal surgery: an evidence-based review,” (in eng). World J Gastroenterol. 2014;20(17):4926–33. https://doi.org/10.3748/wjg.v20.i17.4926.
Masubuchi S, Okuda J, Hamamoto H, Ishii M, Osumi W, Yamamoto M, et al. Intracorporeal versus extracorporeal anastomosis in laparoscopic left colectomy forleft-side colon cancer: a retrospective study. Clin Surg. 2019;4:2506.
Brown RF, Cleary RK. “Intracorporeal anastomosis versus extracorporeal anastomosis for minimally invasive colectomy,” (in eng). J Gastrointest Oncol. 2020;11(3):500–7. https://doi.org/10.21037/jgo.2019.12.02.
Cleary RK, et al. “Intracorporeal versus extracorporeal anastomosis for minimally invasive right colectomy: a multi-center propensity score-matched comparison of outcomes,” (in eng). PLoS ONE. 2018;13(10):e0206277. https://doi.org/10.1371/journal.pone.0206277.
Van Leersum NJ, et al. “The Dutch surgical colorectal audit,” (in eng). Eur J Surg Oncol. 2013;39(10):1063–70. https://doi.org/10.1016/j.ejso.2013.05.008.
Vlug MS, et al. “Laparoscopy in combination with fast track multimodal management is the best perioperative strategy in patients undergoing colonic surgery: a randomized clinical trial (LAFA-study),” (in eng). Ann Surg. 2011;254(6):868–75. https://doi.org/10.1097/SLA.0b013e31821fd1ce.
Kennedy RH, et al. “Multicenter randomized controlled trial of conventional versus laparoscopic surgery for colorectal cancer within an enhanced recovery programme: EnROL,” (in eng). J Clin Oncol. 2014;32(17):1804–11. https://doi.org/10.1200/jco.2013.54.3694.
Samia H, Lawrence J, Nobel T, Stein S, Champagne BJ, Delaney CP. Am J Surg. 2013;205(3):264–7. https://doi.org/10.1016/j.amjsurg.2013.01.006 (discussion 268).
Jian-Cheng T, Shu-Sheng W, Bo Z, Jian F, Liang Z. “Total laparoscopic right hemicolectomy with 3-step stapled intracorporeal isoperistaltic ileocolic anastomosis for colon cancer: an evaluation of short-term outcomes,” (in eng). Medicine. 2016;95(48):e5538–e5538. https://doi.org/10.1097/MD.0000000000005538.
R. F. Brown and R. K. Cleary, "Intracorporeal anastomosis versus extracorporeal anastomosis for minimally invasive colectomy," Journal of Gastrointestinal Oncology; Vol 11, No 3 (June 2020): Journal of Gastrointestinal Oncology, 2020. [Online]. Available: https://jgo.amegroups.com/article/view/35090.
"Intracorporeal versus extracorporeal anastomosis in laparoscopic right colectomy: an updated systematic review and cumulative meta-analysis," J Laparoendosc Ad Surg Tech 2020, 30 4 402–412. https://doi.org/10.1089/lap.2019.0693
Wu Q, Jin C, Hu T, Wei M, Wang Z. “Intracorporeal versus extracorporeal anastomosis in laparoscopic right colectomy: a systematic review and meta-analysis,” (in eng). J Laparoendosc Adv Surg Tech A. 2017;27(4):348–57. https://doi.org/10.1089/lap.2016.0485.
van Oostendorp S, et al. “Intracorporeal versus extracorporeal anastomosis in right hemicolectomy: a systematic review and meta-analysis,” (in eng). Surg Endosc. 2017;31(1):64–77. https://doi.org/10.1007/s00464-016-4982-y.
Feroci F, Lenzi E, Garzi A, Vannucchi A, Cantafio S, Scatizzi M. “Intracorporeal versus extracorporeal anastomosis after laparoscopic right hemicolectomy for cancer: a systematic review and meta-analysis,” (in eng). Int J Colorectal Dis. 2013;28(9):1177–86. https://doi.org/10.1007/s00384-013-1651-7.
• Aiolfi A, et al. “Intracorporeal versus extracorporeal anastomosis in laparoscopic right colectomy: an updated systematic review and cumulative meta-analysis,” (in eng). J Laparoendosc Adv Surg Tech A. 2020;30(4):402–12. https://doi.org/10.1089/lap.2019.0693. (The most recent systematic review conducted at the time of writing this paper. Unlike previous reviews, it reported no significant differences in key outcomes such as anastomotic leakage. Lymph node yield was not analysed.)
Emile SH, et al. “Intracorporeal versus extracorporeal anastomosis in minimally invasive right colectomy: an updated systematic review and meta-analysis,” (in eng). Tech Coloproctol. 2019;23(11):1023–35. https://doi.org/10.1007/s10151-019-02079-7.
Moher D, Liberati A, Tetzlaff J, Altman DG, The PG. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009;6(7):e1000097. https://doi.org/10.1371/journal.pmed.1000097.
Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5(1):13. https://doi.org/10.1186/1471-2288-5-13.
Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. 2014;14(1):135. https://doi.org/10.1186/1471-2288-14-135.
"Wells G SB, O’Connell D, Peterson J, Welch V, Losos M, Tugwell P. (2013) The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. [Online]. Available: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp.
Jadad AR, et al. “Assessing the quality of reports of randomized clinical trials: is blinding necessary?,” (in eng). Control Clin Trials. 1996;17(1):1–12. https://doi.org/10.1016/0197-2456(95)00134-4.
T. C. C. Review Manager (RevMan) [Computer program]. Version 5.3. Copenhagen: The Nordic Cochrane Centre, 2014., 2014.
P. RStudio Team (2020). RStudio: Integrated Development Environment for R. RStudio, Boston, MA URL http://www.rstudio.com/.
DerSimonian R, Laird N. “Meta-analysis in clinical trials,” (in eng). Control Clin Trials. 1986;7(3):177–88. https://doi.org/10.1016/0197-2456(86)90046-2.
Akram WM, et al. “A propensity score-matched comparison of intracorporeal and extracorporeal techniques for robotic-assisted right colectomy in an Enhanced Recovery Pathway,” (in English). Am J Surg, Article. 2018;216(6):1095–100. https://doi.org/10.1016/j.amjsurg.2018.06.010.
Anania G, et al. “A totally mini-invasive approach for colorectal laparoscopic surgery,” (in English). World J Gastroenterol, Article. 2012;18(29):3869–74. https://doi.org/10.3748/wjg.v18.i29.3869.
N. Bou Saleh et al., "Intracorporeal versus extracorporeal anastomosis in laparoscopic right hemicolectomy: results from the CLIMHET study group," (in English). Tech Coloproctol 2020 24 6 585–592. https://doi.org/10.1007/s10151-020-02202-z.
Carlini M, Spoletini D, Castaldi F, Giovannini C, Passaro U. “Laparoscopic resection of splenic flexure tumors,” (in English). Updates Surg, Article. 2016;68(1):77–83. https://doi.org/10.1007/s13304-016-0357-0.
R. K. Cleary et al., "Intracorporeal versus extracorporeal anastomosis for minimally invasive right colectomy: a multi-center propensity scorematched comparison of outcomes," (in English), PLoS One 2018 13 10. https://doi.org/10.1371/journal.pone.0206277.
Fagarasan V, et al. “Which is the optimal method of reconstruction after laparoscopic right hemicolectomy, the intracorporeal or extracorporeal anastomosis technique?,” (in English), Chirurgia (Bucharest, Romania : 1990). Article. 2020;115(4):493–504. https://doi.org/10.21614/chirurgia.115.4.493.
M. E. Franklin Jr et al., "Laparoscopic right hemicolectomy for cancer: 11-year experience," (in English). Rev Gastroenterol Mex 2004 69 Suppl 1 65–72. [Online]. Available: https://www.embase.com/search/results?subaction=viewrecord&id=L40414143&from=export.
Grams J, Tong W, Greenstein AJ, Salky B. “Comparison of intracorporeal versus extracorporeal anastomosis in laparoscopic-assisted hemicolectomy,” (in English). Surg Endosc, Article. 2010;24(8):1886–91. https://doi.org/10.1007/s00464-009-0865-9.
Grieco M, et al. “Intracorporeal versus extracorporeal anastomosis for laparoscopic resection of the splenic flexure colon cancer: a multicenter propensity score analysis,” (in English). Surgical Laparoscopy, Endoscopy and Percutaneous Techniques, Article. 2019;29(6):483–8. https://doi.org/10.1097/SLE.0000000000000653.
Hanna MH, et al. “Laparoscopic right hemicolectomy: short- and long-term outcomes of intracorporeal versus extracorporeal anastomosis,” (in English). Surg Endosc, Article. 2016;30(9):3933–42. https://doi.org/10.1007/s00464-015-4704-x.
M. Hellan, C. Anderson, and A. Pigazzi, "Extracorporeal versus intracorporeal anastomosis for laparoscopic right hemicolectomy," (in English). J Soc Laparoendosc Surg 2009 Article vol. 13, no. 3, pp. 312–317. [Online]. Available: https://www.embase.com/search/results?subaction=viewrecord&id=L355484774&from=exporthttp://docserver.ingentaconnect.com/deliver/connect/sls/10868089/v13n3/s6.pdf?expires=1255556505&id=52532148&titleid=75000538&accname=Elsevier+Science&checksum=C8AA576B818E1B929896AF4D710263AB.
T. Jian-Cheng, W. Shu-Sheng, Z. Bo, F. Jian, and Z. Liang, "Total laparoscopic right hemicolectomy with 3-step stapled intracorporeal isoperistaltic ileocolic anastomosis for colon cancer: an evaluation of short-term outcomes." Medicine 2016 95 48 e5538. [Online]. Available: http://ovidsp.ovid.com/ovidweb.cgi?T=JS&CSC=Y&NEWS=N&PAGE=fulltext&D=med13&AN=27902621http://bb2sz3ek3z.search.serialssolutions.com/?url_ver=Z39.88-2004&rft_val_fmt=info:ofi/fmt:kev:mtx:journal&rfr_id=info:sid/Ovid:med13&rft.genre=article&rft_id=info:doi/10.1097%2FMD.0000000000005538&rft_id=info:pmid/27902621&rft.issn=0025-7974&rft.volume=95&rft.issue=48&rft.spage=e5538&rft.pages=e5538&rft.date=2016&rft.jtitle=Medicine&rft.atitle=Total+laparoscopic+right+hemicolectomy+with+3-step+stapled+intracorporeal+isoperistaltic+ileocolic+anastomosis+for+colon+cancer%3A+An+evaluation+of+short-term+outcomes.&rft.aulast=Jian-Cheng.
Kayano H, et al. “Short- and long-term outcomes of 2-step stapled intracorporeal versus extracorporeal anastomosis in laparoscopic colectomy for colon cancer,” (in English). Anticancer Res, Article. 2019;39(11):6393–401. https://doi.org/10.21873/anticanres.13853.
S. R. Kelley, E. Duchalais, and D. W. Larson, "Short-term outcomes with robotic right colectomy," (in English). Am Surg 2018 84 11 1768–1773. [Online]. Available: https://www.embase.com/search/results?subaction=viewrecord&id=L625451368&from=export.
Krouchev R, Tamana S, Nguyen N, Yorke E, Harris D, Sampath S. “Total intracorporeal anastomosis for right hemicolectomy: experience from a Canadian center,” (in English). Am J Surg, Article. 2018;215(5):905–8. https://doi.org/10.1016/j.amjsurg.2018.02.013.
Kwiatkowski AP, Stępińska G, Stanowski E, Paśnik K, Janik MR. “Intracorporeal versus extracorporeal anastomosis in laparoscopic right hemicolectomy – single center experience,” (in English). Wideochirurgia I Inne Techniki Maloinwazyjne, Article. 2019;14(3):381–6. https://doi.org/10.5114/wiitm.2019.81725.
Lee KH, Ho J, Akmal Y, Nelson R, Pigazzi A. “Short- and long-term outcomes of intracorporeal versus extracorporeal ileocolic anastomosis in laparoscopic right hemicolectomy for colon cancer,” (in English). Surg Endosc, Article. 2013;27(6):1986–90. https://doi.org/10.1007/s00464-012-2698-1.
H. J. Lujan et al., "Advantages of robotic right colectomy with intracorporeal anastomosis". Surg Laparosc Endosc Percutan Tech, Comparative Study 2018 28 1 36–41. [Online]. Available: http://ovidsp.ovid.com/ovidweb.cgi?T=JS&CSC=Y&NEWS=N&PAGE=fulltext&D=med15&AN=28319493http://bb2sz3ek3z.search.serialssolutions.com/?url_ver=Z39.88-2004&rft_val_fmt=info:ofi/fmt:kev:mtx:journal&rfr_id=info:sid/Ovid:med15&rft.genre=article&rft_id=info:doi/10.1097%2FSLE.0000000000000384&rft_id=info:pmid/28319493&rft.issn=1530-4515&rft.volume=28&rft.issue=1&rft.spage=36&rft.pages=36-41&rft.date=2018&rft.jtitle=Surgical+Laparoscopy%2C+Endoscopy+%26+Percutaneous+Techniques&rft.atitle=Advantages+of+Robotic+Right+Colectomy+With+Intracorporeal+Anastomosis.&rft.aulast=Lujan.
Magistro C, et al. “Totally laparoscopic versus laparoscopic-assisted right colectomy for colon cancer: is there any advantage in short-term outcomes? A prospective comparative assessment in our center,” (in English). Surg Endosc, Article. 2013;27(7):2613–8. https://doi.org/10.1007/s00464-013-2799-5.
V. Malerba et al., "Extracorporeal versus intracorporeal anastomosis after laparoscopic right hemicolectomy: cost-effectiveness analysis," (in English). Ann Ital Chir 2020 91 49–54. [Online]. Available: https://www.embase.com/search/results?subaction=viewrecord&id=L631268119&from=export.
Mari GM, et al. “Intracorporeal anastomosis reduces surgical stress response in laparoscopic right hemicolectomy: a prospective randomized trial,” (in English). Surg Laparosc Endosc Percutaneous Tech. 2018;28(2):77–81. https://doi.org/10.1097/SLE.0000000000000506.
Martinek L, You K, Giuratrabocchetta S, Gachabayov M, Lee K, Bergamaschi R. “Does laparoscopic intracorporeal ileocolic anastomosis decreases surgical site infection rate? A propensity score-matched cohort study,” (in English). Int J Colorectal Dis, Article. 2018;33(3):291–8. https://doi.org/10.1007/s00384-017-2957-7.
M. Milone et al., "Intracorporeal versus extracorporeal anastomosis after laparoscopic left colectomy for splenic flexure cancer: results from a multi-institutional audit on 181 consecutive patients." Surg Endosc 2018 Article In Press 1‐7. https://doi.org/10.1007/s00464-018-6065-8.
M. Milone et al., "Intracorporeal versus extracorporeal anastomosis. Results from a multicentre comparative study on 512 right-sided colorectal cancers." Surg Endosc, Comparative Study Multicenter Study 2015 29 8 2314–20. [Online]. Available: http://ovidsp.ovid.com/ovidweb.cgi?T=JS&CSC=Y&NEWS=N&PAGE=fulltext&D=med12&AN=25414066http://bb2sz3ek3z.search.serialssolutions.com/?url_ver=Z39.88-2004&rft_val_fmt=info:ofi/fmt:kev:mtx:journal&rfr_id=info:sid/Ovid:med12&rft.genre=article&rft_id=info:doi/10.1007%2Fs00464-014-3950-7&rft_id=info:pmid/25414066&rft.issn=0930-2794&rft.volume=29&rft.issue=8&rft.spage=2314&rft.pages=2314-20&rft.date=2015&rft.jtitle=Surgical+Endoscopy&rft.atitle=Intracorporeal+versus+extracorporeal+anastomosis.+Results+from+a+multicentre+comparative+study+on+512+right-sided+colorectal+cancers.&rft.aulast=Milone.
Rawlings AL, Woodland JH, Vegunta RK, Crawford DL. “Robotic versus laparoscopic colectomy,” (in English). Surg Endosc, Article. 2007;21(10):1701–8. https://doi.org/10.1007/s00464-007-9231-y.
Reitz ACW, Lin E, Rosen SA. “A single surgeon’s experience transitioning to robotic-assisted right colectomy with intracorporeal anastomosis,” (in English). Surg Endosc, Article. 2018;32(8):3525–32. https://doi.org/10.1007/s00464-018-6074-7.
Scotton G, Contardo T, Zerbinati A, Tosato SM, Orsini C, Morpurgo E. “From laparoscopic right colectomy with extracorporeal anastomosis to robot-assisted intracorporeal anastomosis to totally robotic right colectomy for cancer: the evolution of robotic multiquadrant abdominal surgery,” (in English). J Laparoendosc Ad Surg Tech. 2018;28(10):1216–22. https://doi.org/10.1089/lap.2017.0693.
Shapiro R, Keler U, Segev L, Sarna S, Hatib K, Hazzan D. “Laparoscopic right hemicolectomy with intracorporeal anastomosis: short- and long-term benefits in comparison with extracorporeal anastomosis,” (in English). Surg Endosc, Article. 2016;30(9):3823–9. https://doi.org/10.1007/s00464-015-4684-x.
Swaid F, Sroka G, Madi H, Shteinberg D, Somri M, Matter I. “Totally laparoscopic versus laparoscopic-assisted left colectomy for cancer: a retrospective review,” (in English). Surg Endosc, Article. 2016;30(6):2481–8. https://doi.org/10.1007/s00464-015-4502-5.
Trastulli S, et al. “Robotic right colectomy with intracorporeal anastomosis compared with laparoscopic right colectomy with extracorporeal and intracorporeal anastomosis: a retrospective multicentre study,” (in English). Surg Endosc, Article. 2015;29(6):1512–21. https://doi.org/10.1007/s00464-014-3835-9.
A. S. Vergis, S. N. Steigerwald, F. D. Bhojani, P. A. Sullivan, and K. M. Hardy, "Laparoscopic right hemicolectomy with intracorporeal versus extracorporeal anastamosis: a comparison of short-term outcomes," (in English), Canadian journal of surgery. Journal canadien de chirurgie 2015 58 1 63–68. [Online]. Available: https://www.embase.com/search/results?subaction=viewrecord&id=L609324784&from=export.
Vignali A, Elmore U, Lemma M, Guarnieri G, Radaelli G, Rosati R. “Intracorporeal versus extracorporeal anastomoses following laparoscopic right colectomy in obese patients: a case-matched study,” (in English). Dig Surg, Article. 2018;35(3):236–42. https://doi.org/10.1159/000479241.
Widmar M, et al. Intracorporeal anastomosis in minimally invasive right colectomies is associated with fewer incisional hernias and shorter length of stay. Dis Colon Rectum, Journal: Conference Abstract. 2018;61(5):e231. https://doi.org/10.1097/DCR.0000000000001104.
Fabozzi M, et al. “Comparison of short- and medium-term results between laparoscopically assisted and totally laparoscopic right hemicolectomy: a case-control study,” (in eng). Surg Endosc. 2010;24(9):2085–91. https://doi.org/10.1007/s00464-010-0902-8.
Erguner I, Aytac E, Baca B, Hamzaoglu I, Karahasanoglu T. “Total laparoscopic approach for the treatment of right colon cancer: a technical critique,” (in eng). Asian J Surg. 2013;36(2):58–63. https://doi.org/10.1016/j.asjsur.2012.09.004.
Roscio F, Bertoglio C, De Luca A, Frattini P, Scandroglio I. Totally laparoscopic versus laparoscopic assisted right colectomy for cancer. Int J Surg. 2012;10(6):290–5. https://doi.org/10.1016/j.ijsu.2012.04.020.
Patron Uriburu JC et al (2016) Intracorporeal and Extracorporeal Anastomosis During Right Laparoscopic Colectomy. Comparative Study. EC Gastroenterology and Digestive System 1(3):87–98.
Biondi A, Santocchi P, Pennestrì F, Santullo F, D’Ugo D, Persiani R. “Totally laparoscopic right colectomy versus laparoscopically assisted right colectomy: a propensity score analysis,” (in eng). Surg Endosc. 2017;31(12):5275–82. https://doi.org/10.1007/s00464-017-5601-2.
N. R. Inês Campos Gil, Diana Parente, Inês Sales, Paulo Alves, Paulo Clara, Sandra Amado, Miguel Coelho, Vitor Faria "Intracorporeal versus extracorporeal anastomosis in laparoscopic right colectomy: short-term outcomes." Surg Gastroenterol Oncol 2018 23.
G. Anania et al., "Extracorporeal versus intracorporeal anastomosis in laparoscopic right hemicolectomy for cancer," (in eng). Minim Invasive Ther Allied Technol 2020 1–7. https://doi.org/10.1080/13645706.2020.1757464.
Trépanier M, et al. “Intracorporeal versus extracorporeal anastomosis for right colectomy does not affect gastrointestinal recovery within an enhanced recovery after surgery program,” (in eng). Surg Endosc. 2020;34(10):4601–8. https://doi.org/10.1007/s00464-019-07204-z.
•• Allaix ME, et al. “Intracorporeal or extracorporeal ileocolic anastomosis after laparoscopic right colectomy: a double-blinded randomized controlled trial,” (in English). Ann Surg, Article. 2019;270(5):762–7. https://doi.org/10.1097/SLA.0000000000003519. (One of 3 major RCTs conducted. Showed that lymph node yield was not significantly different between IA and EA.)
Bollo J, et al. “Randomized clinical trial of intracorporeal versus extracorporeal anastomosis in laparoscopic right colectomy (IEA trial),” (in English). Br J Surg, Article. 2020;107(4):364–72. https://doi.org/10.1002/bjs.11389.
A. Vignali, M. Bissolati, P. De Nardi, S. Di Palo, and C. Staudacher, "Extracorporeal vs. intracorporeal ileocolic stapled anastomoses in laparoscopic right colectomy: an interim analysis of a randomized clinical trial," (in English). J Laparoendosc Adv Surg Tech 2016 26 5 343–348. https://doi.org/10.1089/lap.2015.0547.
J. Arredondo Chaves et al., "A case-control study of extracorporeal versus intracorporeal anastomosis in patients subjected to right laparoscopic hemicolectomy," (in Spanish). Cirugia Espanola 2011 89 1 24–30. https://doi.org/10.1016/j.ciresp.2010.10.003.
Morpurgo E, Contardo T, Molaro R, Zerbinati A, Orsini C, D’Annibale A. “Robotic-assisted intracorporeal anastomosis versus extracorporeal anastomosis in laparoscopic right hemicolectomy for cancer: a case control study,” (in English). J Laparoendosc Adv Surg Tech A. 2013;23(5):414–7. https://doi.org/10.1089/lap.2012.0404.
M. Scatizzi, K. C. Kröning, A. Borrelli, G. Andan, E. Lenzi, and F. Feroci, "Extracorporeal versus intracorporeal anastomosis after laparoscopic right colectomy for cancer: a case-control study," (in English). World J Surg 2010 34 12 2902–2908. [Online]. Available: https://www.embase.com/search/results?subaction=viewrecord&id=L360282807&from=export.
Bollo J, et al. “Randomized clinical trial of intracorporeal versus extracorporeal anastomosis in laparoscopic right colectomy (IEA trial),” (in eng). Br J Surg. 2020;107(4):364–72. https://doi.org/10.1002/bjs.11389.
Orsenigo E, Gasparini G, Carlucci M. “Clinicopathological factors influencing lymph node yield in colorectal cancer: a retrospective study,” (in eng). Gastroenterol Res Pract. 2019;2019:5197914. https://doi.org/10.1155/2019/5197914.
Wong SL. “Lymph node counts and survival rates after resection for colon and rectal cancer,” (in eng). Gastrointest Cancer Res. 2009;3(2 Suppl):S33–5.
B. D. Edge SB, Compton CC, "AJCC Cancer Staging Manual," Springer, 2010.
C. C. Ricci, R.: Alagna, V.: Zani, E.: Taffurelli, G.: Pacilio, C. A.: Minni, F., "A critical and comprehensive systematic review and meta-analysis of studies comparing intracorporeal and extracorporeal anastomosis in laparoscopic right hemicolectomy," (in English). Langenbeck's Archives of Surgery 2017 402 3 417–427. https://doi.org/10.1007/s00423-016-1509-x.
Z. G. W. Ow et al., "Comparing complete mesocolic excision versus conventional colectomy for colon cancer: a systematic review and meta-analysis," (in eng). Eur J Surg Oncol 2020. https://doi.org/10.1016/j.ejso.2020.09.007.
Widmar M, et al. “Lymph node yield in right colectomy for cancer: a comparison of open, laparoscopic and robotic approaches,” (in eng). Colorectal Dis. 2017;19(10):888–94. https://doi.org/10.1111/codi.13786.
Krell RW, Girotti ME, Fritze D, Campbell DA, Hendren S. “Hospital readmissions after colectomy: a population-based study,” (in eng). J Am Coll Surg. 2013;217(6):1070–9. https://doi.org/10.1016/j.jamcollsurg.2013.07.403.
Martin ST, Stocchi L. “Laparoscopic colorectal resection in the obese patient,” (in eng). Clin Colon Rectal Surg. 2011;24(4):263–73. https://doi.org/10.1055/s-0031-1295690.
Tonouchi H, Ohmori Y, Kobayashi M, Kusunoki M. “Trocar site hernia,” (in eng). Arch Surg. 2004;139(11):1248–56. https://doi.org/10.1001/archsurg.139.11.1248.
Singh R, Omiccioli A, Hegge S, McKinley C. “Does the extraction-site location in laparoscopic colorectal surgery have an impact on incisional hernia rates?,” (in eng). Surg Endosc. 2008;22(12):2596–600. https://doi.org/10.1007/s00464-008-9845-8.
Cirocchi R, et al. “Intracorporeal versus extracorporeal anastomosis during laparoscopic right hemicolectomy – systematic review and meta-analysis,” (in eng). Surg Oncol. 2013;22(1):1–13. https://doi.org/10.1016/j.suronc.2012.09.002.
Vasiliu EC, Zarnescu NO, Costea R, Neagu S. “Review of risk factors for anastomotic leakage in colorectal surgery,” (in eng). Chirurgia (Bucur). 2015;110(4):319–26.
Kryzauskas M, et al. “Risk factors for anastomotic leakage and its impact on long-term survival in left-sided colorectal cancer surgery,” (in eng). World J Surg Oncol. 2020;18(1):205. https://doi.org/10.1186/s12957-020-01968-8.
Grams J, Tong W, Greenstein AJ, Salky B. “Comparison of intracorporeal versus extracorporeal anastomosis in laparoscopic-assisted hemicolectomy,” (in eng). Surg Endosc. 2010;24(8):1886–91. https://doi.org/10.1007/s00464-009-0865-9.
I. Gil et al., "Intracorporeal versus extracorporeal anastomosis in laparoscopic right colectomy: short-term outcomes". Surg Gastroenterol Oncol 2018 23 71. https://doi.org/10.21614/sgo-23-1-71.
G. C. Scotton, T.: Zerbinati, A.: Tosato, S. M.: Orsini, C.: Morpurgo, E., "From laparoscopic right colectomy with extracorporeal anastomosis to robot-assisted intracorporeal anastomosis to totally robotic right colectomy for cancer: the evolution of robotic multiquadrant abdominal surgery," (in English). J Laparoendosc Ad Surg Tech 2018 28 10 1216–1222. https://doi.org/10.1089/lap.2017.0693.
R. K. Shapiro, U.: Segev, L.: Sarna, S.: Hatib, K.: Hazzan, D., "Laparoscopic right hemicolectomy with intracorporeal anastomosis: short- and long-term benefits in comparison with extracorporeal anastomosis," (in English). Surg Endosc 2016 30 9 3823–3829. https://doi.org/10.1007/s00464-015-4684-x.
S. C. Trastulli, A.: Guarino, S.: Piagnerelli, R.: Annecchiarico, M.: Coratti, F.: Di Marino, M.: Ricci, F.: Desiderio, J.: Cirocchi, R.: Parisi, A., "Robotic right colectomy with intracorporeal anastomosis compared with laparoscopic right colectomy with extracorporeal and intracorporeal anastomosis: a retrospective multicentre study," (in English). Surg Endosc 2015 29 6 1512–1521. https://doi.org/10.1007/s00464-014-3835-9.
L. Y. Martinek, K.: Giuratrabocchetta, S.: Gachabayov, M.: Lee, K.: Bergamaschi, R., "Does laparoscopic intracorporeal ileocolic anastomosis decreases surgical site infection rate? A propensity score-matched cohort study," (in English). Int J Colorectal Dis 2018 33 3 291–298. https://doi.org/10.1007/s00384-017-2957-7.
F. R. Jamali, A. M. Soweid, H. Dimassi, C. Bailey, J. Leroy, and J. Marescaux, "Evaluating the degree of difficulty of laparoscopic colorectal surgery," (in eng). Arch Surg 2008 143 8 762–7. discussion 768. https://doi.org/10.1001/archsurg.143.8.762.
Author information
Authors and Affiliations
Contributions
Conception and Design: Choon Seng Chong.
Acquisition of data: Timothy Jia Rong Lam, Shamill Amedot Udonwa.
Interpretation of data: Clyve Yu Leon Yaow, Kameswara Rishi Yeshayahu Nistala.
Drafting of article: Timothy Jia Rong Lam, Shamill Amedot Udonwa.
Critical revision of article: Clyve Yu Leon Yaow, Kameswara Rishi Yeshayahu Nistala, Choon Seng Chong.
Approval of final manuscript: Timothy Jia Rong Lam, Shamill Amedot Udonwa, Clyve Yu Leon Yaow, Kameswara Rishi Yeshayahu Nistala, Choon Seng Chong.
Corresponding author
Ethics declarations
Conflict of Interest
Timothy Jia Rong Lam, Shamill Amedot Udonwa, Clyve Yu Leon Yaow, Kameswara Rishi Yeshayahu Nistala, and Choon Seng Chong have nothing to disclose.
Human and Animal Rights and Informed Consent This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Surgery and Surgical Innovations in Colorectal Cancer
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lam, T.J.R., Udonwa, S.A., Yaow, C.Y.L. et al. Intracorporeal Versus Extracorporeal Anastomosis in Laparoscopic Colectomy: A Meta-Analysis and Systematic Review. Curr Colorectal Cancer Rep 18, 1–17 (2022). https://doi.org/10.1007/s11888-022-00473-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11888-022-00473-2