Abstract
Echocardiographic evaluation of diastolic properties and assessment of hemodynamic status of the right and the left ventricle have been traditionally applied for many years in clinical practice. Establishment of diagnosis of diastolic dysfunction, grading, and estimation of filling pressures noninvasively adds prognostic information to the clinician, which may affect treatment management. Novel methods, including left atrium strain, left ventricular diastolic strain rate, and left ventricular untwisting rate, have been imported in clinical practice attempting to provide a more comprehensive and more accurate understanding of the mechanisms and diagnosis of diastolic dysfunction.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Left ventricular (LV) diastolic properties were first characterized invasively using the rate of left ventricular pressure decline, LV relaxation time constant, and measurements of myocardial and chamber stiffness. Currently, noninvasive evaluation of diastolic function is used in the day-to-day evaluation of LV function. It is based on Doppler echocardiography and utilizes measurements of transmitral flow, pulmonary vein flow velocities and patterns, mitral annulus tissue Doppler imaging, and pulmonary artery pressure (PAP) estimation by echocardiography. A comprehensive approach that takes into consideration LV ejection fraction (EF), presence or absence of left ventricular hypertrophy, and left atrial (LA) volume is recommended for the grading of diastolic dysfunction and the estimation of LV filling pressures [1]. In addition, there are relatively novel indices including global diastolic strain rate (SR), regional diastolic strain rate, LV untwisting rate, and LA strain based on speckle tracking imaging that can be implemented to study LV diastolic function. In this article, we present a guideline-based approach to assess LV diastolic function and the emerging role for the more recently introduced parameters.
Grading Diastolic Function
The presence of diastolic dysfunction by Doppler echocardiography is not uncommon in the general population and has been associated with increased all-cause mortality [2]. Further, progression of diastolic dysfunction has been related to the development of heart failure symptoms [3] and to higher mortality in patients with normal EF [4•]. In a recent study, a simple marker of diastolic dysfunction, e′ velocity, was shown to be a significant predictor of fatal and nonfatal cardiovascular events in the general population [5•].
The initial step in diastolic function assessment is the evaluation of mitral annular e′ velocity by tissue Doppler, a marker that correlates significantly with the time constant of LV relaxation [6]. This is done in conjunction with biplane LA volume measurements. Patients with impaired diastolic function usually have a septal e′ velocity <8 cm/s and a lateral e′ <10 cm/s, and depending on the stage of the disease, LA volume index may be increased (≥34 ml/m2). However, tissue Doppler annular velocities are not reliable for evaluation of LV relaxation in patients with primary mitral valve disease and normal EF; patients with LBBB, paced rhythms, or prosthetic valves or rings; and patients with constrictive pericarditis.
The American Society of Echocardiography/European Association of Echocardiography (ASE/EAE) guidelines use three grades of diastolic dysfunction: grade I (mild), grade II (moderate), and grade III (severe) [1]. The separation into three grades is primarily based on the mitral inflow pattern. Important measurements of mitral inflow Doppler signal include peak early filling velocity (E), late diastolic filling velocity (A), the E/A ratio, the deceleration time (DT) of the early filling velocity, and the time interval between the aortic closure and mitral valve opening called isovolumic relaxation time (IVRT).
In grade I, an abnormal relaxation pattern is seen and defined by an E/A ratio <0.8 and a DT >200 ms. DT reflects the rise in ventricular pressure during diastole and is related to LV ventricular stiffness. Those patients usually have normal diastolic LV filling pressures. However, there is a subset of patients where LV filling pressures are elevated despite an E/A ratio <1. This is more frequently seen in patients with markedly prolonged LV relaxation, often in the setting of hypertrophic cardiomyopathy and advanced hypertensive cardiovascular disease. These patients can be recognized by an elevated E/e′ ratio.
Patients in grade III diastolic dysfunction have abnormal LV relaxation and elevated filling pressures. By Doppler, E/A ratio is ≥2 and DT is <160 ms. Additional Doppler parameters that can be used to support the grading include E/e′ (using average e′) ≥13 cm/s, Ar-A ≥30 ms (difference in duration between atrial reversal signal in pulmonary veins and mitral A duration at the level of mitral annulus), and change in E/A ratio ≥0.5 with Valsalva maneuver. Moderate or grade II diastolic dysfunction is characterized by a pattern similar to a normal pattern defined by an E/A ratio 0.8–1.5. In those patients, the duration of the mitral inflow (duration of the A wave at the level of the mitral annulus) is usually shorter than the duration of atrial reversal velocity (Ar) in the pulmonary veins by at least 30 ms (Ar-A ≥ 30 ms). The average E/e′ often falls in the range of 9–12, and the change in E/A ratio ≥0.5 with Valsalva maneuver.
Estimation of LV Filling Pressures
LV filling pressures as assessed by E/e′ have been shown to predict outcome in patients following myocardial infarction [7]; in patients with hypertension [8], moderately severe to severe secondary MR [9], heart failure due to reduced EF and heart failure with normal EF [10], end-stage renal disease [11], or atrial fibrillation [12]; in patients with cardiomyopathies [13, 14]; and in patients with aortic stenosis after aortic valve replacement [15]. Several studies have confirmed the significant correlation of this ratio with filling pressures in several patient populations [16–23]. However, Mullens et al. reported that E/e′ ratio may not be as reliable in decompensated patients with advanced systolic heart failure and the presence of resynchronization therapy [24]. Of note, the latter study had several limitations related to pressure and Doppler acquisition signals [25]. In a more recent study, Nagueh et al. showed in a similar population of patients with advanced decompensated heart failure that several Doppler indices including E/e′ ratio have good accuracy in identifying patients with elevated mean wedge pressure (>15 mmHg). Further, the latter study showed that the comprehensive approach recommended by the ASE/EAE guidelines had the highest accuracy [25]. Likewise, Ritzema et al., using direct measurements of LA pressure in patients with various grades of diastolic dysfunction and chronic heart failure due to reduced EF, have shown E/e′ ratio to be the most accurate parameter in identifying patients with increased left atrial pressure (LAP) [26]. Changes in LAP in repeat studies were tracked well by E/e′ ratio [26]. Another recent study successfully applied a simplified approach using E/e′ ratio ≥15, LA area ≥20 cm2, and DT <140 ms for evaluation of filling pressures in a population with heart failure and reduced EF and cardiac transplant patients with normal or reduced EF [27].
While the above presentation discussed the role of the more commonly applied Doppler measurements, it is also important to recognize other parameters which can help in patients with equivocal findings. In the presence of LV diastolic dysfunction, a short DT indicates increased LV chamber stiffness and can be of value even in the presence of significant functional mitral regurgitation [1]. The IVRT is another parameter for differentiating normal from elevated LAP as it is often <60 ms with elevated LAP. In addition, the ratio of IVRT to the time interval delay between onset of E velocity and that of e′ (T E-e′, Fig. 1) relates inversely with mean wedge pressure and can be applied in cases of mitral stenosis, mitral regurgitation, and atrial fibrillation which pose challenges to many Doppler signals [28, 29]. For pulmonary vein signals (1), a decrease in LA compliance along with an increase in LA pressure is associated with reduced forward systolic flow (systolic filling fraction <40 %). As noted above, Ar-A duration >30 ms occurs in the setting of elevated LVEDP (>20 mmHg), but its clinical utility is limited in the presence of atrioventricular block or arrhythmias.
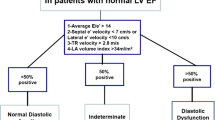
Filling Pressures in Patients with Normal EF
According to the ASE/EAE guidelines [1], LV filling pressures are elevated if septal E/e′ ≥ 15, lateral E/e′ ≥ 12, or average E/e′ ≥ 13. An E/e′ ratio <8 occurs in the presence of a normal LAP, whereas an E/e′ ratio 9–14 requires a more comprehensive approach that includes parameters as LA volume index (increased LAP with ≥34 ml/cm2); pulmonary artery systolic pressure estimate (increased LAP with >35 mmHg); time difference between duration of atrial reversal wave at the pulmonary veins and the duration of the A wave at the level of the mitral annulus, or Ar-A duration; ratio of IVRT to time interval delay between onset of mitral E and annular e′ (increased LAP with IVRT/T E-e′ < 2); and the change in E/A ratio with Valsalva maneuver ≥0.5.
Filling Pressures in Patients with Reduced EF
In patients with depressed EF, the noninvasive LAP estimation algorithm initially uses the mitral inflow pattern E/A ratio to differentiate patients with normal and elevated LAP. Patients with a restrictive filling pattern (E/A ≥ 2, DT < 150 ms) usually have elevated LAP, while those with an impaired relaxation pattern and peak E velocity <50 cm/s have normal LAP [1]. In patients with pseudonormal filling (E/A ratio 1–2 in the setting of impaired LV relaxation) or impaired relaxation pattern with peak E > 50 cm/s, additional parameters are needed. Thus, several other variables are needed to conclude that LAP is elevated and include an average E/e′ ratio ≥15, E/Vp ratio ≥2.5 (Vp stands for flow propagation velocity by color M-mode), pulmonary vein inflow systolic/diastolic ratio <1, Ar-A ≥ 30 ms, IVRT/T E-e′ < 2, change in E/A ratio ≥0.5 with Valsalva maneuver, and noninvasive pulmonary artery systolic pressure estimate >35 mmHg [1]. It should be noted that not all of the above parameters will fall in the range of expected values, and based on technical and pathophysiological factors, one or more of these parameters may have limitations and thus should not be used.
Filling Pressures in Atrial Fibrillation
The accuracy of LV filling pressures in patients with atrial fibrillation has been addressed in several studies. Deceleration time of mitral E velocity (DT) has been shown to correlate well with mean wedge pressure in patients with depressed EF [30]. The authors also demonstrated that peak acceleration rate of mitral diastolic flow velocity (increased LAP with peak acceleration rate ≥1900 cm/s2) correlated well with LV filling pressures. In addition, the ratio of early transmitral flow velocity to flow propagation velocity (increased LAP with E/Vp > 1.4) identified patients with wedge pressure ≥15 mmHg [30]. Furthermore, Sohn et al. have shown that a septal E/e′ ratio ≥11 predicts LV filling pressure ≥15 mmHg with reasonable accuracy [17]. Pulmonary venous diastolic velocity deceleration time ≤220 ms appears to identify patients in atrial fibrillation with mean wedge pressure ≥18 mmHg [31]. T E-e′ >34 ms using dual Doppler assessment has improved the accuracy of evaluation of filling pressures when added to E/e′ ratio in patients with atrial fibrillation [32]. Likewise, single-beat acquisition of mitral E and annular e′ velocities resulted in a better correlation with plasma BNP and mean wedge pressure in patients with atrial fibrillation and normal EF than an E/e′ ratio obtained by velocity averaging from nonsimultaneous beats [33] .
Diastolic Stress Test
Diastolic stress testing has become a valuable method to unveil elevated LV filling pressures in patients with exertional dyspnea of a cardiac etiology. A diastolic stress test is performed using supine bicycle exercise allowing a detailed stage-by-stage analysis of diastolic properties during which mitral inflow velocities, mitral annular velocities by tissue Doppler, and tricuspid valve peak regurgitation velocity are recorded at the different stages and the early recovery period. In patients with diastolic dysfunction, peak E velocity increases more profoundly than the change in annular e′ velocity [34], and thus, E/e′ ratio increases with exercise reflecting the elevated LV filling pressures [35, 36]. Importantly, the elevated E/e′ ratio with exercise has been associated with adverse cardiovascular outcomes even when detectable ischemia is considered [37]. Diastolic stress testing with intravenous dobutamine infusion, although infrequent in current clinical practice, has also been shown to predict clinical events in cardiac patients [38].
Deformation Measurements (Strain and Strain Rate)
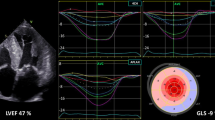
Myocardial deformation and the rate of deformation during the different phases of the cardiac cycle can be currently measured by tissue Doppler imaging and speckle tracking imaging techniques. Strain represents the fractional length change of a myocardial segment with respect to an original length (could be the length in the absence of any stress or at end diastole), and strain rate is the speed that deformation occurs (unit: s−1). Many patients with diastolic dysfunction, including those with heart failure and normal EF, have impaired longitudinal strain (Fig. 2). With respect to relation of diastolic strain and strain rate (Fig. 3), both regional and global measurements have been correlated with the time constant of LV relaxation; this applies to signals during the isovolumic relaxation period as well as during early diastole [39–41]. Regarding segmental function, time to onset of regional relaxation was used to identify ischemic segments during dobutamine stress echocardiography [42]. In another report, early diastolic strain rate predicted segmental viability in STEMI patients with an akinetic apex following reperfusion therapy [43]. Similarly, diastolic strain rates have been used for differentiating segments with myocardial viability in patients with prior myocardial infarction [44]. More recently, regional increased strain in early diastole (described by strain imaging diastolic index and using end systolic strain at aortic valve closure and strain at one-third diastole) was noted in patients undergoing percutaneous coronary revascularization not only during balloon inflation but also after the ischemia has resolved [45]. With respect to outcome studies on diastolic strain rate measurements, strain rate during the isovolumic relaxation period (SRIVR) was an independent predictor of outcome in patients with ST-elevation myocardial infarction in one study [46].
Strain imaging and calculation of global longitudinal strain in a heart failure patient with preserved EF using measurements from apical three-chamber view (a), apical four-chamber view (b), and apical two-chamber view (c). Segmental strain results are displayed in a polar map (d). Global LV longitudinal strain is impaired at −15.7 %.
There are growing data supporting the notion that diastolic strain rate in conjunction with mitral E velocity can provide reasonable estimation of LV filling pressures. In one study, Kimura et al. [47] used the ratio of mitral E velocity to early diastolic global longitudinal strain rate (E/SR E ) as surrogate of LV filling pressures. In another study that included animal experiments and patients’ data [40], the ratio of E velocity to diastolic strain rate during isovolumic relaxation (E/SR IVR ) was a good measure of mean wedge pressure. In addition, there are data [48] that E/SR E and E/SR IVR are significantly related to LV stiffness constant. With respect to diastolic strain, one study used the ratio of mitral E to global longitudinal diastolic strain (and strain rate) during peak mitral filling (E/D S ) to successfully predict LV filling pressures in patients with preserved EF [49]. While most studies used longitudinal deformation, one report utilized the ratio of mitral E velocity to circumferential strain at the time of peak E velocity to identify dilated cardiomyopathy patients with mean wedge pressure >12 mmHg [50]. These ratios provided incremental prognostic information in several patient populations including myocardial infarction and atrial fibrillation [46, 51•, 52•].
Twist and Untwisting
Twisting of the LV results in storage of potential energy during systole, and untwisting is in part related to the recoil forces and favorably influences early diastolic filling. LV twist is obtained as the net difference between anticlockwise rotation of the apex and clockwise rotation of the base [53]. CMR, tissue Doppler imaging, and speckle tracking can be used to study torsional mechanics [54, 55].
Several studies have shown that patients with diastolic dysfunction can have delayed untwisting and a reduced peak untwisting rate with exercise [56–58]. Several factors affect peak untwisting rate and include the diastolic time constant of LV [59], LV early diastolic load which increases it [60••], as well as LV elastic recoil which can result in normal peak untwisting rate in patients with diastolic dysfunction [61]. Recently, 3D speckle tracking has been evaluated to study twist mechanics. However, the low frame rate could lead to underestimation of LV peak twisting and untwisting velocities. At the present time, these measurements are useful in evaluating myocardial function in several disease states but are not essential for day-to-day clinical studies.
2D and 3D LA Volumes
An LA volume index ≥34 ml/cm2 is associated with adverse cardiovascular outcomes [62]. In one study, LA volume index was highly sensitive and specific for detecting diastolic dysfunction in a patient population older than 45 years [63]. Although LA size is usually increased in patients with elevated filling pressures, LA volume may not be increased in early stages of diastolic dysfunction [64]. Notwithstanding the findings in early and mild disease, LA maximal volume can be taken as an index of diastolic dysfunction in the absence of significant mitral regurgitation and atrial arrhythmias. LA enlargement is not specific for diastolic dysfunction as the LA can be enlarged in patients with anemia, high-output states, bradycardia, athletes, mitral valve disease, and atrial arrhythmias [1].
Most labs obtain biplane measurements of LA volumes. However, 3D echocardiography appears to be more accurate in determining true LA size. This is in part due to the asymmetrical enlargement of the left atrium [65••]. Additional studies are needed to examine the advantage of using 3D LA volume measurements in the clinical setting.
LA Strain
LA strain (Fig. 4) provides insights into LA reservoir (LA S ), conduit, and pump (LA A ) functions. Additionally, a surrogate of LA stiffness may be calculated using the ratio of E/e′ to LA S . This index is abnormally elevated in patients with diastolic heart failure when compared to healthy subjects and patients with diastolic dysfunction who are not in heart failure [66]. Importantly, LA S relates inversely to LV filling pressures, especially in patients with depressed EF [67]. Studies have also shown that LA S is a significant predictor of maintenance of sinus rhythm after cardioversion in patients with atrial fibrillation [68]. While promising, measurement of LA strain is not standardized. Similar to LV strain measurements, the results depend on the ultrasound system used for acquisition and the software used for analysis. In addition, the number of sites and the location of sampling in the LA have varied between the studies.
Left atrium (LA) strain curves in a patient with heart failure and preserved EF. Signals are obtained from LA mid-lateral and mid-septal regions in the apical four-chamber view. LA S refers to LA strain during left ventricular systole, and LA A refers to LA strain during atrial systole before the QRS complex
Conclusion
Doppler echocardiography can be used to obtain reliable noninvasive assessment of LV diastolic function. There are several promising new indices, but their ultimate adoption in clinical practice awaits standardization of measurements and additional clinical studies.
References
Papers of particular interest, published recently, have been highlighted as • Of importance •• Of major importance
Nagueh SF, Appleton CP, Gillebert TC, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography. J Am Soc Echocardiogr. 2009;22:107–33.
Redfield MM, Jacobsen SJ, Burnett JC, et al. Burden of systolic and diastolic ventricular dysfunction in the community: appreciating the scope of the heart failure epidemic. JAMA. 2003;289:194–202.
Kane GC, Karon BL, Mahoney DW, et al. Progression of left ventricular diastolic dysfunction and risk of heart failure. JAMA. 2011;306:856–63.
Aljaroudi W, Alraies MC, Halley C, et al. Impact of progression of diastolic dysfunction on mortality in patients with normal ejection fraction. Circulation. 2012;125:782–8. Important study showing the adverse effects of progression of diastolic dysfunction with an increase in total mortality.
Kuznetsova T, Thijs L, Knez J, et al. Prognostic value of left ventricular diastolic dysfunction in a general population. J Am Heart Assoc. 2014;3:e000789. An interesting study showing the prognostic power of a simple diastolic measurement in a general population.
Oki T, Tabata T, Yamada H, et al. Clinical application of pulsed Doppler tissue imaging for assessing abnormal left ventricular relaxation. Am J Cardiol. 1997;79:921–8.
Hillis GS, Moller JE, Pellikka PA, et al. Noninvasive estimation of left ventricular filling pressure by E/e’ is a powerful predictor of survival after acute myocardial infarction. J Am Coll Cardiol. 2004;43:360–7.
Wang M, Yip GW, Wang AY, et al. Tissue Doppler imaging provides incremental prognostic value in patients with systemic hypertension and left ventricular hypertrophy. J Hypertens. 2005;23:183–91.
Bruch C, Klem I, Breithardt G, et al. Diagnostic usefulness and prognostic implications of the mitral E/E’ ratio in patients with heart failure and severe secondary mitral regurgitation. Am J Cardiol. 2007;100:860–5.
Wang M, Yip G, Yu CM, et al. Independent and incremental prognostic value of early mitral annulus velocity in patients with impaired left ventricular systolic function. J Am Coll Cardiol. 2005;45:272–7.
Sharma R, Pellerin D, Gaze DC, et al. Mitral peak Doppler E-wave to peak mitral annulus velocity ratio is an accurate estimate of left ventricular filling pressure and predicts mortality in end-stage renal disease. J Am Soc Echocardiogr. 2006;19:266–73.
Okura H, Takada Y, Kubo T, et al. Tissue Doppler-derived index of left ventricular filling pressure, E/E’, predicts survival of patients with non-valvular atrial fibrillation. Heart. 2006;92:1248–52.
McMahon CJ, Nagueh SF, Eapen RS, et al. Echocardiographic predictors of adverse clinical events in children with dilated cardiomyopathy: a prospective clinical study. Heart. 2004;90:908–15.
McMahon CJ, Nagueh SF, Pignatelli RH, et al. Characterization of left ventricular diastolic function by tissue Doppler imaging and clinical status in children with hypertrophic cardiomyopathy. Circulation. 2004;109:1756–62.
Chang SA, Park PW, Sung K, et al. Noninvasive estimate of left ventricular filling pressure correlated with early and midterm postoperative cardiovascular events after isolated aortic valve replacement in patients with severe aortic stenosis. J Thorac Cardiovasc Surg. 2010;140:1361–6.
Nagueh SF, Middleton KJ, Kopelen HA, et al. Doppler tissue imaging: a noninvasive technique for evaluation of left ventricular relaxation and estimation of filling pressures. J Am Coll Cardiol. 1997;30:1527–33.
Sohn DW, Song JM, Zo JH, et al. Mitral annulus velocity in the evaluation of left ventricular diastolic function in atrial fibrillation. J Am Soc Echocardiogr. 1999;12:927–31.
Nagueh SF, Mikati I, Kopelen HA, et al. Doppler estimation of left ventricular filling pressure in sinus tachycardia. A new application of tissue Doppler imaging. Circulation. 1998;98:1644–50.
Sohn DW, Kim YJ, Kim HC, et al. Evaluation of left ventricular diastolic function when mitral E and A waves are completely fused: role of assessing mitral annulus velocity. J Am Soc Echocardiogr. 1999;12:203–8.
Ommen SR, Nishimura RA, Appleton CP, et al. Clinical utility of Doppler echocardiography and tissue Doppler imaging in the estimation of left ventricular filling pressures: a comparative simultaneous Doppler-catheterization study. Circulation. 2000;102:1788–94.
Kim YJ, Sohn DW. Mitral annulus velocity in the estimation of left ventricular filling pressure: prospective study in 200 patients. J Am Soc Echocardiogr. 2000;13:980–5.
Kasner M, Westermann D, Steendijk P, et al. Utility of Doppler echocardiography and tissue Doppler imaging in the estimation of diastolic function in heart failure with normal ejection fraction: a comparative Doppler-conductance catheterization study. Circulation. 2007;116:637–47.
Rivas-Gotz C, Manolios M, Thohan V, et al. Impact of left ventricular ejection fraction on estimation of left ventricular filling pressures using tissue Doppler and flow propagation velocity. Am J Cardiol. 2003;91:780–4.
Mullens W, Borowski AG, Curtin RJ, et al. Tissue Doppler imaging in the estimation of intracardiac filling pressure in decompensated patients with advanced systolic heart failure. Circulation. 2009;119:62–70.
Nagueh SF, Bhatt R, Vivo RP, et al. Echocardiographic evaluation of hemodynamics in patients with decompensated systolic heart failure. Circ Cardiovasc Imaging. 2011;4:220–7.
Ritzema JL, Richards AM, Crozier IJ, et al. Serial Doppler echocardiography and tissue Doppler imaging in the detection of elevated directly measured left atrial pressure in ambulant subjects with chronic heart failure. JACC Cardiovasc Imaging. 2011;4:927–34.
Rafique AM, Phan A, Tehrani F, et al. Transthoracic echocardiographic parameters in the estimation of pulmonary capillary wedge pressure in patients with present or previous heart failure. Am J Cardiol. 2012;110:689–94.
Rivas-Gotz C, Khoury DS, Manolios M, et al. Time interval between onset of mitral inflow and onset of early diastolic velocity by tissue Doppler: a novel index of left ventricular relaxation: experimental studies and clinical application. J Am Coll Cardiol. 2003;42:1463–70.
Diwan A, McCulloch M, Lawrie GM, et al. Doppler estimation of left ventricular filling pressures in patients with mitral valve disease. Circulation. 2005;111:3281–9.
Nagueh SF, Kopelen HA, Quinones MA. Assessment of left ventricular filling pressures by Doppler in the presence of atrial fibrillation. Circulation. 1996;94:2138–45.
Chirillo F, Brunazzi MC, Barbiero M, et al. Estimating mean pulmonary wedge pressure in patients with chronic atrial fibrillation from transthoracic Doppler indexes of mitral and pulmonary venous flow velocity. J Am Coll Cardiol. 1997;30:19–26.
Wada Y, Murata K, Tanaka T, et al. Simultaneous Doppler tracing of transmitral inflow and mitral annular velocity as an estimate of elevated left ventricular filling pressure in patients with atrial fibrillation. Circ J. 2012;76:675–81.
Kusunose K, Yamada H, Nishio S, et al. Clinical utility of single-beat E/e’ obtained by simultaneous recording of flow and tissue Doppler velocities in atrial fibrillation with preserved systolic function. JACC Cardiovasc Imaging. 2009;2:1147–56.
Ha JW, Choi D, Park S, et al. Left ventricular diastolic functional reserve during exercise in patients with impaired myocardial relaxation at rest. Heart. 2009;95:399–404.
Talreja DR, Nishimura RA, Oh JK. Estimation of left ventricular filling pressure with exercise by Doppler echocardiography in patients with normal systolic function: a simultaneous echocardiographic-cardiac catheterization study. J Am Soc Echocardiogr. 2007;20:477–9.
Burgess MI, Jenkins C, Sharman JE, et al. Diastolic stress echocardiography: hemodynamic validation and clinical significance of estimation of ventricular filling pressure with exercise. J Am Coll Cardiol. 2006;47:1891–900.
Holland DJ, Prasad SB, Marwick TH. Prognostic implications of left ventricular filling pressure with exercise. Circ Cardiovasc Imaging. 2010;3:149–56.
Duncan AM, Lim E, Gibson DC, et al. Effect of dobutamine stress on left ventricular filling in ischemic dilated cardiomyopathy: pathophysiology and prognostic implications. J Am Coll Cardiol. 2005;46:488–96.
Kato T, Noda A, Izawa H, et al. Myocardial velocity gradient as a noninvasively determined index of left ventricular diastolic dysfunction in patients with hypertrophic cardiomyopathy. J Am Coll Cardiol. 2003;42:278–85.
Wang J, Khoury DS, Thohan V, et al. Global diastolic strain rate for the assessment of left ventricular relaxation and filling pressures. Circulation. 2007;115:1376–83.
Wakami K, Ohte N, Sakata S, et al. Myocardial radial strain in early diastole is useful for assessing left ventricular early diastolic function: comparison with invasive parameters. J Am Soc Echocardiogr. 2008;21:446–51.
Abraham TP, Belohlavek M, Thomson HL, et al. Time to onset of regional relaxation: feasibility, variability and utility of a novel index of regional myocardial function by strain rate imaging. J Am Coll Cardiol. 2002;39:1531–7.
Park SM, Miyazaki C, Prasad A, et al. Feasibility of prediction of myocardial viability with Doppler tissue imaging following percutaneous coronary intervention for ST elevation anterior myocardial infarction. J Am Soc Echocardiogr. 2009;22:183–9.
Hoffmann R, Altiok E, Nowak B, et al. Strain rate analysis allows detection of differences in diastolic function between viable and nonviable myocardial segments. J Am Soc Echocardiogr. 2005;18:330–5.
Ishii K, Suyama T, Imai M, et al. Abnormal regional left ventricular systolic and diastolic function in patients with coronary artery disease undergoing percutaneous coronary intervention: clinical significance of post-ischemic diastolic stunning. J Am Coll Cardiol. 2009;54:1589–97.
Shanks M, Ng AC, van de Veire NR, et al. Incremental prognostic value of novel left ventricular diastolic indexes for prediction of clinical outcome in patients with ST-elevation myocardial infarction. Am J Cardiol. 2010;105:592–7.
Kimura K, Takenaka K, Ebihara A, et al. Speckle tracking global strain rate E/E’ predicts LV filling pressure more accurately than traditional tissue Doppler E/E’. Echocardiography. 2012;29:404–10.
Kasner M, Gaub R, Sinning D, et al. Global strain rate imaging for the estimation of diastolic function in HFNEF compared with pressure-volume loop analysis. Eur J Echocardiogr. 2010;11:743–51.
Dokainish H, Sengupta R, Pillai M, et al. Usefulness of new diastolic strain and strain rate indexes for the estimation of left ventricular filling pressure. Am J Cardiol. 2008;101:1504–9.
Meluzin J, Spinarova L, Hude P, et al. Estimation of left ventricular filling pressures by speckle tracking echocardiography in patients with idiopathic dilated cardiomyopathy. Eur J Echocardiogr. 2011;12:11–8.
Hsu PC, Lee WH, Chu CY, et al. The ratio of early mitral inflow velocity to global diastolic strain rate as a useful predictor of cardiac outcomes in patients with atrial fibrillation. J Am Soc Echocardiogr. 2014;27:717–25. Interesting study showing the prognostic power of diastolic strain rate measurements in patients with atrial fibrillation.
Ersboll M, Andersen MJ, Valeur N, et al. Early diastolic strain rate in relation to systolic and diastolic function and prognosis in acute myocardial infarction: a two-dimensional speckle-tracking study. Eur Heart J. 2014;35:648–56. Interesting study showing the prognostic power of diastolic strain rate measurements in patients with acute myocardial infarction.
Sengupta PP, Tajik AJ, Chandrasekaran K, et al. Twist mechanics of the left ventricle: principles and application. JACC Cardiovasc Imaging. 2008;1:366–76.
Notomi Y, Setser RM, Shiota T, et al. Assessment of left ventricular torsional deformation by Doppler tissue imaging: validation study with tagged magnetic resonance imaging. Circulation. 2005;111:1141–7.
Notomi Y, Lysyansky P, Setser RM, et al. Measurement of ventricular torsion by two-dimensional ultrasound speckle tracking imaging. J Am Coll Cardiol. 2005;45:2034–41.
Pacileo G, Baldini L, Limongelli G, et al. Prolonged left ventricular twist in cardiomyopathies: a potential link between systolic and diastolic dysfunction. Eur J Echocardiogr. 2011;12:841–9.
Takeuchi M, Borden WB, Nakai H, et al. Reduced and delayed untwisting of the left ventricle in patients with hypertension and left ventricular hypertrophy: a study using two-dimensional speckle tracking imaging. Eur Heart J. 2007;28:2756–62.
Notomi Y, Martin-Miklovic MG, Oryszak SJ, et al. Enhanced ventricular untwisting during exercise: a mechanistic manifestation of elastic recoil described by Doppler tissue imaging. Circulation. 2006;113:2524–33.
Dong SJ, Hees PS, Siu CO, et al. MRI assessment of LV relaxation by untwisting rate: a new isovolumic phase measure of tau. Am J Physiol Heart Circ Physiol. 2001;281:H2002–9.
Opdahl A, Remme EW, Helle-Valle T, et al. Myocardial relaxation, restoring forces, and early-diastolic load are independent determinants of left ventricular untwisting rate. Circulation. 2012;126:1441–51. An important study looking at the hemodynamic determinants of LV untwisting rate.
Wang J, Khoury DS, Yue Y, et al. Left ventricular untwisting rate by speckle tracking echocardiography. Circulation. 2007;116:2580–6.
Abhayaratna WP, Seward JB, Appleton CP, et al. Left atrial size: physiologic determinants and clinical applications. J Am Coll Cardiol. 2006;47:2357–63.
Pritchett AM, Mahoney DW, Jacobsen SJ, et al. Diastolic dysfunction and left atrial volume: a population-based study. J Am Coll Cardiol. 2005;45:87–92.
Tsang TS, Abhayaratna WP, Barnes ME, et al. Prediction of cardiovascular outcomes with left atrial size: is volume superior to area or diameter? J Am Coll Cardiol. 2006;47:1018–23.
Mor-Avi V, Yodwut C, Jenkins C, et al. Real-time 3D echocardiographic quantification of left atrial volume: multicenter study for validation with CMR. JACC Cardiovasc Imaging. 2012;5:769–77. Important study looking at multicenter LA volume measurements with validation against CMR.
Kurt M, Wang J, Torre-Amione G, et al. Left atrial function in diastolic heart failure. Circ Cardiovasc Imaging. 2009;2:10–5.
Wakami K, Ohte N, Asada K, et al. Correlation between left ventricular end-diastolic pressure and peak left atrial wall strain during left ventricular systole. J Am Soc Echocardiogr. 2009;22:847–51.
Di Salvo G, Caso P, Lo Piccolo R, et al. Atrial myocardial deformation properties predict maintenance of sinus rhythm after external cardioversion of recent-onset lone atrial fibrillation: a color Doppler myocardial imaging and transthoracic and transesophageal echocardiographic study. Circulation. 2005;112:387–95.
Compliance with Ethics Guidelines
Conflict of Interest
Dimitrios Maragiannis and Sherif F. Nagueh declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Echocardiography
Rights and permissions
About this article
Cite this article
Maragiannis, D., Nagueh, S.F. Echocardiographic Evaluation of Left Ventricular Diastolic Function: an Update. Curr Cardiol Rep 17, 3 (2015). https://doi.org/10.1007/s11886-014-0561-9
Published:
DOI: https://doi.org/10.1007/s11886-014-0561-9