Abstract
Purpose of Review
Wall shear stress describes the mechanical influence of blood flow on the arterial wall. In this review, we discuss the role of the wall shear stress in the development of atherosclerosis and its complications.
Recent Findings
Areas with chronically low, oscillating wall shear stress are most prone to plaque development and include outer bifurcation walls and inner walls of arches. In some diseases, patients have lower wall shear stress even in straight arterial segments; also, these findings were associated with atherosclerosis. High wall shear stress develops in the distal part (shoulder) of a stenosis and contributes to plaque destabilization.
Summary
Wall shear stress changes are involved in the development of atherosclerosis. They are not fully understood yet and act in concert with tangential wall stress.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Atherosclerosis and its complications, such as coronary artery disease, cerebrovascular disease, and peripheral artery disease and their acute complications (acute myocardial infarction, ischemic stroke, and acute limb ischemia), are the leading cause of morbidity and mortality in developed countries. Many risk factors have been described, and the traditional ones include smoking, diabetes mellitus, hypercholesterolemia, arterial hypertension, obesity, and others [1]. However, also hemodynamic factors play a role in plaque development or rupture. These include tangential stress (arterial blood pressure), the cyclic stretch by pulsatile cardiac output, and wall shear stress (WSS). WSS alterations are also involved in atherogenesis and the development of complications: chronically low oscillating WSS is the strongest local risk factor of atherosclerosis, while high WSS (due to stenosis) contributes to plaque rupture [2••]. WSS quantifies the parallel frictional drag force that depends on blood flow, arterial diameter, and blood viscosity [3]. WSS is imposed directly on endothelium and modulates both its structure, permeability, and function. Areas with constantly low WSS with changing vector (outer walls of bifurcations, inner wall of the aortic arch) are most frequently affected by atherosclerotic plaques. Briefly, chronically low WSS may induce plaque formation, while high WSS may destabilize plaques and thus induce an acute vascular catastrophe, such as myocardial infarction and stroke [4]. We discuss the role of WSS in atherogenesis current possibilities of WSS evaluation.
Wall Shear Stress Explanation
As fluid flows and two adjacent layers have different velocities, shear stresses are generated at this interface. The shear stress depends on the velocity gradient between these layers and on the viscosity of the liquid. Similarly, there is a velocity gradient between the flowing fluid (blood) and the vascular wall. To describe this interaction, the concept of wall shear stress (WSS) is used. WSS is defined as the shear stress exerted by the fluid tangentially on the wall. The WSS determines the degree of interaction between the fluid and the wall. For simplified WSS assessment, the following equation is used: WSS = (4 μ.v)/r where μ is viscosity, v = velocity, and r = arterial radius. Part of the equation (v/r) represents wall shear rate and is sometimes used as a surrogate of WSS as it bypasses the problems of dynamic viscosity [5, 6].
For a laminar (parabolic) velocity profile in a circular tube with a rigid wall, the shear stress distribution is linear with a maximum along the walls and zero in the tube axis. In the case of a turbulent velocity profile, the shear stress distribution inside the cross section is nonlinear. In the central region of the tube, the values of the shear stress are relatively small because the velocity profile is flat, and therefore the velocity gradients are small. In turbulent flow, WSS increases rapidly toward the wall, and the maximum value of the shear stress is generated at the wall surface. In the case of flow disturbed by stenosis or arterial curvature, the situation is more complex. The shear stress values in these regions are affected by the presence of a secondary flow or a flow separation. We can detect regions with local wall shear stress peaks (e.g., constrictions/stenosis [7]) or sections with low variable shear stress on the wall (e.g., separation areas [8]).
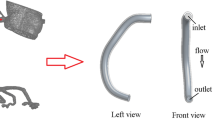
If we focus on the WSS in a symmetric stenosis (such as due to a symmetric atherosclerosis plaque), the flow can be divided into 4 sections: (1) inlet, (2) throttle or stenosis, (3) flow separation (distal plaque shoulder), and (4) outlet—see Fig. 1 for an explanation.
Schematic illustration of the wall shear stress along the idealized symmetrical stenosis. (1) The inlet section, where the nature of the flow and thus the shear stress are determined by the boundary conditions. (2) This is followed by the constriction section, in which the velocity profile is flattened due to acceleration. In this region, the velocity profile can be almost piston-like, and due to the large velocity gradient near the wall, the shear stress values on the wall are relatively high. (3) Beyond the constriction, an accelerated mean flow can be seen, which gradually slows down due to the viscous forces, and its area expands to the point of reattachment to the wall. The wall shear stress in this section is determined by the size of the separation regions and the nature of the flow in them. In general, however, there is a significant reduction in shear stresses compared to the flow in the throat, and the reverse and unstable flow causes the stresses to become negative and oscillating. (4) In the outlet section, the shear stresses return to positive values after the reattachment of the flow [58, 59]. In the case of physiological pulsating flow in the physiological range, the shear stress distribution remains similar [60, 61]. Compared to the stationary flow, the absolute values change, and the positions of the local minima and maxima of the shear stress shift due to pulsations. The eccentricity of the stenosis influences both the shear stress distribution and its maxima [62]. While the shear stress in the neck of symmetric stenosis is constant around the circumference, in the case of an asymmetric stenosis, a maximum is localized at the opposite side of the constriction. This maximum is significantly higher than for the symmetrical stenosis. As in the case of symmetrical stenosis, there is a region just distal to the stenosis where shear stress becomes negative
The Physiological Importance of Wall Shear Stress
In the circulation, endothelial cells (ECs) are permanently exposed to WSS caused by blood flow. The local pattern of blood flow determines the characteristics of WSS, i.e., its intensity, direction, pulsatility, and regularity, which are sensed by endothelium [9]. EC sensitivity to WSS is involved in developmental and physiological vascular processes such as the pattern of vascular architecture during developmental vessel formation, vasoreactivity, and vascular remodeling [10,11,12]. WSS affects the morphology, intimal proliferation, differentiation, metabolism, and communication of endothelial cells (ECs). The endothelium is highly metabolically active and mitotically stable under healthy conditions [13].
In straight arterial segments, the blood flow is laminar, and WSS values are kept within narrow limits of 10–50 dyn/cm2 [14] by the tone of the vascular smooth muscle cells of lamina media. WSS controls the appropriate arterial diameter: in case of sudden need of increased blood flow (e.g., working muscles of lower extremities during jogging), the peripheral vascular resistance decreases. This is followed by higher blood flow velocity, which is a trigger for the release of nitric oxide, strong vasodilating molecule, and the feeding artery dilates, which, in turn, restores the normal WSS value. In vivo experiments have shown that an increase in WSS triggered arterial vasodilation within a few seconds [11]. Short-term effect of WSS (seconds to minutes) on endothelial function includes increase in secretion of prostacyclin and mainly of nitric oxide [15, 16].
The effects of acute WSS increase (and of endothelial response) can be studied by the method of flow-mediated dilatation [17]: the sphygmomanometer cuff is inflated to a suprasystolic pressure over proximal forearm for several minutes. Temporary ischemia due to cuff inflation triggers dilatation of the resistant arterioles distal to the cuff, and after cuff release, the flow increases by several times, which leads to arterial dilatation by nitric oxide. Long-term WSS increase in straight arterial segments is typical for arteriovenous fistulas, including those used as a hemodialysis vascular access. Interestingly, WSS in the feeding artery normalizes years after fistula creation [18] because the arterial wall stroma has to be dissolved by matrix metalloproteinases.
Several studies during the past decades concluded that shear stress induces long-term remodeling of vessel walls and subsequent growth in vessel caliber [19, 20]. As a mechanical consequence of the remodeling and increase in vessel diameter, the WSS that had initially increased returned to its initial value. Inversely, a decrease in blood flow due to a proximal significant stenosis has been shown to induce endothelium-dependent inward remodeling and constitutive reduction in artery diameter [21, 22]. WSS-induced remodeling involves several processes affecting vessel homeostasis such as EC morphology, endothelial permeability, and inflammation.
As far as the flow is pulsatile in arteries, studies are distinguished between peak, mean, and end-diastolic WSS similar to blood flow velocity (Fig. 2).
Common carotid artery wall shear rate. Longitudinal section of the common carotid artery in B-mode (above) and spectral Doppler curve (below). Peak systolic (PS), end-diastolic (ED), and mean (TAMAX) velocities are measured and together with the arterial diameter give peak systolic, end-diastolic and mean wall shear rate values
Age-Related Changes of Arteries
In healthy children free of severe atherosclerosis risk factors, the arteries are relatively straight and have a thin arterial wall with intact, functioning endothelial layer, and bifurcations tend to have sharp angles that are associated with the least energy loss. Aging-related arterial wall structure changes include histological changes (less elastin, more collagen, atherosclerosis, and medial calcinosis) and also arterial dilatation and prolongation. Both central and peripheral conduit artery diameters demonstrate a gradual increase with age. For example, the cross-sectional interadventitial diameter of the common carotid artery increases by 0.017 mm per year in healthy subjects while dilates by 0.03 mm per year in patients with known atherosclerosis [23]. Arterial dilatation is also called redundancy [24•]. Arteries do not only dilate but also elongate with age and with various diseases. As far as the adult human body does not grow, arteries have no space for elongation. Therefore, various arches, tortuosities, kinking, or coiling develop, and the bifurcation angles become more obtuse [24•]. These changes lead to considerable flow alteration (turbulence instead of laminar flow) and thus also to disturbed atherosclerosis-prone WSS [25]. The aforementioned vascular changes are responsible for a higher atherosclerotic burden with age. This is due to longer exposure to the traditional risk factors, such as serum LDL cholesterol, due to age-related processes, such as apoptosis, but, again, due to hemodynamic changes, where wall shear stress plays an important role.
Wall Shear Stress Receptors
As far as the WSS vector is tangential to the arterial wall, it is sensed by its inner layer—endothelium. Therefore, there should be mechanisms that react (chemically) to this physical force. Rather than one specific molecule serving as a WSS receptor, so far the research suggests there might be dozens of proteins and cellular structures participating in sensing and mechanotransmission—a process of transducing a mechanical force into biochemical processes and signaling pathways [26, 27].
Several molecules were proposed to the function of mechanotransduction—primary cilia, integrins, glycocalyx, and ion channels [27, 28]. For example, a protein in the junctional complex in endothelial cells called PECAM-1 (platelet endothelial cell adhesion molecule-1) has been a subject of several studies. Increased tension in PECAM-1 triggers kinases and through phosphorylation activates VEGF (vascular endothelial growth factor) receptors, which have an important role in vasculogenesis [29]. Caveolae, a part of the apical membrane containing nitric oxide, also participate, and their way of sensing is quite unique. The shape of caveolae is similar to a hot-air balloon with the neck being toward the vascular lumen. Due to this neck-like structure, the velocity of blood flow in the internal parts of the caveolae is normally lower compared to the lumen. However, if the velocity in the vessel raises, the neck of the caveola is due to the friction and membrane tension open apart, and the internal part of the caveola is exposed to a higher velocity, which may in turn result, e.g., in a nitric oxide release [30].
Another possible way of sensing WSS has been promoted and is based on a principle of tensegrity [27]. This view focuses the cellular micro-architecture, mainly the cytoskeleton, to be the primary sensor. With tensegrity, one may see the shape of a cell to be a net result of a fine equilibrium between tensile forces acting in various directions. When there is a force pushing from the outside, the deformation is sensed by the cytoskeleton, and biochemical processes are triggered [27, 31].
Low Wall Shear Stress and Atherosclerosis
In straight arterial segments, the flow is laminar, and WSS values remain physiologically within narrow limits of 10–50 dyne/cm2 [14]. When there are increased flow demands, such as in lower extremities during walking, jogging, etc., the peripheral arterial resistance (tones of the arterioles) decreases, while flow velocity and volume increase. Increased velocity means higher WSS. This is sensed by the ECs, and more nitric oxide is released in order to dilate the diameter of the feeding artery—and to normalize WSS. However, this ideal mechanism is not achievable in arterial segments that have relatively fixed geometry, such as in the outer walls of bifurcations (Fig. 3) or along the inner wall of the aortic arch, where the blood flow is slower than along the outer wall. Indeed, these sites are the most frequently affected by atherosclerosis. The explanation is in the changed shape of the endothelial cells leading to the increased permeability of Evans blue (an azo dye) [32] but also of LDL particles and monocytes [33] that constitute the atherosclerotic plaque. Endothelial cells tend to align with wall shear stress: the higher the wall shear stress, the more elongated the cells are and vice versa. In stenosed arterial segments, endothelial cells lose their elongated shape in the separation zones (see Fig. 1 for explanation). An interesting and easily accessible example of low WSS with oscillating vector is the common carotid artery bifurcation. The most proximal part of the internal carotid artery has a highly innervated dilatation, called carotid sinus, and is also a frequent site of plaques. In the carotid sinus, the arterial diameter suddenly becomes too wide for the flow, and the so-called flow-reversal pattern develops and can be visualized (see Fig. 3) or even quantified by duplex Doppler ultrasonography [34].
Flow reversal in carotid bifurcation. Longitudinal section of carotid bifurcation in B-mode (left) and with color Doppler mapping (right). The internal carotid artery begins with a dilated section (bulb or sinus). During the systolic phase, blood flow is accelerated in the straight segments, but flow reversal develops in the carotid sinus (blue area). Low oscillating WSS in the carotid sinus is responsible for earlier atherosclerotic plaque development
The WSS in straight arterial segments is, however, also chronically lower in various disease states such as type 2 diabetes mellitus [6] and end-stage kidney disease [35] in comparison with healthy controls. More importantly, lower common carotid WSS was associated with higher intima-media thickness [36], more carotid bifurcation plaques [35], but also with brain white-matter hyperintensities and cognitive impairment in older subjects [37]. Similarly, areas of low oscillating WSS are first affected by wall thickening in coronary arteries [38].
Why is the WSS lower in the aforementioned patients? In both studies, performed in our institution, diabetic and end-stage kidney disease patients had wider arteries and slower velocity than controls—both determinants of wall shear rate [6, 35]. Age- and disease-related arterial widening is probably related both to the blood pressure and abnormalities of the arterial wall [39]. Internal carotid flow volume is maintained by the brain autoregulation supposing that there is adequate cardiac output, although this old concept is currently debated [40]. The flow volume is directly proportional to the mean velocity and indirectly to the arterial cross-sectional area. Therefore, it seems that the arteries are too wide for the given flow volume, and, secondarily, the flow velocity and WSS decrease, which, in turn, promotes atherosclerosis. Nevertheless, carotid arteries differ from the peripheral ones especially by stable flow volume. Indeed, this speculative mechanism can not explain all findings, nor all types of arteries. One such example is the diffuse narrowing of peripheral arteries in diabetic patients, where the medial arterial layer is significantly affected. Moreover, only very few authors investigated WSS in femoral arteries, but in one study, femoral WSS at rest was significantly lower than carotid WSS in young healthy subjects [41].
High Wall Shear Stress and Atherosclerosis
Areas with high WSS include arterial stenoses (see Fig. 1), ascending aorta in case of severe aortic valve stenosis, and feeding arteries of arteriovenous fistulas or malformations. In segments with stenosis, the stenotic part is followed by the flow separation region, where the WSS becomes low and has an oscillating vector.
The effects of chronically high WSS are both mechanical and biochemical. Mechanical effects include denudation of ECs denudation, fibrous cap thinning, plaque hemorrhage, and calcification [42] but also lumen dilatation when the plaque is not circular [43]. Humoral effects include higher production of nitric oxide but also of growth factors and activation of von Willebrand factor, etc. [44]. In other words, chronically high WSS contributes to plaque instability and rupture.
Wall Shear Stress Visualization In Vivo
As is explained above, WSS depends on diameter, velocity, and viscosity. The latter is frequently not included in in vivo calculations (and in fact, we calculate wall shear rate instead of stress). Therefore, the visualization and quantification of WSS in patients are based on two foundations: anatomy (precise delineation of vessel wall boundaries) and flow (luminal flow profile must be either measured or calculated using computational fluid dynamics). These two inputs are then used to calculate, quantify, and project WSS on the vessel wall.
Ultrasound Doppler imaging and magnetic resonance imaging (MRI) quantitative flow imaging are mostly used for WSS quantification. Detailed arterial wall structure and lumen analysis could be also obtained by computerized tomography (CT), intravascular ultrasound (IVUS), or optical coherence tomography (OCT). Flow characteristics are then added by image fusion.
Flow characteristic is close to laminar in straight arterial segments (common carotid artery, descending aorta, superficial femoral artery, etc.). In this case, a parabolic velocity profile is expected, and it is easier to estimate WSS, such as in the common carotid artery by ultrasound. The situation is much more difficult in non-straight segments, bifurcations, stenoses, arches, etc. Flow dissipation and turbulence occur in such areas, and, therefore, velocity profiling or vector analysis is necessary for the more precise WSS estimation.
Doppler ultrasonography has the advantage of being easily accessible and cheap. Straight segments WSS has been calculated and validated repeatedly by this method [35] [37]; see also Fig. 2. However, due to significant differences in sound velocity, this method cannot “see” through bones or gas and can therefore visualize with the adequate quality only carotid and upper or lower extremity arteries. Some modern devices are equipped with vector flow imaging that can be also used for WSS analysis in bifurcations and arches, where the flow is non-laminar and where the plaques are most frequent [45•].
Small arteries (especially coronary and brain) that are more susceptible to occlusion due to progression of atherosclerotic changes or plaque rupture leading to cardiovascular events are less amenable to noninvasive flow characterization. Computation of shear stress in these small arteries requires high-resolution image and three-dimensional isotropic reconstruction of the vessel lumen which is best achieved by CT.
Coronary CT angiography (CCTA) is the only noninvasive method to depict coronary arteries in isotropic space with a spatial resolution below 0.4 mm and temporal resolution up to 66 ms [46]. Advances in CCTA made it the tool of choice for imaging of coronary vessel lumen and plaque composition and superseded invasive methods such as coronary angiography, IVUS, or OCT in low or moderate risk patients. Computation of flow dynamics has become part of CCTA evaluation with commercially available packages for clinical use [47]. WSS derived from computational models based on the three-dimensional anatomical structure has its clinical mirror. CT-derived characteristics of vulnerable plaques coexist with high shear stress on the plaque surface [4]. High-risk plaques (low-density, positive remodeling, napkin-ring sign, spotty calcification) in the left anterior descending artery had the highest WSS tertile [47].
Compared to CT, MRI has the advantage of flow quantification, the lack of radiation burden, but it has a lower spatial resolution and suffers from artifacts. The presence of different velocities within a single voxel leads to decreasing signal. Phase encoding can be performed along three orthogonal axes (3D phase-contrast angiography, 3D PCA) and synchronized with cardiac cycle by electrocardiogram or peripheral sensing of blood oxygen (cine mode) to capture flow variations during systole and diastole. PCA depicts both vessel lumen and velocity profile of the flow (Fig. 4).
Wall shear stress in the aorta by magnetic resonance. Color-encoded axial wall shear stress map in the aorta derived from four-dimensional quantitative flow (4D flow) magnetic resonance acquisition. The low WSS is depicted in blue as is evident along the inner wall of the aortic arch. Color irregularities in the thoracic and abdominal parts are caused by atherosclerotic plaques
In human volunteers and patients, WSS distribution based on PCA MRI has been most explored in the aorta and the carotid arteries. The clinical utility of WSS derived from PCA MRI is limited by the accuracy of the definition of vessel boundaries, low spatial resolution, background noise, phase offset, fair reproducibility, and limited supportive longitudinal data [48].
MRI-based WSS calculations have attempted to evaluate more complex anatomical structures including aneurysms. However, the reproducibility of these measurements and calculations was limited, and the correlation with clinical significance was fair. Apart from WSS, other parameters have been calculated from the flow profile acquired by four-dimensional magnetic resonance angiography (4D MRA). Pulse wave velocity measures the propagation speed of the systolic pulse, and it is a measure of arterial stiffness and early marker of atherosclerosis. Turbulent kinetic energy estimates the loss of energy or pressure due to turbulent blood flow [49].
Steady/Non-Pulsatile Flow and Wall Shear Stress
Modern medicine brings situations when arterial flow loses its pulsatility and becomes steady as the mechanical circulatory support devices either contribute to the cardiac output or even replace it. These lifesaving methods therefore can also serve as a model of flow changes. Mechanical circulatory support devices with steady/non-pulsatile flow, most commonly represented by cardiopulmonary bypass, centrifugal pumps, venoarterial extracorporeal membrane oxygenation, and left/right ventricular assist devices, provide effective short- or long-term circulatory support with the restoration of adequate end-organ blood flow [50]. However, only limited data exist describing the effect of continuous blood flow on the vascular wall and its clinical consequences, especially in long-term ventricular assist devices and its difficult steady WSS from the lack of pulsatile tangential stress. It has been shown that a reduction of arterial pulse pressure is associated with the development of atrophic changes in the arterial wall, which was documented in in vitro [51] and animal [52] studies with subsequent confirmation in human histopathological studies [53]. The typical aortic pathological changes include increased foci of medial degeneration, smooth muscle cell disorientation and depletion, elastic fiber fragmentation, and medial fibrosis [53]. Contrariwise, cases of renal artery hypertrophy [54] and renal periarteritis [55] with local upregulation of the renin-angiotensin system have been reported. Wall shear stress change caused by non-pulsatile flow during cardiopulmonary bypass in cardiac surgery has been associated with degradation of the endothelial glycocalyx [56]. Impaired vascular function was observed in left ventricular assist device patients [57]. All these findings warrant further studies to clarify the consequences of continuous non-pulsatile flow on long-term outcomes.
Conclusions
WSS changes play a pivotal role in the development of atherosclerosis in concert with risk factors. Arterial sites with chronically low WSS that cannot respond by diameter reduction due to their geometry and wall changes are first affected by plaques. This is due to changed endothelial permeability and function. However, it has been shown that some patients have lower WSS also in straight arterial segments (especially in the common carotid artery). The reason is probably in age- and disease-mediated arterial dilatation. Lower WSS in the common carotid artery predicts the presence of bifurcation plaques and brain white-matter hyperintensities. High WSS occurs in arterial stenoses and seems to contribute to plaque destabilization and coagulation activation.
Abbreviations
- CT:
-
Computerized tomography
- CCTA:
-
Computerized tomography coronary angiography
- ECs:
-
Endothelial cells
- IVUS:
-
Intravascular ultrasonography
- 4D MRA:
-
Four-dimensional magnetic resonance angiography
- MRI:
-
Magnetic resonance imaging
- OCT:
-
Optical coherence tomography
- PECAM-1:
-
Platelet endothelial cell adhesion molecule-1
- VEGF:
-
Vascular endothelial growth factor
- WSS:
-
Wall shear stress
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Goyal A, Cho L. Preventive cardiology and risk assessment: beyond LDL. Curr Atheroscler Rep. 2020;22(10):56. https://doi.org/10.1007/s11883-020-00876-2.
•• Kojima K, Hiro T, Koyama Y, Ohgaku A, Fujito H, Ebuchi Y, et al. High wall shear stress is related to atherosclerotic plaque rupture in the aortic arch of patients with cardiovascular disease: a study with computational fluid dynamics model and non-obstructive general angioscopy. J Atheroscler Thromb. 2021;28(7):742–53. https://doi.org/10.5551/jat.56598. (This study demonstrates plaque aortic rupture in areas of high wall shear stress.)
Cunningham KS, Gotlieb AI. The role of shear stress in the pathogenesis of atherosclerosis. Lab Invest. 2005;85(1):9–23. https://doi.org/10.1038/labinvest.3700215.
Murata N, Hiro T, Takayama T, Migita S, Morikawa T, Tamaki T, et al. High shear stress on the coronary arterial wall is related to computed tomography-derived high-risk plaque: a three-dimensional computed tomography and color-coded tissue-characterizing intravascular ultrasonography study. Heart Vessels. 2019;34(9):1429–39. https://doi.org/10.1007/s00380-019-01389-y.
Aizawa K, Ramalli A, Sbragi S, Tortoli P, Casanova F, Morizzo C, et al. Arterial wall shear rate response to reactive hyperaemia is markedly different between young and older humans. J Physiol-London. 2019;597(16):4151–63. https://doi.org/10.1113/Jp278310.
Chytilova E, Malik J, Kasalova Z, Dolezalova R, Stulc T, Ceska R. Lower wall shear rate of the common carotid artery in treated type 2 diabetes mellitus with metabolic syndrome. Physiol Res. 2009;58(2):185–91. https://doi.org/10.33549/physiolres.931445.
Stroud JS, Berger SA, Saloner D. Numerical analysis of flow through a severely stenotic carotid artery bifurcation. J Biomech Eng. 2002;124(1):9–20. https://doi.org/10.1115/1.1427042.
Razavi A, Shirani E, Sadeghi MR. Numerical simulation of blood pulsatile flow in a stenosed carotid artery using different rheological models. J Biomech. 2011;44(11):2021–30. https://doi.org/10.1016/j.jbiomech.2011.04.023.
Caro C. The Mechanics of the Circulation. 2 ed. Cambridge University Press; 2011.
Franco CA, Jones ML, Bernabeu MO, Geudens I, Mathivet T, Rosa A, et al. Dynamic endothelial cell rearrangements drive developmental vessel regression. PLoS Biol. 2015;13(4):e1002125. https://doi.org/10.1371/journal.pbio.1002125.
Carter HH, Atkinson CL, Heinonen IH, Haynes A, Robey E, Smith KJ, et al. Evidence for shear stress-mediated dilation of the internal carotid artery in humans. Hypertension. 2016;68(5):1217–24. https://doi.org/10.1161/HYPERTENSIONAHA.116.07698.
Poduri A, Chang AH, Raftrey B, Rhee S, Van M, Red-Horse K. Endothelial cells respond to the direction of mechanical stimuli through SMAD signaling to regulate coronary artery size. Development. 2017;144(18):3241–52. https://doi.org/10.1242/dev.150904.
Wragg JW, Durant S, McGettrick HM, Sample KM, Egginton S, Bicknell R. Shear stress regulated gene expression and angiogenesis in vascular endothelium. Microcirculation. 2014;21(4):290–300. https://doi.org/10.1111/micc.12119.
Malek AM, Alper SL, Izumo S. Hemodynamic shear stress and its role in atherosclerosis. JAMA. 1999;282(21):2035–42. https://doi.org/10.1001/jama.282.21.2035.
Frangos JA, Eskin SG, McIntire LV, Ives CL. Flow effects on prostacyclin production by cultured human endothelial cells. Science. 1985;227(4693):1477–9. https://doi.org/10.1126/science.3883488.
Joannides R, Haefeli WE, Linder L, Richard V, Bakkali EH, Thuillez C, et al. Nitric oxide is responsible for flow-dependent dilatation of human peripheral conduit arteries in vivo. Circulation. 1995;91(5):1314–9. https://doi.org/10.1161/01.cir.91.5.1314.
Celermajer DS, Sorensen KE, Bull C, Robinson J, Deanfield JE. Endothelium-dependent dilation in the systemic arteries of asymptomatic subjects relates to coronary risk factors and their interaction. J Am Coll Cardiol. 1994;24(6):1468–74. https://doi.org/10.1016/0735-1097(94)90141-4.
Tuka V, Malik J, Chytilova E, Kudlicka J, Slavikova M, Wijnen E, Tordoir JHM, et al. Long-term arterial adaptation to high blood flow in the feeding artery of vascular access for hemodialysis. J Vasc Access. 2012;13(3):305–9. https://doi.org/10.5301/jva.5000048.
Zarins CK, Zatina MA, Giddens DP, Ku DN, Glagov S. Shear stress regulation of artery lumen diameter in experimental atherogenesis. J Vasc Surg. 1987;5(3):413–20.
Kamiya A, Togawa T. Adaptive regulation of wall shear stress to flow change in the canine carotid artery. Am J Physiol. 1980;239(1):H14-21. https://doi.org/10.1152/ajpheart.1980.239.1.H14.
Langille BL, O’Donnell F. Reductions in arterial diameter produced by chronic decreases in blood flow are endothelium-dependent. Science. 1986;231(4736):405–7. https://doi.org/10.1126/science.3941904.
Langille BL, Bendeck MP, Keeley FW. Adaptations of carotid arteries of young and mature rabbits to reduced carotid blood flow. Am J Physiol. 1989;256(4 Pt 2):H931–9. https://doi.org/10.1152/ajpheart.1989.256.4.H931.
Eigenbrodt ML, Bursac Z, Rose KM, Couper DJ, Tracy RE, Evans GW, et al. Common carotid arterial interadventitial distance (diameter) as an indicator of the damaging effects of age and atherosclerosis, a cross-sectional study of the Atherosclerosis Risk in Community Cohort Limited Access Data (ARICLAD), 1987–89. Cardiovasc Ultrasound. 2006;4:1. https://doi.org/10.1186/1476-7120-4-1.
• Ciurica S, Lopez-Sublet M, Loeys BL, Radhouani I, Natarajan N, Vikkula M, et al. Arterial Tortuosity. Hypertension. 2019;73(5):951–60. https://doi.org/10.1161/HYPERTENSIONAHA.118.11647. (Detailed review about arterial tortuosities.)
Strecker C, Krafft AJ, Kaufhold L, Hüllebrandt M, Weber S, Ludwig U, et al. Carotid geometry is an independent predictor of wall thickness - a 3D cardiovascular magnetic resonance study in patients with high cardiovascular risk. J Cardiovasc Magn Reson. 2020;22(1):67. https://doi.org/10.1186/s12968-020-00657-5.
Zhou J, Li YS, Chien S. Shear stress-initiated signaling and its regulation of endothelial function. Arterioscler Thromb Vasc Biol. 2014;34(10):2191–8. https://doi.org/10.1161/atvbaha.114.303422.
Roux E, Bougaran P, Dufourcq P, Couffinhal T. Fluid shear stress sensing by the endothelial layer. Front Physiol. 2020;11:861. https://doi.org/10.3389/fphys.2020.00861.
Resnick N, Yahav H, Shay-Salit A, Shushy M, Schubert S, Zilberman LC, et al. Fluid shear stress and the vascular endothelium: for better and for worse. Prog Biophys Mol Biol. 2003;81(3):177–99. https://doi.org/10.1016/s0079-6107(02)00052-4.
Woodfin A, Voisin MB, Nourshargh S. PECAM-1: a multi-functional molecule in inflammation and vascular biology. Arterioscler Thromb Vasc Biol. 2007;27(12):2514–23. https://doi.org/10.1161/atvbaha.107.151456.
Shin H, Haga JH, Kosawada T, Kimura K, Li YS, Chien S, et al. Fine control of endothelial VEGFR-2 activation: caveolae as fluid shear stress shelters for membrane receptors. Biomech Model Mechanobiol. 2019;18(1):5–16. https://doi.org/10.1007/s10237-018-1063-2.
Lim YC, Cooling MT, Long DS. Computational models of the primary cilium and endothelial mechanotransmission. Biomech Model Mechanobiol. 2015;14(3):665–78. https://doi.org/10.1007/s10237-014-0629-x.
Caplan BA, Schwartz CJ. Increased endothelial cell turnover in areas of in vivo Evans blue uptake in the pig aorta. Atherosclerosis. 1973;17(3):401–17. https://doi.org/10.1016/0021-9150(73)90031-2.
Silva T, Jäger W, Neuss-Radu M, Sequeira A. Modeling of the early stage of atherosclerosis with emphasis on the regulation of the endothelial permeability. J Theor Biol. 2020;496:110229. https://doi.org/10.1016/j.jtbi.2020.110229.
Zhang B, Ma Y, Ding F. Evaluation of spatial distribution and characterization of wall shear stress in carotid sinus based on two-dimensional color Doppler imaging. Biomed Eng Online. 2018;17(1):141. https://doi.org/10.1186/s12938-018-0589-y.
Malik J, Kudlicka J, Tuka V, Chytilova E, Adamec J, Rocinovaá K, et al. Common carotid wall shear stress and carotid atherosclerosis in end-stage renal disease patients. Physiological Res. 2012;61(4):355–61. https://doi.org/10.33549/physiolres.932259.
Zhang B, Gu J, Qian M, Niu L, Zhou H, Ghista D. Correlation between quantitative analysis of wall shear stress and intima-media thickness in atherosclerosis development in carotid arteries. Biomed Eng Online. 2017;16(1):137. https://doi.org/10.1186/s12938-017-0425-9.
Liu ZD, Zhao YX, Wang XD, Zhang H, Cui Y, Diao YT, et al. Low carotid artery wall shear stress is independently associated with brain white-matter hyperintensities and cognitive impairment in older patients. Atherosclerosis. 2016;247:78–86. https://doi.org/10.1016/j.atherosclerosis.2016.02.003.
Brown AJ, Teng Z, Evans PC, Gillard JH, Samady H, Bennett MR. Role of biomechanical forces in the natural history of coronary atherosclerosis. Nature Rev Cardiol. 2016;13(4):210–20. https://doi.org/10.1038/nrcardio.2015.203.
Thijssen DHJ, Carter SE, Green DJ. Arterial structure and function in vascular ageing: are you as old as your arteries? J Physiol-London. 2016;594(8):2275–84. https://doi.org/10.1113/Jp270597.
Brassard PA-O, Labrecque L, Smirl JA-O, Tymko MM, Caldwell HG, Hoiland RL et al. Losing the dogmatic view of cerebral autoregulation. Physiol Rep. 2021;e14982 https://doi.org/10.14814/phy2.14982.
Wu SP, Ringgaard S, Oyre S, Hansen MS, Rasmus S, Pedersen EM. Wall shear rates differ between the normal carotid, femoral, and brachial arteries: an in vivo MRI study. J Magnet Reson Imaging. 2004;19(2):188–93. https://doi.org/10.1002/jmri.10441.
Urschel K, Tauchi M, Achenbach S, Dietel B. Investigation of wall shear stress in cardiovascular research and in clinical practice-from bench to bedside. Int J Mol Sci. 2021;22(11). https://doi.org/10.3390/ijms22115635.
Samady H, Molony DS, Coskun AU, Varshney AS, De Bruyne B, Stone PH. Risk stratification of coronary plaques using physiologic characteristics by CCTA: focus on shear stress. J Cardiovasc Comput Tomogr. 2020;14(5):386–93. https://doi.org/10.1016/j.jcct.2019.11.012.
Wijeratne SS, Botello E, Yeh HC, Zhou Z, Bergeron AL, Frey EW, Patel JM, et al. Mechanical activation of a multimeric adhesive protein through domain conformational change. Phys Rev Lett. 2013;110(10):108102. https://doi.org/10.1103/PhysRevLett.110.108102.
• Du Y, Goddi A, Bortolotto C, Shen Y, Dell’Era A, Calliada F, et al. Wall shear stress measurements based on ultrasound vector flow imaging: theoretical studies and clinical examples. J Ultrasound Med. 2020;39(8):1649–64. https://doi.org/10.1002/jum.15253. (An interesting study of the carotid flow visualization by means of ultrasound vector imaging.)
Ghekiere O, Salgado R, Buls N, Leiner T, Mancini I, Vanhoenacker P, et al. Image quality in coronary CT angiography: challenges and technical solutions. Br J Radiol. 2017;90(1072):20160567. https://doi.org/10.1259/bjr.20160567.
Park JB, Choi G, Chun EJ, Kim HJ, Park J, Jung JH, et al. Computational fluid dynamic measures of wall shear stress are related to coronary lesion characteristics. Heart. 2016;102(20):1655–61. https://doi.org/10.1136/heartjnl-2016-309299.
Dyverfeldt P, Bissell M, Barker AJ, Bolger AF, Carlhäll CJ, Ebbers T, et al. 4D flow cardiovascular magnetic resonance consensus statement. J Cardiovasc Magn Res. 2015;17(1):72. https://doi.org/10.1186/s12968-015-0174-5.
Morgant MA, Lin S, Marin-Castrillon D, Bernard CA-O, Laubriet A, Cochet A et al. Comparison of two techniques (in vivo and ex-vivo) for evaluating the elastic properties of the ascending aorta: prospective cohort study. PLoS One 2021; e0256278. https://doi.org/10.1371/journal.pone.0256278
Hajjar LA, Teboul JL. Mechanical circulatory support devices for cardiogenic shock: state of the art. Crit Care. 2019;23(1):76. https://doi.org/10.1186/s13054-019-2368-y.
Birukov KG, Shirinsky VP, Stepanova OV, Tkachuk VA, Hahn AW, Resink TJ, et al. Stretch affects phenotype and proliferation of vascular smooth muscle cells. Mol Cell Biochem. 1995;144(2):131–9. https://doi.org/10.1007/BF00944392.
Yao Q, Hayman DM, Dai Q, Lindsey ML, Han HC. Alterations of pulse pressure stimulate arterial wall matrix remodeling. J Biomech Eng. 2009;131(10):101011. https://doi.org/10.1115/1.3202785.
Segura AM, Gregoric I, Radovancevic R, Demirozu ZT, Buja LM, Frazier OH. Morphologic changes in the aortic wall media after support with a continuous-flow left ventricular assist device. J Heart Lung Transplant. 2013;32(11):1096–100. https://doi.org/10.1016/j.healun.2013.07.007.
Kihara S, Litwak Kn Fau - Nichols L, Nichols L Fau - Litwak P, Litwak P Fau - Kameneva MV, Kameneva Mv Fau - Wu Z, Wu Z Fau - Kormos RL et al. Smooth muscle cell hypertrophy of renal cortex arteries with chronic continuous flow left ventricular assist. Ann Thorac Surh 2003;75(1):178–83. https://doi.org/10.1016/s0003-4975(02)04087-0
Ootaki C, Yamashita M Fau - Ootaki Y, Ootaki Y Fau - Kamohara K, Kamohara K Fau - Weber S, Weber S Fau - Klatte RS, Klatte Rs Fau - Smith WA et al. Reduced pulsatility induces periarteritis in kidney: role of the local renin-angiotensin system. J Thorac Cardiovasc Surg 2008;136(1):150–8. https://doi.org/10.1016/j.jtcvs.2007.12.023
He G, Gao Y, Feng L, He G, Wu Q, Gao W, et al. Correlation between wall shear stress and acute degradation of the endothelial glycocalyx during cardiopulmonary bypass. J Cardiovasc Transl Res. 2020;13(6):1024–32. https://doi.org/10.1007/s12265-020-10027-2.
Ivak P, Netuka I, Tucanova Z, Wohlfahrt P, Konarik M, Szarszoi O et al. The effect of artificial pulsatility on the peripheral vasculature in patients with a continuous-flow ventricular assist device. Can J Cardiol 2021;S0828–282X(21)00289–0. https://doi.org/10.1016/j.cjca.2021.05.013
Layek GC, Mukhopadhyay S. Laminar flow separation in an axi-symmetric sudden smooth expanded circular tube. J Appl Math Comput. 2008;28:235–47. https://doi.org/10.1007/s12190-008-0097-5.
Banerjee RK, Back LH, Back MR, Cho YI. Physiological flow analysis in significant human coronary artery stenoses. Biorheology. 2003;40(4):451–76.
Long Q, Xu XY, Ramnarine KV, Hoskins P. Numerical investigation of physiologically realistic pulsatile flow through arterial stenosis. J Biomech. 2001;34(10):1229–42. https://doi.org/10.1016/s0021-9290(01)00100-2.
Deplano V, Siouffi M. Experimental and numerical study of pulsatile flows through stenosis: wall shear stress analysis. J Biomech. 1999;32(10):1081–90. https://doi.org/10.1016/s0021-9290(99)00098-6.
Novakova L, Kolinsky J, Adamec J, Kudlicka J, Malik J. Vascular stenosis asymmetry influences considerably pressure gradient and flow volume. Physiol Res. 2016;65(1):63–9. https://doi.org/10.33549/physiolres.932944.
Funding
This work was supported by MH CZ — DRO (General University Hospital in Prague — VFN, 00064165).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Statin Drugs
Rights and permissions
About this article
Cite this article
J., M., L., N., A., V. et al. Wall Shear Stress Alteration: a Local Risk Factor of Atherosclerosis. Curr Atheroscler Rep 24, 143–151 (2022). https://doi.org/10.1007/s11883-022-00993-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11883-022-00993-0