Abstract
Thrips (Thysanoptera: Thripidae) feed on leaf and flower tissue and some of them can be facultative predators. Caliothrips phaseoli Hood causes severe damage to different species of the Fabaceae family, mainly to Phaseolus vulgaris L. crops. The purpose of this research is to assess the change in feeding damage due to predation on Tetranychus merganser (Acari: Tetranychidae) eggs by the first and second larval stages of C. phaseoli on bean leaf discs, under laboratory conditions. Predation and damage were assessed through functional response and ordinal scale, respectively. The first and second larval stages of C. phaseoli exhibited a type II functional response. The second larval stage of C. phaseoli displayed shorter handling time and higher attack rate in comparison to the first instar. The first and second larval stages of C. phaseoli consumed 3.8 and 4.9 eggs of T. merganser per day, respectively. The second larval stage caused the highest damage (median = 3). The feeding damage of C. phaseoli was negatively associated to the number of eggs offered. The C. phaseoli larvae became more predatory than herbivorous when the number of eggs offered increased; therefore, C. phaseoli can be considered a facultative predator.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The Thripidae family (Thysanoptera: Terebrantia) contains around 2100 species, categorised in 242 genera and divided in four sub-families: Dendrothripinae, Panchaetothripinae, Sericothripinae and Thripinae. The Thripinae sub-family can be found all over the world, most of its species reproduce on flowers and leaves; some of them feed on mosses and few of them are predators (CSIRO 2022). Therefore, it is important to study the omnivory of thrips, which could potencially reduce damage from agricultural pests. Caliothrips phaseoli Hood (Thysanoptera: Thripidae) distribution ranges across the American continent, from the southeast of the United States to Argentina. This species causes damage by feeding on different Fabaceae species, such as bean (Phaseolus vulgaris L.), soybean (Glycine max (L.) Merr), pea (Pisum sativum L.), peanut (Arachis hypogaea L.) and “garapa” or “grapia” [Apuleia leiocarpa (Vogel) J. F. Macbr.] (Boscardin et al. 2019). Also, C. phaseoli, has been reported to cause feeding damage on the leaves of Moringa tree [Moringa oleifera Lam. (Moringaceae)] (Molina-Arjona et al. 2018), corn [Zea mays L. (Poaceae)] and cotton [Gossypium sp. L. (Malvaceae)] (García-Martínez et al. 2011). It is considered an important pest of P. vulgaris crops (Janini et al. 2011; Hoddle et al. 2012; Boscardin et al. 2019; Mound et al. 2019). C. phaseoli feed on the underside of primary leaves of newly emerged plants in both first and second larval stages, as well as adults; while on developed plants C. phaseoli can be found scraping and sucking sap from leaves, flowers, and petioles (Boiça-Júnior et al. 2015; Flores 2021). The damage caused by C. phaseoli feeding habits cause leaf yellowing, leading sometimes to the development of silver spots in leaves and pods. Furthermore, C. phaseoli causes leaf deformation and delays plant growth and development (Beltrán et al. 2004; Janini et al. 2011). Also, C. phaseoli feeds on red spider mites’ eggs as well as adult specimens of Tetranychus merganser Boudreaux (Acari: Tetranychidae) (Molina-Arjona et al. 2018).
The insects that are facultative predators have the capacity to feed on plants and prey, at different stages of insect development (Albajes and Alomar 2008; Molina-Arjona et al. 2018). One of the advantages of facultative predators is their ability to obtain supplementary nutrients such as vitamins, water, minerals, symbionts or enzymes from alternative food sources (either plants or prey), enhancing their development, survival, fertility and longevity rates (Trichilo and Leigh 1988; Milne and Walter 1997; Albajes and Alomar 2008). Multiple studies have reported different species of phytophagous thrips behaving as facultative predators, feeding on eggs of Tetranychus urticae Koch (Acari: Tetranychidae) growing on cotton, under laboratory and field conditions, such as Thrips imaginis Bagnall (Thysanoptera: Thripidae), T. tabaci Lindeman (Thysanoptera: Thripidae) (Wilson et al. 1996), Frankliniella schultzei Trybom (Thysanoptera: Thripidae) (Wilson et al. 1996; Milne and Walter 1997) and F. occidentalis Pergande (Thysanoptera: Thripidae) (González et al. 1982; Gonzalez and Wilson 1982; Trichilo and Leigh 1986, 1988).
T. merganser is found in China, Mexico, United States and Thailand and feeds on 17 plant species (Migeon and Dorkeld 2022). In Mexico, T. merganser is considered a potential pest for agriculture, causing losses that range between $352 and $820 dollars per hectare on prickly pear crops [Opuntia ficus-indica L.) Miller (Cactaceae)] (Lima-Espíndola and Vanegas-Rico 2017). The feeding habits of this mite causes several types of damage: it destroys the epidermal tissue, the parenchyma and the chloroplasts of leaves, affecting also the growth, development and production of the host plant. This damage can be observed as white spots near the leaf veins, and when the red spider mite populations are high, the spots can merge, causing the leaves to turn completely white (López-Bautista 2014; López-Bautista et al. 2016). The control of T. merganser is achieved through insecticides and chemical acaricides. However, the short life cycle and the high reproductive potential of the red spider mite lead to fast development of resistance to these compounds (Ullah et al. 2011). Predator mites, such as Neoseiulius californicus (McGregor) and Amblyseius swirskii (Athias-Henriot) (Gamasida: Phytoseiidae) are used as preventive control methods on the populations of red spider mites attacking papaya crops (López-Bautista et al. 2016). Molina-Arjona et al. (2018) documented that the first and second larval stages of C. phaseoli feed on eggs and adult females of T. merganser; but they did not assess the relationship between feeding damage and mite predation on bean leaf discs. Tetranychidae eggs, such as Panonychus citri McGregor (Cabrera et al 2009) and Tetranychus urticae Koch (Zhong et al. 2015) contain high contents of amino acids, lipids, carbohydrates, vitamins and other nutrients. The aim of this research is to assess the change in feeding damage due to predation on T. merganser eggs by the first and second larval stages of C. phaseoli on bean leaf discs, under laboratory conditions. This can provide a better understanding of the role that C. phaseoli larvae play on agricultural crops as phytophagous and predators.
Materials and methods
Tetranychus merganser eggs
Tetranychus merganser eggs were obtained from a colony in the Laboratory of Population Ecology, Institute of Applied Ecology at the Universidad Autónoma de Tamaulipas in Mexico (IAE-UAT). The original colony was collected from Capsicum annuum var. glabriusculum (23°41′52″ N, 99°11′04″ W). The individual specimens provided by IAE-UAT were part of the 10th generation of red spider mites. In order to improve the reproduction and oviposition of the red spider mites, 100 females and 100 males were placed on bean plants (P. vulgaris L.) under greenhouse conditions of 30 ± 2 °C and 70 ± 10% of relative humidity (RH). To germinate the bean plants we used polyethylene bags (5 × 7 × 10 cm high) with a growth medium of soil: vermiculite (1:0.5), which was fertilised once a week only with HUMIMAX (Humic substances derived from 12% leonardite, 2% soluble potassium, 0.5% amino acids, 3% manganese (Mn), 3% iron (Fe), 1% zinc (Zn), 0.5% boron (B), and 78% humectants, dispersants, and penetrating) at 2.5 ml per liter of water. To maintain the red spider mite colony, clean 20 day old plants were added.
Caliothrips phaseoli larvae
First and second larval stages from C. phaseoli were collected from from moringa trees (M. oleifera), 2 ± 0.15 m-tall, growing under greenhouse conditions at 29 ± 5 °C and 70 ± 10% of HR. Several leaflets of M. oleifera with C. phaseoli larvae were cut and taken to the Laboratory of Population Ecology in IEA-UAT.
Experimental design
Predation
Experiments regarding functional response and feeding damage of the first and second larval stages of C. phaseoli were performed in a bioclimatic chamber using Ahmadi’s modified sand technique (Ahmadi 1983), using 2.0 cm diameter bean discs with the underside facing up, on water-saturated cotton, plated on 5 cm-diameter petri dishes, at a temperature of 28 ± 1 °C with 70–80% of relative humidity and a photoperiod of 12:12 light:darkness. In order to improve the reproduction and oviposition of the females, 10 females and 5 males of T. merganser were placed on a clean leaf disk. After 24 h, the females and males were removed, adjusting the number of eggs per bean leaf disc to densities of 1, 2, 4, 6, 8 and 10. The rest of the eggs were removed with the help of a thin brush. Immediately after removing the mite eggs, one larva (I or II instar) of C. phaseoli was placed on each disc. Eight replicates were made per each treatment (density). The number of eggs consumed by the first and second instar larvae was recorded at 24 h.
In the second experiment of predation, the same Ahmadi’s sand modified technique (1983) described above was used. In order to improve the reproduction and oviposition of the females, 20 females and 10 males of T. merganser were placed on a clean leaf disk. After 24 h the females and males were removed, adjusting the number of eggs per bean leaf disc to 20. The rest of the eggs were removed with the help of a thin brush. Immediately after removing the mite eggs, a first or second instar larva of C. phaseoli was placed on each bean leaf disc. Six replicates were made per each instar (treatments). The number of eggs eaten by the first and second instar larvae of C. phaseoli was recorded at 72 h.
Damage by larval feeding
Predation rates were assessed at 24 and 72 h. The damage caused by feeding of the first and second larval stages of C. phaseoli was visually estimated on each bean disc (where the red spider mite eggs were offered as food). Although in each experiment females and males only remained 24 h on the clean bean disc to improve oviposition, feeding damage caused by thrips larvae differ from mites because thrips larvae scrape the leaf when sucking the sap while mites suck the sap by inserting their stylet in the leaf. The experiment uses the damage scale proposed by van Haperen et al. (2019); in which 1 = no damage (0% feeding damage), 2 = tiny silver spots (< 0.1%), 3 = some small silver spots distributed near the leaf central vein or on the leaf disc edge (0.1–1%), 4 = some small spots evenly distributed all over the leaf disc (1–2%), 5 = moderate number of spots, evenly distributed all over the leaf disc (3–5%), 6 = intermediate between 5 and 7 (6–10%), 7 = many silver spots over all the leaf disc (11–20%), 8 = intermediate between 7 and 9 (21–40%), and 9 = very intense silver color, large damaged leaf area (> 40% of feeding damage). The scale of the damage was recorded at 24 and 72 h.
Analysis of predation data
The type of functional response was determined (shape of the curve) by the analysis of logistic regression, adjusting a polynomial equation of the prey eaten ratio (NA/NO) versus the number of prey offered (NO) (Juliano 2001).
where NA is the number of prey eaten, NO is the number of prey offered, P1, P2 and P3 are the parameters to estimate using the method of maximum likelihood.
The regression started with a cubic model. The higher order coefficients that were not significantly different from zero were eliminated, until all the coefficients of the model were significantly different from zero. To separate the functional response type I from type II and type III, the linear coefficient of the model was used. For the functional response type II, the linear term of the model had to be significantly negative (P1 < 0); for the functional response type III, the linear term had to be significantly positive and the quadratic coefficient significantly negative (P1 > 0, P2 < 0). If the linear coefficient was not significant, then it was considered to be type I (Juliano 2001). The parameters were estimated using “glm” in R software (Dobson & Barnett 2008; R Core Team 2022).
The parameters of the functional response (attack coefficient “a” and handling time “Th”), were estimated using Holling’s model (Holling 1959) for the functional response type II.
in which NA/P is the number of prey consumed (NA) per predator (P), T is the total available time (1 day in this essay), NO is the density of prey offered prey, a is the attack rate and Th is the handling time. The parameters were estimated using the function “nls” in R software (R Core Team 2022). The predation data recorded on the third day for the first and the second larval stages were analyzed through the t-Students’ test.
Analysis of the feeding damage data
The score of feeding damage caused by C. phaseoli was recorded at 24 and 72 h. At 24 h, the Kruskal–Wallis test was used to compare the damage when different T. merganser egg densities were offered to the thrips (1, 2, 4, 6, 8, 10). In order to analyze the leaf damage index recorded on the third day, the Wilcoxon test was used to compare the damage caused by the first and second larval stages when they were offered 20 eggs of T. merganser per bean leaf. Software by R Development Core Team (2022) was used in all the assay.
Results
The logistic regression analysis indicated a functional response of type II for the first and second larval stages of C. phaseoli (Table 1). The linear coefficient resulted significantly negative (P1 < 0). This indicates that the proportion of eaten eggs decreased gradually as the initial number of eggs offered increased.
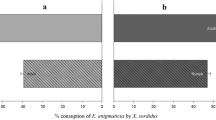
The results show that the first and second larval stages of C. phaseoli ate eggs of T. merganser, causing feeding damage on the same bean leaf disc (Fig. 1). The first and second larval stages of C. phaseoli ate 3.8 and 4.9 eggs of T. merganser when they were offered eight eggs (Fig. 1A, B), respectively. In general, the number of eggs eaten by both larval stages of C. phaseoli hyperbolically approached the asymptote, as the density of eggs offered increased (Fig. 1).
At 24 h, the feeding damage caused by the first and second larval stages of C. phaseoli on the bean discs differed significantly when they were offered different egg densities (H = 16.08, df = 5, P = 0.0018; H = 18.04, df = 5, P = 0.0006), respectively. The first and second larval stages of C. phaseoli caused higher damage on those bean leaf discs in which the number of eggs offered was lower (one up to four eggs) (Fig. 1). The highest level of damage was caused by the second larval stage (median = 3). A negative correlation was found between egg consumption and damage caused in the first and second larval stages. In other words, the higher the egg consumption, the lesser the damage caused by larvae feeding (First larval stage: S = 65.741; Rho = − 0.8783, P = 0.0213; Second larval stage: S = 65.741; Rho = − 0.8783, P = 0.0213), respectively. In general, predation rate tended to decrease with prey density, probably due to an increase in satiety level of C. phaseoli in the areas with higher density of T. mergnaser eggs.
The estimated values for a and Th of the first and second larval stages of C. phaseoli over the eggs of T. merganser are shown in Table 2. An increase in the attack rate “a” and a decrease in Th are observed in the second larval stage, compared to the first larval stage, likely due to the size of the predator. This is due because the second larval stage is larger in size and requires less time to dominate, eat and digest the eggs of T. merganser, as well as to prepare for the next search. Therefore, the attack rate increases. The second larval stage showed a higher upper limit (NA–max = 9) to predation than the first larval stage (NA–max = 7), which was associated with a shorter Th for the second larval stage (Table 2), where NA–max = T/Th.
At 72 h, egg consumption differed significantly between the first and second larval stages of C. phaseoli (t = 5.7565, gl = 10, P = 0.0002). The first larval stage consumed (± SD) less eggs (mean: 7.00 ± 1.63) compared to the second larval stage (12.00 ± 1.47) (Fig. 2).
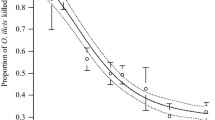
Regarding feeding damage, this differed significantly between the first and second larval stages of C. phaseoli (U = 3, P = 0.0151). The second larval stage caused the highest damage (median = 5.5) in comparison to the first larval stage (median = 3.5) (Fig. 3), suggesting that the second larval stage requires a higher amount of nutrients.
Boxplot comparing the foliar feeding damage score on Phaseolus vulgaris leaf discs caused by Caliothrips phaseoli larvae during a three day period. Box plot elements: Line through the box is median of the damage score. × is mean of the damage score. Top of upper whisker is the maximum value of the damage score. Top of box is the 75th percentile of the damage score. Bottom of the box is the 25th percentile of the of the damage score. Bottom of the lower whisker is the minimum of the of the damage score
Discussion
The first and second larval stages of C. phaseoli caused feeding damage and consumed T. merganser eggs when held on the same bean leaf discs. Also, it was demonstrated that the first and second larval stages decreased their food intake of cell content in bean leaves when the number of eggs of T. merganser increased. According to these results, the phytophagous and predatory behaviour of C. phaseoli differs when this omnivore is found confined to a disc, on a plant or when the plants are grown in a laboratory, greenhouse, cages, or open field. Still, the results of this research bring into view the role of C. phaseoli larvae in agricultural ecosystems and can help predict C. phaseoli impact as a predator of T. merganser population and as a phytophagous in agricultural crops, such as bean crops. This research shows that the functional response of the first and second larval stages of C. phaseoli was of type II and that the consumption rate increases in relation to the increase on the number of offered eggs, which is consistent with the results reported by other researchers (Trichilo and Leigh 1986; Wilson et al. 1996). Trichilo and Leigh (1986) reported a functional response type II of F. occidentalis larvae on eggs of T. urticae. Wilson et al. (1996) found the same type of response in larvae of the first and second larval stages of F. schultzei; as well as in larvae of the second larval stage of T. imaginis and T. tabaci.
The attack rate “a” increased while the handling time “Th” decreased from the first to the second stage of C. phaseoli, which is consistent with the results of Wilson et al. (1996), which describe an increase of “a” and a decrease in “Th” for F. schultzei when it consumed T. urticae eggs. In this regard, Wilson et al. (1996) mentioned that these changes in parameters of functional response are due to the fact that the first stage is too small to deal effectively with mite eggs.
There is no reference in literature regarding the assessment of feeding damage and consumption of eggs of T. merganser or other Tetranychidae by C. phaseoli larvae on the same bean leaf disc. So far, the research has focused only on the predation of C. phaseoli first and second larval stages over the eggs and females of T. merganser on discs of P. vulgaris and M. oleifera leaflets (Molina-Arjona et al. 2018). Those results are similar to the results reported in this work. Molina-Arjona et al. (2018) reported a consumption rate of C. phaseoli in first larval stage of 3.60 ± 1.14 eggs of T. merganser per day on bean discs, and 1.80 ± 0.83 eggs of T. merganser on M. oleifera leaflets; while, the second stage of C. phaseoli consumed 6.20 ± 0.83 eggs of T. merganser per day on bean discs and 4.60 ± 1.14 eggs of T. merganser on M. oleifera leaflets. Besides, these authors reported that larvae of the first stage do not eat red spider mite adult females on moringa tree leaflets; however, they feed on the bean discs, at a consumption rate of 0.40 red spider mite adult females per day; while, the second larval stage predates 1.60 ± 0.54 adult females of T. merganser on beans and 0.40 ± 0.55 on moringa tree leaflets. Chacón-Hernández et al. (2017) documented that C. phaseoli larvae impacted on the growth rate of T. merganser under greenhouse conditions.
Moreover, predation of other phytophagous thrip species on eggs of T. urticae has been studied with results similar to this research. Milne and Walter (1997) reported that the second instar larvae of F. schultzei (22.10 ± 1.75) ate more eggs of T. urticae during 24 h than larvae from the first instar (0.35 ± 0.22). Wilson et al. (1996) documented that the first larval stage of F. schultzei ate less than one egg per day, while the second instar larvae ate between two and six eggs of T. urticae per day, and some larvae ate even nine eggs per day. Trichilo and Leigh (1986) reported that the second larval stage of F. occidentalis ate between 8 and 12 eggs of T. urticae per day. Whereas, Wilson et al. (1996) documented that the second stage of T. imaginis and T. tabaco ate between 1 and 2 eggs per day. Wilson et al. (1996) mention that the differences in the consumption rates among thrip species (Thysanoptera: Thripidae) is due to the nature of each species, regarding the acceptability of mite eggs as prey and the age of each instar, for example, Trichilo and Leigh (1986) used newly emerged larvae from the second instar of F. occidentalis; whereas, Wilson et al. (1996) used larvae from the first and the second instar of F. schultzei, as well as larvae from the second instar of T. tabaci and T. imagmis, all of them collected at the field, so the age of these larvae was unknown. Milne and Walter (1997) used newly emerged larvae of first and second instar of F. schultzei, and found that the consumption rate of the second larval stage of thrips significantly decreases with age.
The feeding damage of the second instar of C. phaseoli was higher than the damage caused by the first instar, which indicates that the second larval stage affects more the plants’physiology, reducing photosynthesis, stomatal conductance and the transpiration rate; which can be translated into crop yield losses (Gamundi and Perotti 2009). Flores (2021) mentioned that the damage caused by C. phaseoli feeding allows the entry of disease-causing agents, such as fungi and bacteria. In both larval stages (first and second) of C. phaseoli, the feeding damage decreased as the number of offered eggs of T. merganser increased. DeLong (2021) mentioned that predators change food sources (prey) because they seek food that can provide more proteins or other specific nutrients. Molina-Arjona et al. (2018) mentioned that the predatory activity of C. phaseoli satisfies certain nutritional needs and eliminates the competition for food resources, in a way that predatory activity can increase the survival rate of thrips populations. In this regard, Milne and Walter (1997) found that the phytophagous thrips F. schultzei enhances its development and fertility rate when it feeds on cotton leaf tissue plus the eggs of T. urticae, in contrast to when it only feeds on leaf tissue. Trichilo and Leigh (1988) documented that F. occidentalis has a higher probability of survival when it feeds on leaf tissue plus the eggs of T. urticae, compared to when it feeds only on leaf tissue. Also Trichilo and Leigh (1988), as well as Milne and Walter (1997) documented that F. occidentalis and F. schultzei larvae supplement their diet (proteins) by eating eggs of T. urticae.
Our results showed that C. phaseoli is an opportunistic insect with a high inclination to become omnivorous, because it feeds from both foliar tissue and prey (eggs of T. merganser). When the number of eggs offered (NO) is low (one to four), the thrip larvae resort to herbivore feeding. Eubanks (2005) mentioned that omnivorous insects survive by feeding only from plants when the abundance of prey is low or non-existent, but their reproductive performance is low. When the number of eggs offered increases, thrip larvae become more predatory than herbivore. In this regard, Eubanks (2005) mentions that omnivorous insects enhance their nutritional quality by consuming prey increasing their diet’s energy value compared to the consumption of plants alone. So, this behaviour causes C. phaseoli to become an omnivore but differs from predators that are strictly or mainly carnivorous, such as Scolothrips sexmaculatus Pergande (Thysanoptera: Thripidae), a predator specialized on mites from the Tetranychidae family (Haviland et al. 2021). Milne and Walter (1997) mention that the relationship between a phytophagous thrip (also predatory) and its host plant may be complicated by the existence of food items on plants. But under particular circumstances, red spider mite eggs may be a regular part of C. phaseoli diet, for instance in bean fields where the reproductive yield of the thrips is low due to the poor quality of bean leaves, which can impact in both C. phaseoli and T. merganser population dynamics. Moreover, the interaction between thrips and red spider mites can reduce the damage caused by both pests, i.e. if the larvae of C. phaseoli change their diet (mite eggs for bean leaves), the larvae can cause less damage to the bean crop. On the other hand, the reduction in the number of eggs due to the predation of C phaseoli reduces the T. merganser population, which imply less damage to bean plants. More research is needed to understand the behaviour of C. phaseoli as facultative predator, including the assessment of biological and demographical parameters of thrips feeding on foliar tissue, thrips feeding on leaves plus red spider mite eggs, and thrips feeding on pollen plus leaf tissue, under different experimental conditions.
References
Ahmadi A (1983) Demographic toxicology as a method for studying the dicofol two spotted spider mite (Acari:Tetranychidae) system. J Econ Entomol 76:239–242. https://doi.org/10.1093/jee/76.2.239
Albajes R, Alomar O (2008) Facultative Predators. In: Capinera JL (ed) Encyclopedia of entomology. Springer, Dordrecht, pp 1400–1405. https://doi.org/10.1007/978-1-4020-6359-6_3742
Beltrán R, Helman S, Peterlin O (2004) Control de Caliothrips phaseoli Hood y Frankliniella schultzei Trybon y Aphis gossypii Glover con insecticidas sistémicos aplicados a las semillas de algodón. RIA Rev Invest Agropecu 33:39–48
Boiça-Júnior AL, Costa EN, Souza BHS, Silva AG, Chiorato AF (2015) Infestation of Caliothrips phaseoli (Thysanoptera: Thripidae) on bean cultivars grown in the winter, rainy, and dry seasons in Brazil. Environ Entomol 44:1139–1148. https://doi.org/10.1093/ee/nvv100
Boscardin J, de Oliveira GG, Costa EC, Aimi SC, Fleck MD, Araujo MM (2019) First record of Caliothrips phaseoli (Hood, 1912) (Thysanoptera: Thripidae) in Apuleia leiocarpa (Fabaceae) seedlings in Rio Grande do Sul, Brazil. Pesqui Forest Bras 39:1–3. https://doi.org/10.4336/2019.pfb.39e201801636
Cabrera AR, Donohue KV, Khalil SMS, Sonenshine DE, Roe RM (2009) Characterization of vitellin protein in the twospotted spider mite, Tetranychus urticae (Acari: Tetranychidae). J Insect Physiol 55:655–661. https://doi.org/10.1016/j.jinsphys.2009.04.006
Chacón-Hernández JC, Castillo-Flores PM, Rocandio-Rodríguez M, Reyes-Zepeda F, Vanoye-Eligio V, Mora-Ravelo SG (2017) Impacto de la depredación de trips Caliothrips phaseoli1 Hood sobre Tetranychus merganser2 Boudreaux sobre Moringa oleifera3 Lamarck. Southwest Entomol 42:477–484. https://doi.org/10.3958/059.042.0217
CSIRO (2022) Thysanoptera (Thrips) of the World - a checklist. http://www.ento.csiro.au/thysanoptera/worldthrips.php. Accessed 15 February 2022
DeLong JP (2021) Predator ecology: evolutionary ecology of the functional response. Oxford University Press, Oxford, United Kingdom
Dobson AJ, Barnett AG (2008) An introduction to generalized linear model. CRC Press Taylor and Francis Group, New York, USA
Eubanks MD (2005) Predaceous herbivores and herbivorous predators: the biology of omnivores and the ecology of omnivore-prey interactions. In: Barbosa P, Castellanos I (eds) Ecology of predator-prey interactions. Oxford University Press, New York, USA, pp 3–16
Flores F (2021) Manejo de trips “Caliothrips phaseoli” en el cultivo de soja. INTA Digital (INTA) 1:40–43
Gamundi JC, Perotti E (2009) Evaluación de daño de Frankliniella schultzei (Trybom) y Caliothrips phaseoli (Hood) en diferentes estados fenológicos del cultivo de soja. Para Mejorar La Producción 42:107–111
García-Martínez O, Johansen-Naime R, Villarreal-Quintanilla R, Cambero-Campos J, Retana-Salazar AP (2011) Contribución al conocimiento de los Thysanoptera de Coahuila, México. Métodos En Ecología y Sistemática 6:15–26
González D, Pallerson BR, Leigh TF, Wilson LT (1982) Mites: a primary food source for two predators in San Joaquin Valley cotton. Calif Agric 36:18–20
Gonzalez D, Wilson LT (1982) A food-web approach to economic thresholds: a sequence of pests/predaceous arthropods on California cotton. Entomophaga 27:31–43
Haviland DR, Rill SM, Gordon CA (2021) Field biology of Scolothrips sexmaculatus (Thysanoptera: Thripidae) as a predator of Tetranychus pacificus (Acari: Tetranychidae) in California Almonds. J Econ Entomol 114:1111–1116. https://doi.org/10.1093/jee/toab036
Hoddle MS, Mound LA, Paris D (2012) Thrips of California 2012. CBIT Publishing, Queensland. https://keys.lucidcentral.org/keys/v3/thrips_of_california/Thrips_of_California.html. Accessed 02 Febrary 2022.
Holling CS (1959) Some characteristics of simple types of predation and parasitism. Can Entomol 91:385–398. https://doi.org/10.4039/Ent91385-7
Janini JC, Boiça Júnio AL, Jesus FG, Silva AG, Carbonell SA, Chiorato AF (2011) Effect of bean genotypes, insecticides, and natural products on the control of Bemisia tabaci (Gennadius) biotype B (Hemiptera: Aleyrodidae) and Caliothrips phaseoli (Hood) (Thysanoptera: Thripidae). Maringá 33:445–450. https://doi.org/10.4025/actasciagron.v33i3.7577
Juliano SA (2001) Nonlinear curve fitting: Predation and functional response curves. In: Scheiner SM, Gurevitch J (eds) Design and analysis of ecological experiments. Oxford University Press, Oxford, UK, pp 159–182
Lima-Espíndola J, Vanegas-Rico JM (2017) Damage caused by Tetranychus merganser Boudreaux (Acari: Tetranychidae) on nopal verdura Opuntia ficus-indica (L.) Miller during winter. J Entomol Acarol Res 49:121–122. https://doi.org/10.4081/jear.2017.6281
López-Bautista E, Santillán-Galicia MT, Suárez-Espinosa J, Cruz-Huerta N, Bautista-Martínez N, Alcántara-Jiménez JA (2016) Damage caused by mite Tetranychus merganser (Trombidiformes: Tetranychidae) on Carica papaya (Violales: Caricaceae) plants and effect of two species of predatory mite. Int J Acarol 42:303–309. https://doi.org/10.1080/01647954.2016.1184714
López-Bautista E (2014) Incidencia de daño y estrategias de control de Tetranychus merganser en el cultivo de papaya (Carica papaya L.). Dissertation, Colegio de Postgraduados Campus Montecillo, Texcoco, Estado de México, Mexico.
Migeon A, Dorkeld F (2022) Spider Mites Web: a comprehensive database for the Tetranychidae. Available from https://www1.montpellier.inra.fr/CBGP/spmweb. Accessed 08 July 2022
Milne M, Walter GH (1997) The significance of prey in the diet of the phytophagous thrips, Frankliniella schultzei. Ecol Entomol 22:74–81. https://doi.org/10.1046/j.1365-2311.1997.00034.x
Molina-Arjona C, Chacón-Hernández JC, Hernández-Juárez A, Anguiano-Cabello JC, Arredondo-Valdés R, Laredo-Alcala EI (2018) Caliothrips phaseoli (Thysanoptera: Thripidae) occurrence on Moringa oleifera (Brassicales: Moringaceae) and its predation of Tetranychus merganser (Acari: Tetranychidae). J Entomol Sci 53:89–92. https://doi.org/10.18474/JES17-76.1
Mound LA, Hoddle MS, Hastings A (2019). Thysanoptera Californica - Thrips of California. Lucidcentral.org, Identic Pty Ltd, Queensland, Australia. https://keys.lucidcentral.org/keys/v3/thrips_of_california_2019/. Accessed 26 February 2022
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/. Accessed 05 March 2022
Trichilo PJ, Leigh TF (1986) Predation on spider mite eggs by the western flower thrips, Frankhniella occidentalis (Thysanoptera: Thnpidae), an opportunist in a cotton agroecosystem. Environ Entomol 15:821–825. https://doi.org/10.1093/ee/15.4.821
Trichilo PJ, Leigh TF (1988) Influence of resource quality on the reproductive fitness of flower thrips (Thysanoptera: Thripidae). Ann Entomol Soc Am 81:64–70. https://doi.org/10.1093/aesa/81.1.64
Ullah MS, Moriya D, Kongchuensin M, Konvipasruang P, Gotoh T (2011) Comparative toxicity of acaricides to Tetranychus merganser Boudreaux and Tetranychus kanzawai Kishida (Acari: Tetranychidae). Int J Acarol 37:535–543. https://doi.org/10.1080/01647954.2010.525531
van Haperen P, Voorrips RE, van Loon JJA, Vosman B (2019) The effect of plant development on thrips resistance in Capsicum. Arthropod-Plant Inte 13:11–18. https://doi.org/10.1007/s11829-018-9645-6
Wilson LJ, Bauer LR, Walter GH (1996) ‘Phytophagous’ thrips are facultative predators of twospotted spider mites (Acari: Tetranychidae) on cotton in Australia. Bull Entomol Res 86:297–305. https://doi.org/10.1017/S0007485300052597
Zhong R, Ding T-B, Niu J-Z, Xia W-K, Liao C-Y, Dou W, Wang J-J (2015) Molecular characterization of vitellogenin and its receptor genes from citrus red mite, Panonychus citri (McGregor). Int J Mol Sci 16:4759–4773. https://doi.org/10.3390/ijms16034759
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Daniel Carillo.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Villagran-Mancilla, C., Chacón-Hernández, J.C., Delgadillo-Ángeles, J.L. et al. Phytophagy and predatory behavior of Caliothrips phaseoli (Thysanoptera: Thripidae) on bean foliage discs with Tetranychus merganser (Acari: Tetranychidae) eggs. Arthropod-Plant Interactions 17, 217–224 (2023). https://doi.org/10.1007/s11829-023-09949-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11829-023-09949-w