Abstract
Purpose
To examine associations between walkability, metropolitan size, and physical activity (PA) among cancer survivors and explore if the association between walkability and PA would vary across United States metropolitan sizes.
Methods
This study used data from the 2020 National Health Interview Survey to examine independent associations of walkability and metropolitan size with engaging in moderate-to-vigorous PA (MVPA) and to explore the effect modification of metropolitan size using log-binomial regression. The dependent variable was dichotomized as < vs. ≥ 150 min/week of MVPA. The predictors were perceived walkability, a total score comprising eight neighborhood attributes, and metropolitan size. Covariates included sociodemographic and health characteristics, geographic region, cancer type, and time since cancer diagnosis.
Results
Engaging in 150 + min/wk of MVPA significantly increased among cancer survivors (n = 3,405) who perceived their neighborhoods as more walkable (prevalence ratio:1.04; p = 0.004). Engaging in 150 + min/wk of MVPA significantly increased among cancer survivors living in medium and small metropolitan areas vs. those living in large central metropolitan areas (prevalence ratio:1.12; p = 0.044). Perceived walkability levels were similar among cancer survivors in nonmetropolitan areas vs. those living in large central metropolitan areas. Association between walkability and PA did not significantly vary across metropolitan sizes.
Conclusions
Perceived neighborhood walkability is positively associated with MVPA among cancer survivors, regardless of metropolitan size.
Implications for Cancer Survivors
Findings highlight the importance of investing in the built environment to increase walkability among this population and translating lessons from medium and small metropolitan areas to other metropolitan areas to address the rural–urban disparity in PA among cancer survivors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
As of January 2022, an estimated 18 million Americans have a history of cancer in the United States (U.S.) [1]. The number of cancer survivors is projected to increase to 26.6 million by the year 2040 [1]. This growth is attributed to factors such as population expansion, aging, enhanced early detection efforts, and advancements in medical treatments. Cancer survivors, defined as individuals who have been diagnosed with cancer and continue to live with it, often grapple with a range of physical and mental health issues that can persist for months or even years after their treatment [2,3,4,5].
The American Cancer Society advocates regular physical activity (PA) for cancer survivors, backed by a substantial body of evidence demonstrating the various health benefits of PA post-diagnosis [6]. These benefits include reduced cancer-specific and overall mortality, decreased cancer recurrence, fewer comorbidities, and improved management of treatment side effects, physical function, and overall quality of life [6,7,8,9,10,11]. A systematic review has shown levels of physical activity recommended in the 2018 Physical Activity Guidelines Advisory Committee Scientific Report are associated with reduced risk and improved survival for several cancers [12]. Despite these advantages, only approximately 38.4% of adult cancer survivors in the U.S. meet the recommended aerobic exercise guidelines [13], and nearly 40% report no physical activity at all [13, 14].
Walkability, which refers to the degree to which the built environment facilitates walking [15], has been associated with increased PA in adults [16, 17], as well as lower body mass index (BMI) [17,18,19], reduced risk of chronic diseases [19,20,21,22], and improved mental health [23]. Nevertheless, studies investigating the influence of the neighborhood environment on PA levels among cancer survivors have yielded mixed results. Three studies linked PA participation to the design and accessibility of the built environment [24,25,26]; however; two studies found no significant association between the neighborhood-built environment and PA among prostate and kidney cancer survivors [27, 28]. Three studies identified the outdoor environment as central to cancer survivors’ PA of choice, with walking being a preferred outdoor activity [29,30,31], but a study among colorectal cancer survivors indicated the neighborhood environment as less critical than the home environment for moderate-to-vigorous PA [32]. Finally, studies report conflicting results on the association between PA and geographic location (i.e., urban versus rural) among cancer survivors [25].
Unlike the general adult population [33, 34], fewer studies have examined environmental determinants of exercise among cancer survivors [35]. This gap in knowledge is significant because rural cancer survivors are known to have lower PA levels and poorer overall health outcomes compared to their urban counterparts [36, 37]. Furthermore, cancer survivors often face additional barriers to engaging in PA due to cancer treatment-related side effects (fatigue, insomnia, peripheral neuropathy, pain, and psychosocial distress) [9, 38,39,40,41,42]. It is important to note that findings on the built environment's influence on PA in the general population may not always apply to cancer survivors because they are typically older and have more chronic conditions, and some features of the built environment may be more or less relevant to their specific needs [43,44,45]. Rural residents encounter additional challenges, such as the absence of sidewalks on roads, which makes it more challenging for cancer survivors to exercise safely. However, research on efforts to enhance walkability is more common in urban areas compared to rural communities [18]. This disparity needs to be urgently addressed as the built environment (e.g., neighborhoods) is considered a social determinant of health. Creating environments and neighborhoods that promote health and safety to increase regular physical activity is an important objective of Healthy People 2030 [46].
This study used cross-sectional data from the 2020 National Health Interview Survey of civilian adults including adult cancer survivors aged 18 and older in the U.S. Its primary objectives were to: 1) Examine the association between perceived neighborhood walkability and PA; 2) Examine the association between metropolitan size and PA; and 3) Determine if the association between perceived walkability and PA differed based on metropolitan size. The hypothesis was that sufficient PA would be positively associated with perceived neighborhood walkability, sufficient PA would be inversely associated with rural areas (i.e., smaller metropolitan size), and the strength of the association between PA and walkability would vary depending on the size of the metropolitan area. This hypothesis was proposed because, in many communities, walking in the neighborhood is the most accessible form of exercise. However, cancer survivors may face hindrances due to the physical effects of cancer and its treatment, and neighborhood characteristics can also affect walking rates (e.g., traffic, crime, sidewalks, destinations). Therefore, the association between neighborhood factors and walking rates may vary across different metropolitan sizes due to varying built environments, population densities, and infrastructure.
Methods
National health interview survey
This study is a secondary analysis using the 2020 National Health Interview Survey (NHIS) cross-sectional data, specifically the adult file. The NHIS is a household survey that employs a complex, multistage probability sampling design, which includes stratification and clustering, to accurately represent the civilian, noninstitutionalized population residing in the 50 states and the District of Columbia at the time of the survey. The National Center for Health Statistics (NCHS), part of the U.S. Centers for Disease Control and Prevention, interviews one participant aged 18 or older from randomly selected households. These interviews cover various aspects, including demographics, health status, illness, disabilities, chronic conditions, access to healthcare, and utilization of health services.
Due to the COVID-19 pandemic, NCHS transitioned from face-to-face to telephone interviews from late March 2020 through June 2020. Data collection from July to December 2020 was primarily through face-to-face visits in areas with low COVID-19 prevalence, with continued reliance on telephone interviews in other regions [47]. For this study, the NHIS adult data file was utilized to obtain a nationally representative sample of noninstitutionalized adults in the U.S. [48].
Study population
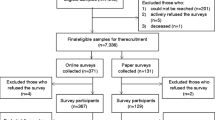
Within the adult NHIS file, 31,568 adults aged 18 and above completed the interviews. Among them, 4,130 participants reported having been previously informed by a healthcare professional that they had cancer. This excluded 34 participants who either refused to answer or were uncertain. Furthermore, pregnant participants (n = 3) were excluded from the analysis, as were individuals classified as having disabilities according to the criteria set by the NCHS, based on the Washington Group Short Set Composite Disability Indicator (n = 722) [49, 50]. Research indicates adults with disabilities exhibit distinct physical activity levels compared to the general adult population. Consequently, the final study sample consisted of 3,405 participants. See Fig. 1.
Measures
Dependent variable
Participants were asked eight questions about exercise, sports, or physically active hobbies during leisure time. These eight variables captured whether participants engaged in moderate or vigorous intensity activities (resulting in moderate or large increases in breathing or heart rate). From participants’ self-reported responses, the NCHS derived a three-category variable defined as inactive, insufficiently active (< 150 min/wk of MVPA), and sufficiently active (150 + min/wk of MVPA). This categorization reflects the recommended exercise guidelines for cancer survivors of 150 + min/week of MVPA as established by the American Cancer Society for aerobic exercise [51,52,53]. The dependent variable was dichotomized as sufficiently active vs. inactive/insufficiently active (the reference category).
Independent variables
Perceived walkability
This variable was derived from the responses to eight binary questions concerning the ease of walking related to various neighborhood characteristics, including traffic, crime, road conditions, availability of shops, public transportation, places for recreation, relaxing areas, and the presence of sidewalks (refer to Table 1 for specific questions). A variable ranging from 0 to 8 was created by summing the responses to these eight survey items. Higher scores indicated a higher level of perceived walkability. The internal consistency of eight survey items captured by a Cronbach’s alpha of 0.71 is considered as acceptably reliable [54]. More information on the perceived walkability variable can be found in a published paper written by the first, second, and last author [17].
Metropolitan size status
This variable is based on participants’ county of residence. It comes from the 2013 NCHS urban–rural classification scheme for U.S. counties, which combines counties of into six urban–rural categories: large central metro, large fringe metro, medium metro, small metro, micropolitan, and non-core [55]. The categorization included on the NHIS dataset groups medium and small metropolitan areas into a single category [47, 55].
Moderator variable
The four-level categorization of metropolitan size was also explored as a potential moderator affecting the relationship between perceived walkability (continuous) and PA (dichotomous).
Covariates
The covariates included several demographic and health-related variables and were chosen a priori based on putative factors known to be associated with PA, neighborhoods as place of residence/walkability (Age [years capped at 85], age-squared, biological sex (male or female), race/ethnicity, marital status, employment status, education level, nativity, federal poverty level, number of children in the household, self-reported health status, obesity status, number of chronic conditions other than cancer, and geographic region), but also variables capturing cancer type and time since cancer diagnosis [24, 56,57,58,59]. Additional details on the measurement of covariates are available in Table 1.
Statistical analyses
To describe the study population, weighted percentages were computed for categorical variables, and weighted means (with 95% confidence intervals [CI]) were calculated for continuous variables. The dependent variable, PA, was examined across all sociodemographic and health-related variables using the Rao-Scott Chi-square test [60] for categorical variables and the adjusted Wald F-Test for continuous variables. These tests were selected to account for the complex survey design of the NHIS [47, 61, 62].
A test for multicollinearity, based on the sample variance inflation factor (VIF), indicated no high correlation between the independent variables (mean VIF 1.35; range: 1.03–2.26). Due to the high prevalence of MVPA (45%), log-binomial regressions were fitted to assess the presence of a statistically significant association between perceived walkability and PA, as well as between metropolitan size and PA. The estimated associations between exposure and outcome were calculated as prevalence ratios (PR) instead of odds ratios [63, 64]. The possibility of metropolitan size acting as an effect modifier in the association between walkability and PA was explored through moderation analysis (a product term between walkability and metropolitan size included in the regression model) and tested using the adjusted Wald test. Model fit was assessed using Somers' Delta [65], with a coefficient closer to 1 indicating a better fit while adjusting for survey sampling weights.
NHIS complex survey design weights were applied to ensure unbiased estimates of descriptive and regression parameters and to account for variance in the weights [66]. Missing data constituted 4.9% of the total analytical sample; therefore, complete case analyses were conducted as this level of missingness would likely not influence the associations examined significantly or introduce large bias in the analysis [67]. All analyses were performed using STATA/SE 17 [68].
Results
Descriptive analysis
Table 2 provides an overview of the characteristics of the entire study population, categorized by their physical activity (PA) outcomes: either insufficiently active (< 150 min/wk of MVPA) or sufficiently active (≥ 150 min/wk of MVPA). For these two categories, the percentages were 55% and 45%, respectively.
The mean age of cancer survivors was 64.5 years (95% CI: 63.9, 65.1), and 55.2% of them were female. The majority of cancer survivors identified as White (84.1%), and there were noticeable disparities in PA levels across different racial/ethnic groups (p < 0.05). Notably, the proportion of Black and Hispanic cancer survivors who reported being sufficiently active was significantly lower than that of their White counterparts (33% and 39.1% vs. 46.2%). Significant sex-differences in PA levels were observed (p < 0.05), with 42.6% of females and 48.0% of males being sufficiently active.
Statistically significant disparities in PA levels were also found in relation to metropolitan size and perceived walkability scores (p < 0.01). Among cancer survivors residing in nonmetropolitan areas, 37.0% were sufficiently active, notably lower than those living in large fringe and medium and small metropolitan areas (47.7% and 46.6%, respectively). Moreover, the mean perceived walkability score was 5.40 (95% CI: 5.26, 5.54) for cancer survivors who were sufficiently active and 4.94 (95% CI: 4.80, 5.07) for those not meeting the activity guidelines, and this difference was statistically significant (p < 0.001).
There were significant differences in PA levels based on cancer type (p < 0.05). For instance, among cancer survivors with non-melanoma skin cancer, 51% reported being sufficiently active, a significantly higher percentage than among cancer survivors with multiple types of cancer (38.9%).
Health indicators for sufficiently active cancer survivors differed significantly from those who were not. For cancer survivors who self-rated their health as fair or poor, only 24.6% were sufficiently active, which was considerably lower than those who reported excellent health (67.8%) (p<0.001). More than a third of cancer survivors with obesity (37.8%) were sufficiently active, a proportion that was significantly lower than those without obesity (48.2%) (p<0.001). Additionally, sufficiently active cancer survivors had a significantly lower average number of chronic health conditions (2.5) compared to those who were not (3.1) (p<0.001).
Regression analysis
After incorporating all covariates in the log-binomial models, we found higher levels of perceived walkability were positively associated with being sufficiently active (adjusted prevalence ratio: 1.04; 95% CI: 1.01, 1.06; p = 0.004). Living in medium and small metropolitan areas vs. living in large central metropolitan areas was positively associated with being sufficiently active (adjusted prevalence ratio: 1.12; 95% CI: 1.00, 1.24; p = 0.044). Notably, cancer survivors living in nonmetropolitan areas and large fringe metropolitan areas reported similar levels of perceived walkability as cancer survivors residing in large central metropolitan areas. The model had a good fit, as indicated by Somers' Delta (0.68; 95% CI: 0.67, 0.69; p < 0.001). Refer to Table 3 for details.
Notably, the association between walkability and PA did not show significant variations based on metropolitan size (Adjusted Wald Test: F3,570 = 0.64, p = 0.592).
Discussion
Based on previous research, we hypothesized that sufficient physical activity (≥ 150 min/wk of MVPA) would be positively associated with perceived neighborhood walkability and negatively associated with metropolitan size (i.e., smaller metropolitan size) and that the strength of the association between PA and walkability would vary by metropolitan size. This research represents an initial attempt to explore how perceived walkability of neighborhoods, metropolitan size (or size of metropolitan areas), and PA levels are interconnected among a large and nationally diverse group of cancer survivors. Results of our study revealed that residing in a neighborhood where walkability is more positively perceived is linked to an increased likelihood of cancer survivors engaging in sufficient MVPA in the U.S. Furthermore, the size of the metropolitan area also plays a role in the likelihood of achieving sufficient PA, as individuals living in medium and small-sized metropolitan areas were more likely to achieve MVPA levels compared with those residing in large central metropolitan regions.
The association between walkability and PA did not vary by metropolitan size of cancer survivors’ residence.
Findings from this study are consistent with results from studies of perceived walkability and PA in adult populations. The positive association between neighborhood walkability and PA among adults has been established using a range of measurements for PA and walkability [19, 33, 34, 69,70,71,72]. Using the same measures of perceived walkability and PA as in this study, our previous research [17] found higher walkability associated with greater odds to engage in adequate PA among a nationally representative sample of U.S. adults with obesity.
This article adds new insights, as previous literature on neighborhood walkability and PA in cancer survivors has been limited to smaller samples, specific types of cancer (e.g., prostate [27], breast [25, 30, 31] or kidney cancer [26]), or specific geographic areas which limit the external validity of these studies’ findings. For example, a recent study by Stevens et al. (2023) investigated whether social and built environment factors relate to leisure-time PA among 219 rural cancer survivors and explored whether survivors’ stage of change for exercise moderated these associations. Similar to our results, study findings demonstrated that the built environment is associated with PA and additionally emphasized the role of family and friends' social support, home environment for exercise, and aesthetic attributes of the neighborhoods as having the strongest associations with PA. Moreover, Stevens et al.’s research showed a stronger positive association between social and built environmental factors and PA among rural cancer survivors in the action or maintenance stages of change vs. those in the precontemplation, contemplation, or preparation stages. However, the study was limited to cancer survivors living in a rural county in Pennsylvania; thus, findings may not be generalizable to other populations of cancer survivors [24].
Previous research has investigated the built environment and PA among survivors with a specific type of cancer. In a study conducted by Trinh and colleagues (2016) using the Alberta Cancer registry of kidney cancer survivors in Canada between 1996–2010, a combination of residential and commercial land use, as measured by the presence of shops within walking distance from ones’ home, was linked to adequate PA. However, no connection was seen between PA levels and built environments characteristics determined through geographic information systems (GIS) [28].
Using the same registry, Tabaczynski et al. (2020) examined correlates of the built (GIS-based measures such as median household income, population/park/local road/intersection density, and number of shopping and recreation centers) and perceived environments (nine self-reported measures such as traffic, neighborhood aesthetics, walking distance to shops, access to exercise attire) with aerobic and strength PA (separately and combined) among 651 kidney cancer survivors. They found survivors living at walking distance to shops and with access to workout attire (two out of nine perceived environment factors examined) were more likely to meet the aerobic-only guidelines, and those with access to workout attire were more likely to meet the combined guidelines. Notably, the nine built environmental GIS factors studied were not significantly associated with meeting either of the PA guidelines [26]. Associations between PA and geographic location (living in an urban area versus living in rural/remote areas based on self-report or country-defined zip codes) were also not found to be significant among 574 female breast cancer survivors who participated in three different PA intervention studies in Australia. These findings exemplify the dissonance between the built environment (GIS) vs. perception of the built environment and their potential differential influence on PA [25].
In a smaller study of 165 prostate cancer survivors (mean age 68.3) in Edmonton, Alberta, Canada, McGowan et al. found no association between PA and built environment measures, including 500 m buffer walkability and 1000 m buffer walkability [27]. One possible reason for the apparent positive links between walkability and PA as revealed in our research could be that neighborhood walkability and the opportunity for outdoor activity play a significant role in promoting walking, a form of PA often preferred by cancer survivors [40, 73, 74]. Supporting this idea, a smaller survey of cancer survivors conducted by Lesser and colleagues in 2021 showed the majority (83.3%) indicated outdoor recreational areas (28.1%) and general residential neighborhood (26.3%) as their preferred locations for engaging in PA [29].
Other studies on metropolitan size and PA have yielded somewhat mixed findings across different study samples. Metropolitan size has been positively associated with sufficient PA and with both leisure and transportation walking among U.S. adults using past years of NHIS data [75]. Using 2015 NHIS data, Whitfield (2019) reported both leisure and transportation walking were less common among rural compared to urban dwellers [76]. With respect to the specific types of walking, the study reported differences between urban and rural dwellers in the built environment features associated with transportation walking, but no difference between rural and urban residents in built environment features associated with leisure walking. Likewise, Mama et al. (2020) reported a significant difference in sufficient PA between rural and urban cancer survivors, with urban cancer survivors 2.6 times more likely to meet aerobic PA recommendations (95% CI 1.1–6.4) compared to rural cancer survivors in a sample of 591 cancer survivors drawn from a state cancer registry in Central Pennsylvania [36].
However, other studies have reported no difference in sufficient PA by metropolitan size or walkability among cancer survivors. Using 2006–2010 NHIS data, Weaver et al. (2013) reported sufficient PA did not differ significantly between rural (18.8%) and urban (22.8%) cancer survivors [77], although physical inactivity (defined as no PA) was significantly higher in rural compared to urban cancer survivors (rural 50.7% vs. urban 38.7%). One possible explanation for the null associations is that some cancer survivors may prefer to be active indoors or at home, depending on the kind of cancer and cancer treatment they received. For example, Lawrence et al. (2017) monitored activity and GPS location in a sample of 31 colorectal cancer survivors ages 50 and over in Nova Scotia, Canada and identified the home environment as the main location for moderate-to-vigorous PA, with a relatively small share of MVPA taking place in the environment outside the home [32]. Another explanation for variation in study findings may include differences in study samples and measures; compared to our study of a nationally representative sample of adult cancer survivors, other study samples focused on older age groups [27, 32], were cancer-type specific [27, 32, 78], were gender-specific [27], were location-specific in non-U.S. locations [27, 32, 78], were drawn from locations offering limited variability in built environment features [27, 32, 36, 78], or were limited to a dichotomous measure of metropolitan size (urban vs. rural) [36], compared to our study’s use of six different categories of metropolitan size.
Strengths and limitations
It is important to evaluate the study’s results while considering its strengths and limitations. This research marks the initial attempts to explore PA in cancer survivors based on their perception of neighborhood walkability using a composite scale of eight neighborhood attributes. This research also delves into PA patterns among cancer survivors with a focus on the size of metropolitan areas, distinguishing between medium and small, large fringe, and large central metropolitan area residency, within a nationally representative sample of cancer survivors in the U.S. This is an important strength because findings from our study are generalizable to U.S. cancer survivor populations, whereas previous comparable studies are small-scale studies with limited external validity or studies with simple convenience design which increases risk of selection bias. Study data are from a large, nationally representative random sample of adult cancer survivors in the U.S., and we had sufficient variability to consider metropolitan size, where previous studies have examined urban vs. rural as a binary variable.
Limitations of the study include the cross-sectional survey design, which limits inferences of causality and directionality of the observed associations. With respect to measures, PA was assessed via self-report and may vary from studies using more objective measures [79, 80]. Additionally, self-reported PA may be overestimated [80] and subject to variations in interpretation [81] or social desirability bias reporting [82]. Our measure of perceived walkability [17] has not been validated across different racial and ethnic groups [17], and responses based on perceived walkability may vary based on comfort level and security [16]. Moreover, findings from this study are limited to perceived neighborhood walkability. It is plausible that objective measures of traffic, crime, road conditions, shops, public transportation, places for recreation, relaxing areas, and presence of sidewalks captured with GPS or GIS technologies would have yielded different results—in terms of magnitude of associations and statistical significance—in relation to MVPA levels among cancer survivors. Furthermore, other important aspects of MVPA such as exercise preferences, adherence, and maintenance were not considered in this study [30, 31, 35].
Policy implications
Findings of this research have important implications for crafting interventions aimed at facilitating moderate-to-vigorous physical activities among cancer survivors. Based on our findings, policies curbing environmental barriers (such as traffic, crime, poor road conditions and sidewalks, and limited public transportation) and promoting places for recreation and relaxation to improve cancer survivors’ perception of walkability and facilitate physical activity in neighborhoods are recommended. The results underscore the significance of dedicating resources to enhance the urban infrastructure for better walkability. They also emphasize the need to extract insights from medium and small metropolitan areas (e.g., walkable community models) and apply them to various metropolitan areas to address the physical activity gap between cancer survivors living in medium and small-size metropolitan areas vs. those living in large central metropolitan areas. Since the average age of cancer survivors was 65 years in our study population, policy recommendations promoting retirement in medium and small metropolitan communities may be an attractive strategy more amenable to the physical activity needs of cancer survivors.
According to the World Health Organization, 30–50% of cancers are preventable [83]. Prevention of cancer will not only be achieved by pressing cancer survivors to adhere to regular MVPA but will depend on action on the social determinants of health such as the built environments and neighborhoods. This will necessitate bold actionable strategies across different sectors of the government (e.g., health, urban planning). The potential positive long-term ramifications are great: reduce inequality in the occurrence of cancer and advance the cause of health equity in the U.S. [84].
Conclusion
Perceived neighborhood walkability is positively associated with MVPA activities among cancer survivors, irrespective of U.S. metropolitan size. Future studies should assess the dissonance between the built environment captured by GPS/GIS-based measures and perception of the built environment so that cancer survivors’ perception of their neighborhood environment aligns better with what is in their neighborhood. Future research could consider the potential moderating effects of other moderating variables such as time since diagnosis, cancer type, chronic diseases, and health status. These variables are unique to cancer survivors and can provide additional insights into the design of future interventions to increase PA. Additional research on MVPA preferences, adherence, and maintenance among cancer survivors is warranted. Finally, since this survey was administered during the COVID-19 pandemic, future studies could investigate whether the associations found in this study have perdured post-pandemic.
Data availability
The 2020 National Health Interview Survey (NHIS) is openly made available by the Centers for Disease Control and Prevention (CDC) at https://www.cdc.gov/nchs/nhis/2020nhis.htm. No datasets were generated for this study. Any analysis, interpretation, and/or conclusion based on the NHIS 2020 data is solely that of the authors. Opinions, conclusions, and recommendations expressed herein do not necessarily represent those of the National Center for Health Statistics or the CDC, which are responsible for the data.
References
American Cancer Society. Cancer treatment & survivorship facts & figures 2022–2024. Atlanta, GA: American Cancer Society; 2022.
Stein K, Syrjala K, Andrykowski M. Physical and psychological long-term and late effects of cancer. Cancer. 2008;112(11 Suppl):2577–92.
van den Beuken-van Everdingen M, et al. Prevalence of pain in patients with cancer: a systematic review of the past 40 years. Ann Oncol. 2007;18(9):1437–49.
Weis J. Cancer-related fatigue: prevalence, assessment and treatment strategies. Expert Rev Pharmacoecon Outcomes Res. 2011;11(4):441–6.
Mitchell A, et al. Depression and anxiety in long-term cancer survivors compared with spouses and healthy controls: a systematic review and meta-analysis. Lancet Oncol. 2013;14(8):721–32.
Rock C, et al. American Cancer Society nutrition and physical activity guideline for cancer survivors. CA: A Cancer J Clin. 2022;72(3):230–62.
Friedenreich C, et al. Physical Activity and Mortality in Cancer Survivors: A Systematic Review and Meta-Analysis. JNCI Cancer Spectr. 2020;4(1):pkz080.
Swain C, et al. Postdiagnosis sedentary behavior and health outcomes in cancer survivors: a systematic review and meta-analysis. Cancer. 2020;126(4):861–9.
Cormie P, et al. The impact of exercise on cancer mortality, recurrence, and treatment-related adverse effects. Epidemiol Rev. 2017;39(1):71–92.
Gerritsen J, Vincent A. Exercise improves quality of life in patients with cancer: a systematic review and meta-analysis of randomised controlled trials. Br J Sports Med. 2016;50(13):796–803.
Burke S, et al. Physical activity and quality of life in cancer survivors: a meta-synthesis of qualitative research. Cancers (Basel). 2017;9(5):53.
McTiernan A, et al. Physical activity in cancer prevention and survival: a systematic review. Med Sci Sports Exerc. 2019;51(6):1252–61.
Arem H, et al. Prevalence of healthy behaviors among cancer survivors in the United States: how far have we come? Cancer Epidemiol Biomarkers Prev. 2020;29(6):1179–87.
National Cancer Institute. Cancer Survivors and Physical Activity. National Institutes of Health. 2022. Available from: https://progressreport.cancer.gov/after/physical_activity. Accessed 12 Apr 2023.
Wang H, Yang Y. Neighbourhood walkability: a review and bibliometric analysis. Cities. 2019;93:43–61.
Liao B, et al. How does walkability change behavior? a comparison between different age groups in the Netherlands. Int J Environ Res Public Health. 2020;17(2):540.
Wang ML, Narcisse MR, McElfish PA. Higher walkability associated with increased physical activity and reduced obesity among United States adults. Obesity (Silver Spring). 2023;31(2):553–64.
Grasser G, et al. Objectively measured walkability and active transport and weight-related outcomes in adults: a systematic review. Int J Public Health. 2013;58(4):615–25.
Sallis J, et al. Role of built environments in physical activity, obesity, and cardiovascular disease. Circulation. 2012;125(5):729–37.
Booth G, et al. Neighbourhood walkability and the incidence of diabetes: an inverse probability of treatment weighting analysis. J Epidemiol Community Health. 2019;73(4):287–94.
Coffee N, et al. Is walkability associated with a lower cardiometabolic risk? Health Place. 2013;21:163–9.
Jones A, et al. Neighborhood walkability as a predictor of incident hypertension in a National Cohort Study. Front Public Health. 2021;9:611895.
Zhao Y, Chung P. Neighborhood environment walkability and health-related quality of life among older adults in Hong Kong. Arch Gerontol Geriatr. 2017;73:182–6.
Stevens CJ, et al. Linking social and built environmental factors to leisure-time physical activity in rural cancer survivors. J Natl Cancer Inst Monogr. 2023;2023(61):125–32.
Kampshoff CS, et al. Demographic, clinical, psychosocial, and environmental correlates of objectively assessed physical activity among breast cancer survivors. Support Care Cancer. 2016;24(8):3333–42.
Tabaczynski A, et al. Demographic, medical, social-cognitive, and environmental correlates of meeting independent and combined physical activity guidelines in kidney cancer survivors. Support Care Cancer. 2020;28(1):43–54.
McGowan E, et al. The role of the built environment in a randomized controlled trial to increase physical activity among men with prostate cancer: the PROMOTE trial. Support Care Cancer. 2017;25(10):2993–6.
Trinh L, et al. Social-ecological correlates of physical activity in kidney cancer survivors. J Cancer Surviv. 2016;10(1):164–75.
Lesser I, Nienhuis C, Belanger L. Active by nature: exploring cancer survivors’ exercise barriers, facilitators, preferences, and psychosocial benefits of engaging in outdoor physical activity. Support Care Cancer. 2021;29(7):4095–103.
Vallance J, et al. Rural and small town breast cancer survivors’ preferences for physical activity. Int J Behav Med. 2013;20(4):522–8.
Rogers LQ, et al. Rural breast cancer survivors: exercise preferences and their determinants. Psychooncology. 2009;18(4):412–21.
Lawrence L, et al. Environments Associated with Moderate-to-Vigorous Physical Activity and Sedentary Behavior of Colorectal Cancer Survivors. Int J Behav Med. 2017;24(1):120–6.
Smith M, et al. Systematic literature review of built environment effects on physical activity and active transport - an update and new findings on health equity. Int J Behav Nutr Phys Act. 2017;14(1):158.
McCormack G, Shiell A. In search of causality: a systematic review of the relationship between the built environment and physical activity among adults. Int J Behav Nutr Phys Act. 2011;8:125.
Kampshoff CS, et al. Determinants of exercise adherence and maintenance among cancer survivors: a systematic review. Int J Behav Nutr Phys Act. 2014;11:80.
Mama S, et al. Rural-urban differences in meeting physical activity recommendations and health status in cancer survivors in central Pennsylvania. Support Care Cancer. 2020;28(10):5013–22.
Weaver K, et al. Rural-urban disparities in health status among US cancer survivors. Cancer. 2013;119(5):1050–7.
NIH News in Health. Health in Rural America: Connecting to Care. 2022. Available from: https://newsinhealth.nih.gov/2022/03/health-rural-america. Accessed 4 Apr 2023.
National Center for Health Statistics. QuickStats: percentage of children and adolescents aged 6–17 years who have roads, sidewalks, paths, or trails where they can walk or ride a bicycle, by urban-rural status and family income — national health interview survey, United States, 2020. MMWR Morb Mortal Wkly Rep. 2022;71(35):1136.
Blaney JM, et al. Cancer survivors’ exercise barriers, facilitators and preferences in the context of fatigue, quality of life and physical activity participation: a questionnaire-survey. Psychooncology. 2013;22(1):186–94.
Cadmus-Bertram L, et al. Understanding the physical activity needs and interests of inactive and active rural women: a cross-sectional study of barriers, opportunities, and intervention preferences. J Behav Med. 2020;43(4):638–47.
Adams J, et al. Determining patient needs to enhance exercise program implementation and uptake in rural settings for women after a cancer diagnosis. Support Care Cancer. 2021;29(8):4641–9.
Frank L, Engelke P, Schmid T. Health and Community Design: The Impact Of The Built Environment On Physical Activity. 1st ed. Washington DC: Island Press; 2003.
Tcymbal A, et al. Effects of the built environment on physical activity: a systematic review of longitudinal studies taking sex/gender into account. Environ Health Prev Med. 2020;25(1):75.
Rock C, et al. American Cancer Society guideline for diet and physical activity for cancer prevention. CA: A Cancer J Clin. 2020;70(4):245–71.
Healthy People 2030. Increase the proportion of adults who walk or bike to get places — PA‑10. Office of Disease Prevention and Health Promotion. Available from: https://health.gov/healthypeople/objectives-and-data/browse-objectives/physical-activity/increase-proportion-adults-who-walk-or-bike-get-places-pa-10. Accessed 1 Mar 2024.
National Health Interview Survey. 2020 Survey Description. National Center for Health Statistics. 2021. Available from: https://ftp.cdc.gov/pub/Health_Statistics/NCHS/Dataset_Documentation/NHIS/2020/srvydesc-508.pdf. Accessed 4 Mar 2024.
National Health Interview Survey. 2019 Survey Description. National Center for Health Statistics. 2019. Available from: https://nhis.ipums.org/nhis/resources/srvydesc2019.pdf. Accessed 4 Mar 2024.
Centers for Disease Control and Prevention. Disability and Obesity. 2019. Available from: https://www.cdc.gov/ncbddd/disabilityandhealth/obesity.html. Accessed 4 Mar 2024.
Fox M, Witten M, Lullo C. Reducing obesity among people with disabilities. J Disabil Policy Stud. 2014;25(3):175–85.
DiPietro L, et al. New scientific basis for the 2018 US Physical Activity Guidelines. J Sport Health Sci. 2019;8(3):197–200.
King AC, et al. Shaping the future of physical activity promotion: highlights from the 2018 pa guidelines scientific advisory committee review. Ann Behav Med. 2018;52:S173–S173.
Vaux-Bjerke A, et al. Strong evidence from the 2018 physical activity guidelines advisory committee. Med Sci Sports Exerc. 2018;50(5):634–5.
Cronbach LJ. Coefficient alpha and the internal structure of tests. Psychometrika. 1951;16:297–334.
Ingram DD, Franco SJ. NCHS urban-rural classification scheme for counties. National Center for Health Statistics. Vital Health Stat 2. 2014;(166):1–73.
Bluethmann SM, et al. Physical activity in older cancer survivors: what role do multimorbidity and perceived disability play? J Aging Phys Act. 2020;28(2):311–9.
Bhatia S, et al. Rural-urban disparities in cancer outcomes: opportunities for future research. J Natl Cancer Inst. 2022;114(7):940–52.
Zahnd WE, et al. The intersection of rural residence and minority race/ethnicity in cancer disparities in the United States. Int J Environ Res Public Health. 2021;18(4):1384.
Leach MJ, et al. Physical activity, obesity, and quality of life among rural Australian cancer survivors: a cross-sectional study. Support Care Cancer. 2023;31(4):222.
Rao J, Scott A. On simple adjustments to chi-square tests with sample survey data. Ann Stat. 1987;15(1):385–97.
Bramlett M, Dahlhamer J, Bose J. Weighting procedures and bias assessment for the 2020 national health interview survey. National center for health statistics. 2021. https://ftp.cdc.gov/pub/Health_Statistics/NCHS/Dataset_Documentation/NHIS/2020/nonresponse-report-508.pdf. Accessed 4 Mar 2024.
National Center for Health Statistics. NHIS data, questionnaires and related documentation. Centers for disease control and prevention. 2019. Available from: https://www.cdc.gov/nchs/nhis/data-questionnaires-documentation.htm. Accessed 9 Oct 2019.
Deddens JA, Petersen MR. Approaches for estimating prevalence ratios. Occup Environ Med. 2008;65(7):481, 501–6.
Zocchetti C, Consonni D, Bertazzi PA. Estimation of prevalence rate ratios from cross-sectional data. Int J Epidemiol. 1995;24(5):1064–7.
Somers RH. A new asymmetric measure of association for ordinal variables. Am Sociol Rev. 1962;27(6):799–811.
Heeringa S, West BT, Berglund PA. Applied survey data analysis. 2nd ed. Boca Raton, FL: CRC Press, Taylor & Francis Group; 2017.
Little RJA, Rubin DB. Statistical analysis with missing data. Hoboken, NJ: Wiley; 2020.
StataCorp. Stata Statistical Software: Release 17. College Station, TX: StataCorp LLC. 2021.
Rundle A, et al. Using GPS data to study neighborhood walkability and physical activity. Am J Prev Med. 2016;50(3):e65–72.
Paul P, Carlson S, Fulton J. Walking and the perception of neighborhood attributes among US adults-2012. J Phys Act Health. 2017;14(1):36–44.
Kerr J, et al. Perceived neighborhood environmental attributes associated with walking and cycling for transport among adult residents of 17 cities in 12 countries: The IPEN study. Environ Health Perspect. 2016;124(3):290–8.
Frank L, et al. Chronic disease and where you live: Built and natural environment relationships with physical activity, obesity, and diabetes. Environ Int. 2022;158:106959.
Buffart LM, et al. Evidence-based physical activity guidelines for cancer survivors: current guidelines, knowledge gaps and future research directions. Cancer Treat Rev. 2014;40(2):327–40.
McGowan EL, et al. Physical activity preferences among a population-based sample of colorectal cancer survivors. Oncol Nurs Forum. 2013;40(1):44–52.
Whitfield G, et al. Trends in meeting physical activity guidelines among urban and rural dwelling adults - United States, 2008–2017. MMWR Morb Mortal Wkly Rep. 2019;68(23):513–8.
Whitfield G, et al. National-level environmental perceptions and walking among urban and rural residents: Informing surveillance of walkability. Prev Med. 2019;123:101–8.
Weaver K, et al. Rural-urban differences in health behaviors and implications for health status among US cancer survivors. Cancer Causes Control. 2013;24(8):1481–90.
Lynch B, et al. Perceived barriers to physical activity for colorectal cancer survivors. Support Care Cancer. 2010;18(6):729–34.
Dowd K, et al. A systematic literature review of reviews on techniques for physical activity measurement in adults: a DEDIPAC study. Int J Behav Nutr Phys Act. 2018;15(1):15.
Troiano R, et al. Physical activity in the United States measured by accelerometer. Med Sci Sports Exerc. 2008;40(1):181–8.
Silsbury Z, Goldsmith R, Rushton A. Systematic review of the measurement properties of self-report physical activity questionnaires in healthy adult populations. BMJ Open. 2015;5(9):e008430.
Adams S, et al. The effect of social desirability and social approval on self-reports of physical activity. Am J Epidemiol. 2005;161(4):389–98.
World Health Organization. Preventing Cancer. Available from: https://www.who.int/activities/preventing-cancer. Accessed 1 Mar 2024.
Marmot M. Health equity, cancer, and social determinants of health. Lancet Global Health. 2018;6(S29).
Funding
Research reported in this publication was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under award number (UL1 TR003107). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
MRN conceptualized the study, conducted the statistical analysis, wrote the Abstract, and revised the whole manuscript. MLW wrote the Introduction and Discussion. MS, PAM, and PD revised the whole manuscript. AGS contributed to the literature review. All authors reviewed the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
N/A.
Consent to participate
N/A.
Consent to publish
N/A.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Narcisse, MR., Wang, M.L., Schootman, M. et al. Physical activity among cancer survivors: do neighborhood walkability and metropolitan size play a role?. J Cancer Surviv (2024). https://doi.org/10.1007/s11764-024-01584-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11764-024-01584-5